Symptomatic relapse in schizophrenia is both distressing and costly. It can devastate the lives not only of patients, but also of their families. The debilitating symptoms require specialist health care interventions and targeted treatments, with potentially high costs. It has been estimated, for example, that relapse cost $2 billion just for readmissions to hospital in the USA, almost a decade ago (Reference Weiden and OlfsonWeiden & Olfson, 1995). There is no equivalent estimate for the UK. This study aimed to compare costs, clinical outcomes and quality of life (QoL) for patients with schizophrenia in the UK according to whether or not they had experienced a relapse in the previous 6 months.
METHOD
Study sample
Patients were randomly selected from current (active) psychiatric case-loads drawn from urban and suburban areas of the English city of Leicester. Consultant psychiatrists or senior responsible medical staff were approached by a project research psychiatrist and asked for a list of patients with a possible diagnosis of schizophrenia. Full lists were obtained from five consultants covering city and suburban catchment areas of Leicester. An additional five consultants were also approached to identify patients with the diagnosis who had experienced a relapse within the past 6 months. Patients were excluded if they were living outside this area when the sampling was undertaken. Patients from rural areas of Leicestershire were excluded. The sampling procedure was designed to recruit equal numbers of relapse and non-relapse cases.
Patients were included as participants if they had received a diagnosis of schizophrenia according to DSM–IV criteria (American Psychiatric Association, 1994), had no other psychosis, were aged 18–64 years, and gave their informed consent. Patients were excluded from the study if they were roofless, continuously hospitalised for 12 months or more, about to move residence, already participating in a clinical trial, or unable to participate for language reasons. Although such biases were not specifically controlled for, clinicians took every step to avoid biases in the socio-economic and demographic profiles of patients.
Relapse criteria
Many alternative definitions of relapse in schizophrenia have been published (see Reference LaderLader, 1995, for review). These include number of admissions to hospital, detention under a section of the Mental Health Act, attendance at an acute day care centre, change of antipsychotic agent, increased staff input and/or more intensive case staff management, and a significant change in accommodation. Relapse was identified retrospectively in this study as the re-emergence or aggravation of psychotic symptoms for at least 7 days during the 6 months prior to the study. In addition to instances of relapse pointed out by clinical staff, recorded changes in mental state were regarded as significant and amounting to relapse if there was a clearly documented assessment of a relapse. A change in management as appropriate might also have occurred but not necessarily, and not all relapses led to readmission. Relapse could thus be identified in cases of patients who had been admitted to hospital in the past 6 months, who had consulted their psychiatrist and had had their medication changed for deterioration in their condition, or who had had an increase in intensive support at home from the community mental health team. A planned hospital admission was not classed as a relapse. A research team specialist registrar advised the researcher on any case-note descriptions or accounts from staff that were unclear.
Instrumentation
Data were collected especially for this study. Data collection was based on information obtained directly from case notes and from interviews with the patients in which rating scales were completed (patients gave informed written consent). The information had not been extracted for any other or prior reason.
We used the Positive and Negative Syndrome Scale (PANSS; Reference Kay, Fiszbein and OplerKay et al, 1987), one question from the Clinical Global Impression scale (CGI; Reference GuyGuy, 1976) covering severity of illness, the Global Assessment of Functioning (GAF; American Psychiatric Association, 1987), the Lehman Quality of Life scale (Reference Lehman, Thornicroft and TansellaLehman, 1996), the visual analogue scale from the EuroQoL EQ–5D health-related quality of life measure (Reference Kind and SpilkerKind, 1996) and the Client Service Receipt Inventory (CSRI; Beecham & Knapp, Reference Beecham, Knapp, Thornicroft, Brewin and Wing1992, Reference Beecham, Knapp and Thornicroft2001). Unit costs attached to services were national average figures for the period over which clinical and service use data were collected, at 1998–9 prices (Reference Netten, Dennett and KnightNetten et al, 1999).
Statistical analyses
Depending on the distribution of key variables, parametric (independent t-test) and non-parametric (Mann–Whitney, Kruskal–Wallis) tests were carried out to check for significant differences in mean costs, clinical and QoL outcomes by relapse status. The Pearson chi-squared statistic was used to test for significant differences between categorical measures and relapse status, and for other relapse criteria.
