The world market for functional foods (FF) and dietary supplements (DS) is expanding rapidly. In 2010 FF are expected to represent 5 % of the total global food market(1) and the market for DS is estimated at more than $60 billion worldwide(Reference Crowley and FitzGerald2). In general, the target population of FF or DS is healthy individuals with slightly elevated risk factors or some physical discomfort. However, due to the fast growing market of FF or DS and the accompanying strong advertising and marketing, also patients on medication may be stimulated to use FF or DS. This may have several consequences for the quality of drug treatment as stated by de Jong et al. with the example of the combined intake of plant sterols and/or stanols and statins(Reference de Jong, Klungel and Verhagen3). Whereas they addressed the additive effect of plant sterols and/or stanols on reducing LDL-cholesterol values in patients on statin treatment, their main focus was the possible negative aspects of the combination, such as unfavourable effects on patient adherence with drug treatment and increasing the potential for food–drug interactions.
In the present review we will focus on the possible beneficial effects that FF or DS may have on drug therapy. Because of the large number of subjects treated suboptimally with statins (hydroxymethylglutaryl CoA (HMG-CoA) reductase inhibitors)(Reference Mantel-Teeuwisse, Verschuren and Klungel4) and the availability of several FF and DS possibly contributing to the beneficial effects of statin treatment, we will put special emphasis on this group of drugs.
In theory, FF or DS may support drug therapy in three different ways.
First, FF or DS may add to the effect that a drug has in reducing risk factors associated with certain conditions or diseases. For the example of statin therapy, statins reduce LDL-cholesterol by 18–55 % (mean absolute LDL-cholesterol reduction: 1·8 mmol/l)(5–Reference Law, Wald and Rudnicka8) and plant sterols and/or stanols and soluble dietary fibres are thought to reduce LDL-cholesterol levels even further when added to the statin treatment.
Second, certain FF or DS may improve risk factors associated with the condition, other than the risk factor that the drug is dealing with. In our example, statins are highly effective in lowering total and LDL-cholesterol, but statin monotherapy may not be sufficient to reach goals for TAG concentrations. Depending on the type of statin and its dose, TAG are lowered only by 7–30 %(5). Supplementing patients with n-3 PUFA will lower TAG and might improve statin therapy, since both cholesterol and TAG levels are lowered.
Third, FF or DS may be capable of reducing drug-associated side effects, for example, by restoring depleted compounds. With statin treatment, adverse events such as musculoskeletal complaints have been reported in 1–7 % of statin users(Reference Kiortsis, Filippatos and Mikhailidis9) and it has been hypothesised that statin-induced coenzyme Q10 (CoQ10) deficiency is involved in this. Supplementing CoQ10 might reduce musculoskeletal complaints. Besides, in patients who reach recommended goals for risk factors but experience side effects with drug use, combination therapy of the drug and a FF or DS might be an alternative with the potential of reducing the drug dose and as a result the side effects, while levels of risk factors remain constant. Subsequently, it is conceivable that patients experiencing fewer side effects will have a better adherence to drug treatment. Adherence might also be higher with the combination therapy of a FF or DS and a statin compared with a multidrug therapy, as patients might be more willing to slightly modify their diet by replacing normal food items with comparable FF, compared with taking another drug; patients' perception of overmedication has been found to correlate with self-report of decreased adherence(Reference Fincke, Miller and Spiro10). Other advantages of the combination therapy with FF or DS compared with multidrug therapy are the lower drug costs and the reduced risk for interactions and serious side effects(Reference Cannon11).
For the present review, we reviewed the data from clinical and observational studies that have investigated the effects of the use of FF or DS in patients on statin treatment. We selected four categories: FF or DS containing (1) plant sterols or stanols, (2) soluble dietary fibre, (3) n-3 PUFA, and (4) CoQ10. We investigated whether these FF or DS have been demonstrated to support statin therapy in one of the three ways described above.
The present review should not be viewed as comprehensive in covering all possible beneficial combination therapies of FF or DS and statins. Rather, the authors' intent is to focus on different mechanisms of action by which FF or DS may support statin treatment and to provide a full coverage of the literature of the examples of combination therapies given.
Literature search
Computerised searches for relevant articles in the PubMed electronic database were performed between March and August 2008, using Medical Subject Heading (MeSH) terms or text words combi*, supple* or interact* with statin*, antilipemic agents, anticholesteremic agents or hydroxymethylglutaryl CoA reductase inhibitors, and combined to one of the search items for the specific FF or DS as noted in Table 1. The search was limited to articles written in English or Dutch and studies performed in human subjects. Studies conducted in patients with medical conditions other than hyperlipidaemia, for example, cancer or diabetics, were excluded. Relevant articles were selected from the title and abstract. Moreover, additional articles were selected from citations in the publications found. Two authors of this report (S. E. and C. R.) independently reviewed the methodological quality of the included trials using the Jadad scoring system to evaluate the effect of study quality on the observed results. This validated scoring system assigns points for randomisation, double-blinding, and documentation of patient withdrawal, as well as additional points for the appropriateness of the randomisation and blinding methods(Reference Jadad, Moore and Carroll12). Trials scoring 3 points or above, out of a maximum of 5, are generally considered to be of good methodological quality. Discrepancies between the two authors were settled through discussion.
Table 1 Literature search
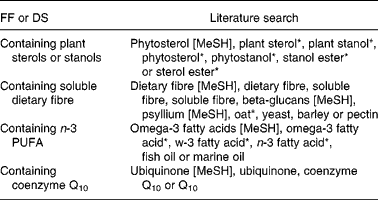
FF, functional food; DS, dietary supplement; MeSH, Medical Subject Heading.
In the following section we will first explain our current understanding of the mechanism of action by which the FF or DS may support statin treatment. Subsequently, the effects of the FF or DS in the healthy population and approved health claims will be discussed and we will summarise the results of clinical and observational studies exploring the combination therapy. Finally, safety aspects of the combination are addressed.
Plant sterols and stanols
Mechanism of supporting statin therapy
Plant sterols and stanols lower serum levels of total cholesterol and LDL-cholesterol through a different mechanism compared with statins. Whereas statins inhibit hepatic cholesterol synthesis, plant sterols and stanols reduce the intestinal absorption of cholesterol. Therefore it is thought that both mechanisms work simultaneously when statins and plant sterols and/or stanols are taken together. It is generally assumed that plant sterols and stanols compete with both dietary and biliary cholesterol for solubilisation into mixed micelles. Because plant sterols and stanols are more hydrophobic than cholesterol, they have a higher affinity for the micelle(Reference Plat and Mensink13, Reference Marinangeli, Varady and Jones14). Other mechanisms proposed are the interference with the cholesteryl ester-mediated hydrolysis process necessary for absorption, and/or stimulation of the ATP-binding cassette (ABC) transporter expression by plant sterols and stanols(Reference Plat and Mensink13, Reference Trautwein15–Reference de Jong, Plat and Mensink17). The ABCG5 and ABCG8 transporters actively transport dietary sterol out of the enterocytes back into the intestinal lumen, thereby limiting the amount of sterol absorbed. ABCA1 may also participate in this process(Reference Hegele and Robinson18). However, studies in plant sterol- and stanol-treated ABCA1- and ABCG5/G8-deficient mice have not demonstrated the involvement of these ABC transporters in the reduction of intestinal cholesterol absorption(Reference Calpe-Berdiel, Escolà-Gil and Blanco-Vaca19). Differences in ABCG5 and ABCG8 genes between humans and murines might (partly) explain these results(Reference Kidambi and Patel20).
Decreased cholesterol absorption is associated with a compensatory increase in cholesterol synthesis and an increase in LDL receptor expression. This elevated expression may not only lead to an increased clearance of LDL from the circulation, but also of intermediate-density lipoprotein (IDL). Because IDL is the precursor of LDL-cholesterol, this may ultimately lead to a decreased LDL production. The net result of the lower cholesterol absorption, higher LDL expression and higher endogenous cholesterol synthesis is a reduction in serum total and LDL-cholesterol concentration(Reference Plat and Mensink13, Reference de Jong, Plat and Mensink17) (Fig. 1).
