Volume 78 - December 2014
Research Article
Mineralogical characterization of calcification in cardiovascular aortic atherosclerotic plaque: A case study
- Yan Li, Xin Wang, Meiqian Zhu, Chong-Qing Yang, Anhuai Lu, Kang Li, Fanlu Meng, Changqiu Wang
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 775-786
-
- Article
- Export citation
-
Calcification in cardiovascular aortic atherosclerotic plaque contains Ca-phosphate minerals. However, most research on cardiovascular calcification has focused on its physiological properties rather than its mineralogical features. In this present study, cardiovascular calcification was characterized by collecting samples from patients’ tissues and applying mineralogical techniques. Synchrotron radiation-based micro-X-ray diffraction showed the calcification had a similar structure to hydroxylapatite (HAp). Transmission electron microscopy showed some structurally HAp-like spherical particles with a diameter of ∼200 nm and acicular crystals ∼100 nm × ∼20 nm in size. Selected-area electron diffraction indicated that these mineral particles belonged to the hexagonal crystal system. Fourier-transform infrared (FTIR) spectroscopy showed three typical peaks at 1469 cm−1, 1455 cm−1 and 1413 cm−1, indicating that the carbonate group in the calcification plaque substituted for a hydroxyl group to form B-type CHAp (Ca10(PO4,CO3)x(OH)y). The FTIR mapping results illustrated the intergrowth of calcification and organic tissues and the inhomogeneous substitution of phosphate by carbonate in the calcification area. X-ray absorption near-edge structure analysis affirmed that the chemical environments of Ca in the calcification were close to those in HAp. Based on these mineralogical characteristics, the calcification in plaque is identified as a mixture phase of HAp and B-type carbonate HAp, which is similar to the composition of bones.
The crystal structure of kiddcreekite solved using micro X-ray diffraction and the EPCryst program
- Liu Wenyuan, Dong Cheng, Gu Xiangping, Liu Yu, Qiu Xiaoping, Chen Yuchuan
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 1517-1525
-
- Article
- Export citation
-
Abundant kiddcreekite grains were identified in the Zijinshan Cu-Au epithermal deposit in Fujian Province, China. The mineral occurs as polycrystalline grains, 5–300 μm in size, associated with colusite, enargite, stannoidite, mawsonite, vinciennite, hemusite, tennantite and wolframite in a predominantly covellite ore. Based on electron microprobe analysis, the empirical formula of the kiddcreekite is Cu6.2Sn0.97W0.95S7.83, without significant Se or Te contents. The crystal structure of kiddcreekite was solved using the direct-space method (EPCryst) from laboratory micro X-ray diffraction (μXRD) data and refined by the Rietveld method. The R values of the final Rietveld refinement were Rp = 9.06%, Rwp = 8.31%, RB = 3.16 and RF = 2.17%. Kiddcreekite has a cubic structure, space group F
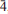 3m and lattice parameter a = 10.8178(3) Å (Z = 4, V = 1265.95(6) Å3). In the unit cell, W, Sn and Cu atoms occupy the 4a, 4c and 24f Wyckoff positions, respectively, and S atoms occupy two sets of 16e Wyckoff positions. The structure of kiddcreekite consists of stacked double MeS4 layers (giving a W–Sn–Cu tier and a Cu–vacancy tier) as in the sphalerite substructure. This study also demonstrates the possibility of using laboratory µXRD data coupled with the direct-space method to solve inorganic structures in cases where samples are too small for conventional powder and single-crystal diffraction.
3m and lattice parameter a = 10.8178(3) Å (Z = 4, V = 1265.95(6) Å3). In the unit cell, W, Sn and Cu atoms occupy the 4a, 4c and 24f Wyckoff positions, respectively, and S atoms occupy two sets of 16e Wyckoff positions. The structure of kiddcreekite consists of stacked double MeS4 layers (giving a W–Sn–Cu tier and a Cu–vacancy tier) as in the sphalerite substructure. This study also demonstrates the possibility of using laboratory µXRD data coupled with the direct-space method to solve inorganic structures in cases where samples are too small for conventional powder and single-crystal diffraction.
Coupled fluctuations in element release during dolomite dissolution
- Christine V. Putnis, Encarnación Ruiz-Agudo, Jörn Hövelmann
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 1355-1362
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Atomic force microscopy has been used to determine more precisely the mechanism of the initial stages of dolomite dissolution. Analysis of outflow solutions initially shows fluctuations of both Ca and Mg release with concentrations of Ca >> Mg. The dolomite surface dissolves congruently in the presence of slightly acidified water as confirmed by the regular spreading of characteristic rhombohedral etch pits. Direct in situ observations show that a new phase precipitates on the dissolving surface simultaneously. As the Ca and Mg release decreases with time, the precipitated phase can be seen to spread across the dolomite surface. These observations indicate that the apparent incongruent dissolution of dolomite is a two-step process involving stoichiometric dissolution with the release of Ca, Mg and CO3 ions to solution at the mineral–fluid interface coupled with precipitation of a new Mg-carbonate phase. The coupled element release confirms the interface-coupled dissolutionprecipitation mechanism.
