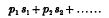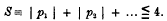43 results
Reducing preventable patient transfers from long-term care facilities to emergency departments: a scoping review – CORRIGENDUM
- Kiran L. Grant, Daniel Dongjoo Lee, Ivy Cheng, G. Ross Baker
-
- Journal:
- Canadian Journal of Emergency Medicine / Volume 22 / Issue 6 / November 2020
- Published online by Cambridge University Press:
- 23 November 2020, E16
- Print publication:
- November 2020
-
- Article
-
- You have access Access
- HTML
- Export citation
Learning from health system reform trajectories in seven Canadian provinces
- Susan Usher, Jean-Louis Denis, Johanne Préval, Ross Baker, Samia Chreim, Sara Kreindler, Mylaine Breton, Élizabeth Côté-Boileau
-
- Journal:
- Health Economics, Policy and Law / Volume 16 / Issue 4 / October 2021
- Published online by Cambridge University Press:
- 06 August 2020, pp. 383-399
-
- Article
- Export citation
-
In publicly funded health systems, reform efforts have proliferated to adapt to increasingly complex demands. In Canada, prior research (Lazar et al., 2013, Paradigm Freeze: Why is it so Hard to Reform Health Care in Canada?, McGill-Queen's Press) found that reforms at the end of the 20th century failed to change the fundamentals of the Canadian system based on physician independence and assured universal coverage only for medical and hospital services. This paper focuses on reforms since the turn of the millennium to explore the transformative capacities developed in seven provinces within this system architecture. Longitudinal case studies, based on scientific and grey literature, and interviews with key informants, trace the patterns of reform in each province and reveal five objectives that, to varying degrees, preoccupied reformers: (1) address chronic disease, (2) align health system actors with provincial objectives, (3) shift from hospital to community-based care, (4) integrate physicians, and (5) develop improvement capacities. The range of strategies adopted to achieve these objectives in different provinces is compared to identify emerging pathways of reform and extract lessons for future reformers. We find significant cross-learning between provinces, but also note an emergent dimension to reforms, where multiple strategies aggregate through time to create unique patterns, presenting their own set of possibilities and limitations for the future.
Reducing preventable patient transfers from long-term care facilities to emergency departments: a scoping review
- Kiran L. Grant, Daniel Dongjoo Lee, Ivy Cheng, G. Ross Baker
-
- Journal:
- Canadian Journal of Emergency Medicine / Volume 22 / Issue 6 / November 2020
- Published online by Cambridge University Press:
- 03 August 2020, pp. 844-856
- Print publication:
- November 2020
-
- Article
-
- You have access Access
- HTML
- Export citation
-
Background
In Canada, there were over 60,000 long-term care facility patient transfers to emergency departments (EDs) in 2014, with up to a quarter of them being potentially preventable. Each preventable transfer exposes the patient to transport- and hospital-related complications, contributes to ED crowding, and adds significant costs to the health care system. There have been many proposed and studied interventions aimed at alleviating the issue, but few attempts to assess and evaluate different interventions across institutions.
MethodsA systematic search of MEDLINE, CINAHL, and EMBASE for studies describing the impact of interventions aimed at reducing preventable transfers from long-term care facilities to EDs on ED transfer rate. Two independent reviewers screened the studies for inclusion and completed a quality assessment. A tabular and narrative synthesis was then completed. This study adhered to the Preferred Reporting Items for Systematic Reviews and Meta-Analysis Extension for Scoping Reviews (PRISMA-ScR) guidelines.
ResultsA total of 26 studies were included (Cohen's k = 0.68). One was of low quality (Cohen's k = 0.58). Studies were summarized into five themes based on intervention type: Telemedicine, Outreach Teams, Interdisciplinary Care, Integrated Approaches, and Other. Effective interventions reported reductions in ED transfer rates post intervention ranging from 10 to 70%. Interdisciplinary health care teams staffed within long-term care facilities were the most effective interventions.
ConclusionThere are several promising interventions that have successfully reduced the number of preventable transfers from long-term care facilities to EDs in a variety of health care settings. Widespread implementation of these interventions has the potential to reduce ED crowding in Canada.
155 Symptom Stability in de Novo and Rollover Patients with Bipolar I Disorder Receiving Aripiprazole Once-monthly in a 52-Week, Open-label Study
- Jessica J Madera, Pedro Such, Maxine Chen, Ross A Baker
-
- Journal:
- CNS Spectrums / Volume 25 / Issue 2 / April 2020
- Published online by Cambridge University Press:
- 24 April 2020, p. 299
-
- Article
-
- You have access Access
- Export citation
-
Introduction:
Long-term maintenance treatment is essential in management of bipolar I disorder (BP-I) to achieve mood stability, prevent recurrence of mood episodes and improve functioning. Aripiprazole once-monthly 400 mg (AOM 400) is a long-acting formulation of aripiprazole for maintenance treatment of BP-I. In a double-blind, placebo-controlled, randomized withdrawal study in adult patients with BP-I after a manic episode (NCT01567527), AOM 400 delayed time to and reduced rate of recurrence of mood episodes and was safe and well tolerated (1). In an open-label, long-term safety study (NCT01710709), AOM 400 was safe and effective as long-term maintenance treatment (2).
Objective:To evaluate the effect of long-term AOM 400 maintenance treatment on manic and depressive symptoms in BP-I patients from the open-label, long term safety study.
Methods:Manic and depressive symptoms were assessed post-hoc by analysis of change from study entry in the Young-Mania Rating Scale (YMRS) and Montgomery-Åsberg Depression Rating Scale (MADRS) total scores and line item scores. The mean changes from baseline at last visit in YMRS and MADRS total scores, and single items were calculated using descriptive statistics, using last observation carried forward for total scores and observed cases for single items.
Results:A total of 464 patients entered the maintenance phase: 379 were de novo and 85 were rollover patients who completed the double-blind, placebo-controlled withdrawal study. Overall, 63% (291/464) completed 52 weeks of open-label treatment. Mean YMRS and MADRS total scores were minimally changed from baseline (YMRS: 2.31, endpoint change -0.30; MADRS: 3.23, endpoint change +1.24) across the study in the total population.
Conclusion:Patients entering an open-label safety study with stable manic and depressive symptoms maintained stability in both types of symptoms, as shown by minimal mean changes from baseline in YMRS and MADRS scores, suggesting that treatment with AOM 400 is effective in preventing re-emergence of both manic and depressive symptoms.
Funding Acknowledgements:The study was supported by Otsuka Pharmaceutical Development & Commercialization, Inc.
154 Functioning in de Novo and Rollover Patients with Bipolar I Disorder Receiving Aripiprazole Once-monthly in a 52-Week, Open-label Study
- Jessica J Madera, Pedro Such, Maxine Chen, Ross A Baker
-
- Journal:
- CNS Spectrums / Volume 25 / Issue 2 / April 2020
- Published online by Cambridge University Press:
- 24 April 2020, pp. 298-299
-
- Article
-
- You have access Access
- Export citation
-
Introduction:
Long-term maintenance treatment is essential in management of bipolar I disorder (BP-I) to achieve mood stability, prevent recurrence of mood episodes and improve functioning. Aripiprazole once-monthly 400 mg (AOM 400) is a long-acting formulation of aripiprazole for maintenance treatment of BP-I. In a double-blind, placebo-controlled, randomized withdrawal study in adult patients with BP-I after a manic episode (NCT01567527), AOM 400 delayed time to and reduced rate of recurrence of mood episodes and was safe and well tolerated (1). In an open-label, long-term safety study (NCT01710709), AOM 400 was safe and effective as long-term maintenance treatment (2).
