71 results
The role of Europe in Global Psychiatry
- P. Falkai
-
- Journal:
- European Psychiatry / Volume 67 / Issue S1 / April 2024
- Published online by Cambridge University Press:
- 27 August 2024, p. S1
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Abstract
The burden of mental illness in Europe: High individual and societal burden, but only 2% spent on mental health. The tradition of European Psychiatry needs to be strengthened in care, research and teaching.Within the long-term Strategic Mental Health Plan of the EPA the improvement of clinical care research, the “mapping excellence” and “developing core treatment guidelines” require further action. Researching the influence of environmental stressors on the development and maintenance of mental illness and fostering stepped care approaches to improve resilience are none the less important. Furthermore, to improve the reliability and especially validity of diagnoses of mental disorders by introducing (bio)markers and defining dimensions of mental illnesses using big data and predictive sciences are just as important as an enforced research on reducing stigma and discrimination of mental disorders.
Disclosure of InterestNone Declared
Preliminary data from the CONNEX-X extension trial examining the long-term safety of iclepertin in patients with schizophrenia who completed Phase III CONNEX trials
- C. Reuteman-Fowler, Z. Blahova, S. Marder, S. Ikezawa, P. Falkai
-
- Journal:
- European Psychiatry / Volume 67 / Issue S1 / April 2024
- Published online by Cambridge University Press:
- 27 August 2024, pp. S91-S92
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Introduction
Cognitive impairment associated with schizophrenia (CIAS) is an important unmet need as there are no effective treatments available. Iclepertin (BI 425809), a glycine transporter-1 inhibitor, has been shown to improve CIAS in Phase II trials, and Phase III trials are underway.
ObjectivesThe ongoing CONNEX-X extension study aims to collect additional safety data relating to iclepertin treatment in patients with CIAS.
MethodsCONNEX-X (NCT05211947/1346-0014) is a multinational, multicentre, open-label, single-arm extension study in patients with CIAS who completed 26 weeks of treatment (iclepertin 10 mg or placebo) in one of 3 Phase III CONNEX parent trials (NCT04846868/1346-0011, NCT04846881/1346-0012, NCT04860830/1346-0013). An estimated 1400 clinically stable outpatients will be treated (iclepertin 10 mg daily) for 1 year, irrespective of previous treatment (iclepertin/placebo). Patients are excluded if any of the following circumstances occur during the parent study and up to Visit 1 of CONNEX-X: suicidal behaviour or ideation (type 5 on the Columbia-Suicide Severity Rating Scale), diagnosis with moderate/severe substance use disorder, diagnosis other than schizophrenia (according to Diagnostic and Statistical Manual of Mental Disorders – Fifth Edition), development of any condition preventing participation, a haemoglobin level decrease (>25% or <100g/L from baseline in parent trial) or haemoglobinopathies. The primary endpoint is the occurrence of treatment-emergent adverse events. The secondary endpoints include change from baseline (CfB) in Clinical Global Impressions-Severity (CGI-S) and CfB in haemoglobin. Further efficacy endpoints include CfB in MATRICS Consensus Cognitive Battery (MCCB) overall composite T-score, CfB in Schizophrenia Cognition Rating Scale total score and CfB in Virtual Reality Functional Capacity Assessment Tool (VRFCAT) total times.
ResultsCurrently, 460 patients have been enrolled and randomised from the parent trials with 0% screening failures (-80% roll-over rate, 30 August 2023). Current study status, including recruitment, screening failures and data collection experiences, are presented.
ConclusionsPatient enrolment rates from the CONNEX trials to the CONNEX-X open-label extension study are stable. CONNEX-X will allow the exploration of long-term safety, as well as descriptive analyses of cognitive and functional endpoints of iclepertin in the treatment of CIAS.
FundingBoehringer Ingelheim
Disclosure of InterestC. Reuteman-Fowler Employee of: Boehringer Ingelheim Pharmaceuticals, Inc., Z. Blahova Employee of: Boehringer Ingelheim RCV GmbH & Co. KG, S. Marder Consultant of: Boehringer Ingelheim Pharma GmbH, Merck, Biogen and Sunovion, S. Ikezawa Consultant of: Boehringer Ingelheim Pharma GmbH, Lundbeck, Takeda Pharma, Sumitomo Dainippon Pharma, Employee of: International University of Health and Welfare, Mita Hospital, Tokyo, Japan, P. Falkai Consultant of: Boehringer Ingelheim Pharma GmbH, Boehringer Ingelheim Pharma advisory board
Does war increases the risk for psychoses?
- P. Falkai
-
- Journal:
- European Psychiatry / Volume 67 / Issue S1 / April 2024
- Published online by Cambridge University Press:
- 27 August 2024, p. S15
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Abstract
The World Health Organization (WHO) has stated that in situations of armed conflict, “Around 10 percent of the people who experience traumatic events will have serious mental health problems, and another 10 percent will develop behavior that will hinder their ability to function effectively.” Problems include post-traumatic stress disorder, anxiety, depression, substance misuse, and possibly precipitation of psychosis. War has a catastrophic effect on the health and well being of nations. Studies have shown that conflict situations cause more mortality and disability than any major disease. Only through a greater understanding of conflicts and the myriad of mental health problems that arise from them, coherent and effective strategies for dealing with such problems can be developed.
Disclosure of InterestNone Declared
Cardiovascular and metabolic issues in the treatment of schizophrenia: focus on the management of negative symptoms
- P. Falkai
-
- Journal:
- European Psychiatry / Volume 67 / Issue S1 / April 2024
- Published online by Cambridge University Press:
- 27 August 2024, p. S18
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Abstract
Mortality from cardiovascular disease is increased in people with mental health disorders in general and schizophrenia in particular. The causes are multifactorial, but it is known that antipsychotic medication can cause cardiac side-effects beyond the traditional coronary risk factors. Schizophrenia itself is a contributor to an increased risk of cardiovascular mortality via cardiac autonomic dysfunction and a higher prevalence of metabolic syndrome, both contributing to a reduced life expectancy.
Overall, management of cardiovascular risk within this population group must be multifaceted and nuanced to allow the most effective treatment of serious mental illness to be conducted within acceptable parameters of cardiovascular risk; some practical measures are presented for the clinical cardiologist.
Disclosure of InterestNone Declared
Exploring Cariprazine’s Potential in Late-Stage Schizophrenia Treatment
- P. Falkai, R. Csehi, K. Acsai, G. Németh
-
- Journal:
- European Psychiatry / Volume 67 / Issue S1 / April 2024
- Published online by Cambridge University Press:
- 27 August 2024, p. S211
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Introduction
Schizophrenia is a chronic neuropsychiatric disorder that often requires long-term pharmacotherapy to manage symptoms and prevent relapse. There are important clinical differences between early-stage versus late-stage schizophrenia, like the predominant symptomatology. In later stages, negative, cognitive, and anxiety/depressive symptoms dominate the clinical picture, with relapses further potentiating the emergence of positive symptoms. Therefore, it is crucial to establish the efficacy of an antipsychotic medication in the later stages of schizophrenia as well. Cariprazine is a novel dopamine D3-preferring D3/D2 receptor partial agonist that has shown efficacy in treating schizophrenia across the symptom spectrum.
ObjectivesThe aim of this poster is to present the findings of cariprazine’s efficacy in treating late-stage schizophrenia, especially in symptoms that are more commonly occurring in this phase of the disorder.
