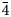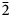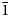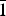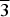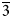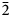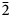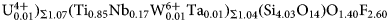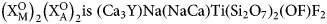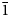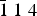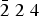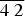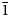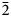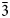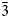Volume 83 - December 2019
Article
The story of one diamond: the heterogeneous distribution of the optical centres within a diamond crystal from the Ichetju placer, northern Urals
- Evgeny Vasilev, Vitaly Petrovsky, Alexander Kozlov, Anton Antonov, Andrey Kudryavtsev, Ksenia Orekhova
-
- Published online by Cambridge University Press:
- 08 May 2019, pp. 515-522
-
- Article
- Export citation
-
We have investigated a diamond crystal that consists of several misorientated subgrains. The main feature of the crystal is the dark areas in the cathodoluminescent core that has ‘estuary-like’ boundaries extending along the subgrain interfaces. The core has >3100 ppm of nitrogen, and the share of the B form is >95%; the absorbance of the centre N3VH at 3107 cm–1 reaches 75 cm–1. The N3 centre absorbance, as well as N3 luminescence, is absent in the core. In the outer part of the crystal, bright blue luminescence of the N3 centre is apparent, and the N3 absorbance reaches 5.3 cm–1. These observations could be explained by the conversion of N3 centres to N3VH after attaching a hydrogen atom. After the full conversion of the N3 centres, the diamond becomes darker under CL. We hypothesise the dark core has a specific shape due to the post-growth diffusion of the hydrogen.
Application of scanning electron diffraction in the transmission electron microscope for the characterisation of dislocations in minerals
- Billy C. Nzogang, Alexandre Mussi, Patrick Cordier
-
- Published online by Cambridge University Press:
- 30 July 2018, pp. 71-79
-
- Article
- Export citation
-
We present an application of scanning electron diffraction for the characterisation of crystal defects in olivine, quartz and phase A (a high pressure hydrated phase). In this mode, which takes advantage of the ASTAR™ module from NanoMEGAS, a slightly convergent probe is scanned over the sample with a short acquisition time (a few tens of ms) and the spot patterns are acquired and stored for further post-processing. Originally, orientation maps were constructed from automatic indexing at each probe location. Here we present another application where images are reconstructed from the intensity of diffraction spots, producing either so-called ‘virtual’ bright- or dark-field images. We show that these images present all the characteristics of contrast (perfect crystal or defects) of conventional transmission electron microscopy images. Data are acquired with a very short time per probe location (a few tens of milliseconds), this technique appears very attractive for the characterisation of beam-sensitive materials. However, as the acquisition is done at a given orientation, fine tuning of the diffraction conditions at a given location for each reflection is not possible. This might present a difficulty for some precise, quantitative contrast analysis.
An in situ study of the phase transitions among CaCO3 high-pressure polymorphs
- Xueyin Yuan, Chao Gao, Jing Gao
-
- Published online by Cambridge University Press:
- 02 July 2018, pp. 191-197
-
- Article
- Export citation
-
The phase transitions involving calcite (CaCO3-I), CaCO3-II, CaCO3-III and CaCO3-IIIb were investigated using a diamond anvil cell and micro-Raman spectroscopy. Based on the results obtained from in situ observations and Raman measurements made with six natural calcite crystals, the phase transition from calcite to CaCO3-II took place between 1.56 and 1.67 GPa under ambient temperature. Under a precise pressure of 1.97 ± 0.03 GPa, three CaCO3 samples were observed to transform from CaCO3-II directly to CaCO3-III, while in the other three samples both CaCO3-III and CaCO3-IIIb crystal structures were detected. Transformation from CaCO3-IIIb to CaCO3-III was completed in a short period in one sample, whereas in the other two samples coexistence of CaCO3-III and CaCO3-IIIb was observed over a wide pressure range from 1.97 to 3.38 GPa, with sluggish transformation from CaCO3-IIIb to CaCO3-III being observed after the samples were preserved under 3.38 GPa for 72 h. Hence, it can be concluded that CaCO3-IIIb is a metastable intermediate phase occurring during the reconstructive transformation from CaCO3-II to CaCO3-III. Splitting of the C–O in-plane bending (ν4) and symmetric stretching (ν1) vibrations and appearance of new lattice vibrations in the Raman spectra of CaCO3-III and CaCO3-IIIb suggest a lowering in crystal symmetry during the transformation from CaCO3-II through CaCO3-IIIb to CaCO3-III, which is in good agreement with the observed sequence of phase symmetries.
Lussierite, a new sodium uranyl sulfate mineral with bidentate UO7–SO4 linkage from the Blue Lizard mine, San Juan County, Utah, USA
- Anthony R. Kampf, Travis A. Olds, Jakub Plášil, Barbara P. Nash, Joe Marty
-
- Published online by Cambridge University Press:
- 03 June 2019, pp. 799-808
-
- Article
- Export citation
-
The new mineral lussierite (IMA2018-101), Na10[(UO2)(SO4)4](SO4)2(H2O)3, was found in the Blue Lizard mine, San Juan County, Utah, USA, where it occurs as pale green–yellow prisms or blades in a secondary assemblage with belakovskiite, ferrinatrite, halite, ivsite, metavoltine and thénardite. The streak is white and the fluorescence is bright cyan under 365 nm ultraviolet light. Crystals are transparent with vitreous lustre. The tenacity is brittle, the Mohs hardness is 2½, the fracture is irregular and no cleavage was observed. The mineral is easily soluble in H2O and has a measured density of 2.87(2) g cm–3. Lussierite is optically biaxial (+), with α = 1.493(1), β = 1.505(1) and γ = 1.518(1) (white light); 2Vmeas. = 88(1)°; dispersion is r > v, moderate; pleochroism: X = colourless, Y and Z = green yellow (X < Y ≈ Z); optical orientation: X = b, Z ∧ a = 44° in obtuse β. Electron microprobe analyses (wavelength-dispersive spectroscopy mode) provided Na10(U0.99O2)(S1.00O4)6·3H2O (+0.06 H for charge balance). The five strongest X-ray powder diffraction lines are [dobs Å(I)(hkl)]: 6.69(95)(
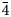 $\bar{1}$11,130), 4.814(100)(150,002,060), 3.461(83)(171,
$\bar{1}$11,130), 4.814(100)(150,002,060), 3.461(83)(171,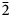 $\bar{2}$02), 2.955(81)(113,330) and 2.882(74)(
$\bar{2}$02), 2.955(81)(113,330) and 2.882(74)(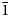 $\bar{1}$91,311,191,0·10·0). Lussierite is monoclinic, Cc, a = 9.3134(4), b = 28.7501(11), c = 9.6346(7) Å, β = 93.442(7)°, V = 2575.1(2) Å3 and Z = 4. The structure (R1 = 0.0298 for 5202 I > 2σI) contains a [(UO2)(SO4)4]6– uranyl sulfate cluster in which one SO4 tetrahedron shares an edge (bidentate linkage) with the UO7 pentagonal bipyramid. The uranyl sulfate clusters occur in layers and are linked through a complex network of bonds involving Na+ cations, isolated SO4 tetrahedra and isolated H2O groups.
