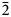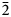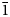8 results
Amableite-(Ce), Na15[(Ce1.5Na1.5)Mn3]Mn2Zr3□Si[Si24O69(OH)3](OH)2⋅H2O, a new eudialyte-group mineral from Saint-Amable Sill, Québec, Canada
- Nikita V. Chukanov, Andrey A. Zolotarev, Christof Schäfer, Dmitry A. Varlamov, Igor V. Pekov, Marina F. Vigasina, Dmitry I. Belakovskiy, Sergey M. Aksenov, Svetlana A. Vozchikova, Sergey N. Britvin
-
- Journal:
- Mineralogical Magazine , FirstView
- Published online by Cambridge University Press:
- 12 April 2024, pp. 1-11
-
- Article
- Export citation
-
The new eudialyte-group mineral amableite-(Ce), ideally Na15[(Ce1.5Na1.5)Mn3]Mn2Zr3□Si[Si24O69(OH)3](OH)2⋅H2O, was discovered in a peralkaline pegmatite at Saint-Amable Sill, Montérégie, Québec, Canada. The associated minerals are albite, microcline, aegirine, serandite, natrolite, yofortierite, and an unidentified titanosilicate forming minute grains. Amableite-(Ce) occurs as yellow equant or thick tabular crystals up to 2 mm across. The observed crystal forms are {0001}; the subordinate forms are {11$\bar{2}$
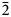 0}, {10$\bar{1}$
0}, {10$\bar{1}$ 1}, and {10$\bar{1}$
1}, and {10$\bar{1}$ 0}. Amableite-(Ce) is brittle, with a Mohs hardness of 5. D(meas) = 2.89(1), D(calc) = 2.899 g⋅cm–3. Amableite-(Ce) is optically anomalously biaxial (+) with α ≈ β = 1.603(2) and γ = 1.608(2). The chemical composition is (wt.%, electron microprobe, H2O measured by means of a modified Penfield method): Na2O 14.20, K2O 0.41, CaO 1.89, MnO 8.25, Fe2O3 2.40, La2O3 3.10, Ce2O3 4.19, Pr2O3 0.16, Nd2O3 0.59, SiO2 49.41, ZrO2 11.17, HfO2 0.24, TiO2 0.68, Nb2О5 1.54, Cl 0.26, H2O 1.70, –O≡Cl –0.06, total 100.13. The crystal structure was determined using single-crystal X-ray diffraction data and refined to R1 = 0.0423. Amableite-(Ce) is trigonal, space group R3, with a = 14.1340(3) Å, c = 30.3780(11) Å and V = 5255.6(3) Å3. The crystal-chemical formula is (Na12.93K0.27Ce0.06)Σ13.26[(Mn2.49Ce0.30Ca0.21)Σ3.00(Ce1.14Na1.04Ca0.82)Σ3.00](Mn1.05Fe0.90□1.05)Σ3.00(Zr2.85Ti0.12Hf0.03)Σ3.00(□0.40Nb0.36Si0.24)Σ1.00(Si0.88□0.12)Σ1.00[Si24(O70.44(OH)1.56)Σ72.00][(OH)2.20(H2O)1.27]Σ3.47Cl0.22 (Z = 3). Infrared and Raman spectra are given. The strongest lines of the powder X-ray diffraction pattern [d, Å (I, %)(hkl)] are: 11.34 (51)(101), 7.06 (76)(110), 4.312 (63)(205), 3.783 (38)(033), 3.538 (43)(027, 220), 2.963 (84)($\bar{3}$
