3 results
865 – Effects Of Repeated Transcranial Direct Current Stimulation (tdcs) On Addiction-related Behaviors In Mice: a Preliminary Study
- S. Pedron, J. Monnin, P. Andrieu, M. Nicolier, L. Jacquot, J.-L. Millot, D. Sechter, P.-Y. Risold, E. Haffen, V. Van Waes
-
- Journal:
- European Psychiatry / Volume 28 / Issue S1 / 2013
- Published online by Cambridge University Press:
- 15 April 2020, 28-E323
-
- Article
-
- You have access Access
- Export citation
-
This study was part of a wider translational project on the efficacy of tDCS in the treatment of major depression and in attempts to facilitate alcohol and smoking cessation. tDCS is a non- invasive, painless and safe brain stimulation procedure capable of modulating cortical excitability. Preliminary clinical studies have indicated that tDCS applied over the dorsal prefrontal cortex of chronically dependent smokers and alcohol users reduces their desire for smoking and drinking, respectively. The neurobiological mechanisms underlying these effects remain unknown. We have developed a model of tDCS in mice that mimics the experimental paradigms used in our clinical research. Anodal tDCS is applied transcranially over the frontal cortex (2 x 20 min/day current for 5 consecutive days, intensity = 0.2 mA). The control group undergoes the same procedure but no current is applied. We evaluated the effects of tDCS on nicotine and alcohol consumption as well as on the rewarding effects of these two drugs of abuse. We assessed the impact of tDCS on stress-related, anxiety-related and depression-related behaviors. We tested the effects of tDCS on mice that had been chronically exposed to nicotine during adolescence. Chronic exposure to nicotine induced depression-like behavior in adult mice. It is thought that this depression- like state may be a symptom of withdrawal that facilitates relapse after abstinence. Our data indicate that tDCS reversed behavioral anomalies. In conclusion, our animal model has potential value as a means for exploring the neurobiological changes that underlie the beneficial effects of tDCS on addiction-related behaviors.
Manganoblödite, Na2Mn(SO4)2·4H2O, and cobaltoblödite, Na2Co(SO4)2·4H2O: two new members of the blödite group from the Blue Lizard mine, San Juan County, Utah, USA
- A. V. Kasatkin, F. Nestola, J. Plášil, J. Marty, D. I. Belakovskiy, A. A. Agakhanov, S. J. Mills, D. Pedron, A. Lanza, M. Favaro, S. Bianchin, I. S. Lykova, V. Goliáš, W. D. Birch
-
- Journal:
- Mineralogical Magazine / Volume 77 / Issue 3 / April 2013
- Published online by Cambridge University Press:
- 05 July 2018, pp. 367-383
-
- Article
- Export citation
-
Two new minerals – manganoblödite (IMA2012–029), ideally Na2Mn(SO4)2·4H2O, and cobaltoblödite (IMA2012–059), ideally Na2Co(SO4)2·4H2O, the Mn-dominant and Co-dominant analogues of blödite, respectively, were found at the Blue Lizard mine, San Juan County, Utah, USA. They are closely associated with blödite (Mn-Co-Ni-bearing), chalcanthite, gypsum, sideronatrite, johannite, quartz and feldspar. Both new minerals occur as aggregates of anhedral grains up to 60 μm (manganoblödite) and 200 μm (cobaltoblödite) forming thin crusts covering areas up to 2 × 2 cm on the surface of other sulfates. Both new species often occur as intimate intergrowths with each other and also with Mn-Co-Ni-bearing blödite. Manganoblödite and cobaltoblödite are transparent, colourless in single grains and reddish-pink in aggregates and crusts, with a white streak and vitreous lustre. Their Mohs' hardness is ∼2½. They are brittle, have uneven fracture and no obvious parting or cleavage. The measured and calculated densities are Dmeas = 2.25(2) g cm−3 and Dcalc = 2.338 g cm−3 for manganoblödite and Dmeas = 2.29(2) g cm−3 and Dcalc = 2.347 g cm−3 for cobaltoblödite. Optically both species are biaxial negative. The mean refractive indices are α = 1.493(2), β = 1.498(2) and γ = 1.501(2) for manganoblödite and α = 1.498(2), β = 1.503(2) and γ = 1.505(2) for cobaltoblödite. The chemical composition of manganoblödite (wt.%, electron-microprobe data) is: Na2O 16.94, MgO 3.29, MnO 8.80, CoO 2.96, NiO 1.34, SO3 45.39, H2O (calc.) 20.14, total 98.86. The empirical formula, calculated on the basis of 12 O a.p.f.u., is: Na1.96(Mn0.44Mg0.29Co0.14Ni0.06)Σ0.93S2.03O8·4H2O. The chemical composition of cobaltoblödite (wt.%, electron-microprobe data) is: Na2O 17.00, MgO 3.42, MnO 3.38, CoO 7.52, NiO 2.53, SO3 45.41, H2O (calc.) 20.20, total 99.46. The empirical formula, calculated on the basis of 12 O a.p.f.u., is: Na1.96(Co0.36Mg0.30Mn0.17Ni0.12)Σ 0.95S2.02O8·4H2O. Both minerals are monoclinic, space group P21/a, with a = 11.137(2), b = 8.279(1), c = 5.5381(9) Å, β = 100.42(1)°, V = 502.20(14) Å3 and Z = 2 (manganoblödite); and a = 11.147(1), b = 8.268(1), C = 5.5396(7) Å, β = 100.517(11)°, V = 501.97(10) Å3 and Z = 2 (cobaltoblödite). The strongest diffractions from X-ray powder pattern [listed as (d,Å(I)(hkl)] are for manganoblödite: 4.556(70)(210, 011); 4.266(45)(
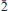 01); 3.791(26)(
01); 3.791(26)(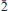 11); 3.338(21)(310); 3.291(100)(220, 021), 3.256(67)(211,
11); 3.338(21)(310); 3.291(100)(220, 021), 3.256(67)(211, 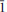 21), 2.968(22)(
21), 2.968(22)(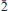 21), 2.647(24)(
21), 2.647(24)(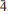 01); for cobaltoblödite: 4.551(80)(210, 011); 4.269(50)(
01); for cobaltoblödite: 4.551(80)(210, 011); 4.269(50)(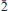 01); 3.795(18)(
01); 3.795(18)(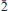 11); 3.339(43)(310); 3.29(100)(220, 021), 3.258(58)(
11); 3.339(43)(310); 3.29(100)(220, 021), 3.258(58)(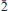 11,
11, 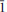 21), 2.644(21)(
21), 2.644(21)(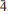 01), 2.296(22)(122). The crystal structures of both minerals were refined by single-crystal X-ray diffraction to R1 = 0.0459 (manganoblödite) and R1 = 0.0339 (cobaltoblödite).
