22 results
Effects of Long-Term Deutetrabenazine Treatment in Patients with Tardive Dyskinesia and Underlying Psychiatric or Mood Disorders
- Robert A. Hauser, Hadas Barkay, Hubert H. Fernandez, Stewart A. Factor, Joohi Jimenez-Shahed, Nicholas Gross, Leslie Marinelli, Amanda Wilhelm, Mark Forrest Gordon, Juha-Matti Savola, Karen E. Anderson
-
- Journal:
- CNS Spectrums / Volume 27 / Issue 2 / April 2022
- Published online by Cambridge University Press:
- 28 April 2022, pp. 245-246
-
- Article
-
- You have access Access
- Export citation
-
Introduction
Deutetrabenazine is FDA-approved for the treatment of tardive dyskinesia (TD) in adults. In two 12-week pivotal trials (ARM-TD/AIM-TD), deutetrabenazine significantly improved Abnormal Involuntary Movement Scale (AIMS) scores and was well-tolerated. This post hoc analysis examined the efficacy and safety of long-term deutetrabenazine treatment in TD patients with comorbid psychiatric illness, including schizophrenia/schizoaffective disorder and mood disorders (bipolar/depression/other).
MethodsPatients who completed ARM-TD or AIM-TD enrolled in the 3-year, open-label extension (OLE) study. Deutetrabenazine was titrated based on dyskinesia control and tolerability. Change from baseline in total motor AIMS score, Patient Global Impression of Change (PGIC), Clinical Global Impression of Change (CGIC), and adverse events (AEs) were analyzed in subgroups by comorbid psychiatric illness.
ResultsA total of 337 patients in the OLE study were included in the analysis: 205 patients with schizophrenia/schizoaffective disorder (mean age, 55 years; 50% male; 6.4 years since diagnosis; 92% taking DRA) and 131 patients with mood disorders (mean age, 60 years; 35% male; 4.6 years since diagnosis; 50% taking DRA). At week 145, mean ± SE dose was 40.4 ± 1.1 mg/day for schizophrenia/schizoaffective disorder (n = 88) and 38.5 ± 1.2 mg/day for mood disorders (n = 72). Mean ± SE change from baseline in AIMS score at week 145 was −6.3 ± 0.49 and −7.1 ± 0.58, 56% and 72% achieved PGIC treatment success, and 66% and 82% achieved CGIC treatment success in schizophrenia/schizoaffective disorder and mood disorder patients, respectively. Overall AE incidence (exposure-adjusted incidence rates [incidence/patient-years]) was low: any, 1.02 and 1.71; serious, 0.10 and 0.12; leading to discontinuation, 0.07 and 0.05).
ConclusionLong-term deutetrabenazine treatment provided clinically meaningful improvements in TD-related movements, with a favorable safety profile, regardless of underlying comorbid psychiatric illness.
FundingTeva Pharmaceutical Industries Ltd., Petach Tikva, Israel
Long-Term Efficacy and Safety of Deutetrabenazine in Patients with Tardive Dyskinesia by Concomitant Dopamine-Receptor Antagonist Use
- Robert A. Hauser, Hadas Barkay, Hubert H. Fernandez, Stewart A. Factor, Joohi Jimenez-Shahed, Nicholas Gross, Leslie Marinelli, Amanda Wilhelm, Mark Forrest Gordon, Juha-Matti Savola, Karen E. Anderson
-
- Journal:
- CNS Spectrums / Volume 27 / Issue 2 / April 2022
- Published online by Cambridge University Press:
- 28 April 2022, p. 246
-
- Article
-
- You have access Access
- Export citation
-
Introduction
Tardive dyskinesia (TD) is an involuntary movement disorder that can result from exposure to dopamine-receptor antagonists (DRAs). Deutetrabenazine demonstrated significant improvements in Abnormal Involuntary Movement Scale (AIMS) scores in the 12-week pivotal trials (ARM-TD/AIM-TD). This post hoc analysis assessed the long-term efficacy and safety of deutetrabenazine by baseline DRA use.
MethodsPatients who completed ARM-TD or AIM-TD enrolled in the 3-year, open-label extension (OLE) study, with deutetrabenazine dose titrated based on dyskinesia control and tolerability. Change from baseline in total motor AIMS score, Patient Global Impression of Change (PGIC), Clinical Global Impression of Change (CGIC), and adverse event (AE) rates were analyzed in subgroups by baseline DRA use.
ResultsOf 337 patients in the OLE study, 254 were taking DRAs at baseline (mean age, 56 years; 48% male; 6.0 years since diagnosis) and 83 were not (mean age, 60 years; 31% male; 4.9 years since diagnosis). Mean ± SE dose at week 145 was 39.9 ± 1.0 mg/day in patients taking DRAs (n = 108) and 38.5 ± 1.5 mg/day in patients not taking DRAs (n = 53). At week 145, mean ± SE change from baseline in AIMS score was −6.1 ± 0.43 and −7.5 ± 0.71; 64% and 62% achieved PGIC treatment success; and 69% and 81% achieved CGIC treatment success, respectively. Overall AE incidence was low (exposure-adjusted incidence rates [incidence/patient-years]: any, 1.08 and 1.97; serious, 0.10 and 0.12; leading to discontinuation, 0.06 and 0.05).
ConclusionThis analysis suggests that deutetrabenazine for long-term treatment of TD is beneficial, with a favorable safety profile, regardless of concomitant DRA use.
FundingTeva Pharmaceutical Industries Ltd., Petach Tikva, Israel
Effect of Deutetrabenazine on Metabolic Parameters in the Treatment of Tardive Dyskinesia
- Joohi Jimenez-Shahed, Hadas Barkay, Karen E. Anderson, Hubert H. Fernandez, Stewart A. Factor, Robert A. Hauser, Maria Wieman, Mark Forrest Gordon, Juha-Matti Savola
-
- Journal:
- CNS Spectrums / Volume 26 / Issue 2 / April 2021
- Published online by Cambridge University Press:
- 10 May 2021, pp. 159-160
-
- Article
-
- You have access Access
- Export citation
-
Background
Deutetrabenazine, a novel vesicular monoamine transporter 2 (VMAT2) inhibitor, is approved by the FDA for treatment of tardive dyskinesia (TD) in adults. Dopamine-receptor antagonists (DRAs) are associated with worsening of metabolic parameters, including weight gain, hyperlipidemia, and elevated blood glucose. This post hoc analysis assessed the short- and long-term effects of deutetrabenazine treatment on weight and metabolic parameters in individuals treated for TD.
MethodsTwo 12-week, randomized placebo-controlled trials (RCTs) of deutetrabenazine for patients with TD evaluated either fixed dosing (AIM-TD; 12, 24, or 36 mg) or dose titration (ARM-TD; max dose, 48 mg/day). Patients completing ARM-TD or AIM-TD were included in an open-label extension (OLE) study, in which all patients underwent response-driven titration of deutetrabenazine from 12 mg/day up to a maximum total dose of 48 mg/day. Weight, body mass index (BMI), serum glucose, serum total cholesterol, and serum triglycerides were evaluated at baseline and during treatment in the RCTs and in the OLE.
ResultsIn the RCTs, 282 and 133 patients received deutetrabenazine or placebo. At baseline, 77% of patients used DRAs. At Week 12, no meaningful changes in weight were observed, with mean (standard error) weight changes of 0.9–1.2 (0.3–0.5) and 0.2 (0.3) kg in the deutetrabenazine and placebo groups, respectively, and mean BMI changes of 0.3–0.5 (0.1–0.2) and 0.1 (0.1) kg/m2. 337 patients were included in the analysis of the OLE study. No meaningful changes were observed in weight (mean change: 0.4 [0.4] kg at Week 54, –0.5 [0.6] kg at Week 106, and –1.1 [0.6] kg at Week 145) or BMI (mean change: 0.1 [0.2] kg/m2 at Week 54, –0.2 [0.2] kg/m2 at Week 106, and –0.3 [0.2] kg/m2 at Week 145). Across the studies, no meaningful changes were observed in triglyceride, cholesterol, or glucose levels.
ConclusionDeutetrabenazine does not affect common metabolic parameters in patients with TD, even during long-term exposure.
