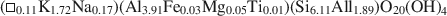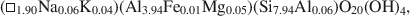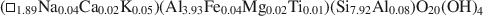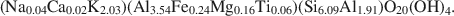11 results
Transmission Electron Microscopic Study of Coexisting Pyrophyllite and Muscovite: Direct Evidence for the Metastability of Illite
- Wei-Teh Jiang, Eric J. Essene, Donald R. Peacor
-
- Journal:
- Clays and Clay Minerals / Volume 38 / Issue 3 / June 1990
- Published online by Cambridge University Press:
- 02 April 2024, pp. 225-240
-
- Article
- Export citation
-
Transmission electron microscopy has been used to characterize coexisting pyrophyllite and muscovite in low-grade metamorphosed pelites from Witwatersrand and northeastern Pennsylvania. The Witwatersrand sample consisted largely of porphyroblasts of interlayered muscovite and pyrophyllite in a fine-grained matrix of the same phases. In both textures, muscovite and pyrophyllite occurred as interlayered packets (with apparently coherent interfaces) from about 300 Å to a few micrometers in thickness, with no mixed layering. Their compositions were determined with a scanning transmission electron microscope to be
$$(\Box{_{{\rm{0}}{\rm{.11}}}}{\rm{ }}{{\rm{K}}_{{\rm{1}}{\rm{.72}}}}{\rm{Na}_{{\rm{0}}{\rm{.17}}})(A}{{\rm{l}}_{{\rm{3}}{\rm{.91}}}}{\rm{ F}}{{\rm{e}}_{{\rm{0}}{\rm{.03}}}}{\rm{M}}{{\rm{g}}_{{\rm{0}}{\rm{.05}}}}{\rm{T}}{{\rm{i}}_{{\rm{0}}{\rm{.01}}}}{\rm{)(S}}{{\rm{i}}_{{\rm{6}}{\rm{.11}}}}{\rm{ Al}}{{\rm{l}}_{{\rm{1}}{\rm{.89}}}}{\rm{)}}{{\rm{O}}_{{\rm{20}}}}{{\rm{(OH)}}_{\rm{4}}}$$and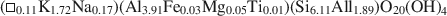 $$(\Box_{{\rm{1}}{\rm{.90}}}{\rm{N}}{{\rm{a}}_{{\rm{0.06}}}}{{\rm{K}}_{{\rm{0}}{\rm{.04}}}}{\rm{)(A}}{{\rm{l}}_{{\rm{3}}{\rm{.94}}}}{\rm{F}}{{\rm{e}}_{{\rm{0}}{\rm{.01}}}}{\rm{M}}{{\rm{g}}_{{\rm{0}}{\rm{.05}}}}{\rm{)(S}}{{\rm{i}}_{{\rm{7}}{\rm{.94}}}}{\rm{A}}{{\rm{l}}_{{\rm{0}}{\rm{.06}}}}{\rm{)}}{{\rm{O}}_{{\rm{20}}}}{{\rm{(OH)}}_{\rm{4}}},$$respectively.