$\bar{1}$91,311,191,0·10·0). Lussierite is monoclinic, Cc, a = 9.3134(4), b = 28.7501(11), c = 9.6346(7) Å, β = 93.442(7)°, V = 2575.1(2) Å3 and Z = 4. The structure (R1 = 0.0298 for 5202 I > 2σI) contains a [(UO2)(SO4)4]6– uranyl sulfate cluster in which one SO4 tetrahedron shares an edge (bidentate linkage) with the UO7 pentagonal bipyramid. The uranyl sulfate clusters occur in layers and are linked through a complex network of bonds involving Na+ cations, isolated SO4 tetrahedra and isolated H2O groups.
Lipuite, a new manganese phyllosilicate mineral from the N'Chwaning III mine, Kalahari Manganese Fields, South Africa
- Xiangping Gu, Hexiong Yang, Xiande Xie, Jaco J. van Nieuwenhuizen, Robert T. Downs, Stanley H. Evans
-
- Published online by Cambridge University Press:
- 26 February 2019, pp. 645-654
-
- Article
- Export citation
-
A new phyllosilicate mineral, lipuite (IMA2014-085), has been discovered from the N'Chwaning III mine, Kalahari Manganese Fields, Northern Cape Province, Republic of South Africa. It occurs as platy, tabular, or granular crystals and veined agglomerate in association with Mn-bearing sugilite, taniajacoite, pectolite, richterite, norrishite and namansilite. Lipuite is dark red–brown with vitreous lustre, red streak, an estimated Mohs hardness of 5 and the measured density is 2.83(3) g/cm3. It is biaxial (+) and characterised by bright red to dark red colour in thin section with measured refractive indices in white light: α = 1.635(1), β = 1.653(1), γ = 1.670(1) and 2V = 86(2)°. The Raman spectra of lipuite are composed of over 21 bands at 109, 146, 162, 183, 206, 244, 288, 342, 362, 455, 496, 520, 552, 613, 669, 886, 930, 971, 1097, 3487 and 3540 cm–1. The empirical formula from microprobe analyses is (based on total number of cations = 27.5 and structural refinement): K1.12Na8.16(Mn4.77Fe0.07)Σ4.84Mg0.44[Si11.97O30(OH)4](PO4)0.94O2(OH)2·4H2O. The idealised formula is: KNa8Mn3+5Mg0.5[Si12O30(OH)4](PO4)O2(OH)2·4H2O.
Lipuite is orthorhombic, space group Pnnm, a = 9.080(3), b = 12.222(3), c = 17.093(5) Å, V = 1897.0(9) Å3 and Z = 2. The strongest powder X-ray diffraction peaks [d, Å (I) (hkl)] are: 9.965(40)(011), 2.938(33)(310), 2.895(100)(311), 2.777(38)(224), 2.713(53)(320), 2.483(32)(126), 2.086(35)(046) and 1.534(40)(446). The crystal structure of lipuite is characterised by sheets of SiO4 tetrahedra that are linked together along [010] by K+, Na+, Mn3+, Mg2+ and P5+ cations, as well as hydrogen bonds. These tetrahedral sheets consist of 14-membered rings of SiO4 tetrahedra that zigzag along [100]. The two independent Mn3+ cations are both octahedrally coordinated. They form five-membered, edge-shared octahedral clusters between the SiO4 tetrahedral sheets. Lipuite represents a rather unique structure type and its silicate tetrahedral sheets can be considered a derivative of the silicate sheets in mica.
From structure topology to chemical composition. XXVI. Crystal structure and chemical composition of a possible new mineral of the murmanite group (seidozerite supergroup), ideally Na2CaTi4(Si2O7)2O4(H2O)4, from the Lovozero alkaline massif, Kola Peninsula, Russia
- Elena Sokolova, Frank C. Hawthorne
-
- Published online by Cambridge University Press:
- 29 May 2018, pp. 199-207
-
- Article
- Export citation
-
The crystal structure of a murmanite-related mineral (MRM) of the murmanite group (seidozerite supergroup), ideally Na2CaTi4(Si2O7)2O4(H2O)4, from Mt. Pyalkimpor, the Lovozero alkaline massif, Kola Peninsula, Russia, was refined in space group P
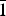 $ {\bar 1} $ with a = 5.363(2), b = 7.071(2), c = 12.176(5) Å, α = 92.724(3), β = 107.542(7), γ = 90.13(2)°, V = 439.7(4) Å3 and R1 = 5.72%. On the basis of electron-microprobe analysis, the empirical formula calculated on 22 (O + F), with two constraints derived from structure refinement, OH = 0.11 per formula unit (pfu) and H2O = 3.89 pfu, is (Na2.12K0.07Sr0.01)Σ2.20Ca0.85(Ti3.01Nb0.39Mn0.20Fe2+0.19Mg0.17Zr0.01Al0.01)Σ3.98(Si4.20O14)[O3.90F0.10]Σ4[(H2O)3.89(OH)0.11]Σ4{P0.03}, with Z = 1. It seems unlikely that {P0.03} belongs to MRM itself. The crystal structure of MRM is an array of TS blocks (Titanium-Silicate) connected via hydrogen bonds. The TS block consists of HOH sheets (H = heteropolyhedral, O = octahedral) parallel to (001). In the O sheet, the Ti-dominant MO1 site and Ca-dominant MO2 site give ideally (Ca□)Ti2 pfu. In the H sheet, the Ti-dominant MH site and Na-dominant AP site give ideally Na2Ti2 pfu. The MH and AP polyhedra and Si2O7 groups constitute the H sheet. The ideal structural formula of MRM of the form AP2MH2MO4(Si2O7)2(XOM,A)4(XOA)2(XPM,A)4 is Na2Ti2(Ca□)Ti2(Si2O7)2O4(H2O)4. MRM is a Ca-rich and Na-poor analogue of murmanite, ideally Na2Ti2Na2Ti2(Si2O7)2O4(H2O)4 and a Na-rich and (OH)-poor analogue of calciomurmanite, ideally (Ca□)Ti2(Na□)Ti2(Si2O7)2O2[O(OH)](H2O)4. MRM and (murmanite and calciomurmanite) are related by the following substitutions: O(Ca2+□)MRM ↔ O(Na+2)mur and O(Ca2+□)MRM + H(Na+2)MRM + O(O2–)MRM ↔ O(Na+□)cal + H(Ca2+□)cal + O[(OH)–]cal. MRM is a possible new mineral of the murmanite group (seidozerite supergroup) where Ti + Mn + Mg = 4 apfu.