MethodsThis poster reports the results of a post-hoc pooled analysis of three 6-week, double-blind, placebo-controlled trials (NCT01104766, NCT01104779, NCT00694707) that assessed the efficacy of cariprazine in schizophrenia. The primary outcome was the change in Positive and Negative Syndrome Scale (PANSS) Total Scores from baseline to endpoint. The analysis focused on patients with late-stage schizophrenia (defined as having an illness-duration of more than 15 years) who received cariprazine at doses between 1.5 mg/day to 6.0 mg/day. The changes in PANSS-derived Marder Factor Scores for Negative, Disorganised Thought (i.e., Cognitive) and Anxiety/Depression symptoms were further examined. The least square mean differences (LSMDs) between cariprazine and placebo groups were calculated using mixed-models for repeated measures (MMRM).
ResultsAltogether, 128 placebo-, and 286 cariprazine-treated patients were identified as having schizophrenia for more than 15 years. The mean age of patients was about 45 years, while the mean illness-duration was about 24 years. The mean baseline PANSS scores were the same between the two groups. In the late-stage schizophrenia population, at Week 6, cariprazine yielded statistically significantly greater reductions on the PANSS Total Score (LSMD -6.7, p<0.01). Cariprazine further showed superiority over placebo in reducing negative (LSMD -1.4, p<0.05), disorganised thought (LSMD -1.3, p<0.01), and anxiety/depression (LSMD -0.9, p<0.05) symptoms.
ConclusionsCariprazine showed efficacy in treating patients with late-stage schizophrenia. It improved overall schizophrenia symptoms, as well as the negative, cognitive and anxiety/depression symptoms that are more prevalent in this phase of the disorder.
Disclosure of InterestP. Falkai Consultant of: Janssen-Cilag, AstraZeneca, Lilly, and Lundbeck, Speakers bureau of: AstraZeneca, Bristol Myers Squibb, Lilly, Essex, GE Healthcare, GlaxoSmithKline, Gedeon Richter, Janssen Cilag, Lundbeck, Otsuka, Pfizer, Servier, and Takeda, R. Csehi Employee of: Gedeon Richter Plc, K. Acsai Employee of: Gedeon Richter Plc, G. Németh Employee of: Gedeon Richter Plc
How good is the clinical diagnosis in schizophrenia? Reliability and validity
- P. Falkai
-
- Journal:
- European Psychiatry / Volume 67 / Issue S1 / April 2024
- Published online by Cambridge University Press:
- 27 August 2024, p. S13
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Abstract
Several changes to the classification of mental disorders have been made during the past half century to increase the reliability, clinical use and validity of the diagnostic classification. Despite the high expansion of knowledge about mental disorders, understanding of their components and processes still requires fine-tuning. This symposium identifies key issues on different classification systems with different purposes relevant to understanding and classifying mental disorders. We discuss how key issues such as ICD-11, RDoC or Biomarkers correspond or diverge because of their different purposes, and constituencies. Although these approaches have varying degrees of overlap and distinguishing features, they share the goal of reducing the burden of suffering due to mental disorder.
Disclosure of InterestNone Declared
Multivariate associations between psychiatric drug intake and grey matter volume changes in individuals at early stages of psychosis and depression
- C. Weyer, D. Popovic, A. Ruef, L. Hahn, E. Sarişik, J. Fanning, J. Kambeitz, R. K. Salokangas, J. Hietala, A. Bertolino, S. Borgwardt, P. Brambilla, R. Upthegrove, S. J. Wood, R. Lencer, E. Meisenzahl, P. Falkai, N. Koutsouleris
-
- Journal:
- European Psychiatry / Volume 67 / Issue S1 / April 2024
- Published online by Cambridge University Press:
- 27 August 2024, pp. S272-S273
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Introduction
Psychiatric drugs, including antipsychotics and antidepressants, are widely prescribed, even in young and adolescent populations at early or subthreshold disease stages. However, their impact on brain structure remains elusive. Elucidating the relationship between psychotropic medication and structural brain changes could enhance the understanding of the potential benefits and risks associated with such treatment.
ObjectivesInvestigation of the associations between psychiatric drug intake and longitudinal grey matter volume (GMV) changes in a transdiagnostic sample of young individuals at early stages of psychosis or depression using an unbiased data-driven approach.
MethodsThe study sample comprised 247 participants (mean [SD] age = 25.06 [6.13] years, 50.61% male), consisting of young, minimally medicated individuals at clinical high-risk states for psychosis, individuals with recent-onset depression or psychosis, and healthy control individuals. Structural magnetic resonance imaging was used to obtain whole-brain voxel-wise GMV for all participants at two timepoints (mean [SD] time between scans = 11.15 [4.93] months). The multivariate sparse partial least squares (SPLS) algorithm (Monteiro et al. JNMEDT 2016; 271:182-194) was embedded in a nested cross-validation framework to identify parsimonious associations between the cumulative intake of psychiatric drugs, including commonly prescribed antipsychotics and antidepressants, and change in GMV between both timepoints, while additionally factoring in age, sex, and diagnosis. Furthermore, we correlated the retrieved SPLS results to personality domains (NEO-FFI) and childhood trauma (CTQ).
ResultsSPLS analysis revealed significant associations between the antipsychotic classes of benzamides, butyrophenones and thioxanthenes and longitudinal GMV decreases in cortical regions including the insula, posterior superior temporal sulcus as well as cingulate, postcentral, precentral, orbital and frontal gyri (Figure 1A-C). These brain regions corresponded most closely to the dorsal and ventral attention, somatomotor, salience and default network (Figure 1D). Furthermore, the medication signature was negatively associated with the personality domains extraversion, agreeableness and conscientiousness and positively associated with the CTQ domains emotional and physical neglect.
Image:
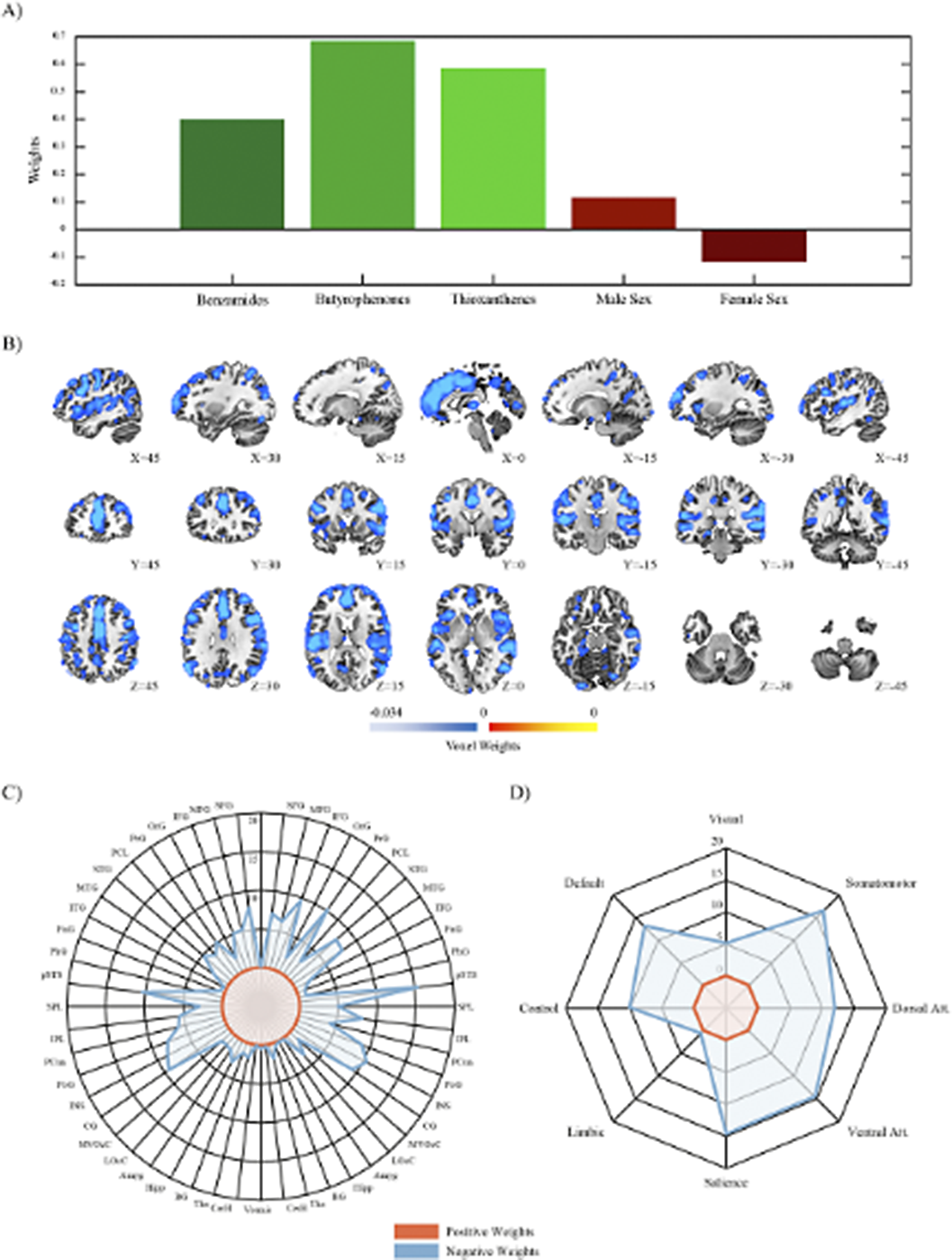 Conclusions
ConclusionsPsychiatric drug intake over a period of one year was linked to distinct GMV reductions in key cortical hubs. These patterns were already visible in young individuals at early or subthreshold stages of mental illness and were further linked to childhood neglect and personality traits. Hence, a better and more in-depth understanding of the structural brain implications of medicating young and adolescent individuals might lead to more cautious, sustainable and targeted treatment strategies.