$$(\Box_{{\rm{1}}{\rm{.90}}}{\rm{N}}{{\rm{a}}_{{\rm{0.06}}}}{{\rm{K}}_{{\rm{0}}{\rm{.04}}}}{\rm{)(A}}{{\rm{l}}_{{\rm{3}}{\rm{.94}}}}{\rm{F}}{{\rm{e}}_{{\rm{0}}{\rm{.01}}}}{\rm{M}}{{\rm{g}}_{{\rm{0}}{\rm{.05}}}}{\rm{)(S}}{{\rm{i}}_{{\rm{7}}{\rm{.94}}}}{\rm{A}}{{\rm{l}}_{{\rm{0}}{\rm{.06}}}}{\rm{)}}{{\rm{O}}_{{\rm{20}}}}{{\rm{(OH)}}_{\rm{4}}},$$respectively.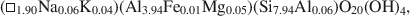
The pyrophyllite and muscovite in the Pennsylvania shale likewise occurred only as coexisting coherent to sub-parallel packets as thin as 200 Å, with compositions of
$$(\Box_{{\rm{1}}{\rm{.89}}}{\rm{N}}{{\rm{a}}_{{\rm{0.04}}}}{\rm{C}}{{\rm{a}}_{{\rm{0}}{\rm{.02}}}}{{\rm{K}}_{{\rm{0}}{\rm{.05}}}}{\rm{)(A}}{{\rm{l}}_{{\rm{3}}{\rm{.93}}}}{\rm{F}}{{\rm{e}}_{{\rm{0}}{\rm{.04}}}}{\rm{M}}{{\rm{g}}_{{\rm{0}}{\rm{.02}}}}{\rm{T}}{{\rm{i}}_{{\rm{0}}{\rm{.01}}}}{\rm{)(S}}{{\rm{i}}_{{\rm{7}}{\rm{.92}}}}{\rm{A}}{{\rm{l}}_{{\rm{0}}{\rm{.08}}}}{\rm{)}}{{\rm{O}}_{{\rm{20}}}}{{\rm{(OH)}}_{\rm{4}}}$$and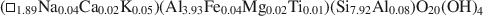 $$({\rm{N}}{{\rm{a}}_{{\rm{0}}{\rm{.04}}}}{\rm{C}}{{\rm{a}}_{{\rm{0}}{\rm{.02}}}}{{\rm{K}}_{{\rm{2}}{\rm{.03}}}}{\rm{)(A}}{{\rm{l}}_{{\rm{3}}{\rm{.54}}}}{\rm{F}}{{\rm{e}}_{{\rm{0}}{\rm{.24}}}}{\rm{M}}{{\rm{g}}_{{\rm{0}}{\rm{.16}}}}{\rm{T}}{{\rm{i}}_{{\rm{0}}{\rm{.06}}}}{\rm{)(S}}{{\rm{i}}_{{\rm{6}}{\rm{.09}}}}{\rm{A}}{{\rm{l}}_{{\rm{1}}{\rm{.91}}}}{\rm{)}}{{\rm{O}}_{{\rm{20}}}}{{\rm{(OH)}}_{\rm{4}}}.$$The textures of both samples were consistent with an equilibrium relationship between pyrophyllite and muscovite. The Pennsylvania sample also contained NH4-rich illite, kaolinite, and an illite-like phase having intermediate Na/K, which collectively imply non-equilibrated low-grade conditions.
$$({\rm{N}}{{\rm{a}}_{{\rm{0}}{\rm{.04}}}}{\rm{C}}{{\rm{a}}_{{\rm{0}}{\rm{.02}}}}{{\rm{K}}_{{\rm{2}}{\rm{.03}}}}{\rm{)(A}}{{\rm{l}}_{{\rm{3}}{\rm{.54}}}}{\rm{F}}{{\rm{e}}_{{\rm{0}}{\rm{.24}}}}{\rm{M}}{{\rm{g}}_{{\rm{0}}{\rm{.16}}}}{\rm{T}}{{\rm{i}}_{{\rm{0}}{\rm{.06}}}}{\rm{)(S}}{{\rm{i}}_{{\rm{6}}{\rm{.09}}}}{\rm{A}}{{\rm{l}}_{{\rm{1}}{\rm{.91}}}}{\rm{)}}{{\rm{O}}_{{\rm{20}}}}{{\rm{(OH)}}_{\rm{4}}}.$$The textures of both samples were consistent with an equilibrium relationship between pyrophyllite and muscovite. The Pennsylvania sample also contained NH4-rich illite, kaolinite, and an illite-like phase having intermediate Na/K, which collectively imply non-equilibrated low-grade conditions.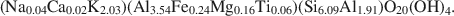
The compositions of these coexisting pyrophyllite and muscovite define a solvus with steep limbs and extremely limited solid solution. Illite is a white mica, intermediate in composition between pyrophyllite and muscovite, formed at much lower temperatures than muscovite. These relations show that illite is metastable relative to pyrophyllite + muscovite in all of its diagenetic and low-grade metamorphic occurrences. This further implies that illite precursor phases, such as smectite, are also metastable. The prograde reactions involving smectite, illite, and muscovite are therefore inferred to represent Ostwald-step-rule-like advances through a series of metastable phases toward the equilibrium states attained in the greenschist facies. “Illite crystallinity” can therefore be a measure of reaction progress, for which temperature is only one of several determining factors.
