Meals with lactic acid-fermented vegetables and cereals have shown a significant increase in Fe absorption in human subjectsReference Derman, Bothwell, Torrance, Bezwoda, Macphail, Kew, Sayers, Disler and Charlton1–Reference Bering, Suchdev, Sjøltov, Berggren, Tetens and Bukhave4. This increase may be caused mainly by the small organic acids that are produced during the lactic acid fermentation, with lactic acid as the major product. Lactic acid may act by (a) lowering pH, which activates endogenous phytases present and consequently releases Fe from insoluble Fe–phytate complexesReference Sandberg and Andersson5, (b) increasing the buffer capacity in the pH-range 3–5, which keep Fe in a bioavailable formReference Hallberg and Rossander3, (c) formation of soluble Fe-ligand complexes at intestinal pHReference Bergqvist, Sandberg, Andlid and Wessling-Resnick6, and (d) delaying the gastric emptying rateReference Liljeberg and Bjorck7, which increases the time that dietary Fe is in contact with the duodenal surface.
Fe absorption is believed to occur mainly in the duodenum and to diminish from the proximal jejunum to the ileumReference Miret, Simpson and McKie8. However, the distal part of the intestine may also play an important role in the absorption of FeReference Bouglé, Vaghefi-Vaezzadeh, Roland, Bouvard, Arhan, Bureau, Neuville and Maubois9–Reference Johnston, Johnson, Marks, Srai, Debnam and Sharp11. Animal studies have shown a significant increase in Fe absorption in the proximal colon compared to the ileum on a surface basis. Due to the larger surface area of the human colon compared to the duodenum, and the longer retention time in the colon, the colon may add significantly to the absorption of FeReference Bouglé, Vaghefi-Vaezzadeh, Roland, Bouvard, Arhan, Bureau, Neuville and Maubois9. Although very little is known about the mechanisms of Fe absorption in the colon, animal studies indicate that the colonic mucosa is highly permeable for Fe both via active transport and passive diffusionReference Campos, Gómes-Ayala, López-Aliaga, Pallarés, Hartiti, Alférez, Barrionuevo, Rodríguez-Matas and Lisbona10. The luminal pH, the caecal surface area, and the concentration of organic acids in the colon are closely associated with the quantities of fermentable carbohydrates (prebiotics), as well as mineral concentrationsReference Demigne, Levrat and Remesy12; thus, microbial fermentation could play a major role in Fe absorption from the colonReference Yeung, Glahn, Welch and Miller13. Ingestion of prebiotics, in form of non-digestible oligosaccharides that stimulate fermentation by the intestinal flora, have been shown to increase Fe absorption in ratsReference Sakai, Ohta, Shiga, Takasaki, Tokunaga and Hara14, Reference Lopez, Levrat-Verny, Coudray, Besson, Krespine, Messager, Demigne and Remesy15, but this effect has so far not been confirmed in human subjectsReference Coudray, Bellanger, Castiglia-Delavaud, Remesy, Vermorel and Rayssignuier16, Reference van den Heuvel, Schaafsma, Muys and van Dokkum17. An increase in Fe absorption from probiotic foods has also been shown in ratsReference Bouglé, Vaghefi-Vaezzadeh, Roland, Bouvard, Arhan, Bureau, Neuville and Maubois9, Reference Oda, Kado-oka and Hashiba18, and the effect has been addressed both to the fermentation products and to the viable bacteriaReference Bouglé, Vaghefi-Vaezzadeh, Roland, Bouvard, Arhan, Bureau, Neuville and Maubois9. However, the rat is not an optimal model for human Fe absorption, since they are able to synthesize ascorbic acid and possess phytase activity in the intestinal lumenReference Wienk, Marx and Beynen19.
A probiotic non-dairy food product based on oatmeal fermented with the defined probiotic strain L. plantarum 299v (Probi AB, Lund, Sweden) has shown several of the health benefits attributed to probioticsReference Molin20. The strain originates from the human intestinal mucosa and has been shown to survive the low pH of the stomach, to tolerate the bile salts in the small intestine, and to colonize the intestinal mucosa, including the jejunum, after oral administrationReference Johansson, Molin, Jeppsson, Nobaek, Ahrne and Bengmark21.
