‘Betel chewing’ describes the practice of masticating a quid of ingredients, including the seed of the Areca catechu palm (betel nut), the leaf of the creeping vine Piper betle and lime, usually in the form of burnt shell or coral. Betel nut is humanity's fourth most widely used drug after nicotine, ethanol and caffeine, and is chewed by millions of people living between the east coast of Africa and the western Pacific (Reference Marshall and LindstromMarshall, 1987).
Nine alkaloids constitute the active ingredients of betel nut (Reference FarnworthFarnworth, 1976), the most abundant of which is arecoline - a potent muscarinic agonist that rapidly crosses the blood-brain barrier and induces a range of parasympathetic effects (Reference Asthana, Greig and HollowayAsthana et al, 1996). Such cholinergic agents are again receiving attention as potential treatments for psychosis (Reference Bodick, Offen and LeveyBodick et al, 1997; Reference TandonTandon, 1999). Our principal hypothesis is that the muscarinic action of betel nut may exert a beneficial effect on the symptoms of people with schizophrenia. Since millions of people with schizophrenia live in betel-chewing regions, an increased understanding of the interaction between betel chewing and schizophrenia should benefit clinical treatment.
METHOD
Research setting
The study was conducted in the Republic of Palau (population 17 000), the westernmost island group in Micronesia. As betel chewing is an integral cultural activity practised by more than 70% of the population (Reference Ysaol, Chilton and CallaghanYsaol et al, 1996; Reference Futterman and LymanFutterman & Lyman, 1998), Palau is an ideal study context, combining a well-described and accessible schizophrenia population (Reference Myles-Worsley, Coon and TiobechMyles-Worsley et al, 1999) and a modern American-style health service.
Subjects
Following ethical approval, the study was carried out at the Belau National Hospital between June and October 1998. The inclusion criteria were chronic schizophrenia or schizoaffective disorder (with mainly schizophrenic course), with an established DSM-III or DSM-IV (American Psychiatric Association, 1980, 1994) diagnosis.
Seventy-six informed and consenting out-patients, all indigenous Palauans, completed the study and were paid for their participation. Five subjects with bipolar disorder and one with acute schizophrenia were excluded, leaving a final pool of 70 subjects (49 men and 21 women). Fifty-four subjects were being treated with either haloperidol or fluphenazine (mainly by depot injection) and 48 were receiving anticholingeric medication. No participants were treated with atypical medications.
A recently completed genetic epidemiological study had identified and diagnosed 160 people with ‘strictly defined’ schizophrenia in Palau (Reference Myles-Worsley, Coon and TiobechMyles-Worsley et al, 1999). A number of these people were now deceased or ‘off-island’, leaving 122 people from the original group; the study sample therefore includes about 57% of the known Palauan schizophrenia population.
Fifty-two subjects (74.3% of the sample) chewed betel nut. However, this group included a proportion of casual users. The ‘serious’ betel chewer carries a kit of ingredients and is readily distinguishable from the ‘social’ user, who does not carry chewing paraphernalia but accepts a quid from peers in social situations. The casual users were included in the non-chewing group. After local advice on defining ‘casual user’, an arbitrary cut-off point was made at two or fewer betel nuts per day as the criterion for inclusion in the non-chewing group. This cut-off produced a chewing group of 40 and a non-chewing group of 30.
A subgroup of 16 subjects (10 chewers and 6 non-chewers) were not receiving antipsychotic pharmacotherapy and were used as a comparison group to control for the effects of medication.
Instruments
The symptomatology of betel chewers was compared with non-chewers using the Positive and Negative Syndrome Scale (PANSS; Reference Kay, Opler and FiszbeinKay et al, 1992), the Extrapyramidal Symptom Rating Scale (ESRS; Reference Chouinard and Ross-ChouinardChouinard & Ross-Chouinard, 1979) and a self-report questionnaire of substance-using habits. In conjunction with demographic details, the substance-use questionnaire asked about consumption of betel nut, cigarettes, alcohol and marijuana. Self-reports were supplemented with reference to chart histories of substance misuse and consultation with case workers.