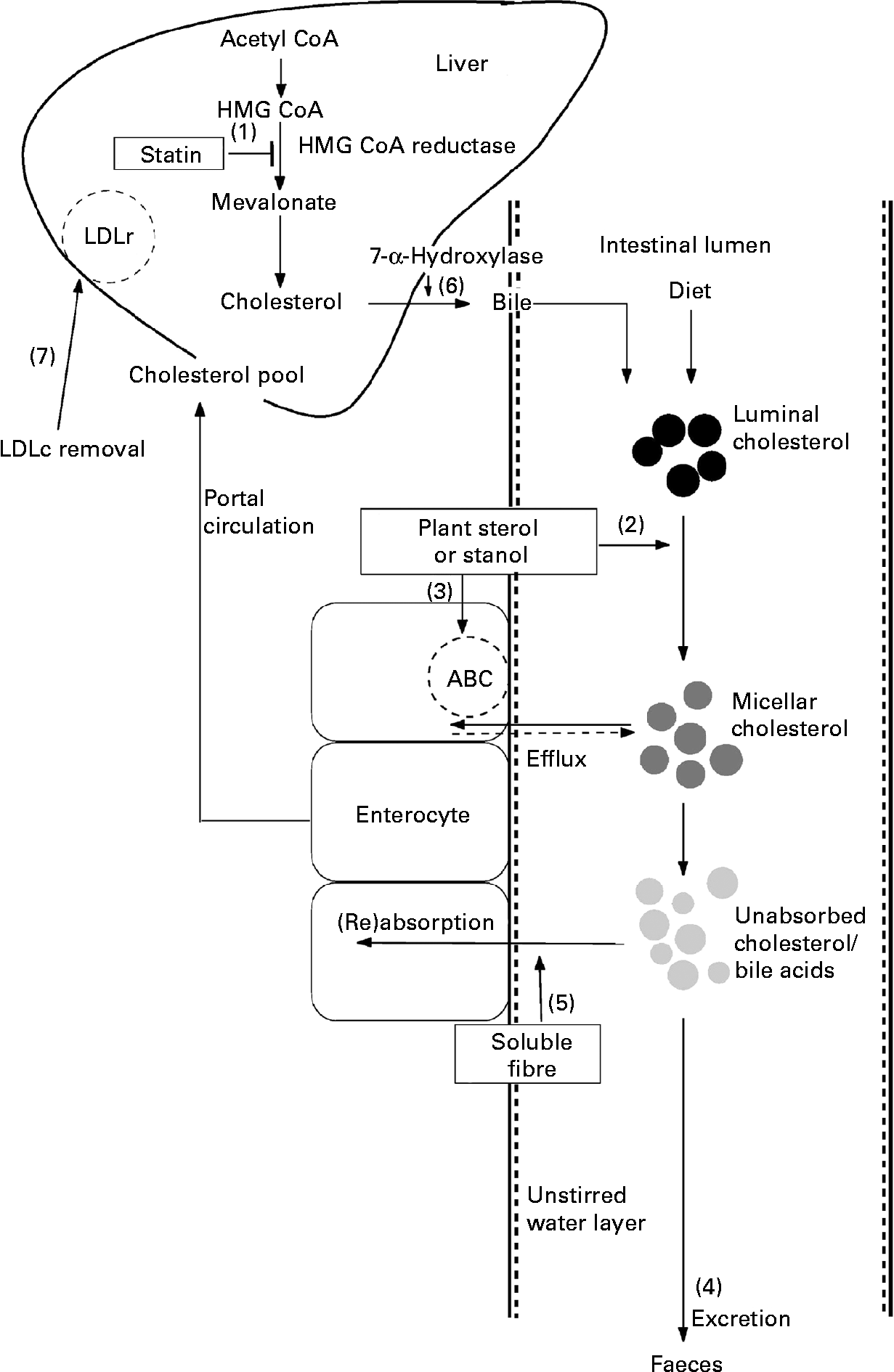
Fig. 1 Postulated cholesterol-lowering mechanisms of statins, plant sterols and stanols and soluble dietary fibre. Statins inhibit the enzyme hydroxymethylglutaryl CoA (HMG-CoA) reductase (1). Plant sterols and stanols compete with cholesterol for solubilisation into mixed micelles (2), leading to a reduced luminal absorption of cholesterol and/or they induce a higher expression of the ATP-binding cassette (ABC) transporter (3), resulting in an efflux of cholesterol back into the intestinal lumen. Both mechanisms lead to an increased faecal output (4)(Reference Plat and Mensink13, Reference de Jong, Plat and Mensink17). Soluble dietary fibre interrupts with cholesterol and/or bile acid (re)absorption (5), either by binding bile acids or by forming a thick unstirred water layer in the intestinal lumen, leading to an increased faecal output (4). Compensatory up-regulation of the enzyme cholesterol 7-α-hydroxylase (6) increases the conversion of cholesterol into bile acids. All processes will result in a reduction in the cholesterol content of liver cells what will lead to an up-regulation of LDL receptors (LDLr) and ultimately in an increased clearance of circulation LDL-cholesterol (LDLc) (7)(Reference Bell, Hectorn and Reynolds59, Reference Brown, Rosner and Willett66, Reference Theuwissen and Mensink68).
Estimated effects of plant sterols and stanols on lipid levels and health claims
Plant sterol and stanol esters have been incorporated in dairy products such as low-fat margarine, milk and yoghurt. Also cereals, bread and orange juice containing esterified or non-esterified plant sterols or stanols are available on the market. Since 2001, the Adult Treatment Panel of the US National Cholesterol Education Program has recommended the use of plant sterols or stanols (2 g/d) in conjunction with other lifestyle changes to enhance LDL-cholesterol reduction. The panel states that daily intake of 2–3 g plant sterol or stanol esters will reduce LDL-cholesterol by 6–15 %(5). Plant sterols and stanols do not have an effect on TAG or HDL-cholesterol levels(Reference Castro, Barroso and Sinnecker21, Reference de Graaf and Stalenhoef22). However, two recent meta-analyses evaluated the LDL-cholesterol-lowering effects of plant sterols and/or stanols. Both found that LDL-cholesterol reduction was approximately 0·33 mmol/l for a mean daily intake of 2·1–2·5 g plant sterols and/or stanols(Reference Abumweis, Barake and Jones23, Reference Demonty, Ras and van der Knaap24).
Plant sterols and stanols have approved health claims in the USA and in Europe. According to the United States Food and Drug Administration (FDA) there is significant scientific agreement for a consistent, clinically significant effect of plant sterols and stanols on blood total and LDL-cholesterol in both mildly and moderately hypercholesterolaemic (HC) populations. Therefore it has authorised the use of health claims on the association between plant sterol and stanol esters and reduced risk of CHD on food labels. The claims states that ‘Diets low in saturated fat and cholesterol that include two servings of foods that provide a daily total of at least 1·3 g of plant sterol esters or 3·4 g of plant stanol esters, may reduce the risk of heart disease’(25, 26). Based on the scientific evidence available at the time of evaluation, the FDA made a distinction between the amount of plant sterols and plant stanols necessary to lower total and LDL-cholesterol. However, in a clinical trial comparing the cholesterol-lowering efficacy of plant sterols and plant stanols, published shortly after the claim authorisation, no significant difference between esterified plant sterols and plant stanols was found(Reference Hallikainen, Sarkkinen and Gylling27).
Since January 2007, Regulation 1924/2006 applies to nutrition and health claims made in commercial communications in all European Union countries(28). The European Food Safety Authority (EFSA) was requested to evaluate scientific data on plant sterols and stanols in accordance with the Regulation and approved in 2008 health claims stating: ‘Plant sterols and plant stanol esters have been shown to lower/reduce blood cholesterol. Blood cholesterol lowering may reduce the risk of coronary heart disease’(29, 30). This advice has been provided to the European Commission and member states who will adopt and authorise the health claims(Reference Verhagen31).
As concerns safety, the Scientific Committee on Food has assessed plant sterol-enriched foods under the novel foods procedure (European Union Regulation 258/97)(32). They concluded that a maximum level of 8 % non-esterified plant sterols, consisting of 30–65 % β-sitosterol, 10–40 % campesterol, 6–30 % stigmasterol and a total of 5 % other plant sterols, is safe for human use, also stating that patients on cholesterol-lowering medication should only consume the enriched products under medical supervision(33). Plant stanols were not assessed through the novel foods procedure as these products were consumed in Finland already before 1997(Reference Verhagen, Te Boekhorst and Kamps34).
