Introduction
Alzheimer’s disease (AD) is a devastating, incurable, neurodegenerative condition experienced by more than 600,000 Canadians. 1 The disease is prevalent in 10%–30% of people over 65 years of age. Reference Masters, Bateman, Blennow, Rowe, Sperling and Cummings2,3 More than 95% of cases are sporadic and occur late in life, with pathophysiological hallmarks of accumulated amyloid-β (Aβ) peptide and neurofibrillary tangles of tau protein in the brain. Reference Masters, Bateman, Blennow, Rowe, Sperling and Cummings2 Preclinical disease may be present for decades before the onset of clinical signs and symptoms which, once overt, typically progress over 8–10 years, ultimately leading to death from complications. Reference Masters, Bateman, Blennow, Rowe, Sperling and Cummings2
The burden of neurological conditions is high, with affected Canadians utilizing more healthcare personnel support than Canadians with other chronic health condition(s). 4 The patient, caregiver, and health system burden of dementia is heavy. Patients with dementia experience worsening deficits in cognitive, emotional, and physical function, and loss of independence. Reference Tahami Monfared, Byrnes, White and Zhang5 Caregivers, many of whom are elderly and also at risk, contribute substantial hours to the care of people with AD and experience negative quality of life impacts as the disease progresses. Reference Tahami Monfared, Byrnes, White and Zhang5,6 A 2016 study estimated combined Canadian healthcare system costs and out-of-pocket caregiver costs to be $10.4 billion; an amount expected to grow to $16.6 billion by 2031. 6 Growth in burden is primarily attributed to a growing prevalence of disease, with case numbers expected to double over a 20 year period. Reference Manuel, Garner and Finès7 A 2019 global burden of disease study estimated that in Canada, dementia-related spending accounted for 2.3% of total healthcare spending and would increase by 5.5% annually. Reference Pedroza, Miller-Petrie and Chen8
While clinical criteria have been the primary basis for the diagnosis of AD dementia, Reference McKhann, Knopman and Chertkow9 biological definitions of AD, based on biomarkers of Aβ deposition, pathologic tau, and neurodegeneration, have been used in research settings. Reference Ismail, Black and Camicioli10 In Canada, national guidelines for the diagnosis of AD suggest that most screening and clinical workup be completed by a primary care physician (PCP), and that more advanced diagnostics require referral to specialty clinics. Reference Ismail, Black and Camicioli10 Sadly, studies suggest that less than half of those living with dementia receive a diagnosis, so many progress while awaiting a diagnosis. Reference McGrath, Robinson-Lane, Clark, Suhr, Giordani and Vincent11–Reference Russell, Banerjee and Watt13 Furthermore, current Canadian guidelines discourage screening for mild cognitive impairment (MCI), a common precursor to AD dementia. 14
AD management mainly involves the treatment of comorbid illnesses and extensive social support. Reference Masters, Bateman, Blennow, Rowe, Sperling and Cummings2 Until recently, although there had been continued progress in understanding AD pathophysiology, a disease-modifying therapy (DMT) remained elusive. Reference Anderson, Sathe and Polacek15,Reference Morató, Pytel, Jofresa, Ruiz and Boada16 However, a number of drugs aimed at modifying the disease in its earlier stages (i.e. MCI due to AD and mild AD dementia) are currently under investigation or have recently reported promising results. Reference Morató, Pytel, Jofresa, Ruiz and Boada16–Reference van Dyck, Swanson and Aisen18 Many of these treatments target Aβ peptide, two of which have received US Food and Drug Administration approval for AD treatment: aducanumab and lecanemab. 19,20 Aβ-targeted therapies require detection of Aβ aggregation using cerebrospinal fluid (CSF) or positron emission tomography (PET) to determine treatment eligibility. Reference Masters, Bateman, Blennow, Rowe, Sperling and Cummings2,Reference McDade21–Reference Gauthier, Rosa-Neto, Morias and Webster25 Unfortunately, low numbers of dementia specialists and access to imaging already lengthen wait times for such services in Canada. Reference Mattke and Hanson26
The promise of DMTs for AD is bittersweet, considering the challenges anticipated from a health system perspective. Experts across the globe have raised concerns that current infrastructure is not sufficient for the anticipated demand for early AD (eAD) diagnostics. Reference Gauthier, Rosa-Neto, Morias and Webster25–27 Furthermore, in Canada, healthcare resources in neurology and dementia care are already strained or scarce, as has been highlighted in numerous reports. Reference Aminzadeh, Molnar, Dalziel and Ayotte28–Reference Meloff33
In alignment with implementation science and practice improvement, Reference Leeman, Rohweder and Lee34 we sought to quantify and characterize the expected capacity constraints related to DMT availability for patients with eAD in Canada. By understanding where bottlenecks exist, health systems and policy makers can make more informed decisions related to future models of care and care investment.