The objectives of the present study were to examine the effect of a heat-inactivated fermented oat gruel with added viable, lyophilized L. plantarum 299v on non-haem Fe absorption from a meal with low Fe bioavailability compared to the heat-inactivated fermented oat gruel without added viable L. plantarum 299v. In addition, the study allowed for an investigation of the absorption of non-haem Fe in the ileum and colon separate from that occurring in the proximal small intestine. The study was designed as a randomized, double-blinded crossover trial with eighteen healthy young women.
Subjects and methods
Subjects
Eighteen women aged 22 (sd 3) years with a body weight of 64 (sd 7) kg and BMI of 21·9 (sd 1·9) kg/m2 participated in the study. All subjects were non-smokers and were not pregnant or lactating, and none of the women took any vitamin or mineral supplements for ≤ 2 months before and during the study. Fifteen subjects used oral contraceptives, but none of the subjects were routinely taking any other medication. Blood donation was not allowed for at least 2 months prior to and during the study. The participants were recruited from universities in the nearby area and received oral and written information about the study before they gave their written consent. The study was approved by the Municipal Ethical Committee of Copenhagen and Frederiksberg, Denmark (file no. KF 01-219/03), and the National Institute of Radiation Hygiene, Denmark, and was registered at the ClinicalTrials.gov database (NCT00283491).
Experimental design
The study was a randomized, double-blinded crossover trial, in which each subject was served two test meals: A, a heat-inactivated lactic acid-fermented oat gruel with added lyophilized viable L. plantarum 299v and B, a heat-inactivated lactic acid-fermented oat gruel.
The non-haem Fe absorption from the two test meals was determined by extrinsic labelling of the oat gruels with 59Fe, while absorption from the distal part of the intestine was elucidated by 55Fe in enterocoated capsules designed to disintegrate in the ileum. Each test meal was served within one of two periods separated by a wash-out period of 18 d to avoid any possible carry-over effect of the meal with the viable, colonizing L. plantarum 299v, and the test meals were served twice on two consecutive mornings to minimize day-to-day variation. The subjects were assigned randomly to each of two groups, which were given meal A and B in different orders.
The retention of 59Fe was measured in a whole-body counter at baseline and 15 d after intake of the test meals, and the activity of both isotopes, 55Fe and 59Fe, was measured in a blood sample drawn 18 d after intake of the test mealsReference Bukhave, Sørensen and Hansen22. Residual isotope activities from the first period were subtracted from the isotope activity levels in the second period and Fe excretion between the two measurements was assumed negligible.
The stability of the enterocoated capsules and therefore length of time before disintegration in the gastrointestinal tract was estimated from in vitro pepsin and pancreatin-bile digestions.
Composition of test meals and serving procedure
Probi AB (Lund, Sweden) supplied the oat gruels for the test meals. Whole grain oatmeal (Kungsörnen AB, Järna, Sweden) and water was mixed and a blend of different enzymes was added, followed by a specific heat treatment to reduce viscosity. The oat gruel was heated to 90°C for 1 h to inactivate the enzymes and to kill any contaminating microorganisms already present in the gruel, followed by inoculation with L. plantarum 299v (DSM 9843)Reference Molin, Albertsson, Bengmark and Larsson23. After fermentation, the oat gruel was pasteurized (100°C for 1 h, to kill the relatively heat resistant L. plantarum 299v) and stored frozen ( − 20°C) with a final DM of 15 g/100 g (oat gruel B). The fermented oat gruel with viable L. plantarum 299v (A) was produced by inoculation of oat gruel B at 4°C with viable lyophilized L. plantarum 299v (10Reference Bouglé, Vaghefi-Vaezzadeh, Roland, Bouvard, Arhan, Bureau, Neuville and Maubois9 cfu/g) 1 d before serving and stored at 4°C to minimize further fermentation and production of organic acids compared to the control gruel (B).
