2 results
Efficacy of two intensive and spaced protocols of theta burst transcranial magnetic stimulation in treatment-resistant depression: a double-blind randomized trial
- Y. Cañada, P. Navalón, P. Benavent, A. Sabater, J. Ribes, L. Livianos, P. Sierra
-
- Journal:
- European Psychiatry / Volume 66 / Issue S1 / March 2023
- Published online by Cambridge University Press:
- 19 July 2023, pp. S608-S609
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Introduction
Depression is the disease with the greatest burden of disability. Despite pharmacological options, up to 30% of depressions are considered resistant to treatment (RTD). Theta burst transcranial magnetic stimulation (TBS) on the dorsolateral prefrontal cortex allows the application of shorter protocols and with longer-lasting effects than conventional TMS. Its implementation in the Public Health System requires the design of efficient, cost-effective and accessible protocols for patients.
ObjectivesThe objective of this project is to assess the efficacy and safety of two intensive and spaced protocols (unilateral and bilateral) of 1800 pulses TBS compared to sham stimulation in outpatients with unipolar and bipolar TRD in a public hospital.
MethodsThis project is the 1st double-blind placebo-controlled RCT with TBS in Spain. It is now in the recruitment phase. Patients receive a total of 22 sessions of 1800 pulses in 6 weeks: 5 days/week the 1st and 2nd weeks (10 sessions) and 3 sessions/week the following weeks (12 sessions). Patients are randomized into three groups: i) bilateral, ii) left unilateral, and iii) sham. The main variable is the change in the HDRS-17 score at the end of treatment compared to baseline. The results were analyzed with a general linear model of HDRS, using time as the intrasubject factor and randomization group as the intersubject factor, using resistance to treatment (Maudsley Score) and diagnosis (bipolar, unipolar) as covariates.
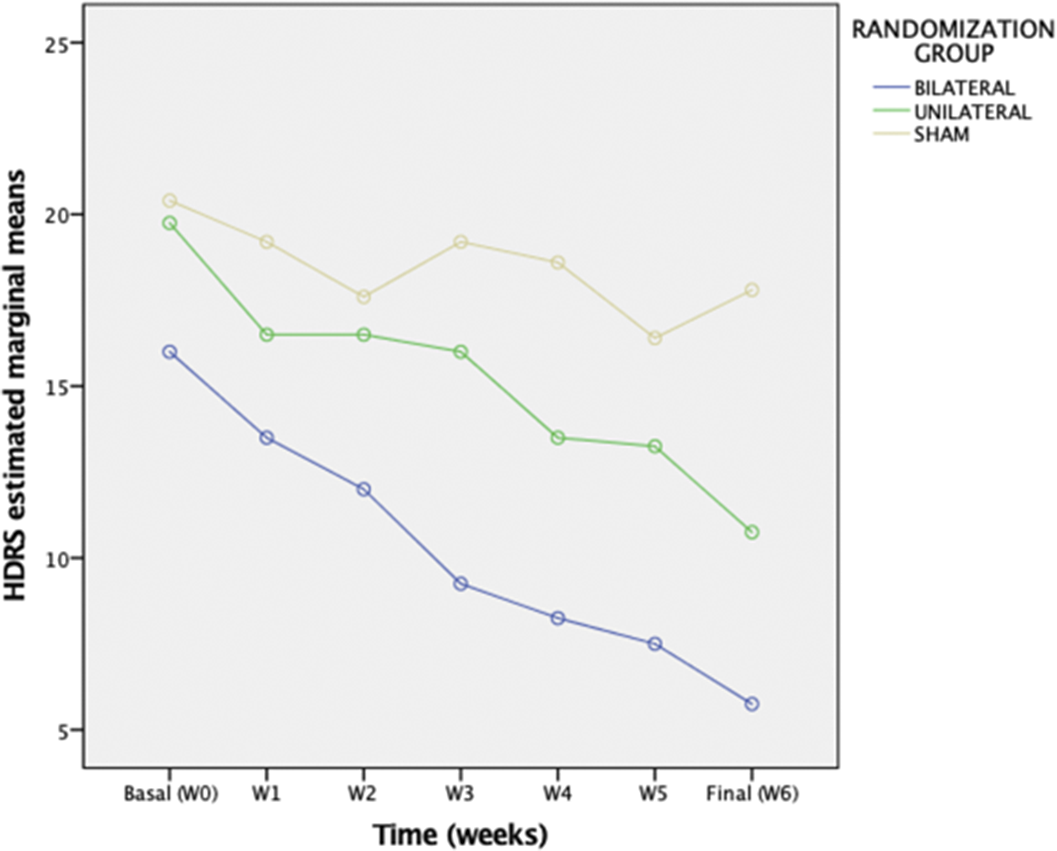
ResultsPreliminary results from 13 participants (nbil=4, nuni=4, nsham=5) reveal a significant effect of (group x time) on HDRS change (p= 0.020) with no influence of Maudsley Score or diagnosis. The bilateral group presented a greater decrease in the HDRS with a mean difference of 4,38 points [CI95% 0,17-8,58), (p=0.043)] with respect to the unilateral group and a difference of 8.23 [CI95% 4,24-12,21)(p =0.001)] compared to the sham group.