FundingTeva Pharmaceutical Industries Ltd., Petach Tikva, Israel
Long-Term Deutetrabenazine Treatment Is Associated With Continued Improvement in Tardive Dyskinesia in the Completed 3-Year Open-Label Extension Study
- Robert A. Hauser, Hadas Barkay, Hubert H. Fernandez, Stewart A. Factor, Joohi Jimenez-Shahed, Nicholas Gross, Leslie Marinelli, Amanda Wilhelm, Mark Forrest Gordon, Juha-Matti Savola, Karen E. Anderson
-
- Journal:
- CNS Spectrums / Volume 26 / Issue 2 / April 2021
- Published online by Cambridge University Press:
- 10 May 2021, p. 162
-
- Article
-
- You have access Access
- Export citation
-
Background
The 12-week ARM-TD and AIM-TD studies in tardive dyskinesia (TD) patients showed statistically significant improvements in TD symptoms with deutetrabenazine. The completed open-label extension (OLE) study (SD-809-C−20) evaluated long-term efficacy and safety of deutetrabenazine in TD.
MethodsPatients who completed ARM-TD or AIM-TD enrolled in the OLE study, with deutetrabenazine dose titrated based on dyskinesia control and tolerability. Change from baseline in Abnormal Involuntary Movement Scale (AIMS) score was assessed by local site raters. Treatment success was evaluated locally as patients being “much improved” or “very much improved” on Clinical Global Impression of Change (CGIC).
Results343 patients enrolled in the OLE study; 6 patients were excluded from analyses. At Week 54 (n=249; dose [mean±SE]: 38.7±0.66mg/day), mean change from baseline in AIMS score was –4.8±0.28; 66% of patients experienced treatment success. At Week 106 (n=194; dose: 39.3±0.75mg/day), mean change from baseline in AIMS score was –5.4±0.33; 65% of patients experienced treatment success. At Week 145 (n=160; dose: 39.4±0.83mg/day), mean change from baseline in AIMS score was –6.6±0.37; 73% of patients experienced treatment success. Treatment was generally well tolerated across 723 patient-years of exposure through Week 158, and exposure-adjusted incidence rates (incidence/patient-years) for akathisia/restlessness were 0.01, somnolence/sedation were 0.07, and symptoms which may represent parkinsonism or depression were 0.08 each.
ConclusionsPatients who received long-term treatment with deutetrabenazine achieved sustained improvement in AIMS scores. Findings from this open-label trial with response-driven dosing suggest the possibility of increasing benefit over time.
FundingTeva Pharmaceutical Industries Ltd., Petach Tikva, Israel
Comparison of Safety and Tolerability of Deutetrabenazine During Titration and Maintenance in Patients with Tardive Dyskinesia
- Amanda Wilhelm, Karen E. Anderson, Hubert H. Fernandez, Hadas Barkay, Nayla Chaijale, Alexander F. Send, Juha-Matti Savola, Mark Forrest Gordon
-
- Journal:
- CNS Spectrums / Volume 26 / Issue 2 / April 2021
- Published online by Cambridge University Press:
- 30 April 2021, p. 164
-
- Article
-
- You have access Access
- Export citation
-
Background
Deutetrabenazine is approved to treat tardive dyskinesia (TD) in adults and is titrated weekly by 6 mg/day, from 12 to 48 mg/day, based on dyskinesia control and tolerability. This analysis compared the safety of deutetrabenazine during titration versus maintenance.
MethodsSafety was assessed during titration versus maintenance using integrated data from two 12-week placebo-controlled studies (ARM-TD and AIM-TD) and the open-label extension study. Rates were compared for overall and serious adverse events (AEs), AEs leading to discontinuation, treatment-related AEs, common AEs (≥4%), and specific AEs (parkinsonism, suicidal ideation, akathisia, restlessness).
ResultsIn titration versus maintenance, AE rates with placebo (n=130) were: overall, 43.1% vs 25.4%; serious, 4.6% vs 2.3%; leading to discontinuation, 3.1% vs 0; treatment-related, 26.9% vs 10.0%. For placebo, common AEs during titration were somnolence, headache, nausea, fatigue, and dry mouth; none occurred during maintenance. In titration versus maintenance, AE rates in fixed-dose deutetrabenazine 12–36 mg (n=216) were: overall, 33.3–38.9% vs 22.2–29.2%; serious, 2.8–6.9% vs 0–1.4%; leading to discontinuation, 2.8–5.6% vs 0; treatment-related, 8.3–16.7% vs 8.3–13.9%. For fixed-dose deutetrabenazine, common AEs during titration were headache, diarrhea, nasopharyngitis, depression, hypertension, and dry mouth; headache was the only common AE during maintenance. In titration versus maintenance, AE rates with flexible-dose deutetrabenazine (n=168) were: overall, 49.4% vs 32.7%; serious, 3.6% vs 2.4%; leading to discontinuation, 2.4% vs 0.6%. For flexible-dose deutetrabenazine, the only common AE during titration was somnolence; none occurred during maintenance. Rates of parkinsonism, suicidal ideation, akathisia, and restlessness were low and comparable in titration and maintenance.
ConclusionsDeutetrabenazine was well-tolerated, with AE rates similar to placebo during both phases; AE rates were higher during titration and decreased during maintenance.
FundingTeva Pharmaceutical Industries Ltd., Petach Tikva, Israel
The Rapid ASKAP Continuum Survey I: Design and first results
- Part of
- D. McConnell, C. L. Hale, E. Lenc, J. K. Banfield, George Heald, A. W. Hotan, James K. Leung, Vanessa A. Moss, Tara Murphy, Andrew O’Brien, Joshua Pritchard, Wasim Raja, Elaine M. Sadler, Adam Stewart, Alec J. M. Thomson, M. Whiting, James R. Allison, S. W. Amy, C. Anderson, Lewis Ball, Keith W. Bannister, Martin Bell, Douglas C.-J. Bock, Russ Bolton, J. D. Bunton, A. P. Chippendale, J. D. Collier, F. R. Cooray, T. J. Cornwell, P. J. Diamond, P. G. Edwards, N. Gupta, Douglas B. Hayman, Ian Heywood, C. A. Jackson, Bärbel S. Koribalski, Karen Lee-Waddell, N. M. McClure-Griffiths, Alan Ng, Ray P. Norris, Chris Phillips, John E. Reynolds, Daniel N. Roxby, Antony E. T. Schinckel, Matt Shields, Chenoa Tremblay, A. Tzioumis, M. A. Voronkov, Tobias Westmeier
-
- Journal:
- Publications of the Astronomical Society of Australia / Volume 37 / 2020
- Published online by Cambridge University Press:
- 30 November 2020, e048
-
- Article
-
- You have access Access
- HTML
- Export citation
-
The Rapid ASKAP Continuum Survey (RACS) is the first large-area survey to be conducted with the full 36-antenna Australian Square Kilometre Array Pathfinder (ASKAP) telescope. RACS will provide a shallow model of the ASKAP sky that will aid the calibration of future deep ASKAP surveys. RACS will cover the whole sky visible from the ASKAP site in Western Australia and will cover the full ASKAP band of 700–1800 MHz. The RACS images are generally deeper than the existing NRAO VLA Sky Survey and Sydney University Molonglo Sky Survey radio surveys and have better spatial resolution. All RACS survey products will be public, including radio images (with
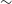 $\sim$
15 arcsec resolution) and catalogues of about three million source components with spectral index and polarisation information. In this paper, we present a description of the RACS survey and the first data release of 903 images covering the sky south of declination
$\sim$
15 arcsec resolution) and catalogues of about three million source components with spectral index and polarisation information. In this paper, we present a description of the RACS survey and the first data release of 903 images covering the sky south of declination 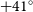 $+41^\circ$
made over a 288-MHz band centred at 887.5 MHz.
$+41^\circ$
made over a 288-MHz band centred at 887.5 MHz.
151 Confirmed Safety of Deutet.rabenazine for Tardive Dyskinesia in a 3-Year Open-Label Extension Study
- Hubert H. Fernandez, Hadas Barkay, Robert A. Hauser, Stewart A. Factor, Joohi Jimenez-Shahed, Nicholas Gross, Leslie Marinelli, Mark Forrest Gordon, Juha-Matti Savola, Karen E. Anderson
-
- Journal:
- CNS Spectrums / Volume 25 / Issue 2 / April 2020
- Published online by Cambridge University Press:
- 24 April 2020, pp. 296-297
-
- Article
-
- You have access Access
- Export citation
-
Background:
Deutetrabenazine (Austedo) is approved by the FDA for treatment of tardive dyskinesia (TD) in adults. In the 12-week ARM-TD and AIM-TD studies, deutetrabenazine showed clinically significant improvements in Abnormal Involuntary Movement Scale (AIMS) scores compared with placebo, and there were low rates of overall adverse events (AEs) and discontinuations associated with deutetrabenazine. The objective of this study was to evaluate the long-term safety and tolerability of deutetrabenazine in patients with TD at 3 years.