Disclosure of InterestNone Declared
The Phase III CONNEX programme assessing the efficacy and safety of iclepertin in patients with schizophrenia: Trial design and recruitment update
- C. Reuteman-Fowler, Z. Blahova, S. Ikezawa, S. Marder, P. Falkai, J. H. Krystal
-
- Journal:
- European Psychiatry / Volume 67 / Issue S1 / April 2024
- Published online by Cambridge University Press:
- 27 August 2024, pp. S87-S88
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Introduction
In a 12-week, Phase II (NCT02832037) trial, iclepertin (BI 425809), an inhibitor of glycine transporter-1, was generally well tolerated and significantly improved cognition in 509 patients with schizophrenia.
ObjectivesThe Phase III CONNEX programme aims to confirm the efficacy, safety and tolerability of iclepertin in improving cognition and functioning across a larger cohort of patients with schizophrenia.
MethodsThe CONNEX programme includes 3 randomised, double-blind, placebo-controlled parallel group trials in patients with schizophrenia (NCT04846868, NCT04846881, NCT04860830) receiving stable antipsychotic treatment. Each trial aims to recruit ˜586 patients, 18–50 years old, treated with 1–2 antipsychotic medications (≥12 weeks on current drug and ≥35 days on current dose before treatment) who have functional impairment in day-to-day activities and interact ≥1 hour per week with a designated study partner. Patients with cognitive impairment due to developmental, neurological or other disorders, with a current DSM-5 diagnosis other than schizophrenia or receiving cognitive remediation therapy within 12 weeks prior to screening, will be excluded. Patients will be recruited from multiple centres across 41 countries in Asia, North and South America, Europe and the Asia-Pacific Region, and randomised 1:1 to receive either iclepertin 10 mg (oral administration; n=293), or placebo (n=293) once daily for 26 weeks. The primary endpoint is change from baseline in overall composite T-score of the Measurement and Treatment Research to Improve Cognition in Schizophrenia Consensus Cognitive Battery. The key secondary endpoints are change from baseline in total score on the Schizophrenia Cognition Rating Scale and change from baseline in the adjusted total time T-score in the Virtual Reality Functional Capacity Assessment Tool.
ResultsThe CONNEX programme is currently recruiting (Table); the first patients were enrolled in Aug–Sept 2021 and completion is expected in Q1 2025. The presentation will describe the current study status, information relating to screening failures, and the experience of collecting these data as part of a large multi-country, multicentre study.
Table. The number of patients recruited by 31 August 2023
CONNEX 1 CONNEX 2 CONNEX 3 Screened 565 521 493 Randomised 409 360 350 Completed trial medication 202 184 191 ConclusionsIclepertin may represent the first efficacious medication for cognitive impairment associated with schizophrenia.
FundingBoehringer Ingelheim
Disclosure of InterestC. Reuteman-Fowler Employee of: Boehringer Ingelheim, Z. Blahova Employee of: Boehringer Ingelheim, S. Ikezawa Consultant of: Boehringer Ingelheim Pharma GmbH, Lundbeck, Takeda Pharma, Sumitomo Dainippon Pharma, Employee of: International University of Health and Welfare, Mita Hospital, Tokyo, Japan, S. Marder Consultant of: Boehringer Ingelheim Pharma GmbH, Merck, Biogen and Sunovion, P. Falkai Consultant of: Boehringer Ingelheim Pharma GmbH, Boehringer Ingelheim Pharma Advisory Board, J. H. Krystal Shareolder of: Freedom Biosciences, Inc., Biohaven Pharmaceuticals, Sage Pharmaceuticals, Spring Care, Biohaven Pharmaceuticals Medical Sciences, EpiVario, RBNC Therapeutics, Terran Biosciences and Tempero Bio, Consultant of: Aptinyx, Atai Life Sciences, AstraZeneca Pharmaceuticals, Biogen, Biomedisyn Corporation, Bionomics, Boehringer Ingelheim International, Cadent Therapeutics, Clexio Bioscience, COMPASS Pathways, Concert Pharmaceuticals, Epiodyne, EpiVario, Greenwich Biosciences, Heptares Therapeutics, Janssen, Jazz Pharmaceuticals, Otsuka America Pharmaceutical, Perception Neuroscience Holdings, Spring Care, Sunovion Pharmaceuticals, Takeda Industries, Taisho Pharmaceutical Co.; Biohaven Pharmaceuticals, BioXcel Therapeutics, Cadent Therapeutics, Cerevel Therapeutics, Delix Therapeutics, EpiVario, Eisai, Jazz Pharmaceuticals, Novartis, PsychoGenics, RBNC Therapeutics, Tempero Bio and Terran Biosciences Advisory Boards
Exploring Associations between Grey Matter Volume and Clinical High-Risk for Psychosis: A Transdiagnostic Study Utilizing the NAPLS-2 Risk Calculator in the PRONIA Cohort
- L.-M. Neuner, L. Hahn, J. Kambeitz, R. K. Salokangas, J. Hietala, A. Bertolino, S. Borgwardt, P. Brambilla, R. Upthegrove, S. J. Wood, R. Lencer, E. Meisenzahl, P. Falkai, T. D. Cannon, N. Koutsouleris
-
- Journal:
- European Psychiatry / Volume 67 / Issue S1 / April 2024
- Published online by Cambridge University Press:
- 27 August 2024, pp. S271-S272
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Introduction
The clinical high-risk state for psychosis (CHR) is associated with alterations in grey matter volume (GMV) in various regions such as the hippocampus (Vissink et al. BP:GOS 2022; 2(2) 147-152). Within the scope of the North American Prodrome Longitudinal Study (NAPLS-2; Cannon et al. AM J Psychiatry 2016; 173(10), 980-988), a publicly available risk calculator based on clinical variables was developed to assess the likelihood of individuals to transition to psychosis within a 2-year period.