All rating was carried out by R.J.S. To avoid rater bias, the interviewer was blind to the chewing status of subjects until symptom rating was completed. The test batteries were conducted in English with the assistance of the study participant's case worker - either a psychiatric nurse or social worker. English is the language of instruction in Palauan schools and all subjects spoke English with varying degrees of fluency. The case worker helped each participant to complete the substance-use questionnaire and acted as interpreter when required during the PANSS interview and ESRS assessment that followed. Background information on the participant's social functioning required for the PANSS was obtained from the participant's chart, case worker and family.
Palauan case workers were consulted on the range of PANSS items as they related to each subject, particularly delusional content, communication and cognitive agility, and interpretation of affect. The Structured Clinical Interview for the PANSS (SCI-PANSS; Reference Kay, Opler and FiszbeinKay et al, 1992) was translated into Palauan, then back-translated into English to provide a transcultural reference text for the rater and case workers. The westernised ‘similarities’ and ‘proverbs’ items of the ‘abstract thinking’ section of the PANSS were substituted with Palauan expressions and proverbs.
Statistical tests
Differences in scale scores between sample groups were compared using the independent samples t-test. Non-parametric data were assessed using the χ2-test and the independent samples Mann-Whitney U-test. Correlations between continuous variables were assessed using Pearson's r. All tests were two-tailed. The 95th percentile (0.05) was considered the minimum level of statistically significant difference in all tests.
RESULTS
Demographic and clinical data by chewing status
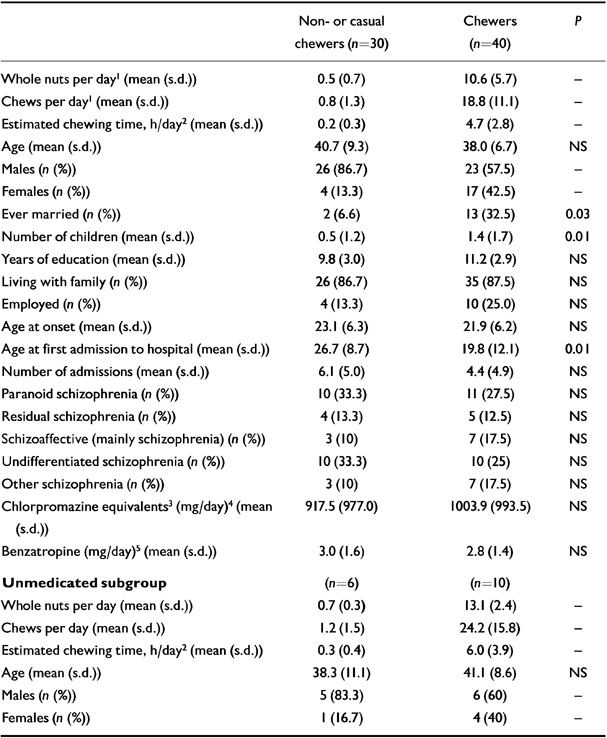
Chewers and non-chewers were significantly different in the proportions who had ever married, in mean number of off-spring and mean age at first admission to hospital (Table 1). With the exception of age at first admission, these differences are an artefact of the uneven gender distribution in the non-chewing group (87% male). The sample exhibits characteristic gender differences in marital status and number of children: 48% (10) of the women were or had been married, v. 10% (5) of the men (P=0.002); and women averaged 2.3 (1.7 s.d.) children v. 0.5 (1.1 s.d.) children per man (P<0.001). However, there were no significant differences in marital status or number of children in intra-gender comparisons (data not shown).