METHODS:Patients who completed ARM-TD or AIM-TD were included in this open-label, single-arm extension study, in which all patients restarted/started deutetrabenazine 12 mg/day, titrating up to a maximum total daily dose of 48 mg/day based on dyskinesia control and tolerability. The study comprised a 6-week titration period and a long-term maintenance phase. Safety measures included incidence of AEs, serious AEs (SAEs), and AEs leading to withdrawal, dose reduction, or dose suspension. Exposure-adjusted incidence rates (EAIRs; incidence/patient-years) were used for calculating AE frequencies. This analysis reports results up to Week 158.
RESULTS:A total of 343 patients were enrolled (111 received placebo and 232 received deutetrabenazine in the parent studies). At the time of this analysis, 183 patients were still receiving treatment; 259 completed 1 year, 172 completed 2 years, and 41 completed 3 years. There were 623 patient-years of exposure. More than 40% of patients reached the maximum dose. EAIRs of AEs were comparable to or lower than those observed in the ARM-TD and AIM-TD short-term randomized trials of deutetrabenazine vs. placebo. The frequency of SAEs (EAIR 0.10) was similar to that observed with short-term placebo (0.33) and short-term deutetrabenazine (range 0.06–0.33) treatment. AEs leading to withdrawal (0.06), dose reduction (0.10), and dose suspension (0.05) were uncommon.
CONCLUSION:These results support the safety outcomes observed in the ARM-TD and AIM-TD parent studies and the safety of deutetrabenazine for long-term use in patients with TD.
Funding Acknowledgements: This study was funded by Teva Pharmaceuticals, Petach Tikva, Israel
134 Long-Term Deutetrabenazine Treatment Is Associated with Sustained Treatment Response in Tardive Dyskinesia: Results from an Open-Label Extension Study
- Robert A. Hauser, Hadas Barkay, Hubert H. Fernandez, Stewart A. Factor, Joohi Jimenez-Shahed, Nicholas Gross, Leslie Marinelli, Mark Forrest Gordon, Juha-Matti Savola, Karen E. Anderson
-
- Journal:
- CNS Spectrums / Volume 25 / Issue 2 / April 2020
- Published online by Cambridge University Press:
- 24 April 2020, pp. 284-285
-
- Article
-
- You have access Access
- Export citation
-
Background:
In the 12-week ARM-TD and AIM-TD studies evaluating deutetrabenazine for the treatment of tardive dyskinesia (TD), the percentage of patients achieving ≥50% response was higher in the deutetrabenazine-treated group than in the placebo group. These studies also showed low rates of overall adverse events (AEs) and discontinuations associated with deutetrabenazine. The current open-label study evaluated the long-term efficacy and safety of deutetrabenazine in patients with TD.
Methods:Patients with TD who completed ARM-TD or AIM-TD could enroll in this open-label, single-arm extension study, titrating up over 6 weeks to a maximum total daily dose of deutetrabenazine 48 mg/day on the basis of dyskinesia control and tolerability. The proportion of Abnormal Involuntary Movement Scale (AIMS; items 1-7) responders was assessed based on response rates for achieving ≥50% improvement from baseline in the open-label extension study. AlMS score was assessed by local site raters for this analysis.
Results:343 patients enrolled in the extension study. At Week 54 (n=249; total daily dose [mean ± standard error]: 38.6±0.66 mg), the mean percentage change from baseline in AIMS score was –40%; 48% of patients achieved a ≥50% response and 59% of those had already achieved a ≥50% response at Week 15. Further, 34% of those who had not achieved a ≥50% response at Week 15 achieved a ≥50% response at Week 54. At Week 106 (n=169; total daily dose: 39.6±0.77 mg), the mean percentage change from baseline in AIMS score was –45%; 55% of patients achieved a ≥50% response, 59% of those patients had already achieved a ≥50% response at Week 15, and 41% of those who had not achieved a ≥50% response at Week 15 but who reached Week 106 achieved a ≥50% response. At Week 132 (n=109; total daily dose: 39.7±0.97 mg), the mean percentage change from baseline in AIMS score was –61%; 55% of patients achieved a ≥50% response, 61% of those patients had already achieved a ≥50% response at Week 15, and 43% of those who had not achieved a ≥50% response at Week 15 but who reached Week 132 achieved a ≥50% response. Completer analysis suggests that long-term efficacy was not due to dose increases over time. Treatment with deutetrabenazine was generally well tolerated. There were 623 patient-years of exposure through Week 158, and exposure-adjusted incidence rates (incidence/patient-years) of adverse events of special interest were 0.01 for akathisia and restlessness, 0.07 for somnolence and sedation, 0.04 for parkinsonism, and 0.05 for depression.
Conclusions:Patients who received long-term treatment with deutetrabenazine achieved response rates that were indicative of clinically meaningful long-term benefit. Results from this open-label trial suggest the possibility of increasing benefit over time with individual dose titration of deutetrabenazine.
Funding Acknowledgements:This study was funded by Teva Pharmaceuticals, Petach Tikva, Israel.
34 Long-Term Deutetrabenazine Treatment Response in Tardive Dyskinesia by Concomitant Dopamine-Receptor Antagonists and Baseline Comorbidities
- Karen E. Anderson, David Stamler, Mat D. Davis, Robert A. Hauser, L. Fredrik Jarskog, Joohi Jimenez-Shahed, Rajeev Kumar, Stanislaw Ochudlo, Joseph McEvoy, Hubert H. Fernandez
-
- Journal:
- CNS Spectrums / Volume 24 / Issue 1 / February 2019
- Published online by Cambridge University Press:
- 12 March 2019, p. 193
-
- Article
-
- You have access Access
- Export citation
-
Background
Tardive dyskinesia (TD) results from exposure to dopamine-receptor antagonists (DRAs), such as typical and atypical antipsychotics. Clinicians commonly manage TD by reducing the dose of or stopping the causative agent; however, this may cause psychiatric relapse and worsen quality of life. In the 12-week ARM-TD and AIM-TD trials, deutetrabenazine demonstrated statistically significant improvements in Abnormal Involuntary Movement Scale (AIMS) scores versus placebo and was generally well tolerated, regardless of baseline DRA use or comorbidities.
Study ObjectiveTo evaluate the impact of underlying disease and current DRA use on efficacy and safety of long-term therapy of deutetrabenazine in patients with TD.
MethodPatients with TD who completed ARM-TD or AIM-TD were eligible to enter this open-label, single-arm, long-term extension after completing the 1-week washout period and final evaluation in the blinded portion of the trial. Change in AIMS scores from baseline to Week 54 and patients “Much Improved” or “Very Much Improved” (treatment success) on the Clinical Global Impression of Change (CGIC) and Patient Global Impression of Change (PGIC) at Week 54 were analyzed by baseline psychiatric illness type, including mood disorders (bipolar disorder/depression/other) or psychotic disorders (schizophrenia/schizoaffective disorder), and presence or absence of current DRA use.
ResultsAt Week 54, meaningful improvements from baseline in mean (standard error) AIMS scores were observed for patients with baseline mood disorders (–5.2[0.93]) and psychotic disorders (–5.0[0.63]), and in patients currently using DRAs (–4.6[0.54]) or not using DRAs (–6.4[1.27]). Most patients with mood disorders (73%) and psychotic disorders (71%) were “Much Improved” or “Very Much Improved” on CGIC at Week 54, similar to patients currently using (71%) or not using (74%) DRAs. The majority of patients with mood disorders (62%) and psychotic disorders (57%), as well as patients currently using (58%) or not using (63%) DRAs, were also “Much Improved” or “Very Much Improved” on PGIC at Week 54. Prior treatment in ARM-TD and AIM-TD did not impact the long-term treatment response. Underlying psychiatric disorder and concomitant DRA use did not impact the occurrence of adverse events (AEs). The frequencies of dose reductions, dose suspensions, and withdrawals due to AEs were low, regardless of baseline psychiatric comorbidities and DRAuse.
ConclusionsLong-term deutetrabenazine treatment demonstrated meaningful improvements in abnormal movements in TD patients, which were recognized by clinicians and patients, regardless of underlying psychiatric illness or DRAuse.
Presented at: American Psychiatric Association Annual Meeting; May 5–9, 2018, New York, New York, USA
Funding Acknowledgements: This study was supported by Teva Pharmaceuticals, Petach Tikva, Israel.
