14 results
Cost consequence analysis of Apathy in Dementia Methylphenidate Trial 2 (ADMET 2)
- Krista L. Lanctôt, Clara Chen, Ethan Mah, Alex Kiss, Abby Li, Dave Shade, Roberta W. Scherer, Danielle Vieira, Hamadou Coulibaly, Paul B. Rosenberg, Alan J. Lerner, Prasad R. Padala, Olga Brawman-Mintzer, Christopher H. van Dyck, Anton P. Porsteinsson, Suzanne Craft, Allan Levey, William J. Burke, Jacobo Mintzer, Nathan Herrmann
-
- Journal:
- International Psychogeriatrics / Volume 35 / Issue 11 / November 2023
- Published online by Cambridge University Press:
- 17 April 2023, pp. 664-672
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
-
Background:
This paper used data from the Apathy in Dementia Methylphenidate Trial 2 (NCT02346201) to conduct a planned cost consequence analysis to investigate whether treatment of apathy with methylphenidate is economically attractive.
Methods:A total of 167 patients with clinically significant apathy randomized to either methylphenidate or placebo were included. The Resource Utilization in Dementia Lite instrument assessed resource utilization for the past 30 days and the EuroQol five dimension five level questionnaire assessed health utility at baseline, 3 months, and 6 months. Resources were converted to costs using standard sources and reported in 2021 USD. A repeated measures analysis of variance compared change in costs and utility over time between the treatment and placebo groups. A binary logistic regression was used to assess cost predictors.
Results:Costs were not significantly different between groups whether the cost of methylphenidate was excluded (F(2,330) = 0.626, ηp2 = 0.004, p = 0.535) or included (F(2,330) = 0.629, ηp2 = 0.004, p = 0.534). Utility improved with methylphenidate treatment as there was a group by time interaction (F(2,330) = 7.525, ηp2 = 0.044, p < 0.001).
Discussion:Results from this study indicated that there was no evidence for a difference in resource utilization costs between methylphenidate and placebo treatment. However, utility improved significantly over the 6-month follow-up period. These results can aid in decision-making to improve quality of life in patients with Alzheimer’s disease while considering the burden on the healthcare system.
Adolescents and adults with Fontan circulation: insights from the PREpArE-Fontan registry
- Part of
- Lars Søndergaard, Jamil Aboulhosn, Yves d’Udekem, Céline Faure, Wayne J Franklin, Alfred Hager, Yuli Y Kim, Erwan Muros-Le Rouzic, Daniel Rosenberg, Markus Schwerzmann, Paul Clift
-
- Journal:
- Cardiology in the Young / Volume 32 / Issue 4 / April 2022
- Published online by Cambridge University Press:
- 23 July 2021, pp. 597-605
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
-
The Patient Registry for Adolescents and Adults with Stable Fontan Circulation aims to describe a contemporary cohort of Fontan patients who could be eligible for a clinical trial investigating macitentan, an endothelin receptor antagonist. This international, non-interventional, multicentre, cross-sectional, observational registry enrolled patients with “stable” Fontan circulation ≥10 years following extra-cardiac conduit or lateral tunnel procedure. Main exclusion criteria were NYHA functional class IV, reoperation of Fontan circulation, or signs of disease worsening. Patient characteristics at enrolment are described; available data were collected during a single registration visit. Of the 266 screened patients, 254 were included in this analysis. At enrolment, median (interquartile range) age was 24 (20;30) years, 37%/63% of patients were from the USA/Europe, 54% were male, 54%/47% had undergone extra-cardiac conduit/lateral tunnel procedures, and 95% were in NYHA functional class I or II. History of arrhythmia was more common in older patients and patients with lateral tunnel; overall prevalence was 19%. Most laboratory values were within the normal range but mean creatinine clearance was abnormally low (87.7 ml/min). Angiotensin-converting enzyme inhibitors were used by 48% of patients and their use was associated with creatinine clearance <90 ml/min (p = 0.007), as was Fontan completion at an older age (p = 0.007). 53.4% of patients had clinical characteristics that could potentially meet an endothelin receptor antagonist trial’s eligibility criteria. The PREpArE-Fontan registry describes a cohort of patients who could potentially participate in an endothelin receptor antagonist trial and identified early subtle signs of Fontan failure, even in “stable” patients.
Alcohol-impregnated caps and ambulatory central-line–associated bloodstream infections (CLABSIs): A randomized clinical trial
- Aaron M. Milstone, Carol Rosenberg, Gayane Yenokyan, Danielle W. Koontz, Marlene R. Miller, for the CCLIP Authorship Group
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 42 / Issue 4 / April 2021
- Published online by Cambridge University Press:
- 12 October 2020, pp. 431-439
- Print publication:
- April 2021
-
- Article
- Export citation
-
Objective:
To evaluate the effect of 70% isopropyl alcohol–impregnated central venous catheter caps on ambulatory central-line–associated bloodstream infections (CLABSIs) in pediatric hematology-oncology patients.
Design:This study was a 24-month, cluster-randomized, 2 period, crossover clinical trial.
Setting:The study was conducted in 15 pediatric healthcare institutions, including 16 pediatric hematology-oncology clinics.
Participants:All patients with an external central line followed at 1 of the 16 hematology-oncology clinics.
Intervention:Usual ambulatory central-line care per each institution using 70% isopropyl alcohol–impregnated caps at home compared to usual ambulatory central-line care in each institution without using 70% isopropyl alcohol–impregnated caps.