0}. Amableite-(Ce) is brittle, with a Mohs hardness of 5. D(meas) = 2.89(1), D(calc) = 2.899 g⋅cm–3. Amableite-(Ce) is optically anomalously biaxial (+) with α ≈ β = 1.603(2) and γ = 1.608(2). The chemical composition is (wt.%, electron microprobe, H2O measured by means of a modified Penfield method): Na2O 14.20, K2O 0.41, CaO 1.89, MnO 8.25, Fe2O3 2.40, La2O3 3.10, Ce2O3 4.19, Pr2O3 0.16, Nd2O3 0.59, SiO2 49.41, ZrO2 11.17, HfO2 0.24, TiO2 0.68, Nb2О5 1.54, Cl 0.26, H2O 1.70, –O≡Cl –0.06, total 100.13. The crystal structure was determined using single-crystal X-ray diffraction data and refined to R1 = 0.0423. Amableite-(Ce) is trigonal, space group R3, with a = 14.1340(3) Å, c = 30.3780(11) Å and V = 5255.6(3) Å3. The crystal-chemical formula is (Na12.93K0.27Ce0.06)Σ13.26[(Mn2.49Ce0.30Ca0.21)Σ3.00(Ce1.14Na1.04Ca0.82)Σ3.00](Mn1.05Fe0.90□1.05)Σ3.00(Zr2.85Ti0.12Hf0.03)Σ3.00(□0.40Nb0.36Si0.24)Σ1.00(Si0.88□0.12)Σ1.00[Si24(O70.44(OH)1.56)Σ72.00][(OH)2.20(H2O)1.27]Σ3.47Cl0.22 (Z = 3). Infrared and Raman spectra are given. The strongest lines of the powder X-ray diffraction pattern [d, Å (I, %)(hkl)] are: 11.34 (51)(101), 7.06 (76)(110), 4.312 (63)(205), 3.783 (38)(033), 3.538 (43)(027, 220), 2.963 (84)($\bar{3}$ 45), 2.837 (100)(404). The mineral is named after the discovery locality.
45), 2.837 (100)(404). The mineral is named after the discovery locality.
Selsurtite, (H3O)12Na3(Ca3Mn3)(Na2Fe)Zr3□Si[Si24O69(OH)3](OH)Cl⋅H2O, a new eudialyte-group mineral from the Lovozero alkaline massif, Kola Peninsula, Russia
- Nikita V. Chukanov, Sergey M. Aksenov, Olga N. Kazheva, Igor V. Pekov, Dmitry A. Varlamov, Marina F. Vigasina, Dmitry I. Belakovskiy, Svetlana A. Vozchikova, Sergey N. Britvin
-
- Journal:
- Mineralogical Magazine / Volume 87 / Issue 2 / April 2023
- Published online by Cambridge University Press:
- 09 December 2022, pp. 241-251
-
- Article
- Export citation
-
The new eudialyte-group mineral selsurtite, ideally (H3O)12Na3(Ca3Mn3)(Na2Fe)Zr3□Si[Si24O69(OH)3](OH)Cl⋅H2O, was discovered in metasomatic peralkaline rock from the Flora mountain, northern spur of the Selsurt mountain, Lovozero alkaline massif, Kola Peninsula, Russia. The associated minerals are aegirine, albite and orthoclase, as well as accessory lorenzenite, calciomurmanite, natrolite, lamprophyllite and sergevanite. Selsurtite occurs as brownish-red to reddish-orange, equant or flattened on (0001) crystals up to 2 mm across and elongate crystals up to 3 cm long. The main crystal forms are {0001}, {11$\bar{2}$
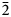 0}, and {10$\bar{1}$