45 Long-term Treatment with Deutetrabenazine Is Associated with Continued Improvement in Tardive Dyskinesia: Results from an Open-label Extension Study
- Robert A. Hauser, Hubert H. Fernandez, David Stamler, Mat D. Davis, Stewart A. Factor, Joohi Jimenez-Shahed, William G. Ondo, L. Fredrik Jarskog, Scott W. Woods, Mark S. LeDoux, David R. Shprecher, Karen E. Anderson
-
- Journal:
- CNS Spectrums / Volume 24 / Issue 1 / February 2019
- Published online by Cambridge University Press:
- 12 March 2019, pp. 200-201
-
- Article
-
- You have access Access
- Export citation
-
Study Objective
To evaluate long-term efficacy of deutetrabenazine in patients with tardive dyskinesia (TD) by examining response rates from baseline in Abnormal Involuntary Movement Scale (AIMS) scores. Preliminary results of the responder analysis are reported in this analysis.
BackgroundIn the 12-week ARM-TD and AIM-TD studies, the odds of response to deutetrabenazine treatment were higher than the odds of response to placebo at all response levels, and there were low rates of overall adverse events and discontinuations associated with deutetrabenazine.
MethodPatients with TD who completed ARM-TD or AIM-TD were included in this open-label, single-arm extension study, in which all patients restarted/started deutetrabenazine 12mg/day, titrating up to a maximum total daily dose of 48mg/day based on dyskinesia control and tolerability. The study comprised a 6-week titration and a long-term maintenance phase. The cumulative proportion of AIMS responders from baseline was assessed. Response was defined as a percent improvement from baseline for each patient from 10% to 90% in 10% increments. AlMS score was assessed by local site ratings for this analysis.
Results343 patients enrolled in the extension study (111 patients received placebo in the parent study and 232 patients received deutetrabenazine). At Week 54 (n=145; total daily dose [mean±standard error]: 38.1±0.9mg), 63% of patients receiving deutetrabenazine achieved ≥30% response, 48% of patients achieved ≥50% response, and 26% achieved ≥70% response. At Week 80 (n=66; total daily dose: 38.6±1.1mg), 76% of patients achieved ≥30% response, 59% of patients achieved ≥50% response, and 36% achieved ≥70% response. Treatment was generally well tolerated.
ConclusionsPatients who received long-term treatment with deutetrabenazine achieved response rates higher than those observed in positive short-term studies, indicating clinically meaningful long-term treatment benefit.
Presented at: American Academy of Neurology Annual Meeting; April 21–27, 2018, Los Angeles, California, USA.
Funding Acknowledgements: This study was supported by Teva Pharmaceuticals, Petach Tikva, Israel.
46 Confirmed Safety of Deutetrabenazine for Tardive Dyskinesia in a 2-Year Open-label Extension Study
- Hubert H. Fernandez, David Stamler, Mat D. Davis, Stewart A. Factor, Robert A. Hauser, Joohi Jimenez-Shahed, William G. Ondo, L. Fredrik Jarskog, Scott W. Woods, Mark S. LeDoux, David R. Shprecher, Karen E. Anderson
-
- Journal:
- CNS Spectrums / Volume 24 / Issue 1 / February 2019
- Published online by Cambridge University Press:
- 12 March 2019, p. 201
-
- Article
-
- You have access Access
- Export citation
-
Study Objective
To evaluate the long-term safety and tolerability of deutetrabenazine in patients with tardive dyskinesia (TD) at 2years.
BackgroundIn the 12-week ARM-TD and AIM-TD studies, deutetrabenazine showed clinically significant improvements in Abnormal Involuntary Movement Scale scores compared with placebo, and there were low rates of overall adverse events (AEs) and discontinuations associated with deutetrabenazine.
MethodPatients who completed ARM-TD or AIM-TD were included in this open-label, single-arm extension study, in which all patients restarted/started deutetrabenazine 12mg/day, titrating up to a maximum total daily dose of 48mg/day based on dyskinesia control and tolerability. The study comprised a 6-week titration period and a long-term maintenance phase. Safety measures included incidence of AEs, serious AEs (SAEs), and AEs leading to withdrawal, dose reduction, or dose suspension. Exposure-adjusted incidence rates (EAIRs; incidence/patient-years) were used to compare AE frequencies for long-term treatment with those for short-term treatment (ARM-TD and AIM-TD). This analysis reports results up to 2 years (Week106).
Results343 patients were enrolled (111 patients received placebo in the parent study and 232 received deutetrabenazine). There were 331.4 patient-years of exposure in this analysis. Through Week 106, EAIRs of AEs were comparable to or lower than those observed with short-term deutetrabenazine and placebo, including AEs of interest (akathisia/restlessness [long-term EAIR: 0.02; short-term EAIR range: 0–0.25], anxiety [0.09; 0.13–0.21], depression [0.09; 0.04–0.13], diarrhea [0.06; 0.06–0.34], parkinsonism [0.01; 0–0.08], somnolence/sedation [0.09; 0.06–0.81], and suicidality [0.02; 0–0.13]). The frequency of SAEs (EAIR 0.15) was similar to those observed with short-term placebo (0.33) and deutetrabenazine (range 0.06–0.33) treatment. AEs leading to withdrawal (0.08), dose reduction (0.17), and dose suspension (0.06) were uncommon.
ConclusionsThese results confirm the safety outcomes seen in the ARM-TD and AIM-TD parent studies, demonstrating that deutetrabenazine is well tolerated for long-term use in TD patients.
Presented at: American Academy of Neurology Annual Meeting; April 21–27, 2018, Los Angeles, California,USA
Funding Acknowledgements: Funding: This study was supported by Teva Pharmaceuticals, Petach Tikva, Israel
35 Long-term Improvements in Site-Rated Outcomes with Deutetrabenazine Treatment in Patients with Tardive Dyskinesia
- Karen E. Anderson, David Stamler, Mat D. Davis, Nicholas Gross, Robert A. Hauser, L. Fredrik Jarskog, Joohi Jimenez-Shahed, Rajeev Kumar, Stanislaw Ochudlo, Joseph McEvoy, Hubert H. Fernandez
-
- Journal:
- CNS Spectrums / Volume 24 / Issue 1 / February 2019
- Published online by Cambridge University Press:
- 12 March 2019, pp. 193-194
-
- Article
-
- You have access Access
- Export citation
-
Background
Tardive dyskinesia (TD) is an often-irreversible movement disorder that may intensify the stigma of patients with psychiatric disorders and worsen quality of life. In two randomized, double-blind, placebo (PBO)-controlled, 12-week trials, ARM-TD and AIM-TD (‘parent studies’), deutetrabenazine (DTB) demonstrated statistically significant improvements in centrally read Abnormal Involuntary Movement Scale (AIMS) scores at Week 12 compared with PBO and was generally well tolerated.
Study ObjectiveTo evaluate the long-term efficacy of DTB in an open-label safety study following double-blind treatment using site-rated efficacy measures: AIMS, the Clinical Global Impression of Change (CGIC) and the Patient Global Impression of Change (PGIC), which may be used in real-world clinical practice settings.
MethodPatients with TD who completed the parent studies were eligible to enter this open-label, long-term extension (OLE) after completing the 1-week washout period and final evaluation in the blinded portion of the trial. This extension comprised a 6-week titration period followed by a long-term maintenance phase. Patients began DTB at 12mg/day, titrating up to a maximum total dose of 48mg/day based on dyskinesia control and tolerability. Efficacy endpoints included in this analysis are the change in site-rated AIMS score (items 1–7) from parent study baseline, and the proportion of patients who were “Much Improved” or “Very Much Improved” (treatment success) on the CGIC and PGIC from OLE baseline.
ResultsAt the end of the parent studies (Week 12), patients treated with DTB had experienced greater mean (standard error) improvements in site-rated AIMS score (–5.0[0.40]) than patients given PBO (–3.2[0.47]). With long-term DTB treatment, both groups experienced improvements in site-rated AIMS scores (prior DTB, –7.9[0.62]; prior placebo, –6.6[0.64]) compared with parent study baseline. Similarly, at the end of the parent studies, a greater proportion of patients treated with DTB had treatment success on the CGIC (DTB, 51%; PBO, 32%) and the PGIC (DTB, 46%; PBO: 33%); whereas at Week 54 of the OLE study, treatment success on CGIC and PGIC were similar in both the CGIC (prior DTB: 66%; prior PBO: 68%) and PGIC (prior DTB: 62%; prior PBO: 62%) groups. DTB was generally well tolerated.
ConclusionsPatients treated with DTB showed improvements in abnormal movements, as measured by site-rated AIMS, CGIC, and PGIC scores, which may be used in real-world clinical practice settings. These results corroborate the previously reported efficacy of DTB as observed in the 12-week, double-blind ARM-TD and AIM-TD trials, in which central raters were used to evaluate AIMS scores.