The survey design also permitted multivariate analysis to examine simultaneously some of the potential correlates of relapse status and costs, although it should be noted that the study did not include a full range of possible associations with relapse (see for example, Reference Robinson, Woerner and MaRobinson et al, 1999). First, a generalised linear model (GLM) with a logit link function was used to predict whether a patient had experienced a relapse or not. The logit GLM is similar to the standard logistic model but also produces a measure of dispersion (the variance of the unexplained part of the model). Odds ratios are presented which show the likelihood of relapse given particular patient characteristics. Second, because costs were skewed to the right (although only 5% were zero values), standard ordinary least squares estimates were inappropriate (cf. Reference Dunn, Mirandola and AmaddeoDunn et al, 2003). The results presented are based on a reduced-form GLM model, with a log link function and a Gaussian variance function. Compared with other standard GLM specifications, this produced the best-fitting model in terms of mean predicted cost levels. It also produced the most efficient estimates in terms of lower standard errors and smaller confidence intervals. The statistical analyses were carried out using the Statistical Package for the Social Sciences version 9 for descriptive comparisons and STATA version 6 for the multivariate analyses.
RESULTS
Sample
We identified 257 patients potentially eligible to participate in the study. Of these, 12 refused to take part, 67 were not interviewed because of staff concerns, 12 could not be contacted, and 9 were judged by the interviewer to be too ill; in three cases it was felt to be unsafe to see the patient at home.
A total of 145 patients completed interviews in the study: 77 relapse cases and 68 non-relapse cases. Another 9 patients who were also interviewed were excluded because of incomplete records or inconsistent data. The limited information available on them suggests that most would have been assigned to the non-relapse group and, if included, their cases would have had little impact on average costs.
Relapse and patient characteristics
Relapse status was defined on the basis of re-emergence or aggravation of psychotic symptoms. Table 1 lists other patient characteristics previously employed to define relapse (Reference LaderLader, 1995). Not surprisingly, relapse cases were characterised by higher rates of hospitalisation (63%), reemergence of psychotic symptoms (60%) and aggravation of positive or negative symptoms (43%), and an increased level of staff input or more intensive case staff management (33%) (all P<0.05).
Table 1 Criteria for assignment to relapse or non-relapse study group

| Variable | Non-relapse (n=68) % | Relapse (n=77) % |
|---|---|---|
| Significant change in management directly related to illness or | 0 | 100 |
| treatment side-effects1 | ||
| Change in clinical state | ||
| Re-emergence of psychotic symptoms2 | 0 | 60 |
| Aggravation of positive or negative symptoms2 | 0 | 43 |
| Change in management | ||
| Hospital admission in past 6 months2 | 0 | 63 |
| Detention under section of Mental Health Act2 | 0 | 20 |
| Acute day care3 | 0 | 5 |
| Change of antipsychotic agent2 | 0 | 21 |
| Increased staff input, more intensive case staff management2 | 0 | 33 |
| Significant change in accommodation3 | 0 | 5 |
Compared with the non-relapse group, patients who had recently experienced a relapse had been more recently admitted to a psychiatric ward (using actual years: 1997 and 1992, P<0.05), and experienced a higher number of admissions (5.6 and 3.3, P<0.05). Although patients in the non-relapse group appeared to have spent longer in hospital, the difference was not significant (Table 2). There was no difference between the relapse and non-relapse groups with respect to gender, ethnic group, marital status, employment status or highest level of education (Table 3). Relapse patients were more likely to be living alone (P<0.05). Mean ages were 37.9 (s.d.=10.7) years for relapse patients and 41.1 (s.d.=11.1) years for non-relapse patients (not significantly different).