Transmission Electron Microscope Observations of Illite Polytypism
- Susan M. Baxter Grubb, Donald R. Peacor, Wei-Teh Jiang
-
- Journal:
- Clays and Clay Minerals / Volume 39 / Issue 5 / October 1991
- Published online by Cambridge University Press:
- 02 April 2024, pp. 540-550
-
- Article
- Export citation
-
Transmission electron microscopy (TEM), including selected area electron diffraction (SAED), has been used to identify polytypes in illite, phengite and muscovite from samples representing a wide range of diagenesis and low-temperature metamorphism. Samples include Gulf Coast sediments, sediments from the Salton Sea region, California, the Martinsburg Formation at Lehigh Gap, Pennsylvania, the Kalkberg Formation at Catskill, New York, Otago Schists from southern New Zealand, pelites from the Gaspé Peninsula in Quebec, Canada, shales and slates from Wales, sediments from the Barbados accretionary complex, and synthetic hydrothermal illite.
Samples from rocks of lowest grades, including those representing a range of sedimentary diagenesis, invariably give SAED patterns with few, complex non-00l reflections which are diffuse and ill-defined and that represent largely disordered stacking sequences. Corresponding XRD patterns are consistent with 1Md polytypism. The term 1Md is therefore retained for this material. Higher grade samples, including those in which slaty cleavage is developed, and detrital grains in low-grade sediments invariably give diffraction patterns of well-ordered 2- or 3-layer polytypes. Of all samples and localities studied, only one diffraction pattern, from a sample in the Gaspé sequence, was found to be predominantly 1M. In none of the other sequences included in this study were any 1M or predominantly 1M electron diffraction patterns obtained for illite grains.
Where illite is in its original state of formation, it is consistently 1Md, whether it originates as a result of direct crystallization from solution or as a replacement of smectite. Where illite has apparently undergone subsequent change, presumably through dissolution and crystallization representing an Ostwald-step-rule-like change, it occurs as a well-ordered 2-layer (inferred to be 2M1) or, less commonly, a 3T polytype. On the basis of this limited survey, the state of polytypism appears to directly identify illite as either being in, or changed from, its initial state of formation.
Transmission Electron Microscopic Study of the Kaolinitization of Muscovite
- Wei-Teh Jiang, Donald R. Peacor
-
- Journal:
- Clays and Clay Minerals / Volume 39 / Issue 1 / February 1991
- Published online by Cambridge University Press:
- 02 April 2024, pp. 1-13
-
- Article
- Export citation
-
Hydrothermally kaolinitized muscovite from the Otago schist of Brighton, New Zealand, has been studied by transmission and analytical electron microscopy (TEM and AEM) to determine the mechanism of alteration and to compare reactant-product relations for di- and trioctahedral micas. The muscovite is a primary metamorphic phase having a phengitic composition. It occurs as well-ordered, two- and three-layer polytypes, in grains as thick as 30 μm. Kaolinite occurs as packets of layers, each about 100–600 Å thick, which alternate with packets of muscovite or smectite-like layers. Most of the kaolinite is highly disordered in stacking sequence, although a one-layer polytype is also present, occurring as relatively thick sequences of layers. Phase boundaries between kaolinite and muscovite are invariably parallel to the 001 lattice fringes with no strain contrast; i.e., no transitions exist along layers. Parallelism of 00l and 11l reflection rows of both kaolinite and muscovite implies a topotaxial intergrowth. A smectitelike phase is also present, occurring as packets of wavy layers, which locally have periodic contrast that may reflect R1 ordering of illite/smectite. This material appears to be a direct, “along-layer” alteration product of muscovite. Electron diffraction data and lattice-fringe images imply that kaolinite alternates with micaceous phase(s) with some regularity; i.e., micaceous layers are separated by approximately equal numbers of kaolinite layers. Similar long-range periodicity occurs in contrast variations within packets of kaolinite layers.