01), 2.296(22)(122). The crystal structures of both minerals were refined by single-crystal X-ray diffraction to R1 = 0.0459 (manganoblödite) and R1 = 0.0339 (cobaltoblödite).
Tazzoliite: a new mineral with a pyrochlore-related structure from the Euganei Hills, Padova, Italy
- F. Cámara, F. Nestola, L. Bindi, A. Guastoni, F. Zorzi, L. Peruzzo, D. Pedron
-
- Journal:
- Mineralogical Magazine / Volume 76 / Issue 4 / August 2012
- Published online by Cambridge University Press:
- 05 July 2018, pp. 827-838
-
- Article
- Export citation
-
Tazzoliite, ideally Ba2CaSr0.5Na0.5Ti2Nb3SiO17[PO2(OH)2]0.5, is a new mineral (IMA 2011-018) from Monte delle Basse, Euganei Hills, Galzignano Terme, Padova, Italy. It occurs as lamellar pale orange crystals, which are typically a few m m thick and up to 0.4 mm long, closely associated with a diopsidic pyroxene and titanite. Tazzoliite is transparent. It has a white streak, a pearly lustre, is not fluorescent and has a hardness of 6 (Mohs' scale). The tenacity is brittle and the crystals have a perfect cleavage along {010}. The calculated density is 4.517 g cm–3. Tazzoliite is biaxial (–) with 2Vmeas of ~50º, it is not pleochroic and the average refractive index is 2.04. No twinning was observed. Electronmicroprobe analyses gave the following chemical formula: (Ba1.93Ca1.20Sr0.52Na0.25Fe0.102+)Σ4 (Nb2.88Ti2.05Ta0.07Zr0.01V0.015+)Σ5.02SiO17[(P0.13Si0.12S0.07)Σ0.32O0.66(OH)0.66][F0.09(OH)0.23]Σ0.32.
Tazzoliite is orthorhombic, space group Fmmm, with unit-cell parameters a = 7.4116(3), b = 20.0632(8), c = 21.4402(8) Å, V = 3188.2(2) Å3 and Z = 8. The crystal structure, obtained from single-crystal X-ray diffraction data, was refined to R1(F2) = 0.063. It consists of a framework of Nb(Ti) octahedra and BaO7 polyhedra sharing apexes or edges, and Si tetrahedra sharing apexes with Nb(Ti) octahedra and BaO7 polyhedra. The structure, which is related to the pyrochlore structure, contains three Nb(Ti) octahedra: two are Nb dominant and one is Ti dominant. Chains of A2O8 polyhedra [A2 being occupied by Sr(Ca, Fe)] extend along [100] and are surrounded by Nb octahedra. Channels formed by six Nb(Ti) octahedra and two tetrahedra, or four A1O8(OH) polyhedra (A1 being occupied by Ba), alternate along [100]. The channels are partially occupied by [PO2(OH)2] in two possible mutually exclusive positions, alternating with fully occupied A3O7 polyhedral pairs [A3 being occupied by Ca(Na)]. The seven strongest X-ray powder diffraction lines [d in Å (I/I0) (hkl)] are: 3.66 (60) (044), 3.16 (30) (153), 3.05 (100) (204), 2.98 (25) (240), 2.84 (50) (064), 1.85 (25) (400) and 1.82 (25) (268). Raman spectra of tazzoliite were collected in the range 150–3700 cm–1 and confirm the presence of OH groups. Tazzoliite is named in honour of Vittorio Tazzoli in recognition of his contributions to the fields of mineralogy and crystallography.




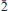 01); 3.791(26)(
01); 3.791(26)(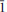 21), 2.968(22)(
21), 2.968(22)( 01); for cobaltoblödite: 4.551(80)(210, 011); 4.269(50)(
01); for cobaltoblödite: 4.551(80)(210, 011); 4.269(50)(