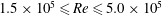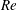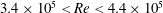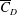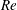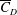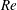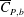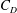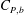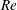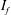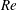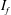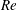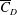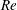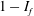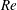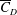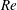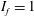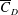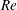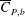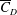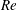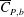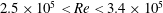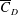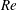4 results
52350 PKM2 mediates anti-tumor immunity and T cell dysfunction
- Geoffrey Markowitz, Yi Ban, Michael Crowley, Diamile Tavarez, Stephen T.C. Wong, Kenneth Chang, Andrea Schietinger, Nasser Altorki, Vivek Mittal
-
- Journal:
- Journal of Clinical and Translational Science / Volume 5 / Issue s1 / March 2021
- Published online by Cambridge University Press:
- 31 March 2021, p. 89
-
- Article
-
- You have access Access
- Open access
- Export citation
-
ABSTRACT IMPACT: T cell dysfunction is a dominant suppressor of anti-tumor immunity, reducing immunotherapeutic efficacy and benefit to patients; our work will identify novel mediators of this process for both therapeutic potential and underlying mechanism, allowing for both potential immediate clinical utility and identification of future targets based on new mechanistic insights. OBJECTIVES/GOALS: T cell dysfunction is a dominant suppressor of anti-tumor immunity, reducing immunotherapeutic efficacy and clinical benefit to the majority of patients. We aim to interrogate a novel mediator of dysfunction identified from transcriptome analyses, pyruvate kinase muscle isozyme isoform 2 (PKM2), for therapeutic utility and underlying mechanism. METHODS/STUDY POPULATION: Transcriptome analyses of CD8+ lymphocytes from tumor-bearing lungs from both murine KrasG12D p53-/- and human non-small cell lung cancer (NSCLC) patients were performed, and differentially expressed genes identified. Flow cytometric analyses for PKM isoform expression and effects of target knockdown on accumulation of dysfunctional characteristics, including checkpoint and transcription factor expression, proliferation, and cytokine production, were performed using an in vitro co-culture of murine antigen-specific T (OT-I) cells and antigen-expressing NSCLC (HKP1-ova) cells. In vivo examination of the same was performed using adoptive transfer of OT-I cells into immunocompetent recipient mice with engraftment of HKP1-ova cells, and subsequent evaluation of mouse survival and T cell phenotypes. RESULTS/ANTICIPATED RESULTS: Transcriptome analyses demonstrated that PKM expression was upregulated in dysfunctional T cells from both murine and human samples. This was confirmed both in vitro with co-culture and in vivo with adoptive transfer approaches, with both activated and dysfunctional OT-I cells expressing higher levels of isoform 2 of PKM than naive OT-I cells. Expression of PKM2 mimicked the kinetics of the transcription factor Tox, a known driver of dysfunction, and knockdown of PKM2 resulted in reduced granzyme B expression, and increased proportions of progenitors with fewer terminally differentiated dysfunctional cells. Knockdown of PKM2 in adoptively-transferred OT-I cells led to enhanced tumor control; results are being extended to other tumor models, and T cells metabolically profiled with PKM2 manipulation. DISCUSSION/SIGNIFICANCE OF FINDINGS: This work identified a novel mediator of dysfunction whose targeting has the potential to enhance anti-tumor immunity. Mechanistically, targeting PKM2 led to altered T cell differentiation to a dysfunctional state, linking metabolic phenotypes to these traits and underlining the importance and therapeutic potential of T cell metabolic pathways.