$ {\bar 1} $ with a = 5.363(2), b = 7.071(2), c = 12.176(5) Å, α = 92.724(3), β = 107.542(7), γ = 90.13(2)°, V = 439.7(4) Å3 and R1 = 5.72%. On the basis of electron-microprobe analysis, the empirical formula calculated on 22 (O + F), with two constraints derived from structure refinement, OH = 0.11 per formula unit (pfu) and H2O = 3.89 pfu, is (Na2.12K0.07Sr0.01)Σ2.20Ca0.85(Ti3.01Nb0.39Mn0.20Fe2+0.19Mg0.17Zr0.01Al0.01)Σ3.98(Si4.20O14)[O3.90F0.10]Σ4[(H2O)3.89(OH)0.11]Σ4{P0.03}, with Z = 1. It seems unlikely that {P0.03} belongs to MRM itself. The crystal structure of MRM is an array of TS blocks (Titanium-Silicate) connected via hydrogen bonds. The TS block consists of HOH sheets (H = heteropolyhedral, O = octahedral) parallel to (001). In the O sheet, the Ti-dominant MO1 site and Ca-dominant MO2 site give ideally (Ca□)Ti2 pfu. In the H sheet, the Ti-dominant MH site and Na-dominant AP site give ideally Na2Ti2 pfu. The MH and AP polyhedra and Si2O7 groups constitute the H sheet. The ideal structural formula of MRM of the form AP2MH2MO4(Si2O7)2(XOM,A)4(XOA)2(XPM,A)4 is Na2Ti2(Ca□)Ti2(Si2O7)2O4(H2O)4. MRM is a Ca-rich and Na-poor analogue of murmanite, ideally Na2Ti2Na2Ti2(Si2O7)2O4(H2O)4 and a Na-rich and (OH)-poor analogue of calciomurmanite, ideally (Ca□)Ti2(Na□)Ti2(Si2O7)2O2[O(OH)](H2O)4. MRM and (murmanite and calciomurmanite) are related by the following substitutions: O(Ca2+□)MRM ↔ O(Na+2)mur and O(Ca2+□)MRM + H(Na+2)MRM + O(O2–)MRM ↔ O(Na+□)cal + H(Ca2+□)cal + O[(OH)–]cal. MRM is a possible new mineral of the murmanite group (seidozerite supergroup) where Ti + Mn + Mg = 4 apfu.
New minerals with modular structure derived from hatrurite from the pyrometamorphic rocks. Part IV: Dargaite, BaCa12(SiO4)4(SO4)2O3, from Nahal Darga, Palestinian Autonomy
- Irina O. Galuskina, Frank Gfeller, Evgeny V. Galuskin, Thomas Armbruster, Yevgeny Vapnik, Mateusz Dulski, Mariusz Gardocki, Lidia Jeżak, Mikhail Murashko
-
- Published online by Cambridge University Press:
- 21 May 2018, pp. 81-88
-
- Article
- Export citation
-
Dargaite, ideally BaCa12(SiO4)4(SO4)2O3, is an additional member of the arctite group belonging to minerals with a modular intercalated antiperovskite structure derived from hatrurite. The holotype specimen was found at a small outcrop of larnite pseudoconglomerates in the Judean Mts, West Bank, Palestinian Autonomy. Larnite, fluorellestadite–fluorapatite, brownmillerite, fluormayenite–fluorkyuygenite and ye'elimite are the main minerals of the holotype specimen; ternesite, shulamitite and periclase are noted rarely. Dargaite, nabimusaite and gazeevite occur in linear zones with higher porosity within larnite rocks. Pores are filled with ettringite and Ca-hydrosilicates, less commonly with gibbsite, brucite, baryte, katoite and calciolangbeinite. Dargaite is colourless, transparent with a white streak and has a vitreous lustre. It exhibits pronounced parting and imperfect cleavage along (001). Mohs’ hardness is ~4.5–5.5. The empirical formula is (Ba0.72K0.24Na0.04)Σ1(Ca11.95Mg0.04Na0.01)Σ12([SiO4]0.91 [PO4]0.05[AlO4]0.03[Ti4+O4]0.01)Σ4([SO4]0.84[PO4]0.14[CO3]0.02)Σ2(O2.54F0.46)Σ3. Dargaite is trigonal R
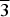 $\overline 3 $m, the unit-cell parameters are: a = 7.1874(4) Å, c = 41.292(3) Å, V = 1847.32(19) Å3 and Z = 3. The crystal structure of dargaite was refined from X-ray single-crystal data to R1 = 3.79%. The calculated density is 3.235 g cm–3. The following main Raman bands are distinguished on the holotype dargaite (cm–1): 122, 263, 323, 464, 523, 563, 641 and 644, 829 and 869, 947, 991 and 1116. The formation conditions of dargaite are linked to the local occurrence of pyrometamorphic by-products (gases, fluids and melts) transforming earlier mineral associations at ~900°C.
$\overline 3 $m, the unit-cell parameters are: a = 7.1874(4) Å, c = 41.292(3) Å, V = 1847.32(19) Å3 and Z = 3. The crystal structure of dargaite was refined from X-ray single-crystal data to R1 = 3.79%. The calculated density is 3.235 g cm–3. The following main Raman bands are distinguished on the holotype dargaite (cm–1): 122, 263, 323, 464, 523, 563, 641 and 644, 829 and 869, 947, 991 and 1116. The formation conditions of dargaite are linked to the local occurrence of pyrometamorphic by-products (gases, fluids and melts) transforming earlier mineral associations at ~900°C.
Mitrofanovite, Pt3Te4, a new mineral from the East Chuarvy deposit, Fedorovo–Pana intrusion, Kola Peninsula, Russia
- Victor V. Subbotin, Anna Vymazalová, František Laufek, Yevgeny E. Savchenko, Chris J. Stanley, Dmitry A. Gabov, Jakub Plášil
-
- Published online by Cambridge University Press:
- 03 October 2018, pp. 523-530
-
- Article
- Export citation
-
Mitrofanovite, Pt3Te4, is a new telluride discovered in low-sulfide disseminated ore in the East Chuarvy deposit, Fedorovo–Pana intrusion, Kola Peninsula, Russia. It forms anhedral grains (up to ~20 μm × 50 μm) commonly in intergrowths with moncheite in aggregates with lukkulaisvaaraite, kotulskite, vysotskite, braggite, keithconnite, rustenburgite and Pt–Fe alloys hosted by a chalcopyrite–pentlandite–pyrrhotite matrix. Associated silicates are: orthopyroxene, augite, olivine, amphiboles and plagioclase. Mitrofanovite is brittle; it has a metallic lustre and a grey streak. Mitrofanovite has a good cleavage, along {001}. In plane-polarised light, mitrofanovite is bright white with medium to strong bireflectance, slight pleochroism, and strong anisotropy on non-basal sections with greyish brown rotation tints; it exhibits no internal reflections. Reflectance values for the synthetic analogue of mitrofanovite in air (Ro, Re’ in %) are: 58.4, 54.6 at 470 nm; 62.7, 58.0 at 546 nm; 63.4, 59.1 at 589 nm; and 63.6, 59.5 at 650 nm. Fifteen electron-microprobe analyses of mitrofanovite gave an average composition: Pt 52.08, Pd 0.19, Te 47.08 and Bi 0.91, total 100.27 wt.%, corresponding to the formula (Pt2.91Pd0.02)Σ2.93(Te4.02Bi0.05)Σ4.07 based on 7 atoms; the average of eleven analyses on synthetic analogue is: Pt 52.57 and Te 47.45, total 100.02 wt.%, corresponding to Pt2.94Te4.06. The density, calculated on the basis of the formula, is 11.18 g/cm3. The mineral is trigonal, space group R
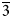 $\overline 3 $m, with a = 3.9874(1), c = 35.361(1) Å, V = 486.91(2) Å3 and Z = 3. The crystal structure was solved and refined from the powder X-ray-diffraction data of synthetic Pt3Te4. Mitrofanovite is structurally and chemically related to moncheite (PtTe2). The strongest lines in the powder X-ray diffraction pattern of synthetic mitrofanovite [d in Å (I) (hkl)] are: 11.790(23)(003), 5.891(100)(006), 2.851(26)(107), 2.137(16)(1013), 2.039(18)(0114), 1.574(24)(0120), 1.3098(21)(0027). The structural identity of natural mitrofanovite with synthetic Pt3Te4 was confirmed by electron backscatter diffraction measurements on the natural sample. The mineral name is chosen to honour Felix P. Mitrofanov, a Russian geologist who was among the first to discover platinum-group element mineralisation in the Fedorova–Pana complex.