Table 1 Demographic and clinical data by chewing status (n=70). ‘Chewers’ defined as > 2 betel nuts per day
| Non- or casual chewers (n=30) | Chewers (n=40) | P | |
|---|---|---|---|
| Whole nuts per day1 (mean (s.d.)) | 0.5 (0.7) | 10.6 (5.7) | — |
| Chews per day1 (mean (s.d.)) | 0.8 (1.3) | 18.8 (11.1) | — |
| Estimated chewing time, h/day2 (mean (s.d.)) | 0.2 (0.3) | 4.7 (2.8) | — |
| Age (mean (s.d.)) | 40.7 (9.3) | 38.0 (6.7) | NS |
| Males (n (%)) | 26 (86.7) | 23 (57.5) | — |
| Females (n (%)) | 4 (13.3) | 17 (42.5) | — |
| Ever married (n (%)) | 2 (6.6) | 13 (32.5) | 0.03 |
| Number of children (mean (s.d.)) | 0.5 (1.2) | 1.4 (1.7) | 0.01 |
| Years of education (mean (s.d.)) | 9.8 (3.0) | 11.2 (2.9) | NS |
| Living with family (n (%)) | 26 (86.7) | 35 (87.5) | NS |
| Employed (n (%)) | 4 (13.3) | 10 (25.0) | NS |
| Age at onset (mean (s.d.)) | 23.1 (6.3) | 21.9 (6.2) | NS |
| Age at first admission to hospital (mean (s.d.)) | 26.7 (8.7) | 19.8 (12.1) | 0.01 |
| Number of admissions (mean (s.d.)) | 6.1 (5.0) | 4.4 (4.9) | NS |
| Paranoid schizophrenia (n (%)) | 10 (33.3) | 11 (27.5) | NS |
| Residual schizophrenia (n (%)) | 4 (13.3) | 5 (12.5) | NS |
| Schizoaffective (mainly schizophrenia) (n (%)) | 3 (10) | 7 (17.5) | NS |
| Undifferentiated schizophrenia (n (%)) | 10 (33.3) | 10 (25) | NS |
| Other schizophrenia (n (%)) | 3 (10) | 7 (17.5) | NS |
| Chlorpromazine equivalents3 (mg/day)4 (mean (s.d.)) | 917.5 (977.0) | 1003.9 (993.5) | NS |
| Benzatropine (mg/day)5 (mean (s.d.)) | 3.0 (1.6) | 2.8 (1.4) | NS |
| Unmedicated subgroup | (n=6) | (n=10) | |
| Whole nuts per day (mean (s.d.)) | 0.7 (0.3) | 13.1 (2.4) | — |
| Chews per day (mean (s.d.)) | 1.2 (1.5) | 24.2 (15.8) | — |
| Estimated chewing time, h/day2 (mean (s.d.)) | 0.3 (0.4) | 6.0 (3.9) | — |
| Age (mean (s.d.)) | 38.3 (11.1) | 41.1 (8.6) | NS |
| Males (n (%)) | 5 (83.3) | 6 (60) | — |
| Females (n (%)) | 1 (16.7) | 4 (40) | — |
Among chewers the average betel nut consumption was 10.6 (5.7 s.d.) whole nuts (18.8 (11.1 s.d.) quids) per day. This figure is probably conservative, as an uncharacteristic dry season, attributed popularly to El Niño, resulted in a shortage of betel nut over the first few months of the study period: 29 members of the chewing group (72.5%) said that they were chewing less frequently than usual.
With the exception of the positive and negative sub-scales (r=0.18), the PANSS sub-scales and total score are significantly intercorrelated. Therefore, although all PANSS sub-scale and symptom cluster data are reported with associated P values, statistically valid scale comparisons should be limited to those between the positive and negative sub-scales.
The mean PANSS scores for the chewing group were significantly lower than those for the non-chewing group on the positive, negative and general psychopathology sub-scales, as was the total score (Table 2). This trend was repeated in symptom cluster measurements of thought disturbance, paranoid belligerence and anergia. Scores on the ESRS of extrapyramidal symptoms (EPS) and tardive dyskinesia (TD) were not significantly different between the two groups. In comparison to a normative United States PANSS sample of 240 medicated North American patients with schizophrenia (Reference Kay, Opler and FiszbeinKay et al, 1992), the positive and negative scale scores of non-chewers were ‘average’, and those of chewers were ‘slightly below’ to ‘below average’, suggesting that these group-symptomatology profiles are broadly comparable transculturally.