Presented at: American Psychiatric Association Annual Meeting; May 5–9, 2018, New York, New York, USA
Funding Acknowledgements: Funding: This study was supported by Teva Pharmaceuticals, Petach Tikva, Israel.
158 Long-Term Safety of Deutetrabenazine for the Treatment of Tardive Dyskinesia: Results From an Open-Label, Long-Term Study
- Karen E. Anderson, Mat D. Davis, Stewart A. Factor, Robert A. Hauser, L. Fredrik Jarskog, Joohi Jimenez-Shahed, Rajeev Kumar, Stanislaw Ochudlo, William G. Ondo, Hubert H. Fernandez
-
- Journal:
- CNS Spectrums / Volume 23 / Issue 1 / February 2018
- Published online by Cambridge University Press:
- 15 June 2018, p. 97
-
- Article
-
- You have access Access
- Export citation
-
Introduction
In the 12-week ARM-TD and AIM-TD studies, deutetrabenazine showed clinically significant improvements in Abnormal Involuntary Movement Scale (AIMS) scores at Week 12 compared with placebo, and was generally well tolerated.
ObjectiveTo evaluate the long-term safety/tolerability and efficacy of deutetrabenazine in patients with TD. Week 54 open-labelresults are reported in this interim analysis.
MethodsPatients with TD who completed ARM-TD or AIM-TD were included in this open-label, single-arm extension study, in which all patients restarted/started deutetrabenazine 12 mg/day, titrating up to a maximum total daily dose of 48 mg/day based on dyskinesia control and tolerability. The study comprised a 6-week titration period and a long-term maintenance phase. Safetymeasures included incidence of adverse events (AEs), serious AEs (SAEs), drug-related AEs, and AEs leading to withdrawal, dose reduction, or dose suspension. This analysis reports results up to Week 54.
Results304 patients enrolled in the extension study. There were 215 patient-years of exposure in this analysis, and exposure-adjusted incidence rates (EAIRs) of AEs (incidence/patient-years) were comparable to or lower than those observed with short-term deutetrabenazine treatment and placebo. The frequency of SAEs (EAIR 0.14) was similar to rates observed with short-termplacebo (EAIR 0.33) and deutetrabenazine (EAIR range 0.06–0.33) treatment. AEs leading to study discontinuation (EAIR 0.08), dose reduction (EAIR 0.17), and dose suspension (EAIR 0.09) were uncommon.
ConclusionsLong-term treatment with deutetrabenazine was generally safe and well tolerated in patients with TD, and did not result in cumulative toxicity.
Presented at: The American Psychiatric Association 2017 Annual Meeting; May 20–24, 2017; San Diego, California, USA.
Funding AcknowledgementsThis study was funded by Teva Pharmaceutical Industries, Petach Tikva, Israel.
149 Deutetrabenazine for the Treatment of Tardive Dyskinesia: Results From an Open-Label, Long-Term Study
- Karen E. Anderson, Mat D. Davis, Stewart A. Factor, Robert A. Hauser, L. Fredrik Jarskog, Joohi Jimenez-Shahed, Rajeev Kumar, FRCPC, Stanislaw Ochudlo, William G. Ondo, Hubert H. Fernandez
-
- Journal:
- CNS Spectrums / Volume 23 / Issue 1 / February 2018
- Published online by Cambridge University Press:
- 15 June 2018, pp. 92-93
-
- Article
-
- You have access Access
- Export citation
-
Introduction
Tardive dyskinesia (TD) is an involuntary movement disorder resulting from exposure to dopamine-receptor antagonists. In the 12-week ARM-TD and AIM-TD studies, deutetrabenazine demonstrated significant improvements in Abnormal Involuntary Movement Scale (AIMS) scores at Week 12 compared with placebo, and was generally well tolerated.
ObjectiveTo evaluate the efficacy and safety of long-term deutetrabenazine therapy in patients with TD.
MethodsPatients with TD who completed the ARM-TD or AIM-TD studies were eligible to enter this open-label, single-arm, long-term safety study after they completed the 1-week washout period and final evaluation in the blinded portion of the trial. Efficacy endpoints included the change in AIMS score from baseline, and treatment success (defined as “much improved” or “very much improved”) on the Clinical Global Impression of Change (CGIC) and Patient Global Impression of Change (PGIC). This analysis reports results up to Week 54.
Results304 patients enrolled in the extension study. At Week 54, the mean (standard error) change in AIMS score was –5.1 (0.52). After 6 weeks of deutetrabenazine treatment, the proportion of patients who achieved treatment success was 58% per the CGIC and 53% per the PGIC, and by Week 54 was 72% per the CGIC and 59% per the PGIC, thus demonstrating maintenance or enhancement of benefit over time. Deutetrabenazine was well tolerated for up to 54 weeks, and compared with the ARM-TD and AIM-TD studies, no new safety signals were detected.
Conclusions54 weeks of deutetrabenazine treatment was generally efficacious, safe, and well tolerated in patients with TD.
Presented at: The American Psychiatric Association 2017 Annual Meeting; May 20–24, 2017; San Diego, California, USA.
Funding AcknowledgementsThis study was funded by Teva Pharmaceutical Industries, Petach Tikva, Israel.
134 Improvements in Clinical Global Impression of Change With Deutetrabenazine Treatment in Tardive Dyskinesia From the ARM-TD and AIM-TD Studies
- Hubert H. Fernandez, Mat D. Davis, Stewart A. Factor, Robert A. Hauser, L. Fredrik Jarskog, Joohi Jimenez-Shahed, Rajeev Kumar, Stanislaw Ochudlo, William G. Ondo, Karen E. Anderson
-
- Journal:
- CNS Spectrums / Volume 23 / Issue 1 / February 2018
- Published online by Cambridge University Press:
- 15 June 2018, p. 84
-
- Article
-
- You have access Access
- Export citation
-
Introduction
Tardive dyskinesia (TD) is an involuntary movement disorder that is often irreversible, can affect any body region, and can be debilitating. In the ARM-TDand AIM-TD studies, deutetrabenazine treatment demonstrated statistically and clinically significant reductions in Abnormal Involuntary Movement Scale (AIMS) scores at Week 12 compared with placebo (primary endpoint).
ObjectiveTo evaluate the efficacy of deutetrabenazine, as measured by the Clinical Global Impression of Change (CGIC) scale, in patients with TD from the pooled ARM-TDand AIM-TD (24 and 36 mg/day doses) data sets, as compared with the pooled placebo cohort.
MethodsARM-TD and AIM-TD were 12-week, randomized, double-blind, placebo-controlled studies that evaluated the safety and efficacy of deutetrabenazine for thetreatment of TD. The key secondary endpoint of each study was the proportion of patients “much improved” or “very much improved” (treatment success) at Week 12 on theCGIC.
ResultsAt Week 12, the odds of treatment success among patients treated with deutetrabenazine (n=152) was more than double that of patients given placebo (n=107; odds ratio: 2.12; P=0.005). In a categorical analysis of CGIC ratings, patients treated with deutetrabenazine showed greater improvement than patients given placebo (P=0.003). Patients treated with deutetrabenazine also had a significantly better treatment response than those given placebo (least-squares mean CGIC score treatment difference: –0.4; P=0.006).
ConclusionsDeutetrabenazine treatment led to statistically and clinically significant improvements in TD symptoms based on the CGIC result, suggesting that clinicians were able to recognize the benefit in patients treated with deutetrabenazine.
Presented at: The International Congress of Parkinson’s Disease and Movement Disorders; June 4–8, 2017; Vancouver, British Columbia, Canada.
Funding AcknowledgementsThese studies were funded by Teva Pharmaceutical Industries, Petach Tikva, Israel.
Continent-wide estimates of Antarctic strain rates from Landsat 8-derived velocity grids
- KAREN E. ALLEY, TED A. SCAMBOS, ROBERT S. ANDERSON, HARIHAR RAJARAM, ALLEN POPE, TERRY M. HARAN
-
- Journal:
- Journal of Glaciology / Volume 64 / Issue 244 / April 2018
- Published online by Cambridge University Press:
- 19 March 2018, pp. 321-332
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
-
Strain rates are fundamental measures of ice flow and are used in a wide variety of glaciological applications including investigations of bed properties, calculations of basal mass balance on ice shelves, and constraints on ice rheological models. However, despite their extensive application, strain rates are calculated using a variety of methods and length scales and the details are often not specified. In this study, we compare the results of nominal and logarithmic strain-rate calculations based on a satellite-derived velocity field of the Antarctic ice sheet generated from Landsat 8 satellite data. Our comparison highlights the differences between the two common approaches in the glaciological literature. We evaluate the errors introduced by each approach and their impacts on the results. We also demonstrate the importance of choosing and specifying a length scale over which strain-rate calculations are made, which can strongly influence other derived quantities such as basal mass balance on ice shelves. Finally, we present strain-rate data products calculated using an approximate viscous length-scale with satellite observations of ice velocity for the Antarctic continent.