Objective:To evaluate the effect of long-term AOM 400 maintenance treatment on manic and depressive symptoms in BP-I patients from the open-label, long term safety study.
Methods:Manic and depressive symptoms were assessed post-hoc by analysis of change from study entry in the Young-Mania Rating Scale (YMRS) and Montgomery-Åsberg Depression Rating Scale (MADRS) total scores and line item scores. The mean changes from baseline at last visit in YMRS and MADRS total scores, and single items were calculated using descriptive statistics, using last observation carried forward for total scores and observed cases for single items.
Results:A total of 464 patients entered the maintenance phase: 379 were de novo and 85 were rollover patients who completed the double-blind, placebo-controlled withdrawal study. Overall, 63% (291/464) completed 52 weeks of open-label treatment. Mean YMRS and MADRS total scores were minimally changed from baseline (YMRS: 2.31, endpoint change -0.30; MADRS: 3.23, endpoint change +1.24) across the study in the total population.
Conclusion:Patients entering an open-label safety study with stable manic and depressive symptoms maintained stability in both types of symptoms, as shown by minimal mean changes from baseline in YMRS and MADRS scores, suggesting that treatment with AOM 400 is effective in preventing re-emergence of both manic and depressive symptoms.
Funding Acknowledgements:The study was supported by Otsuka Pharmaceutical Development & Commercialization, Inc.
47 Sustained Functional Recovery and Symptom Remission After Maintenance Treatment with Aripiprazole Once-Monthly for Patients with Bipolar I Disorder
- Eduard Vieta, Ross A. Baker, Jessica J. Madera, Peter Zhang, Pedro Such, Maxine Chen, Joseph Calabrese
-
- Journal:
- CNS Spectrums / Volume 24 / Issue 1 / February 2019
- Published online by Cambridge University Press:
- 12 March 2019, pp. 201-202
-
- Article
-
- You have access Access
- Export citation
-
Study Objectives
To report functional recovery, symptomatic remission, and sustained symptomatic remission rates after treatment with aripiprazole once-monthly 400mg (AOM 400) administered every 4weeks for up to 52weeks as maintenance treatment in a mixed cohort of AOM 400 naïve (de novo) and experienced adults (rollover) with bipolar I disorder (BP-I).
MethodThis open-label study (NCT01710709) enrolled de novo patients with a diagnosis of BP-I and ≥1 previous manic or mixed episode and rollover patients who completed a randomized, double-blind, placebo-controlled study assessing the efficacy and safety of AOM 400 (NCT01567527). Efficacy was assessed by mean changes from baseline in Young-Mania Rating Scale (YMRS), Montgomery-Asberg Depressive Rating Scale (MADRS), and Clinical Global Impression- Bipolar Version-Severity of Illness (CGI-BP-S) scores. Sustained functional recovery was defined as a total score of ≤11 on the Functioning Assessment Short Test (FAST) for ≥8 consecutive weeks. Remission was defined as YMRS and MADRS total scores ≤12, and sustained remission was defined as meeting criteria for remission for 8 consecutive weeks. The study included a screening phase (6weeks) for de novo patients, an oral aripiprazole conversion phase (4–6weeks), an oral stabilization phase (4–12weeks), and an AOM 400 maintenance phase (up to 52weeks). Rollover patients entered directly into the AOM 400 maintenance phase.
ResultsA total of 464 subjects entered the maintenance phase and 63% (291/464) completed the trial. Of patients entering the maintenance phase, 379 (82%) were de novo and 85 (18%) were rollover. The most frequent reasons for discontinuation were withdrawal of consent (11%) and adverse events (AEs) (10%). Weight increase (1.5%, 7/464) and BP-I (0.9%, 4/464) were the most common reasons for discontinuation due to AEs. Improvements in mean YMRS, MADRS, CGI-BP-S, and FAST scores achieved in previous phases were maintained over 52weeks. Treatment-emergent AEs experienced by >10% of the patients were akathisia (14.7%), weight increased (13.4%), nasopharyngitis (12.1%), and insomnia (11.0%). A high proportion of de novo patients met the criteria for symptomatic remission (87.2%, 328/376) and sustained remission (77%, 292/379) by last visit. Rollover patients’ remission rate remained stable (98.8%, 84/85) by last visit. Of the rollover patients, 35/85 (43%) and 35/116 (36%) of de novo subjects met the criteria for sustained functional recovery after study completion.
ConclusionsPatients treated with AOM 400 maintained symptomatic and functional stability for up to 52weeks. Importantly, more than one-third of patients achieved sustained functional recovery using a strict criterion. Overall, AOM 400 was safe and well tolerated in patients with BP-I. Results support AOM 400 as a viable once-monthlyoption for maintenance treatment of BP-I.
These data were previously presented at the 31st ECNP Congress, 2018, Barcelona,Spain.
Funding Acknowledgements: The study was supported by Otsuka Pharmaceutical Development & Commercialization, Inc.
14 Long-term Efficacy of Brexpiprazole in Patients with Schizophrenia with Clinically Relevant Levels of Negative Symptoms
- Catherine Weiss, Peter Zhang, Ross A Baker, Mary Hobart, Nanco Hefting, Stine R Meehan
-
- Journal:
- CNS Spectrums / Volume 24 / Issue 1 / February 2019
- Published online by Cambridge University Press:
- 12 March 2019, p. 180
-
- Article
-
- You have access Access
- Export citation
-
Background
Effective treatments for patients with high levels of negative symptoms of schizophrenia are lacking. Brexpiprazole is a serotonin–dopamine activity modulator that is a partial agonist at 5-HT1A and dopamine D2 receptors, and an antagonist at 5-HT2A and noradrenaline alpha1B/2C receptors, all with subnanomolar potency. Long-term treatment with brexpiprazole demonstrated broad efficacy across all five Marder factor groupings, including positive, negative, disorganized thoughts, uncontrolled hostility/excitement, and anxiety/depression. This post-hoc analysis of long-term effects of brexpiprazole in patients with clinically relevant levels of negative symptoms of schizophrenia is based on data from two similarly designed short-term, placebo-controlled studies (Vector; NCT01396421 or Beacon; NCT01393613) for the brexpiprazole-treated patients who continued into an open-label extension study (Zenith; NCT01397786).
MethodsIn the short-term studies, patients with acute schizophrenia were randomly assigned to fixed once-daily doses of brexpiprazole 0.25mg (Vector), 1mg (Beacon), 2mg , 4mg or placebo for 6weeks. The long-term study was an open-label, 52-week (amended to 26weeks), safety extension study with flexible-dose (1–4mg/day) brexpiprazole. The post-hoc analyses were performed on brexpiprazole-treated patients from the short-term studies who continued into the long-term study, and who had clinically relevant negative symptoms, defined as PANSS Factor Score for Negative Symptoms (PANSS-FSNS; N1, N2, N3, N4, G7, G16) of ≥24, and score of ≥4 on at least two of three core negative symptom PANSS items at randomization in the parent study. The outcome of the analysis included change from baseline to up to 58weeks in PANSS-FSNS, PANSS Total, and PSP. Safety was also assessed.