Methods
Study Design and Objectives
A comprehensive capacity model, similar to system dynamics modeling, Reference Cassidy, Singh and Schiratti35 was developed in Microsoft Excel to model the healthcare capacity in Canada for treatment of eAD with Aβ-targeted DMT. The primary objective of the study was to characterize the healthcare capacity to diagnose, assess eligibility, and treat prodromal and mild AD patients with an Aβ-targeted DMT in Canada. Secondary objectives included estimating the number of patients who would not receive a DMT due to resource constraints, identifying the resource bottlenecks constraining the flow of patients along the AD treatment journey, and quantifying the healthcare resources (personnel and infrastructure) needed to overcome any identified bottlenecks.
The research question, study design, and data inputs were validated by a steering committee, which included three AD experts from major regions in Canada (Western Canada - HN, Ontario - SB, Quebec - LV) and one representative from the patient support community (LTW). The study was designed between May and October 2022 with data outputs finalized in December 2022. The protocol for this modeling project was reviewed by Advara, an institutional review board (IRB), and was deemed exempt from IRB oversight.
Study Setting
The model was based on health system configurations and delivery of care common in Canada, where health care is largely public-funded and provincially delivered. Reference Martin, Miller, Quesnel-Vallée, Caron, Vissandjée and Marchildon36 Canada, the second largest country in the world, is a geographically vast and regionally diverse country. This study included all Canadian provinces; however, due to lack of available data, it excluded the territories in the less populated northern areas of the country.
Overall, model assumptions were based on steering committee input, clinical trial design of Aβ-targeted DMTs under investigation for eAD, 37 and current Canadian guidelines. Reference Ismail, Black and Camicioli10 Healthcare innovations not yet available for clinical use (e.g., diagnostic blood-based biomarkers, BBBMs) were not included. Reference Ismail, Black and Camicioli10,Reference Hansson, Edelmayer and Boxer38 Similarly, the clinical trial population was used as reference for patients eligible for treatment. For the model, patients were considered eligible for Aβ-targeted DMT if they had MCI or mild AD, and tested Aβ positive (i.e., by increased amyloid PET or decreased CSF-Aβ42).
Generally, each input variable required data supported by, in order of priority, published scientific literature, clinical trial protocol, public data sources, or steering committee consensus. Every effort was made to identify the most relevant and recent data specific to Canada, first provincially, and then nationally. Average consensus of the steering committee was used for parameters where no data were currently available (e.g., time to treat an AD patient with Aβ-targeted DMT).
Model configuration and assumptions
The model calculated the capacity gap as the difference between the total required resources and the currently available resources, for each step of the patient journey (diagnosis, eligibility, and treatment/monitoring). The model used a 3-year time horizon, assuming introduction of Aβ-targeted DMTs in year 1.
There were three essential elements of the model:
[ Number of patients (Capacity demand) x Resource required per patient ] – Available healthcare capacity = Capacity Gap
-
1. Healthcare capacity demand (i.e., prevalence, diagnostic rate, Aβ positivity rate, treatment rate, market shares; see Fig. 1),
-
2. Healthcare resources required (i.e., assignment of resource allocations or patient slots to various personnel or infrastructure)
-
3. Available healthcare capacity (i.e. number of health system personnel and infrastructure resources available, estimate of time available).