Other
Celebrating the 2014 UNESCO International Year of Crystallography: Preface to the April 2014 special issue of Mineralogical Magazine
- S. J. Mills
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. i-iii
-
- Article
- Export citation
Research Article
Kihlmanite-(Ce), Ce2TiO2[SiO4](HCO3)2(H2O), a new rare-earth mineral from the pegmatites of the Khibiny alkaline massif, Kola Peninsula, Russia
- V. N. Yakovenchuk, S.V. Krivovichev, G. Y. Ivanyuk, Ya. A. Pakhomovsky, E.A. Selivanova, E. A. Zhitova, G. O. Kalashnikova, A. A. Zolotarev, J. A. Mikhailova, G. I. Kadyrova
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 483-496
-
- Article
- Export citation
-
Kihlmanite-(Ce), Ce2TiO2[SiO4](HCO3)2(H2O), is a new rare-earth titanosilicate carbonate, closely related to tundrite-(Ce). It is triclinic, P
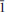 , a = 4.994(2), b = 7.54(2), c = 15.48(4) Å, α = 103.5(4), β = 90.7(2), γ = 109.2(2)o , V = 533(1) Å3, Z = 2 (from powder diffraction data) or a = 5.009(5), b = 7.533(5), c = 15.407(5) Å, α = 103.061(5), β = 91.006(5), γ = 109.285(5)°, V = 531.8(7) Å3, Z = 2 (from single-crystal X-ray diffraction data). The mineral was found in the arfvedsonite-aegirine-microcline vein in fenitized metavolcanic rock at the foot of the Mt Kihlman (Chil’man), near the western contact of the Devonian Khibiny alkaline massif and the Proterozoic Imandra-Varzuga greenstone belt. It forms brown spherulites (up to 2 cm diameter) and sheaf-like aggregates of prismatic crystals, flattened on {010} and up to 0.5 mm diameter. Both spherulites and aggregates occur in interstices in arfvedsonite and microcline, in intimate association with golden-green tundrite-(Ce). Kihlmanite-(Ce) is brown, with a vitreous lustre and a pale yellowish-brown streak. The cleavage is perfect on {010}, parting is perpendicular to c and the fracture is stepped. Mohs hardness is ∼3. In transmitted light, the mineral is yellowish brown; pleochroism and dispersion were not observed. Kihlmanite-(Ce) is biaxial (+), α = 1.708(5), β = 1.76(1), γ = 1.82(1) (589 nm), 2Vcalc = 89°. The optical orientation is Y ^ c = 5°, other details are unclear. The calculated and measured densities are 3.694 and 3.66(2) g cm−3, respectively. The mean chemical composition, determined by electron microprobe, is: Na2O 0.13, Al2O3 0.24, SiO2 9.91, CaO 1.50, TiO2 11.04, MnO 0.26, Fe2O3 0.05, Nb2O5 2.79, La2O3 12.95, Ce2O3 27.33, Pr2O3 2.45, Nd2O3 8.12, Sm2O3 1.67, Gd2O3 0.49 wt.%, with CO2 15.0 and H2O 6.0 wt.% (determined by wet chemical and Penfield methods, respectively), giving a total of 99.93 wt.%. The empirical formula calculated on the basis of Si + Al = 1 atom per formula unit is (Ca0.16Na0.11Mn0.02)∑0.29[(Ce0.98La0.47Pr0.09Nd0.29Sm0.06Gd0.02)∑1.91(Ti0.82Nb0.12)∑0.94O2 (Si0.97Al0.03)∑1O4.02(HCO3)2.01](H2O)0.96. The simplified formula is Ce2TiO2(SiO4)(HCO3)2·H2O. The mineral reacts slowly in cold 10% HCl with weak effervescence and fragmentation into separate plates. The strongest X-ray powder-diffraction lines [listed as d in Å(I) (hkl)] are as follows: 15.11(100)(00
, a = 4.994(2), b = 7.54(2), c = 15.48(4) Å, α = 103.5(4), β = 90.7(2), γ = 109.2(2)o , V = 533(1) Å3, Z = 2 (from powder diffraction data) or a = 5.009(5), b = 7.533(5), c = 15.407(5) Å, α = 103.061(5), β = 91.006(5), γ = 109.285(5)°, V = 531.8(7) Å3, Z = 2 (from single-crystal X-ray diffraction data). The mineral was found in the arfvedsonite-aegirine-microcline vein in fenitized metavolcanic rock at the foot of the Mt Kihlman (Chil’man), near the western contact of the Devonian Khibiny alkaline massif and the Proterozoic Imandra-Varzuga greenstone belt. It forms brown spherulites (up to 2 cm diameter) and sheaf-like aggregates of prismatic crystals, flattened on {010} and up to 0.5 mm diameter. Both spherulites and aggregates occur in interstices in arfvedsonite and microcline, in intimate association with golden-green tundrite-(Ce). Kihlmanite-(Ce) is brown, with a vitreous lustre and a pale yellowish-brown streak. The cleavage is perfect on {010}, parting is perpendicular to c and the fracture is stepped. Mohs hardness is ∼3. In transmitted light, the mineral is yellowish brown; pleochroism and dispersion were not observed. Kihlmanite-(Ce) is biaxial (+), α = 1.708(5), β = 1.76(1), γ = 1.82(1) (589 nm), 2Vcalc = 89°. The optical orientation is Y ^ c = 5°, other details are unclear. The calculated and measured densities are 3.694 and 3.66(2) g cm−3, respectively. The mean chemical composition, determined by electron microprobe, is: Na2O 0.13, Al2O3 0.24, SiO2 9.91, CaO 1.50, TiO2 11.04, MnO 0.26, Fe2O3 0.05, Nb2O5 2.79, La2O3 12.95, Ce2O3 27.33, Pr2O3 2.45, Nd2O3 8.12, Sm2O3 1.67, Gd2O3 0.49 wt.%, with CO2 15.0 and H2O 6.0 wt.% (determined by wet chemical and Penfield methods, respectively), giving a total of 99.93 wt.%. The empirical formula calculated on the basis of Si + Al = 1 atom per formula unit is (Ca0.16Na0.11Mn0.02)∑0.29[(Ce0.98La0.47Pr0.09Nd0.29Sm0.06Gd0.02)∑1.91(Ti0.82Nb0.12)∑0.94O2 (Si0.97Al0.03)∑1O4.02(HCO3)2.01](H2O)0.96. The simplified formula is Ce2TiO2(SiO4)(HCO3)2·H2O. The mineral reacts slowly in cold 10% HCl with weak effervescence and fragmentation into separate plates. The strongest X-ray powder-diffraction lines [listed as d in Å(I) (hkl)] are as follows: 15.11(100)(00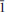 ), 7.508(20)(00
), 7.508(20)(00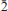 ), 6.912(12)(0
), 6.912(12)(0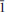 1), 4.993(14)(00
1), 4.993(14)(00 ), 3.563(15)(0
), 3.563(15)(0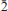 1), 2.896(15)(1
1), 2.896(15)(1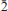
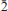 ). The crystal structure of kihlmanite-(Ce) was refined to R1 = 0.069 on the basis of 2441 unique observed reflections (MoKα, 293 K). It is closely related to the crystal structure of tundrite-(Ce) and is based upon [Ce2TiO2(SiO4)(HCO3)2] layers parallel to (001). Kihlmanite-(Ce) can be considered as a cationdeficient analogue of tundrite-(Ce). The mineral is named in honour of Alfred Oswald Kihlman (1858–1938), a remarkable Finnish geographer and botanist who participated in the Wilhelm Ramsay expeditions to the Khibiny Mountains in 1891–1892. The mineral name also reflects its occurrence at the Kihlman (Chil’man) Mountain.