Table 2 Characteristics of service contact prior to study entry
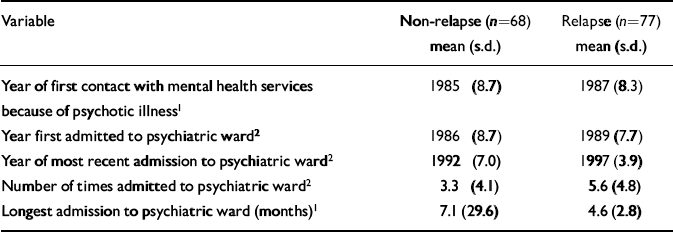
| Variable | Non-relapse (n=68) mean (s.d.) | Relapse (n=77) mean (s.d.) |
|---|---|---|
| Year of first contact with mental health services because of psychotic illness1 | 1985 (8.7) | 1987 (8.3) |
| Year first admitted to psychiatric ward2 | 1986 (8.7) | 1989 (7.7) |
| Year of most recent admission to psychiatric ward2 | 1992 (7.0) | 1997 (3.9) |
| Number of times admitted to psychiatric ward2 | 3.3 (4.1) | 5.6 (4.8) |
| Longest admission to psychiatric ward (months)1 | 7.1 (29.6) | 4.6 (2.8) |
Table 3 Socio-economic and demographic characteristics of the participants

| Variable | Non-relapse (n=68) % | Relapse (n=77) % |
|---|---|---|
| Gender | ||
| Female | 47.1 | 32.8 |
| Ethnic group1 | ||
| White | 82.4 | 83.1 |
| Black Caribbean | 4.4 | 2.6 |
| Indian | 11.8 | 13.0 |
| Other | 1.4 | 1.3 |
| Marital status1 | ||
| Single | 55.9 | 74.0 |
| Married/cohabiting | 26.5 | 11.7 |
| Divorced/separated | 16.2 | 10.4 |
| Widowed | 1.4 | 3.9 |
| Highest educational level1 | ||
| Primary | 4.4 | 1.3 |
| Secondary | 88.2 | 76.6 |
| Tertiary/further | 4.4 | 13.0 |
| Other (not specified) | 2.9 | 9.1 |
| Living arrangements2 | ||
| Alone at home | 19.1 | 37.7 |
| With family/others | 53.0 | 35.1 |
| Collective accommodation | 22.1 | 11.7 |
| Other (not specified) | 5.8 | 15.6 |
| Employment1 | ||
| Not working | 94.1 | 97.4 |
Clinical health and quality of life
Although higher scores on the PANSS and the CGI suggested worse symptoms for relapse compared with non-relapse cases, the differences were not statistically significant. However, GAF scores indicated worse symptoms for relapse patients (P<0.05; Table 4).
Table 4 Clinical characteristics and quality of life
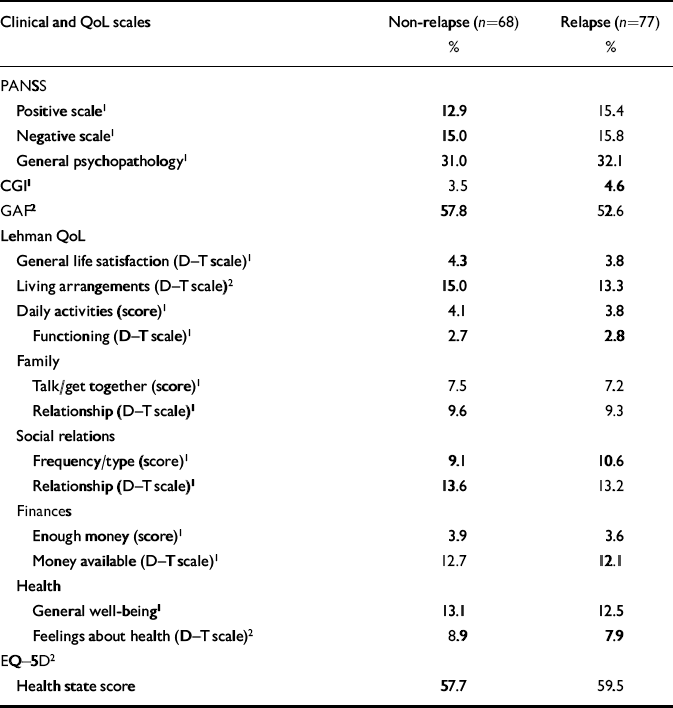
| Clinical and QoL scales | Non-relapse (n=68) % | Relapse (n=77) % |
|---|---|---|
| PANSS | ||
| Positive scale1 | 12.9 | 15.4 |
| Negative scale1 | 15.0 | 15.8 |
| General psychopathology1 | 31.0 | 32.1 |
| CGI1 | 3.5 | 4.6 |
| GAF2 | 57.8 | 52.6 |
| Lehman QoL | ||
| General life satisfaction (D—T scale)1 | 4.3 | 3.8 |
| Living arrangements (D—T scale)2 | 15.0 | 13.3 |
| Daily activities (score)1 | 4.1 | 3.8 |
| Functioning (D—T scale)1 | 2.7 | 2.8 |
| Family | ||
| Talk/get together (score)1 | 7.5 | 7.2 |
| Relationship (D—T scale)1 | 9.6 | 9.3 |
| Social relations | ||
| Frequency/type (score)1 | 9.1 | 10.6 |
| Relationship (D—T scale)1 | 13.6 | 13.2 |
| Finances | ||
| Enough money (score)1 | 3.9 | 3.6 |
| Money available (D—T scale)1 | 12.7 | 12.1 |
| Health | ||
| General well-being1 | 13.1 | 12.5 |
| Feelings about health (D—T scale)2 | 8.9 | 7.9 |
| EQ—5D2 | ||
| Health state score | 57.7 | 59.5 |
Using the Lehman ‘delighted–terrible’ (D–T) scale and scores, relapse patients appeared to experience lower QoL than non-relapse patients on most dimensions, but the differences were small and not statistically significant, except for the items ‘living arrangements’ and ‘feelings about current health’ (P<0.05). There was perhaps some inconsistency in the QoL findings since relapse patients scored slightly better on the EQ–5D visual analogue scale compared with non-relapse patients (P<0.05). However, the EQ–5D measures own health state today, whereas the Lehman score covers broader dimensions of quality of life.