ObjectivesIn the current study, we aim to examine the association between GMV and NAPLS-2 risk scores calculated for individuals with CHR and recent-onset depression (ROD), taking a transdiagnostic approach on the transition to psychosis.
MethodsThe sample consisted of 315 CHR (M = 23.85, SD = ± 5.64; female: 164) and 295 ROD (M = 25.11, SD = ± 6.21; female: 144) patients from the multi-site Personalised Prognostic Tools for Early Psychosis Management (PRONIA) Study (Koutsouleris et al. JAMA Psychiatry 2018; 57(11), 1156-1172). Risk scores were calculated using the six clinical and neurocognitive variables included in the NAPLS-2 risk calculator that were significant for predicting psychosis. Further, we derived smoothed GMV maps from T1-weighted structural magnetic resonance imaging using a full width at half maximum kernel size of 8 mm. We employed a multiple regression design in SPM12 to examine associations between risk scores and GMV. On the whole-brain level, we calculated permutation-based threshold-free cluster enhancement (TFCE) contrasts using the TFCE toolbox. Additionally, we calculated t-contrasts within a region-of-interest (ROI) analysis encompassing the hippocampus. All results were thresholded at p < 0.05 with family wise error correction to address multiple comparisons.
ResultsOur analysis revealed that linear GMV increases in the right middle and superior frontal gyrus (kE= 2726 voxels) were significantly associated with higher risk for psychosis transition within two years (see figure 1, highlighted in blue). In the ROI analysis, we found a significant negative linear association between GMV decreases in the left hippocampus (kE = 353 voxels) and higher risk for psychosis transition (see figure 1, highlighted in red).
Image:
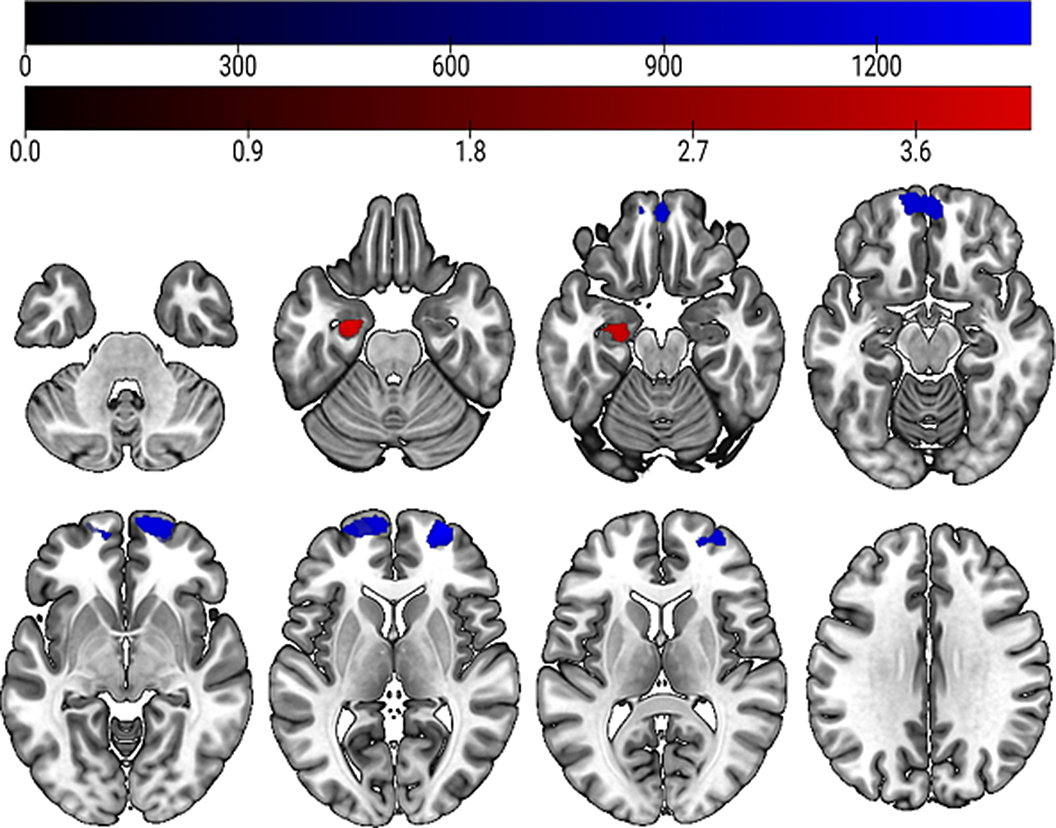 Conclusions
ConclusionsGMV reductions in the hippocampus have frequently been observed in CHR and psychosis patients (Vissink et al. BP:GOS 2022; 2(2) 147-152), therefore our results further highlight the crucial role of this region in the progression of the disease. There is limited evidence on GMV increases in CHR patients. However, the GMV increase we found in the frontal pole may reflect compensatory mechanisms of the brain in the development of psychosis. In addition, we were able to provide biological validation of the NAPLS-2 risk calculator and its assessment of risk for transition to psychosis.
Disclosure of InterestNone Declared
Efficacy and safety of iclepertin (BI 425809) in patients with schizophrenia: CONNEX, a Phase III randomised controlled trial programme
- P. Falkai, C. Reuteman-Fowler, Z. Blahova, S. Ikezawa, S. R. Marder, J. H. Krystal
-
- Journal:
- European Psychiatry / Volume 66 / Issue S1 / March 2023
- Published online by Cambridge University Press:
- 19 July 2023, p. S637
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Introduction
Cognitive impairment (CI) is a major determinant of poor functional outcome in schizophrenia and there are currently no available pharmacotherapies. Deficits in glutamatergic signalling play a key role in the neuropathology of cognitive symptoms. Iclepertin (BI 425809), an inhibitor of glycine transporter-1, enhances glutamatergic signalling by increasing synaptic levels of the N-methyl-D-aspartate receptor co-agonist, glycine. A 12-wk, Phase II trial (NCT02832037) in 509 patients (pts) with schizophrenia demonstrated that iclepertin was well tolerated and significantly improved cognition.
ObjectivesThe Phase III CONNEX programme aims to confirm the efficacy, safety and tolerability of iclepertin in improving cognition and functioning in a larger cohort of pts.
MethodsCONNEX consists of three replicate randomised, double-blind, placebo-controlled parallel-group trials in pts with schizophrenia (NCT04846868, NCT04846881, NCT04860830) currently stable on antipsychotic treatment. Each trial aims to recruit ~586 pts, 18–50 years old, treated with 1–2 antipsychotic medications (≥12 wks on current drug; ≥35 days on current dose prior to treatment), who have functional impairment in day-to-day activities and interact ≥1 hr per wk with a designated study partner. Pts with CI due to developmental, neurological or other disorders, or receiving cognitive remediation therapy within 12 wks prior to screening, will be excluded. Pts will be recruited from 39 countries in Asia, Australia, New Zealand, North and South America and Europe, and randomised 1:1 to receive either oral iclepertin 10 mg (n=293) or placebo (n=293) once daily over 26 wks. The primary efficacy endpoint is change from baseline (CfB) in the MATRICS Consensus Cognitive Battery overall composite T-score. Key secondary efficacy endpoints are CfB in Schizophrenia Cognition Rating Scale total score and CfB in the adjusted total time in the Virtual Reality Functional Capacity Assessment Tool. Long-term safety and tolerability data will be collected in an open-label safety extension study (CONNEX-X).