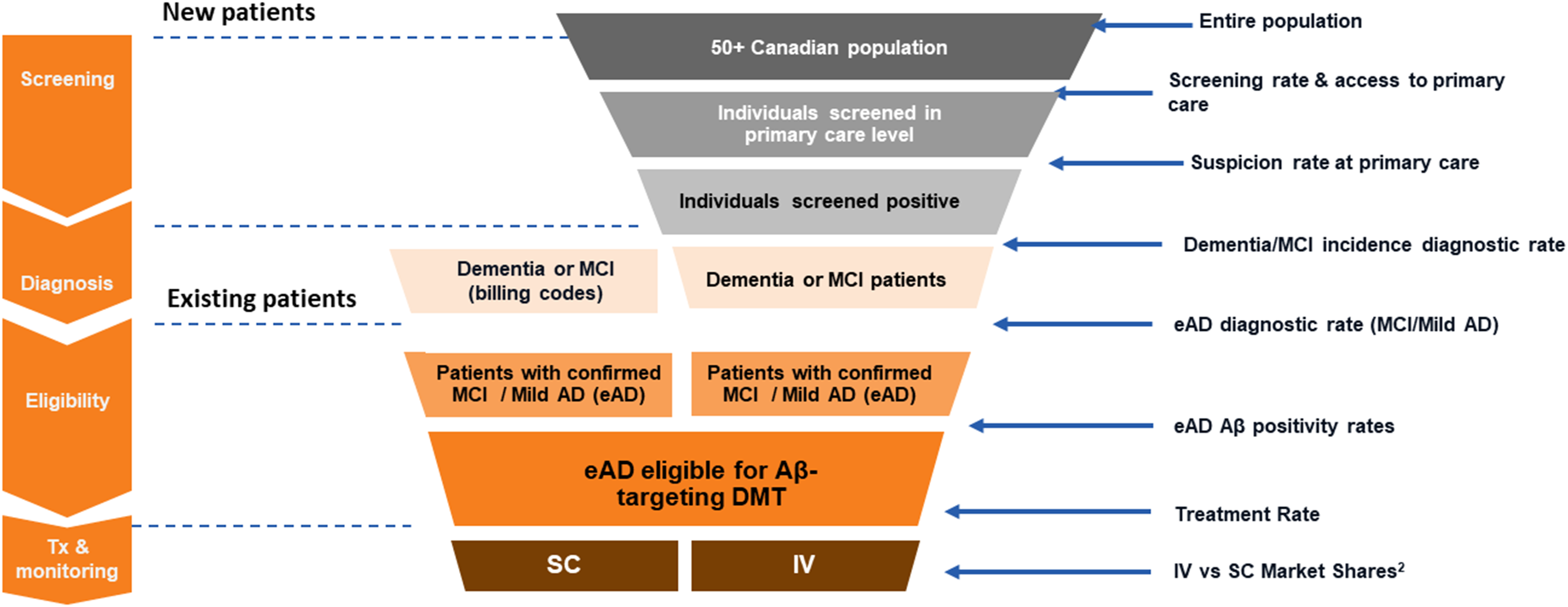
Figure 1: An illustration of the patient journey, from screening and diagnosis of early AD (eAD) to assessment of eligibility and treatment with an Aß-targeted DMT, estimating the expected demand at each step of the funnel.
Healthcare capacity demand . The demand on the health system was determined based on the number of individuals expected to require access to healthcare personnel or resources. A patient journey funnel (Fig. 1) was designed to quantify the number of individuals flowing through the healthcare system toward treatment with an Aβ-targeted DMT. The model had a total of four stages (screening, diagnosis, eligibility, and treatment/monitoring) and two streams of patients. The two streams of patients were either new prevalent patients (i.e., an undiagnosed, prevalent pool of patients not currently managed by the healthcare system) or existing prevalent patients (i.e., diagnosed). Incident new patients were not captured in the model. Within the new patient stream, demand and associated rates (e.g., screening, suspicion, diagnosis) were assumed to be stable over the 3-year horizon of the model.