Resources and costs
Six-month service use rates and costs per patient are summarised in Table 5. Costs for relapse cases were four times higher than those for non-relapse cases – £ 8212 compared with £1899 (P<0.05) – with much of the cost difference accounted for by in-patient days. During the 6 months prior to the study, patients in the relapse group spent a mean of 58 days in hospital – although this figure was inflated by six patients who were continuously in hospital for the entire period. By design and selection, nobody in the non-relapse group experienced any hospitalisation in this period.
Table 5 Mean 6-month service use and costs (£, 1998) per patient by relapse status
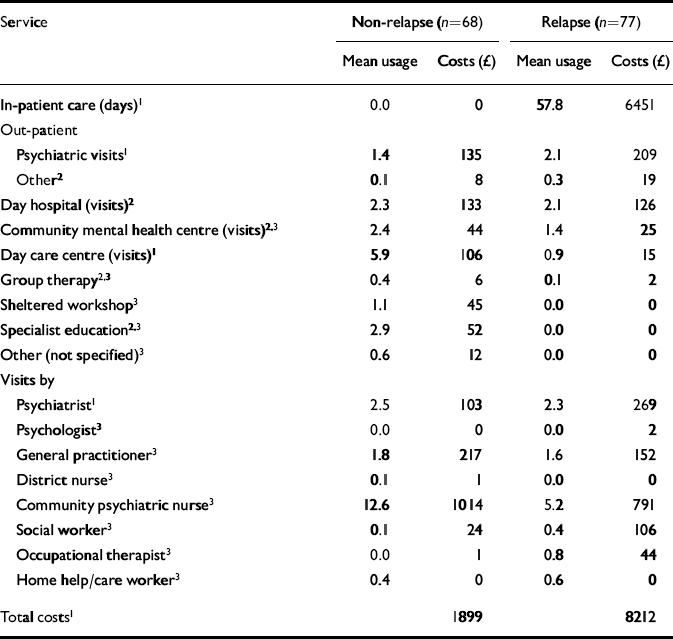
| Service | Non-relapse (n=68) | Relapse (n=77) | ||
|---|---|---|---|---|
| Mean usage | Costs (£) | Mean usage | Costs (£) | |
| In-patient care (days)1 | 0.0 | 0 | 57.8 | 6451 |
| Out-patient | ||||
| Psychiatric visits1 | 1.4 | 135 | 2.1 | 209 |
| Other2 | 0.1 | 8 | 0.3 | 19 |
| Day hospital (visits)2 | 2.3 | 133 | 2.1 | 126 |
| Community mental health centre (visits)2,3 | 2.4 | 44 | 1.4 | 25 |
| Day care centre (visits)1 | 5.9 | 106 | 0.9 | 15 |
| Group therapy2,3 | 0.4 | 6 | 0.1 | 2 |
| Sheltered workshop3 | 1.1 | 45 | 0.0 | 0 |
| Specialist education2,3 | 2.9 | 52 | 0.0 | 0 |
| Other (not specified)3 | 0.6 | 12 | 0.0 | 0 |
| Visits by | ||||
| Psychiatrist1 | 2.5 | 103 | 2.3 | 269 |
| Psychologist3 | 0.0 | 0 | 0.0 | 2 |
| General practitioner3 | 1.8 | 217 | 1.6 | 152 |
| District nurse3 | 0.1 | 1 | 0.0 | 0 |
| Community psychiatric nurse3 | 12.6 | 1014 | 5.2 | 791 |
| Social worker3 | 0.1 | 24 | 0.4 | 106 |
| Occupational therapist3 | 0.0 | 1 | 0.8 | 44 |
| Home help/care worker3 | 0.4 | 0 | 0.6 | 0 |
| Total costs1 | 1899 | 8212 | ||
Psychiatric out-patient visits were also significantly more common in relapse than in non-relapse cases (mean cost £209 v. £ 135, P<0.05). On the other hand, there was slightly higher use by patients in the non-relapse group of day care centres, group therapy, sheltered workshops, specialist education, general practitioners and community psychiatric nurse (CPN) visits, but apart from day care centres none of the differences was statistically significant at the 5% level. Services are complements, in the sense that patients with greater morbidity are likely to use more of a number of services, but are also substitutes, in that (for example) hospital in-patients will have less need and less opportunity to use day care, primary care and CPN support. These two tendencies may have cancelled out for this sample.