0}, and {10$\bar{1}$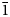 1}. Selsurtite is brittle, with the Mohs’ hardness of 5. No cleavage is observed. Parting is distinct on (001). D(meas) = 2.73(2) and D(calc) = 2.722 g⋅cm–3. Selsurtite is optically uniaxial (–), with ω = 1.598(2) and ɛ = 1.595(2). The chemical composition is (wt.%, electron microprobe): Na2O 6.48, K2O 0.27, MgO 0.10, CaO 6.83, MnO 4.73, FeO 1.18, SrO 1.88, La2O3 0.57, Ce2O3 1.07, Pr2O3 0.20, Nd2O3 0.44, Al2O3 0.29, SiO2 50.81, ZrO2 13.50, HfO2 0.45, TiO2 0.61, Nb2О5 1.10, Cl 1.01, SO3 0.29, H2O 8.10, –O≡Cl –0.23, total 99.68. The empirical formula is H25.94Na6.03K0.16Mg0.07Ca3.51Sr0.52Ce0.19La0.10Nd0.08Pr0.03Mn1.91Fe0.47Ti0.22Zr3.16Hf0.06Nb0.24Si24.40Al0.16S0.10Cl0.82O79.13. The crystal structure was determined using single-crystal X-ray diffraction data and refined to R = 0.0484. Selsurtite is trigonal, space group R3, with a = 14.1475(7) Å, c = 30.3609(12) Å, V = 5262.65(7) Å3 and Z = 3. Infrared and Raman spectra show that hydronium cations are involved in very strong hydrogen bonds and form Zundel- and Eigen-like complexes. The strongest lines of the powder X-ray diffraction pattern [d, Å (I, %)(hkl)] are: 11.38 (56)(101), 7.08 (59)(110), 5.69 (36)(202), 4.318 (72)(205), 3.793 (36)(303), 3.544 (72)(027, 220, 009), 2.970 (100)(315) and 2.844 (100)(404). The mineral is named after the discovery locality.
1}. Selsurtite is brittle, with the Mohs’ hardness of 5. No cleavage is observed. Parting is distinct on (001). D(meas) = 2.73(2) and D(calc) = 2.722 g⋅cm–3. Selsurtite is optically uniaxial (–), with ω = 1.598(2) and ɛ = 1.595(2). The chemical composition is (wt.%, electron microprobe): Na2O 6.48, K2O 0.27, MgO 0.10, CaO 6.83, MnO 4.73, FeO 1.18, SrO 1.88, La2O3 0.57, Ce2O3 1.07, Pr2O3 0.20, Nd2O3 0.44, Al2O3 0.29, SiO2 50.81, ZrO2 13.50, HfO2 0.45, TiO2 0.61, Nb2О5 1.10, Cl 1.01, SO3 0.29, H2O 8.10, –O≡Cl –0.23, total 99.68. The empirical formula is H25.94Na6.03K0.16Mg0.07Ca3.51Sr0.52Ce0.19La0.10Nd0.08Pr0.03Mn1.91Fe0.47Ti0.22Zr3.16Hf0.06Nb0.24Si24.40Al0.16S0.10Cl0.82O79.13. The crystal structure was determined using single-crystal X-ray diffraction data and refined to R = 0.0484. Selsurtite is trigonal, space group R3, with a = 14.1475(7) Å, c = 30.3609(12) Å, V = 5262.65(7) Å3 and Z = 3. Infrared and Raman spectra show that hydronium cations are involved in very strong hydrogen bonds and form Zundel- and Eigen-like complexes. The strongest lines of the powder X-ray diffraction pattern [d, Å (I, %)(hkl)] are: 11.38 (56)(101), 7.08 (59)(110), 5.69 (36)(202), 4.318 (72)(205), 3.793 (36)(303), 3.544 (72)(027, 220, 009), 2.970 (100)(315) and 2.844 (100)(404). The mineral is named after the discovery locality.