Table 2 All subjects with schizophrenia: betel chewers (> 2 betel nuts per day) v. non-chewers (n=70)

| Chewers (mean (s.d.)) | Non-chewers (mean (s.d.)) | t | P | |
|---|---|---|---|---|
| Positive and Negative Syndrome Scale (PANSS) | (n=40) | (n=30) | ||
| Positive syndrome scale1 | 14.3 (5.7) | 19.2 (6.6) | -3.3 | 0.001 |
| Negative syndrome scale1 | 14.4 (6.7) | 19.7 (7.1) | -3.2 | 0.002 |
| General psychopathology scale | 31.7 (8.1) | 38.4 (9.0) | -3.2 | 0.002 |
| Composite scale | -0.2 (8.1) | -0.6 (10.0) | 0.2 | NS |
| Total PANSS score | 60.5 (16.5) | 77.3 (17.3) | -4.1 | <0.001 |
| Thought disturbance symptom cluster | 8.4 (4.1) | 10.8 (4.4) | -2.3 | 0.02 |
| Paranoid belligerence symptom cluster | 5.1 (2.1) | 7.3 (3.2) | -3.2 | 0.002 |
| Anergia symptom cluster | 8.5 (3.8) | 10.5 (4.4) | -2.0 | 0.05 |
| Activation symptom cluster | 8.9 (1.6) | 9.6 (1.9) | -1.6 | NS |
| Depression symptom cluster | 9.0 (3.6) | 9.5 (3.7) | -0.5 | NS |
| Extrapyramidal Symptom Rating Scale (ESRS) | (n=39) | (n=29) | ||
| Parkinsonism | 11.4 (9.1) | 10.4 (5.8) | 0.5 | NS |
| Tardive dyskinesia | 3.1 (3.4) | 2.4 (2.6) | 0.9 | NS |
In the unmedicated subgroup, chewers scored significantly lower on the scale for negative symptoms and the anergia symptom cluster, on the general psychopathology scale and in total score (Table 3). There were no significant between-group differences in positive symptoms. The unmedicated chewers consumed more betel nut than medicated chewers (24.2 v. 18.8 quids/day) with an associated increase in estimated chewing time from 4.7 to 6 h/day (Table 1). No significant differences in EPS or TD scores emerged between the two groups.
Table 3 Subjects with schizophrenia not receiving medication: betel chewers (> 2 betel nuts per day) v. non-chewers (n=16)

| Chewers (mean (s.d.)) | Non-chewers (mean (s.d.)) | t | P | |
|---|---|---|---|---|
| Positive and Negative Syndrome Scale (PANSS) | (n=10) | (n=6) | ||
| Positive syndrome scale1 | 15.4 (7.2) | 17.8 (7.2) | -0.6 | NS |
| Negative syndrome scale1 | 12.7 (5.8) | 24.0 (8.2) | -3.2 | 0.006 |
| General psychopathology scale | 29.6 (9.5) | 42.2 (13.9) | -2.2 | 0.05 |
| Composite scale | 2.7 (5.4) | -6.2 (10.2) | 2.0 | NS |
| Total PANSS score | 57.7 (20.8) | 84.0 (24.4) | -2.3 | 0.04 |
| Thought disturbance symptom cluster | 8.7 (5.0) | 9.6 (3.6) | -0.4 | NS |
| Paranoid belligerence symptom cluster | 6.0 (2.8) | 7.8 (5.2) | -0.9 | NS |
| Anergia symptom cluster | 7.4 (3.0) | 14.3 (5.1) | -3.4 | 0.004 |
| Activation symptom cluster | 8.0 (1.4) | 9.3 (2.6) | -1.1 | NS |
| Depression symptom cluster | 8.4 (4.5) | 9.2 (4.5) | -0.3 | NS |
| Extrapyramidal Symptom Rating Scale (ESRS) | (n=9) | (n=5) | ||
| Parkinsonism | 13.3 (13.1) | 4.2 (4.0) | 1.49 | NS |
| Tardive dyskinesia | 1.4 (2.4) | 3.0 (3.3) | -1.0 | NS |
Other substances
Betel chewing was not associated with the use of other recreational substances, whereas nicotine, alcohol and marijuana consumption were significantly positively correlated (Table 4).