). The crystal structure of kihlmanite-(Ce) was refined to R1 = 0.069 on the basis of 2441 unique observed reflections (MoKα, 293 K). It is closely related to the crystal structure of tundrite-(Ce) and is based upon [Ce2TiO2(SiO4)(HCO3)2] layers parallel to (001). Kihlmanite-(Ce) can be considered as a cationdeficient analogue of tundrite-(Ce). The mineral is named in honour of Alfred Oswald Kihlman (1858–1938), a remarkable Finnish geographer and botanist who participated in the Wilhelm Ramsay expeditions to the Khibiny Mountains in 1891–1892. The mineral name also reflects its occurrence at the Kihlman (Chil’man) Mountain.
GEOLIFE – Geomaterials for the environment, technology and human activities. Preface to the October 2014 special set of papers arising from presentations at the Goldschmidt 2013 conference
- R. Giere, R. Oberti, S. Quartieri, R. Wogelius
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. i-iii
-
- Article
- Export citation
-
This special issue of the Mineralogical Magazine springs from the session we organized as part of the Goldschmidt 2013 conference in Florence. The title we proposed “GEOLIFE – Geomaterials for environment, technology and human activities” reflects our feeling that a wise use of the expertise gained by mineralogists while studying the crystal chemistry, as well as the chemical and physical properties of minerals in rocks and soils, still plays an important role in what is called “The cutting edge in Mineralogy and Mineral Physics”.
Indeed, the current expertise in the physical, chemical and structural characterization of geomaterials, i.e. minerals, rocks and their synthetic analogues, now allows us to design innovative applications both for technological and environmental purposes. During the meeting we discussed some frontier work on phases produced naturally by the Earth ‘laboratory’ or fabricated within academic laboratories, which have the potential to improve the quality of life for current and future generations.
Tl-bearing sulfosalt from the Lengenbach quarry, Binn Valley, Switzerland: Philrothite, TlAs3S5
- L. Bindi, F. Nestola, E. Makovicky, A. Guastoni, L. De Battisti
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 1-9
-
- Article
- Export citation
-
Philrothite, ideally TlAs3S5, is a new mineral from the Lengenbach quarry in the Binn Valley, Valais, Switzerland. It occurs as very rare crystals up to 200 mm across on realgar associated with smithite, rutile and sartorite. Philrothite is opaque with a metallic lustre and shows a dark brown streak. It is brittle; the Vickers hardness (VHN25) is 128 kg/mm2 (range: 120–137) (Mohs hardness of 3–3½). In reflected light philrothite is moderately bireflectant and weakly pleochroic from dark grey to light grey. Under crossed polars it is anisotropic with grey to bluish rotation tints. Internal reflections are absent. Reflectance percentages for the four COM wavelengths (Rmin and Rmax) are: 26.5, 28.8 (471.1 nm), 25.4, 27.2 (548.3 nm), 24.6, 26.3 (586.6 nm) and 24.0, 25.1 (652.3 nm), respectively.
Philrothite is monoclinic, space group P21/c, with a = 8.013(2), b = 24.829(4), c = 11.762(3) Å, β = 132.84(2)°, V = 1715.9(7) Å3, Z = 8. It represents the N = 4 homologue of the sartorite homologous series. In the crystal structure [R1 = 0.098 for 1217 reflections with I > 2σ(I)], Tl assumes tricapped prismatic sites alternating to form columns perpendicular to the b axis. Between the zigzag walls of Tl coordination prisms, coordination pyramids of As(Sb) form diagonally-oriented double layers separated by broader interspaces which house the lone electron pairs of these elements.
The eight strongest calculated powder-diffraction lines [d in Å(I/I0) (hkl)] are: 12.4145 (52) (020); 3.6768 (100) (
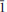 61); 3.4535 (45) (131); 3.0150 (46) (
61); 3.4535 (45) (131); 3.0150 (46) (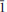 53); 2.8941 (52) (
53); 2.8941 (52) (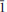 81); 2.7685 (76) (230); 2.7642 (77) (
81); 2.7685 (76) (230); 2.7642 (77) (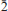 34); 2.3239 (52) (092). A mean of five electron microprobe analyses gave Tl 26.28(12), Pb 6.69(8), Ag 2.50(4), Cu 0.04(2), Hg 0.07(2), As 32.50(13), Sb 3.15(3), S 26.35(10), total 97.58 wt.%, corresponding, on the basis of a total of nine atoms, to (Tl0.789Pb0.198)∑=0.987 (As2.662Sb0.159Ag0.142Cu0.004Hg0.002)∑=2.969S5.044. The new mineral has been approved by the Commission on New Minerals, Nomenclature and Classification (CNMNC) of the International Mineralogical Association (2013-066) and named for Philippe Roth (b. 1963), geophysicist and well known mineral expert on the Lengenbach minerals for more than 25 years.
34); 2.3239 (52) (092). A mean of five electron microprobe analyses gave Tl 26.28(12), Pb 6.69(8), Ag 2.50(4), Cu 0.04(2), Hg 0.07(2), As 32.50(13), Sb 3.15(3), S 26.35(10), total 97.58 wt.%, corresponding, on the basis of a total of nine atoms, to (Tl0.789Pb0.198)∑=0.987 (As2.662Sb0.159Ag0.142Cu0.004Hg0.002)∑=2.969S5.044. The new mineral has been approved by the Commission on New Minerals, Nomenclature and Classification (CNMNC) of the International Mineralogical Association (2013-066) and named for Philippe Roth (b. 1963), geophysicist and well known mineral expert on the Lengenbach minerals for more than 25 years.
Evidence of interspersed co-existing CaCO3-III and CaCO3-IIIb structures in polycrystalline CaCO3 at high pressure
- M. Merlini, W. A. Crichton, J. Chantel, J. Guignard, S. Poli
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 225-233
-
- Article
- Export citation
-
New experimental data are reported on high-pressure polymorphism of CaCO3. The CaCO3-III phase was stabilized using a large-volume press device and high-resolution X-ray powder diffraction (XRPD) patterns were collected from a few mm3 of powder sample. The interpretation of XRPD indicates that CaCO3-III and CaCO3-IIIb structures are present simultaneously and are in similar proportions. The lack of any unindexed peaks demonstrates that these two polymorphs are the only phases in this experiment, indicating that CaCO3-III and CaCO3-IIIb are the structures most likely to occur above 2.5 GPa. Relevant co-axial crystallographic matrix transformations from lower-pressure polymorphs to both CaCO3-III and CaCO3-IIIb are discussed to illustrate a further possible occurrence of co-existing and interspersed stable polymorphs in carbonate systems.