Columbite supergroup of minerals: nomenclature and classification
- Nikita V. Chukanov, Marco Pasero, Sergey M. Aksenov, Sergey N. Britvin, Natalia V. Zubkova, Li Yike, Thomas Witzke
-
- Journal:
- Mineralogical Magazine / Volume 87 / Issue 1 / February 2023
- Published online by Cambridge University Press:
- 08 September 2022, pp. 18-33
-
- Article
- Export citation
-
The columbite supergroup is established. It includes five mineral groups (ixiolite, wolframite, samarskite, columbite and wodginite) and one ungrouped species (lithiotantite). The criteria for a mineral to belong to the columbite supergroup are: the general stoichiometry MO2; the crystal structure based on the hexagonal close packing (hcp) of anions (or close to it); the six-fold coordination number of M-type cations (augmented to eight-fold in the case of slight distortion of hcp); and the presence of zig-zag chains of edge-sharing M-centred polyhedra. The ixiolite-type structure is considered as an aristotype with the space group Pbcn, the smallest unit cell volume, and the basic vectors a0, b0 and c0. Based on the multiplying of the ixiolite-type unit cell the following derivatives are distinguished: ixiolite type [ixiolite-group minerals; a = a0, b = b0 and c = c0; space group Pbcn; the members are ixiolite-(Mn2+), ixiolite-(Fe2+), scrutinyite, seifertite and srilankite]; wolframite type [wolframite-group minerals, ordered analogues of the ixiolite type with a = a0, b = b0 and c = c0; P2/c; the members are ferberite, hübnerite, huanzalaite, sanmartinite, heftetjernite, nioboheftetjernite, rossovskyite and riesite]; samarskite type [samarskite-group minerals; a = 2a0, b = b0 and c = c0; P2/c; the members are samarskite-(Y), ekebergite and shakhdaraite-(Y)]; columbite type [columbite-group minerals; a = 3a0, b = b0 and c = c0; Pbcn; the members are columbite-(Fe), columbite-(Mn), columbite-(Mg), tantalite-(Fe), tantalite-(Mn), tantalite-(Mg), fersmite, euxenite-(Y), tanteuxenite-(Y) and uranopolycrase]; and wodginite type [wodginite-group minerals; a = 2a0, b = 2b0 and c = c0; C2/c; the members are wodginite, ferrowodginite, titanowodginite, ferrotitanowodginite, tantalowodginite, lithiowodginite and achalaite]. Samarskite-(Yb), ishikawaite and calciosamarskite are insufficiently studied, tentatively considered as possible members of the samarskite supergroup. Qitianlingite, yttrocolumbite-(Y), yttrotantalite-(Y) and yttrocrasite-(Y) are questionable and need further studies. Polycrase-(Y) is discredited as identical to euxenite-(Y). Ixiolite has been renamed as ixiolite-(Mn2+), with the end-member formula (Ta2/3Mn2+1/3)O2. Ta- and Nb-dominant analogues of ixiolite with different schemes of charge balancing have the end-member formulae (M15+0.5M23+0.5)O2, M15+2/3M22+1/3)O2, M15+0.75M2+0.25)O2 or M15+0.8□0.2)O2 and the root name ‘ixiolite’ (for M1 = Ta) or ‘nioboixiolite’ (for M1 = Nb).
Crystal chemistry and origin of REE-bearing mukhinite from carbonate veins of the Svetlinsky gold deposit, South Urals, Russia
- Victor G. Korinevsky, Nikita V. Chukanov, Sergey M. Aksenov, Evgeniy V. Korinevsky, Vasiliy A. Kotlyarov, Dmitry А. Zamyatin, Anastasiya D. Ryanskaya, Sergey V. Kolisnichenko, Svetlana M. Lebedeva, Vera N. Ermolaeva
-
- Journal:
- Mineralogical Magazine / Volume 86 / Issue 5 / October 2022
- Published online by Cambridge University Press:
- 12 May 2022, pp. 821-833
-
- Article
- Export citation
-
A rare earth element (REE)-, Cr- and Mg-bearing variety of the vanadium epidote-group mineral mukhinite occurs in a calcite–dolomite carbonatite dyke cutting metamorphosed volcano-sedimentary rocks exposed in the walls of the quarry of the Svetlinsky gold deposit, South Urals. This mineral was found in a paragene assemblage including native sulphur, phlogopite and fluorophlogopite, together with accessory pyrite, other sulfides and sulfosalts, gold, Cr- and V-bearing muscovite, margarite, Cr- and V-bearing dravite, fluoro-tremolite, actinolite, fluoro-pargasite, anhydrite, apatite, uranium hydroxides, V-rich titanite, V- and Nb-rich rutile, spinel and corundum. The contents of ΣREE2O3 and V2O3 in mukhinite vary in the ranges of 4.01–9.69 and 5.34–7.46 wt.%, respectively. A Raman spectrum of REE-rich mukhinite is provided. The main schemes of isomorphic substitutions in mukhinite are ΣREE + Mg ↔ Ca + Al and V+Cr ↔ Al. The crystal structure of REE-rich mukhinite has been studied by single-crystal X-ray diffraction analysis. The mineral is monoclinic, with the space group P21/m, and unit-cell parameters are: a = 8.8972(11) Å, b = 5.6221(6) Å, c = 10.1519(12) Å, β = 115.169° and V = 459.60(11) Å3. The crystal structure of REE-rich mukhinite is similar to that of its synthetic analogue; the refined crystal-chemical formula of the sample studied is (Z = 2): {A1CaA2(Ca0.8REE0.2)}{M1(Al0.95Cr0.05)M2AlM3[(V,Cr)3+0.40Al0.35Mg0.25]}(Si2O7)(SiO4)O(OH).