$\overline 3 $m, with a = 3.9874(1), c = 35.361(1) Å, V = 486.91(2) Å3 and Z = 3. The crystal structure was solved and refined from the powder X-ray-diffraction data of synthetic Pt3Te4. Mitrofanovite is structurally and chemically related to moncheite (PtTe2). The strongest lines in the powder X-ray diffraction pattern of synthetic mitrofanovite [d in Å (I) (hkl)] are: 11.790(23)(003), 5.891(100)(006), 2.851(26)(107), 2.137(16)(1013), 2.039(18)(0114), 1.574(24)(0120), 1.3098(21)(0027). The structural identity of natural mitrofanovite with synthetic Pt3Te4 was confirmed by electron backscatter diffraction measurements on the natural sample. The mineral name is chosen to honour Felix P. Mitrofanov, a Russian geologist who was among the first to discover platinum-group element mineralisation in the Fedorova–Pana complex.
The effect of cooling rate on immiscible silicate liquid microstructure: an example from the Palaeogene dykes of Northeast England
- Victoria C. Honour, Marian B. Holness, Michael J. Stock
-
- Published online by Cambridge University Press:
- 06 November 2019, pp. 809-820
-
- Article
- Export citation
-
The migration and accumulation of immiscible silicate liquids may play a significant role in the differentiation of crustal magma bodies and the formation of some economic mineral deposits. However, our understanding of the processes that control the segregation of these liquids is currently limited by the short timescales of petrological experiments. Detailed microstructural investigations of Palaeogene basaltic dykes from Northeast England, coupled with simple 1D thermal models, constrain the effects of cooling rate on the microstructure of unmixed immiscible silicate liquids under natural conditions. The size of unmixed Fe-rich droplets within a continuous silicic phase is related to the cooling rate by a power law, with droplet diameter increasing with decreasing cooling rate, accompanied by an increase in the number of droplets. Fe-rich droplet coarsening is a result of diffusion-controlled growth. The average apparent aspect ratio and grain size of matrix plagioclase crystals indicate that nucleation and growth of these grains probably occurred in a static (or only weakly convecting) fluid dynamical regime.
Rinkite-(Y), Na2Ca4YTi(Si2O7)2OF3, a seidozerite-supergroup TS-block mineral from the Darai-Pioz alkaline massif, Tien-Shan mountains, Tajikistan: Description and crystal structure
- Leonid A. Pautov, Atali A. Agakhanov, Vladimir Yu. Karpenko, Yulia A. Uvarova, Elena Sokolova, Frank C. Hawthorne
-
- Published online by Cambridge University Press:
- 29 June 2018, pp. 373-380
-
- Article
- Export citation
-
Rinkite-(Y), ideally Na2Ca4YTi(Si2O7)2OF3, is a new rinkite-group (seidozerite-supergroup) TS-block mineral from the Darai-Pioz alkaline massif, Tian-Shan mountains, Tajikistan. The mineral is of hydrothermal origin. It occurs as aggregates (up to 1.5 cm long) of acicular crystals 0.1–1.0 mm thick, and as separate elongated columnar, flattened-prismatic crystals up to 1 cm long with rectangular or rhombic sections up to 0.5 mm across. Associated minerals are quartz, aegirine, microcline, neptunite, pectolite, calcite, eudialyte-group minerals, fluorite, titanite, turkestanite, kupletskite, galena, albite and pyrochlore-group minerals. Crystals are transparent and colourless to occasionally white, with a vitreous lustre. Rinkite-(Y) has a white streak, uneven, conchoidal fracture and does not fluoresce under a cathode or ultraviolet light. Cleavage is very good on {100}, no parting was observed, Mohs hardness is ~5, and it is brittle, Dmeas. = 3.44(2) g/cm3, Dcalc. = 3.475 g/cm3. It is biaxial (+) with refractive indices (λ = 590 nm) α = 1.662(2), β = 1.666(2), γ = 1.685(5); 2Vmeas. = 50(3) and 2Vcalc. = 49.7°. It is nonpleochroic. Rinkite-(Y) is monoclinic, space group P21/c, a = 7.3934(5), b = 5.6347(4), c = 18.713(1) Å, β = 101.415(2)° and V = 764.2(2) Å3. The six strongest reflections in the X-ray powder diffraction data [d(Å), I, (hkl)] are: 3.057, 100, (006,
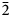 $\bar{2}$12, 210); 2.688, 28, (016); 9.18, 24, (002); 2.929, 17, (
$\bar{2}$12, 210); 2.688, 28, (016); 9.18, 24, (002); 2.929, 17, (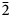 $\bar{2}$13, 211); 3.559, 15, (104, 014) and 2.783, 14, (021). The empirical formula calculated on 18 (O + F) is Na2.11(Ca3.74Sr0.03Mn0.03)Σ3.80(Y0.50Nd0.16Ce0.16Gd0.07Dy0.06Sm0.05Pr0.03La0.03
$\bar{2}$13, 211); 3.559, 15, (104, 014) and 2.783, 14, (021). The empirical formula calculated on 18 (O + F) is Na2.11(Ca3.74Sr0.03Mn0.03)Σ3.80(Y0.50Nd0.16Ce0.16Gd0.07Dy0.06Sm0.05Pr0.03La0.03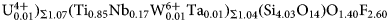 ${\rm U}_{0.01}^{{\rm 4 + }} {\rm )}_{\Sigma 1.07}{\rm (T}{\rm i}_{0.85}{\rm N}{\rm b}_{0.17}{\rm W}^{6+}_{0.01}{\rm T}{\rm a}_{0.01}{\rm )}_{\Sigma 1.04}\left( {{\rm S}{\rm i}_{4.03}{\rm O}_{14}} \right){\rm O}_{1.40}{\rm F}_{2.60}$ with Z = 2. The ideal formula is Na2Ca4YTi(Si2O7)2OF3. The crystal structure was refined on a twinned crystal to R1 = 4.59% on the basis of 1489 unique reflections (F > 4σF) and is a framework of TS (Titanium-Silicate) blocks. The TS block consists of HOH sheets (H – heteropolyhedral, O – octahedral) parallel to (100). In the O sheet, the Ti-dominant [6]MO1 site ideally gives 1 Ti apfu. The [8]MO2 and [6]MO3 sites are ideally occupied by Na and (NaCa) apfu. In the H sheet, the [7]MH site is occupied by Ca1.13Y0.50REE0.37, (REE = rare-earth element), ideally (CaY), <MH–φ> = 2.415 Å and the [7]AP site is occupied by Ca1.81REE0.19, ideally Ca2, <AP–φ> = 2.458 Å. The MH + AP sites ideally give (Ca3Y) apfu. The MH and AP polyhedra and Si2O7 groups constitute the H sheet. Linkage of H and O sheets via common vertices of MH and AP polyhedra and Si2O7 groups with MO1–3 polyhedra results in a TS block. The TS block in rinkite-(Y) exhibits linkage 1 and stereochemistry typical for the rinkite group (Ti = 1 apfu) of the seidozerite supergroup. For rinkite-(Y), the ideal structural formula of the form AP2MH2MO4(Si2O7)2