ResultsThe studies are currently recruiting (first pts enrolled Aug–Sept 2021), with completion expected in Q2 2024. Here we present an overview of the current study status, including any information relating to screening failures and the experience of collecting these data as part of a large multicountry, multicentre study.
ConclusionsTo date, most large, industry-sponsored studies testing various compounds to address cognitive function have failed to show proof-of-clinical concept. Demonstration of efficacy of iclepertin in improving cognition in this Phase III programme would provide important insight into the role of glutamate in cognitive symptoms, that may also have relevance for other cognitive disorders. Iclepertin may represent the first efficacious medication for CI associated with schizophrenia.
Disclosure of InterestP. Falkai Consultant of: Advistory board Boehringer Ingelheim Pharma GmBH & Co. KG, C. Reuteman-Fowler Employee of: Boehringer Ingelheim Pharma GmBH & Co. KG, Z. Blahova Employee of: Boehringer Ingelheim Pharma GmBH & Co. KG, S. Ikezawa Consultant of: Financial agreements with Boehringer Ingelheim Pharma GmbH, Merck, Biogen and Sunovion, S. Marder Grant / Research support from: Boehringer Ingelheim, GW Pharma, Consultant of: Boehringer Ingelheim, Merck, Sunovion, J. Krystal Shareolder of: Co-founder of Freedom Biosciences, Inc Investments in Biohaven Pharmaceuticals, Sage Pharmaceuticals, Spring Care, Biohaven Pharmaceuticals Medical Sciences, EpiVario, RBNC Therapeutics, Terran Biosciences and Tempero Bio., Consultant of: Aptinyx, Atai Life Sciences, AstraZeneca Pharmaceuticals, Biogen, Biomedisyn Corporation, Bionomics, Boehringer Ingelheim International, Cadent Therapeutics, Clexio Bioscience, COMPASS Pathways, Concert Pharmaceuticals, Epiodyne, EpiVario, Greenwich Biosciences, Heptares Therapeutics, Janssen, Jazz Pharmaceuticals, Otsuka America Pharmaceutical, Perception Neuroscience Holdings, Spring Care, Sunovion Pharmaceuticals, Takeda Industries, Taisho Pharmaceutical Co Advistory board: Biohaven Pharmaceuticals, BioXcel Therapeutics, Cadent Therapeutics, Cerevel Therapeutics, Delix Therapeutics, EpiVario, Eisai, Jazz Pharmaceuticals, Novartis, PsychoGenics, RBNC Therapeutics, Tempero Bio and Terran Biosciences;
Characteristics of Adults Hospitalized for a Major Depressive Disorder: Results from the Multicenter OASIS-D Study
- C. U. Correll, F. Bermpohl, N. Schoofs, R. Bathe-Peters, K. Pfeifer, P. Falkai, C. Schüle, F. Pan-Montojo, E. Y. M. Wang, A. Reif, C. Reif-Leonhard, S. Schillo, P. Getty, M. Adli, R. Papenfuß, F. Jessen, F. Salimi-Dafsari, M. Bauer, U. Lewitzka, C. Otte, L. Graumann, D. Piber, S. Weyn-Banningh, A. Meyer-Lindenberg, A. Böhringer, F. Heuer, V. B. Nöhles
-
- Journal:
- European Psychiatry / Volume 66 / Issue S1 / March 2023
- Published online by Cambridge University Press:
- 19 July 2023, pp. S346-S347
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Introduction
Major Depressive Disorder (MDD) is one of the most common mental illnesses worldwide and is strongly associated with suicidality. Commonly used treatments for MDD with suicidality include crisis intervention, oral antidepressants (although risk of suicidal behavior is high among non-responders and during the first 10-14 days of the treatment) benzodiazepines and lithium. Although several interventions addressing suicidality exist, only few studies have characterized in detail patients with MDD and suicidality, including treatment, clinical course and outcomes. Patient Characteristics, Validity of Clinical Diagnoses and Outcomes Associated with Suicidality in Inpatients with Symptoms of Depression (OASIS-D)-study is an investigator-initiated trial funded by Janssen-Cilag GmbH.
ObjectivesFor population 1 out of 3 OASIS-D populations, to assess the sub-population of patients with suicidality and its correlates in hospitalized individuals with MDD.
MethodsThe ongoing OASIS-D study consecutively examines hospitalized patients at 8 German psychiatric university hospitals treated as part of routine clinical care. A sub-group of patients with persistent suicidality after >48 hours post-hospitalization are assessed in detail and a sub-group of those are followed for 6 months to assess course and treatment of suicidality associated with MDD. The present analysis focuses on a preplanned interim analysis of the overall hospitalized population with MDD.
ResultsOf 2,049 inpatients (age=42.5±15.9 years, females=53.2%), 68.0% had severe MDD without psychosis and 21.2% had moderately severe MDD, with 16.7% having treatment-resistant MDD. Most inpatients referred themselves (49.4%), followed by referrals by outpatient care providers (14.6%), inpatient care providers (9.0%), family/friends (8.5%), and ambulance (6.8%). Of these admissions, 43.1% represented a psychiatric emergency, with suicidality being the reason in 35.9%. Altogether, 72.4% had at least current passive suicidal ideation (SI, lifetime=87.2%), including passive SI (25.1%), active SI without plan (15.5%), active SI with plan (14.2%), and active SI with plan+intent (14.1%), while 11.5% had attempted suicide ≤2 weeks before admission (lifetime=28.7%). Drug-induced mental and behavioral disorders (19.6%) were the most frequent comorbid disorders, followed by personality disorders (8.2%). Upon admission, 64.5% were receiving psychiatric medications, including antidepressants (46.7%), second-generation antipsychotics (23.0%), anxiolytics (11.4%) antiepileptics (6.0%), and lithium (2.8%). Altogether, 9.8% reported nonadherence to medications within 6 months of admission.
ConclusionsIn adults admitted for MDD, suicidality was common, representing a psychiatric emergency in 35.9% of patients. Usual-care treatments and outcomes of suicidality in hospitalized adults with MDD require further study.
Disclosure of InterestNone Declared
Covid-19: Lessons for Mental and Brain Health
- P. Falkai
-
- Journal:
- European Psychiatry / Volume 65 / Issue S1 / June 2022
- Published online by Cambridge University Press:
- 01 September 2022, p. S9
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Psychiatry is facing major challenges during times of a pandemic as illustrated by the current COVID-19 pandemic. The challenges involve its actual and perceived role within the medical system, in particular how psychiatric hospitals can maintain their core mission of attending to the mentally ill while at the same time providing relief to general medicine. Since psychiatric disorders are the top leading causes of global burden of disease, we need to strengthen mental health care in the wake of the massive onslaught of the pandemic. While nobody can deny the need to act decisively and swiftly and ramp up intensive care readiness, we believe that the immediate availability of psychiatric care is just as important. In order to provide the best possible treatment conditions for people suffering from mental illness but as well for those suffering from the immediate pandemic’s consequences such as isolation, reduced social interaction etc. instant and comprehensive provision is inevitable.
DisclosureNo significant relationships.
