37 results
Numerical simulation of bubble dynamics and segregation in binary heptane/dodecane mixtures
- J.M. Bermudez-Graterol, R. Skoda
-
- Journal:
- Journal of Fluid Mechanics / Volume 947 / 25 September 2022
- Published online by Cambridge University Press:
- 17 August 2022, A9
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
-
A mathematical model for the simulation of spherical bubble dynamics in binary alkane mixtures is presented. Detailed heat and mass transfer and phase transition are resolved, and air absorption and desorption are considered. As example mixtures, high-volatile heptane and low-volatile dodecane are investigated. The low-volatile component shows a convective counter-intuitive mass transport opposite to the diffusion flux. A staggered mass flux of heptane and dodecane is associated with a local segregation of mixture components in the surrounding liquid and a distinctive species distribution within the bubble. A comparative study with a pseudo-fluid where alkane species interdiffusion is absent and whose components cannot segregate reveals that for an oscillating bubble, the local segregation hardly affects bubble dynamics, while for a continuously growing bubble in a superheated liquid, the growth rate is considerably affected, particularly for a low heptane mixture percentage. This study demonstrates limitations of treating mixtures by a single-component surrogate fluid and may serve as a starting point for the development of multi-component cavitation models for computational fluid dynamics applications in real fluid mixtures as, e.g. fuels or hydraulic oils.
Paramarkeyite, a new calcium–uranyl–carbonate mineral from the Markey mine, San Juan County, Utah, USA
- Anthony R. Kampf, Travis A. Olds, Jakub Plášil, Peter C. Burns, Radek Škoda, Joe Marty
-
- Journal:
- Mineralogical Magazine / Volume 86 / Issue 1 / February 2022
- Published online by Cambridge University Press:
- 13 December 2021, pp. 27-36
-
- Article
- Export citation
-
The new mineral paramarkeyite (IMA2021-024), Ca2(UO2)(CO3)3⋅5H2O, was found in the Markey mine, San Juan County, Utah, USA, where it occurs as a secondary phase on gypsum-coated asphaltum in association with andersonite, calcite, gypsum and natromarkeyite. Paramarkeyite crystals are transparent, pale green-yellow, striated tablets, up to 0.11 mm across. The mineral has white streak and vitreous lustre. It exhibits moderate bluish-white fluorescence (405 nm laser). It is very brittle with irregular, curved fracture and a Mohs hardness of 2½. It has an excellent {100} cleavage and probably two good cleavages on {010} and {001}. The measured density is 2.91(2) g cm–3. Optically, the mineral is biaxial (–) with α = 1.550(2), β = 1.556(2), γ = 1.558(2) (white light); 2V = 60(2)°; strong r > v dispersion; orientation: Y = b; nonpleochroic. The Raman spectrum exhibits bands consistent with UO22+, CO32– and O–H. Electron microprobe analysis provided the empirical formula (Ca1.83Na0.20Sr0.03)Σ2.05(UO2)(CO3)3⋅5H2O (+0.07 H). Paramarkeyite is monoclinic, P21/n, a = 17.9507(7), b = 18.1030(8), c = 18.3688(13) Å, β = 108.029(8)°, V = 5676.1(6) Å3 and Z = 16. The structure of paramarkeyite (R1 = 0.0647 for 6657 I > 2σI) contains uranyl tricarbonate clusters that are linked by Ca–O polyhedra to form heteropolyhedral layers. The structure of paramarkeyite is very similar to those of markeyite, natromarkeyite and pseudomarkeyite.