${\rm U}_{0.01}^{{\rm 4 + }} {\rm )}_{\Sigma 1.07}{\rm (T}{\rm i}_{0.85}{\rm N}{\rm b}_{0.17}{\rm W}^{6+}_{0.01}{\rm T}{\rm a}_{0.01}{\rm )}_{\Sigma 1.04}\left( {{\rm S}{\rm i}_{4.03}{\rm O}_{14}} \right){\rm O}_{1.40}{\rm F}_{2.60}$ with Z = 2. The ideal formula is Na2Ca4YTi(Si2O7)2OF3. The crystal structure was refined on a twinned crystal to R1 = 4.59% on the basis of 1489 unique reflections (F > 4σF) and is a framework of TS (Titanium-Silicate) blocks. The TS block consists of HOH sheets (H – heteropolyhedral, O – octahedral) parallel to (100). In the O sheet, the Ti-dominant [6]MO1 site ideally gives 1 Ti apfu. The [8]MO2 and [6]MO3 sites are ideally occupied by Na and (NaCa) apfu. In the H sheet, the [7]MH site is occupied by Ca1.13Y0.50REE0.37, (REE = rare-earth element), ideally (CaY), <MH–φ> = 2.415 Å and the [7]AP site is occupied by Ca1.81REE0.19, ideally Ca2, <AP–φ> = 2.458 Å. The MH + AP sites ideally give (Ca3Y) apfu. The MH and AP polyhedra and Si2O7 groups constitute the H sheet. Linkage of H and O sheets via common vertices of MH and AP polyhedra and Si2O7 groups with MO1–3 polyhedra results in a TS block. The TS block in rinkite-(Y) exhibits linkage 1 and stereochemistry typical for the rinkite group (Ti = 1 apfu) of the seidozerite supergroup. For rinkite-(Y), the ideal structural formula of the form AP2MH2MO4(Si2O7)2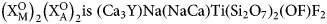 $ \left( {{\rm X}_{\rm M}^{\rm O} } \right)_2\left( {{\rm X}_{\rm A}^{\rm O} } \right)_2{\rm is }\;\left( {{\rm C}{\rm a}_3{\rm Y}} \right){\rm Na}\left( {{\rm NaCa}} \right){\rm Ti}\left( {{\rm S}{\rm i}_2{\rm O}_7} \right)_2\left( {{\rm OF}} \right){\rm F}_2 $ with Z = 2. The mineral is named rinkite-(Y) as it is structurally identical to rinkite-(Ce) and Y is the dominant rare-earth element.
$ \left( {{\rm X}_{\rm M}^{\rm O} } \right)_2\left( {{\rm X}_{\rm A}^{\rm O} } \right)_2{\rm is }\;\left( {{\rm C}{\rm a}_3{\rm Y}} \right){\rm Na}\left( {{\rm NaCa}} \right){\rm Ti}\left( {{\rm S}{\rm i}_2{\rm O}_7} \right)_2\left( {{\rm OF}} \right){\rm F}_2 $ with Z = 2. The mineral is named rinkite-(Y) as it is structurally identical to rinkite-(Ce) and Y is the dominant rare-earth element.
Platinum-group minerals from Seyba, Eastern Sayans, Russia, and substitutions in the PGE-rich pentlandite and ferhodsite series
- Andrei Y. Barkov, Gennadiy I. Shvedov, Andrey A. Nikiforov, Robert F. Martin
-
- Published online by Cambridge University Press:
- 12 April 2019, pp. 531-538
-
- Article
- Export citation
-
Chromitite zones associated with ultramafic units of the Lysanskiy layered complex of dunite–peridotite–gabbro composition could well represent the primary source for the placers bearing platinum-group minerals (PGM) of the entire drainage of the River Sisim and its tributaries, the rivers Ko and Seyba, eastern Sayans. Alluvial gold present in the placers of River Seyba, as elsewhere in the Sisim Placer Zone, reflects mineralisation during a recent period of tectonic activity. We focus on the PGM in the Seyba suite, and in particular on the attributes of pentlandite enriched in platinum-group-elements (PGE) and the compositionally similar and recently defined ferhodsite, which were trapped in host grains of Os–Ir–Ru alloy. Both minerals formed from small volumes of fractionated Fe–Ni–Cu melt considerably enriched in the PGE. In the Seyba suite, as in several others, the amounts of PGE in ferhodsite exceeds that in pentlandite, which results in a greater proportion of vacancies than in pentlandite.
Russoite, NH4ClAs23+O3(H2O)0.5, a new phylloarsenite mineral from Solfatara Di Pozzuoli, Napoli, Italy
- Italo Campostrini, Francesco Demartin, Marco Scavini
-
- Published online by Cambridge University Press:
- 15 May 2018, pp. 89-94
-
- Article
- Export citation
-
The new mineral russoite (IMA2015-105), NH4ClAs23+O3(H2O)0.5, was found at the Solfatara di Pozzuoli, Pozzuoli, Napoli, Italy, as a fumarolic phase associated with alacránite, dimorphite, realgar, mascagnite, salammoniac and an amorphous arsenic sulfide. It occurs as hexagonal plates up to ~300 µm in diameter and 15 µm thick, in rosette-like intergrowths. On the basis of powder X-ray diffraction measurements and chemical analysis, the mineral was recognised to be identical to the corresponding synthetic phase NH4ClAs2O3(H2O)0.5. Crystals are transparent and colourless, with vitreous lustre and white streak. Tenacity is brittle and fracture is irregular. Cleavage is perfect on {001}. The measured density is 2.89(1) g/cm3; the calculated density is 2.911 g/cm3. The empirical formula, (based on 4.5 anions per formula unit) is [(NH4)0.94,K0.06]Σ1.00(Cl0.91,Br0.01)Σ0.92As2.02O3(H2O)0.5. Russoite is hexagonal, space group P622, with a = 5.2411(7), c = 12.5948(25) Å, V = 299.62(8) Å3 and Z = 2. The eight strongest X-ray powder diffraction lines are [dobs Å(I)(hkl)]: 12.63(19)(001), 6.32(100)(002), 4.547(75)(100), 4.218(47)(003), 3.094(45)(103), 2.627(46)(110), 2.428(31)(112) and 1.820(28)(115). The structure, was refined to R = 0.0518 for 311 reflections with I > 2σ(I) and shows a different location of the ammonium cation and water molecules with respect to that reported for the synthetic analogue. The mineral belongs to a small group of phylloarsenite minerals (lucabindiite, torrecillasite and gajardoite). It contains electrically neutral As2O3 layers, topologically identical to those found in lucabindiite and gajardoite between which are ammonium cations and outside of which Cl– anions. Water molecules and additional ammonium cations are located in a layer between two levels of chloride anions.