Results:Of the 16 participating clinics, 15 clinics completed both assignment periods. As assigned, there was no reduction in CLABSI incidence in clinics using 70% isopropyl alcohol–impregnated caps (1.23 per 1,000 days) compared with standard practices (1.38 per 1,000 days; adjusted incidence rate ratio [aIRR], 0.83; 95% CI, 0.63–1.11). In the per-protocol population, there was a reduction in positive blood culture incidence in clinics using 70% isopropyl alcohol-impregnated caps (1.51 per 1,000 days) compared with standard practices (1.88 per 1,000 days; aIRR, 0.72; 95% CI, 0.52–0.99). No adverse events were reported.
Conclusions:Isopropyl alcohol–impregnated central-line caps did not lead to a statistically significant reduction in CLABSI rates in ambulatory hematology-oncology patients. In the per-protocol analysis, there was a statistically significant decrease in positive blood cultures. Larger trials are needed to elucidate the impact of 70% isopropyl alcohol–impregnated caps in the ambulatory setting.
Registration:ClinicalTrials.gov; NCT02351258
Change in agitation in Alzheimer's disease in the placebo arm of a nine-week controlled trial
- Paul B. Rosenberg, Lea T. Drye, Anton P. Porsteinsson, Bruce G. Pollock, D.P. Devanand, Constantine Frangakis, Zahinoor Ismail, Christopher Marano, Curtis L. Meinert, Jacobo E. Mintzer, Cynthia A. Munro, Gregory Pelton, Peter V. Rabins, Lon S. Schneider, David M. Shade, Daniel Weintraub, Jeffery Newell, Jerome Yesavage, Constantine G. Lyketsos, for the CitAD Research Group
-
- Journal:
- International Psychogeriatrics / Volume 27 / Issue 12 / December 2015
- Published online by Cambridge University Press:
- 25 August 2015, pp. 2059-2067
-
- Article
- Export citation
-
Background:
Placebo responses raise significant challenges for the design of clinical trials. We report changes in agitation outcomes in the placebo arm of a recent trial of citalopram for agitation in Alzheimer's disease (CitAD).
Methods:In the CitAD study, all participants and caregivers received a psychosocial intervention and 92 were assigned to placebo for nine weeks. Outcomes included Neurobehavioral Rating Scale agitation subscale (NBRS-A), modified AD Cooperative Study-Clinical Global Impression of Change (CGIC), Cohen-Mansfield Agitation Inventory (CMAI), the Neuropsychiatric Inventory (NPI) Agitation/Aggression domain (NPI A/A) and Total (NPI-Total) and ADLs. Continuous outcomes were analyzed with mixed-effects modeling and dichotomous outcomes with logistic regression.
Results:Agitation outcomes improved over nine weeks: NBRS-A mean (SD) decreased from 7.8 (3.0) at baseline to 5.4 (3.2), CMAI from 28.7 (6.7) to 26.7 (7.4), NPI A/A from 8.0 (2.4) to 4.9 (3.8), and NPI-Total from 37.3 (17.7) to 28.4 (22.1). The proportion of CGI-C agitation responders ranged from 21 to 29% and was significantly different from zero. MMSE improved from 14.4 (6.9) to 15.7 (7.2) and ADLs similarly improved. Most of the improvement was observed by three weeks and was sustained through nine weeks. The major predictor of improvement in each agitation measure was a higher baseline score in that measure.
Conclusions:We observed significant placebo response which may be due to regression to the mean, response to a psychosocial intervention, natural course of symptoms, or nonspecific benefits of participation in a trial.
Contributors
-
- By Mitchell Aboulafia, Frederick Adams, Marilyn McCord Adams, Robert M. Adams, Laird Addis, James W. Allard, David Allison, William P. Alston, Karl Ameriks, C. Anthony Anderson, David Leech Anderson, Lanier Anderson, Roger Ariew, David Armstrong, Denis G. Arnold, E. J. Ashworth, Margaret Atherton, Robin Attfield, Bruce Aune, Edward Wilson Averill, Jody Azzouni, Kent Bach, Andrew Bailey, Lynne Rudder Baker, Thomas R. Baldwin, Jon Barwise, George Bealer, William Bechtel, Lawrence C. Becker, Mark A. Bedau, Ernst Behler, José A. Benardete, Ermanno Bencivenga, Jan Berg, Michael Bergmann, Robert L. Bernasconi, Sven Bernecker, Bernard Berofsky, Rod Bertolet, Charles J. Beyer, Christian Beyer, Joseph Bien, Joseph Bien, Peg Birmingham, Ivan Boh, James Bohman, Daniel Bonevac, Laurence BonJour, William J. Bouwsma, Raymond D. Bradley, Myles Brand, Richard B. Brandt, Michael E. Bratman, Stephen E. Braude, Daniel Breazeale, Angela Breitenbach, Jason Bridges, David O. Brink, Gordon G. Brittan, Justin Broackes, Dan W. Brock, Aaron Bronfman, Jeffrey E. Brower, Bartosz