Table 1: Sample description. Data shown are means and standar deviations. In bold significant diferences p<0,05. Bilateral TBS (n=4) Unilateral TBS (n=4) Sham TBS (n=5) Age [M (SD)] 55,50 (5,80) 48,50 (16,86) 56,40 (5,86) Male: Female [n] 0:4 2:2 3:2 Depression:Bipolar disorder [n] 3:1 3:1 4:1 Length of depresive episode (months) [M (SD)] 12,00 (4,08) 16,50 (6,61) 15,00 (5,83) Current number of antidepressants [M (SD)] 2,00 (0,82) 1,75 (0,50) 2,60 (0,55) Maudsley score [M (SD)] 7,00 (1,82) 8,75 (1,5) 7,90 (1,30) Basal HDRS basal [M (SD)] 16,00 (1,82) 19,75 (4,03) 20,40 (3,21) Final HDRS [M (SD)] 5,75 (3,30) 10,75 (3,10) 17,80 (2,49) Response /Remission[n (%)] 3:3 (75%) 2:0(50:0%) 0:0 (0%) Image:
 Conclusions
ConclusionsThe results demonstrate the preliminary efficacy of intensive TBS protocols relative to sham.
Disclosure of InterestNone Declared
Efficacy and clinical and neuroimaging biomarkers of response of two intensive and spaced treatment protocols of theta burst magnetic stimulation in treatment resistant depression
- Y. Cañada, P. Benavent, A. Sabater, P. Navalón, M. Beser, L. Martí-Bonmatí, P. Sierra
-
- Journal:
- European Psychiatry / Volume 66 / Issue S1 / March 2023
- Published online by Cambridge University Press:
- 19 July 2023, p. S608
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Introduction
Resistant to treatment depression (RTD) is a prevalent disease that implies functional impairment and high resources consumption. Theta Burst Transcranial Magnetic Stimulation (TBS) in dorsolateral prefrontal cortex (DLPFC) is a novel therapy that has shown experimental efficacy and as an adjuvant strategy in RTD. The implementation of TBS in the Public National Health Service requires cost-effective protocols that achieve earlier responses and higher rates of effectiveness, and whose design is based on biomarkers of response so as to adequately select candidate patients.
ObjectivesTo assess the efficacy and safety of novel bilateral and unilateral intensive/spaced protocols of TBS in outpatients with unipolar/bipolar RTD compared with sham stimulation. Specific objectives: I) Comparison of mood change at the end of TBS protocol in the groups and maintenance of its effect at 3 months; II) Characterization of cerebral connectivity and metabolism patterns related to the effects of TBS; III) Analysis of the interaction between clinical and neuroimaging predictors so as to determine a RTD profile of patient that can benefit from TBS.
MethodsA two-year randomized double-blind clinical trial with 96 outpatients with TRD will be carried out. Participants will be randomized in three groups (active bilateral, active left and sham right and sham bilateral) to receive 22 active/sham sessions of continuous TBS (right DLPFC) and intermittent TBS (left DLPFC) during 6 weeks (w 1-2: 5 sessions/w, w 3-6: 3 sessions/w). Assessments of mood and side-effects will be carried out weekly. Functional neuroimaging will be a a simultaneous PET/MR acquisition previous and at the end of TBS treatment. Between-group comparisons of efficacy in terms of Hamilton Depression Rating Scale (HDRS-17) from basal to 6th week will be performed using controlled mixed regression models. Between-group comparisons will be made at baseline and after treatment, studying the imaging biomarkers obtained. Clinical and neuroimaging predictors of response will be integrated in machine learning models.
ResultsThe expected results of the project are summarized in the following hypotheses: 1) The intensive and spaced protocols of TBS as an adjuvant antidepressant treatment will have greater efficacy than sham stimulation in patients with TRD. 2) Both protocols will be safe, with mild side effects. 3) Unilateral and bilateral TBS protocols will involve changes in connectivity and cerebral metabolic consumption mainly in regions of the fronto-cingulo-temporal circuitry. 4) PET/MR imaging biomarkers will allow us to differentiate whether patients have responded to treatment with TBS.
ConclusionsThis project may help to improve resistant to treatment depression management by personalizing TMS treatment with the use of neuroimaging biomarkers.
Disclosure of InterestNone Declared