ResultsA total of 187 patients with clinically relevant levels of negative symptoms in the parent study rolled-over into the open-label extension study and were available for analysis. Eighty-three of these patients remained in the studies for 58weeks. Due to the study amendment, not all patients had the opportunity of complete 52weeks of open-label treatment. Baseline PANSS Total score was 104.4, while baseline PANSS-FSNS was 27.6 and baseline PSP Total score was 41.3. Mean change (SD) from baseline in PANSS-FSNS was –10.9 (5.0), and –44.2 (17.5) for PANSS Total score at Week 58. Change from baseline (SD) to Week 58 for PSP Total score was 24.8 (12.9) with improvement in all domains (socially useful activities, personal and social relationship, self-care, and disturbing and aggressive behaviors). The TEAEs reported ≥5% were schizophrenia (18.9%), insomnia (8.6%), weight increased (5.9%) and akathisia (5.9%).
ConclusionThis post-hoc analysis suggests that brexpiprazole has long-term effectiveness on negative symptoms and functioning in patients with schizophrenia and clinically relevant levels of negative symptoms.
Funding Acknowledgements: The study was funded by Otsuka Pharmaceutical Development & Commercialization Inc. and H. Lundbeck A/S
Agitation in patients with dementia: a systematic review of epidemiology and association with severity and course
- Milena Anatchkova, Anne Brooks, Laura Swett, Ann Hartry, Ruth A. Duffy, Ross A. Baker, Lene Hammer-Helmich, Myrlene Sanon Aigbogun
-
- Journal:
- International Psychogeriatrics / Volume 31 / Issue 9 / September 2019
- Published online by Cambridge University Press:
- 11 March 2019, pp. 1305-1318
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
-
Objectives:
More than 90% of individuals with Alzheimer’s disease (AD) experience behavioral and neuropsychiatric symptoms (NPS), such as agitation. However, little is known regarding the specific burden of agitation for Alzheimer’s patients.
Design:A global systematic literature review was conducted in MEDLINE and Embase for studies of clinical, humanistic, and economic burden of agitation in AD/dementia published from 2006–2016. References of identified papers and related literature reviews were examined. Studies meeting predetermined inclusion criteria for burden of agitation/NPS were summarized.
Results:Eighty papers met the inclusion criteria for burden of agitation in dementia. Wide ranges of agitation prevalence were reported, but few papers provided information on incidence. The association of agitation with AD severity was presented in multiple studies; a few suggested positive association of agitation with mortality.
Conclusions:High prevalence of agitation is consistent with earlier reports, but several gaps in understanding of agitation in AD need further exploration.
Successful switching of patients with acute schizophrenia from another antipsychotic to brexpiprazole: comparison of clinicians’ choice of cross-titration schedules in a post hoc analysis of a randomized, double-blind, maintenance treatment study
- Christoph U. Correll, Lily Shi, Catherine Weiss, Mary Hobart, Anna Eramo, Ruth A. Duffy, Emmanuelle Weiller, Ross A. Baker
-
- Journal:
- CNS Spectrums / Volume 24 / Issue 5 / October 2019
- Published online by Cambridge University Press:
- 11 October 2018, pp. 507-517
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
-
Objective
To compare the tolerability and efficacy of different antipsychotic cross-titration schedules, using data from a brexpiprazole study (Equator; NCT01668797).
MethodsPatients with schizophrenia were cross-titrated from other antipsychotics to brexpiprazole monotherapy in a 1–4 week open-label conversion phase, then entered a single-blind brexpiprazole treatment phase. Patients were stratified into four “conversion groups,” according to the amount of time spent in the conversion phase. Discontinuation rates, treatment-emergent adverse events (TEAEs), and efficacy (Positive and Negative Syndrome Scale [PANSS]) were compared between conversion groups.
ResultsOf the 404 patients treated with brexpiprazole, the majority (72.0%) spent 22–33 days in the conversion phase. Discontinuation rates due to lack of efficacy or adverse events were low in all conversion groups. Of the 292 patients who successfully switched and completed 8 weeks of brexpiprazole treatment, most were converted to brexpiprazole over 22–33 days (80.1%), and fewer were converted over 1–7 days (2.4%), 8–14 days (6.5%), or 15–21 days (11.0%). The incidence of TEAEs over 8 weeks was lower among those converted over 22–33 days (44.4%) than in other conversion groups (62.5–84.2%), although low patient numbers with shorter conversion times limit the generalizability of this finding. Each conversion group showed comparable improvement in PANSS total score from baseline.
ConclusionThe majority of patients were cross-titrated to brexpiprazole over a period of 22–33 days, by investigators’ choice. Additional data on shorter conversions may help clinicians to choose a switching paradigm that best meets their patients’ needs.
Aripiprazole once-monthly for treatment of schizophrenia: double-blind, randomised, non-inferiority study
- W. Wolfgang Fleischhacker, Raymond Sanchez, Pamela P. Perry, Na Jin, Timothy Peters-Strickland, Brian R. Johnson, Ross A. Baker, Anna Eramo, Robert D. McQuade, William H. Carson, David Walling, John M. Kane
-
- Journal:
- The British Journal of Psychiatry / Volume 205 / Issue 2 / August 2014
- Published online by Cambridge University Press:
- 02 January 2018, pp. 135-144
- Print publication:
- August 2014
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
-
Background
Long-acting injectable formulations of antipsychotics are treatment alternatives to oral agents.
AimsTo assess the efficacy of aripiprazole once-monthly compared with oral aripiprazole for maintenance treatment of schizophrenia.
MethodA 38-week, double-blind, active-controlled, non-inferiority study; randomisation (2:2:1) to aripiprazole once-monthly 400 mg, oral aripiprazole (10–30 mg/day) or aripiprazole once-monthly 50mg (a dose below the therapeutic threshold for assay sensitivity). (Trial registration: clinicaltrials.gov, NCT00706654.)
ResultsA total of 1118 patients were screened, and 662 responders to oral aripiprazole were randomised. Kaplan–Meier estimated impending relapse rates at week 26 were 7.12% for aripiprazole once-monthly 400mg and 7.76% for oral aripiprazole. This difference (−0.64%, 95% CI −5.26 to 3.99) excluded the predefined non-inferiority margin of 11.5%. Treatments were superior to aripiprazole once-monthly 50mg (21.80%, P⩽0.001).
ConclusionsAripiprazole once-monthly 400mg was non-inferior to oral aripiprazole, and the reduction in Kaplan–Meier estimated impending relapse rate at week 26 was statistically significant v. aripiprazole once-monthly 50 mg.
Efficacy of adjunctive aripiprazole in patients with major depressive disorder whose symptoms worsened with antidepressant monotherapy
- J. Craig Nelson, Zia Rahman, Kimberly K. Laubmeier, James M. Eudicone, Robert D. McQuade, Robert M. Berman, Ronald N. Marcus, Ross A. Baker, John J. Sheehan
-
- Journal:
- CNS Spectrums / Volume 19 / Issue 6 / December 2014
- Published online by Cambridge University Press:
- 18 March 2014, pp. 528-534
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
-
Introduction
Efficacy of depression treatments, including adjunctive antipsychotic treatment, has not been explored for patients with worsening symptoms after antidepressant therapy (ADT).