Structural and chemical complexity of minerals: an update
- Sergey V. Krivovichev, Vladimir G. Krivovichev, Robert M. Hazen, Sergey M. Aksenov, Margarita S. Avdontceva, Alexander M. Banaru, Liudmila A. Gorelova, Rezeda M. Ismagilova, Ilya V. Kornyakov, Ivan V. Kuporev, Shaunna M. Morrison, Taras L. Panikorovskii, Galina L. Starova
-
- Journal:
- Mineralogical Magazine / Volume 86 / Issue 2 / April 2022
- Published online by Cambridge University Press:
- 04 April 2022, pp. 183-204
-
- Article
-
- You have access Access
- HTML
- Export citation
-
The complexities of chemical composition and crystal structure are fundamental characteristics of minerals that have high relevance to the understanding of their stability, occurrence and evolution. This review summarises recent developments in the field of mineral complexity and outlines possible directions for its future elaboration. The database of structural and chemical complexity parameters of minerals is updated by H-correction of structures with unknown H positions and the inclusion of new data. The revised average complexity values (arithmetic means) for all minerals are 3.54(2) bits/atom and 345(10) bits/cell (based upon 4443 structure reports). The distributions of atomic information amounts, chemIG and strIG, versus the number of mineral species fit the normal modes, whereas the distributions of total complexities, chemIG,total and strIG,total, along with numbers of atoms per formula and per unit cell are log normal. The three most complex mineral species known today are ewingite, morrisonite and ilmajokite, all either discovered or structurally characterised within the last five years. The most important complexity-generating mechanisms in minerals are: (1) the presence of isolated large clusters; (2) the presence of large clusters linked together to form three-dimensional frameworks; (3) formation of complex three-dimensional modular frameworks; (4) formation of complex modular layers; (5) high hydration state in salts with complex heteropolyhedral units; and (6) formation of ordered superstructures of relatively simple structure types. The relations between symmetry and complexity are considered. The analysis of temporal dynamics of mineralogical discoveries since 1875 with the step of 25 years show the increasing chemical and structural complexities of human knowledge of the mineral kingdom in the history of mineralogy. In the Earth's history, both diversity and complexity of minerals experience dramatic increases associated with the formation of Earth's continental crust, initiation of plate tectonics and the Great Oxidation event.