Effects of combination therapy with plant sterols and/or stanols and statins
Vanhanen(Reference Vanhanen35) was the first to conduct a clinical trial towards the effects of sitostanol esters on lipid levels in patients on pravastatin treatment. It was found that the daily addition of 1·5 g sitostanol ester did not lower serum total or LDL-cholesterol after 6 weeks of supplementation. In contrast, subsequent studies, using higher doses, all reported that plant sterols or stanols in combination with various statins have additive effects on total and LDL-cholesterol reduction in patients with (familial) hypercholesterolaemia (FH), as summarised in Tables 2–4.
Table 2 Clinical studies towards the effects on lipid levels (total, LDL- and HDL-cholesterol and TAG) of the combination therapy with statins and plant sterols or stanols: effects of plant sterols or stanols in statin users
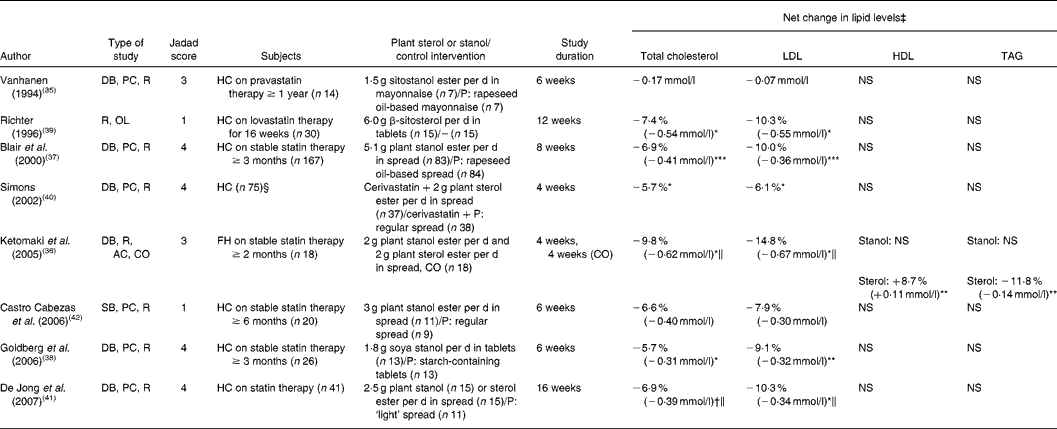
DB, double-blind; PC, placebo-controlled; R, randomised; HC, hypercholesterolaemic; P, placebo; OL, open-label; AC, active controlled; CO, cross-over; FH, familial hypercholesterolaemia; SB, single-blind.
* P < 0·05, ** P < 0·01, *** P < 0·001.
† Borderline significant.
‡ The net change in lipid levels was calculated by subtracting the mean change from baseline after control intervention from the mean change from baseline after plant sterol or stanol intervention, except for the study of Ketomaki et al. (Reference Ketomaki, Gylling and Miettinen36) where the net change is the mean change from baseline after plant sterol and stanol intervention.
§ The study of Simons(Reference Simons40) is a 2 × 2 factorial design study with four parallel arms. In this table the net change is calculated by subtracting the mean change from baseline after statin intervention from the mean change from baseline after combined intervention of plant sterols and statins.
∥ No significant difference between sterol and stanol esters.
Table 3 Clinical studies towards the effects on lipid levels (total, LDL- and HDL-cholesterol and TAG) of the combination therapy with statins and plant sterols or stanols: difference in effects of plant sterols or stanols between statin users and non-statin users

DB, double-blind; R, randomised; CHD, patients with coronary artery disease; OL, open-label; FH, familial hypercholesterolaemia; PC, placebo-controlled; HC, hypercholesterolaemic; P, placebo.
** P < 0·01, *** P < 0·001.
† The net change in lipid levels was calculated by subtracting the mean change from baseline after plant sterol or stanol intervention in the non-statin users from the mean change from baseline after plant sterol or stanol intervention in the statin users.
‡ The study of Simons(Reference Simons40) is a 2 × 2 factorial design study with four parallel arms. In this table the net change is calculated by subtracting the mean change from baseline after plant sterol intervention in patients on placebo drug from the mean change from baseline after plant sterol intervention in patients on cerivastatin.
§ In both groups significant reduction.
∥ Simvastatin-treated patients 7 weeks, not treated patients 12 weeks.
¶ Simvastatin-treated patients 6 weeks, not treated patients 12 weeks.
Table 4 Clinical studies towards the effects on lipid levels (total, LDL- and HDL-cholesterol and TAG) of the combination therapy with statins and plant sterols or stanols: effects of plant sterols or stanols in combined groups of statin users and non-statin users

DB, double-blind; PC, placebo-controlled; R, randomised; FH, familial hypercholesterolaemia; HC, hypercholesterolaemic; P, placebo; OL, open-label.
* P < 0·05, ** P < 0·01, *** P < 0·001.
† The net change in lipid levels was calculated by subtracting the mean change from baseline after control intervention from the mean change from baseline after plant sterol or stanol intervention in a combined group of statin users and non-statin users, except for the studies of Amundsen et al. (Reference Amundsen, Ntanios and Put47) and O'Neill et al. (Reference O'Neill, Brynes and Mandeno49) where the net change is the mean change from baseline after plant sterol or stanol intervention.
‡ No significant difference between sterol or stanol ester or between high- and low-dose stanol.
In Table 2, results of clinical studies are presented that investigated the effects of adding plant sterols or stanols, either in tablet form or incorporated into food products, on lipid levels in patients on (stable) statin treatment. In seven studies, using doses of plant sterols or stanols varying from 1·8 g/d to 6·0 g/d and with intervention periods between 4 and 16 weeks, effects were found ranging from a 6 to 10 % decrease for total cholesterol and from a 6 to 15 % decrease for LDL-cholesterol. Absolute reductions in total and LDL-cholesterol ranged from 0·31 to 0·62 mmol/l and from 0·30 to 0·67 mmol/l, respectively. The largest reductions were found in a cross-over trial conducted in patients with FH(Reference Ketomaki, Gylling and Miettinen36), although these reductions are probably partly caused by the low-fat spread as the results were not corrected for changes in a placebo-controlled group and no run-in period on placebo spread was used.
The results for total cholesterol were statistically significant for five out of seven studies(Reference Ketomaki, Gylling and Miettinen36–Reference Simons40), and either borderline significant (P = 0·052)(Reference De Jong, Plat and Bast41) or non-significant(Reference Castro Cabezas, de Vries and Van Oostrom42) for the two remaining studies. Reductions in LDL-cholesterol were not significantly different between the intervention and control group only in a single-blind study performed by Castro Cabezas et al. (Reference Castro Cabezas, de Vries and Van Oostrom42). This may have been due to the significant reduction in LDL-cholesterol in both the intervention and the control group, caused by the nutritional guidelines and low-fat margarines given to both groups, which may have made it more difficult to find significant differences in reductions between the two groups. The methodological quality of this clinical trial was poor based on components assessed by the Jadad scale. The majority of the studies did not find any significant effects of plant sterols or stanols on HDL-cholesterol or TAG, nor were the effects of plant sterols different compared with the effects of plant stanols. However, Ketomaki et al. found in a study consisting of two consecutive 4-week intervention periods with either a plant stanol ester or a plant sterol ester that only during the sterol ester period HDL-cholesterol increased and TAG levels decreased significantly(Reference Ketomaki, Gylling and Miettinen36). This study achieved a Jadad score of 3; no placebo-controlled group was included in this study, possibly leading to flawed results.
Table 3 shows the results of studies investigating the differences in effects that plant sterols or stanols have on lipid levels in statin users and non-statin users. All studies have demonstrated that if plant sterols or stanols are added to a statin, the effect on cholesterol reduction is similar(Reference Simons40, Reference Gylling, Radhakrishnan and Miettinen43) or even higher(Reference Vuorio, Gylling and Turtola44) compared with that observed with the use of the plant sterols or stanols alone.