MethodsThis post-hoc analysis utilized pooled data from 3 similarly designed, randomized, double-blind, placebo-controlled trials that assessed the efficacy, safety, and tolerability of adjunctive aripiprazole in patients with major depressive disorder with inadequate response to ADT. The studies had 2 phases: an 8-week prospective ADT phase and 6-week adjunctive (aripiprazole or placebo) treatment phase. This analysis focused on patients whose symptoms worsened during the prospective 8-week ADT phase (worsening defined as >0% increase in Montgomery–Åsberg Depressive Rating Scale [MADRS] Total score). During the 6-week, double-blind, adjunctive phase, response was defined as ≥50% reduction in MADRS Total score and remission as ≥50% reduction in MADRS Total score and MADRS score ≤10.
ResultsOf 1065 patients who failed to achieve a response during the prospective phase, 160 exhibited worsening of symptoms (ADT-Worseners), and 905 exhibited no change/reduction in MADRS scores (ADT-Non-worseners). Response rates for ADT-Worseners at endpoint were 36.6% (adjunctive aripiprazole) and 22.5% (placebo). Similarly, response rates at endpoint for ADT-Non-worseners were 37.5% (adjunctive aripiprazole) and 22.5% (placebo). Remission rates at endpoint for ADT-Worseners were 25.4% (adjunctive aripiprazole) and 12.4% (placebo). For ADT-Non-worseners, remission rates were 29.9% (adjunctive aripiprazole) and 17.4% (placebo).
ConclusionThese results suggest that adjunctive aripiprazole is an effective intervention for patients whose symptoms worsen during antidepressant monotherapy. The results challenge the view that benefits of adjunctive therapy with aripiprazole are limited to partial responders to ADT.
Evaluation of Organizational Culture among Different Levels of Healthcare Staff Participating in the Institute for Healthcare Improvement's 100,000 Lives Campaign
- Ronda L. Sinkowitz-Cochran, Amanda Garcia-Williams, Andrew D. Hackbarth, Bonnie Zell, G. Ross Baker, C. Joseph McCannon, Elise M. Beltrami, John A. Jernigan, L. Clifford McDonald, Donald A. Goldmann
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 33 / Issue 2 / February 2012
- Published online by Cambridge University Press:
- 02 January 2015, pp. 135-143
- Print publication:
- February 2012
-
- Article
- Export citation
-
Background.
Little is known about how hospital organizational and cultural factors associated with implementation of quality initiatives such as the Institute for Healthcare Improvement's (IHI) 100,000 Lives Campaign differ among levels of healthcare staff.
Design.Evaluation of a mixed qualitative and quantitative methodology (“trilogic evaluation model”).
Setting.Six hospitals that joined the campaign before June 2006.
Participants.Three strata of staff (executive leadership, midlevel, and frontline) at each hospital.
Results.Surveys were completed in 2008 by 135 hospital personnel (midlevel, 43.7%; frontline, 38.5%; executive, 17.8%) who also participated in 20 focus groups. Overall, 93% of participants were aware of the IHI campaign in their hospital and perceived that 58% (standard deviation, 22.7%) of improvements in quality at their hospital were a direct result of the campaign. There were significant differences between staff levels on the organizational culture (OC) items, with executive-level staff having higher scores than midlevel and frontline staff. All 20 focus groups perceived that the campaign interventions were sustainable and that data feedback, buy-in, hardwiring (into daily activities), and leadership support were essential to sustainability.
Conclusions.The trilogic model demonstrated that the 3 levels of staff had markedly different perceptions regarding the IHI campaign and OC. A framework in which frontline, midlevel, and leadership staff are simultaneously assessed may be a useful tool for future evaluations of OC and quality initiatives such as the IHI campaign.
Infect Control Hosp Epidemiol 2012;33(2):135-143
Contributors
-
- By Aakash Agarwala, Linda S. Aglio, Rae M. Allain, Paul D. Allen, Houman Amirfarzan, Yasodananda Kumar Areti, Amit Asopa, Edwin G. Avery, Patricia R. Bachiller, Angela M. Bader, Rana Badr, Sibinka Bajic, David J. Baker, Sheila R. Barnett, Rena Beckerly, Lorenzo Berra, Walter Bethune, Sascha S. Beutler, Tarun Bhalla, Edward A. Bittner, Jonathan D. Bloom, Alina V. Bodas, Lina M. Bolanos-Diaz, Ruma R. Bose, Jan Boublik, John P. Broadnax, Jason C. Brookman, Meredith R. Brooks, Roland Brusseau, Ethan O. Bryson, Linda A. Bulich, Kenji Butterfield, William R. Camann, Denise M. Chan, Theresa S. Chang, Jonathan E. Charnin, Mark Chrostowski, Fred Cobey, Adam B. Collins, Mercedes A. Concepcion, Christopher W. Connor, Bronwyn Cooper, Jeffrey B. Cooper, Martha Cordoba-Amorocho, Stephen B. Corn, Darin J. Correll, Gregory J. Crosby, Lisa J. Crossley, Deborah J. Culley, Tomas Cvrk, Michael N. D'Ambra, Michael Decker, Daniel F. Dedrick, Mark Dershwitz, Francis X. Dillon, Pradeep Dinakar, Alimorad G. Djalali, D. John Doyle, Lambertus Drop, Ian F. Dunn, Theodore E. Dushane, Sunil Eappen, Thomas Edrich, Jesse M. Ehrenfeld, Jason M. Erlich, Lucinda L. Everett, Elliott S. Farber, Khaldoun Faris, Eddy M. Feliz, Massimo Ferrigno, Richard S. Field, Michael G. Fitzsimons, Hugh L. Flanagan Jr., Vladimir Formanek, Amanda A. Fox, John A. Fox, Gyorgy Frendl, Tanja S. Frey, Samuel M. Galvagno Jr., Edward R. Garcia, Jonathan D. Gates, Cosmin Gauran, Brian J. Gelfand, Simon Gelman, Alexander C. Gerhart, Peter Gerner, Omid Ghalambor, Christopher J. Gilligan, Christian D. Gonzalez, Noah E. Gordon, William B. Gormley, Thomas J. Graetz, Wendy L. Gross, Amit Gupta, James P. Hardy, Seetharaman Hariharan, Miriam Harnett, Philip M. Hartigan, Joaquim M. Havens, Bishr Haydar, Stephen O. Heard, James L. Helstrom, David L. Hepner, McCallum R. Hoyt, Robert N. Jamison, Karinne Jervis, Stephanie