4 results
Transition to Psychosis in Individuals at Clinical High Risk: Meta-analysis
- G. Salazar De Pablo, J. Radua, I. Bonoldi, V. Arienty, F. Besana, A. Cabras, A. Catalan, P. Fusar-Poli
-
- Journal:
- European Psychiatry / Volume 66 / Issue S1 / March 2023
- Published online by Cambridge University Press:
- 19 July 2023, p. S367
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Introduction
Estimating the current likelihood of transitioning from a clinical high risk for psychosis (CHR-P) to psychosis holds paramount importance for preventive care and applied research.
ObjectivesOur aim was to quantitatively examine the consistency and magnitude of transition risk to psychosis in individuals at CHR-P.
MethodsThis meta-analysis is compliant with Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) and Meta-analysis of Observational Studies in Epidemiology (MOOSE) reporting guidelines. PubMed and Web of Science databases were searched for longitudinal studies reporting transition risks in individuals at CHR-P.
Primary effect size was cumulative risk of transition to psychosis at 0.5, 1, 1.5, 2, 2.5, 3, 4, and more than 4 years’ follow-up, estimated using the numbers of individuals at CHR-P transitioning to psychosis at each time point. Random-effects meta-analysis were conducted.
ResultsA total of 130 studies and 9222 individuals at CHR-P were included. The mean (SD) age was 20.3 (4.4) years, and 5100 individuals (55.3%) were male.
The cumulative transition risk was 9% (95% CI = 7-10% k = 37; n = 6485) at 0.5 years, 15% (95% CI = 13-16%; k = 53; n = 7907) at 1 year, 20% (95% CI = 17%-22%; k = 30; n = 5488) at 1.5 years, 19% (95% CI = 17-22%; k = 44; n = 7351) at 2 years, 25% (95% CI, 21-29%) at 2.5 years, 25% (95% CI = 22-29%; k = 29; n = 4029) at 3 years, 27% (95% CI = 23-30%; k = 16; n = 2926) at 4 years, and 28% (95% CI = 20-37%; k = 14; n = 2301) at more than 4 years.
Meta-regressions showed that a lower proportion of female individuals (β = -0.02; 95% CI, -0.04 to -0.01) and a higher proportion of brief limited intermittent psychotic symptoms (β = 0.02; 95% CI, 0.01-0.03) were associated with an increase in transition risk. Other predictors were not statistically significant (p > 0.05).
Heterogeneity across the studies was high (I2 range, 77.91% to 95.73%).
ConclusionsIn this meta-analysis, 25% of individuals at CHR-P developed psychosis within 3 years. Transition risk continued increasing in the long term. Extended clinical monitoring and preventive care may be beneficial in this patient population.
Disclosure of InterestNone Declared
Lurasidone augmentation to clozapine in treatment resistant schizophrenia: A pilot study
- V. Arienti, S. C. Civardi, F. Besana, F. Mazzoni, G. Carnevale Miacca, N. Brondino, P. Politi, M. Olivola
-
- Journal:
- European Psychiatry / Volume 66 / Issue S1 / March 2023
- Published online by Cambridge University Press:
- 19 July 2023, pp. S306-S307
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Introduction
Treatment resistant schizophrenia still represents a major clinical and pharmacological challenge.30% of patients diagnosed with schizophrenia is characterised by a poor response to at least two different antipsychotics administered for a proper period of time and at adequate doses. Clozapine still represents the gold standard for treatment resistant patients. Unfortunately, a significant percentage of these are only partial responders. Augmentation strategies must be set up and atypical antipsychotic drugs are used in clinical practice. Promising findings have been observed in patients treated with Lurasidone as an add-on therapy with Clozapine. This novel second-generation antipsychotic has a unique receptor profile, showing 5-HT1a partial agonism and 5HT7 antagonism. These properties could also explain its procognitive effect, as several preclinical studies in literature have demonstrated.
ObjectivesThe aim of our study is to highlight the advantages of add on therapy with Lurasidone compared with treatment as usual (i.e. Clozapine + another atypical antipsychotic) in treatment resistant schizophrenia patients.
MethodsWe conducted an observational study in a sample of 20 patients diagnosed with treatment resistant schizophrenia, based on DSM-5 diagnostic criteria and psychopharmacologic history. Treatment choices were taken independently by clinicians in charge of each patient. 10 subjects underwent Lurasidone augmentation of Clozapine, whereas the remaining 10 subjects were treated as usual with Clozapine and another atypical antipsychotic. PANSS and BPRS scales to assess general psychopathology and UKU side effects scale were administered both at baseline and at follow-up (T1= 1 month; T2=6 months).
ResultsAll patients treated with Lurasidone augmentation strategy achieved a significant reduction of both positive and negative symptoms, with no significant adverse effects to be reported. In particular, Lurasidone showed no impact on metabolic parameters nor on ECG features, namely the QTc interval. The psychopathological improvement appeared higher in patients who received Lurasidone than in those treated as usual. This was particularly evident in cognitive domains.