The role of Th-U minerals in assessing the performance of nuclear waste forms
- G. R. Lumpkin, Yan Gao, R. Gieré, C. T. Williams, A. N. Mariano, T. Geisler
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 1071-1095
-
- Article
- Export citation
-
Materials designed for nuclear waste disposal include a range of ceramics, glass ceramics and glass waste forms. Those with crystalline phases have provided the momentum for studies of minerals as a means to understand aspects of waste-form crystal chemistry, behaviour in aqueous systems and radiation damage over geological periods of time. Although the utility of natural analogue studies varies, depending upon the degree of analogy to the proposed geological repository and other factors such as chemical composition, the available data suggest that Th-U host phases such as brannerite, monazite, pyrochlore, zircon and zirconolite are resistant generally to dissolution in aqueous fluids at low temperatures. Geochemical durability may or may not extend to hydrothermal systems depending on the specifics of fluid composition, temperature and pressure. At elevated temperatures, for example, davidite may break down to new phase assemblages including titanite, ilmenite and rutile. Perovskite is generally less resistant to dissolution at low temperatures and breaks down to TiO2, releasing A-site cations to the aqueous fluid. Studies of radiation damage indicate that the oxide and silicate phases become amorphous as a result of the gradual accumulation of alpha-recoil collision cascades. Monazite tends to remain crystalline on geological time scales, a very attractive property that potentially eliminates major changes in physical properties such as density and volume, thereby reducing the potential for cracking, which is a major concern for zircon. In spite of recent success in describing the behaviour of Th-U minerals in geological systems, considerable work remains in order to understand the P-T-X conditions during alteration and T-t history of the host rocks.
Canutite, NaMn3[AsO4][AsO3(OH)]2, a new protonated alluaudite-group mineral from the Torrecillas mine, Iquique Province, Chile
- A. R. Kampf, S. J. Mills, F. Hatert, B. P. Nash, M. Dini, A. A. Molina Donoso
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 787-795
-
- Article
- Export citation
-
The new mineral canutite (IMA2013-070), NaMn3[AsO4][AsO3(OH)]2, was found at two different locations at the Torrecillas mine, Salar Grande, Iquique Province, Chile, where it occurs as a secondary alteration phase in association with anhydrite, halite, lavendulan, magnesiokoritnigite, pyrite, quartz and scorodite. Canutite is reddish brown in colour. It forms as prisms elongated on [20
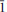 ] and exhibiting the forms {010}, {100}, {10
] and exhibiting the forms {010}, {100}, {10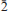 }, {201} and {102}, or as tablets flattened on {102} and exhibiting the forms {102} and {110}. Crystals are transparent with a vitreous lustre. The mineral has a pale tan streak, Mohs hardness of 2½, brittle tenacity, splintery fracture and two perfect cleavages, on {010} and {101}. The calculated density is 4.112 g cm−3. Optically, canutite is biaxial (+) with α = 1.712(3), β = 1.725(3) and γ = 1.756(3) (measured in white light). The measured 2V is 65.6(4)°, the dispersion is r < v (slight), the optical orientation is Z = b; X ^ a = 18° in obtuse β and pleochroism is imperceptible. The mineral is slowly soluble in cold, dilute HCl. The empirical formula (for tabular crystals from near the mineshaft), determined from electron - microprobe analyses, is (Na1.05Mn2.64Mg0.34Cu0.14Co0.03)∑4.20As3O12H1.62. Canutite is monoclinic, C2/c, a = 12.3282(4), b = 12.6039(5), c = 6.8814(5) Å, β = 113.480(8)°, V = 980.72(10) Å3 and Z = 4. The eight strongest X-ray powder diffraction lines are [dobs Å(I)(hkl)]: 6.33(34)(020), 4.12(26)(
}, {201} and {102}, or as tablets flattened on {102} and exhibiting the forms {102} and {110}. Crystals are transparent with a vitreous lustre. The mineral has a pale tan streak, Mohs hardness of 2½, brittle tenacity, splintery fracture and two perfect cleavages, on {010} and {101}. The calculated density is 4.112 g cm−3. Optically, canutite is biaxial (+) with α = 1.712(3), β = 1.725(3) and γ = 1.756(3) (measured in white light). The measured 2V is 65.6(4)°, the dispersion is r < v (slight), the optical orientation is Z = b; X ^ a = 18° in obtuse β and pleochroism is imperceptible. The mineral is slowly soluble in cold, dilute HCl. The empirical formula (for tabular crystals from near the mineshaft), determined from electron - microprobe analyses, is (Na1.05Mn2.64Mg0.34Cu0.14Co0.03)∑4.20As3O12H1.62. Canutite is monoclinic, C2/c, a = 12.3282(4), b = 12.6039(5), c = 6.8814(5) Å, β = 113.480(8)°, V = 980.72(10) Å3 and Z = 4. The eight strongest X-ray powder diffraction lines are [dobs Å(I)(hkl)]: 6.33(34)(020), 4.12(26)(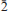 21), 3.608(29)(310,
21), 3.608(29)(310,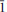 31), 3.296(57)(
31), 3.296(57)(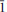 12), 3.150(28)(002,131), 2.819(42)(400,041,330), 2.740(100)(240,
12), 3.150(28)(002,131), 2.819(42)(400,041,330), 2.740(100)(240,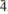 02,112) and 1.5364(31)(multiple). The structure, refined to R1 = 2.33% for 1089 Fo > 4σF reflections, shows canutite to be isostructural with protonated members of the alluaudite group.
02,112) and 1.5364(31)(multiple). The structure, refined to R1 = 2.33% for 1089 Fo > 4σF reflections, shows canutite to be isostructural with protonated members of the alluaudite group.