Brozek, Anthony Brueckner, Jeffrey Bub, Lara Buchak, Otavio Bueno, Ann E. Bumpus, Robert W. Burch, John Burgess, Arthur W. Burks, Panayot Butchvarov, Robert E. Butts, Marina Bykova, Patrick Byrne, David Carr, Noël Carroll, Edward S. Casey, Victor Caston, Victor Caston, Albert Casullo, Robert L. Causey, Alan K. L. Chan, Ruth Chang, Deen K. Chatterjee, Andrew Chignell, Roderick M. Chisholm, Kelly J. Clark, E. J. Coffman, Robin Collins, Brian P. Copenhaver, John Corcoran, John Cottingham, Roger Crisp, Frederick J. Crosson, Antonio S. Cua, Phillip D. Cummins, Martin Curd, Adam Cureton, Andrew Cutrofello, Stephen Darwall, Paul Sheldon Davies, Wayne A. Davis, Timothy Joseph Day, Claudio de Almeida, Mario De Caro, Mario De Caro, John Deigh, C. F. Delaney, Daniel C. Dennett, Michael R. DePaul, Michael Detlefsen, Daniel Trent Devereux, Philip E. Devine, John M. Dillon, Martin C. Dillon, Robert DiSalle, Mary Domski, Alan Donagan, Paul Draper, Fred Dretske, Mircea Dumitru, Wilhelm Dupré, Gerald Dworkin, John Earman, Ellery Eells, Catherine Z. Elgin, Berent Enç, Ronald P. Endicott, Edward Erwin, John Etchemendy, C. Stephen Evans, Susan L. Feagin, Solomon Feferman, Richard Feldman, Arthur Fine, Maurice A. Finocchiaro, William FitzPatrick, Richard E. Flathman, Gvozden Flego, Richard Foley, Graeme Forbes, Rainer Forst, Malcolm R. Forster, Daniel Fouke, Patrick Francken, Samuel Freeman, Elizabeth Fricker, Miranda Fricker, Michael Friedman, Michael Fuerstein, Richard A. Fumerton, Alan Gabbey, Pieranna Garavaso, Daniel Garber, Jorge L. A. Garcia, Robert K. Garcia, Don Garrett, Philip Gasper, Gerald Gaus, Berys Gaut, Bernard Gert, Roger F. Gibson, Cody Gilmore, Carl Ginet, Alan H. Goldman, Alvin I. Goldman, Alfonso Gömez-Lobo, Lenn E. Goodman, Robert M. Gordon, Stefan Gosepath, Jorge J. E. Gracia, Daniel W. Graham, George A. Graham, Peter J. Graham, Richard E. Grandy, I. Grattan-Guinness, John Greco, Philip T. Grier, Nicholas Griffin, Nicholas Griffin, David A. Griffiths, Paul J. Griffiths, Stephen R. Grimm, Charles L. Griswold, Charles B. Guignon, Pete A. Y. Gunter, Dimitri Gutas, Gary Gutting, Paul Guyer, Kwame Gyekye, Oscar A. Haac, Raul Hakli, Raul Hakli, Michael Hallett, Edward C. Halper, Jean Hampton, R. James Hankinson, K. R. Hanley, Russell Hardin, Robert M. Harnish, William Harper, David Harrah, Kevin Hart, Ali Hasan, William Hasker, John Haugeland, Roger Hausheer, William Heald, Peter Heath, Richard Heck, John F. Heil, Vincent F. Hendricks, Stephen Hetherington, Francis Heylighen, Kathleen Marie Higgins, Risto Hilpinen, Harold T. Hodes, Joshua Hoffman, Alan Holland, Robert L. Holmes, Richard Holton, Brad W. Hooker, Terence E. Horgan, Tamara Horowitz, Paul Horwich, Vittorio Hösle, Paul Hoβfeld, Daniel Howard-Snyder, Frances Howard-Snyder, Anne Hudson, Deal W. Hudson, Carl A. Huffman, David L. Hull, Patricia Huntington, Thomas Hurka, Paul Hurley, Rosalind Hursthouse, Guillermo Hurtado, Ronald E. Hustwit, Sarah Hutton, Jonathan Jenkins Ichikawa, Harry A. Ide, David Ingram, Philip J. Ivanhoe, Alfred L. Ivry, Frank Jackson, Dale Jacquette, Joseph Jedwab, Richard Jeffrey, David Alan Johnson, Edward Johnson, Mark D. Jordan, Richard Joyce, Hwa Yol Jung, Robert Hillary Kane, Tomis Kapitan, Jacquelyn Ann K. Kegley, James A. Keller, Ralph Kennedy, Sergei Khoruzhii, Jaegwon Kim, Yersu Kim, Nathan L. King, Patricia Kitcher, Peter D. Klein, E. D. Klemke, Virginia Klenk, George L. Kline, Christian Klotz, Simo Knuuttila, Joseph J. Kockelmans, Konstantin Kolenda, Sebastian Tomasz Kołodziejczyk, Isaac Kramnick, Richard Kraut, Fred Kroon, Manfred Kuehn, Steven T. Kuhn, Henry E. Kyburg, John Lachs, Jennifer Lackey, Stephen E. Lahey, Andrea Lavazza, Thomas H. Leahey, Joo Heung Lee, Keith Lehrer, Dorothy Leland, Noah M. Lemos, Ernest LePore, Sarah-Jane Leslie, Isaac Levi, Andrew Levine, Alan E. Lewis, Daniel E. Little, Shu-hsien Liu, Shu-hsien Liu, Alan K. L. Chan, Brian Loar, Lawrence B. Lombard, John Longeway, Dominic McIver Lopes, Michael J. Loux, E. J. Lowe, Steven Luper, Eugene C. Luschei, William G. Lycan, David Lyons, David Macarthur, Danielle Macbeth, Scott MacDonald, Jacob L. Mackey, Louis H. Mackey, Penelope Mackie, Edward H. Madden, Penelope Maddy, G. B. Madison, Bernd Magnus, Pekka Mäkelä, Rudolf A. Makkreel, David Manley, William E. Mann (W.E.M.), Vladimir Marchenkov, Peter Markie, Jean-Pierre Marquis, Ausonio Marras, Mike W. Martin, A. P. Martinich, William L. McBride, David McCabe, Storrs