The data collectively suggest that the alteration interface was self-perpetuating and that alteration proceeded rapidly along layers once it initiated in 2:1 layers at crystal edges or strained areas, with no observable component normal to the layers. They also suggest that smectite may have formed as an intermediate phase during the hydrothermal kaolinitization of muscovite. In the previous study of alteration of biotite in the same sample, “along-layer” transition boundaries were commonly observed, and a second, intermediate product phase was not detected, implying a relation between the alteration mechanisms and the chemical differences between reactants and products.
Transmission and Analytical Electron Microscopic Study of Mixed-Layer Illite/Smectite Formed as an Apparent Replacement Product of Diagenetic Illite
- Wei-Teh Jiang, Donald R. Peacor, R. J. Merriman, B. Roberts
-
- Journal:
- Clays and Clay Minerals / Volume 38 / Issue 5 / October 1990
- Published online by Cambridge University Press:
- 02 April 2024, pp. 449-468
-
- Article
- Export citation
-
Ordered illite/smectite (I/S) and illite in a pelitic rock from a prograde metamorphic sequence in North Wales were observed by transmission electron microscopy. The dominant phyllosilicate noted was diagenetic-metamorphic illite, occurring as subparallel packets of layers, each about a few hundred Ångstroms thick. It exhibited two-layer polytypism (presumably 2M1) and numerous strain features and had a composition of (K1.21Na0.12)(Al3.36Fe0.31Mg0.33)(Si6.28Al1.72)O20(OH)4.
I/S occurred as thick packets of wavy layers having 10-Å subperiodicity and sharp differences in contrast in successive lattice fringes. All stages in a replacement series were noted, from one or two layers of smectite within illite, through thin packets of I/S, to thick packets that contained inherited deformation textures of diagenetic-metamorphic illite. Deformed illite was replaced by I/S more commonly than was undeformed illite. The I/S replacing undeformed original illite had significantly greater order, primarily of R1 type (ISISIS…), than that replacing deformed illite. R> 1 I/S occurred as small crystallites and contained relatively less smectite than the ordered I/S, Single smectite layers were spaced within several illite layers, forming curved packets of layers. IISIIS… (R2) and IIISIIIS… (R3) ordering were present locally, as was discrete smectite. Analytical electron microscopic analyses indicated that the I/S, (K0.46Na0.43)(Al3.75Fe0.06Mg0.19)(Si6.26Al1.74)O20(OH)4, was rectorite-like in composition and had smaller (Mg + Fe) contents and greater Al/Si ratios than the coexisting illite, which was also anomalous in terms of general crystal-chemical relationships between coexisting illite and I/S in burial diagenesis environments. The I/S appears to have formed by replacement of diagenetic-metamorphic illite, presumably at very low temperatures under hydrous conditions via dissolution and crystallization.