The crystal structure of camerolaite and structural variation in the cyanotrichite family of merotypes
- S. J. Mills, A. G. Christy, C. Schnyder, G. Favreau, J. R. Price
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 1527-1552
-
- Article
- Export citation
-
We present Raman data for camerolaite, cyanotrichite and carbonatecyanotrichite, and using synchrotron single-crystal X-ray diffraction have solved the structure of camerolaite from the Tistoulet Mine, Padern, Aude Department, France. Camerolaite crystallizes in space group P1 with the unit-cell parameters: a = 6.3310(13) Å, b = 2.9130(6) Å, c = 10.727(2) Å, α = 93.77(3)°, β = 96.34(3)°, γ =79.03(3)º, V = 192.82(7) Å3 and Z = ⅓, with respect to the ideal formula from the refinement, Cu6Al3(OH)18(H2O)2[Sb(OH)6](SO4). The crystal structure was solved to R1 = 0.0890 for all 1875 observed reflections [Fo > 4σFo] and 0.0946 for all 2019 unique reflections. The P cell has been transformed into a C-centred cell that aids comparison with that of the structurally related khaidarkanite by aC = 2aP – bP, giving parameters a = 12.441(3), b = 2.9130(6), c = 10.727(2) Å, α = 93.77(3), β = 95.57(3), γ = 92.32(3)º and Z = ⅔ in C1. Edge-sharing octahedral ribbons Cu2Al(O,OH,H2O)8 form hydrogen-bonded layers || (001), as in khaidarkanite. The partially occupied interlayer Sb and S sites of the average structure are in octahedral and tetrahedral coordination by oxygen, respectively. They cannot be occupied simultaneously, which leads to regular alternation of [Sb(OH)6]– and SO42– groups in rods || y, resulting in local tripling of the periodicity along y for the Sb(OH)6–SO4 rods. Thus, camerolaite has a ‘host–guest’ structure in which an invariant host module (layers of Cu–Al ribbons) has embedded rod-like guest modules with a longer periodicity. Coupling between the phases of these rods is only short-range, resulting in diffuse X-ray scattering rather than sharp superstructure reflections. Similar disorder is known for parnauite, and is deduced for other members of the cyanotrichite group (cyanotrichite, carbonatecyanotrichite and khaidarkanite). Group members all share the Cu–Al ribbon module but have interlayer rods of different compositions and topologies; thus, they form a merotypic family. The low symmetry of the camerolaite average structure suggests other possibilities for structure variation in the group, which are discussed.
Kinetics of the solvent-mediated transformation of hydromagnesite into magnesite at different temperatures
- F. Di Lorenzo, R. M. Rodríguez-Galán, M. Prieto
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 1363-1372
-
- Article
-
- You have access Access
- Open access
- Export citation
-
The process of transforming hydromagnesite to magnesite is analysed in the context of the theory of solvent-mediated transformations. A series of experiments at 120, 150 and 180ºC with different heating times was designed to determine, by powder X-ray diffraction, the amount of magnesite generated as a function of time. The aqueous-phase composition was monitored by inductively coupled plasma-mass spectrometry and carbonate alkalimetry. From the analytical data, the evolution of saturation indexes with respect to both phases was determined using the geochemical code PHREEQC. Finally, two different methods were applied to obtain the activation energy of the process and a TTT (Temperature- Transformation-Time) graph was constructed to define suitable conditions in which to obtain magnesite.
Nestolaite, CaSeO3·H2O, a new mineral from the Little Eva mine, Grand County, Utah, USA
- A. V. Kasatkin, J. Plášil, J. Marty, A. A. Agakhanov, D. I. Belakovskiy, I. S. Lykova
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 497-505
-
- Article
- Export citation
-
Nestolaite (IMA 2013-074), CaSeO3·H2O, is a new mineral species from the Little Eva mine, Grand County, Utah, USA. It is named in honour of the prominent Italian mineralogist and crystallographer Fabrizio Nestola. The new mineral was found on sandstone matrix as rounded aggregates up to 2 mm across and up to 0.05 μm thick consisting of tightly intergrown oblique-angled, flattened to acicular crystals up to 30 μm long and up to 7 μm (very rarely up to 15 μm) thick. Nestolaite associates with cobaltomenite, gypsum, metarossite, orschallite and rossite. The new mineral is light violet and transparent with a white streak and vitreous lustre. The Mohs hardness is 2½. Nestolaite is brittle, has uneven fracture and perfect cleavage on {100}. The measured and calculated densities are Dmeas. = 3.18(2) g/cm3 and Dcalc. = 3.163 g/cm3. Optically, nestolaite is biaxial positive. The refractive indices are α = 1.642(3), β = 1.656(3), γ = 1.722(6). The measured 2V is 55(5)° and the calculated 2V is 51°. In transmitted light nestolaite is colourless. It does not show pleochroism but has strong pseudoabsorption caused by high birefringence. The chemical composition of nestolaite (wt.%, electronmicroprobe data) is: CaO 28.97, SeO2 61.14, H2O (calc.) 9.75, total 99.86. The empirical formula calculated on the basis of 4 O a.p.f.u. (atoms per formula unit) is Ca0.96Se1.02O3·H2O. The Raman spectrum is dominated by the Se–O stretching and O–Se–O bending vibrations of the pyramidal SeO3 groups and O–H stretching modes of the H2O molecules. The mineral is monoclinic, space group P21/c, with a = 7.6502(9), b = 6.7473(10), c = 7.9358(13) Å, β = 108.542 (12)°, V = 388.37(10) Å3 and Z = 4. The eight strongest powder X-ray diffraction lines are [dobs in Å(hkl) (Irel)]: 7.277 (100)(100), 4.949 (110)(37), 3.767 (002)(29), 3.630 (200)(58), 3.371 (020)(24), 3.163 (
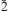 02)(74), 2.9783 (
02)(74), 2.9783 (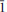 21)(74) and 2.7231 (112)(31). The crystal structure of nestolaite was determined by means of the Rietveld refinement from the powder data to Rwp = 0.019. Nestolaite possesses a layered structure consisting of CaΦ–SeO3 sheets, composed of edge-sharing polyhedra. Adjacent sheets are held by H bonds emanating from the single (H2O) group within the sheets. The nestolaite structure is topologically unique.
21)(74) and 2.7231 (112)(31). The crystal structure of nestolaite was determined by means of the Rietveld refinement from the powder data to Rwp = 0.019. Nestolaite possesses a layered structure consisting of CaΦ–SeO3 sheets, composed of edge-sharing polyhedra. Adjacent sheets are held by H bonds emanating from the single (H2O) group within the sheets. The nestolaite structure is topologically unique.