McCall, Hugh J. McCann, Robert N. McCauley, John J. McDermott, Sarah McGrath, Ralph McInerny, Daniel J. McKaughan, Thomas McKay, Michael McKinsey, Brian P. McLaughlin, Ernan McMullin, Anthonie Meijers, Jack W. Meiland, William Jason Melanson, Alfred R. Mele, Joseph R. Mendola, Christopher Menzel, Michael J. Meyer, Christian B. Miller, David W. Miller, Peter Millican, Robert N. Minor, Phillip Mitsis, James A. Montmarquet, Michael S. Moore, Tim Moore, Benjamin Morison, Donald R. Morrison, Stephen J. Morse, Paul K. Moser, Alexander P. D. Mourelatos, Ian Mueller, James Bernard Murphy, Mark C. Murphy, Steven Nadler, Jan Narveson, Alan Nelson, Jerome Neu, Samuel Newlands, Kai Nielsen, Ilkka Niiniluoto, Carlos G. Noreña, Calvin G. Normore, David Fate Norton, Nikolaj Nottelmann, Donald Nute, David S. Oderberg, Steve Odin, Michael O’Rourke, Willard G. Oxtoby, Heinz Paetzold, George S. Pappas, Anthony J. Parel, Lydia Patton, R. P. Peerenboom, Francis Jeffry Pelletier, Adriaan T. Peperzak, Derk Pereboom, Jaroslav Peregrin, Glen Pettigrove, Philip Pettit, Edmund L. Pincoffs, Andrew Pinsent, Robert B. Pippin, Alvin Plantinga, Louis P. Pojman, Richard H. Popkin, John F. Post, Carl J. Posy, William J. Prior, Richard Purtill, Michael Quante, Philip L. Quinn, Philip L. Quinn, Elizabeth S. Radcliffe, Diana Raffman, Gerard Raulet, Stephen L. Read, Andrews Reath, Andrew Reisner, Nicholas Rescher, Henry S. Richardson, Robert C. Richardson, Thomas Ricketts, Wayne D. Riggs, Mark Roberts, Robert C. Roberts, Luke Robinson, Alexander Rosenberg, Gary Rosenkranz, Bernice Glatzer Rosenthal, Adina L. Roskies, William L. Rowe, T. M. Rudavsky, Michael Ruse, Bruce Russell, Lilly-Marlene Russow, Dan Ryder, R. M. Sainsbury, Joseph Salerno, Nathan Salmon, Wesley C. Salmon, Constantine Sandis, David H. Sanford, Marco Santambrogio, David Sapire, Ruth A. Saunders, Geoffrey Sayre-McCord, Charles Sayward, James P. Scanlan, Richard Schacht, Tamar Schapiro, Frederick F. Schmitt, Jerome B. Schneewind, Calvin O. Schrag, Alan D. Schrift, George F. Schumm, Jean-Loup Seban, David N. Sedley, Kenneth Seeskin, Krister Segerberg, Charlene Haddock Seigfried, Dennis M. Senchuk, James F. Sennett, William Lad Sessions, Stewart Shapiro, Tommie Shelby, Donald W. Sherburne, Christopher Shields, Roger A. Shiner, Sydney Shoemaker, Robert K. Shope, Kwong-loi Shun, Wilfried Sieg, A. John Simmons, Robert L. Simon, Marcus G. Singer, Georgette Sinkler, Walter Sinnott-Armstrong, Matti T. Sintonen, Lawrence Sklar, Brian Skyrms, Robert C. Sleigh, Michael Anthony Slote, Hans Sluga, Barry Smith, Michael Smith, Robin Smith, Robert Sokolowski, Robert C. Solomon, Marta Soniewicka, Philip Soper, Ernest Sosa, Nicholas Southwood, Paul Vincent Spade, T. L. S. Sprigge, Eric O. Springsted, George J. Stack, Rebecca Stangl, Jason Stanley, Florian Steinberger, Sören Stenlund, Christopher Stephens, James P. Sterba, Josef Stern, Matthias Steup, M. A. Stewart, Leopold Stubenberg, Edith Dudley Sulla, Frederick Suppe, Jere Paul Surber, David George Sussman, Sigrún Svavarsdóttir, Zeno G. Swijtink, Richard Swinburne, Charles C. Taliaferro, Robert B. Talisse, John Tasioulas, Paul Teller, Larry S. Temkin, Mark Textor, H. S. Thayer, Peter Thielke, Alan Thomas, Amie L. Thomasson, Katherine Thomson-Jones, Joshua C. Thurow, Vzalerie Tiberius, Terrence N. Tice, Paul Tidman, Mark C. Timmons, William Tolhurst, James E. Tomberlin, Rosemarie Tong, Lawrence Torcello, Kelly Trogdon, J. D. Trout, Robert E. Tully, Raimo Tuomela, John Turri, Martin M. Tweedale, Thomas Uebel, Jennifer Uleman, James Van Cleve, Harry van der Linden, Peter van Inwagen, Bryan W. Van Norden, René van Woudenberg, Donald Phillip Verene, Samantha Vice, Thomas Vinci, Donald Wayne Viney, Barbara Von Eckardt, Peter B. M. Vranas, Steven J. Wagner, William J. Wainwright, Paul E. Walker, Robert E. Wall, Craig Walton, Douglas Walton, Eric Watkins, Richard A. Watson, Michael V. Wedin, Rudolph H. Weingartner, Paul Weirich, Paul J. Weithman, Carl Wellman, Howard Wettstein, Samuel C. Wheeler, Stephen A. White, Jennifer Whiting, Edward R. Wierenga, Michael Williams, Fred Wilson, W. Kent Wilson, Kenneth P. Winkler, John F. Wippel, Jan Woleński, Allan B. Wolter, Nicholas P. Wolterstorff, Rega Wood, W. Jay Wood, Paul Woodruff, Alison Wylie, Gideon Yaffe, Takashi Yagisawa, Yutaka Yamamoto, Keith E. Yandell, Xiaomei Yang, Dean Zimmerman, Günter Zoller, Catherine Zuckert, Michael Zuckert, Jack A. Zupko (J.A.Z.)