Effects of Exercise in People with Severe Mental Illness and Recommendations for its Implementation as Add-on Therapy
- I. Maurus, L. Röll, D. Keeser, A. Schmitt, A. Hasan, D. Hirjak, A. Meyer-Lindenberg, P. Falkai
-
- Journal:
- European Psychiatry / Volume 65 / Issue S1 / June 2022
- Published online by Cambridge University Press:
- 01 September 2022, p. S21
-
- Article
-
- You have access Access
- Open access
- Export citation
-
There are many reasons for people with (and without) severe mental illness to exercise regularly. In people with schizophrenia, major depression and bipolar disorder, it has already been shown that regular physical activity as an add-on therapy can improve quality of life and symptom severity. This is particularly important in domains that standard therapy is currently not able to treat sufficiently, such as cognitive deficits. Postulated underlying neurobiological effects include increased volume in hippocampal areas as demonstrated by data of a current clinical trial in people with schizophrenia.
Furthermore, regular exercise is essential to counteract the increased cardiovascular morbidity and mortality of people with severe mental illness. However, most people with severe mental illness do not achieve the recommended amount of physical activity and the potential of exercise as an add-on therapy is currently not even close to being fully realized. On the one hand, it is important that mental health staff also considers the physical condition of patients with mental illnesses and counsels them on their health behavior. On the other hand, there is a need for individually adapted training programs delivered by qualified exercise professionals that incorporate motivational and adherence strategies. Examples of barriers and facilitators for the implementation of exercise as an add-on therapy are discussed on the basis of current local projects.
DisclosureNo significant relationships.
The efficacy of cariprazine in chronic schizophrenia – post hoc analyses of phase II/III clinical trials
- P. Falkai, Z. Dombi, K. Acsai, Á. Barabássy, G. Németh
-
- Journal:
- European Psychiatry / Volume 65 / Issue S1 / June 2022
- Published online by Cambridge University Press:
- 01 September 2022, p. S323
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Introduction
Chronic schizophrenia patients are experiencing persistent and severe illness for more than 15-20 years and are usually suffering from long-term negative symptoms. Cariprazine, a novel D3-D2 partial agonist has been proven to be effective in the treatment of acute schizophrenia, however its ability to treat chronic patients has not been assessed yet.
ObjectivesThe primary aim of the present post-hoc analysis is to assess the efficacy of cariprazine in treating patients with chronic schizophrenia (late-stage and residual schizophrenia patients).
MethodsData from 3 phase II/III 6-week, randomized, double-blind, placebo-controlled trials with similar design in patients with acute exacerbation of schizophrenia were pooled and patients with more than 15 years of schizophrenia were analysed (late-stage patients). Furthermore, schizophrenia patients experiencing predominantly negative symptoms from a 26-week, randomized, double-blind, active-controlled, fixed-flexible-dose trial with an ICD-10 code of F20.5 were analysed post-hoc (residual patients).
ResultsAltogether, 414 late stage (286 cariprazine and 128 placebo) and 35 residual (23 cariprazine and 12 risperidone) patients were identified. The pooled analysis evaluating mean change from baseline to week 6 in the PANSS total score indicated statistically significant difference in favour of cariprazine in the late stage (LSMD -6.7, p<0.01) subpopulation compared to placebo. The mean change from baseline in patients with residual schizophrenia in the cariprazine arm was -9.6 on the PANSS-FSNS scale, while -7.9 in the risperidone arm.
ConclusionsBased on the results, it seems that cariprazine might be a good treatment option for patients with chronic schizophrenia. Nonetheless, further studies are needed to confirm this.
DisclosureI am an employee of Gedeon Richter Plc.
How to Improve the Physical Health in Patients with Severe Mental Disorders
- P. Falkai
-
- Journal:
- European Psychiatry / Volume 65 / Issue S1 / June 2022
- Published online by Cambridge University Press:
- 01 September 2022, pp. S16-S17
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Schizophrenia is a severe brain disorder characterised by positive, negative, affective and cognitive symptoms and can be regarded as a disorder of impaired neural plasticity. This lecture focusses on the beneficial role of exercise in schizophrenia and its underlying mechanisms.Apart from the established pharmacological treatments in schizophrenia, aerobic exercise has a profound impact on the plasticity of the brain of both rodents and humans such as inducing the proliferation and differentiation of neural progenitor cells of the hippocampus in mice and rats. Aerobic exercise enhances LTP and leads to a better performance in hippocampus related memory tasks, eventually by increasing metabolic and synaptic plasticity related proteins in the hippocampus. In healthy humans, regular aerobic exercise increases hippocampal volume and seems to diminish processes of ageing like brain atrophy and cognitive decline.Several meta-analyses demonstrate the beneficial effect of exercise on function, positive as well as negative symptoms and brain structure in multi-episode schizophrenia.
DisclosureNo significant relationships.
Integration of real-world clinical data into the Munich Mental Health Biobank – clinical and scientific potential and challenges
- J. Kálmán, G. Burkhardt, O. Pogarell, F. Padberg, T. Schulze, P. Falkai
-
- Journal:
- European Psychiatry / Volume 65 / Issue S1 / June 2022
- Published online by Cambridge University Press:
- 01 September 2022, p. S568
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Introduction
New insights into the pathophysiology of mental disorders and innovations in psychiatric care depend on the availability of representative, longitudinal and multidimensional datasets across diverse, transdiagnostic populations. Biobanks usually attempt to collect such data in parallel to clinical routine, which is resource-intensive, puts additional burden on health-care providers, and may reduce the generalizability of the results. Despite containing rich phenotypic and biological information, data generated in routine clinical care is seldomly used for research purposes, because it is usually unstructured and locked in data silos. To truly link clinical practice and research, solutions that optimize the generation and scientific utilization of real-world clinical data are needed.
ObjectivesEvaluation of a new digital infrastructure which warrants the efficient, automatized, and structured collection of real-world data in psychiatric care, and integrates the generated data into existing biobanking efforts.
MethodsWe have developed a new documentation system which augments the existing IT-structures, enables the collection of routine clinical data in a structured format and involves patients in the data generation process. In an implementation science approach, to replicate and extend the findings of Blitz et al. (JMIR Ment Health 2021), we are investigating the acceptance, efficacy, and safety of the system in our outpatient clinic for affective disorders.
ResultsFirst results describing the technical safety, usage metrics, and acceptance of the system, and the quality of the collected data will be presented.
ConclusionsChallenges of collecting real-world data for biobanking and research purposes and perspectives on future digital solutions will be discussed.
DisclosureNo significant relationships.
COVID-19 vaccination rates in hospitalized mentally ill patients compared to the general population in Germany: Results from the COVID Ψ Vac study
- Hauke F. Wiegand, Birgit Maicher, Mike Rueb, Paula Wessels, Bianca Besteher, Sabine Hellwig, Andrea Pfennig, Henrik Rohner, Stefan Unterecker, Lars P. Hölzel, Alexandra Philipsen, Katharina Domschke, Peter Falkai, Klaus Lieb, Kristina Adorjan
-
- Journal:
- European Psychiatry / Volume 65 / Issue 1 / 2022
- Published online by Cambridge University Press:
- 28 June 2022, e41
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
-
Background
Mental illness is known to come along with a large mortality gap compared to thegeneral population and it is a risk for COVID-19 related morbidity andmortality. Achieving high vaccination rates in people with mental illness is therefore important. Reports are conflicting on whether vaccination rates comparable to those of the general population can be achieved and which variables represent risk factors for nonvaccination in people with mental illness.
MethodsThe COVID Ψ Vac study collected routine data on vaccination status, diagnostic groups, sociodemographics, and setting characteristics from in- and day-clinic patients of 10 psychiatric hospitals in Germany in August 2021. Logistic regression modeling was used to determine risk factors for nonvaccination.