Characterisation and possible hazard of an atypical asbestiform sepiolite associated with aliphatic hydrocarbons from Sassello, Ligurian Apennines, Italy
- Roberto Giustetto, Loredana Macaluso, Gloria Berlier, Yadolah Ganjkhanlou, Luca Barale
-
- Published online by Cambridge University Press:
- 08 October 2018, pp. 209-222
-
- Article
- Export citation
-
An unusual occurrence of asbestiform sepiolite, filling veins in the antigorite serpentinites of the Voltri Unit exposed in a borrow pit (now reclaimed) in the Deiva forest, near Sassello, NW Italy, was investigated with an in-depth analytical approach aimed at studying its crystal-chemistry and structure and evaluating its possible hazards for human health. Optical microscopy and scanning electron microscopy (energy-dispersive spectroscopy mode) proved that these sepiolite fibres, apparently up to several cm long, are made up of bundles of thinner fibrils (or laths: average length > 100 µm; thickness ≈ 80 nm), with a composition consistent to that reported in the literature. The dehydration process was monitored through thermo-gravimetric analyses and Fourier-transform infrared spectroscopy, performed at increasing T; the latter, in particular, showed the presence of moderate amounts of aliphatic hydrocarbons – not yet identified thoroughly – associated with the sample. The crystal structure refinement with the Rietveld method showed no relevant difference from the literature models, although a peculiar distribution of zeolitic H2O molecules was observed. The geological context suggests that the Sassello sepiolite precipitated from hydrothermal fluids, which were saturated in Mg and silica by the interaction of the host serpentinites. The same setting favoured formation of abiotic hydrocarbons, by means of the Fischer–Tropsch reaction. The extremely long and flexible fibrils (length/width aspect ratio >> 3) of this sepiolite specimen could represent a serious hazard for human health if air dispersed and inhaled; also, its atypical association with hydrocarbons (only reported once previously) might even favour further fragmentation in thinner units.
Subaerial sulfate mineral formation related to acid aerosols at the Zhenzhu Spring, Tengchong, China
- Lianchao Luo, Huaguo Wen, Rongcai Zheng, Ran Liu, Yi Li, Xiaotong Luo, Yaxian You
-
- Published online by Cambridge University Press:
- 14 January 2019, pp. 381-392
-
- Article
- Export citation
-
The Zhenzhu Spring, located in the Tengchong volcanic field, Yunnan, China, is an acid hot spring with high SO42− concentrations and intense acid aerosol generation. In order to understand the formation mechanism of sulfate minerals at the Zhenzhu Spring and provide a better insight into the sulfur isotope geochemistry of the associated Rehai hydrothermal system, we investigated the spring water hydrochemistry, mineralogy and major-element geochemistry of sulfate minerals at the Zhenzhu Spring together with the sulfur-oxygen isotope geochemistry of sulfur-containing materials at the Rehai geothermal field and compared the isotope results with those in other steam-heated environments. Subaerial minerals include a wide variety of sulfate minerals (gypsum, alunogen, pickeringite, tamarugite, magnesiovoltaite and a minor Mg–S–O phase) and amorphous SiO2. The δ34S values of the subaerial sulfate minerals at the Zhenzhu Spring varied subtly from –0.33 to 1.88‰ and were almost consistent with the δ34S values of local H2S (–2.6 to 0.6‰) and dissolved SO42− (–0.2 to 5.8‰), while the δ18O values (–8.94 to 20.1‰) were between that of the spring waters (–10.19 to –6.7‰) and atmospheric O2 (~23.88‰). The results suggest that most of the sulfate minerals are derived from the oxidation of H2S, similar to many sulfate minerals from modern steam-heated environments. However, the rapid environmental change (different ratio of atmospheric and water oxygen) at the Zhenzhu Spring accounts for the large variation of δ18O. The formation of subaerial sulfate minerals around the Zhenzhu Spring is related to acid aerosols (vapour and acid water droplets). The intense activity of spring water around vents supply the aerosol with H2SO4 (H2S oxidation and acid water droplets formed by bubble bursting) and few cations. Deposition of the acid sulfate aerosol forms the acid condensate, which attacks the underlying rocks and releases many cations and anions to form subaerial sulfate minerals at the Zhenzhu Spring.
Bavsiite, Ba2V2O2[Si4O12], mineral data and crystal structure
- Hans-Peter Bojar, Franz Walter, Judith Baumgartner
-
- Published online by Cambridge University Press:
- 12 September 2019, pp. 821-827
-
- Article
- Export citation
-
Bavsiite from the type locality Gun Claim, Yukon Territory, Canada, occurs as millimetre-sized sky-blue platy crystals in a barium-rich low-temperature skarn related to a porphyritic quartz monzonite stock. Associated minerals are alstonite, baryte, celsian, diopside, fresnoite, mica, suzukiite, walstromite, witherite and minerals of the cerchiaraite group. Bavsiite is optical uniaxial (+), with ω = 1.725(3) and ε = 1.750(3) (589 nm) and pleochroic. Electron microprobe analyses yielded the empirical formula Na0.02Ba1.98Ti0.16Fe2+0.03V4+1.80 Al0.05Si4.00O14 based on 14 oxygen atoms, the simplified chemical formula is Ba2V2Si4O14. Bavsiite is tetragonal, space group I4/m, a = 7.043(1), c = 11.444(2) Å and Z = 2 obtained from single crystal data at 100 K, which are in good agreement with cell parameters from powder diffraction data at 293 K: a = 7.051(1) Å and c = 11.470(1) Å. The eight strongest lines of the powder X-ray diffraction pattern are [d, Å (I,%)(hkl)]: 3.76(30)(112), 3.36(44)(013), 3.004(100)(022), 2.493(43)(220), 2.486(67)(114), 2.286(24)(222), 1.785(39)(116) and 1.763(25)(040). The crystal structure was refined to R = 0.0159 based upon 312 unique reflections with I > 2σ(I). The crystal structure of bavsiite comprises unbranched single [Si4O12]8– rings connected by [VO5]6– square pyramids and BaO12 polyhedra. It can also be considered as cage–like [Si4V2O18]12– clusters built by four SiO4 tetrahedra and two VO5 square pyramids. These clusters are cross–linked to form a pseudo-two-dimensional network (2D) parallel to (001) containing empty channels along the a axis and the 2D networks held together by Ba2+ ions located in channels parallel to the c axis. The structural formula is Ba2V2O2[Si4O12]. Bavsiite is polymorphic to suzukiite, BaVSi2O7, which is orthorhombic.