De Jong et al. (Reference de Jong, Zuur and Wolfs45) and Wolfs et al. (Reference Wolfs, de Jong and Ocke46) also investigated the cholesterol-lowering effects of plant sterol- and stanol-enriched margarine (no differentiation between plant sterols and stanols) between statin users and non-statin users in a post-marketing surveillance setting over 5 years. These authors suggest that plant sterols and stanols have an additive effect to the drug, although significance levels were not reached because of the small number of combination users in the studies. Moreover, Simons(Reference Simons40) performed a 2 × 2 factorial study with four parallel treatment arms, aiming to distinguish between an additive effect and an interactive effect between plant sterol ester margarine and cerivastatin. Statistical analysis showed no evidence of an interactive effect and therefore the authors concluded that, although a small interaction between the two compounds could not be excluded, it is unlikely that this interaction is of any clinical importance.
In Table 4, the pooled results are given of studies not differentiating between statin users and non-statin users. Moreover, statin use was not quantified and cholesterol-lowering effects of plant sterols and stanols in normal, HC or FH patients were put together. All studies described significant reductions in total and LDL-cholesterol levels and suggested that the plant sterols and stanols were effective in both statin users as well as non-statin users(Reference Amundsen, Ntanios and Put47–Reference O'Neill, Brynes and Mandeno49).
In summary it can be concluded that plant sterol and plant stanol esters are an effective approach to lower cholesterol levels in addition to statin treatment in both HC and FH patients on statin treatment. Cholesterol lowering is at least equally effective in statin users compared with non-users. The addition of 2–5 g plant sterol or stanol esters per d to statins will result in an additive LDL- and total cholesterol reduction of roughly 10 (or 0·40 mmol/l) and 6 % (or 0·35 mmol/l) respectively, without significant changes in HDL-cholesterol or TAG levels. Effects in FH patients might even be slightly greater, although well-designed randomised double-blind trials are needed to confirm this hypothesis.
Differences in cholesterol-lowering effects of plant sterols or stanols between the different studies might be explained by baseline cholesterol levels, because it has been hypothesised that patients with high baseline cholesterol levels experience a larger reduction in cholesterol levels after sterol or stanol ester consumption(Reference Ketomaki, Gylling and Miettinen36). Moreover, it is suggested that patients with high ratios of serum cholestanol and plant sterols to cholesterol (markers for cholesterol absorption) may benefit the most from plant stanol or sterol intake(Reference Gylling and Miettinen50). A synergistic effect between statins and plant sterols and/or stanols should not be expected(Reference Katan, Grundy and Jones51).
Safety aspects of combination therapy with plant sterols and/or stanols and statins
In none of the studies were adverse effects related to the use of plant sterol- or stanol-enriched products in combination with statin therapy found. However, in studies towards the effects of plant sterols alone, it has been found that serum plant sterol concentration is elevated after consumption of plant sterols (unlike plant stanols) with potential atherogenic effects(Reference Patel and Thompson52). Normally, only 5–15 % of the plant sterols are absorbed in the intestinal tract(Reference Katan, Grundy and Jones51, Reference Heinemann, Axtmann and von Bergmann53). Patients with the rare autosomal recessive disease phytosterolaemia, however, are hyperabsorbers of plant sterols and should therefore not consume products containing high amounts of plant sterols, whether added to statin therapy or not. In healthy subjects, it is assumed that the beneficial effects on cholesterol levels of plant sterols outweigh any potential atherosclerotic risk, although additional research on this topic is urgently warranted(Reference Goldstein, Mascitelli and Pezzetta54, Reference John, Sorokin and Thompson55).
Furthermore, both plant sterols and stanols are associated with reductions in plasma concentrations of α-carotene, β-carotene, lycopene and α-tocopherol. Reductions in all vitamins, except for β-carotene, can be explained by reductions in LDL-cholesterol, the main lipoprotein carrier. Negative health effects related to these reductions are not expected(Reference Katan, Grundy and Jones51), although it might be a concern for groups with high nutritional needs such as elderly and pregnant women. These groups can be advised to add an extra amount of fruits and vegetables to the diet.
In addition, post-marketing surveillance of plant sterols and stanols upon request of the European Commission did not indicate adverse effects(Reference Lea and Hepburn56), thereby supporting the safety of these products(33).
Soluble dietary fibre
Mechanism of supporting statin therapy
Dietary fibres are associated with a reduced risk of CHD. Soluble fibre appears to be primarily responsible for the cholesterol-lowering effect of dietary fibre intake(Reference Van Horn, McCoin and Kris-Etherton57). Studies in HC patients without treatment with cardiovascular drugs showed that addition of soluble fibres (psyllium(Reference Anderson, Allgood and Lawrence58, Reference Bell, Hectorn and Reynolds59), β-glucan(Reference Chen, He and Wildman60–Reference Van Horn, Emidy and Liu63), guar gum(Reference Butt, Shahzadi and Sharif64, Reference Todd, Benfield and Goa65), pectin(Reference Brown, Rosner and Willett66)) to a low-fat, low-cholesterol diet was an effective approach to reduce total and LDL-cholesterol. The mechanisms involved are not completely understood, but it is suggested that soluble fibres reduce plasma cholesterol by interruption with cholesterol and/or bile acid (re)absorption(Reference Vergara-Jimenez, Furr and Fernandez67). Some authors suggest that soluble fibres bind bile acids; others assume that water-soluble fibres form a thick unstirred water layer in the intestinal lumen. Both proposed mechanisms will lead to an increased faecal output of bile acids, resulting in a reduction in bile acids available for transport back to the liver. Compensatory up-regulation of hepatic enzymes such as cholesterol 7-α-hydroxylase, the rate-limiting enzyme in bile acid biosynthesis, results in a reduction in the cholesterol content of liver cells. This leads to an up-regulation of the LDL receptors and the enzyme HMG-CoA reductase to re-establish hepatic cholesterol stores, ultimately resulting in an increased clearance of circulating LDL-cholesterol(Reference Bell, Hectorn and Reynolds59, Reference Brown, Rosner and Willett66, Reference Theuwissen and Mensink68) (Fig. 1). Other suggested mechanisms include the inhibition of cholesterol synthesis by SCFA (mainly propionate), which are the major fermentation products of soluble fibre, the increased intestinal viscosity causing lowered glucose absorption and thereby improving insulin sensitivity, and the increased satiety leading to lower overall energy intake(Reference Brown, Rosner and Willett66, Reference Theuwissen and Mensink68). These postulated mechanisms differ from the cholesterol-lowering mechanism of statins, and therefore both compounds may decrease cholesterol levels simultaneously.
Estimated effects of soluble dietary fibre on lipid levels and health claims
In a meta-analysis, it was estimated that 2–10 g soluble fibre per d significantly lowers total and LDL-cholesterol concentrations by 0·045 and 0·057 mmol/l, respectively(Reference Brown, Rosner and Willett66). Various soluble fibres, including oat products, psyllium, pectin and guar gum, reduce total and LDL-cholesterol by similar amounts; the effects depend on the food matrix used, the method of food processing and the concentration, water-solubility and molecular weight of the fibres.
In 1997 the FDA adopted health claims on the labels of foods containing β-glucan soluble fibre of whole oats noting that these foods, as part of a diet low in saturated fat and cholesterol, may reduce the risk of heart disease by reducing total and LDL-cholesterol. Since then, this claim was extended by adding psyllium seed husk, whole-grain barley products and barley β-fibre as additional eligible sources of soluble fibre. The FDA states that the food products must provide at least 0·75 g of β-glucan soluble fibre or 1·7 g of psyllium soluble fibre per serving(69, 70).
In Europe, member states such as Sweden, The Netherlands and UK have also approved claims linking oat soluble fibre consumption and reduced total and LDL-cholesterol(71–73). This has led to the introduction of several food products enriched with soluble fibre, including bread, cereals and cookies. However, in the context of Regulation 1924/2006, the claims currently used in the different member states need to be reviewed by the EFSA(28).
Effects of combination therapy with soluble dietary fibre and statins
Results of studies exploring the combination therapy with soluble dietary fibre and statins are shown in Tables 5 and 6. All studies scored less than 4 points on the Jadad scale.
