2 results
Mycobacterium chimaera infections in cardiothoracic surgery patients exposed to heating and cooling devices despite infection control measures
- Jensie Burton, Yosra Alkabab, Susan Dorman, Jeremy D. Moore, Danny Nixon, Cassandra Salgado, Scott Curry
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, pp. s15-s16
-
- Article
-
- You have access Access
- Open access
- Export citation
-
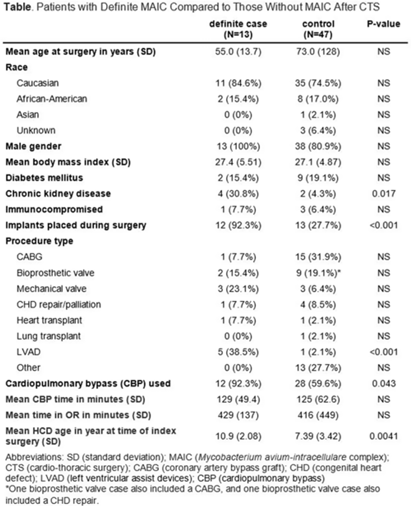
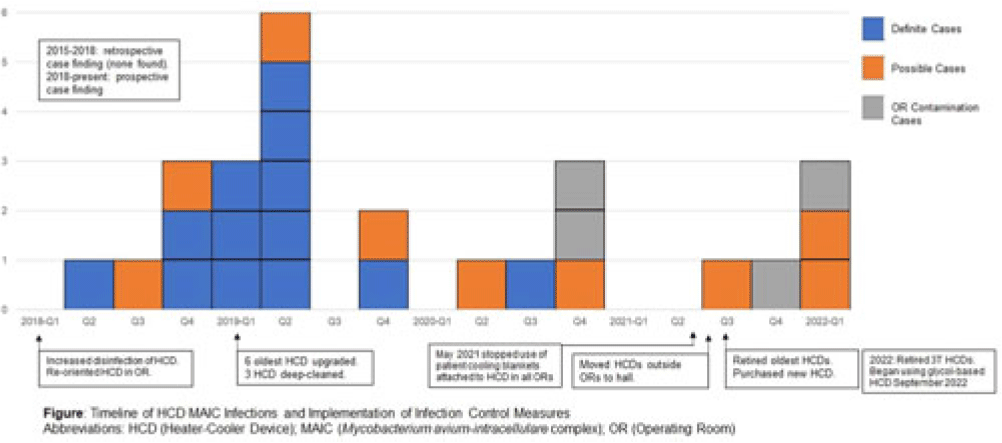
Background: LivaNova 3T heating and cooling devices (HCDs) have been associated with Mycobacterium chimaera, a Mycobacterium avium-intracellulare (MAIC) species, infections after cardiothoracic surgery. We describe our outbreak, which persisted despite escalating infection control measures. Methods: We identified patients with a positive MAIC culture following cardiothoracic surgery from January 2015 to the present at our institution. We classified these as “definite,” “possible,” or “operating room contamination” cases based on positive cultures from sterile sites, airway, or surgical specimens without evidence of infection. To identify patient or surgery characteristics associated with risk for MAIC infection, we conducted a case–control study comparing definite cases to randomly selected unmatched controls of patients over the same period without a positive MAIC culture after cardiothoracic surgery. Results: We identified 26 patients with a positive MAIC culture after cardiothoracic surgery: 13 definite, 9 possible, and 4 contamination cases. Among definite cases, the most common surgeries were valve replacements and left ventricular assist devices (5 cases each). The mean time from cardiothoracic surgery to diagnosis was 525 days. Overall, 10 (77%) cases occurred after exposure to our oldest HCDs (manufactured in 2013 or earlier). To date, 16 (62%) have undergone or are undergoing treatment for MAIC infection, and 4 (15%) have died due to NTM infection or complications. Compared to 47 controls, definite cases were associated with chronic kidney disease, implants, procedure type, use of cardiopulmonary bypass, and HCD age. Cases were not associated with time on bypass, time in the operating room, or other comorbid conditions (Table). All cases occurred despite enhanced disinfection and reorienting the HCD within the operating room, according to manufacturer recommendations. Moreover, 18 cases, including 7 definite cases, occurred after most HCDs were either deep cleaned or upgraded by the manufacturer. Also, 5 cases, including 3 possible cases and 2 contamination cases, occurred after physical separation of the HCD from the operating room. In August 2022, we purchased a fleet of glycol-cooled HCDs, and we have not identified additional MAIC cases since their deployment (Fig.). Conclusions: MAIC infections after cardiothoracic surgery were associated with procedure type, especially implants, use of cardiopulmonary bypass, and HCD age. Contrary to prior reports, neither operative nor CPB time was associated with MAIC infection after cardiothoracic surgery. The outbreak persisted despite disinfection and/or deep cleaning and reorienting HCDs within the operating room; some possible and contamination cases occurred even after moving HCDs outside the operating room. Thus, HCD water contamination events in the operating room (eg, spills from HCD tubing) may be a route of exposure, and different infection prevention measures are needed.