Cobaltite-rich mineralization in the iron skarn deposit of Traversella (Western Alps, Italy)
- P. Nimis, L. Dalla Costa, A. Guastoni
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 11-27
-
- Article
- Export citation
-
Cobaltite-rich mineralization from the iron skarns of the Traversella magnetite mine (Western Alps, Italy) was studied by reflected-light microscopy, scanning electron microscopy and electron microprobe analysis. Cobaltite is found in carbonate-chlorite-rich rocks at the margins of the main magnetite masses, where it forms disseminations and metasomatic veinlets that postdate the formation of magnetite. The paragenesis includes cobaltite (± arsenopyrite), bismuthinite, pyrrhotite and/or pyrite, chalcopyrite, carbonates, talc, chlorite and native gold, and is indicative of a low-sulfidation environment. The sulfarsenides show oscillatory and sector zoning, which indicates disequilibrium during crystal growth. Compositional variations are mainly due to variations in the Co/Fe ratio of arsenopyrite and in either the Co/Fe or the Ni/(Fe + Co) ratios of the coexisting cobaltite. The Ni contents are low to very low in the cobaltites (<2.4 wt.%) and very low in the arsenopyrites (<0.16 wt.%). The As/S molar ratios in the cobaltites are highly variable (0.59−1.00) and show a broad negative correlation with the Fe contents. The formation of cobaltite is related to circulation of relatively low-temperature (<∼300°C), (Co,As,Bi)-rich fluids during the retrograde sulfidation stage which followed the formation of magnetite. The apparent restriction of cobaltite (+ bismuthinite ± arsenopyrite) to the margins of the main magnetite columns may reflect the establishment of thermochemical gradients around the main direction of infiltration of the retrograde metasomatic fluids.
Alpine oxidation of lithium micas in Permian S-type granites (Gemeric unit, Western Carpathians, Slovakia)
- I. Petrík, Š. Čík, M. Miglierini, T. Vaculovič, I. Dianiška, D. Ozdín
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 507-533
-
- Article
- Export citation
-
Lithium micas of the zinnwaldite and phengite–Li-phengite series occur as characteristic minerals in Permian Li-F-(P) granites of the western Gemeric unit (Western Carpathians) accompanied by topaz, tourmaline, Nb, Ta, Ti, Sn oxides and aluminophosphates. The calculated Li2O contents of all the mica analysed, together with Rb2O and Cs2O were confirmed by LA-ICP-MS analyses for all the identified micas. Samples from three localities were investigated: two surficial (Surovec, Vrchsúl’ová); and one drill hole (Dlhá dolina). Zinnwaldite (polylithionite) occurs in the upper level of the Dlhá dolina granitic intrusion and in the nearby shallow satellite body of Surovec. The lower level porphyritic granites contain only siderophyllite. The Vrchsúl’ ová micas are closer in composition to Li-annite and siderophyllite. Dioctahedral micas are mostly phengites, although zinnwaldite-bearing granites are rich in late-crystallizing Li-phengite, which extensively replaces earlier zinnwaldite. The secondary Liphengite and phengite are interpreted as products of Alpine metamorphism during Cretaceous burial and subsequent exhumation of the Gemeric unit. Reactions are suggested explaining the formation of Li-phengite by reaction of zinnwaldite with phengite or with muscovite. All mica types were investigated by Mössbauer spectroscopy, which showed high degrees of oxidation (25–50% Fe3+ of total Fe) with the exception of zinnwaldite from Vrchsúl’ová, which may have preserved an original, reduced value of 10%. The metamorphic assemblage present permitted calculation of P-T-X conditions: T = 184°C, P = 320 MPa, with oxidation of siderophyllite to phengite + goethite and fO2 at ΔNN = 4.7, confirming the low-grade conditions of the Alpine metamorphism in agreement with previous estimates.
IMA Commission on New Minerals, Nomenclature and Classification (CNMNC) NewsLetter 21
New minerals and nomenclature modifications approved in 2014
- P. A. Williams, F. Hatert, M. Pasero, S. J. Mills
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 797-804
-
- Article
- Export citation
Research Article
Hilgardite polytype distribution in Sussex, New Brunswick evaporite deposits
- J. D. Grice, R. Rowe
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 235-252
-
- Article
- Export citation
-
Hilgardite, Ca2B5O9Cl·H2O, has a zeolite-type framework structure consisting of cross-linked (010) layers of borate sheets having the fundamental building block (FBB) = 2Δ3☐:<Δ2☐>-<Δ2☐>. These layers are polar and the orientation, plus the number of layers in a sequence, determine the polytype. To date three hilgardite polytypes have been described: hilgardite-1A, -3A and -4M. A reliable means of polytype identification by micro X-ray powder diffraction (mXRPD) is outlined. Two polytypes are identified in the Sussex New Brunswick potash deposits: hilgardite-1A and -3A. Both cells are refined with XRPD data: hilgardite-1A, a 6.456(1), b 6.558(1), c 6.295(1) Å, α 61.62(1), β 118.76(1), γ 108.82(2)°, V 205.23(4) Å3 and hilgardite-3A, a 17.484(6), b 6.487(3), c 6.321(3) Å, α 60.78(3), β 79.58(3), γ 84.16(3)°, V 615.3(3) Å3. Hilgardite polytype assemblages are determined for 24 Millstream deposit core samples and 43 Penobsquis deposit core samples. The Millstream deposit occurs as an isolated, inner basin during evaporite formation. Borate formation from this marine environment was during the last stages of evaporation. Simple Mg-Ca borates precipitated from this highly saline brine. It underwent subsequent minor folding and all hilgardite samples are the hilgardite-1A polytype. The Penobsquis deposit occurs as an open fore-basin. This larger, less saline basin also precipitated borate minerals during the final stages of evaporation. The borate assemblage here is much more complex with borate minerals that have essential Ca, K, Fe, Mg and Sr. This deposit was subjected to major folding and recrystallization. Both hilgardite-1A and -3A occur here. Polytype-3A is concentrated in areas of maximum folding and it is proposed that pressure is the main control of hilgardite polytypes. Strontium substitution has no effect on polytype formation.