The enterocoated capsules were prepared the day before serving by filling hard gelatine capsules (0·50 ml, Wepa, Hillscheid, Germany) with 250 mg potato flour and adding either 28 kBq 55Fe(II)Cl2 and 25 μg ascorbic acid to avoid oxidation or 55Fe(III)Cl3 (Risø National Laboratory, Roskilde, Denmark) in 25 μl 0·1 m-HCl. The capsules were then coated with cellulose acetate phthalate by dipping each capsule half fixed to a disc eight times into the film solution (1·0 kg cellulose acetate phthalate, 0·05 kg virgin castor oil, and 8·95 kg acetone)Reference Jacobsen, Hojgaard, Hylander, Wielandt, Thale, Jarnum and Krag24.
For each test meal 100 g oat gruel (A or B) plus two enterocoated capsules with 55Fe(II) and 55Fe(III), respectively, were served with a 140 g whole-wheat roll (60·0 g wheat flour, 20·0 g whole-wheat flour, 2·0 g salt, 2·0 g yeast, 16·0 g rapeseed oil, 40·0 g ultra pure water) with 10 g butter and a glass of ultra pure water (200 ml). The whole-wheat rolls were prepared in one batch, stored at − 20°C and reheated in an oven at 200°C for 10 min before serving.
The test meals were served in the morning after 12 h fasting. Intake of a maximum of 0·5 litre water was allowed overnight. Moderate or hard physical activity or the intake of alcohol was not allowed during the 12 h before intake of the test meals. After consuming the test meals, the subjects were not allowed to eat or drink for 2 h and intake of alcohol was prohibited for 24 h. The subjects filled in a questionnaire in connection with each test meal to ensure that they adhered to all procedures, and they were instructed to eat and drink alternately and to rinse the glass containing the oat gruel thoroughly with the water to ensure complete intake of the isotope dose. A staff member ensured that the protocol was followed.
Isotopes and labelling procedure
The oat gruels were extrinsically labelled by adding 1 ml isotope solution (57 kBq 59FeCl3 in 0·1 m-HCl (Risø National Laboratory)] directly to the oat gruels 18 h before serving for isotope exchange.
Dietary analyses
The oat gruels and the bread were freeze-dried, homogenized, and analyzed in duplicates for total Fe, Ca, Zn, phytate, and polyphenols. The energy content was calculated using a national food-composition database (Danish Tables of Food Composition, DANKOST 2000, version 1.20, Herlev, Denmark). Total Fe, Ca, and Zn were determined by atomic absorption spectrophotometry (Spectra-AA 200, Varian, Mulgrave, Australia) after wet-ashing in a MES 1000 Solvent Extraction System (CEM Corp., Matthews, NC, USA) with 650 g suprapure nitric acid/l (Merck KgaA, Darmstadt, Germany). A typical diet (Standard Reference Material 1548a, National Institute of Standards and Technology, Gaithersburg, MD, USA) was used as the reference for Fe (35·3 (sd 3·77) μg/g), Ca (1·96 (sd 0·11) mg/g), and Zn (24·6 (sd 1·79) μg/g), and the analysed values were 34·7 μg/g, 1·83 mg/g, and 23·3 μg/g, respectively. Phytate was analyzed as individual inositol tri- to hexaphosphates by high-performance ion chromatographyReference Carlsson, Bergman, Skoglund, Hasselblad and Sandberg25. Polyphenols were analyzed as total polyphenols by the Folin–Ciocalteu method and as proanthocyanidines using the vanillin assay with sulphuric acidReference Scalbert, Hemingway and Laks26, and the results expressed as mg gallic acid and mg (+)catechin equivalents, respectively. The concentration of organic acids in the oat gruels was determined by capillary gas chromatographyReference Richardson, Calder, Stewart and Smith27.