Table 4 Correlation coefficients: substance consumption per day

| Betel | Cigarettes | Alcohol | Marijuana | |
|---|---|---|---|---|
| Betel | 1.0 | -0.43* | -0.04 | -0.17 |
| Cigarettes | 1.0 | 0.29* | 0.44** | |
| Alcohol | 1.0 | 0.53** | ||
| Marijuana | 1.0 |
A significant negative correlation between cigarette smoking and betel chewing was found, that is, subjects tended to be either exclusively chewers or smokers (Table 4). However, most betel chewers included tobacco as an ingredient of their chewing quid, resulting in a majority of subjects (91.4%) consuming tobacco either as part of a chewing quid or as smoked cigarettes. Smokers, none the less, consumed more tobacco, at an average of 13.8 cigarettes/day v. 6.1 for those who included tobacco in the betel quid.
Alcohol and marijuana consumption were not significantly related to PANSS symptom scores. The relationship between cigarette smoking and schizophrenia symptoms was a reversal of the betel data: the total PANSS score of the smoking group was significantly higher than that of the non-smoking group (t=3.13, P=0.002).
DISCUSSION
Although it has been previously suggested that betel nut alkaloids should be considered in the search for pharmacological treatments for schizophrenia (Reference SmythiesSmythies, 1977), to our knowledge this suggestion has not been pursued and this is the first study to investigate the effects of betel nut directly on the symptoms of people with schizophrenia.
Our results indicated that betel chewing is associated with less severe symptoms of schizophrenia as measured by the PANSS. Chewers scored significantly lower than non-chewers on the positive and negative symptom measures of the PANSS. The symptom score differences between groups were modest for the total group and balanced between positive and negative symptoms. When only subjects not receiving medication were considered, the group difference was substantial, mainly for negative symptoms. Among all subjects, the group total scores of chewers were significantly lower than those of non-chewers (60.5 v. 77.3, t= ‒4.1, P≤0.001) and among unmedicated subjects the difference in mean total PANSS score between chewers and non-chewers was dramatic (57.7 v. 84.0, t= ‒2.3, P=0.04).
Muscarinic agonists in schizophrenia
The main study hypothesis, that betel chewing may exert a beneficial effect on the primary symptoms of schizophrenia, is supported by these results, and the muscarinic agonist action of the most abundant betel nut alkaloid, arecoline (Reference FarnworthFarnworth, 1976), provides the most promising pharmacological explanation for this effect.
Despite ambiguous results in early research (Reference Davis, Berger and HollisterDavis et al, 1978), a number of researchers propose that cholinergic agents may modulate dopaminergic hyperactivity and prevent the emergence of positive symptoms (Reference Friedhoff and AlpertFriedhoff & Alpert, 1973; Reference Davis, Berger and HollisterDavis et al, 1978; Reference Tandon and GredenTandon & Greden, 1989; Reference TandonTandon, 1999). Research suggests that muscarinic agonist derivatives of arecoline may exert an atypical-like action, ameliorating both negative and positive symptoms. Bodick et al (Reference Bodick, Offen and Levey1997) report that the selective M1 agonist xanomeline, a thiadiazole derivative of arecoline (Reference Moltzen and BjornholmMoltzen & Bjornholm, 1995), produced dose-dependent reductions in delusions, hallucinations and other psychotic behaviours in a clinical trial with patients diagnosed with Alzheimer's disease. Shannon et al (Reference Shannon, Bymaster and Bodick1998) performed preclinical rodent studies assessing the use of xanomeline as an antipsychotic and produced results consistent with the performance of atypical agents. They conclude that “xanomeline may provide a novel approach to the treatment of psychosis with potential for a rapid onset of action, efficacy against positive and negative symptoms, and with little or no liability to produce extra-pyramidal side-effects” (see also studies on other muscarinic agents by Bymaster et al (Reference Bymaster, Shannon and Rasmussen1998) and Shannon et al (Reference Shannon, Bymaster and Calligaro1999)).
Betel nut arecoline may have similar effects to those of its derivatives described above. Betel chewers hold the betel quid in the buccal cheek cavity, utilising an absorption route that avoids first-pass metabolism and maintaining betel alkaloids in the blood stream for extended periods. The non-selective agonist action of arecoline may exert a crude atypical-like antipsychotic effect, in conjunction with the parasympathetic effects routinely tolerated by habitual users. Such an action may explain the favourable effect on negative symptoms and the generally mild EPS and TD among betel-chewing subjects with schizophrenia.