Assumptions related to healthcare capacity demand are detailed in Supplementary Table S1. Potential patients were assigned provincially, based on population distributions 39 after which applicable rates related to screening, diagnosis, eligibility, and treatment were applied nationally.
Healthcare resources required . At each stage of the demand funnel, individuals were assumed to require access to various healthcare resources, captured in units of time (personnel) or slots (infrastructure) required per patient. Personnel included AD specialists (neurologists, geriatricians, or psychiatrists with a focus in dementia care), AD nurses, pharmacists, imaging specialists (nuclear medicine specialists and radiologists), and technologists. For simplicity, some supportive resources (e.g., administrative, lab, and social services, intravenous infusion) were assumed to be unconstrained and were not included in the model. Infrastructure requirements included magnetic resonance imaging (MRI) and PET exam slots.
Assumptions related to healthcare resources required are detailed in Supplementary Table S2.
Available healthcare capacity . For each resource, an estimation of available time or exams/tests, based on the current health system, was incorporated in the model. Available resources were distributed to each step of the patient journey by type (i.e., AD specialist, nurse, PET, etc.), in a ratio proxying the required resources for each step. With the exception of PCPs, all resources were allocated provincially.
Inputs related to available healthcare capacity are detailed in Supplementary Table S3. After quantification of each available resource, an assumption regarding the proportion of time dedicated to AD was required. The steering committee agreed on an allocation for AD specialists: 10% of all neurologists and geriatricians and 2.3% of psychiatrists. Reference Pedroza, Miller-Petrie and Chen8 For all other healthcare resources (personnel and infrastructure), allocation of resources to dementia care was estimated using Canadian AD expenditure as a fraction of entire healthcare expenditure (2.3%), as a proxy estimate. Reference Pedroza, Miller-Petrie and Chen8
Model simulation / Data analysis reporting
Microsoft Excel was used for modeling, data analysis, and visualization.
Flow through the system was supported or constrained by the resources required and available within each step. The model used the total potential patient demand, regardless of prior bottleneck (i.e., satisfied + unsatisfied) as the denominator to begin each step of the patient journey. The capacity gap within each step was computed as the number of patients for whom demand was satisfied versus unsatisfied in that step, with proportions reported as a function of the potential patients that could have entered that step. The unsatisfied demand of patients forming in a single treatment journey step (i.e., waitlist) was added to the following year’s demand for that step.
Modeling assumed that a DMT treatment was only prescribed to a patient if there was available capacity for monitoring that patient. Mortality and treatment discontinuation were not included in the model, as they were assumed to be negligible over the short 3-year model horizon. Neither endogenous increases in available resources over time nor resources required for non-DMT eligible patients were included in the model.
For national analyses, data from all provinces were combined to provide Canadian capacity estimates. Results are reported using descriptive statistics. Human resource requirements are reported in units of full-time equivalent (FTE) with one FTE equal to 37.5 hours/week and 48 weeks/year. Reported patient numbers are rounded to the nearest 1,000 with the intention to illustrate directional estimates rather than specific numbers generated from modeling using extensive input assumptions with inherent variance.
Sensitivity Analysis
Based on the design of the model, there was an opportunity to perform sensitivity analyses where assumptions were uncertain or where changes in the care configuration were expected to result in capacity improvements. Two such scenarios were tested, independently. In one analysis, allocation of healthcare resource time (FTE) and infrastructure (slots) was increased from the base assumption of 2.3% to the upper limit of the reported range for percentage of total healthcare expenditure (i.e., 4.6%). Reference Pedroza, Miller-Petrie and Chen8 In the second scenario, Aβ testing was shifted from 50% PET and 50% CSF to 100% CSF. Increased potential capacity was reported for each of these scenarios as described above.
Results
Capacity Gaps Related to the Provision of Aβ-targeted DMTs in eAD
In modeling resources available, anticipated patient demand, and required resources for the treatment journey, the model estimated that only 6.89%, 1.15%, and 1.14% of potential patients nationally would have access to diagnosis, DMT eligibility assessment, and DMT treatment/monitoring, respectively, in year 1 (Fig. 2).