B. Jones, Swaminathan Karthik, Richard M. Kaufman, Shubjeet Kaur, Lee A. Kearse Jr., John C. Keel, Scott D. Kelley, Albert H. Kim, Amy L. Kim, Grace Y. Kim, Robert J. Klickovich, Robert M. Knapp, Bhavani S. Kodali, Rahul Koka, Alina Lazar, Laura H. Leduc, Stanley Leeson, Lisa R. Leffert, Scott A. LeGrand, Patricio Leyton, J. Lance Lichtor, John Lin, Alvaro A. Macias, Karan Madan, Sohail K. Mahboobi, Devi Mahendran, Christine Mai, Sayeed Malek, S. Rao Mallampati, Thomas J. Mancuso, Ramon Martin, Matthew C. Martinez, J. A. Jeevendra Martyn, Kai Matthes, Tommaso Mauri, Mary Ellen McCann, Shannon S. McKenna, Dennis J. McNicholl, Abdel-Kader Mehio, Thor C. Milland, Tonya L. K. Miller, John D. Mitchell, K. Annette Mizuguchi, Naila Moghul, David R. Moss, Ross J. Musumeci, Naveen Nathan, Ju-Mei Ng, Liem C. Nguyen, Ervant Nishanian, Martina Nowak, Ala Nozari, Michael Nurok, Arti Ori, Rafael A. Ortega, Amy J. Ortman, David Oxman, Arvind Palanisamy, Carlo Pancaro, Lisbeth Lopez Pappas, Benjamin Parish, Samuel Park, Deborah S. Pederson, Beverly K. Philip, James H. Philip, Silvia Pivi, Stephen D. Pratt, Douglas E. Raines, Stephen L. Ratcliff, James P. Rathmell, J. Taylor Reed, Elizabeth M. Rickerson, Selwyn O. Rogers Jr., Thomas M. Romanelli, William H. Rosenblatt, Carl E. Rosow, Edgar L. Ross, J. Victor Ryckman, Mônica M. Sá Rêgo, Nicholas Sadovnikoff, Warren S. Sandberg, Annette Y. Schure, B. Scott Segal, Navil F. Sethna, Swapneel K. Shah, Shaheen F. Shaikh, Fred E. Shapiro, Torin D. Shear, Prem S. Shekar, Stanton K. Shernan, Naomi Shimizu, Douglas C. Shook, Kamal K. Sikka, Pankaj K. Sikka, David A. Silver, Jeffrey H. Silverstein, Emily A. Singer, Ken Solt, Spiro G. Spanakis, Wolfgang Steudel, Matthias Stopfkuchen-Evans, Michael P. Storey, Gary R. Strichartz, Balachundhar Subramaniam, Wariya Sukhupragarn, John Summers, Shine Sun, Eswar Sundar, Sugantha Sundar, Neelakantan Sunder, Faraz Syed, Usha B. Tedrow, Nelson L. Thaemert, George P. Topulos, Lawrence C. Tsen, Richard D. Urman, Charles A. Vacanti, Francis X. Vacanti, Joshua C. Vacanti, Assia Valovska, Ivan T. Valovski, Mary Ann Vann, Susan Vassallo, Anasuya Vasudevan, Kamen V. Vlassakov, Gian Paolo Volpato, Essi M. Vulli, J. Matthias Walz, Jingping Wang, James F. Watkins, Maxwell Weinmann, Sharon L. Wetherall, Mallory Williams, Sarah H. Wiser, Zhiling Xiong, Warren M. Zapol, Jie Zhou
- Edited by Charles Vacanti, Scott Segal, Pankaj Sikka, Richard Urman
-
- Book:
- Essential Clinical Anesthesia
- Published online:
- 05 January 2012
- Print publication:
- 11 July 2011, pp xv-xxviii
-
- Chapter
- Export citation
Contributors
-
- By Rose Teteki Abbey, K. C. Abraham, David Tuesday Adamo, LeRoy H. Aden, Efrain Agosto, Victor Aguilan, Gillian T. W. Ahlgren, Charanjit Kaur AjitSingh, Dorothy B E A Akoto, Giuseppe Alberigo, Daniel E. Albrecht, Ruth Albrecht, Daniel O. Aleshire, Urs Altermatt, Anand Amaladass, Michael Amaladoss, James N. Amanze, Lesley G. Anderson, Thomas C. Anderson, Victor Anderson, Hope S. Antone, María Pilar Aquino, Paula Arai, Victorio Araya Guillén, S. Wesley Ariarajah, Ellen T. Armour, Brett Gregory Armstrong, Atsuhiro Asano, Naim Stifan Ateek, Mahmoud Ayoub, John Alembillah Azumah, Mercedes L. García Bachmann, Irena Backus, J. Wayne Baker, Mieke Bal, Lewis V. Baldwin, William Barbieri, António Barbosa da Silva, David Basinger, Bolaji Olukemi Bateye, Oswald Bayer, Daniel H. Bays, Rosalie Beck, Nancy Elizabeth Bedford, Guy-Thomas Bedouelle, Chorbishop Seely Beggiani, Wolfgang Behringer, Christopher M. Bellitto, Byard Bennett, Harold V. Bennett, Teresa Berger, Miguel A. Bernad, Henley Bernard, Alan E. Bernstein, Jon L. Berquist, Johannes Beutler, Ana María Bidegain, Matthew P. Binkewicz, Jennifer Bird, Joseph Blenkinsopp, Dmytro Bondarenko, Paulo Bonfatti, Riet en Pim Bons-Storm, Jessica A. Boon, Marcus J. Borg, Mark Bosco, Peter C. Bouteneff, François Bovon, William D. Bowman, Paul S. Boyer, David Brakke, Richard E. Brantley, Marcus Braybrooke, Ian Breward, Ênio José da Costa Brito, Jewel Spears Brooker, Johannes Brosseder, Nicholas Canfield Read Brown, Robert F. Brown, Pamela K. Brubaker, Walter Brueggemann, Bishop Colin O. Buchanan, Stanley M. Burgess, Amy Nelson Burnett, J. Patout Burns, David B. Burrell, David Buttrick, James P. Byrd, Lavinia Byrne, Gerado Caetano, Marcos Caldas, Alkiviadis Calivas, William J. Callahan, Salvatore Calomino, Euan K. Cameron, William S. Campbell, Marcelo Ayres Camurça, Daniel F. Caner, Paul E. Capetz, Carlos F. Cardoza-Orlandi, Patrick W. Carey, Barbara Carvill, Hal Cauthron, Subhadra Mitra Channa, Mark D. Chapman, James H. Charlesworth, Kenneth R. Chase, Chen Zemin, Luciano Chianeque, Philip Chia Phin Yin, Francisca H. Chimhanda, Daniel Chiquete, John T. Chirban, Soobin Choi, Robert Choquette, Mita Choudhury, Gerald Christianson, John Chryssavgis, Sejong Chun, Esther Chung-Kim, Charles M. A. Clark, Elizabeth A. Clark, Sathianathan Clarke, Fred Cloud, John B. Cobb, W. Owen Cole, John A Coleman, John J. Collins, Sylvia Collins-Mayo, Paul K. Conkin, Beth A. Conklin, Sean Connolly, Demetrios J. Constantelos, Michael A. Conway, Paula M. Cooey, Austin Cooper, Michael L. Cooper-White, Pamela Cooper-White, L. William Countryman, Sérgio Coutinho, Pamela Couture, Shannon Craigo-Snell, James L. Crenshaw, David Crowner, Humberto Horacio Cucchetti, Lawrence S. Cunningham, Elizabeth Mason Currier, Emmanuel Cutrone, Mary L. Daniel, David D. Daniels, Robert Darden, Rolf Darge, Isaiah Dau, Jeffry C. Davis, Jane Dawson, Valentin Dedji, John W. de Gruchy, Paul DeHart, Wendy J. Deichmann Edwards, Miguel A. De La Torre, George E. Demacopoulos, Thomas de Mayo, Leah DeVun, Beatriz de Vasconcellos Dias, Dennis C. Dickerson, John M. Dillon, Luis Miguel Donatello, Igor Dorfmann-Lazarev, Susanna Drake, Jonathan A. Draper, N. Dreher Martin, Otto Dreydoppel, Angelyn Dries, A. J. Droge, Francis X. D'Sa, Marilyn Dunn, Nicole