Table 5 Clinical studies towards the effects on lipid levels (total, LDL- and HDL-cholesterol and TAG) of the combination therapy with statins and soluble dietary fibre: effects of soluble dietary fibre in statin users

OL, open-label; HC, hypercholesterolaemic; NM, not measured or calculated.
*** P < 0·001.
† The net change in lipid levels is the mean change from baseline after soluble dietary fibre intervention.
‡ Jadad score was not estimated because description of the study design has not been published (study was interrupted after three patients).
Table 6 Clinical studies towards the effects on lipid levels (total, LDL- and HDL-cholesterol and TAG) of the combination therapy with statins and soluble dietary fibre: difference in effects of a statin plus soluble dietary fibre v. a statin alone
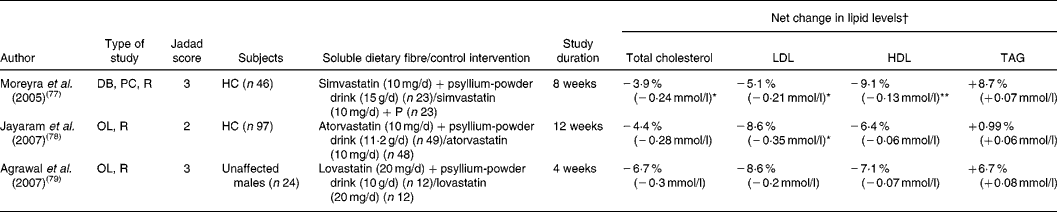
DB, double-blind; PC, placebo-controlled; R, randomised; HC, hypercholesterolaemic; P, placebo; OL, open-label.
* P < 0·05, ** P < 0·01.
† The net change in lipid levels was calculated by subtracting the mean change from baseline after control intervention from the mean change from baseline after soluble dietary fibre intervention.
Table 5 shows the effects on lipid levels of soluble fibre in statin users. One of the first studies performed towards this combination found that in three female HC patients, the addition of pectin daily to treatment with lovastatin resulted in an average rise in LDL-cholesterol of 42 %. After the intake of pectin was stopped, levels returned to normal. Also after the addition of oat bran to lovastatin the LDL-cholesterol levels rose strikingly in two patients in the same study. The authors concluded that both fibres might reduce the bioavailability of the statin(Reference Richter, Jacob and Schwandt74). Further studies towards this specific combination have not been performed, but Uusitupa et al. studied the effects of guar gum in a population of both FH and non-FH patients on lovastatin treatment and reported that adding guar gum resulted in significant reductions in serum total and LDL-cholesterol. However, the results might be flawed because no placebo group was included and no correction for food intake was made(Reference Uusitupa, Ebeling and Happonen75, Reference Uusitupa, Miettinen and Happonen76).
Table 6 presents the results of three studies comparing the effects on lipid values of the combination therapy of a statin and soluble dietary fibre v. statin treatment alone. Whereas total and LDL-cholesterol-lowering effects of the soluble fibres reached statistical significance in the studies performed by Moreya et al. (Reference Moreyra, Wilson and Koraym77) and Jayaram et al. (Reference Jayaram, Prasad and Sovani78), only trends towards an additive effect were observed by Agrawal et al. (Reference Agrawal, Tandon and Sharma79). It should be noted that this last trial was conducted in healthy adult men, and therefore results may differ from effects observed in the other studies performed in HC patients. Of note is that in two out of the three studies a blunting of the statin-associated increase in HDL-cholesterol was observed after addition of the soluble dietary fibre(Reference Moreyra, Wilson and Koraym77, Reference Agrawal, Tandon and Sharma79).
In conclusion we can say that studies towards the possible beneficial effects of soluble dietary fibre on statin therapy are scarce. Most clinical studies have reported negative associations between the use of soluble fibre supplements in combination with statins and LDL- or total cholesterol concentrations. However, also unfavourable reductions in statin bioavailability and reductions in HDL-cholesterol have been described after high intake of soluble fibre. At this moment, there is not sufficient evidence to recommend the use of FF or DS enriched with soluble fibres in patients using statins. Clinical studies are warranted to further elucidate the potentials of the combination therapy with soluble dietary fibre and statins. Research should focus on the effects of different sources of soluble fibre in combination with various statins on lipoprotein subclasses and drug bioavailability. Caution should be taken to interpret the direct effects of fibre supplements instead of possible accompanying effects of reduced dietary fat and cholesterol intake. Also studies investigating the mechanisms of combined action and a possible dose–response relationship between combination therapy and cholesterol levels are needed.
Safety aspects of combination therapy with soluble dietary fibre and statins
Soluble fibre supplementation is generally considered as well tolerated. Side effects observed are mostly related to the gastrointestinal tract, such as abdominal distention, flatulence and diarrhoea. Also some negative nutritional impacts of high soluble fibre intake have been reported, as soluble fibres may interact with vitamins and minerals, resulting in a lower bioavailability of these compounds. However, there are insufficient data to firmly draw conclusions about this matter. Most likely, the effect of the fibre depends on the type of mineral or vitamin, the intestinal transit time and the degree of bacterial fibre degradation in the gut(Reference Butt, Shahzadi and Sharif64, Reference Borel80, Reference Riedl, Linseisen and Hoffmann81).
The combination therapy with soluble fibre and statins may also have some safety limits, while unfavourable reductions in HDL-cholesterol have been described and in one study reduced statin absorption from the gut was suggested after a high intake of soluble fibre(Reference Richter, Jacob and Schwandt74). Studies towards the effects of soluble fibres on the bioavailability of statins and other drugs are scarce and results depend greatly upon the type of drug and fibre. Also the time of drug administration in relation to food intake may influence the bioavailability of the drug. Soluble fibres may influence the bioavailability of statins and other drugs by direct binding or by altering luminal pH, gastric emptying, intestinal transit, mucosal absorption and metabolism of the drug(Reference Anderson, Allgood and Lawrence58, Reference Garcia, Fernandez and Diez82).
n-3 PUFA
Mechanism of supporting statin therapy
In recent years a lot of research has been performed towards the association between intake of n-3 PUFA and reduction in CHD. n-3 PUFA operate via several mechanisms. One of the most important is the favourable effect of n-3 PUFA on VLDL-cholesterol and TAG levels; in a meta-analysis of seventeen population-based prospective studies it was estimated that after adjustment for other risk factors, a 1 mmol/l increase in serum TAG is associated with a 14 % increase in CVD risk in men and 37 % in women(Reference Hokanson and Austin83). Statins efficiently reduce total and LDL-cholesterol, but have only limited TAG-lowering effects; thus a combined intake of n-3 PUFA and a statin might be beneficial in improving the lipid profile in patients with high TAG levels. The favourable decrease in TAG levels caused by n-3 PUFA is probably due to reduced hepatic VLDL and TAG synthesis and secretion, and enhanced TAG clearance from chylomicrons and VLDL particles. Reduced synthesis might be due to increased rates of mitochondrial and/or peroxisomal β-oxidation or a decreased expression of sterol regulatory element-binding protein-1c, a transcription factor involved in the regulation of fatty acid-synthesising enzymes. Both mechanisms will result in a reduction in the availability of the substrate, i.e. fatty acids. Increased clearance is possibly caused by increased lipoprotein lipase activity due to increased PPAR-γ and/or PPAR-α gene expression. Activation of PPAR leads to increased fatty acid β-oxidation in the liver and skeletal muscle(Reference Harris, Miller and Tighe84–Reference Harris and Bulchandani86).
Other mechanisms by which n-3 PUFA may lower the risk of CHD include reductions in platelet aggregation, blood viscosity and ischaemia and their anti-thrombotic, fibrinolytic and anti-inflammatory activities. Moreover, n-3 PUFA appear to play an important role in the prevention of arrhythmias(Reference Connor and Connor87, Reference Demaison and Moreau88).