Relation Between Interlayer Composition of Authigenic Smectite, Mineral Assemblages, I/S Reaction Rate and Fluid Composition in Silicic Ash of the Nankai Trough
- Harue Masuda, James R. O'Neil, Wei-Teh Jiang, Donald R. Peacor
-
- Journal:
- Clays and Clay Minerals / Volume 44 / Issue 4 / August 1996
- Published online by Cambridge University Press:
- 28 February 2024, pp. 443-459
-
- Article
- Export citation
-
The compositions, fabrics and structures of authigenic minerals that formed recently from silicic volcanic ash layers from a 1300-meter sediment column obtained at ODP Site 808 of the Nankai Trough were studied using XRD, STEM, AEM and SEM. Smectite and zeolites were first detected as alteration products of volcanic glass with increasing depth, as follows: smectite at 200 m below seafloor (mbsf) (20 °C), clinoptilolite at 640 mbsf (60 °C) and analcime at 810 mbsf (75 °C).
A primitive clay precursor to smectite was observed as a direct alteration product of glass at 366 mbsf (approximately 30 °C). High defect smectite with lattice fringe spacings of 12 to 17 Å and having a cellular texture filling pore space between altering glass shards occurs at 630 mbsf. Packets of smectite become larger and less disordered with increasing depth and temperature. The smectite that forms as a direct alteration product of volcanic glass has K as the dominant interlayer cation.
With increasing depth, smectite becomes depleted in K as the proportion of clinoptilolite increases, and then becomes depleted in Na as the proportion of analcime increases. The composition of the exchangeable interlayer of smectite appears to be controlled by the formation first of K-rich clinoptilolite and then Na-rich analcime, via the pore fluid, giving rise at depth to Ca-rich smectite. Smectite reacted to form illite in the interbedded shales but not in the bentonites. Paucity of K in smectite and pore fluids, due to formation of clinoptilolite under closed system conditions, is believed to have inhibited the reaction relative to shales.
Microstructures, Mixed Layering, and Polymorphism of Chlorite and Retrograde Berthierine in the Kidd Creek Massive Sulfide Deposit, Ontario
- Wei-Teh Jiang, Donald R. Peacor, John F. Slack
-
- Journal:
- Clays and Clay Minerals / Volume 40 / Issue 5 / October 1992
- Published online by Cambridge University Press:
- 28 February 2024, pp. 501-514
-
- Article
- Export citation
-
Transmission electron microscopy (TEM) was utilized to determine the origins of berthierine and chlorite in the core of the footwall alteration zone of the Kidd Creek massive sulfide deposit, Ontario. TEM images show lamellar intergrowths of packets of berthierine, mixed-layer chlorite/berthierine, Fe-Mg chlorite, and relatively Fe-rich chlorite that contain dislocations, stacking faults, kink bands, and gliding along (001). Interstratification of packets of berthierine and chlorite with one to several tens of layers commonly is associated with terminations of a layer of chlorite by two layers of berthierine. Layers in adjacent domains of berthierine and chlorite are continuous across interfaces that transect their common {001} planes. High-strain zones that cut across cleavage planes, consisting of distorted layers and lens-shaped pores, are associated with stacking faults and gliding along cleavage planes in chlorite crystals. Similar features separate interstratified chlorite/berthierine of different structures and textures, implying development of such composite grains after deformation of chlorite. Electron diffraction patterns show that the chlorite is an ordered one- or two-layer polytype or a one-layer polytype with semi-random stacking, and that the berthierine is a one-layer polytype with semi-random stacking epitaxially intergrown with chlorite.
Coexisting chlorite and berthierine have nearly identical ranges of compositions, containing Si ≅ 5, Al ≅ 6, and Fe ≅ 6.5–8.5 pfu, and minor, variable Mg and Mn contents, in formulae normalized on the basis of 20 total cations. This implies polymorphism among Fe,Al-rich members of the serpentine and chlorite groups. In one of the samples, berthierine and mixed-layer chlorite/berthierine coexist with chlorite having two compositional ranges, including Fe-rich chlorite with a relatively wide range of Fe-Mg contents, and a dominant Fe-Mg chlorite. In another sample, compositionally homogeneous Fe-rich chlorite is associated with berthierine and mixed-layer chlorite/berthierine; Fe-Mg chlorite was not detected.