- Edited by Robert Audi, University of Notre Dame, Indiana
-
- Book:
- The Cambridge Dictionary of Philosophy
- Published online:
- 05 August 2015
- Print publication:
- 27 April 2015, pp ix-xxx
-
- Chapter
- Export citation
Contributors
-
- By Agoston T. Agoston, Syed Z. Ali, Mahul B. Amin, Daniel A. Arber, Pedram Argani, Sylvia L. Asa, Rebecca N. Baergen, Zubair W. Baloch, Andrew M. Bellizzi, Kurt Benirschke, Allen Burke, Kenneth B. Calder, Karen L. Chang, Rebecca D. Chernock, Wang Cheung, Thomas V. Colby, Byron P. Croker, Ronald A. DeLellis, Edward F. DiCarlo, Ralph C. Eagle, Hormoz Ehya, Brett M. Elicker, Tarik M. Elsheikh, Robert E. Fechner, Linda D. Ferrell, Melina B. Flanagan, Douglas B. Flieder, Christopher S. Foster, Lillian Gaber, Karuna Garg, Kim R. Geisinger, Ryan M. Gill, Eric F. Glassy, David J. Glembocki, Zachary D. Goodman, Robert O. Greer, David J. Grignon, Gerardo E. Guiter, Kymberly A. Gyure, Ian S. Hagemann, Michael R. Henry, Jason L. Hornick, Ralph H. Hruban, Phyllis C. Huettner, Peter A. Humphrey, Olga B. Ioffe, Edward C. Klatt, Michael J. Klein, Ernest E. Lack, James N. Lampros, Lester J. Layfield, Robin D. LeGallo, Kevin O. Leslie, James S. Lewis, Virginia A. LiVolsi, Alberto M. Marchevsky, Anne Marie McNicol, Mitra Mehrad, Elizabeth Montgomery, Cesar A. Moran, Christopher A. Moskaluk, George J. Netto, G. Petur Nielsen, Robert D. Odze, Arthur S. Patchefsky, James W. Patterson, Elizabeth N. Pavlisko, John D. Pfeifer, Celeste N. Powers, Richard A. Prayson, Anja C. Roden, Victor L. Roggli, Andrew E. Rosenberg, Sherif Said, Margie A. Scott, Raja R. Seethala, Carlie S. Sigel, Jan F. Silverman, Bruce R. Smoller, Edward B. Stelow, Nora C. J. Sun, Mark W. Teague, Satish K. Tickoo, Thomas M. Ulbright, Paul E. Wakely, Jun Wang, Lawrence M. Weiss, Mark R. Wick, Howard H. Wu, Rhonda K. Yantiss, Charles Zaloudek, Yaxia Zhang, Xiaohui Sheila Zhao
- Edited by Mark R. Wick, University of Virginia, Virginia A. LiVolsi, University of Pennsylvania School of Medicine, John D. Pfeifer, Washington University School of Medicine, St Louis, Edward B. Stelow, University of Virginia, Paul E. Wakely, Jr
-
- Book:
- Silverberg's Principles and Practice of Surgical Pathology and Cytopathology
- Published online:
- 13 March 2015
- Print publication:
- 26 March 2015, pp vii-x
-
- Chapter
- Export citation
Consumption of soda and other sugar-sweetened beverages by 2-year-olds: findings from a population-based survey
- Bernice Raveche Garnett, Kenneth D Rosenberg, Daniel S Morris
-
- Journal:
- Public Health Nutrition / Volume 16 / Issue 10 / October 2013
- Published online by Cambridge University Press:
- 04 October 2012, pp. 1760-1767
-
- Article
-
- You have access Access
- HTML
- Export citation
-
Objective
To determine risk factors for consumption of soda and other sugar-sweetened beverages (SSB) among 2-year-old children.
DesignThe analysis was performed using three linked data sets: the 2004–2005 Oregon Pregnancy Risk Assessment Monitoring Survey (PRAMS); its longitudinal follow-up, 2006–2007 Oregon PRAMS-2; and 2004–2005 Oregon birth certificates.