Estimated effects of n-3 PUFA on lipid levels and health claims
In a recent meta-analysis of twenty-one randomised controlled trials it was estimated that n-3 PUFA consumption resulted in significant changes in TAG of − 0·31 mmol/l, in HDL-cholesterol of+0·04 mmol/l and in LDL-cholesterol of+0·16 mmol/l. There was no effect on total cholesterol(Reference Balk, Lichtenstein and Chung89). It has been suggested that the unfavourable increase in LDL-cholesterol is attributable to the increased conversion of VLDL to IDL and LDL, and the conversion of IDL to LDL after n-3 PUFA supplementation(Reference Harris90, Reference McKenney and Sica91).
In September 2004 the FDA announced a qualified health claim for the use of food products containing both EPA and DHA n-3 PUFA for FF and DS(92). According to the FDA there is supportive, but not conclusive, scientific evidence that suggests a reduction in CHD as a result of eating food or supplements rich in n-3 PUFA. The FDA judges that n-3 PUFA generally reduce TAG and VLDL-cholesterol, and have no effect on total or HDL-cholesterol in both general and diseased populations. The EFSA has not yet evaluated health claims on n-3 PUFA and cardiovascular function.
Effects of combination therapy with n-3 PUFA and statins
Results of clinical studies that have investigated the combination therapy with n-3 PUFA and statins are summarised in Tables 7 and 8. Contacos et al. were the first to demonstrate a beneficial effect of the combination of n-3 PUFA and statin therapy in HC patients(Reference Contacos, Barter and Sullivan93). They found that in patients randomised to either pravastatin, n-3 PUFA or placebo for 6 weeks, an additional 12 weeks of combination therapy with n-3 PUFA and pravastatin further decreased plasma TAG and LDL-cholesterol by 33 % (P < 0·05) and 26 % (P < 0·01), respectively, in patients in the placebo group, whereas in patients already on pravastatin only TAG levels were non-significantly decreased by 33 % and in patients in the n-3 PUFA group only LDL-cholesterol levels were decreased by 24 % (P < 0·05). Total cholesterol levels showed similar changes to LDL-cholesterol after combination therapy. This study indeed showed that statins particularly lowered total and LDL-cholesterol, whereas n-3 PUFA lowered TAG and not cholesterol levels. Combination therapy reduced both cholesterol and TAG concentrations. These beneficial effects of n-3 PUFA on TAG levels have been confirmed in later studies(Reference Chan, Watts and Barrett94–Reference Yokoyama, Origasa and Matsuzaki104).
Table 7 Clinical studies towards the effects on lipid levels (total, LDL- and HDL-cholesterol and TAG) of the combination therapy with statins and n-3 PUFA: effects of n-3 PUFA in statin users
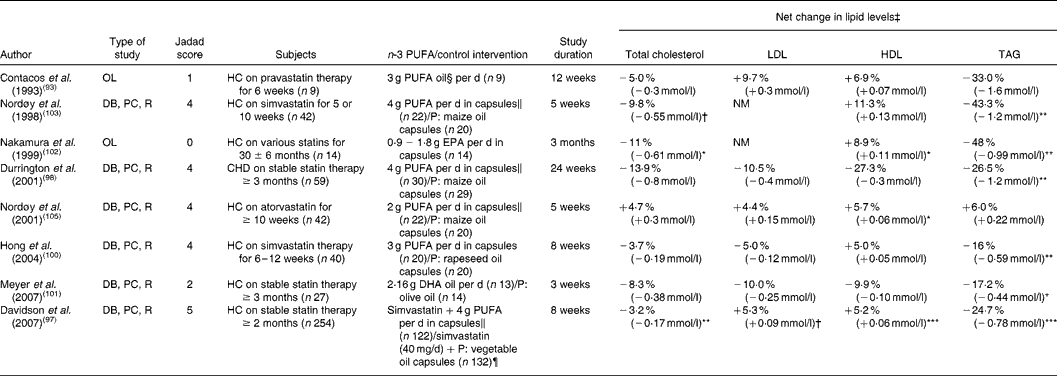
OL, open-label; HC, hypercholesterolaemic; DB, double-blind; PC, placebo-controlled; R, randomised; P, placebo; NM, not measured or calculated; CHD, patients with coronary artery disease.
* P < 0·05, ** P < 0·01, *** P < 0·001.
† Borderline significant.
‡ The net change in lipid levels was calculated by subtracting the mean change from baseline after control intervention from the mean change from baseline after n-3 PUFA intervention, except for the studies of Contacos et al. (Reference Contacos, Barter and Sullivan93) and Nakamura et al. (Reference Nakamura, Hamazaki and Ohta102) where the net change is the mean change from baseline after n-3 intervention.
§ EPA 67 %, DHA 33 %.
∥ EPA 45–48 %, DHA 36–39 %.
¶ At inclusion simvastatin replaced any previous statin.
Table 8 Clinical studies towards the effects on lipid levels (total, LDL- and HDL-cholesterol and TAG) of the combination therapy with statins and n-3 PUFA: difference in effects of a statin plus n-3 PUFA v. a statin alone

DB, double-blind; PC, placebo-controlled; R, randomised; HC, hypercholesterolaemic; P, placebo; OL, open-label; IR, insulin-resistant.
* P < 0·05, ** P < 0·01, *** P < 0·001.
† Borderline significant.
‡ The net change in lipid levels was calculated by subtracting the mean change from baseline after control intervention from the mean change from baseline after n-3 PUFA intervention.
§ EPA 60 %, DHA 40 %.
∥ EPA 45–48 %, DHA 36–39 %.
¶ Values are non-HDL-cholesterol.
†† All patients received the same therapeutic protocol consisting of 4 weeks of diet, 8 weeks of diet+statin, 4 weeks of diet, 8 weeks of diet+statin+PUFA.
Table 7 shows the results of studies examining the effects of supplementing patients on statin therapy with n-3 PUFA(Reference Contacos, Barter and Sullivan93, Reference Davidson, Stein and Bays97, Reference Durrington, Bhatnagar and Mackness98, Reference Hong, Xu and Pang100–Reference Nordøy, Bønaa and Nilsen103, Reference Nordøy, Hansen and Brox105). All studies used EPA and/or DHA, in doses varying from 0·9 to 1·8 g/d and 0·78 to 2·16 g/d for EPA and DHA, respectively. All studies found significant reductions in TAG, ranging from 16 (or 0·44 mmol/l) to 48 % (or 1·2 mmol/l), after supplementing n-3 PUFA, except one study performed by Nordøy et al. in which no TAG-lowering effect was attributable to the n-3 PUFA(Reference Nordøy, Hansen and Brox105). In this study relatively low doses of n-3 PUFA (0·9 g/d EPA, 0·78 g/d DHA) were used, which could explain these results. However, one small, uncontrolled study (Jadad score = 0) in which twelve patients were supplemented with 0·9 g EPA per d and two patients with 1·8 g EPA per d showed highly significant reductions in TAG. In addition, in this study it was found that total cholesterol levels were significantly reduced and HDL-cholesterol was significantly increased after EPA supplementation(Reference Nakamura, Hamazaki and Ohta102). Most studies performed in patients on statin therapy did not find any significant changes in total, LDL- or HDL-cholesterol, although in some studies VLDL-cholesterol was decreased(Reference Contacos, Barter and Sullivan93, Reference Durrington, Bhatnagar and Mackness98, Reference Meyer, Hammervold and Rustan101). In the COMBOS (COMBination of prescription Omega-3 with Simvastatin) study(Reference Davidson, Stein and Bays97), administration of n-3-acid ethyl esters plus simvastatin improved, besides TAG levels, also total, HDL- and VLDL-cholesterol to a greater extent than simvastatin alone. On the unfavourable side, a trend was observed towards a greater reduction in LDL-cholesterol in the simvastatin-only group (0·7 v. − 2·8 %; P = 0·052).