ConclusionsOur observation suggests that augmentation strategy with Lurasidone to Clozapine can lead to clinically significant improvements in psychopathology when compared to Clozapine combined with another atypical antipsychotic, with a good tolerability profile. In future we will increase the number of our sample and the duration of follow-up time. In order to have more relevant statistical results, further research on this topic is needed.
Disclosure of InterestNone Declared
Efficacy and tolerability of brexpiprazole in patients with psychotic and mood disorders: a pilot study
- S.C. Civardi, N. Bassetti, V. Arienti, F. Besana, P. Politi, N. Brondino, M. Olivola
-
- Journal:
- European Psychiatry / Volume 65 / Issue S1 / June 2022
- Published online by Cambridge University Press:
- 01 September 2022, pp. S346-S347
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Introduction
Brexpiprazole is a novel antipsychotic drug. It exerts antagonistic activity at the serotonin 5HT2A, 5HT2B, 5HT7 and noradrenaline alpha 1b/2c receptors; it also acts as partial agonist of serotonin 5HT1 and dopamine D2, D3 receptors. Brexpiprazole is approved for the treatment of schizophrenia and as an add-on therapy for major depression.
ObjectivesThis pilot study aims at exploring efficacy and tolerability of Brexpiprazole in a small sample of patients diagnosed with either a psychotic or a mood disorder.
MethodsThis observational study was conducted at our Acute Psychiatric Inpatient Unit. We included 7 patients (5 males, 2 females) hospitalized between 2020 and 2021, diagnosed with schizophrenia spectrum disorders or mood disorders with psychotic symptoms confirmed by Mini International Neuropsychiatric Interview. Patients who participated signed an informed consent. Information concerning diagnosis, demographic characteristics (age, sex, education, marital status) and pharmacological therapy were collected examining clinical records. The average lenght of hospitalization was 13.4 days. Psychopathology was assessed by means of the PANSS and the severity of the illness was evaluated with CGI severity scale (CGI-S), both on admission and discharge. We also administered the UKU scale to evaluate the tolerability profile.
Results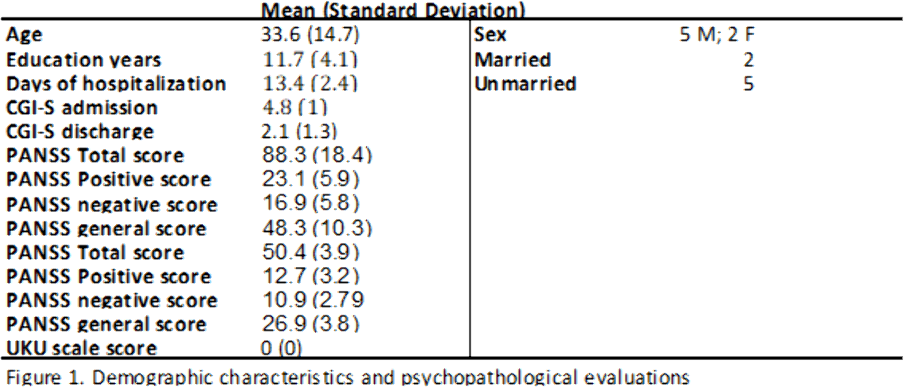

.
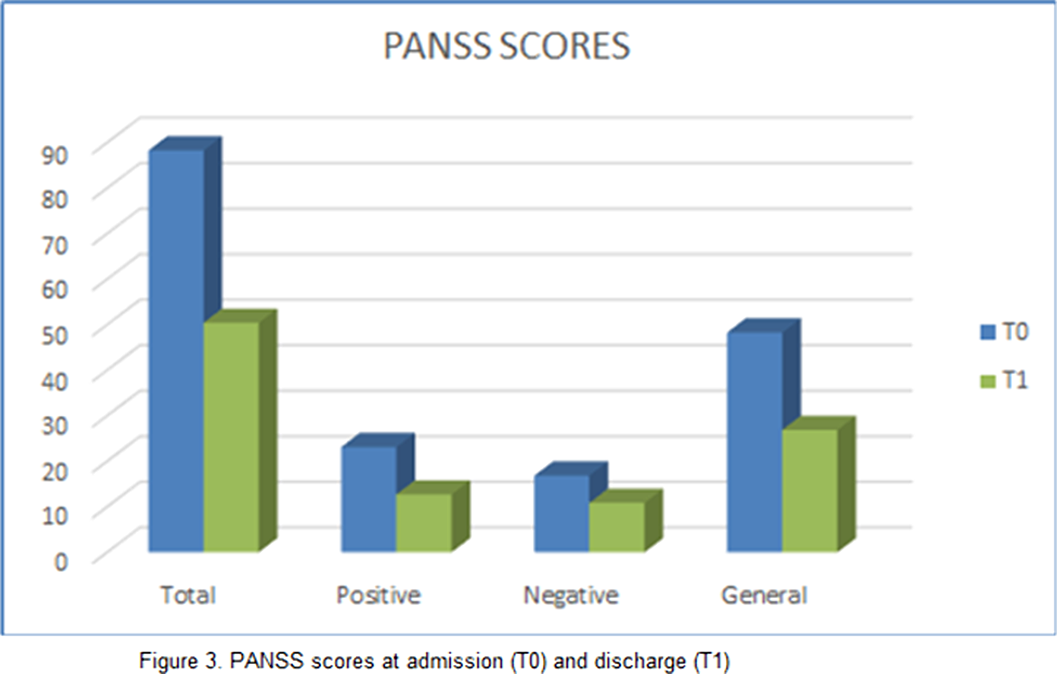
Results can be seen in figures 1, 2, 3
ConclusionsOur study found a significant improvement in both positive and negative symptoms, with good tolerability. Limitations of our study are: small sample size and limited period of observation. These premises suggest that further research is needed in order to elucidate the exact mechanisms underlying Brexpiprazole’s action and the possible implication in mood disorders.