Determination of Fe status
Blood samples were drawn from the cubital vein after the subjects had rested for 10 min in a supine position. Hb analysis was carried out on blood (3·5 ml) collected in tubes containing dissolved EDTA (Vacutainer system, Becton Dickinson, Franklin Lakes, NJ, USA) using a Sysmex KX-21 automated hematology analyzer (Sysmex America Inc., Mundelein, IL, USA) with appropriate controls (Eight check-3WP, 22490822, Sysmex America Inc.). Intra-assay and inter-assay variations were 0·5 % (n 12) and 0·6 % (n 27), respectively. Serum ferritin and α1-antichymotrypsin analyses were carried out on serum from blood (5·0 ml) collected in plain tubes (Vacutainer system, Becton Dickinson). Ferritin was determined by a chemiluminescent immunometric assay using an Immulite 1000 analyzer (Diagnostic Products Corporation, Los Angeles, CA, USA) with appropriate reference sera (3rd International standard for ferritin (80/578);WHO, NIBSC, South Mimms, UK). Intra-assay and inter-assay variations were 2·7 % (n 15) and 5·0 % (n 15), respectively. α1-Antichymotrypsin was determined by an immunoturbidimetric technique using a Cobas Mira analyzer (Roche Diagnostic Systems, F. Hoffman-La Roche Ltd., Basel, Switzerland) with appropriate reference sera (European Commission certified reference material 470, no. 11 924, IRMM, Geel, Belgium). Intra-assay and inter-assay variations were 1·4 % (n 12) and 3·2 % (n 14), respectively.
Determination of non-haem Fe absorption
Activities of 55Fe and 59Fe were determined as double determinations from blood samples (2 × 20 ml) collected in tubes containing heparin as anticoagulant (Vacutainer system, Becton Dickinson) by dry-ashing followed by recrystallization and solubilization before counting in a Tricarb 2100TR Liquid Scintillation Analyzer (Packard Instruments, Meriden, CT, USA) with automatic quench correction as described previouslyReference Bukhave, Sørensen and Hansen22. The whole-body retention of 59Fe was measured in a lead-lined steel chamber with 4 NE110 plastic scintillator blocks (Nuclear Enterprises Ltd, Edinburgh, UK) connected to conventional nuclear electronic modules and a multichannel analyzer system. The counting efficiency and energy window settings were established through measurements of water-filled phantoms whose outlines and weights were approximately equal to those of human subjects. The phantoms were filled with known concentrations of the isotope. In the actual setup and with the energy window used, the overall counting efficiency for 59Fe evenly distributed in a 77 kg phantom was approximately 20 %. To minimize the contamination by atmospheric background activity, all subjects had a shower, washed their hair, and were dressed in hospital clothing before each measurement. The counting time was 10 min, and the results were corrected for chamber background radiation and for the individual background radiation level of the subjects, which was determined 1 d before beginning of the first intervention period. All results from determination of radioactivity were corrected for the physical decay of the isotope. Fe absorption data are expressed as 59Fe whole-body retention measured directly from whole-body counting and as 55Fe whole-body retention determined from the relative activities of 55Fe and 59Fe in blood and the 59Fe whole-body retention using the following equation, which is based on the assumption that the fractions of 55Fe and 59Fe in blood are similarReference Hallberg and Cook28:
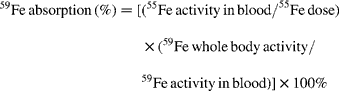
Stability of 55Fe(II) in enterocoated capsules
The stability of 55Fe(II) in the enterocoated capsules was determined by separation of the Fe according to oxidation states by paper chromatographyReference Stevens29. Capsules from the same batch as given in the intervention period were analyzed following 7 d. The 55Fe(II)-capsules were cut through with a scalpel under 8 ml water and the content was mixed with the water in the closed container. After 2 d aliquots were taken from the supernatants for determination of 55Fe(III) by paper chromatography. Briefly, Whatman filter paper (no. 1, Whatman Inc., Florham Park, NJ, USA) was washed with 1 m-HCl and dried followed by application of 20 μl sample. Upward chromatography was carried out with equal volumes of butanol, acetic acid, and ethanol (Merck) as eluant in a closed container and allowed to develop approximately 16 cm. After drying, the paper was scanned for 2 h on a TLC analyzer (miniGita scanner, Raytest Inc., Wilmington, NC, USA). The original solutions of 55Fe(II) and 55Fe(III) in 0·1 m-HCl (Risø National Laboratory) were used as references. The detection limit was approximately 160 dpm.