Figure 2: Percentage of patients with access to diagnosis, eligibility assessment, and treatment/monitoring with an Aß-targeted DMT for early AD, in Canada (year 1 following the introduction of a DMT). Nationally, <2% of patients will have access to the required healthcare resources for treatment with an Aß-targeted DMT in year 1.
Ontario, Manitoba, and Saskatchewan were the provinces with the highest proportion of patients (8.6%, 8.6%, and 8.3%, respectively) with access to resources for diagnosis. In contrast, these provinces were among those with the lowest proportion of patients (< 1.0%) having access to eligibility assessment (see Supplementary Table S2). This drop in capacity was a result of increased patient demand, owing to the entry of prevalent patients with existing MCI or mild AD diagnoses into the model. Eligibility assessment, which could include either PET or CSF testing (including imaging specialist and technologist time), AD specialist assessment, and AD nurse support, represented the step with the largest capacity gap across all provinces (see Supplementary Table S4).
The number of Canadians anticipated to be waiting for eligibility assessment after year 1 was approximately 382,000. This number grows annually for each step of the patient journey (data not shown). All provinces are expected to have waitlists for eligibility in year 1, with the highest being in Ontario (approximately 148,000 patients) (Fig. 3).

Figure 3: Anticipated provincial waitlist for eligibility assessment in year 1, following Aß-targeted DMT introduction. Approximately 382,000 Canadians could be awaiting eligibility assessment following introduction of a DMT.
Bottlenecks Constraining Capacity and Top Resources Required for Aβ-targeted DMT Provision in eAD
Nationally, and considering the entire patient journey, AD specialist time represents the largest capacity gap, equating to a 14-times increase in FTE, amounting to over 3,500 more AD specialists, required to satisfy demand in year 1. However, MRI and PET slot availability is the largest limiting resource constraining diagnosis and eligibility along the patient journey. This finding was largely similar when detailed by province (data not shown).
The difference between required and available resources (i.e., incremental resource required) for MRI and PET slots equates to nearly 26- and 86-fold increases in slots, respectively, in year 1 (data not shown).
Sensitivity Analysis Findings
Shifting from 50% PET / 50% CSF to 100% CSF for detection of Aβ resulted in an increased capacity to assess patients for DMT eligibility across all provinces (Fig. 4), opening access for nearly 47,000 patients nationally in year 1.

Figure 4: Impact of shifting from 50% CSF / 50% PET for Aβ determination compared to 100% CSF.
Increasing the time that ancillary resources are dedicated to AD care also increased capacity to assess patients for DMT eligibility across all provinces (Fig. 5), opening access for nearly 6,000 patients nationally in year 1.

Figure 5: Impact of increased ancillary resources on capacity to assess eligibility for an Aß-targeted DMT (year 1).
Interpretation
Summary of Key Findings
Results of this model simulation show the Canadian healthcare system has an extremely low capacity to diagnose, assess eligibility, and treat (<7% of potential patients) eAD with an Aβ-targeted DMT. Importantly, our findings magnify the issues highlighted by a previous Canadian model 32 and corroborate findings from other countries, including the USA Reference Anderson, Sathe and Polacek15 and Brazil. Reference Mattke and Hanson26 Indeed, our data underscore the need for AD specialist resources called for by Liu et al. 32 in their modeling, especially considering only a fraction of Canadian neurologists, geriatricians, and psychologists specialize in AD care. Combined, these modeling data demonstrate that the capacity of the Canadian healthcare system is grossly inadequate across all stages of the eAD patient journey and that national policy and investment supporting AD care will be required to realize the benefit of DMTs in this space.
Further, considering the large bolus of prevalent patients expected to present for Aβ testing initially, eligibility assessment represents the most constrained step along the patient journey to treatment with an Aβ-targeted DMT. In this step, imaging specialist FTE and PET imaging slots impart the greatest limitations to the flow of patients. In fact, capacity limitations for eligibility assessment predict a wait list of nearly 382,000 patients just 1 year after the introduction of an Aβ-targeted DMT. These findings differ in scale compared to the findings from the disease state and system dynamics model reported by Liu et al. 32 In their study, the year 1 waitlist was less than 100,000 patients for biomarker (eligibility) assessment. This gap widens when accounting for imaging specialist time and AD sub-specialization; additional considerations that reflect important constraints with respect to available and required resources within our model.