Surgical Site Infections at a Level I Trauma Center in India: Data From an Indigenously Developed, e-SSI Surveillance System
- Ayush Lohiya, Samarth Mittal, Vivek Trikha, Surbhi Khurana, Sonal Katyal, Sushma Sagar, Subodh Kumar, Rajesh Malhotra, Purva Mathur
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s397-s398
- Print publication:
- October 2020
-
- Article
-
- You have access Access
- Export citation
-
Background: Globally, surgical site infections (SSIs) not only complicate the surgeries but also lead to $5–10 billion excess health expenditures, along with the increased length of hospital stay. SSI rates have become a universal measure of quality in hospital-based surgical practice because they are probably the most preventable of all healthcare-associated infections. Although, many national regulatory bodies have made it mandatory to report SSI rates, the burden of SSI is still likely to be significant underestimated due to truncated SSI surveillance as well as underestimated postdischarge SSIs. A WHO survey found that in low- to middle-income countries, the incidence of SSIs ranged from 1.2 to 23.6 per 100 surgical procedures. This contrasted with rates between 1.2% and 5.2% in high-income countries. Objectives: We aimed to leverage the existing surveillance capacities at our tertiary-care hospital to estimate the incidence of SSIs in a cohort of trauma patients and to develop and validate an indigenously developed, electronic SSI surveillance system. Methods: A prospective cohort study was conducted at a 248-bed apex trauma center for 18 months. This project was a part of an ongoing multicenter study. The demographic details were recorded, and all the patients who underwent surgery (n = 770) were followed up until 90 days after discharge. The associations of occurrence of SSI and various clinico-microbiological variables were studied. Results: In total, 32 (4.2%) patients developed SSI. S. aureus (28.6%) were the predominant pathogen causing SSI, followed by E. coli (14.3%) and K. pneumoniae (14.3%). Among the patients who had SSI, higher SSI rates were associated in patients who were referred from other facilities (P = .03), had wound class-CC (P < .001), were on HBOT (P = .001), were not administered surgical antibiotics (P = .04), were not given antimicrobial coated sutures (P = .03) or advanced dressings (P = .02), had a resurgery (P < .001), had a higher duration of stay in hospital from admission to discharge (P = .002), as well as from procedure to discharge (P = .002). SSI was cured in only 16 patients (50%) by 90 days. SSI data collection, validation, and analyses are essential in developing countries like India. Thus, it is very crucial to implement a surveillance system and a system for reporting SSI rates to surgeons and conduct a robust postdischarge surveillance using trained and committed personnel to generate, apply, and report accurate SSI data.
Funding: None
Disclosures: None
Intermittency of laminar separation bubble on a sphere during drag crisis
- Rahul Deshpande, Vivek Kanti, Aditya Desai, Sanjay Mittal
-
- Journal:
- Journal of Fluid Mechanics / Volume 812 / 10 February 2017
- Published online by Cambridge University Press:
- 05 January 2017, pp. 815-840
-
- Article
- Export citation
-
The phenomenon of drag crisis for uniform flow past a smooth sphere is investigated via experiments in a low-turbulence wind tunnel for
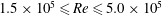 $1.5\times 10^{5}\leqslant Re\leqslant 5.0\times 10^{5}$. The Reynolds number,
$1.5\times 10^{5}\leqslant Re\leqslant 5.0\times 10^{5}$. The Reynolds number, 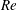 $Re$, is based on the free-stream speed and the diameter of the sphere. Based on the activity related to the laminar separation bubble (LSB), the critical regime for the occurrence of drag crisis (
$Re$, is based on the free-stream speed and the diameter of the sphere. Based on the activity related to the laminar separation bubble (LSB), the critical regime for the occurrence of drag crisis (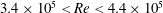 $3.4\times 10^{5}<Re<4.4\times 10^{5}$) is further divided into three subregimes. The gradual decrease of mean drag coefficient (
$3.4\times 10^{5}<Re<4.4\times 10^{5}$) is further divided into three subregimes. The gradual decrease of mean drag coefficient (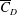 $\overline{C}_{D}$) with
$\overline{C}_{D}$) with 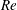 $Re$, in subregime I, is due to the increase of base pressure and suction near the shoulder of the sphere. The flow is devoid of an LSB in this regime. The coefficient
$Re$, in subregime I, is due to the increase of base pressure and suction near the shoulder of the sphere. The flow is devoid of an LSB in this regime. The coefficient 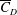 $\overline{C}_{D}$ decreases very rapidly with increase in