Disclosure: None
A single-center experience with microbiologic surveillance of LivaNova 3T heater-cooler devices (HCDs)
- Scott Curry, Yosra Alkabab, Danny Nixon, Susan Dorman, Cassandra Salgado
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, p. s91
-
- Article
-
- You have access Access
- Open access
- Export citation
-
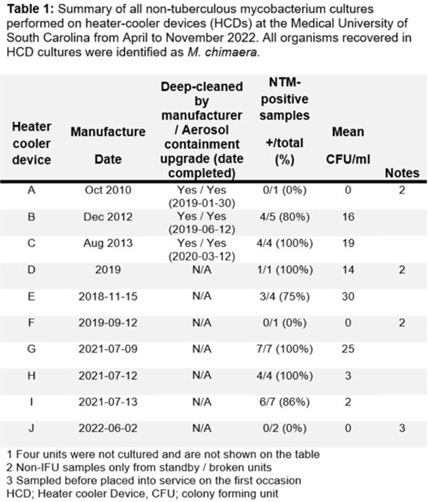
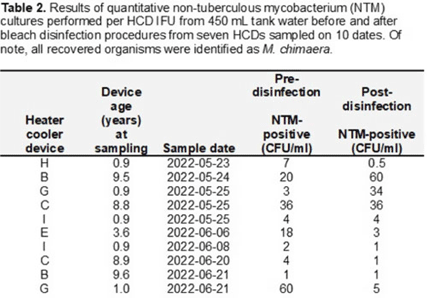
Background: The global outbreak of Mycobacterium chimaera infections associated with HCDs resulted in new maintenance recommendations. Since 2018, HCDs have been disinfected according to instructions for use (IFU), including twice-monthly bleach disinfection and monitoring hydrogen peroxide (H2O2) to maintain a minimum daily concentration of 100 ppm. In February 2020, the IFU added the recommendation to perform microbiologic surveillance of HCD tank water to ensure effectiveness of disinfection to levels of <1 colony forming unit per milliliter (CFU/mL) of nontuberculous mycobacterium (NTM). We report our experience with this microbiologic surveillance as well as that of culturing the HCD environment to investigate modes of transmission. Methods: In 2022, we began culturing tank water in 10 HCDs for NTM. For a subset of 6 HCDs, quantitative NTM culturing of tank water before and after bleach disinfection was done. After initial results indicated widespread-contamination of HCDs with M. chimaera, we performed fill water cultures from 5 sinks in 4 HCD maintenance rooms. We also conducted 20 two-hour NTM settle-plate cultures of a cardiac operating room (OR) at different sites both inside (n = 7) and outside (n = 3) the OR: 10 with the HCD (located outside the OR) turned off (controls) and 10 with HCD turned on (exposure). A paired t test was used to evaluate differences in mean recovery of NTM in tank water samples. Results: Cultures from 7 (70%) of 10 HCDs were positive, with a mean of 13.6 CFU/mL M. chimaera (Table 1). There was no significant difference between the 10 pairs of pre- and postdisinfection NTM cultures done according to the IFU from 6 HCDs: mean predisinfection cultures (15.5 CFU/mL) versus mean postdisinfection cultures (12 CFU/mL) (P = .90) (Table 2). For fill water, 1 of 7 sink samples in 1 of 4 rooms was positive for M. chimaera ( <1 CFU/mL) from a specimen from a fresh 0.2-µm filter that had been stored in the fill-sink splash zone. OR settle-plate cultures showed 0 (0%) of 10 control sites and 1 (10 %) of 10 exposure sites inside the OR positive for NTM, with a single CFU of M. avium–intracellulare complex. Conclusions: Our data cannot clearly refute either of 2 possible scenarios for HCD contamination: cross contamination during device maintenance versus at the point of manufacture. Despite the IFU guidance or disinfection being implemented, disinfection procedures failed to suppress NTM contamination, and tank water within most HCDs was contaminated with M. chimaera regardless of age or whether it was deep cleaned or upgraded with an aerosol containment device.
Disclosures: None