Almeidaite, Pb(Mn,Y)Zn2(Ti,Fe3+)18O36(O,OH)2, a new crichtonite-group mineral, from Novo Horizonte, Bahia, Brazil
- Luiz A. D. Menezes Filho, Nikita V. Chukanov, Ramiza K. Rastsvetaeva, Sergey M. Aksenov, Igor V. Pekov, Mário L. S. C. Chaves, R. Peter Richards, Daniel Atencio, Paulo R. G. Brandão, Ricardo Scholz, Klaus Krambrock, Roberto L. Moreira, Frederico S. Guimarães, Antônio W. Romano, Aba C. Persiano, Luiz C. A. de Oliveira, José D. Ardisson
-
- Journal:
- Mineralogical Magazine / Volume 79 / Issue 2 / April 2015
- Published online by Cambridge University Press:
- 02 January 2018, pp. 269-283
-
- Article
- Export citation
-
Almeidaite (IMA 2013-020), ideally Pb(Mn, Y)Zn2(Ti,Fe3+)18O36(O, OH)2, from Novo Horizonte, Bahia, Brazil, occurs in association with quartz, rutile, anatase, hematite, kaolinite, muscovite, xenotime-(Y) and bastnaesite-(La). Almeidaite forms isolated, black, opaque, sub-metallic, platy crystals flattened on [0001], measuring up to 30 mm 6 30 mm 6 6 mm in size, dominated by the basal pinacoid {0001}, which is bounded by various, mostly steep, rhombohedra and the hexagonal prism {1120}. Most of the crystals are multiply twinned, with non-planar contact surfaces that are approximately parallel to the c axis. The streak is brown. Reflectance values are [(Ro, Re) λ (nm)]: (12.78, 15.39) 470; (12.86, 15.43) 546; (12.91, 15.55) 589; (13.04, 15.75) 650. The empirical formula is (Pb0.59Sr0.12Ca0.04La0.03)Σ0.78(Mn0.54Y0.46)Σ1.00Zn1.43(Ti13.02Fe4.983+)Σ18.00(Fe0.323+Mn0.15)Σ0.47[O37.18(OH)0.82]Σ38.00. It is trigonal, space group R3, with the unit-cell parameters a = 10.4359(2), c = 21.0471(4) Å, V = 1985.10(7) Å3 and Z = 3. The crystal structure was solved (R1 = 0.039) using 2110 unique reflections with I > 3σ(I). Almeidaite is a member of the crichtonite group with Pb dominant in the A site (with 12-fold coordination) and Zn dominant in the T site (with 4-fold coordination). It is a Zn analogue of senaite and a Pb analogue of landauite. The mineral is named after Professor Fernando Flávio Marques de Almeida (1916–2013).
Eleonorite, Fe63+(PO4)4O(OH)4·6H2O: validation as a mineral species and new data
- Nikita V. Chukanov, Sergey M. Aksenov, Ramiza K. Rastsvetaeva, Christof Schäfer, Igor V. Pekov, Dmitriy I. Belakovskiy, Ricardo Scholz, Luiz C.A. de Oliveira, Sergey N. Britvin
-
- Journal:
- Mineralogical Magazine / Volume 81 / Issue 1 / February 2017
- Published online by Cambridge University Press:
- 02 January 2018, pp. 61-76
-
- Article
- Export citation
-
Eleonorite, ideally Fe63+(PO4)4O(OH)4·6H2O, the analogue of beraunite Fe2+Fe53+(PO4)4O(OH)5·6H2O with Fe2+ completely substituted by Fe3+, has been approved by the International Mineralogical Association Commission on New Minerals, Nomenclature and Classification as a mineral species (IMA 2015-003). The mineral was first described on material from the Eleonore Iron mine, Dünsberg, near Giessen, Hesse, Germany, but during this study further samples were required and a neotype locality is the Rotläufchen mine, Waldgirmes, Wetzlar, Hesse, Germany, where eleonorite is associated with goethite, rockbridgeite, dufrénite, kidwellite, variscite, matulaite, planerite, cacoxenite, strengite and wavellite. Usually eleonorite occurs as red-brown prismatic crystals up to 0.2 mm × 0.5 mm × 3.5 mm in size and in random or radial aggregates up to 5 mm across encrusting cavities in massive 'limonite'. The mineral is brittle. Its Mohs hardness is 3. Dmeas = 2.92(1), Dcalc = 2.931 g cm–3. The IR spectrum is given. Eleonorite is optically biaxial (+), α = 1.765(4), β = 1.780(5), γ = 1.812(6), 2Vmeas = 75(10)°, 2Vcalc = 70°. The chemical composition (electron microprobe data, H2O analysed by chromatography of products of ignition at 1200°C, wt.