Extrapyramidal symptoms and tardive dyskinesia
Extrapyramidal symptoms resulting from betel nut consumption have been reported previously (Reference DeahlDeahl, 1989). As discussed above, no significant differences emerged in ratings of EPS or TD between chewers and non-chewers. Additionally, no significant differences emerged in dosages of neuroleptic or anticholinergic medication.
Despite compliant (i.e. mainly depot) long-term neuroleptic medication with substantial dosages for many subjects, symptoms of TD were fairly infrequent among the study participants. Unambiguous TD symptoms, such as choreoathetoid or bucco-lingual movements, were seen in only 7 of the 70 participants (10%). In comparison, a previous analysis of 76 studies (n=39187) has reported a TD prevalence of 24.2% cross-culturally (Reference Yassa and JesteYassa & Jeste, 1992).
Other substances
In accordance with findings reported elsewhere (Reference Chong and ChooChong & Choo, 1996), our results show that smokers' PANSS scores were significantly higher than non-smokers'. The possibility that the favourable association between PANSS score and betel chewing is an artefact of non-smoking is unlikely, because most chewers consumed tobacco in their quid.
Similarly, the finding that betel nut tends to be used to the exclusion of other substances is of interest, but is unlikely to explain the favourable association between betel chewing and milder symptoms of schizophrenia, as neither marijuana nor alcohol consumption were significantly related to group PANSS scores.
Social variability
Betel chewing is a social activity in Micronesia and it may be associated with milder symptomatology simply because the practice itself is indicative of, or marks a return to, ‘normal’ social functioning (Reference WilsonWilson, 1979). However, a social functionality explanation for group differences in scale scores is not supported by the data, since there were no significant chewing v. non-chewing group differences in regard to demographic indicators of social functionality - marital status, number of children, living situation or employment status. Additionally, an assessment of social functioning is implicit in the structure of the PANSS instrument via input from family members and case workers. However, a suitable social functioning instrument is recommended in any subsequent research to more directly clarify associations between social functioning and betel chewing.
CLINICAL IMPLICATIONS AND LIMITATIONS
CLINICAL IMPLICATIONS
-
▪ In a sample comprising more than half of the known schizophrenia population in Palau, the symptoms of betel chewers as measured by total Positive and Negative Syndrome Scale (PANSS) scores were significantly milder than those of non-chewers. The differences were most dramatic in a small group of unmedicated subjects.
-
▪ Chewing was not significantly associated with increases in extrapyramidal symptoms (EPS), despite the cholinergic effects of betel chewing. However, EPS may have been masked in medicated subjects, as the majority were treated with anticholinergic medication.
-
▪ Betel chewing was not associated with the use of alcohol or marijuana and was inversely related to cigarette smoking.
LIMITATIONS
-
▪ The relationship between milder symptomatology as measured by the PANSS and betel chewing is associative rather than causal. The study results highlight the need for pharmacokinetic/dynamic research of betel nut alkaloids via the buccal route, in conjunction with further research on the muscarinic cholinergic aspects of schizophrenia.
-
▪ The cross-sectional study design has methodological limitations and a prospective design is recommended for subsequent research.
-
▪ The analysis of social factors was limited to demographic data and the PANSS instrument. A suitable social functioning instrument is recommended in subsequent research, to clarify associations between social functioning and betel chewing.
ACKNOWLEDGEMENTS
The authors thank the participating subjects and the many people instrumental in the completion of this study. In New Zealand: Dr J. Collier and Dr A. Futterman-Collier, M. Harrison, Professor R. Kydd, Professor R. Miller, Dr G. Robinson. In Micronesia: P. Aribuk, Dr G. Dever, A. Franz, Rev. F. Hezel, G. Johanes, H. Masayos, Dr M. Myles-Worsley, H. Ngiralmau, A. Lyman, D. Patris, Dr A. Polloi, Senator Peter and Akiko Sugiyama, Minister M. Ueda and the staff at Belau National Hospital. This research was supported by grants from the New Zealand Schizophrenia Fellowship and The University of Auckland Research Committee. This study is dedicated to the memory of Dr A. Polloi.







eLetters
No eLetters have been published for this article.