In vitro digestion of enterocoated capsules
Enterocoated capsules filled with either 55Fe(II) and 55Fe(III) as described or the water-soluble marker [14C]mannitol (19 kBq, 2·1 GBq/mmol, PerkinElmer Life and Analytical Sciences, Boston, MA, USA) were digested in vitro with the oat gruels by adding the capsule to a mixture of 50 ml oat gruel (A and B, respectively) and 50 ml pepsin solution (9·1 ml 5 mg porcine pepsin/ml, 1,100 units/mg protein, Sigma-Aldrich, St. Louis, MO, USA, in 50 mm-acetic acid buffer (Sigma-Aldrich), pH 4·5, and 40·9 ml, 50 mm-HCl (Riedel-de-Haën, Seelze, Germany)). The solutions were incubated on a shaking water bath (37°C) and aliquots (100 μl) were taken every 30 min for liquid scintillation counting. After 4 h the capsules were removed and the solutions were added to 0·5 ml 1 m-NaOH (J.T. Baker, Deventer, Holland), 4 ml 1·2 m-NaHCO3 (Merck), and pH was raised to 7·0 with 1 m-NaOH. Thereafter, 60 mg pancreatin (porcine, 3 × U.S.P. specifications, Sigma-Aldrich) and 185 mg porcine bile extract (Sigma-Aldrich) was added followed by the capsules, and the solutions were incubated on a shaking water bath (37°C) with drawing of aliquots (100 μl) every 30 min until the capsules disrupted. For capsules with 55Fe additional aliquots were drawn and centrifuged (8000 g, 10 min) to determine the degree of soluble 55Fe in the digest solution. The time course for the activity of [14C]mannitol and 55Fe in the digest solutions was determined by liquid scintillation counting with automatic quench correction with 5 ml InstaGel Plus (PerkinElmer Life and Analytical Sciences).
Statistical analyses
The sample size was calculated from Fe absorption data from a previous study of ours using similar test meals, i.e. 100 g fermented oat gruel and a 140 g whole-wheat roll with butterReference Bering, Suchdev, Sjøltov, Berggren, Tetens and Bukhave4. To obtain a power of 90 %, a calculated sixteen subjects were needed to detect a 2 % (1 × sd) change in Fe absorption at a significance level of 0·01Reference Altman30. To ensure enough power in case of dropouts a total of eighteen subjects were included in the study.
Non-haem Fe absorption data were converted to logarithms before the statistical analyses, and the results were reconverted to antilogarithms. All data used for statistical analyses were normally distributed, with variance homogeneity tested by plots of residuals. The non-haem Fe absorption from the two test meals was compared using a linear mixed model with log (non-haem Fe absorption) as the dependent variable, meal and serum ferritin as independent fixed variables, and subject as a random effect:
The carry-over effect from the meal with the viable bacteria in the first period to the meal in the second period was analyzed by inserting the alternate meal as a fixed variable in the model. Data are presented as estimates of least-squares means and differences between estimates of means with 95 % CI.
The statistical analyses were performed with the SAS statistical software package, version 9.1 (SAS Institute Inc., Cary, NC USA), and values were considered significantly different for P < 0·05.
Results
Composition of the test meals
The composition of the test meals as regards the content of minerals, inhibitors of Fe absorption, and organic acids in the oat gruels are given in Table 1, and the content of all analysed components were similar when comparing the two test meals.
Table 1 Composition of the test meals (including the whole-wheat roll with butter), and concentrations of organic acids and pH in the fermented oat gruels (Mean values and standard deviations for four meals)

* Represents individual inositol tri- to hexaphosphates.