Relapse correlates
Given the (expected) high costs associated with illness relapse, correlates of relapse and non-relapse status were examined. The odds ratios in Table 6 indicate that, controlling for all other explanatory factors, there was an increased risk of relapse associated with:
-
(a) each year of age (OR=1.07);
-
(b) fewer years since recent hospital admission (converting the tabulated OR: 1/0.79=1.27);
-
(c) previous suicide or self-harm attempts (OR=3.93);
-
(d) increased social functioning (OR=1.29);
-
(e) lower scores on the GAF (converting the tabulated OR: 1/0.93=1.08) (all P<0.05).
Table 6 Factors associated with relapse status: multivariate analyses (n=131)1

| Variable | Odds ratio2 | 95% CI |
|---|---|---|
| Age (years) | 1.07 | 1.01-1.13 |
| Number of years since most recent hospital admission | 0.79 | 0.69-0.90 |
| Previous suicide or self-harm attempts | 3.93 | 1.39-11.07 |
| Social relationships score (Lehman) | 1.29 | 1.13-1.48 |
| GAF score | 0.93 | 0.87-0.98 |
Cost correlates
The log link method of GLM estimation was used to examine the factors associated with cost differences (Table 7). Coefficient values represent the percentage change in total costs (from the average) following a one-unit change in the explanatory variable (compared with a reference category if the variable is categorical). Holding constant all other explanatory factors in the model, average costs were increased by patients who relapsed (147%), and were reduced by patients who were older (3.6% per year of age), and living with family/others compared with those in collective accommodation (58%).
Table 7 Factors associated with differences in costs: multivariate analyses (n=145)

| Variable | Coefficient (β)1 | 95% CI |
|---|---|---|
| Age (years) | -0.04 | -0.06 to -0.16 |
| Gender (male) | 0.08 | 0.32 to 0.48 |
| Ethnicity (White) | -0.11 | -0.64 to 0.43 |
| Ethnicity (Black Caribbean) | 0.99 | -0.15 to 2.12 |
| Marital status (single) | -0.16 | -0.70 to 0.38 |
| Marital status (married/cohabiting) | 0.35 | -0.33 to 1.03 |
| Further education (higher) | 0.26 | -0.44 to 0.94 |
| Living alone at home | -0.05 | -0.58 to 0.48 |
| Living with family/relatives | -0.58 | -1.07 to -0.08 |
| Relapse status | 1.47 | 1.88 to 1.06 |
| Constant | 9.15 | 8.07 to 10.14 |
DISCUSSION
Costs of relapse of schizophrenia
Studies of the overall costs of schizophrenia in the UK (Reference Davies and DrummondDavies & Drummond, 1993) and in other countries (Reference Knapp, Mangalore and SimonKnapp et al, 2004) confirm the high proportion of the total that is attributable to in-patient care. This study shows that illness relapse is a major factor in generating these high hospitalisation rates and costs. We have gone further, however, in providing an estimate of the full service costs of schizophrenia relapse in the UK. Patients who experienced a relapse during the 6 months prior to data collection had mean service costs of £8212 compared with £1899 for those who had no relapse during this period. The only previous UK estimate of the costs of relapse of which we are aware was based on expert opinion and assumed (rather than observed) service utilisation in a simulation model that compared three antipsychotic drugs (Reference Almond and O'DonnellAlmond & O’Donnell, 2000). Average relapse costs at 1997 prices were estimated to be just over £10 000 per patient during three monthly cycles and included both service use costs and accommodation costs (the latter not included here).