SettingPRAMS is a surveillance programme supported by the federal Centers for Disease Control and Prevention and implemented by participating state health departments. Using mixed methods, PRAMS surveys women 2–6 months after a live birth. Oregon PRAMS-2 re-interviews respondents shortly after the index child's second birthday. Oregon PRAMS oversamples minority women.
SubjectsUsing monthly cohorts, we randomly selected 5851 women from the 2004–2005 birth certificates. In total 1911 women completed both PRAMS and PRAMS-2. The weighted response rate of PRAMS-2 was 43·5 %.
ResultsAlmost half of mothers (49·9 %) reported that their child drank SSB on at least 1 d/week. Mothers whose children drank SSB at least once weekly were more likely to have low income (adjusted OR = 2·83, 95 % CI 2·09, 3·83) and to eat out on ≥2 d/week (OR = 2·11 %, 95 % CI 1·66, 2·70). Hispanic and non-Hispanic black women were most likely to report that their child drank SSB at least once weekly.
ConclusionsHalf of mothers reported that their 2-year-old children drank SSB at least once weekly. Public health interventions and policies should address childhood SSB consumption including educating health-care providers and parents.
Management of children undergoing cardiac transplantation with high Panel Reactive Antibodies
- Alfred Asante-Korang, Jeffrey P. Jacobs, Jeremy Ringewald, Jennifer Carapellucci, Kristin Rosenberg, Daniel McKenna, Jorge McCormack, Ivan Wilmot, Abigail Gjeldum, Mayra Lopez-Cepero, John Sleasman
-
- Journal:
- Cardiology in the Young / Volume 21 / Issue S2 / 13 December 2011
- Published online by Cambridge University Press:
- 13 December 2011, pp. 124-132
-
- Article
- Export citation
-
Highly sensitised children in need of cardiac transplantation have overall poor outcomes because of increased risk for dysfunction of the cardiac allograft, acute cellular and antibody-mediated rejection, and vasculopathy of the cardiac allograft. Cardiopulmonary bypass and the frequent use of blood products in the operating room and cardiac intensive care unit, as well as the frequent use of homografts, have predisposed potential recipients of transplants to allosensitisation. The expansion in the use of ventricular assist devices and extracorporeal membrane oxygenation has also contributed to increasing rates of allosensitisation in candidates for cardiac transplantation. Antibodies to Human Leukocyte Antigen can be detected before transplantation using several different techniques, the most common being the “complement-dependent lymphocytotoxicity assays”. “Solid-phase assays”, particularly the “Luminex® single antigen bead method”, offer improved specificity and more detailed information regarding specificities of antibodies, leading to improved matching of donors with recipients. Allosensitisation prolongs the time on the waiting list for potential recipients of transplantation and increases the risk of complications and death after transplantation. Aggressive reduction of antibodies to Human Leukocyte Antigen in these high-risk patients is therefore of vital importance for long-term survival of the patient and cardiac allograft. Strategies to decrease Panel Reactive Antibody or percent reactive antibody before transplantation include plasmapheresis, intravenous administration of immunoglobulin, and specific treatment to reduce B-cells, particularly Rituximab. These strategies have resulted in varying degrees of success. Antibody-mediated rejection and cardiac allograft vasculopathy are two of the most important complications of transplantation in patients with high Panel Reactive Antibody. The treatment of antibody-mediated rejection in recipients of cardiac transplants is largely empirical and includes the use of high-dose corticosteroids, plasmapheresis, intravenous administration of immunoglobulins, anti-thymocyte globulin, and Rituximab. Cardiac allograft vasculopathy is believed to be secondary to chronic complement-mediated endothelial injury and chronic vascular rejection. The use of proliferation signal inhibitors, such as sirolimus and everolimus, has been shown to delay the progression of cardiac allograft vasculopathy. In some non-sensitised recipients of cardiac transplants, the de novo formation of antibodies to Human Leukocyte Antigen after transplantation may increase the likelihood of adverse clinical outcomes. The use of serial testing for donor-specific antibodies after cardiac transplantation may be advisable in patients with frequent episodes of rejection and patients with history of sensitisation. Allosensitisation before transplantation can negatively influence outcomes after transplantation. A high incidence of antibody-mediated rejection and graft vasculopathy can result in graft failure and decreased survival. Current strategies to decrease allosensitisation have helped to expand the pool of donors, improve times on the waiting list, and decrease mortality. Centres of transplantation offering desensitisation are currently using plasmapheresis to remove circulating antibodies; intravenous immunoglobulin to inactivate antibodies; cyclophosphamide to suppress B-cell proliferation; and Rituximab to deplete B-lymphocytes. Similar approaches are also used to treat antibody-mediated rejection after transplantation with promising results.