† Measured as catechin equivalents.
‡ Measured as gallic acid equivalents.
Fe status and non-haem Fe absorption
All subjects had relatively low Fe stores, but were not anaemic as indicated by their serum ferritin concentrations in the range 13–29 μg/l, and Hb concentrations in the range 116–135 g/l. The concentrations of the acute phase protein α1-antichymotrypsin (0·17–0·47 g/l) were in the normal range, i.e. < 0·6 g/l and therefore, serum ferritin was a valid measurement of the Fe status in these subjectsReference Wieringa, Dijkhuizen, West, Northrop-Clewes and Muhilal31.
Determination of 55Fe(III) in the 55Fe(II)-capsules revealed that no significant amount of 55Fe(II) was oxidized to 55Fe(III) in the capsules, as the content of 55Fe(III) was below the detection limit, i.e. < 160 dpm corresponding to less than 4 % conversion of 55Fe(II) (n 4).
The overall non-haem Fe absorption from the fermented oat gruels with and without added viable, lyophilized L. plantarum 299v, respectively, calculated from the mixed linear model analysis is given in Table 2. The results show no significant difference in Fe absorption from the fermented oat gruel with or without added viable, lyophilized bacteria, and the ratio of Fe absorption from the two meals was not different from unity.
Table 2 Non-haem iron absorption from the fermented oat gruels with viable, lyophilized L. plantarum 299v and without L. plantarum 299v, respectively

* Geometric means of least squares estimates from the mixed linear model analysis with 95 % CI (n 18).
† Geometric means of estimates of differences from the mixed linear model analysis with 95 % CI (n 18).
The absorption of 55Fe delivered to the ileum and colon from the enterocoated capsules was below the detection limit, i.e. below 0·1 % of the administered dose.
In vitro digestion of enterocoated capsules
The enterocoated capsules were stable for >4 h when digesting with pepsin at pH 2 (37°C). The time course for disintegration of the enterocoated capsules during the pancreatin-bile digestion phase (pH 7, 37°C) following the 4 h digestion with pepsin showed that the capsules were resistant to the pancreatin-bile solution for about 90–120 min before disruption as shown by release of [14C]mannitol (Fig. 1). No difference in the disintegration rate was observed for capsules digested in media of oat gruels with viable bacteria (n 6) and without bacteria (n 6), respectively. The release of 55Fe(II) and 55Fe(III) from the capsules (n 4 each) was similar, why the data were pooled, and following 20 h, 84 (sem 3) %, (n 8) of the radioactivity was recovered, of which 63 (sem 3) % (n 8) was in the supernatant after centrifugation (8000 g, 10 min).

Fig. 1 Time course for disintegration of enterocoated capsules during digestion with pancreatin-bile (pH 7, 37°C) after 4 h pepsin digestion (pH 2, 37°C) in oat gruel medium with or without viable L. plantarum 299v, respectively (n 6, each). The disintegration was demonstrated by the release of [14C]mannitol from the capsules into the digest solution, and there was no difference between gruels with and without viable bacteria. The lines represent the disruption of the individual capsules.
Discussion
In the present study non-haem Fe absorption was determined from a heat-inactivated lactic acid-fermented oat gruel and from the same gruel where viable, lyophilized L. plantarum 299v (total 10Reference Johnston, Johnson, Marks, Srai, Debnam and Sharp11 cfu) was added. The fermented oat gruel with the viable L. plantarum 299v was prepared by an addition of viable, lyophilized L. plantarum 299v to the heat-inactivated lactic acid-fermented oat gruel, which was kept at 4°C until serving 1 d later, allowing for a minimal change in the content of organic acids, minerals, and inhibitors of Fe absorption between the two test gruels (Table 1). This is in contrast to an earlier study performed by our group, where the active fermented oat gruel with L. plantarum 299v was stored at 4°C between the two periods (25 d in total), leading to an increase in the concentration of lactic acid of 19 % during this periodReference Bering, Suchdev, Sjøltov, Berggren, Tetens and Bukhave4.