The Differential Diagnosis of Pseudobulbar Affect (PBA): Distinguishing PBA Among Disorders of Mood and Affect
- David B. Arciniegas, Edward C. Lauterbach, David L. Ginsberg, Karen E. Anderson, Tiffany W. Chow, Laura A. Flashman, Robin A. Hurley, Daniel I. Kaufer, Thomas W. McAllister, Alison Reeve, Randolph B. Schiffer, Jonathan M. Silver
-
- Journal:
- CNS Spectrums / Volume 10 / Issue S5 / 2005
- Published online by Cambridge University Press:
- 07 November 2014, pp. 1-16
-
- Article
- Export citation
-
This monograph summarizes the proceedings of a roundtable meeting convened to discuss pseudobulbar affect (PBA). Two didactic lectures were presented, followed by a moderated discussion among 11 participants. Post-meeting manuscript development synthesized didactic- and discussion-based content and incorporated additional material from the neuroscience literature. A conceptual framework with which to distinguish between disorders of mood and affect is presented first, and disorders of affect regulation are then reviewed briefly. A detailed description of the most common of these disorders, PBA, is the focus of the remainder of the monograph. The prevalence, putative neuranatomic and neurochemical bases of PBA are reviewed, and current and emerging methods of evaluation and treatment of persons with PBA are discussed. The material presented in this monograph will help clinicians better recognize, diagnose, and treat PBA, and will form a foundation for understanding and interpreting future studies of this condition.
Contributors
-
- By Robert S. Anderson, (Mary) Colleen Bhalla, Michelle Blanda, Christopher Carpenter, Chris Chauhan, Paul L. DeSandre, Maura Dickinson, Jonathan A. Edlow, Dany Elsayegh, Kara Iskyan Geren, Peter J. Gruber, Jin H. Han, Marianne Haughey, Teresita M. Hogan, Ula Hwang, Lindsay Jin, Michael P. Jones, Joseph H. Kahn, Keli M. Kwok, Denise Law, Megan M. Leo, Stephen Y. Liang, Judith A. Linden, Brendan G. Magauran Jr, Joseph P. Martinez, Amal Mattu, Karen M. May, Aileen McCabe, Kerry K. McCabe, Jolion McGreevy, Ron Medzon, Ravi K. Murthy, Aneesh T. Narang, Lauren M. Nentwich, David E. Newman-Toker, Jonathan S. Olshaker, Joseph R. Pare, Thomas Perera, Joanna Piechniczek-Buczek, Jesse M. Pines, Timothy Platts-Mills, Suzanne Michelle Rhodes, Lynne Rosenberg, Mark Rosenberg, Todd C. Rothenhaus, Kristine Samson, Arthur B. Sanders, Jeffrey I. Schneider, Rishi Sikka, Kirk A. Stiffler, Morsal R. Tahouni, Mary E. Tanski, Abel Wakai, Scott T. Wilber, Deborah R. Wong
- Edited by Joseph H. Kahn, Brendan G. Magauran, Jr, Jonathan S. Olshaker
-
- Book:
- Geriatric Emergency Medicine
- Published online:
- 05 January 2014
- Print publication:
- 16 January 2014, pp vii-x
-
- Chapter
- Export citation
Contributors
-
- By Christopher J. Anderson, Candy Gunther Brown, Ted A. Campbell, Morris L. Davis, Wendy J. Deichmann, Dennis C. Dickerson, Maura Jane Farrelly, E. Brooks Holifield, Stan Ingersol, Douglas M. Koskela, Russell E. Richey, Douglas M. Strong, Michael K. Turner, Jason E. Vickers, Laceye C. Warner, Karen B. Westerfield Tucker, Andrew J. Wood, Jennifer L. Woodruff Tait
- Edited by Jason E. Vickers, United Theological Seminary, Ohio
-
- Book:
- The Cambridge Companion to American Methodism
- Published online:
- 18 December 2013
- Print publication:
- 07 October 2013, pp xi-xiv
-
- Chapter
- Export citation
Contributors
-
- By Rose Teteki Abbey, K. C. Abraham, David Tuesday Adamo, LeRoy H. Aden, Efrain Agosto, Victor Aguilan, Gillian T. W. Ahlgren, Charanjit Kaur AjitSingh, Dorothy B E A Akoto, Giuseppe Alberigo, Daniel E. Albrecht, Ruth Albrecht, Daniel O. Aleshire, Urs Altermatt, Anand Amaladass, Michael Amaladoss, James N. Amanze, Lesley G. Anderson, Thomas C. Anderson, Victor Anderson, Hope S. Antone, María Pilar Aquino, Paula Arai, Victorio Araya Guillén, S. Wesley Ariarajah, Ellen T. Armour, Brett Gregory Armstrong, Atsuhiro Asano, Naim Stifan Ateek, Mahmoud Ayoub, John Alembillah Azumah, Mercedes L. García Bachmann, Irena Backus, J. Wayne Baker, Mieke Bal, Lewis V. Baldwin, William Barbieri, António Barbosa da Silva, David Basinger, Bolaji Olukemi Bateye, Oswald Bayer, Daniel H. Bays, Rosalie Beck, Nancy Elizabeth Bedford, Guy-Thomas Bedouelle, Chorbishop Seely Beggiani, Wolfgang Behringer, Christopher M. Bellitto, Byard Bennett, Harold V. Bennett, Teresa Berger, Miguel A. Bernad, Henley Bernard, Alan E. Bernstein, Jon L. Berquist, Johannes Beutler, Ana María Bidegain, Matthew P. Binkewicz, Jennifer Bird, Joseph Blenkinsopp, Dmytro Bondarenko, Paulo Bonfatti, Riet en Pim Bons-Storm, Jessica A. Boon, Marcus J. Borg, Mark Bosco, Peter C. Bouteneff, François Bovon, William D. Bowman, Paul S. Boyer, David Brakke, Richard E. Brantley, Marcus Braybrooke, Ian Breward, Ênio José da Costa Brito, Jewel Spears Brooker, Johannes Brosseder, Nicholas Canfield Read Brown, Robert F. Brown, Pamela K. Brubaker, Walter Brueggemann, Bishop Colin O. Buchanan, Stanley M. Burgess, Amy Nelson Burnett, J. Patout Burns, David B. Burrell, David Buttrick, James P. Byrd, Lavinia Byrne, Gerado Caetano, Marcos Caldas, Alkiviadis Calivas, William J. Callahan, Salvatore Calomino, Euan K. Cameron, William S. Campbell, Marcelo Ayres Camurça, Daniel F. Caner, Paul E. Capetz, Carlos F. Cardoza-Orlandi, Patrick W. Carey, Barbara Carvill, Hal Cauthron, Subhadra Mitra Channa, Mark D. Chapman, James H. Charlesworth, Kenneth R. Chase, Chen Zemin, Luciano Chianeque, Philip Chia Phin Yin, Francisca H. Chimhanda, Daniel Chiquete, John T. Chirban, Soobin Choi, Robert Choquette, Mita Choudhury, Gerald Christianson, John Chryssavgis, Sejong Chun, Esther Chung-Kim, Charles M. A. Clark, Elizabeth A. Clark, Sathianathan Clarke, Fred Cloud, John B. Cobb, W. Owen Cole, John A Coleman, John J. Collins, Sylvia Collins-Mayo, Paul K. Conkin, Beth A. Conklin, Sean Connolly, Demetrios J. Constantelos, Michael A. Conway, Paula M. Cooey, Austin Cooper, Michael L. Cooper-White, Pamela Cooper-White, L. William Countryman, Sérgio Coutinho, Pamela Couture, Shannon Craigo-Snell, James L. Crenshaw, David Crowner, Humberto Horacio Cucchetti, Lawrence S. Cunningham, Elizabeth