The microstructural relations and the presence of coexisting polymorphs, complex mixed layering, heterogeneous polytypism, and wide ranges of mineral compositions are consistent with replacement of chlorite by berthierine under non-equilibrium retrograde conditions, in contrast to the generally assumed prograde origin for other berthierine occurrences.
Chlorite Geothermometry?—Contamination and Apparent Octahedral Vacancies
- Wei-Teh Jiang, Donald R. Peacor, Peter R. Buseck
-
- Journal:
- Clays and Clay Minerals / Volume 42 / Issue 5 / October 1994
- Published online by Cambridge University Press:
- 28 February 2024, pp. 593-605
-
- Article
- Export citation
-
The large contents of octahedral vacancies in published formulae of chlorite from hydrothermal systems and clastic sequences are shown to be largely caused by inclusion of other minerals. Verification is provided by analytical electron microscope (AEM) analyses of chlorite in pelitic rocks from the Gaspé Peninsula, Quebec and the Gulf Coast, Texas. The Gaspé chlorite occurs as discrete crystals locally coexisting with corrensite, and the Gulf Coast chlorite is free of mixed layers other than local serpentine-like 7-Å layers. Unlike most electron microprobe analyses (EMPA), but like other AEM analyses, the reported chlorite formulae do not have significant octahedral vacancies, are not Si-rich and (Fe + Mg)-poor relative to classic metamorphic chlorite, and have nearly equal amounts of tetrahedral and octahedral Al. The studied chlorites and those in metabasites and clastic rocks that could be positively identified as containing no or minimal mixed layering or submicroscopic intergrowths have little or no Ca or alkalis. In contrast, EMPA of chlorite reported for other clastic sequences show variable amounts of Na + K + 2Ca that exhibit a poorly defined positive correlation with the proportion of octahedral vacancies. The EMPA of chlorite from the Salton Sea and Los Azufres geothermal fields that were suggested to contain temperature-dependent amounts of tetrahedral Al (and thus used as “chlorite geothermometers”) show compositional characteristics similar to those reported for several saponite-chlorite transition series in metabasites.
Continuous increases in octahedral occupancy and tetrahedral Al with increasing metamorphic grade are attributed to decreases in abundance of mixed layers or fine-grained intergrown minerals that commonly occur as a result of increasing crystal size and homogeneity in prograde sequences. Use of “chlorite geothermometry” based on the proportion of apparent octahedral vacancies or tetrahedral Al is therefore unwarranted and leads to inaccurate temperature estimates.
Clay Minerals in the MacAdams Sandstone, California: Implications for Substitution of H3O+ and H2O and Metastability of Illite
- Wei-Teh Jiang, Donald R. Peacor, Eric J. Essene
-
- Journal:
- Clays and Clay Minerals / Volume 42 / Issue 1 / February 1994
- Published online by Cambridge University Press:
- 28 February 2024, pp. 35-45
-
- Article
- Export citation
-
Clay minerals from the MacAdams Sandstone, Kettleman North Dome, California, have been studied by electron microscopy. The clay minerals fill pore space associated with fractured and brecciated clasts of K-feldspar. Curved packets of muscovite and kaolinite are caused by deformation of detrital muscovite that resulted in opening of fissures subsequently filled with dominant kaolinite and minor intergrown mixed-layer illite/smectite (I/S). Regions of authigenic R1 I/S (rectorite) with characteristic ~20 Å periodicity are intergrown with kaolinite in microfissures within K-feldspar or detrital muscovite. Clusters of small grains of muscovite with nearly ideal composition occur as stacks and intergrown with kaolinite and are tentatively inferred to be authigenic. Contrary to previous reports, no illite was found in these samples.