Crystal structure and topological affinities of magbasite, KBaFe3+Mg7Si8O22(OH)2F6: a trellis structure related to amphibole and carpholite
- M. D. Welch, R. H. Mitchell, A. R. Kampf, A. R. Chakhmouradian, D. Smith, M. Carter
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 29-45
-
- Article
- Export citation
-
The crystal structure of magbasite from the Eldor carbonatite complex, Quebec, Canada, has been determined and indicates that the currently accepted formula should be revised to KBaFe3+Mg7Si8O22(OH)2F6. Magbasite is orthorhombic, space group Cmme (Cmma), with unit-cell parameters a 18.9506(3) Å, b 22.5045(3) Å, c 5.2780(1) Å, V 2250.93(6) Å3 (Z = 4). The structure has been solved and refined to final agreement indices R1 = 0.026, wR2 = 0.052, GooF = 1.116 for a total of 2379 unique reflections, and is a new kind of trellis motif related to amphibole and carpholite topologies. An amphibole-like I-beam ‖(100) of edge-sharing octahedrally-coordinated M(1,2,3) sites, which are filled by Mg, is sandwiched between double-chains of SiO4 tetrahedra ‖c. This I-beam is connected to side-ribbons ‖(010) of edge-sharing (Mg,Fe2+)O4(OH,F)2 and Fe3+O4(OH)2 octahedra to form a tunnelled box or trellis structure very like that of carpholite, for which the I-beams are pyroxene-like. K occupies a tunnel site analogous to the A site of amphibole. Ba occupies a cavity site at the corners where the I-beam and side-ribbon meet, and corresponds to the A site of carpholite. The structural relations between magbasite and carpholite are discussed.
New arsenate minerals from the Arsenatnaya fumarole, Tolbachik volcano, Kamchatka, Russia. II. Ericlaxmanite and kozyrevskite, two natural modifications of Cu4O(AsO4)2
- I. V. Pekov, N. V. Zubkova, V. O. Yapaskurt, D. I. Belakovskiy, M. F. Vigasina, E. G. Sidorov, D. Yu. Pushcharovsky
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 1553-1569
-
- Article
- Export citation
-
Two new minerals, ericlaxmanite and kozyrevskite, dimorphs of Cu4O(AsO4)2, were found in sublimates of the Arsenatnaya fumarole at the Second scoria cone of the Northern Breakthrough of the Great Tolbachik Fissure Eruption, Tolbachik volcano, Kamchatka, Russia. They are associated with each other and with urusovite, lammerite, lammerite-b, popovite, alarsite, tenorite, hematite, aphthitalite, langbeinite, As-bearing orthoclase, etc. Ericlaxmanite occurs as tabular, lamellar, equant or short prismatic crystals up to 0.1 mm in size, their clusters and pseudomorphs after urusovite crystal crusts up to 1.5 cm × 2 cm in area. Kozyrevskite occurs as prismatic crystals up to 0.3 mm long in clusters and as individual crystals. Both minerals are transparent with a vitreous lustre. They are brittle, with Mohs’ hardness ~3–. Ericlaxmanite is green to dark green. Kozyrevskite is bright grass green to light yellowish green; Dcalc is 5.036 (ericlaxmanite) and 4.934 (kozyrevskite) g cm–3. Both minerals are optically biaxial (–); ericlaxmanite: α = 1.870(10), β = 1.900(10), γ = 1.915(10), 2Vmeas = 60(15)º; kozyrevskite: α = 1.885(8), β = 1.895(8), γ = 1.900(8), 2Vmeas. = 75(10)º. The Raman spectra are given. Chemical data (wt.%, electron microprobe; the first value is for ericlaxmanite, the second for kozyrevskite): CuO 57.55, 58.06; ZnO 0.90, 1.04; Fe2O3 0.26, 0.12; SiO2 n.d., 0.12; P2O5 0.23, 1.23; V2O5 0.14, 0.37; As2O5 40.57, 38.78; SO3 0.17, 0.43; total 99.82, 100.15. The empirical formulae, based on 9 O a.p.f.u., are: ericlaxmanite: (Cu3.97Zn0.06Fe0.02)Σ4.05(As1.94P0.02V0.01S0.01)Σ1.98O9 and kozyrevskite: (Cu3.95Zn0.07Fe0.01)Σ4.03(As1.83P0.09S0.03V0.02Si0.01)Σ1.98O9. Ericlaxmanite is triclinic, P
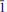 , a = 6.4271(4), b = 7.6585(4), c = 8.2249(3) Å , α = 98.396(4), β = 112.420(5), γ = 98.397(5)º, V = 361.11(3) Å3 and Z = 2. Kozyrevskite is orthorhombic, Pnma, a = 8.2581(4), b = 6.4026(4), c = 13.8047(12) Å , V = 729.90(9) Å3 and Z = 4. The strongest reflections in the X-ray powder patterns [d Å (I)(hkl)] are: ericlaxmanite: 3.868(46)(101), 3.685(100)(020), 3.063(71)(012), 2.957(58)(0
, a = 6.4271(4), b = 7.6585(4), c = 8.2249(3) Å , α = 98.396(4), β = 112.420(5), γ = 98.397(5)º, V = 361.11(3) Å3 and Z = 2. Kozyrevskite is orthorhombic, Pnma, a = 8.2581(4), b = 6.4026(4), c = 13.8047(12) Å , V = 729.90(9) Å3 and Z = 4. The strongest reflections in the X-ray powder patterns [d Å (I)(hkl)] are: ericlaxmanite: 3.868(46)(101), 3.685(100)(020), 3.063(71)(012), 2.957(58)(0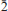 2), 2.777(98)(
2), 2.777(98)(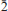 12,
12, 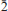
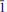 1), 2.698(46)(
1), 2.698(46)(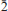
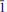 2) and 2.201(51)(013, 031); kozyrevskite: 3.455(100)(004), 3.194(72)(020, 104), 2.910(69)(022), 2.732(82)(122), 2.712(87)(301) and 2.509(92)(123). Their crystal structures, solved from single-crystal X-ray diffraction data [R = 0.0358 (ericlaxmanite) and 0.1049 (kozyrevskite)], are quite different. The ericlaxmanite structure is based on an interrupted framework built by edge- and corner-sharing Cu-centred, distorted tetragonal pyramids, trigonal bipyramids and octahedra. The kozyrevskite structure is based on complicated ribbons of Cu-centred distorted tetragonal pyramids and trigonal bipyramids. Ericlaxmanite is named in honour of the Russian mineralogist, geologist, geographer, biologist and chemist Eric Laxman (1737–1796). Kozyrevskite is named in honour of the Russian geographer, traveller and military man Ivan Petrovich Kozyrevskiy (1680–1734), one of the first researchers of Kamchatka.