Mason Currier, Emmanuel Cutrone, Mary L. Daniel, David D. Daniels, Robert Darden, Rolf Darge, Isaiah Dau, Jeffry C. Davis, Jane Dawson, Valentin Dedji, John W. de Gruchy, Paul DeHart, Wendy J. Deichmann Edwards, Miguel A. De La Torre, George E. Demacopoulos, Thomas de Mayo, Leah DeVun, Beatriz de Vasconcellos Dias, Dennis C. Dickerson, John M. Dillon, Luis Miguel Donatello, Igor Dorfmann-Lazarev, Susanna Drake, Jonathan A. Draper, N. Dreher Martin, Otto Dreydoppel, Angelyn Dries, A. J. Droge, Francis X. D'Sa, Marilyn Dunn, Nicole Wilkinson Duran, Rifaat Ebied, Mark J. Edwards, William H. Edwards, Leonard H. Ehrlich, Nancy L. Eiesland, Martin Elbel, J. Harold Ellens, Stephen Ellingson, Marvin M. Ellison, Robert Ellsberg, Jean Bethke Elshtain, Eldon Jay Epp, Peter C. Erb, Tassilo Erhardt, Maria Erling, Noel Leo Erskine, Gillian R. Evans, Virginia Fabella, Michael A. Fahey, Edward Farley, Margaret A. Farley, Wendy Farley, Robert Fastiggi, Seena Fazel, Duncan S. Ferguson, Helwar Figueroa, Paul Corby Finney, Kyriaki Karidoyanes FitzGerald, Thomas E. FitzGerald, John R. Fitzmier, Marie Therese Flanagan, Sabina Flanagan, Claude Flipo, Ronald B. Flowers, Carole Fontaine, David Ford, Mary Ford, Stephanie A. Ford, Jim Forest, William Franke, Robert M. Franklin, Ruth Franzén, Edward H. Friedman, Samuel Frouisou, Lorelei F. Fuchs, Jojo M. Fung, Inger Furseth, Richard R. Gaillardetz, Brandon Gallaher, China Galland, Mark Galli, Ismael García, Tharscisse Gatwa, Jean-Marie Gaudeul, Luis María Gavilanes del Castillo, Pavel L. Gavrilyuk, Volney P. Gay, Metropolitan Athanasios Geevargis, Kondothra M. George, Mary Gerhart, Simon Gikandi, Maurice Gilbert, Michael J. Gillgannon, Verónica Giménez Beliveau, Terryl Givens, Beth Glazier-McDonald, Philip Gleason, Menghun Goh, Brian Golding, Bishop Hilario M. Gomez, Michelle A. Gonzalez, Donald K. Gorrell, Roy Gottfried, Tamara Grdzelidze, Joel B. Green, Niels Henrik Gregersen, Cristina Grenholm, Herbert Griffiths, Eric W. Gritsch, Erich S. Gruen, Christoffer H. Grundmann, Paul H. Gundani, Jon P. Gunnemann, Petre Guran, Vidar L. Haanes, Jeremiah M. Hackett, Getatchew Haile, Douglas John Hall, Nicholas Hammond, Daphne Hampson, Jehu J. Hanciles, Barry Hankins, Jennifer Haraguchi, Stanley S. Harakas, Anthony John Harding, Conrad L. Harkins, J. William Harmless, Marjory Harper, Amir Harrak, Joel F. Harrington, Mark W. Harris, Susan Ashbrook Harvey, Van A. Harvey, R. Chris Hassel, Jione Havea, Daniel Hawk, Diana L. Hayes, Leslie Hayes, Priscilla Hayner, S. Mark Heim, Simo Heininen, Richard P. Heitzenrater, Eila Helander, David Hempton, Scott H. Hendrix, Jan-Olav Henriksen, Gina Hens-Piazza, Carter Heyward, Nicholas J. Higham, David Hilliard, Norman A. Hjelm, Peter C. Hodgson, Arthur Holder, M. Jan Holton, Dwight N. Hopkins, Ronnie Po-chia Hsia, Po-Ho Huang, James Hudnut-Beumler, Jennifer S. Hughes, Leonard M. Hummel, Mary E. Hunt, Laennec Hurbon, Mark Hutchinson, Susan E. Hylen, Mary Beth Ingham, H. Larry Ingle, Dale T. Irvin, Jon Isaak, Paul John Isaak, Ada María Isasi-Díaz, Hans Raun Iversen, Margaret C. Jacob, Arthur James, Maria Jansdotter-Samuelsson, David Jasper, Werner G. Jeanrond, Renée Jeffery, David Lyle Jeffrey, Theodore W. Jennings, David H. Jensen, Robin Margaret Jensen, David Jobling, Dale A. Johnson, Elizabeth A. Johnson, Maxwell E. Johnson, Sarah Johnson, Mark D. Johnston, F. Stanley Jones, James William Jones, John R. Jones, Alissa Jones Nelson, Inge Jonsson, Jan Joosten, Elizabeth Judd, Mulambya Peggy Kabonde, Robert Kaggwa, Sylvester Kahakwa, Isaac Kalimi, Ogbu U. Kalu, Eunice Kamaara, Wayne C. Kannaday, Musimbi Kanyoro, Veli-Matti Kärkkäinen, Frank Kaufmann, Léon Nguapitshi Kayongo, Richard Kearney, Alice A. Keefe, Ralph Keen, Catherine Keller, Anthony J. Kelly, Karen Kennelly, Kathi Lynn Kern, Fergus Kerr, Edward Kessler, George Kilcourse, Heup Young Kim, Kim Sung-Hae, Kim Yong-Bock, Kim Yung Suk, Richard King, Thomas M. King, Robert M. Kingdon, Ross Kinsler, Hans G. Kippenberg, Cheryl A. Kirk-Duggan, Clifton Kirkpatrick, Leonid Kishkovsky, Nadieszda Kizenko, Jeffrey Klaiber, Hans-Josef Klauck, Sidney Knight, Samuel Kobia, Robert Kolb, Karla Ann Koll, Heikki Kotila, Donald Kraybill, Philip D. W. Krey, Yves Krumenacker, Jeffrey Kah-Jin Kuan, Simanga R. Kumalo, Peter Kuzmic, Simon Shui-Man Kwan, Kwok Pui-lan, André LaCocque, Stephen E. Lahey, John Tsz Pang Lai, Emiel Lamberts, Armando Lampe, Craig Lampe, Beverly J. Lanzetta, Eve LaPlante, Lizette Larson-Miller, Ariel Bybee Laughton, Leonard Lawlor, Bentley Layton, Robin A. Leaver, Karen Lebacqz, Archie Chi Chung Lee, Marilyn J. Legge, Hervé LeGrand, D. L. LeMahieu, Raymond Lemieux, Bill J. Leonard, Ellen M. Leonard, Outi Leppä, Jean Lesaulnier, Nantawan Boonprasat Lewis, Henrietta Leyser, Alexei Lidov, Bernard Lightman, Paul Chang-Ha Lim, Carter Lindberg, Mark R. Lindsay, James R. Linville, James C. Livingston, Ann Loades, David Loades, Jean-Claude Loba-Mkole, Lo Lung Kwong, Wati Longchar, Eleazar López, David W. Lotz, Andrew Louth, Robin W. Lovin, William Luis, Frank D. Macchia, Diarmaid N. J. MacCulloch, Kirk R. MacGregor, Marjory A. MacLean, Donald MacLeod, Tomas S. Maddela, Inge Mager, Laurenti Magesa, David G. Maillu, Fortunato Mallimaci, Philip Mamalakis, Kä Mana, Ukachukwu Chris Manus, Herbert Robinson Marbury, Reuel Norman Marigza, Jacqueline Mariña, Antti Marjanen, Luiz C. L. Marques, Madipoane Masenya (ngwan'a Mphahlele), Caleb J. D. Maskell, Steve Mason, Thomas Massaro, Fernando Matamoros Ponce, András Máté-Tóth, Odair Pedroso Mateus, Dinis Matsolo, Fumitaka Matsuoka, John D'Arcy May, Yelena Mazour-Matusevich, Theodore Mbazumutima, John S. McClure, Christian McConnell, Lee Martin McDonald, Gary B. McGee, Thomas McGowan, Alister E. McGrath, Richard J. McGregor, John A. McGuckin, Maud Burnett McInerney, Elsie Anne McKee, Mary B. McKinley, James F. McMillan, Ernan McMullin, Kathleen E. McVey, M. Douglas Meeks, Monica Jyotsna Melanchthon, Ilie Melniciuc-Puica, Everett Mendoza, Raymond A. Mentzer, William W. Menzies, Ina Merdjanova, Franziska Metzger, Constant J. Mews, Marvin Meyer, Carol Meyers, Vasile Mihoc, Gunner Bjerg