Electron microprobe analyses previously obtained on Kettleman Dome “illite” and subsequently used as a prime example of analyses of illite rich in excess interlayer water (H2O) and hydronium ion (H3O+) are shown to have been obtained on mixtures, and are not representative of the actual clay mineral compositions. Previous conclusions regarding significant H3O+ and H2O contents of illite are invalid because of inaccuracies inherent in bulk and EMPA analyses of illite, and do not affect arguments regarding the metastability of illite. Hydronium substitution should be favored via the reaction H2O + H+ = H3O+ only in highly acidic fluids. Ordinary illite forming in sedimentary environments with carbonates and iron oxides is unlikely to have significant H3O+ substituted for K+.
Prograde Transitions of Corrensite and Chlorite in Low-Grade Pelitic Rocks from the Gaspé Peninsula, Quebec
- Wei-Teh Jiang, Donald R. Peacor
-
- Journal:
- Clays and Clay Minerals / Volume 42 / Issue 5 / October 1994
- Published online by Cambridge University Press:
- 28 February 2024, pp. 497-517
-
- Article
- Export citation
-
A prograde sequence of corrensite and chlorite in pelitic rocks of the diagenetic zone, anchizone, and epizone (illite crystallinity indices = 0.17–0.58°Δ2θ) of the Gaspé Peninsula, Quebec, was studied by analytical and transmission electron microscopy (AEM and TEM). The data collectively suggest that diagenesis/metamorphism of chlorite and corrensite follows a sequence of phase transitions, compositional homogenization, and recrystallization, approaching a state of equilibrium for which chlorite is the stable phase.
Corrensite occurs as coalescing, wavy packets of layers intergrown with chlorite and illite in the diagenetic and low-grade anchizonal rocks. Intergrowths of discrete chlorite and corrensite crystals, interstratified packets of chlorite and corrensite layers, terminations of smectite-like layers by chlorite layers, and 2–3 repeats of R2- and R3-ordered chlorite-smectite mixed layers occur. These materials are alteration products of detrital biotite or other precursor phases like trioctahedral smectite. The crystal size and proportion of corrensite decrease significantly from the diagenetic zone to the anchizone. Deformed corrensite is crosscut by straight packets of chlorite and corrensite in the diagenetic sample. Some chlorite occurs as discrete, euhedral to subhedral crystals intergrown with or enclosed by other phases in the absence of corrensite. The crystal size of chlorite and definition of crystal boundaries increase whereas density of crystal imperfections and randomness in orientation decrease with increase in grade of diagenesis/metamorphism. Crystals that are kinked or bent, or display gliding along (001) form low-angle boundaries with relatively defect-free crystals, implying deformation during crystal growth. Abundant well-defined low-angle boundaries associated with dislocations are observed in the higher grade rocks, consistent with a stage of readjustment of crystal boundaries during crystal growth. The AEM analyses show that the corrensite has lower Fe/(Mg + Fe) and Al/(Si + Al) than the coexisting chlorite in the diagenetic sample, and that the ranges of composition of chlorite of different grades overlap and become smaller with increasing grade, implying prograde homogenization.
The data imply that corrensite is a unique phase that is metastable relative to chlorite: its conversion to chlorite occurred at a grade as low as that of the high-grade diagenetic zone. The textural relations suggest that the metamorphic crystallization and recrystallization were coeval with deformation processes due to tectonism, partially modified by subsequent contact metamorphism. The data, combined with those of previous reports, suggest that the Gaspé Ordovician rocks constitute a part of a regional distribution of trioctahedral phyllosilicate-rich rocks in the northern Appalachians. The regional occurrence of abundant chloritic minerals is thus directly related to a specific tectonic regime with precursor sediments largely derived from an andesitic arc system(s).