$\overline{C}_{D}$ decreases very rapidly with increase in 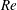 $Re$ in subregime II primarily due to the increase in mean base pressure (
$Re$ in subregime II primarily due to the increase in mean base pressure (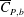 $\overline{C}_{P,b}$). This subregime is characterized by intermittent switching of
$\overline{C}_{P,b}$). This subregime is characterized by intermittent switching of 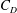 $C_{D}$ and
$C_{D}$ and 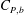 $C_{P,b}$ between bistable states. Statistical analysis of the surface-pressure and force coefficients relates this behaviour to the intermittent appearance/disappearance of the LSB. The two states of the flow are referred to as the LSB and non-LSB states. The frequency of appearance of the LSB and the duration of its stay increase with increase in
$C_{P,b}$ between bistable states. Statistical analysis of the surface-pressure and force coefficients relates this behaviour to the intermittent appearance/disappearance of the LSB. The two states of the flow are referred to as the LSB and non-LSB states. The frequency of appearance of the LSB and the duration of its stay increase with increase in 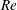 $Re$. An intermittency factor
$Re$. An intermittency factor 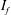 $I_{f}$, defined as the fraction of time during which the LSB exists in the flow, is estimated at each
$I_{f}$, defined as the fraction of time during which the LSB exists in the flow, is estimated at each 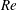 $Re$. The value of
$Re$. The value of 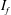 $I_{f}$ is zero in subregime I and increases from zero to one, with increase in
$I_{f}$ is zero in subregime I and increases from zero to one, with increase in 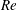 $Re$, in subregime II. The variation of
$Re$, in subregime II. The variation of 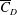 $\overline{C}_{D}$ with
$\overline{C}_{D}$ with 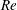 $Re$ is found to follow the variation of (
$Re$ is found to follow the variation of (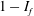 $1-I_{f}$) with
$1-I_{f}$) with 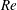 $Re$. This shows that the decrease of
$Re$. This shows that the decrease of 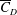 $\overline{C}_{D}$ with increase in
$\overline{C}_{D}$ with increase in 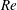 $Re$, during drag crisis, is primarily due to the increased probability of the LSB state as opposed to the non-LSB state. A spatio-temporal analysis of the surface pressure measured at various polar locations on the surface of the sphere confirms the axisymmetric nature of the intermittent LSB. In subregime III of the critical regime, the LSB exists at all time instants (
$Re$, during drag crisis, is primarily due to the increased probability of the LSB state as opposed to the non-LSB state. A spatio-temporal analysis of the surface pressure measured at various polar locations on the surface of the sphere confirms the axisymmetric nature of the intermittent LSB. In subregime III of the critical regime, the LSB exists at all time instants (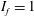 $I_{f}=1$). The
$I_{f}=1$). The 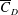 $\overline{C}_{D}$ value continues to decrease with
$\overline{C}_{D}$ value continues to decrease with 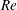 $Re$ in this subregime due to increase in
$Re$ in this subregime due to increase in 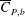 $\overline{C}_{P,b}$. Unlike the general belief that the decrease in
$\overline{C}_{P,b}$. Unlike the general belief that the decrease in 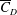 $\overline{C}_{D}$ with increase in
$\overline{C}_{D}$ with increase in 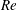 $Re$ is due only to the increase in
$Re$ is due only to the increase in 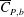 $\overline{C}_{P,b}$, it is found that the increase in suction upstream of the shoulder of the sphere, with increase in
$\overline{C}_{P,b}$, it is found that the increase in suction upstream of the shoulder of the sphere, with increase in 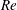 $Re$, also plays an important role. In particular, in the high subcritical regime (
$Re$, also plays an important role. In particular, in the high subcritical regime (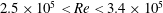 $2.5\times 10^{5}<Re<3.4\times 10^{5}$), the gradual decrease in
$2.5\times 10^{5}<Re<3.4\times 10^{5}$), the gradual decrease in 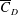 $\overline{C}_{D}$ with increase in
$\overline{C}_{D}$ with increase in 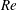 $Re$ is due solely to the increase in suction in a region upstream of the shoulder of the sphere.