Our analysis also provides a unique view of the provincial nuances related to capacity constraints. Given the diversity of provincial challenges and health system models, solutions for increasing capacity will need to be customized and implemented at a provincial level but should focus on AD specialist resourcing and improving access to imaging. 40 Of particular concern and need for consideration will be the equitable provision of care, including access to imaging for diagnosis or eligibility assessment, in remote or rural populations. Reference Leeman, Rohweder and Lee34
One solution to this bottleneck is to decrease reliance on PET (and thereby imaging specialist time) and increase the use of CSF for Aβ assessment, as explored in the sensitivity analysis. Such a shift allowed the assessment of nearly 47,000 additional patients nationally in year 1. Alternately, increasing the proportion of time ancillary resources allocate to dementia care (proxied by an increase in the proportion of healthcare expenditure for dementia care) allowed for close to 6,000 additional patients to be assessed for Aβ-targeted DMT eligibility. While these analyses highlighted potential priority areas for action, it should be recognized that major gaps in capacity remained, and the solutions required to adequately support disease-modifying treatment in eAD will be varied and systemic.
Limitations of the Study
Modeling complex systems has inherent limitations as flows are simplified and best assumptions are made. In this model, key limitations included the focus on a single, presumably prominent model of care. This meant potential capacity gains or losses true for other models of care (e.g., family health teams, centralized image interpretation, etc.) were not accounted for. In addition, the assumptions made for each variable provincially/nationally may not be accurate representations of the provincial/national reality. For example, a national assumption of 50% PET / 50% CSF does not accurately reflect the clinical reality of care in a province where PET is not funded. As was seen with the shift to 100% CSF testing in the sensitivity analysis, alternate resourcing configurations can appreciably impact patient access. Importantly, as data become available at more accurate and detailed levels, the model can be adjusted and re-run.
Another limitation is of course the scope of the model and resources not considered. As an example, dementia care in the Yukon (and other northern regions not included in the model) is unique (e.g., prominent role of nurse practitioner) and remote/rural access challenges are expected to be compounded in these areas. Likewise, resources not accounted for in the model, such as social worker support, should be considered as capacity solutions are contemplated. Additional questions require further study, including the capacity and need for social support, the resources required to manage diagnosed but non-DMT eligible patients, the impact of the pandemic on various assumptions, and the potential influence of blood-based biomarker use for clinical AD diagnosis.
Next Steps
This study can serve as a framework for modeling capacity needs related to future DMTs and changes to clinical care (e.g., BBBMs for the identification of Aβ) for AD, other neurological conditions, and beyond. Presumably, the future clinical use of validated and robust blood-based biomarkers in AD has the potential to not only simplify the diagnosis and evaluation of DMT eligibility but also enable a timelier assessment at the primary care level and a triaging of demand for amyloid PET and specialty consult. With the diagnostic accuracy of assays for phosphorylated-tau improving (particularly p-tau217), Reference Hansson, Blennow, Zetterberg and Dage41 the reality of BBBMs impacting the AD patient pathway to treatment is near. In theory, this would lessen the strain on key bottlenecks identified in our model (i.e., imaging resources and AD specialist time) but would require thorough consideration of other resource capacities (i.e., primary care). For now, stakeholders involved in AD care must heed the red flags raised by this and other research and consider the multitude of health system changes required to provide eligible patients with DMT early in their disease trajectory.