DisclosureNo significant relationships.
Clinical outcomes in individuals at clinical high risk of psychosis who do not transition to psychosis: a meta-analysis
- Gonzalo Salazar de Pablo, Livia Soardo, Anna Cabras, Joana Pereira, Simi Kaur, Filippo Besana, Vincenzo Arienti, Francesco Coronelli, Jae Il Shin, Marco Solmi, Natalia Petros, Andre F. Carvalho, Philip McGuire, Paolo Fusar-Poli
-
- Journal:
- Epidemiology and Psychiatric Sciences / Volume 31 / 2022
- Published online by Cambridge University Press:
- 19 January 2022, e9
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
-
Aims
The clinical outcomes of individuals at clinical high risk of psychosis (CHR-P) who do not transition to psychosis are heterogeneous and inconsistently reported. We aimed to comprehensively evaluate longitudinally a wide range of outcomes in CHR-P individuals not developing psychosis.
Methods“Preferred Reporting Items for Systematic reviews and Meta-Analyses” and “Meta-analysis Of Observational Studies in Epidemiology”-compliant meta-analysis (PROSPERO: CRD42021229212) searching original CHR-P longitudinal studies in PubMed and Web of Science databases up to 01/11/2021. As primary analysis, we evaluated the following outcomes within CHR-P non-transitioning individuals: (a) change in the severity of attenuated psychotic symptoms (Hedge's g); (b) change in the severity of negative psychotic symptoms (Hedge's g); (c) change in the severity of depressive symptoms (Hedge's g); (d) change in the level of functioning (Hedge's g); (e) frequency of remission (at follow-up). As a secondary analysis, we compared these outcomes in those CHR-P individuals who did not transition vs. those who did transition to psychosis at follow-up. We conducted random-effects model meta-analyses, sensitivity analyses, heterogeneity analyses, meta-regressions and publication bias assessment. The risk of bias was assessed using a modified version of the Newcastle-Ottawa Scale (NOS).
ResultsTwenty-eight studies were included (2756 CHR-P individuals, mean age = 20.4, 45.5% females). The mean duration of follow-up of the included studies was of 30.7 months. Primary analysis: attenuated psychotic symptoms [Hedges’ g = 1.410, 95% confidence interval (CI) 1.002–1.818]; negative psychotic symptoms (Hedges’ g = 0.683, 95% CI 0.371–0.995); depressive symptoms (Hedges’ g = 0.844, 95% CI 0.371–1.317); and functioning (Hedges’ g = 0.776, 95% CI 0.463–1.089) improved in CHR-P non-transitioning individuals; 48.7% remitted at follow-up (95% CI 39.3–58.2%). Secondary analysis: attenuated psychotic symptoms (Hedges’ g = 0.706, 95% CI 0.091–1.322) and functioning (Hedges’ g = 0.623, 95% CI 0.375–0.871) improved in CHR-P individuals not-transitioning compared to those transitioning to psychosis, but there were no differences in negative or depressive symptoms or frequency of remission (p > 0.05). Older age was associated with higher improvements of attenuated psychotic symptoms (β = 0.225, p = 0.012); publication years were associated with a higher improvement of functioning (β = −0.124, p = 0.0026); a lower proportion of Brief Limited Intermittent Psychotic Symptoms was associated with higher frequencies of remission (β = −0.054, p = 0.0085). There was no metaregression impact for study continent, the psychometric instrument used, the quality of the study or proportion of females. The NOS scores were 4.4 ± 0.9, ranging from 3 to 6, revealing the moderate quality of the included studies.
ConclusionsClinical outcomes improve in CHR-P individuals not transitioning to psychosis but only less than half remit over time. Sustained clinical attention should be provided in the longer term to monitor these outcomes.