ResultsComplete vaccination rates were 59% (n = 776) for the hospitalized patients with mental illness versus 64% for the regionally and age-matched general population. Partial vaccination rates were 68% (n = 893) for the hospitalised patients with mental illness versus 67% for the respective general population and six percentage (n = 74) of this hospitalized population were vaccinated during the hospital stay. Rates showed a large variation between hospital sites. An ICD-10 group F1, F2, or F4 main diagnosis, younger age, and coercive accommodation were further risk factors for nonvaccination in the model.
ConclusionsVaccination rates were lower in hospitalized people with mental illness than in the general population. By targeting at-risk groups with low-threshold vaccination programs in all health institutions they get in contact with, vaccination rates comparable to those in the general population can be achieved.
Using polygenic scores and clinical data for bipolar disorder patient stratification and lithium response prediction: machine learning approach – CORRIGENDUM
- Micah Cearns, Azmeraw T. Amare, Klaus Oliver Schubert, Anbupalam Thalamuthu, Joseph Frank, Fabian Streit, Mazda Adli, Nirmala Akula, Kazufumi Akiyama, Raffaella Ardau, Bárbara Arias, JeanMichel Aubry, Lena Backlund, Abesh Kumar Bhattacharjee, Frank Bellivier, Antonio Benabarre, Susanne Bengesser, Joanna M. Biernacka, Armin Birner, Clara Brichant-Petitjean, Pablo Cervantes, HsiChung Chen, Caterina Chillotti, Sven Cichon, Cristiana Cruceanu, Piotr M. Czerski, Nina Dalkner, Alexandre Dayer, Franziska Degenhardt, Maria Del Zompo, J. Raymond DePaulo, Bruno Étain, Peter Falkai, Andreas J. Forstner, Louise Frisen, Mark A. Frye, Janice M. Fullerton, Sébastien Gard, Julie S. Garnham, Fernando S. Goes, Maria Grigoroiu-Serbanescu, Paul Grof, Ryota Hashimoto, Joanna Hauser, Urs Heilbronner, Stefan Herms, Per Hoffmann, Andrea Hofmann, Liping Hou, Yi-Hsiang Hsu, Stephane Jamain, Esther Jiménez, Jean-Pierre Kahn, Layla Kassem, Po-Hsiu Kuo, Tadafumi Kato, John Kelsoe, Sarah Kittel-Schneider, Sebastian Kliwicki, Barbara König, Ichiro Kusumi, Gonzalo Laje, Mikael Landén, Catharina Lavebratt, Marion Leboyer, Susan G. Leckband, Mario Maj, the Major Depressive Disorder Working Group of the Psychiatric Genomics Consortium, Mirko Manchia, Lina Martinsson, Michael J. McCarthy, Susan McElroy, Francesc Colom, Marina Mitjans, Francis M. Mondimore, Palmiero Monteleone, Caroline M. Nievergelt, Markus M. Nöthen, Tomas Novák, Claire O'Donovan, Norio Ozaki, Vincent Millischer, Sergi Papiol, Andrea Pfennig, Claudia Pisanu, James B. Potash, Andreas Reif, Eva Reininghaus, Guy A. Rouleau, Janusz K. Rybakowski, Martin Schalling, Peter R. Schofield, Barbara W. Schweizer, Giovanni Severino, Tatyana Shekhtman, Paul D. Shilling, Katzutaka Shimoda, Christian Simhandl, Claire M. Slaney, Alessio Squassina, Thomas Stamm, Pavla Stopkova, Fasil TekolaAyele, Alfonso Tortorella, Gustavo Turecki, Julia Veeh, Eduard Vieta, Stephanie H. Witt, Gloria Roberts, Peter P. Zandi, Martin Alda, Michael Bauer, Francis J. McMahon, Philip B. Mitchell, Thomas G. Schulze, Marcella Rietschel, Scott R. Clark, Bernhard T. Baune
-
- Journal:
- The British Journal of Psychiatry / Volume 221 / Issue 2 / August 2022
- Published online by Cambridge University Press:
- 04 May 2022, p. 494
- Print publication:
- August 2022
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
Using polygenic scores and clinical data for bipolar disorder patient stratification and lithium response prediction: machine learning approach
- Micah Cearns, Azmeraw T. Amare, Klaus Oliver Schubert, Anbupalam Thalamuthu, Joseph Frank, Fabian Streit, Mazda Adli, Nirmala Akula, Kazufumi Akiyama, Raffaella Ardau, Bárbara Arias, Jean-Michel Aubry, Lena Backlund, Abesh Kumar Bhattacharjee, Frank Bellivier, Antonio Benabarre, Susanne Bengesser, Joanna M. Biernacka, Armin Birner, Clara Brichant-Petitjean, Pablo Cervantes, Hsi-Chung Chen, Caterina Chillotti, Sven Cichon, Cristiana Cruceanu, Piotr M. Czerski, Nina Dalkner, Alexandre Dayer, Franziska Degenhardt, Maria Del Zompo, J. Raymond DePaulo, Bruno Étain, Peter Falkai, Andreas J. Forstner, Louise Frisen, Mark A. Frye, Janice M. Fullerton, Sébastien Gard, Julie S. Garnham, Fernando S. Goes, Maria Grigoroiu-Serbanescu, Paul Grof, Ryota Hashimoto, Joanna Hauser, Urs Heilbronner, Stefan Herms, Per Hoffmann, Andrea Hofmann, Liping Hou, Yi-Hsiang Hsu, Stephane Jamain, Esther Jiménez, Jean-Pierre Kahn, Layla Kassem, Po-Hsiu Kuo, Tadafumi Kato, John Kelsoe, Sarah Kittel-Schneider, Sebastian Kliwicki, Barbara König, Ichiro Kusumi, Gonzalo Laje, Mikael Landén, Catharina Lavebratt, Marion Leboyer, Susan G. Leckband, Mario Maj, the Major Depressive Disorder Working Group of the Psychiatric Genomics Consortium, Mirko Manchia, Lina Martinsson, Michael J. McCarthy, Susan McElroy, Francesc Colom, Marina Mitjans, Francis M. Mondimore, Palmiero Monteleone, Caroline M. Nievergelt, Markus M. Nöthen, Tomas Novák, Claire O'Donovan, Norio Ozaki, Vincent Millischer, Sergi Papiol, Andrea Pfennig, Claudia Pisanu, James B. Potash, Andreas Reif, Eva Reininghaus, Guy A. Rouleau, Janusz K. Rybakowski, Martin Schalling, Peter R. Schofield, Barbara W. Schweizer, Giovanni Severino, Tatyana Shekhtman, Paul D. Shilling, Katzutaka Shimoda, Christian Simhandl, Claire M. Slaney, Alessio Squassina, Thomas Stamm, Pavla Stopkova, Fasil Tekola-Ayele, Alfonso Tortorella, Gustavo Turecki, Julia Veeh, Eduard Vieta, Stephanie H. Witt, Gloria Roberts, Peter P. Zandi, Martin Alda, Michael Bauer, Francis J. McMahon, Philip B. Mitchell, Thomas G. Schulze, Marcella Rietschel, Scott R. Clark, Bernhard T. Baune
-
- Journal:
- The British Journal of Psychiatry / Volume 220 / Issue 4 / April 2022
- Published online by Cambridge University Press:
- 28 February 2022, pp. 219-228
- Print publication:
- April 2022
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
-
Background
Response to lithium in patients with bipolar disorder is associated with clinical and transdiagnostic genetic factors. The predictive combination of these variables might help clinicians better predict which patients will respond to lithium treatment.
AimsTo use a combination of transdiagnostic genetic and clinical factors to predict lithium response in patients with bipolar disorder.