The non-haem Fe absorption in the present study was 1·4 and 1·3 % from the heat-inactivated fermented oat gruels with and without viable L. plantarum 299v, respectively (Table 2). The aforementioned study indicated an effect of the viable L. plantarum 299v on Fe absorption beyond that caused by the lactic acid produced during the fermentationReference Bering, Suchdev, Sjøltov, Berggren, Tetens and Bukhave4. However, we were not able to confirm this effect in the present study with viable, lyophilized bacteria.
To be able to ensure detection of even small effects of L. plantarum 299v on Fe absorption a very low Fe absorption ratio was chosen by constructing the meals with a high content of phytate from the dose–response curve for phytate in relation to Fe absorptionReference Brune, Rossander-Hultén, Hallberg, Gleerup and Sandberg32. Furthermore, no enhancers were added. This has been shown to be a relevant level of phytate, as we in the previous study were able to significantly detect small differences in Fe absorption from the same basic mealsReference Bering, Suchdev, Sjøltov, Berggren, Tetens and Bukhave4. Previous studies on Fe absorption from oat porridge meals with similar contents of phytate but also containing ascorbic acid as an enhancer have also shown relatively low Fe absorption values of 3·2–4·0 %, which would have reach levels comparable to this study if no inhibitors were addedReference Larsson, Rossander-Hulten, Sandstrom and Sandberg33, Reference Rossander-Hulten, Gleerup and Hallberg34.
From the putative effect of viable L. plantarum 299vReference Bering, Suchdev, Sjøltov, Berggren, Tetens and Bukhave4 and the hypothesis that the colon may function as a significant site of Fe absorption in response to stimulation by pre- and probiotics, especially during Fe depletionReference Bouglé, Vaghefi-Vaezzadeh, Roland, Bouvard, Arhan, Bureau, Neuville and Maubois9, Reference Yeung, Glahn, Welch and Miller13, Fe absorption was selectively determined from the distal part of the small intestine and the colon using enterocoated capsules containing 55Fe as a tracer for Fe absorption. To our knowledge this is the first study to measure Fe absorption from a meal specifically in the distal part of the human small intestine and colon since only one study describes the attempt to measure Fe absorption from the human colon by infusion of radiolabelled ferrous and ferric chloride solutions directly through a sigmoidoscope in non-anaemic subjectsReference Ohkawara, Bamba, Nakai, Kinka and Masuda35. The stability of the capsules was tested by in vitro digestion. The capsules were stable for more than 4 h under gastric conditions, and disintegration of the capsules under conditions mimicking the lumen in the small intestine did not occur until after 90–120 min (Fig. 1), corresponding roughly to the transit time for the luminal content from pylorus to ileum after a meal. This is in accordance with Jacobsen and co-workers, who determined the disintegration of enterocoated tablets (cellulose acetate phthalate) containing BaSO4 by fluoroscopy in patients who had undergone ileal resectionReference Jacobsen, Hojgaard, Hylander, Wielandt, Thale, Jarnum and Krag24. The tablets disintegrated in the most distal part of the remaining ileum or caecum and in only three of fourteen patients did disintegration not occur within the 8 h after ingestion followed by fluoroscopy. Furthermore, as 84 % 55Fe was recovered from the disintegrated capsules after 20 h of which 63 % was in the supernatant after centrifugation, these data clearly indicate that Fe released from the capsules will mix with Fe in the luminal content and be potentially available for absorption. Together, these observations strongly indicate that the enterocoated capsules with 55Fe disintegrated and released their content of 55Fe in the ileum, and 55Fe thereby acted as a tracer for Fe absorption in the ileum and colon.