Camanchacaite, chinchorroite, espadaite, magnesiofluckite, picaite and ríosecoite: six new hydrogen-arsenate minerals from the Torrecillas mine, Iquique Province, Chile
- Anthony R. Kampf, Barbara P. Nash, Aaron J. Celestian, Maurizio Dini, Arturo A. Molina Donoso
-
- Published online by Cambridge University Press:
- 08 May 2019, pp. 655-671
-
- Article
- Export citation
-
The new minerals camanchacaite, NaCaMg2[AsO4]2[AsO3(OH)2], chinchorroite, Na2Mg5(As2O7)2(AsO3OH)2(H2O)10, espadaite, Na4Ca3Mg2[AsO3(OH)]2[AsO2(OH)2]10(H2O)6·H2O, magnesiofluckite, CaMg(AsO3OH)2(H2O)2, picaite, NaCa[AsO3OH][AsO2(OH)2] and ríosecoite, Ca2Mg(AsO3OH)3(H2O)2, were discovered on two closely related specimens collected from the Torrecillas mine, Iquique Province, Chile. These minerals occur as secondary phases on massive quartz–hematite also in association with anhydrite, gypsum, halite and talmessite. Camanchacaite is monoclinic, C2/c, a = 12.470(9), b = 12.554(9), c = 6.848(9) Å, β = 113.75(2)°, V = 981.3(16) Å3 and Z = 4. It has a protonated alluaudite-type structure. Chinchorroite is triclinic, P
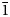 $\bar{1}$, a = 8.7777(2), b = 8.8570(3), c = 9.7981(7) Å, α = 91.097(6), β = 110.544(8), γ = 103.167(7)°, V = 690.43(7) Å3 and Z = 1. The structure contains abbreviated chains of five edge-sharing Mg octahedra that are linked by pyroarsenate and hydrogen-arsenate groups. Espadaite is orthorhombic, Ccca, a = 12.3649(10), b = 22.181(2), c = 18.3292(13) Å, V = 5027.1(7) Å3 and Z = 4. The structure is based on heteropolyhedral sheets of formula {Ca3Mg2[AsO3(OH)]2[AsO2(OH)2]10}4− that contain large voids; NaO6 polyhedra occupy the interlayer region. Magnesiofluckite is triclinic, P
$\bar{1}$, a = 8.7777(2), b = 8.8570(3), c = 9.7981(7) Å, α = 91.097(6), β = 110.544(8), γ = 103.167(7)°, V = 690.43(7) Å3 and Z = 1. The structure contains abbreviated chains of five edge-sharing Mg octahedra that are linked by pyroarsenate and hydrogen-arsenate groups. Espadaite is orthorhombic, Ccca, a = 12.3649(10), b = 22.181(2), c = 18.3292(13) Å, V = 5027.1(7) Å3 and Z = 4. The structure is based on heteropolyhedral sheets of formula {Ca3Mg2[AsO3(OH)]2[AsO2(OH)2]10}4− that contain large voids; NaO6 polyhedra occupy the interlayer region. Magnesiofluckite is triclinic, P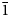 $\bar{1}$, a = 8.4143(6), b = 7.5321(5), c = 6.8917(4) Å, α = 82.477(6), β = 97.682(6), γ = 95.379(6)°, V = 427.84(5) Å3 and Z = 2. It is isostructural with fluckite. Picaite is monoclinic, P21/c, a = 7.2474(4), b = 14.6547(7), c = 7.2624(5) Å, β = 99.520(7)°, V = 760.70(8) Å3 and Z = 4. The structure contains chains of edge-sharing Na− and Ca octahedra with bridging AsO3(OH) and AsO2(OH)2 tetrahedra. Ríosecoite is triclinic, P
$\bar{1}$, a = 8.4143(6), b = 7.5321(5), c = 6.8917(4) Å, α = 82.477(6), β = 97.682(6), γ = 95.379(6)°, V = 427.84(5) Å3 and Z = 2. It is isostructural with fluckite. Picaite is monoclinic, P21/c, a = 7.2474(4), b = 14.6547(7), c = 7.2624(5) Å, β = 99.520(7)°, V = 760.70(8) Å3 and Z = 4. The structure contains chains of edge-sharing Na− and Ca octahedra with bridging AsO3(OH) and AsO2(OH)2 tetrahedra. Ríosecoite is triclinic, P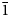 $\bar{1}$, a = 6.8110(9), b = 7.3156(12), c = 11.7773(17) Å, α = 83.466(6), β = 84.394(6), γ = 79.779(6)°, V = 571.95(15) Å3 and Z = 2. The structure contains tetramers of edge-sharing CaO7 and CaO8 polyhedra linked by MgO6 octahedra and bridging AsO3(OH) groups to form chains.
$\bar{1}$, a = 6.8110(9), b = 7.3156(12), c = 11.7773(17) Å, α = 83.466(6), β = 84.394(6), γ = 79.779(6)°, V = 571.95(15) Å3 and Z = 2. The structure contains tetramers of edge-sharing CaO7 and CaO8 polyhedra linked by MgO6 octahedra and bridging AsO3(OH) groups to form chains.
The new mineral novograblenovite, (NH4,K)MgCl3·6H2O from the Tolbachik volcano, Kamchatka, Russia: mineral description and crystal structure
- Viktor M. Okrugin, Sharapat S. Kudaeva, Oxana V. Karimova, Olga V. Yakubovich, Dmitry I. Belakovskiy, Nikita V. Chukanov, Andrey A. Zolotarev, Vladislav V. Gurzhiy, Nina G. Zinovieva, Andrey A. Shiryaev, Pavel M. Kartashov
-
- Published online by Cambridge University Press:
- 24 May 2018, pp. 223-231
-
- Article
- Export citation
-
The new mineral novograblenovite, (NH4,K)MgCl3·6H2O, was found on basaltic lava from the 2012–2013 Tolbachik fissure eruption at the Plosky Tolbachik volcano, Kamchatka Peninsula, Russia. It occurs as prismatic, needle-like transparent crystals together with gypsum and halite. Novograblenovite was formed due to the exposure of the host rocks to eruptive gas exhalations enriched in HCl and NH3. Basalt was the source of potassium and magnesium for the mineral formation. Novograblenovite crystallises in the monoclinic space group C2/c, with unit-cell parameters a = 9.2734(3) Å, b = 9.5176(3) Å, c = 13.2439(4) Å, β = 90.187(2)°, V = 1168.91(2) Å3 and Z = 4. The five strongest reflections in the powder X-ray diffraction pattern [dobs, Å (I, %) (h k l)] are: 3.330 (100) (2 2 0), 2.976 (45) (
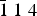 $\bar{1}\; 1\; 4$), 2.353 (29) (
$\bar{1}\; 1\; 4$), 2.353 (29) (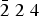 $\bar {2}\; 2\; 4$), 3.825 (26) (2 0 2), 1.997 (25) (
$\bar {2}\; 2\; 4$), 3.825 (26) (2 0 2), 1.997 (25) (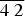 $\overline {4\; 2} $ 2). The density calculated from the empirical formula and the X-ray data is 1.504 g cm–3. The mineral is biaxial (+) with α = 1.469(2), β = 1.479(2) and γ = 1.496(2) (λ = 589 nm); 2Vmeas. = 80(10)° and 2Vcalc. = 75.7°. The crystal structure (solved and refined using single-crystal X-ray diffraction data, R1 = 0.0423) is based on the perovskite-like network of (NH4,K)Cl6-octahedra sharing chlorine vertices, and comprises [Mg(H2O)6]2+ groups in framework channels. The positions of all independent H atoms were obtained by difference-Fourier techniques and refined isotropically. All oxygen, nitrogen and chlorine atoms are involved in the system of hydrogen bonding, acting as donors or acceptors. The formula resulting from the structure refinement is [(NH4)0.7K0.3]MgCl3·6H2O. The mineral is named after Prokopiy Trifonovich Novograblenov, one of the researchers of Kamchatka Peninsula, a teacher, naturalist, geographer and geologist.