We also encourage complementary research such as investigating alternative models of care to help identify impactful interventions for capacity building. Reference DeCorby-Watson, Mensah, Bergeron, Abdi, Rempel and Manson42,Reference Crisp, Swerissen and Duckett43 Further, while some models have predicted overall economic benefit associated with DMT-driven delays in disease progression Reference Tahami Monfared, Tafazzoli, Ye, Chavan, Deger and Zhang44–Reference Boustani, Doty and Garrison46 it will be important to evaluate the economic implications of modified models of care or resource allocation in the context of the clinical benefit realized by a DMT. Reference Angrist, Yang, Kantor and Chiba-Falek47 Furthermore, the true impact of new interventions for eAD will only become clear once people living with AD enter into and pressure the existing health system. With an abundance of health, social, and economic considerations at play, the reshaping of AD care infrastructure and appropriate DMT provision will require a variety of solutions, including informed resource allocation, in order to deliver value.
Conclusion
In Canada, the modeled system of AD care and currently allocated resources are insufficient to support the provision of Aβ-targeted DMTs for patients with or seeking an eAD diagnosis. These capacity constraints are driven by the increased demand for services and treatment, coupled with limited resources. Resources particularly lacking include AD specialists and imaging availability. This model highlights the urgent need for increased national policy and provincial resource allocation to support AD diagnosis, eligibility assessment, and treatment/monitoring.
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1017/cjn.2023.270.
Acknowledgments
The authors thank Hany Khalifa, Debbie Knifton, Mikele Epperly, Ilke Mirik, and Valerie Crowell (F. Hoffmann-La Roche Ltd), as well as the team at Life Science Consulting for their contributions to the study design, execution, and data analysis. We also appreciate the support provided by Stevie Kenyon of Placencia Holdings Ltd (Hamilton, ON, Canada) for data analysis and medical writing.
Statement of authorship
NB, SV, and MW contributed substantially to study conception and data acquisition. SEB, NB, HBN, LV, SV, LTW, and MW contributed to the study design and confirmation of acquired data. NB and MW conducted the primary data analysis. SEB, NB, HBN, LV, SV, LTW, and MW were responsible for data interpretation and the critical review of the draft and final manuscript, and all approved the final version for submission.
Funding
This study and its publication, including medical writing provided by Stevie Kenyon (Placencia Holdings Ltd.), were funded by F. Hoffmann-La Roche Ltd. (Mississauga, ON, Canada).
Author disclosures
Dr Sandra Black has received consultancy fees from Biogen, Hoffmann-La Roche Ltd., and NovoNordisk; speaker fees from Biogen; and support for this study, as noted above. She has also received in kind support from GE Healthcare and Lilly-Avid pharmaceuticals for observational studies using amyloid PET across different dementias. On behalf of her institution, Dr Black has acted as investigator for contract research and a peer reviewer, supporting various private and public organizations. Dr Black is also an unpaid board member of the Conference Board of Canada World Dementia Council, the University of Rochester Contribution to the Mission and Scientific Leadership of the Small Vessel VCID Biomarker Validation Consortium, the National Institute of Neurological Disorders and Stroke, and the Ontario Dementia Care Alliance.
Dr Haakon Nygaard has received consultancy fees from Hoffmann-La Roche Ltd and support for this study, as noted above.
Laura Tamblyn Watts is an unpaid board member of Elder Abuse Ontario and is a partial recipient of an academic grant from Alzheimer’s Society of Canada. She has received paid travel expenses from Hoffmann-La Roche Ltd and Pfizer to attend and present at meetings. Laura is also president and CEO of CanAge, a not-for-profit organization which has received unrestricted and targeted educational grants from Hoffmann-La Roche Ltd., Pfizer, Sanofi, GlaxoSmithKline, Johnson & Johnson; speaker fees from various community-based organizations, professional associations, regulators, and for-profit organizations; and consultancy fees from Hoffmann-La Roche Ltd. CanAge also received support for this study, as noted above.
Dr Louis Verret has acted as Principal Investigator in clinical trials in Alzheimer’s disease and was accordingly paid by Hoffmann-La Roche Ltd., Biogen, IntelGenX, and NovoNordisk; he received personal fees for consulting work and advisory boards, speaker bureaus, and lectures from Hoffmann-La Roche Ltd and Biogen; and has received support for this study, as noted above.
Nathalie Budd, Shikha Virdi, and Melanie Wilson are employed by Hoffmann-La Roche Ltd (Mississauga, ON, Canada).