Clinical and QoL correlates
Surprisingly, perhaps, there were few differences in clinical and QoL outcomes between patients who had relapsed and those who had not. However, some of the patients in the former group would have recovered well from their relapse by the time these clinical and QoL instruments were administered. This time lapse is probably the reason for the lack of difference.
Associations
Multivariate analyses confirmed some significant correlates of relapse, and a reduced-form cost equation found, as expected, that relapse status significantly increased total costs. The cost equation was estimated in reduced form for two main reasons. First, relapse status as a regressor captured some of the important partial effects already identified in the relapse function – for example, suicide attempts, previous hospital admissions and social functioning – and reduced the need to include these variables further as independent effects in the cost analyses. Second, clinical and QoL variables were excluded from the cost equation because it was difficult to relate current measures with costs in the previous 6 months. This is a problem of endogeneity: it is difficult to ascertain the direction of causation between variables. Although higher levels of service use (and costs) might have improved health and reduced the likelihood of relapse, relapse status might have increased service use and costs. However, given that relapse often resulted in hospitalisation (for about two-thirds of the people in the relapse group) and in-patient costs accounted for around three-quarters of total costs, the problem of endogeneity with relapse status was less of an issue.
Finally, a cautionary note is required on measuring differences in costs and health outcomes between the relapse and non-relapse groups. Although this method is valid, a superior comparison would come from panel or longitudinal data that measure changes in outcomes prospectively for a given population (cf. Reference Robinson, Woerner and MaRobinson et al, 1999). The costs of relapse would then be estimated by examining the differences in costs, before, during and after relapse. Cost-effectiveness comparisons are also required based on experimental evaluations of relapse minimisation strategies.
Policy implications
The significant costs found to be associated with relapse confirm the scale of the impact – in this case measured by service uptake – of a worsening of symptoms for people with schizophrenia. These costs will be of interest to clinicians and other decision-makers who face difficult choices about new but more expensive treatments for patients with schizophrenia. Subject to the above cautionary comment, delaying the time to relapse should mean delaying the escalation of costs. More importantly, a slower or reduced rate of relapse means slower or reduced damage to the health and quality of life of patients, and in some cases also less adverse impact on their families.
Psychoeducation and related programmes have been shown to reduce medication non-adherence, detect prodromal symptoms of relapse and reduce the rate of hospitalisation (e.g. Reference Birchwood, Smith and MacmillanBirchwood et al, 1989; Reference Kemp, Hayward and ApplewhaiteKemp et al, 1996; Reference Herz, Lamberti and MintxHerz et al, 2000). A relatively inexpensive evidence-based intervention for reducing relapse is family work for patients with schizophrenia living with a relative with high levels of expressed emotion (e.g. Reference Xiong, Phillips and HuXiong et al, 1994). There is no evidence that these effective interventions have yet come into widespread use.
If new antipsychotic treatments in schizophrenia can improve efficacy and compliance rates compared with conventional neuroleptic therapy, and thereby reduce relapse rates, this might bring about reductions in the service costs of schizophrenia. In turn, as demonstrated in some international studies (Reference Hamilton, Revicki and EdgellHamilton et al, 1999), and as concluded by the National Institute for Clinical Excellence (2002), the overall costs of the treatment could be reduced.
Clinical Implications and Limitations
CLINICAL IMPLICATIONS
-
▪ Compared with the non-relapse group, patients who relapsed scored higher on the Positive and Negative Syndrome Scale and Clinical Global Impression scale, and lower on the Global Assessment of Functioning, but only the latter was statistically significant.
-
▪ The strongest predictor of illness relapse was associated with patients who had made previous suicide or self-harm attempts.
-
▪ Differences in the quality of life between relapse and non-relapse cases, as measured by the Lehman scale, were generally not significant.
LIMITATIONS
-
▪ The study was cross-sectional, and limited to one geographical area of the UK.
-
▪ Relapse was not studied as it occurred, but in a retrospective design.
-
▪ Multivariate analysis was applied to a small sample of patients and with only a few explanatory variables.
Acknowledgements
We would like to thank Lundbeck SA, Paris, France, for providing financial. support to Dr Almond and Professor Brugha for this study; the staff and. R&D Directorate of the Leicestershire Partnership National Health Service. Trust for their support throughout field work; and José Fernández of the London School of Economics for econometric advice.










eLetters
No eLetters have been published for this article.