Contributors
-
- By Robert Acosta, Elizabeth M. Alderman, Dan Barlev, Stephen M. Blumberg, Katherine J. Chou, Anthony J. Ciorciari, Christina M. Coyle, Ellen F. Crain, Sandra J. Cunningham, Joan Di Martino-Nardi, Nancy Dougherty, Glenn Fennelly, Sheila Fallon Friedlander, Jeffrey C. Gershel, Michael H. Gewitz, Beatrice Goilav, Michael Gorn, Waseem Hafeez, Dominic Hollman, Olga Jimenez, Carl Kaplan, Jeffrey Keller, Sergey Kunkov, Carolyn Lederman, Martin Lederman, Stephanie R. Lichten, Julie Lin, Stephen Ludwig, Svetlana Lvovich, Frank A. Maffei, Soe Mar, Robert W. Marion, Morri Markowitz, Daniel Mason, Teresa McCann, Alexandra D. McCollum, Mary Mehlman, James Meltzer, Scott Miller, Kirsten Roberts, Michael Rosenberg, Joy Samanich, David P. Sole, Preeti Venkataraman, Joshua Vova, Mark Weinblatt, Paul K. Woolf, Loren Yellin
- Edited by Ellen F. Crain, Albert Einstein College of Medicine, New York, Jeffrey C. Gershel, Albert Einstein College of Medicine, New York
- Edited in association with Sandra J. Cunningham
-
- Book:
- Clinical Manual of Emergency Pediatrics
- Published online:
- 10 January 2011
- Print publication:
- 02 December 2010, pp x-xiv
-
- Chapter
- Export citation
Traditional knowledge and satellite tracking as complementary approaches to ecological understanding
- HENRY P. HUNTINGTON, ROBERT S. SUYDAM, DANIEL H. ROSENBERG
-
- Journal:
- Environmental Conservation / Volume 31 / Issue 3 / September 2004
- Published online by Cambridge University Press:
- 11 January 2005, pp. 177-180
-
- Article
- Export citation
-
The integration or co-application of traditional knowledge and scientific knowledge has been the subject of considerable research and discussion (see Johannes 1981; Johnson 1992; Stevenson 1996; McDonald et al. 1997; Huntington et al. 1999, 2002), with emphasis on various specific topics including environmental management and conservation (see Freeman & Carbyn 1988; Ferguson & Messier 1997; Ford & Martinez 2000; Usher 2000; Albert 2001). In most cases, examples of successful integration compare traditional and scientific observations at similar spatial scales to increase confidence in understanding or to fill gaps that appear from either perspective. We present a different approach to integration, emphasizing complementarity rather than concordance in spatial perspective, using two migratory species as examples.
Looking Backward, Looking Forward: MLA Members Speak
- April Alliston, Elizabeth Ammons, Jean Arnold, Nina Baym, Sandra L. Beckett, Peter G. Beidler, Roger A. Berger, Sandra Bermann, J.J. Wilson, Troy Boone, Alison Booth, Wayne C. Booth, James Phelan, Marie Borroff, Ihab Hassan, Ulrich Weisstein, Zack Bowen, Jill Campbell, Dan Campion, Jay Caplan, Maurice Charney, Beverly Lyon Clark, Robert A. Colby, Thomas C. Coleman III, Nicole Cooley, Richard Dellamora, Morris Dickstein, Terrell Dixon, Emory Elliott, Caryl Emerson, Ann W. Engar, Lars Engle, Kai Hammermeister, N. N. Feltes, Mary Anne Ferguson, Annie Finch, Shelley Fisher Fishkin, Jerry Aline Flieger, Norman Friedman, Rosemarie Garland-Thomson, Sandra M. Gilbert, Laurie Grobman, George Guida, Liselotte Gumpel, R. K. Gupta, Florence Howe, Cathy L. Jrade, Richard A. Kaye, Calhoun Winton, Murray Krieger, Robert Langbaum, Richard A. Lanham, Marilee Lindemann, Paul Michael Lützeler, Thomas J. Lynn, Juliet Flower MacCannell, Michelle A. Massé, Irving Massey, Georges May, Christian W. Hallstein, Gita May, Lucy McDiarmid, Ellen Messer-Davidow, Koritha Mitchell, Robin Smiles, Kenyatta Albeny, George Monteiro, Joel Myerson, Alan Nadel, Ashton Nichols, Jeffrey Nishimura, Neal Oxenhandler, David Palumbo-Liu, Vincent P. Pecora, David Porter, Nancy Potter, Ronald C. Rosbottom, Elias L. Rivers, Gerhard F. Strasser, J. L. Styan, Marianna De Marco Torgovnick, Gary Totten, David van Leer, Asha Varadharajan, Orrin N. C. Wang, Sharon Willis, Louise E. Wright, Donald A. Yates, Takayuki Yokota-Murakami, Richard E. Zeikowitz, Angelika Bammer, Dale Bauer, Karl Beckson, Betsy A. Bowen, Stacey Donohue, Sheila Emerson, Gwendolyn Audrey Foster, Jay L. Halio, Karl Kroeber, Terence Hawkes, William B. Hunter, Mary Jambus, Willard F. King, Nancy K. Miller, Jody Norton, Ann Pellegrini, S. P. Rosenbaum, Lorie Roth, Robert Scholes, Joanne Shattock, Rosemary T. VanArsdel, Alfred Bendixen, Alarma Kathleen Brown, Michael J. Kiskis, Debra A. Castillo, Rey Chow, John F. Crossen, Robert F. Fleissner, Regenia Gagnier, Nicholas Howe, M. Thomas Inge, Frank Mehring, Hyungji Park, Jahan Ramazani, Kenneth M. Roemer, Deborah D. Rogers, A. LaVonne Brown Ruoff, Regina M. Schwartz, John T. Shawcross, Brenda R. Silver, Andrew von Hendy, Virginia Wright Wexman, Britta Zangen, A. Owen Aldridge, Paula R. Backscheider, Roland Bartel, E. M. Forster, Milton Birnbaum, Jonathan Bishop, Crystal Downing, Frank H. Ellis, Roberto Forns-Broggi, James R. Giles, Mary E. Giles, Susan Blair Green, Madelyn Gutwirth, Constance B. Hieatt, Titi Adepitan, Edgar C. Knowlton, Jr., Emanuel Mussman, Sally Todd Nelson, Robert O. Preyer, David Diego Rodriguez, Guy Stern, James