2) and 2.201(51)(013, 031); kozyrevskite: 3.455(100)(004), 3.194(72)(020, 104), 2.910(69)(022), 2.732(82)(122), 2.712(87)(301) and 2.509(92)(123). Their crystal structures, solved from single-crystal X-ray diffraction data [R = 0.0358 (ericlaxmanite) and 0.1049 (kozyrevskite)], are quite different. The ericlaxmanite structure is based on an interrupted framework built by edge- and corner-sharing Cu-centred, distorted tetragonal pyramids, trigonal bipyramids and octahedra. The kozyrevskite structure is based on complicated ribbons of Cu-centred distorted tetragonal pyramids and trigonal bipyramids. Ericlaxmanite is named in honour of the Russian mineralogist, geologist, geographer, biologist and chemist Eric Laxman (1737–1796). Kozyrevskite is named in honour of the Russian geographer, traveller and military man Ivan Petrovich Kozyrevskiy (1680–1734), one of the first researchers of Kamchatka.
Uranium and technetium interactions with wüstite [Fe1–xO] and portlandite [Ca(OH)2] surfaces under geological disposal facility conditions
- A. Van Veelen, O. Preedy, J. Qi, G. T. W. Law, K. Morris, J. F. W. Mosselmans, M. P. Ryan, N. D. M. Evans, R. A. Wogelius
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 1097-1113
-
- Article
- Export citation
-
Iron oxides resulting from the corrosion of large quantities of steel that are planned to be installed throughout a deep geological disposal facility (GDF) are expected to be one of the key surfaces of interest for controlling radionuclide behaviour under disposal conditions. Over the lengthy timescales associated with a GDF, the system is expected to become anoxic so that reduced Fe(II) phases will dominate. Batch experiments have therefore been completed in order to investigate how a model reduced Fe-oxide surface (wüstite, Fe1–xO) alters as a function of exposure to aqueous solutions with compositions representative of conditions expected within a GDF. Additional experiments were performed to constrain the effect that highly alkaline solutions (up to pH 13) have on the adsorption behaviour of the uranyl (UO22+) ion onto the surfaces of both wüstite and portlandite [Ca(OH)2; representative of the expected cementitious phases]. Surface co-ordination chemistry and speciation were determined by ex situ X-ray absorption spectroscopy measurements (both X-ray absorption near-edge structure analysis (XANES) and extended X-ray absorption fine structure analysis (EXAFS)). Diffraction, elemental analysis and XANES showed that the bulk solid composition and Fe oxidation state remained relatively unaltered over the time frame of these experiments (120 h), although under alkaline conditions possible surface hydroxylation is observed, due presumably to the formation of surface hydroxyl complexes. The surface morphology, however, is altered significantly with a large degree of roughening and an observed decrease in the average particle size. Reduction of U(VI) to U(IV) occurs during adsorption in almost all cases and this is interpreted to indicate that wüstite may be an effective reductant of U during surface adsorption. This work also shows that increasing the carbonate concentration in reactant solutions dramatically decreases the adsorption coefficients for U on both wüstite and portlandite, consistent with U speciation and surface reactivity determined in other studies. Finally, the EXAFS results include new details about exactly how U bonds to this metal oxide surface.

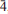 3
3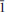 ,
, 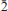 ), 6.912(12)(0
), 6.912(12)(0 ), 3.563(15)(0
), 3.563(15)(0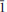 61); 3.4535 (45) (131); 3.0150 (46) (
61); 3.4535 (45) (131); 3.0150 (46) (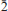 34); 2.3239 (52) (092). A mean of five electron microprobe analyses gave Tl 26.28(12), Pb 6.69(8), Ag 2.50(4), Cu 0.04(2), Hg 0.07(2), As 32.50(13), Sb 3.15(3), S 26.35(10), total 97.58 wt.%, corresponding, on the basis of a total of nine atoms, to (Tl
34); 2.3239 (52) (092). A mean of five electron microprobe analyses gave Tl 26.28(12), Pb 6.69(8), Ag 2.50(4), Cu 0.04(2), Hg 0.07(2), As 32.50(13), Sb 3.15(3), S 26.35(10), total 97.58 wt.%, corresponding, on the basis of a total of nine atoms, to (Tl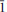 ] and exhibiting the forms {010}, {100}, {10
] and exhibiting the forms {010}, {100}, {10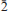 }, {201} and {102}, or as tablets flattened on {102} and exhibiting the forms {102} and {110}. Crystals are transparent with a vitreous lustre. The mineral has a pale tan streak, Mohs hardness of 2½, brittle tenacity, splintery fracture and two perfect cleavages, on {010} and {101}. The calculated density is 4.112 g cm
}, {201} and {102}, or as tablets flattened on {102} and exhibiting the forms {102} and {110}. Crystals are transparent with a vitreous lustre. The mineral has a pale tan streak, Mohs hardness of 2½, brittle tenacity, splintery fracture and two perfect cleavages, on {010} and {101}. The calculated density is 4.112 g cm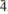 02,112) and 1.5364(31)(multiple). The structure, refined to
02,112) and 1.5364(31)(multiple). The structure, refined to 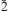 02)(74), 2.9783 (
02)(74), 2.9783 (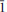 21)(74) and 2.7231 (112)(31). The crystal structure of nestolaite was determined by means of the Rietveld refinement from the powder data to
21)(74) and 2.7231 (112)(31). The crystal structure of nestolaite was determined by means of the Rietveld refinement from the powder data to 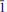 , a = 6.4271(4), b = 7.6585(4), c = 8.2249(3) Å , α = 98.396(4), β = 112.420(5), γ = 98.397(5)º, V = 361.11(3) Å
, a = 6.4271(4), b = 7.6585(4), c = 8.2249(3) Å , α = 98.396(4), β = 112.420(5), γ = 98.397(5)º, V = 361.11(3) Å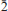 2), 2.777(98)(
2), 2.777(98)(