Wilkinson Duran, Rifaat Ebied, Mark J. Edwards, William H. Edwards, Leonard H. Ehrlich, Nancy L. Eiesland, Martin Elbel, J. Harold Ellens, Stephen Ellingson, Marvin M. Ellison, Robert Ellsberg, Jean Bethke Elshtain, Eldon Jay Epp, Peter C. Erb, Tassilo Erhardt, Maria Erling, Noel Leo Erskine, Gillian R. Evans, Virginia Fabella, Michael A. Fahey, Edward Farley, Margaret A. Farley, Wendy Farley, Robert Fastiggi, Seena Fazel, Duncan S. Ferguson, Helwar Figueroa, Paul Corby Finney, Kyriaki Karidoyanes FitzGerald, Thomas E. FitzGerald, John R. Fitzmier, Marie Therese Flanagan, Sabina Flanagan, Claude Flipo, Ronald B. Flowers, Carole Fontaine, David Ford, Mary Ford, Stephanie A. Ford, Jim Forest, William Franke, Robert M. Franklin, Ruth Franzén, Edward H. Friedman, Samuel Frouisou, Lorelei F. Fuchs, Jojo M. Fung, Inger Furseth, Richard R. Gaillardetz, Brandon Gallaher, China Galland, Mark Galli, Ismael García, Tharscisse Gatwa, Jean-Marie Gaudeul, Luis María Gavilanes del Castillo, Pavel L. Gavrilyuk, Volney P. Gay, Metropolitan Athanasios Geevargis, Kondothra M. George, Mary Gerhart, Simon Gikandi, Maurice Gilbert, Michael J. Gillgannon, Verónica Giménez Beliveau, Terryl Givens, Beth Glazier-McDonald, Philip Gleason, Menghun Goh, Brian Golding, Bishop Hilario M. Gomez, Michelle A. Gonzalez, Donald K. Gorrell, Roy Gottfried, Tamara Grdzelidze, Joel B. Green, Niels Henrik Gregersen, Cristina Grenholm, Herbert Griffiths, Eric W. Gritsch, Erich S. Gruen, Christoffer H. Grundmann, Paul H. Gundani, Jon P. Gunnemann, Petre Guran, Vidar L. Haanes, Jeremiah M. Hackett, Getatchew Haile, Douglas John Hall, Nicholas Hammond, Daphne Hampson, Jehu J. Hanciles, Barry Hankins, Jennifer Haraguchi, Stanley S. Harakas, Anthony John Harding, Conrad L. Harkins, J. William Harmless, Marjory Harper, Amir Harrak, Joel F. Harrington, Mark W. Harris, Susan Ashbrook Harvey, Van A. Harvey, R. Chris Hassel, Jione Havea, Daniel Hawk, Diana L. Hayes, Leslie Hayes, Priscilla Hayner, S. Mark Heim, Simo Heininen, Richard P. Heitzenrater, Eila Helander, David Hempton, Scott H. Hendrix, Jan-Olav Henriksen, Gina Hens-Piazza, Carter Heyward, Nicholas J. Higham, David Hilliard, Norman A. Hjelm, Peter C. Hodgson, Arthur Holder, M. Jan Holton, Dwight N. Hopkins, Ronnie Po-chia Hsia, Po-Ho Huang, James Hudnut-Beumler, Jennifer S. Hughes, Leonard M. Hummel, Mary E. Hunt, Laennec Hurbon, Mark Hutchinson, Susan E. Hylen, Mary Beth Ingham, H. Larry Ingle, Dale T. Irvin, Jon Isaak, Paul John Isaak, Ada María Isasi-Díaz, Hans Raun Iversen, Margaret C. Jacob, Arthur James, Maria Jansdotter-Samuelsson, David Jasper, Werner G. Jeanrond, Renée Jeffery, David Lyle Jeffrey, Theodore W. Jennings, David H. Jensen, Robin Margaret Jensen, David Jobling, Dale A. Johnson, Elizabeth A. Johnson, Maxwell E. Johnson, Sarah Johnson, Mark D. Johnston, F. Stanley Jones, James William Jones, John R. Jones, Alissa Jones Nelson, Inge Jonsson, Jan Joosten, Elizabeth Judd, Mulambya Peggy Kabonde, Robert Kaggwa, Sylvester Kahakwa, Isaac Kalimi, Ogbu U. Kalu, Eunice Kamaara, Wayne C. Kannaday, Musimbi Kanyoro, Veli-Matti Kärkkäinen, Frank Kaufmann, Léon Nguapitshi Kayongo, Richard Kearney, Alice A. Keefe, Ralph Keen, Catherine Keller, Anthony J. Kelly, Karen Kennelly, Kathi Lynn Kern, Fergus Kerr, Edward Kessler, George Kilcourse, Heup Young Kim, Kim Sung-Hae, Kim Yong-Bock, Kim Yung Suk, Richard King, Thomas M. King, Robert M. Kingdon, Ross Kinsler, Hans G. Kippenberg, Cheryl A. Kirk-Duggan, Clifton Kirkpatrick, Leonid Kishkovsky, Nadieszda Kizenko, Jeffrey Klaiber, Hans-Josef Klauck, Sidney Knight, Samuel Kobia, Robert Kolb, Karla Ann Koll, Heikki Kotila, Donald Kraybill, Philip D. W. Krey, Yves Krumenacker, Jeffrey Kah-Jin Kuan, Simanga R. Kumalo, Peter Kuzmic, Simon Shui-Man Kwan, Kwok Pui-lan, André LaCocque, Stephen E. Lahey, John Tsz Pang Lai, Emiel Lamberts, Armando Lampe, Craig Lampe, Beverly J. Lanzetta, Eve LaPlante, Lizette Larson-Miller, Ariel Bybee Laughton, Leonard Lawlor, Bentley Layton, Robin A. Leaver, Karen Lebacqz, Archie Chi Chung Lee, Marilyn J. Legge, Hervé LeGrand, D. L. LeMahieu, Raymond Lemieux, Bill J. Leonard, Ellen M. Leonard, Outi Leppä, Jean Lesaulnier, Nantawan Boonprasat Lewis, Henrietta Leyser, Alexei Lidov, Bernard Lightman, Paul Chang-Ha Lim, Carter Lindberg, Mark R. Lindsay, James R. Linville, James C. Livingston, Ann Loades, David Loades, Jean-Claude Loba-Mkole, Lo Lung Kwong, Wati Longchar, Eleazar López, David W. Lotz, Andrew Louth, Robin W. Lovin, William Luis, Frank D. Macchia, Diarmaid N. J. MacCulloch, Kirk R. MacGregor, Marjory A. MacLean, Donald MacLeod, Tomas S. Maddela, Inge Mager, Laurenti Magesa, David G. Maillu, Fortunato Mallimaci, Philip Mamalakis, Kä Mana, Ukachukwu Chris Manus, Herbert Robinson Marbury, Reuel Norman Marigza, Jacqueline Mariña, Antti Marjanen, Luiz C. L. Marques, Madipoane Masenya (ngwan'a Mphahlele), Caleb J. D. Maskell, Steve Mason, Thomas Massaro, Fernando Matamoros Ponce, András Máté-Tóth, Odair Pedroso Mateus, Dinis Matsolo, Fumitaka Matsuoka, John D'Arcy May, Yelena Mazour-Matusevich, Theodore Mbazumutima, John S. McClure, Christian McConnell, Lee Martin McDonald, Gary B. McGee, Thomas McGowan, Alister E. McGrath, Richard J. McGregor, John A. McGuckin, Maud Burnett McInerney, Elsie Anne McKee, Mary B. McKinley, James F. McMillan, Ernan McMullin, Kathleen E. McVey, M. Douglas Meeks, Monica Jyotsna Melanchthon, Ilie Melniciuc-Puica, Everett Mendoza, Raymond A. Mentzer, William W. Menzies, Ina Merdjanova, Franziska Metzger, Constant J. Mews, Marvin Meyer, Carol Meyers, Vasile Mihoc, Gunner Bjerg Mikkelsen, Maria Inêz de Castro Millen, Clyde Lee Miller, Bonnie J. Miller-McLemore, Alexander Mirkovic, Paul Misner, Nozomu Miyahira, R. W. L. Moberly, Gerald Moede, Aloo Osotsi Mojola, Sunanda Mongia, Rebeca Montemayor, James Moore, Roger E. Moore, Craig E. Morrison O.Carm, Jeffry H. Morrison, Keith