$Re$ is due solely to the increase in suction in a region upstream of the shoulder of the sphere.
81 - Signal transduction in tumor angiogenesis
- from Part 4 - Pharmacologic targeting of oncogenic pathways
-
- By Timothy Hla, Center of Vascular Biology, Department of Pathology and Laboratory Medicine,Weill Medical College of Cornell University, New York, NY, USA, Nasser Altorki, Department of Cardiothoracic Surgery and Neuberger Berman Lung Cancer Research Center,Weill Medical College of Cornell University, New York, NY, USA, Vivek Mittal, Department of Cardiothoracic Surgery and Neuberger Berman Lung Cancer Research Center, and Department of Cell and Developmental Biology, Weill Medical College of Cornell University, New York, NY, USA
- Edited by Edward P. Gelmann, Columbia University, New York, Charles L. Sawyers, Memorial Sloan-Kettering Cancer Center, New York, Frank J. Rauscher, III
-
- Book:
- Molecular Oncology
- Published online:
- 05 February 2015
- Print publication:
- 19 December 2013, pp 861-871
-
- Chapter
- Export citation
-
Summary
Angiogenesis is the formation of nascent blood vessels from existing vasculature. It is a crucial step in physiological conditions such as normal growth, embryonic development, female estrous cycle, and wound healing, as well as in pathological scenarios such as tumor growth, diabetic retinopathy, and rheumatoid arthritis (1). During cancer progression, the angiogenic vasculature is needed for the supply of oxygen and nutrients that sustain tumor growth, and eventually acts as a conduit for metastatic dissemination of tumor cells to distant organs (2,3). Accordingly, tumor angiogenesis remains an important area of cancer research, and understanding its mechanistic basis is critical for the development of effective anti-angiogenic therapy.
Under normal physiological conditions, angiogenesis is well controlled by pro- and anti-angiogenic factors. However, in cancer, this balance of pro- and anti-angiogenic factors is perturbed, resulting in the so-called “angiogenic switch.” Multiple signals trigger the angiogenic switch, including genetic mutations, metabolic and mechanical stresses, and inflammatory responses (4–9; Figure 81.1). Growing tumors progressively become hypoxic, leading to stabilization of the hypoxia inducible factor 1α (HIF-1α) which, in turn, stimulates production of key angiogenic growth factors, including vascular endothelial growth factor (VEGF), platelet-derived growth factor (PDGF), placental growth factor (PLGF), granulocyte colony-stimulating factor (G-CSF), interleukin 8 (IL8), and hepatocyte growth factor (HGF). VEGF-A has been heralded as the most potent endothelial-specific angiogenic factor, which recognizes cognate tyrosine-kinase receptors such as VEGFR-2 and -3 on the endothelial cells, resulting in downstream activation of signal-transduction cascades (10), which induce endothelial cell activation and sprouting of new capillaries. In addition to the pro-angiogenic factors, there are various endogenous angiogenesis-inhibitor proteins including endostatin, angiostatin, thrombospondin-1 (Tsp-1), tumstatin, platelet factor 4, and certain interleukins, including IL-12. De novo blood-vessel formation results from a complex interplay of pro- and anti-angiogenic regulators, and dysregulation of the balance between these factors is the hallmark of tumor angiogenesis. In addition to the participation of vascular endothelial-derived vessels, the generation of new lymphatic vessels by a process referred to as lymphangiogenesis has also been implicated in tumor progression and metastasis (11,12).