Table 8 shows the results of studies comparing the effects on lipid values of a combination therapy of a statin and n-3 PUFA v. statin treatment alone. Davidson et al. (Reference Davidson, Macariola-Coad and McDonald96) found that after treating HC patients with n-3 PUFA and/or simvastatin for 12 weeks, the TAG responses were similar in the EPA/DHA-group ( − 25·3 %) and the combined group ( − 28·8 %), and borderline significantly lower in the simvastatin group ( − 18·5 %), whereas decreases in non-HDL-cholesterol and increases in HDL-cholesterol were statistically significant only for the combined (non-HDL: − 24·8 %, HDL: +10·4 %) and simvastatin group (non-HDL: − 25·8 %, HDL: +7·2 %). All other studies found significant improvements of TAG with a combination therapy compared with the statin therapy alone(Reference Chan, Watts and Barrett94, Reference Chan, Watts and Mori95, Reference Davidson, Stein and Bays97, Reference Grekas, Kassimatis and Makedou99, Reference Yokoyama, Origasa and Matsuzaki104). Study populations included, besides HC patients, renal transplant patients with persistent hypercholesterolaemia(Reference Grekas, Kassimatis and Makedou99) and insulin-resistant obese men with dyslipidaemia(Reference Chan, Watts and Mori95). In this last study, also atorvastatin alone significantly decreased TAG levels. The authors suggest that the two compounds reduce TAG levels through different mechanisms. Whereas n-3 PUFA reduced the hepatic secretion of VLDL-apoB, atorvastatin enhanced the clearance of all apo B-containing lipoproteins, resulting in an additive effect(Reference Chan, Watts and Barrett94, Reference Chan, Watts and Mori95). Aligeti et al. performed a retrospective cohort study in which they compared the change in plasma TAG levels between patients taking fish oil as monotherapy and patients who added fish oil to their usual lipid-lowering drugs, including statins(Reference Aligeti, Gandhi and Braden106). They found that adding fish oil to a statin alone or to multiple lipid-lowering drugs (combination of niacin, statin and/or fibrates) did not alter the TAG-lowering effects of fish oil and effects are therefore additive.
One study compared the effects of the combination therapy and statins as monotherapy on clinical endpoints and found in the combined group a statistically significant 19 % relative reduction in major coronary events, particularly unstable angina and non-fatal coronary events. This applied in both patients taking statins for primary prevention as for secondary prevention(Reference Yokoyama, Origasa and Matsuzaki104). Few studies have examined the effects of combined treatment of n-3 PUFA and statins in patients with FH. Sandset et al. found no additional effects of adding n-3 PUFA (4 g/d for 6 weeks) to simvastatin (40 mg/d) in a small uncontrolled study (n 13); TAG even tended to increase on additional n-3 PUFA(Reference Sandset, Lund and Norseth107). Also in another small study (n 14) in FH patients on chronic simvastatin treatment, TAG were not significantly decreased after n-3 PUFA supplementation (5·1 g/d)(Reference Balestrieri, Maffi and Sleiman108). Small sample sizes or the population under study may explain these results.
In conclusion we can say that all clinical studies conducted in HC patients suggest that after combined intake of n-3 PUFA and statins no diminution of the separate effects of the compounds is expected, but that they improve lipid levels simultaneously through different mechanisms. Whereas statins alone have little effect on TAG levels, adding n-3 PUFA to the statin regimen lowered TAG significantly in most of the studies. Higher doses of n-3 PUFA and higher baseline TAG levels appear to be associated with greater reductions. In some, but not all studies, HDL-cholesterol was significantly increased and VLDL-cholesterol was significantly decreased after supplementation with n-3 PUFA. None of the studies found a significant favourable effect of n-3 PUFA on LDL-cholesterol and in some studies LDL-cholesterol even tended to increase after n-3 PUFA supplementation, contributing to the hypothesis that n-3 PUFA increase the conversion of VLDL to LDL. Effects of combined treatment with n-3 PUFA and statins in FH patients are less clear and studies examining these effects in larger populations are warranted.
Safety aspects of combination therapy with n-3 PUFA and statins
In none of the studies were adverse effects seen after combination therapy of n-3 PUFA and statins other than the adverse effects caused by monotherapy of the compound. n-3 PUFA were usually well tolerated and serious events have not been observed. Potential adverse effects related to n-3 PUFA include an increased bleeding time because of interference with platelet function, gastrointestinal disturbances and increases in LDL-cholesterol(Reference Davidson, Stein and Bays97, Reference Bays109–Reference Yang and Kenny111).
Coenzyme Q10
Mechanism of supporting statin therapy
Statins act by inhibiting the activity of HMG-CoA reductase, the rate-limiting enzyme in cholesterol biosynthesis that catalyses the conversion of HMG-CoA to mevalonate. Besides being an intermediate in cholesterol synthesis, mevalonate is also a precursor of CoQ10; statins thus lower CoQ10 levels(Reference Littarru and Langsjoen112, Reference Nawarskas113) (Fig. 2). CoQ10 is known for its enzymic role in the production of energy within human cells, so CoQ10 deficiency may impair muscle energy metabolism and contribute to the development of myalgia, a frequently reported adverse effect of statin treatment(Reference Caso, Kelly and McNurlan114). Supplementation with CoQ10 can raise the circulating levels of CoQ10 and might therefore be efficient in alleviating myopathic symptoms.
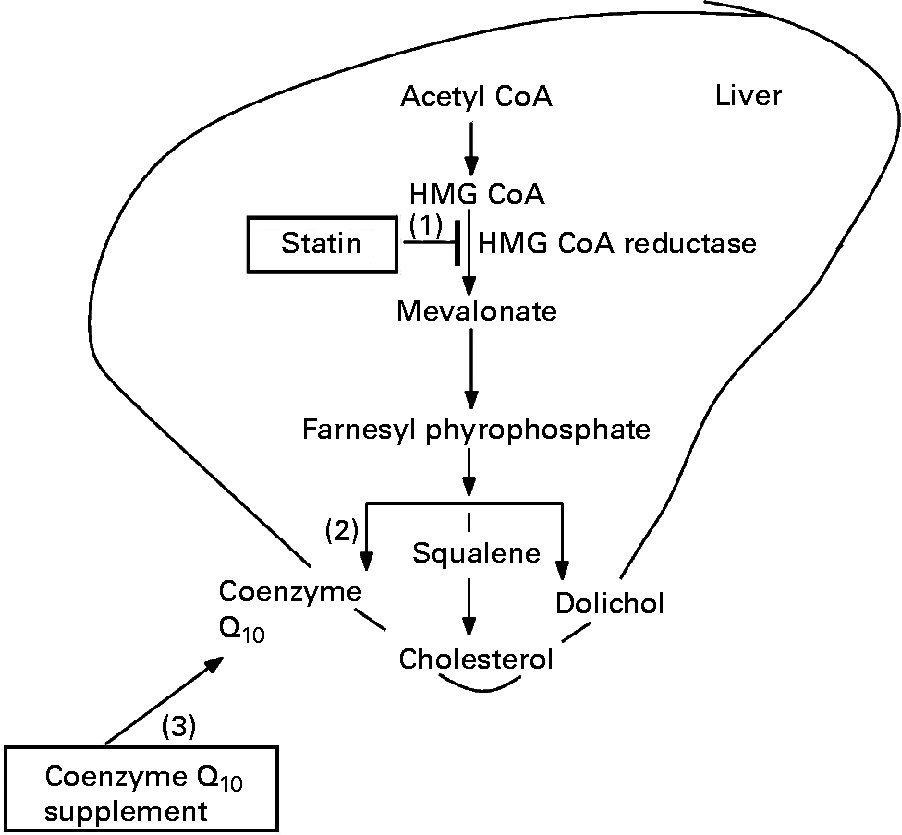
Fig. 2 Proposed mechanism by which coenzyme Q10 supplements may support statin therapy. Statins inhibit the enzyme hydroxymethylglutaryl CoA (HMG-CoA) reductase (1) in the mevalonate pathway. The same pathway is shared by coenzyme Q10 and as a consequence coenzyme Q10 synthesis is inhibited (2)(Reference Littarru and Langsjoen112, Reference Nawarskas113). Coenzyme Q10 supplements may raise the levels of coenzyme Q10 (3) in plasma and platelets.
Coenzyme Q10 and health claims
Dietary supplements containing CoQ10 have not been evaluated for safety and effectiveness and there are no approved health claims for the use of CoQ10, neither in the USA nor in Europe.