Mikkelsen, Maria Inêz de Castro Millen, Clyde Lee Miller, Bonnie J. Miller-McLemore, Alexander Mirkovic, Paul Misner, Nozomu Miyahira, R. W. L. Moberly, Gerald Moede, Aloo Osotsi Mojola, Sunanda Mongia, Rebeca Montemayor, James Moore, Roger E. Moore, Craig E. Morrison O.Carm, Jeffry H. Morrison, Keith Morrison, Wilson J. Moses, Tefetso Henry Mothibe, Mokgethi Motlhabi, Fulata Moyo, Henry Mugabe, Jesse Ndwiga Kanyua Mugambi, Peggy Mulambya-Kabonde, Robert Bruce Mullin, Pamela Mullins Reaves, Saskia Murk Jansen, Heleen L. Murre-Van den Berg, Augustine Musopole, Isaac M. T. Mwase, Philomena Mwaura, Cecilia Nahnfeldt, Anne Nasimiyu Wasike, Carmiña Navia Velasco, Thulani Ndlazi, Alexander Negrov, James B. Nelson, David G. Newcombe, Carol Newsom, Helen J. Nicholson, George W. E. Nickelsburg, Tatyana Nikolskaya, Damayanthi M. A. Niles, Bertil Nilsson, Nyambura Njoroge, Fidelis Nkomazana, Mary Beth Norton, Christian Nottmeier, Sonene Nyawo, Anthère Nzabatsinda, Edward T. Oakes, Gerald O'Collins, Daniel O'Connell, David W. Odell-Scott, Mercy Amba Oduyoye, Kathleen O'Grady, Oyeronke Olajubu, Thomas O'Loughlin, Dennis T. Olson, J. Steven O'Malley, Cephas N. Omenyo, Muriel Orevillo-Montenegro, César Augusto Ornellas Ramos, Agbonkhianmeghe E. Orobator, Kenan B. Osborne, Carolyn Osiek, Javier Otaola Montagne, Douglas F. Ottati, Anna May Say Pa, Irina Paert, Jerry G. Pankhurst, Aristotle Papanikolaou, Samuele F. Pardini, Stefano Parenti, Peter Paris, Sung Bae Park, Cristián G. Parker, Raquel Pastor, Joseph Pathrapankal, Daniel Patte, W. Brown Patterson, Clive Pearson, Keith F. Pecklers, Nancy Cardoso Pereira, David Horace Perkins, Pheme Perkins, Edward N. Peters, Rebecca Todd Peters, Bishop Yeznik Petrossian, Raymond Pfister, Peter C. Phan, Isabel Apawo Phiri, William S. F. Pickering, Derrick G. Pitard, William Elvis Plata, Zlatko Plese, John Plummer, James Newton Poling, Ronald Popivchak, Andrew Porter, Ute Possekel, James M. Powell, Enos Das Pradhan, Devadasan Premnath, Jaime Adrían Prieto Valladares, Anne Primavesi, Randall Prior, María Alicia Puente Lutteroth, Eduardo Guzmão Quadros, Albert Rabil, Laurent William Ramambason, Apolonio M. Ranche, Vololona Randriamanantena Andriamitandrina, Lawrence R. Rast, Paul L. Redditt, Adele Reinhartz, Rolf Rendtorff, Pål Repstad, James N. Rhodes, John K. Riches, Joerg Rieger, Sharon H. Ringe, Sandra Rios, Tyler Roberts, David M. Robinson, James M. Robinson, Joanne Maguire Robinson, Richard A. H. Robinson, Roy R. Robson, Jack B. Rogers, Maria Roginska, Sidney Rooy, Rev. Garnett Roper, Maria José Fontelas Rosado-Nunes, Andrew C. Ross, Stefan Rossbach, François Rossier, John D. Roth, John K. Roth, Phillip Rothwell, Richard E. Rubenstein, Rosemary Radford Ruether, Markku Ruotsila, John E. Rybolt, Risto Saarinen, John Saillant, Juan Sanchez, Wagner Lopes Sanchez, Hugo N. Santos, Gerhard Sauter, Gloria L. Schaab, Sandra M. Schneiders, Quentin J. Schultze, Fernando F. Segovia, Turid Karlsen Seim, Carsten Selch Jensen, Alan P. F. Sell, Frank C. Senn, Kent Davis Sensenig, Damían Setton, Bal Krishna Sharma, Carolyn J. Sharp, Thomas Sheehan, N. Gerald Shenk, Christian Sheppard, Charles Sherlock, Tabona Shoko, Walter B. Shurden, Marguerite Shuster, B. Mark Sietsema, Batara Sihombing, Neil Silberman, Clodomiro Siller, Samuel Silva-Gotay, Heikki Silvet, John K. Simmons, Hagith Sivan, James C. Skedros, Abraham Smith, Ashley A. Smith, Ted A. Smith, Daud Soesilo, Pia Søltoft, Choan-Seng (C. S.) Song, Kathryn Spink, Bryan Spinks, Eric O. Springsted, Nicolas Standaert, Brian Stanley, Glen H. Stassen, Karel Steenbrink, Stephen J. Stein, Andrea Sterk, Gregory E. Sterling, Columba Stewart, Jacques Stewart, Robert B. Stewart, Cynthia Stokes Brown, Ken Stone, Anne Stott, Elizabeth Stuart, Monya Stubbs, Marjorie Hewitt Suchocki, David Kwang-sun Suh, Scott W. Sunquist, Keith Suter, Douglas Sweeney, Charles H. Talbert, Shawqi N. Talia, Elsa Tamez, Joseph B. Tamney, Jonathan Y. Tan, Yak-Hwee Tan, Kathryn Tanner, Feiya Tao, Elizabeth S. Tapia, Aquiline Tarimo, Claire Taylor, Mark Lewis Taylor, Bishop Abba Samuel Wolde Tekestebirhan, Eugene TeSelle, M. Thomas Thangaraj, David R. Thomas, Andrew Thornley, Scott Thumma, Marcelo Timotheo da Costa, George E. “Tink” Tinker, Ola Tjørhom, Karen Jo Torjesen, Iain R. Torrance, Fernando Torres-Londoño, Archbishop Demetrios [Trakatellis], Marit Trelstad, Christine Trevett, Phyllis Trible, Johannes Tromp, Paul Turner, Robert G. Tuttle, Archbishop Desmond Tutu, Peter Tyler, Anders Tyrberg, Justin Ukpong, Javier Ulloa, Camillus Umoh, Kristi Upson-Saia, Martina Urban, Monica Uribe, Elochukwu Eugene Uzukwu, Richard Vaggione, Gabriel Vahanian, Paul Valliere, T. J. Van Bavel, Steven Vanderputten, Peter Van der Veer, Huub Van de Sandt, Louis Van Tongeren, Luke A. Veronis, Noel Villalba, Ramón Vinke, Tim Vivian, David Voas, Elena Volkova, Katharina von Kellenbach, Elina Vuola, Timothy Wadkins, Elaine M. Wainwright, Randi Jones Walker, Dewey D. Wallace, Jerry Walls, Michael J. Walsh, Philip Walters, Janet Walton, Jonathan L. Walton, Wang Xiaochao, Patricia A. Ward, David Harrington Watt, Herold D. Weiss, Laurence L. Welborn, Sharon D. Welch, Timothy Wengert, Traci C. West, Merold Westphal, David Wetherell, Barbara Wheeler, Carolinne White, Jean-Paul Wiest, Frans Wijsen, Terry L. Wilder, Felix Wilfred, Rebecca Wilkin, Daniel H. Williams, D. Newell Williams, Michael A. Williams, Vincent L. Wimbush, Gabriele Winkler, Anders Winroth, Lauri Emílio Wirth, James A. Wiseman, Ebba Witt-Brattström, Teofil Wojciechowski, John Wolffe, Kenman L. Wong, Wong Wai Ching, Linda Woodhead, Wendy M. Wright, Rose Wu, Keith E. Yandell, Gale A. Yee, Viktor Yelensky, Yeo Khiok-Khng, Gustav K. K. Yeung, Angela Yiu, Amos Yong, Yong Ting Jin, You Bin, Youhanna Nessim Youssef, Eliana Yunes, Robert Michael Zaller, Valarie H. Ziegler, Barbara Brown Zikmund, Joyce Ann Zimmerman, Aurora Zlotnik, Zhuo Xinping
- Edited by Daniel Patte, Vanderbilt University, Tennessee
-
- Book:
- The Cambridge Dictionary of Christianity
- Published online:
- 05 August 2012
- Print publication:
- 20 September 2010, pp xi-xliv
-
- Chapter
- Export citation