%) is: Al2O3 1.03, Mn2O3 0.82, Fe2O3 51.34, P2O5 31.06, H2O 16.4, total 99.58. All iron was determined as being trivalent from a Mössbauer analysis. The empirical formula (based on 27 O apfu) is (Fe5.763+Al0.18Mn0.093+)∑6.03(PO4)3.92O(OH)4.34·5.98H2O. The crystal structure (R = 0.0633) is similar to that of beraunite and is based on a heteropolyhedral framework formed by M(1–4)Ø6-octahedra (where M = Fe3+; Ø = O2–, OH– or H2O) and isolated PO4 tetrahedra, with a wide channel occupied by H2O molecules. Eleonorite is monoclinic, space group C2/c, a = 20.679(10), b = 5.148(2), c = 19.223(9) Å, β = 93.574(9)°, V = 2042.5(16) Å3 and Z = 4. The strongest reflections of the powder X-ray diffraction pattern [d, Å (I,%) (Hkl)] are 10.41 (100) (200), 9.67 (38) (002), 7.30 (29) (202̄), 4.816 (31) (111, 004), 3.432 (18) (600, 114, 404, 313), 3.197 (18) (510, 511̄, 006, 314̄, 602), 3.071 (34) (314, 115̄).
Antipinite, KNa3Cu2(C2O4)4, a new mineral species from a guano deposit at Pabellón de Pica, Chile
- Nikita V. Chukanov, Sergey M. Aksenov, Ramiza K. Rastsvetaeva, Konstantin A. Lyssenko, Dmitriy I. Belakovskiy, Gunnar Färber, Gerhard Möhn, Konstantin V. Van
-
- Journal:
- Mineralogical Magazine / Volume 79 / Issue 5 / October 2015
- Published online by Cambridge University Press:
- 02 January 2018, pp. 1111-1121
-
- Article
- Export citation
-
The new oxalate mineral antipinite is found in a guano deposit located on the Pabellón de Pica Mountain, Iquique Province, Tarapacá Region, Chile. Associated minerals are halite, salammoniac, chanabayaite, joanneumite and clays. Antipinite occurs as blue, imperfect, short prismatic crystals up to 0.1 mm × 0.1 mm × 0.15 mm in size, as well as their clusters and random aggregates. The mineral is brittle. Mohs hardness is 2; Dmeas = 2.53(3), Dcalc = 2.549 g cm–3. The infrared spectrum shows the presence of oxalate anions and the absence of absorptions associated with H2O molecules, C–H bonds, CO32–, NO3– and OH– ions. Antipinite is optically biaxial (+), α = 1.432(3), β = 1.530(1), γ = 1.698(5), 2Vmeas = 75(10)°, 2Vcalc = 82°. The chemical composition (electron-microprobe data, C measured by gas chromatography of products of ignition at 1200°C, wt.%) is Na2O 15.95, K2O 5.65, CuO 27.34, C2O3 48.64, total 99.58. The empirical formula is K0.96Na3.04Cu2.03(C2.00O4)4 and the idealized formula is KNa3Cu2(C2O4)4. The crystal structure was solved and refined to R = 0.033 based upon 4085 unique reflections with I > 2σ(I). Antipinite is triclinic, space group P1, a = 7.1574(5), b = 10.7099(8), c = 11.1320(8) Å, α = 113.093(1), β = 101.294(1), γ = 90.335 (1)°, V = 766.51(3) Å3, Z = 2. The strongest reflections of the powder X-ray diffraction pattern [d, Å (I,%) (hkl)] are 5.22 (40) (111), 3.47 (100) (032), 3.39 (80) (210), 3.01 (30) (033, 220), 2.543 (40) (122, 034, 104), 2.481 (30) (213), 2.315 (30) (143, 310), 1.629 (30) (146, 414, 243, 160).