Effects of combination therapy with coenzyme Q10 and statins
Although studies have repeatedly demonstrated reduced levels of plasma CoQ10 with statin therapy(Reference Nawarskas113, Reference Levy and Kohlhaas115–Reference Pepe, Marasco and Haas117) and restored levels after oral CoQ10 supplementation(Reference Nawarskas113, Reference Bargossi, Grossi and Fiorella118), large randomised controlled trials towards the impact of CoQ10 supplementation on statin-induced myalgia in patients with hyperlipidaemia are lacking. Only two double-blind controlled clinical trials investigating this area have been performed. The first study, assigned a Jadad score of 2, was a pilot study in forty-four patients with self-reported myalgia. Patients were randomised to supplementation with 200 mg CoQ10 per d or placebo in combination with an upward dose of simvastatin (starting dose of 10 or 20 mg/d up to 40 mg/d) for 12 weeks. Results showed no difference between the groups in severity of myalgia, in the number of patients tolerating the highest dose of simvastatin, or in the number of patients remaining on therapy(Reference Young, Florkowski and Molyneux119). The second study, assigned a Jadad score of 4, was performed in thirty-two patients with myopathic symptoms taking varying doses of statins, and supplemented with CoQ10 (100 mg/d) or vitamin E (400 IU/d) for 30 d(Reference Caso, Kelly and McNurlan114). This study showed a significant 40 and 38 % reduction in pain severity and pain interference with daily activities, respectively, in the group treated with CoQ10. Vitamin E did not affect pain severity or pain interference. In this study, the benefit of CoQ10 supplementation on improving pain was not stratified by statin type or dose.
In a third trial towards the effects of CoQ10 supplementation on statin-induced myopathic symptoms, statin therapy was discontinued upon initial visit in all patients and no control group was included, so it is not clear what role CoQ10 had in decreasing the incidence of myalgia(Reference Langsjoen, Langsjoen and Langsjoen120).
In summary it can be concluded that although some trial evidence exists about the effectiveness of CoQ10 supplementation on myopathic symptoms, it is too early to recommend its routine use in clinical practice. The only randomised controlled clinical trials investigating this area showed contrasting results and further well-performed clinical trials are needed to investigate whether CoQ10 can be used to support statin therapy. Although several studies have shown that plasma CoQ10 levels are decreased after statin therapy, existing evidence also suggests that skeletal muscle CoQ10 levels are not affected or even increased after statins(Reference Nawarskas113, Reference Levy and Kohlhaas115, Reference Marcoff and Thompson116).
Alternative explanations for the myotoxic adverse effects of statins include instability of skeletal muscle cells due to reduction in cholesterol content of the membranes and inhibited production of GTP-binding proteins involved in cell growth and apoptosis. Apoptosis is a critical mechanism in the remodelling and maintenance of tissue structure and inappropriate apoptosis can produce pathological conditions(Reference Thompson, Clarkson and Karas121, Reference Ucar, Mjorndal and Dahlqvist122). Some of the decrease in CoQ10 can probably be explained by the reduction in LDL-cholesterol levels after statin therapy, since CoQ10 is transported in the LDL particle(Reference Thompson, Clarkson and Karas121).
Safety aspects of combination therapy with coenzyme Q10 and statins
CoQ10 is widely recognised as safe with no reported toxicity(Reference Hidaka, Fujii and Funahashi123). It has been shown that CoQ10 supplementation (100 mg/d) in HC patients treated with atorvastatin (10 mg/d) did not have an effect on statin-induced reductions in total or LDL-cholesterol, or TAG levels(Reference Mabuchi, Nohara and Kobayashi124).
Discussion
The main objective of this review was to present options for the support of drug therapy with FF or DS. We focused on the support of statin therapy with plant sterols and stanols, soluble fibre, n-3 PUFA or CoQ10, because many subjects are treated suboptimally with statins and there are indications supporting combined use with one of these FF or DS.
There is substantial evidence that adding plant sterols or stanols to statin therapy reduces total and LDL-cholesterol, and that adding n-3 PUFA to statins reduces plasma TAG. Both combination treatments are without any changes in HDL-cholesterol. Neither supplementation of plant sterols or stanols nor supplementation with n-3 PUFA had any known clinical significant side effects, although n-3 PUFA supplementation tended to increase LDL-cholesterol and plant sterol and stanol supplementation is associated with a reduction of β-carotene. Also the potential atherogenicity of elevated serum plant sterol concentrations needs to be further investigated.
Information about the combination therapy with either soluble dietary fibres or CoQ10 and statins is less clear. Soluble dietary fibre and statins may have additive effects on reducing total and LDL-cholesterol levels. However, also an antagonistic effect of soluble fibre supplementation on statin therapy might be expected due to a reduced drug bioavailability. Furthermore, soluble fibre supplementation has been associated with a blunting of the HDL-cholesterol-increasing effect of statins. CoQ10 may counteract the adverse myalgic effect produced by statins, but further studies are needed to confirm this hypothesis. Despite the safety and low costs of CoQ10, thus far it should not be recommended as a routine supplement with statin therapy in clinical practice. In the present review we discussed the (limited) available literature on the effectiveness of CoQ10 supplementation in reducing myopathic symptoms. Also other functional foods or dietary supplements might be helpful in reducing statin-induced side effects. Se supplementation has been suggested to reduce both statin-induced liver injury(Reference Kromer and Moosmann125, Reference Moosmann and Behl126) and myotoxicity(Reference Moosmann and Behl126), and l-carnitine might improve statin-associated myotoxicity(Reference Arduini, Peschechera and Giannessi127). However, current research is limited to cell-culture and animal experiments, and human studies should be performed to assess the potential protective effects of these compounds in man. In the present review we have limited our literature search to human studies.
In conclusion it can be stated that using FF or DS might be an effective and safe approach to support drug therapy, especially when drugs alone are insufficient to achieve desirable effects on risk factors or when drug use is associated with side effects. In our example, FF or DS fortified with plant sterols or stanols or n-3 PUFA are a good option for supporting statin therapy. However, every combination of a drug and a FF or DS has to be investigated separately to draw conclusions about the type of effect: additive, synergistic, antagonistic or no effect. In our example of statin therapy, it is possible that various statins have different effects when combined to FF or DS, as statins vary in intestinal absorption and bioavailability. Also studies towards the effects of genetic polymorphisms are warranted as indicated by, for example, the association between variants in SLCO1B1 (solute carrier organic anion transporter family, member 1B1) and increased risk of statin-induced myopathy(Reference Link, Parish and Armitage128), the association between ABCA1 expression and cholesterol absorption after intake of plant stanols(Reference Plat and Mensink16), and the association between polymorphisms in the fatty acid desaturase (FADS) genes and fatty acid concentrations in plasma and erythrocyte membranes(Reference Schaeffer, Gohlke and Muller129–Reference Xie and Innis131, Reference Manolio132).
More research is needed towards the effect that a FF or DS has on side effects caused by drugs, and whether side effects can be reduced by replacing some dose of the drugs with FF or DS, without altering the effects on risk factors. Post-marketing surveillance studies are required to assess the long-term safety of the combination therapies and the safety in specific risk groups; clinical trials do often not attain adequate power for evaluating rare events and interactions. Moreover, the effectiveness of the combination therapies under customary conditions should be addressed as adherence to drugs is known to be suboptimal(Reference Herings, Leufkens and Heerdink133, Reference Mantel-Teeuwisse, Goettsch and Klungel134) and recommended doses of FF and DS might not be consumed(Reference de Jong, Zuur and Wolfs45, Reference Wolfs, de Jong and Ocke46, Reference De Jong, Ros and Ocke135).
Acknowledgements
The present review was funded by a grant from the National Institute for Public Health and the Environment (RIVM).
It was written by S. E., with O. K., J. G., H. V., H. vK., H. vL. and C. R. all providing comments. All authors reviewed the contents of the paper, approved its contents and validated the accuracy of the data.
The division of Pharmacoepidemiology and Pharmacotherapy employing authors S. E. and O. K. received unrestricted funding for pharmacoepidemiological research from GlaxoSmithKline, Novo Nordisk, the private–public funded Top Institute Pharma (www.tipharma.nl, includes co-funding from universities, government and industry), the Dutch Medicines Evaluation Board and the Dutch Ministry of Health.
J. G., H. V., H. vK., H. vL. and C. R. have no conflicts of interest to disclose.