Morrison, Wilson J. Moses, Tefetso Henry Mothibe, Mokgethi Motlhabi, Fulata Moyo, Henry Mugabe, Jesse Ndwiga Kanyua Mugambi, Peggy Mulambya-Kabonde, Robert Bruce Mullin, Pamela Mullins Reaves, Saskia Murk Jansen, Heleen L. Murre-Van den Berg, Augustine Musopole, Isaac M. T. Mwase, Philomena Mwaura, Cecilia Nahnfeldt, Anne Nasimiyu Wasike, Carmiña Navia Velasco, Thulani Ndlazi, Alexander Negrov, James B. Nelson, David G. Newcombe, Carol Newsom, Helen J. Nicholson, George W. E. Nickelsburg, Tatyana Nikolskaya, Damayanthi M. A. Niles, Bertil Nilsson, Nyambura Njoroge, Fidelis Nkomazana, Mary Beth Norton, Christian Nottmeier, Sonene Nyawo, Anthère Nzabatsinda, Edward T. Oakes, Gerald O'Collins, Daniel O'Connell, David W. Odell-Scott, Mercy Amba Oduyoye, Kathleen O'Grady, Oyeronke Olajubu, Thomas O'Loughlin, Dennis T. Olson, J. Steven O'Malley, Cephas N. Omenyo, Muriel Orevillo-Montenegro, César Augusto Ornellas Ramos, Agbonkhianmeghe E. Orobator, Kenan B. Osborne, Carolyn Osiek, Javier Otaola Montagne, Douglas F. Ottati, Anna May Say Pa, Irina Paert, Jerry G. Pankhurst, Aristotle Papanikolaou, Samuele F. Pardini, Stefano Parenti, Peter Paris, Sung Bae Park, Cristián G. Parker, Raquel Pastor, Joseph Pathrapankal, Daniel Patte, W. Brown Patterson, Clive Pearson, Keith F. Pecklers, Nancy Cardoso Pereira, David Horace Perkins, Pheme Perkins, Edward N. Peters, Rebecca Todd Peters, Bishop Yeznik Petrossian, Raymond Pfister, Peter C. Phan, Isabel Apawo Phiri, William S. F. Pickering, Derrick G. Pitard, William Elvis Plata, Zlatko Plese, John Plummer, James Newton Poling, Ronald Popivchak, Andrew Porter, Ute Possekel, James M. Powell, Enos Das Pradhan, Devadasan Premnath, Jaime Adrían Prieto Valladares, Anne Primavesi, Randall Prior, María Alicia Puente Lutteroth, Eduardo Guzmão Quadros, Albert Rabil, Laurent William Ramambason, Apolonio M. Ranche, Vololona Randriamanantena Andriamitandrina, Lawrence R. Rast, Paul L. Redditt, Adele Reinhartz, Rolf Rendtorff, Pål Repstad, James N. Rhodes, John K. Riches, Joerg Rieger, Sharon H. Ringe, Sandra Rios, Tyler Roberts, David M. Robinson, James M. Robinson, Joanne Maguire Robinson, Richard A. H. Robinson, Roy R. Robson, Jack B. Rogers, Maria Roginska, Sidney Rooy, Rev. Garnett Roper, Maria José Fontelas Rosado-Nunes, Andrew C. Ross, Stefan Rossbach, François Rossier, John D. Roth, John K. Roth, Phillip Rothwell, Richard E. Rubenstein, Rosemary Radford Ruether, Markku Ruotsila, John E. Rybolt, Risto Saarinen, John Saillant, Juan Sanchez, Wagner Lopes Sanchez, Hugo N. Santos, Gerhard Sauter, Gloria L. Schaab, Sandra M. Schneiders, Quentin J. Schultze, Fernando F. Segovia, Turid Karlsen Seim, Carsten Selch Jensen, Alan P. F. Sell, Frank C. Senn, Kent Davis Sensenig, Damían Setton, Bal Krishna Sharma, Carolyn J. Sharp, Thomas Sheehan, N. Gerald Shenk, Christian Sheppard, Charles Sherlock, Tabona Shoko, Walter B. Shurden, Marguerite Shuster, B. Mark Sietsema, Batara Sihombing, Neil Silberman, Clodomiro Siller, Samuel Silva-Gotay, Heikki Silvet, John K. Simmons, Hagith Sivan, James C. Skedros, Abraham Smith, Ashley A. Smith, Ted A. Smith, Daud Soesilo, Pia Søltoft, Choan-Seng (C. S.) Song, Kathryn Spink, Bryan Spinks, Eric O. Springsted, Nicolas Standaert, Brian Stanley, Glen H. Stassen, Karel Steenbrink, Stephen J. Stein, Andrea Sterk, Gregory E. Sterling, Columba Stewart, Jacques Stewart, Robert B. Stewart, Cynthia Stokes Brown, Ken Stone, Anne Stott, Elizabeth Stuart, Monya Stubbs, Marjorie Hewitt Suchocki, David Kwang-sun Suh, Scott W. Sunquist, Keith Suter, Douglas Sweeney, Charles H. Talbert, Shawqi N. Talia, Elsa Tamez, Joseph B. Tamney, Jonathan Y. Tan, Yak-Hwee Tan, Kathryn Tanner, Feiya Tao, Elizabeth S. Tapia, Aquiline Tarimo, Claire Taylor, Mark Lewis Taylor, Bishop Abba Samuel Wolde Tekestebirhan, Eugene TeSelle, M. Thomas Thangaraj, David R. Thomas, Andrew Thornley, Scott Thumma, Marcelo Timotheo da Costa, George E. “Tink” Tinker, Ola Tjørhom, Karen Jo Torjesen, Iain R. Torrance, Fernando Torres-Londoño, Archbishop Demetrios [Trakatellis], Marit Trelstad, Christine Trevett, Phyllis Trible, Johannes Tromp, Paul Turner, Robert G. Tuttle, Archbishop Desmond Tutu, Peter Tyler, Anders Tyrberg, Justin Ukpong, Javier Ulloa, Camillus Umoh, Kristi Upson-Saia, Martina Urban, Monica Uribe, Elochukwu Eugene Uzukwu, Richard Vaggione, Gabriel Vahanian, Paul Valliere, T. J. Van Bavel, Steven Vanderputten, Peter Van der Veer, Huub Van de Sandt, Louis Van Tongeren, Luke A. Veronis, Noel Villalba, Ramón Vinke, Tim Vivian, David Voas, Elena Volkova, Katharina von Kellenbach, Elina Vuola, Timothy Wadkins, Elaine M. Wainwright, Randi Jones Walker, Dewey D. Wallace, Jerry Walls, Michael J. Walsh, Philip Walters, Janet Walton, Jonathan L. Walton, Wang Xiaochao, Patricia A. Ward, David Harrington Watt, Herold D. Weiss, Laurence L. Welborn, Sharon D. Welch, Timothy Wengert, Traci C. West, Merold Westphal, David Wetherell, Barbara Wheeler, Carolinne White, Jean-Paul Wiest, Frans Wijsen, Terry L. Wilder, Felix Wilfred, Rebecca Wilkin, Daniel H. Williams, D. Newell Williams, Michael A. Williams, Vincent L. Wimbush, Gabriele Winkler, Anders Winroth, Lauri Emílio Wirth, James A. Wiseman, Ebba Witt-Brattström, Teofil Wojciechowski, John Wolffe, Kenman L. Wong, Wong Wai Ching, Linda Woodhead, Wendy M. Wright, Rose Wu, Keith E. Yandell, Gale A. Yee, Viktor Yelensky, Yeo Khiok-Khng, Gustav K. K. Yeung, Angela Yiu, Amos Yong, Yong Ting Jin, You Bin, Youhanna Nessim Youssef, Eliana Yunes, Robert Michael Zaller, Valarie H. Ziegler, Barbara Brown Zikmund, Joyce Ann Zimmerman, Aurora Zlotnik, Zhuo Xinping
- Edited by Daniel Patte, Vanderbilt University, Tennessee
-
- Book:
- The Cambridge Dictionary of Christianity
- Published online:
- 05 August 2012
- Print publication:
- 20 September 2010, pp xi-xliv
-
- Chapter
- Export citation