MethodThis study utilised genetic and clinical data (n = 1034) collected as part of the International Consortium on Lithium Genetics (ConLi+Gen) project. Polygenic risk scores (PRS) were computed for schizophrenia and major depressive disorder, and then combined with clinical variables using a cross-validated machine-learning regression approach. Unimodal, multimodal and genetically stratified models were trained and validated using ridge, elastic net and random forest regression on 692 patients with bipolar disorder from ten study sites using leave-site-out cross-validation. All models were then tested on an independent test set of 342 patients. The best performing models were then tested in a classification framework.
ResultsThe best performing linear model explained 5.1% (P = 0.0001) of variance in lithium response and was composed of clinical variables, PRS variables and interaction terms between them. The best performing non-linear model used only clinical variables and explained 8.1% (P = 0.0001) of variance in lithium response. A priori genomic stratification improved non-linear model performance to 13.7% (P = 0.0001) and improved the binary classification of lithium response. This model stratified patients based on their meta-polygenic loadings for major depressive disorder and schizophrenia and was then trained using clinical data.
ConclusionsUsing PRS to first stratify patients genetically and then train machine-learning models with clinical predictors led to large improvements in lithium response prediction. When used with other PRS and biological markers in the future this approach may help inform which patients are most likely to respond to lithium treatment.
Characterisation of age and polarity at onset in bipolar disorder
- Janos L. Kalman, Loes M. Olde Loohuis, Annabel Vreeker, Andrew McQuillin, Eli A. Stahl, Douglas Ruderfer, Maria Grigoroiu-Serbanescu, Georgia Panagiotaropoulou, Stephan Ripke, Tim B. Bigdeli, Frederike Stein, Tina Meller, Susanne Meinert, Helena Pelin, Fabian Streit, Sergi Papiol, Mark J. Adams, Rolf Adolfsson, Kristina Adorjan, Ingrid Agartz, Sofie R. Aminoff, Heike Anderson-Schmidt, Ole A. Andreassen, Raffaella Ardau, Jean-Michel Aubry, Ceylan Balaban, Nicholas Bass, Bernhard T. Baune, Frank Bellivier, Antoni Benabarre, Susanne Bengesser, Wade H Berrettini, Marco P. Boks, Evelyn J. Bromet, Katharina Brosch, Monika Budde, William Byerley, Pablo Cervantes, Catina Chillotti, Sven Cichon, Scott R. Clark, Ashley L. Comes, Aiden Corvin, William Coryell, Nick Craddock, David W. Craig, Paul E. Croarkin, Cristiana Cruceanu, Piotr M. Czerski, Nina Dalkner, Udo Dannlowski, Franziska Degenhardt, Maria Del Zompo, J. Raymond DePaulo, Srdjan Djurovic, Howard J. Edenberg, Mariam Al Eissa, Torbjørn Elvsåshagen, Bruno Etain, Ayman H. Fanous, Frederike Fellendorf, Alessia Fiorentino, Andreas J. Forstner, Mark A. Frye, Janice M. Fullerton, Katrin Gade, Julie Garnham, Elliot Gershon, Michael Gill, Fernando S. Goes, Katherine Gordon-Smith, Paul Grof, Jose Guzman-Parra, Tim Hahn, Roland Hasler, Maria Heilbronner, Urs Heilbronner, Stephane Jamain, Esther Jimenez, Ian Jones, Lisa Jones, Lina Jonsson, Rene S. Kahn, John R. Kelsoe, James L. Kennedy, Tilo Kircher, George Kirov, Sarah Kittel-Schneider, Farah Klöhn-Saghatolislam, James A. Knowles, Thorsten M. Kranz, Trine Vik Lagerberg, Mikael Landen, William B. Lawson, Marion Leboyer, Qingqin S. Li, Mario Maj, Dolores Malaspina, Mirko Manchia, Fermin Mayoral, Susan L. McElroy, Melvin G. McInnis, Andrew M. McIntosh, Helena Medeiros, Ingrid Melle, Vihra Milanova, Philip B. Mitchell, Palmiero Monteleone, Alessio Maria Monteleone, Markus M. Nöthen, Tomas Novak, John I. Nurnberger, Niamh O'Brien, Kevin S. O'Connell, Claire O'Donovan, Michael C. O'Donovan, Nils Opel, Abigail Ortiz, Michael J. Owen, Erik Pålsson, Carlos Pato, Michele T. Pato, Joanna Pawlak, Julia-Katharina Pfarr, Claudia Pisanu, James B. Potash, Mark H Rapaport, Daniela Reich-Erkelenz, Andreas Reif, Eva Reininghaus, Jonathan Repple, Hélène Richard-Lepouriel, Marcella Rietschel, Kai Ringwald, Gloria Roberts, Guy Rouleau, Sabrina Schaupp, William A Scheftner, Simon Schmitt, Peter R. Schofield, K. Oliver Schubert, Eva C. Schulte, Barbara Schweizer, Fanny Senner, Giovanni Severino, Sally Sharp, Claire Slaney, Olav B. Smeland, Janet L. Sobell, Alessio Squassina, Pavla Stopkova, John Strauss, Alfonso Tortorella, Gustavo Turecki, Joanna Twarowska-Hauser, Marin Veldic, Eduard Vieta, John B. Vincent, Wei Xu, Clement C. Zai, Peter P. Zandi, Psychiatric Genomics Consortium (PGC) Bipolar Disorder Working Group, International Consortium on Lithium Genetics (ConLiGen), Colombia-US Cross Disorder Collaboration in Psychiatric Genetics, Arianna Di Florio, Jordan W. Smoller, Joanna M. Biernacka, Francis J. McMahon, Martin Alda, Bertram Müller-Myhsok, Nikolaos Koutsouleris, Peter Falkai, Nelson B. Freimer, Till F.M. Andlauer, Thomas G. Schulze, Roel A. Ophoff
-
- Journal:
- The British Journal of Psychiatry / Volume 219 / Issue 6 / December 2021
- Published online by Cambridge University Press:
- 25 August 2021, pp. 659-669
- Print publication:
- December 2021
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
-
Background
Studying phenotypic and genetic characteristics of age at onset (AAO) and polarity at onset (PAO) in bipolar disorder can provide new insights into disease pathology and facilitate the development of screening tools.
AimsTo examine the genetic architecture of AAO and PAO and their association with bipolar disorder disease characteristics.
MethodGenome-wide association studies (GWASs) and polygenic score (PGS) analyses of AAO (n = 12 977) and PAO (n = 6773) were conducted in patients with bipolar disorder from 34 cohorts and a replication sample (n = 2237). The association of onset with disease characteristics was investigated in two of these cohorts.
ResultsEarlier AAO was associated with a higher probability of psychotic symptoms, suicidality, lower educational attainment, not living together and fewer episodes. Depressive onset correlated with suicidality and manic onset correlated with delusions and manic episodes. Systematic differences in AAO between cohorts and continents of origin were observed. This was also reflected in single-nucleotide variant-based heritability estimates, with higher heritabilities for stricter onset definitions. Increased PGS for autism spectrum disorder (β = −0.34 years, s.e. = 0.08), major depression (β = −0.34 years, s.e. = 0.08), schizophrenia (β = −0.39 years, s.e. = 0.08), and educational attainment (β = −0.31 years, s.e. = 0.08) were associated with an earlier AAO. The AAO GWAS identified one significant locus, but this finding did not replicate. Neither GWAS nor PGS analyses yielded significant associations with PAO.
ConclusionsAAO and PAO are associated with indicators of bipolar disorder severity. Individuals with an earlier onset show an increased polygenic liability for a broad spectrum of psychiatric traits. Systematic differences in AAO across cohorts, continents and phenotype definitions introduce significant heterogeneity, affecting analyses.