Very little is known about the mechanisms of Fe absorption in the large intestine, but there seems to be a preference for ferrous FeReference Ohkawara, Bamba, Nakai, Kinka and Masuda35 as in the proximal small intestine, and as the expression of the apical ferric reductase, duodenal cytochrome B (DcytB), involved in reduction of ferric to ferrous Fe in the duodenum is significantly reduced with a downward gradient along the length of the gutReference Miret, Simpson and McKie8, the uptake is dependent on other factors, such as lowering of luminal pH by fermentationReference Yeung, Glahn, Welch and Miller13. It is however uncertain whether both oxidation states of Fe are absorbable in the colon. Therefore, to ensure that if any Fe absorption was to occur distally it would not remain undetected due to a wrong oxidation state of the Fe used as tracer, the Fe in the enterocoated capsules were given as both ferrous and ferric Fe. However, no absorption of 55Fe was observed from either of the test meals, indicating that there was no absorption of Fe in the distal intestine.
An explanation for the lack of effect of the viable L. plantarum 299v in the present study could be that the bacteria were not in a comparable active state, as they were added in a lyophilized condition to a cold product to avoid any changes in composition of the test gruel. This is in sharp contrast to the earlier studyReference Bering, Suchdev, Sjøltov, Berggren, Tetens and Bukhave4 where the lactobacillus was metabolically active during the fermentation process and not heat-inactivated but stored cold until serving.
A subsequent analysis of the activity of the bacteria in an oat gruel fermented with L. plantarum 299v similar to the one used in the aforementioned studyReference Bering, Suchdev, Sjøltov, Berggren, Tetens and Bukhave4 has been undertaken and compared with the activity of the bacteria in the present heat-inactivated oat gruel with added lyophilized L. plantarum 299v. The result showed that the metabolic activity at 37°C (lowering of pH in an oat based medium) was retarded by approximately 1 h from lactic acid bacteria that were added to the oat gruel in a lyophilized form (data not shown). Since no Fe absorption seems to occur in the ileum and colon, it could be hypothesized that the bacteria needs to be in an active stage in the most proximal small intestine to exert a direct effect on Fe absorption beyond the one caused by the organic acids present in the oat gruel. The activity test indicates that the lactobacillus added in a lyophilized form less quickly reaches an active stage, which potentially could affect the metabolic activity of the bacteria in the duodenum and thereby explain the lack of effect of L. plantarum 299v in this study. Furthermore this would support the conclusion in the aforementioned study4 that a specific effect of viable L. plantarum 299v contributed to the increased Fe absorption. The effect of the gastric residence has though not been taken into account, and the lag phase for this adaptation is not known for either of the formulations tested.
Although the subjects had low Fe stores, i.e. serum ferritin < 30 μg/l, the Fe absorption from the fermented oat gruels was very low, 1·3–1·4 %, corresponding to 0·02–0·03 mg Fe in total amounts. This very low absorption was presumably due to the presence of high amounts of phytate and phenolic compounds in the meals (Table 1), and the lack of enhancers such as ascorbic acid, which was deliberately avoided in this study, as we wished a low basal absorption to ensure detection of even small effects of viable L. plantarum 299v on Fe absorption. Previous studies on Fe absorption from oat porridge meals with similar contents of phytate have also shown relatively low Fe absorption values of 3·2–4·0 %Reference Larsson, Rossander-Hulten, Sandstrom and Sandberg33, Reference Rossander-Hulten, Gleerup and Hallberg34. In both studies, the Fe absorption was about double that found in the present study, most likely due to the serving of orange juice, corresponding to 70 mg ascorbic acid, with these meals.
In conclusion, the addition of viable, lyophilized L. plantarum 299v to a heat-inactivated lactic acid-fermentedoat gruel did not affect non-haem Fe absorption from a low Fe bioavailability meal, and no absorption of Fe occurred in the ileum and the colon, at least in young women with low Fe status.
Acknowledgements
We thank Inge Rasmussen, Hanne Lysdal Petersen, and Leif Jakobsen, Department of Human Nutrition, University of Copenhagen, Frederiksberg, Denmark, and Susanne Svalling, Department of Clinical Physiology and Nuclear Medicine, The National University Hospital, Copenhagen, Denmark, for providing excellent technical assistance. This work was funded by The Ministry of Science, Technology, and Innovation, Denmark, and Vinnova, Sweden.