$\overline {4\; 2} $ 2). The density calculated from the empirical formula and the X-ray data is 1.504 g cm–3. The mineral is biaxial (+) with α = 1.469(2), β = 1.479(2) and γ = 1.496(2) (λ = 589 nm); 2Vmeas. = 80(10)° and 2Vcalc. = 75.7°. The crystal structure (solved and refined using single-crystal X-ray diffraction data, R1 = 0.0423) is based on the perovskite-like network of (NH4,K)Cl6-octahedra sharing chlorine vertices, and comprises [Mg(H2O)6]2+ groups in framework channels. The positions of all independent H atoms were obtained by difference-Fourier techniques and refined isotropically. All oxygen, nitrogen and chlorine atoms are involved in the system of hydrogen bonding, acting as donors or acceptors. The formula resulting from the structure refinement is [(NH4)0.7K0.3]MgCl3·6H2O. The mineral is named after Prokopiy Trifonovich Novograblenov, one of the researchers of Kamchatka Peninsula, a teacher, naturalist, geographer and geologist.
Crystal chemistry and nomenclature of rhodonite-group minerals
- Nadezhda V. Shchipalkina, Igor V. Pekov, Nikita V. Chukanov, Cristian Biagioni, Marco Pasero
-
- Published online by Cambridge University Press:
- 09 October 2019, pp. 829-835
-
- Article
- Export citation
-
This paper presents the nomenclature of the rhodonite group accepted by the Commission on New Minerals, Nomenclature and Classification of the International Mineralogical Association (IMA). An overview of the previous studies of triclinic (space group P
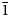 $\bar{1}$) pyroxenoids belonging to the rhodonite structure type, with a focus on their crystal chemistry, is given. These minerals have the general structural formula VIIM(5)VIM(1)VIM(2)VIM(3)VIM(4)[Si5O15]. The following dominant cations at the M sites are known at present: M(5) = Ca or Mn2+, M(1–3) = Mn2+; and M(4) = Mn2+ or Fe2+. In accordance with the nomenclature, the rhodonite group consists of three IMA-approved mineral species having the following the general chemical formulae: M(5)AM(1–3)B3M(4)C[Si5O15], where A = Ca or Mn2+; B = Mn2+; and C = Mn2+ or Fe2+. The end-member formulae of approved rhodonite-group minerals are as follows: rhodonite CaMn3Mn[Si5O15]; ferrorhodonite CaMn3Fe[Si5O15]; and vittinkiite MnMn3Mn[Si5O15].
$\bar{1}$) pyroxenoids belonging to the rhodonite structure type, with a focus on their crystal chemistry, is given. These minerals have the general structural formula VIIM(5)VIM(1)VIM(2)VIM(3)VIM(4)[Si5O15]. The following dominant cations at the M sites are known at present: M(5) = Ca or Mn2+, M(1–3) = Mn2+; and M(4) = Mn2+ or Fe2+. In accordance with the nomenclature, the rhodonite group consists of three IMA-approved mineral species having the following the general chemical formulae: M(5)AM(1–3)B3M(4)C[Si5O15], where A = Ca or Mn2+; B = Mn2+; and C = Mn2+ or Fe2+. The end-member formulae of approved rhodonite-group minerals are as follows: rhodonite CaMn3Mn[Si5O15]; ferrorhodonite CaMn3Fe[Si5O15]; and vittinkiite MnMn3Mn[Si5O15].
Pampaloite, AuSbTe, a new mineral from Pampalo gold mine, Finland
- Anna Vymazalová, Kari Kojonen, František Laufek, Bo Johanson, Chris J. Stanley, Jakub Plášil, Patricie Halodová
-
- Published online by Cambridge University Press:
- 04 July 2018, pp. 393-400
-
- Article
- Export citation
-
Pampaloite, AuSbTe, is a new mineral discovered in the Pampalo gold mine, 65 km east of Joensuu, Finland. It forms anhedral grains (up to ~20 μm) intergrown with gold, frohbergite and altaite. Pampaloite is brittle and has a metallic lustre. Values of VHN25 lie between 245 and 295 kg/mm2, with a mean value of 276 kg/mm2, corresponding to a Mohs hardness of ~4–5 (measured on synthetic material). In plane-polarised light, pampaloite is white with medium to strong bireflectance, weak reflectance pleochroism from slightly pinkish brown to slightly bluish white (only visible in grains of synthetic material containing multiple orientations), and strong anisotropy, with blue to light brown rotation tints; it exhibits no internal reflections. Reflectance values of pampaloite in air (R1,R2 in %) are: 60.0, 62.5 at 470 nm, 62.5, 64.8 at 546 nm, 63.2, 65.6 at 589 nm and 63.7, 66.0 at 650 nm. Ten electron-microprobe analyses of natural pampaloite give an average composition: Au 44.13, Sb 27.44 and Te 28.74, total 100.31 wt.%, corresponding to the empirical formula Au1.00Sb1.00Te1.00 based on 3 atoms; the average of eleven analyses on synthetic pampaloite is: Au 44.03, Sb 27.26, and Te 29.08, total 100.38 wt.%, corresponding to Au0.99Sb1.00Te1.01. The density, calculated on the basis of the empirical formula, is 9.33 g/cm3.The mineral is monoclinic, space group C2/c, with a = 11.947(3), b = 4.481(1) Å, c = 12.335(3) Å, β = 105.83(2)°, V = 635.3(3) Å3 and Z = 8. The crystal structure was solved and refined from the single-crystal X-ray-diffraction data of synthetic AuSbTe. The pampaloite crystal structure can be considered as a monoclinic derivative of the CdI2 structure composed of [AuTe3Sb3] octahedra. The strongest lines in the powder X-ray diffraction pattern of synthetic pampaloite [d in Å (I) (hkl)] are: 4.846(24)(
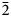 $\bar{2}$02), 3.825(18)(111), 2.978(100)(
$\bar{2}$02), 3.825(18)(111), 2.978(100)(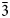 $\bar{3}$11), 2.968(50)(004), 2.242(25)(020), 2.144(55)(313), 2.063(33)(
$\bar{3}$11), 2.968(50)(004), 2.242(25)(020), 2.144(55)(313), 2.063(33)(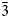 $\bar{3}$15) and 1.789(18)(024).
$\bar{3}$15) and 1.789(18)(024).
Apatite with lamellae of sulfide and other phases in ultrahigh-pressure eclogites from Nové Dvory, Moldanubian Zone, Czech Republic
- Shah Wali Faryad, Radim Jedlicka, Maria Perraki
-
- Published online by Cambridge University Press:
- 26 July 2018, pp. 95-105
-
- Article
- Export citation
-
Exsolution lamellae of baryte, Fe sulfides, Cu sulfides and Fe oxides were observed in apatite enclosed in garnet and omphacite and their intergranular spaces in ultrahigh-pressure eclogite in the Moldanubian Zone, Czech Republic. Micro-textural relations and compositional mapping of the apatite indicates a close relationship between the density of the exsolution lamellae and compositional domains that are rich in sulfur and iron. No relation between compositional domains and fluorine or chlorine content or any evidence of apatite metasomatisation was observed. On the basis of cathodoluminescence images, the compositional domains reflect sector zoning in apatite crystals by preferential uptake of elements due to differences in surface charge and morphology on the growth plane. It is concluded that the lamellae are products of exsolution in a closed system resulting from temperature decrease during metamorphism.