FTIR and XRD Investigations of Tetracycline Intercalation in Smectites
- Zhaohui Li, Vera M. Kolb, Wei-Teh Jiang, Hanlie Hong
-
- Journal:
- Clays and Clay Minerals / Volume 58 / Issue 4 / August 2010
- Published online by Cambridge University Press:
- 01 January 2024, pp. 462-474
-
- Article
- Export citation
-
Due to swelling, smectite minerals are capable of intercalating many organic molecules in their interlayer space. Tetracycline (TC) is a group of antibiotics used extensively in human and veterinary medicine. The great aqueous solubility and long environmental half life of TC mean that the study of interactions between swelling clay minerals and TC are of great importance in TC transport and retention in subsurface soils. In the present study, the intercalation of TC molecules at different levels into smectites was investigated using Fourier transform infrared spectroscopy (FTIR) and X-ray diffraction (XRD). The shift of the FTIR bands of amide I and II in comparison to crystalline TC suggested a strong interaction between the amide groups and the clay surfaces. The band at 1455 cm−1 remained the same after TC intercalation into SAz-1, SWy-2, and SYn-1, suggesting that complexation was not a dominant mechanism of TC uptake by these minerals. With cation exchange as the major mechanism of TC intercalation into these minerals, simultaneous removal of H+ from solution protonated the TC molecules and provided a positive charge to interact with negatively charged mineral surfaces even in neutral to slightly alkaline conditions. The increase in interlayer distance after intercalation by TC, as revealed by XRD, suggested a tilted orientation of the intercalated TC molecules in both twisted conformation in acidic condition and extended conformation in alkaline condition.
Interlayer conformations of intercalated dodecyltrimethylammonium in rectorite as determined by FTIR, XRD, and TG analyses
- Zhaohui Li, Wei-Teh Jiang
-
- Journal:
- Clays and Clay Minerals / Volume 57 / Issue 2 / April 2009
- Published online by Cambridge University Press:
- 01 January 2024, pp. 194-204
-
- Article
- Export citation
-
Intercalation of a wide variety of alkylammonium cations into the interlayer spaces of swelling clay minerals leads to many different applications, ranging from surface-charge measurements to rendering the clay compatible for the preparation of clay nanocomposites, but knowledge of the exact conformation of the intercalated organic species is still incomplete, thus preventing a full understanding of this process. The purpose of this study was to investigate the interlayer conformation of dodecyltrimethylammonium (DDTMA) bromide in rectorite as affected by the amounts of DDTMA intercalated, using a battery of physical and spectroscopic methods. The capacity of rectorite to intercalate DDTMA was equivalent to 1.67 times the cation exchange capacity (1.67 CEC) of the mineral even though the initial input was as much as 5.00 CEC. When the DDTMA intercalated was <0.50 CEC of the mineral, minimal counterion sorption was associated with DDTMA intercalation. Derivative thermogravimetric (DTG) analyses revealed a single-peak decomposition temperature (Tpeak) at 430°C. X-ray diffraction (XRD) analyses indicated a flat-lying monolayer of the intercalated molecules, while shifts in Fourier Transform infrared (FTIR) bands confirmed gauche conformation. These results suggested that cation exchange was the dominant mechanism. At the maximum intercalation, the DDTMA adopted a horizontal trilayer arrangement with mainly gauche conformation as determined by FTIR and XRD. Meanwhile a second Tpeak appeared at 255°C, similar to the Tpeak of solid DDTMA. Counterion bromide sorption accompanying DDTMA intercalation reached a capacity of 310 mmol/kg. The results indicated that van der Waals interaction was responsible for the DDTMA uptake at the amount beyond 0.50 CEC. When the amount of DDTMA intercalated was between 0.5 and 1.67 CEC, the XRD patterns showed non-integrality, i.e. the 002 reflection was split into two non-integral peaks with 2 × d002 < d001 and 3 × d003 > d001. They became integral at 1.67 CEC. The results suggest that the mineral might be composed of mixed layers of a monolayer intercalated rectorite and a trilayer intercalated rectorite, without a bilayer as intermediate, when the amount of DDTMA intercalated was between 0.5 and 1.67 CEC.