Article contents
Synthesis of LiFePO4 powder by the organic–inorganic steric entrapment method
Published online by Cambridge University Press: 27 July 2015
Abstract
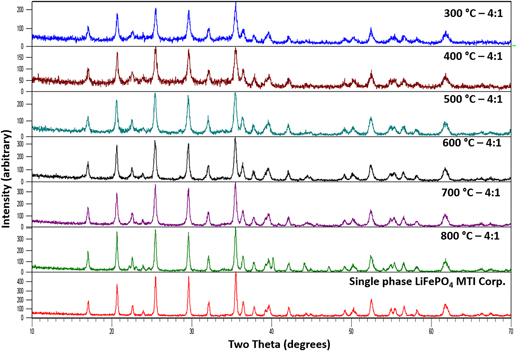
A nanoscale and pure, olivine-structured LiFePO4 was synthesized at ∼300 °C using an organic–inorganic steric entrapment method. Normally, when calcined and crystallized in air, this method leads to the synthesis of compounds where the cations are in their highest oxidation state. However, in this study, we found a way to produce compounds having lower oxidation states (e.g., compounds containing Fe2+), which may have wider applications in the synthesis of other compounds with complex chemistry that have variable oxidation states and, therefore, potential applications in electronic ceramics. The resulting LiFePO4 or (Li2O·2FeO·P2O5) was characterized by thermogravimetric analysis/differential thermal analysis, x-ray diffractometry, scanning electron microscopy, transmission electron microscopy, inductively coupled plasma emission spectroscopy, specific surface area by Brunauer–Emmett–Teller nitrogen absorption, and particle size analysis.
- Type
- Articles
- Information
- Copyright
- Copyright © Materials Research Society 2015
References
REFERENCES
- 9
- Cited by