An improved retinal densitometer: Design concepts and experimental applications
- Howard D. Baker, Ross Henderson, Lawrence P. O'Keefe
-
- Journal:
- Visual Neuroscience / Volume 3 / Issue 1 / July 1989
- Published online by Cambridge University Press:
- 02 June 2009, pp. 71-80
-
- Article
- Export citation
-
A photon-counting retinal densitometer is described that has been designed optically and electronically for improved sensitivity and reliability. The device allows measurement of visual pigments through the undilated natural pupils of subjects at relatively low levels of measuring lights, and serves also as an adaptometer for direct comparisons between pigment bleaching or regeneration and light or dark adaptation. Instrumental control and data collection are by computer to permit rapid and simple data analysis and comparisons between subjects. The methods by which the sensitivity and reliability have been enhanced are described in detail, and some examples of experimental results are presented.
Contributors
-
- By Donald Addington, Jean Addington, Kelly Allott, Amanda Baker, Gregor Berger, Michael Berk, Max Birchwood, Warrick J. Brewer, Peter Burnett, Tyrone Cannon, Andrew Chanen, Philippe Conus, Barbara Cornblatt, Thomas Craig, Alex Fornito, David Fowler, Shona M. Francey, John Gleeson, Susy Harrigan, Meredith Harris, Leanne Hides, Christian G. Huber, Henry J. Jackson, Anthony F. Jorm, Eóin Killackey, Joachim Klosterkötter, Martin Lambert, Tim Lambert, Shon Lewis, Don Linszen, Dan Lubman, Nellie Lucas, Craig Macneil, Ashok K. Malla, Max Marshall, Louise K. McCutcheon, Patrick D. McGorry, Catharine McNab, Maria Michail, Anthony P. Morrison, Merete Nordentoft, Ross M. G. Norman, Keith H. Nuechterlein, Christos Pantelis, Lisa J. Phillips, Richie Poulton, Paddy Power, Jo Robinson, Frauke Schultze-Lutter, Jim van Os, José Luis Vázquez-Barquero, Dennis Velakoulis, Darryl Wade, Daniel Weinberger, Durk Wiersma, Stephen J. Wood, Annemarie Wright, Murat Yücel, Alison R. Yung, Robert B. Zipursky
- Edited by Henry J. Jackson, University of Melbourne, Patrick D. McGorry
-
- Book:
- The Recognition and Management of Early Psychosis
- Published online:
- 10 August 2009
- Print publication:
- 19 February 2009, pp xi-xvi
-
- Chapter
- Export citation
The Vibrations of a Particle about a Position of Equilibrium
- Bevan B. Baker, E. B. Ross
-
- Journal:
- Proceedings of the Edinburgh Mathematical Society / Volume 39 / February 1920
- Published online by Cambridge University Press:
- 20 January 2009, pp. 34-57
-
- Article
-
- You have access Access
- Export citation
-
1. In obtaining a solution of the differential equations corresponding to the motion of a particle about a position of equilibrium, it is usual to express the displacements in terms of a series of periodic terms, each sine or cosine having for its coefficient a series of powers of small quantities. Korteweg has discussed the general form of such solutions, and, from the developments in series which he has obtained, has deduced certain features of interest. In particular, he has shown that, under certain circumstances, it is possible that certain vibrations of higher order, which are normally of small intensity compared with the principal vibrations, may acquire an abnormally large intensity. Considering the oscillations of a dynamical system having a number of degrees of freedom, and supposing
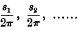 to be the frequencies corresponding to infinitesimal oscillations in the different normal coordinates, Korteweg has shown that these cases of interest arise only when
to be the frequencies corresponding to infinitesimal oscillations in the different normal coordinates, Korteweg has shown that these cases of interest arise only when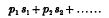
is zero or very small, where p1, p2,… are small integers, positive or negative; the most important cases occur when
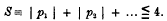
Partners and Rivals: Representation in U.S. Senate Delegations. By Wendy J. Schiller. Princeton: Princeton University Press, 2000. 199p. $49.50 cloth, $18.95 paper
- Ross K. Baker
-
- Journal:
- Perspectives on Politics / Volume 1 / Issue 1 / March 2003
- Published online by Cambridge University Press:
- 09 January 2004, pp. 195-196
- Print publication:
- March 2003
-
- Article
- Export citation
-
Within the past five years, the scholarly literature on the U.S. Senate has been immeasurably enriched by the appearance of a half-dozen books that are noteworthy, not merely for the quality of the research involved but for the perspective they share. Directing their attention to different features of the institution, all emphasize what is truly distinctive about the Senate. In spite of powerful evidence that the hardedged partisanship that has long been a feature of the House has made strong inroads in the Senate, and in the face of evidence provided by Barbara Sinclair's Unorthodox Lawmaking (1997) and others that Senate leadership, at least in the postcommittee phase of legislation, has gotten more muscular in the manner of party leaders in the House, the two chambers are in no danger of becoming indistinguishable one from the other anytime soon.
Actors, Athletes, and Astronauts: Political Amateurs in the United States Congress. By David T. Canon. Chicago: University of Chicago Press, 1990.180p. $32.00 cloth, $14.95 paper.
- Ross K. Baker
-
- Journal:
- American Political Science Review / Volume 85 / Issue 3 / September 1991
- Published online by Cambridge University Press:
- 01 August 2014, pp. 1017-1018
- Print publication:
- September 1991
-
- Article
- Export citation


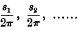 to be the frequencies corresponding to infinitesimal oscillations in the different normal coordinates,
to be the frequencies corresponding to infinitesimal oscillations in the different normal coordinates,