Thorpe, Robert J. Wilson, Rebecca S. Beal, Joyce Simutis, Betsy Bowden, Sara Cooper, Wheeler Winston Dixon, Tarek el Ariss, Richard Jewell, John W. Kronik, Wendy Martin, Stuart Y. McDougal, Hugo Méndez-Ramírez, Ivy Schweitzer, Armand E. Singer, G. Thomas Tanselle, Tom Bishop, Mary Ann Caws, Marcel Gutwirth, Christophe Ippolito, Lawrence D. Kritzman, James Longenbach, Tim McCracken, Wolfe S. Molitor, Diane Quantic, Gregory Rabassa, Ellen M. Tsagaris, Anthony C. Yu, Betty Jean Craige, Wendell V. Harris, J. Hillis Miller, Jesse G. Swan, Helene Zimmer-Loew, Peter Berek, James Chandler, Hanna K. Charney, Philip Cohen, Judith Fetterley, Herbert Lindenberger, Julia Reinhard Lupton, Maximillian E. Novak, Richard Ohmann, Marjorie Perloff, Mark Reynolds, James Sledd, Harriet Turner, Marie Umeh, Flavia Aloya, Regina Barreca, Konrad Bieber, Ellis Hanson, William J. Hyde, Holly A. Laird, David Leverenz, Allen Michie, J. Wesley Miller, Marvin Rosenberg, Daniel R. Schwarz, Elizabeth Welt Trahan, Jean Fagan Yellin
-
- Journal:
- PMLA / Publications of the Modern Language Association of America / Volume 115 / Issue 7 / December 2000
- Published online by Cambridge University Press:
- 23 October 2020, pp. 1986-2078
- Print publication:
- December 2000
-
- Article
- Export citation
Rb1C60: Linear Polymer Chains and Dimers
- Michael C. Martin, Daniel Koller, A. Rosenberg, C. Kendziora, L. Mihaly
-
- Journal:
- MRS Online Proceedings Library Archive / Volume 359 / 1994
- Published online by Cambridge University Press:
- 15 February 2011, 319
- Print publication:
- 1994
-
- Article
- Export citation
-
The infrared- and Raman-active vibrational modes of C60 were measured in the various structural states of Rb1C60. According to earlier studies, Rb1 C60 has an f cc structure at temperatures above ∼ 100°C, a linear chain polymer orthorhombic structure when slowly cooled, and an as yet undetermined structure when very rapidly cooled (“quenched”). We show that the spectra obtained in the polymer state are consistent with each C60 molecule having bonds to two diametrically opposite neighbors. In the quenched state, we find evidence for further symmetry breaking, implying a lower symmetry structure than the polymer state. The spectroscopic data of the quenched phase are shown to be consistent with Rb2(C60)2, a dimerization of C60.
Hospital-Sponsored Preferred Provider Organizations
- Daniel T. Roble, William A. Knowlton, Gary A. Rosenberg
-
- Journal:
- Law, Medicine and Health Care / Volume 12 / Issue 5 / October 1984
- Published online by Cambridge University Press:
- 28 April 2021, pp. 204-209
- Print publication:
- October 1984
-
- Article
- Export citation
-
The recent growth of new forms of health care delivery has been encouraged by the increase in competition in the health care industry. Several factors account for this increase in competition. First, there is an overabundance of physicians in many areas of the United States. In addition, many overbedded hospitals now compete for the same patients to fill their beds. Finally, health maintenance organizations and other alternative delivery systems, such as ambulatory surgical care centers, ‘surgicenters,” and birthing centers, are capturing more of the patient market and reducing hospital inpatient utilization.
Employers, insurers, and the government, as the third-party payers who most often pay for an individual's medical care, hope to take advantage of this new competitive environment and to moderate the increase in their health care expenditures.
On the Dimension of Modules and Algebras, VIII. Dimension of Tensor Products
- Samuel Eilenberg, Alex Rosenberg, Daniel Zelinsky
-
- Journal:
- Nagoya Mathematical Journal / Volume 12 / December 1957
- Published online by Cambridge University Press:
- 22 January 2016, pp. 71-93
- Print publication:
- December 1957
-
- Article
-
- You have access Access
- Export citation
-
The questions concerning the dimension of the tensor product
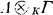 of two K-algebras have turned out to be surprisingly difficult. In this paper we follow a method using spectral sequences (§§1-3) which in some concrete cases yields complete results (§§4-5). In particular, complete results are obtained when r is a ring of matrices, triangular matrices, polynomials or rational functions, so that in the first three cases
of two K-algebras have turned out to be surprisingly difficult. In this paper we follow a method using spectral sequences (§§1-3) which in some concrete cases yields complete results (§§4-5). In particular, complete results are obtained when r is a ring of matrices, triangular matrices, polynomials or rational functions, so that in the first three cases 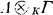 is respectively the ring of matrices, triangular matrices or polynomials with coefficients in the arbitrary algebra A.
is respectively the ring of matrices, triangular matrices or polynomials with coefficients in the arbitrary algebra A.




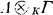 of two
of two