Klajite, MnCu4(AsO4)2(AsO3OH)2(H2O)10, from Jáchymov (Czech Republic): the second world occurrence
- J. Plášil, A. V. Kasatkin, R. Škoda, P. Škácha
-
- Journal:
- Mineralogical Magazine / Volume 78 / Issue 1 / February 2014
- Published online by Cambridge University Press:
- 05 July 2018, pp. 119-129
-
- Article
- Export citation
-
Klajite, MnCu4(AsO4)2(AsO3OH)2(H2O)10, the Mn-Cu-bearing member of the lindackerite group, was found in Jáchymov, Czech Republic, as the second world occurrence. It is associated with ondrušite and other arsenate minerals growing on the quartz gangue with disseminated primary sulfides, namely tennantite and chalcopyrite. Electron-microprobe data showed klajite aggregates to be chemically inhomogeneous at larger scales, varying from Mn-Ca-rich to Cu-rich domains. The chemical composition of the the Mn-rich parts of aggregates can be expressed by the empirical formula (Mn0.46Ca0.22Cu0.07Mg0.02)∑0.77(Cu3.82Mg0.14Ca0.03Zn0.01)∑4.00(As1.94Si0.06)∑2.00O8[AsO2.73(OH)1.27]2(H2O)10 (mean of seven representative spots; calculated on the basis of As + Si + P = 4 a.p.f.u. (atoms per formula unit) and 10 H2O from ideal stoichiometry), showing a slight cationic deficiency at the key Me-site. According to single-crystal X-ray diffraction, klajite from Jáchymov is triclinic, P
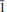 , with a = 6.4298(8), b = 7.9716(8), c = 10.707(2) Å, α = 85.737(12)°, β = 80.994(13)°, γ = 84.982(10)°, and V = 538.85(14) Å3, Z = 1. The crystal structure was refined to R1 = 0.0628 for 1034 unique observed reflections (with Iobs > 3σ(I)), confirming that klajite (Mn-Cu member) and ondrušite (Ca-Cu member) are isostructural. The current data-set allowed determination of the positions of several hydrogen atoms. Discussion on hydrogen bonding networks in the structure of klajite as well as detailed bond-valence analysis are provided.
, with a = 6.4298(8), b = 7.9716(8), c = 10.707(2) Å, α = 85.737(12)°, β = 80.994(13)°, γ = 84.982(10)°, and V = 538.85(14) Å3, Z = 1. The crystal structure was refined to R1 = 0.0628 for 1034 unique observed reflections (with Iobs > 3σ(I)), confirming that klajite (Mn-Cu member) and ondrušite (Ca-Cu member) are isostructural. The current data-set allowed determination of the positions of several hydrogen atoms. Discussion on hydrogen bonding networks in the structure of klajite as well as detailed bond-valence analysis are provided.
Crystal structure and formula revision of deliensite, Fe[(UO2)2(SO4)2(OH)2](H2O)7
- J. Plášil, J. Hauser, V. Petříček, N. Meisser, S. J. Mills, R. Škoda, K. Fejfarová, J. Čejka, J. Sejkora, J. Hloušek, J.-M. Johannet, V. Machovič, L. Lapčák
-
- Journal:
- Mineralogical Magazine / Volume 76 / Issue 7 / December 2012
- Published online by Cambridge University Press:
- 05 July 2018, pp. 2837-2860
-
- Article
- Export citation
-
The crystal structure of deliensite, Fe[(UO2)2(SO4)2(OH)2](H2O)7, was solved by direct methods and refined to R1 = 6.24% for 5211 unique observed reflections [Iobs > 3σ(I)], on a crystal that was found to consist of rotational and inversion (merohedral) twins, from Jeroným mine, Abertamy in the Czech Republic. The presence of four twin domains was taken into account in the refinement. The structure is orthorhombic, space group Pnn2, with unit-cell parameters a = 15.8514(9), b = 16.2478(7), c = 6.8943(3) Å, V = 1775.6(1) Å3 and Z = 4. The crystal structure of deliensite contains uranyl-sulfate sheets with a phosphuranylite topology, consisting of dimers of edge-sharing uranyl pentagonal bipyramids linked by corner-sharing with sulfate tetrahedra. The sheets lie in the (100) plane and are decorated by [Fe2+O(H2O)5] octahedra; two weakly bonded H2O molecules are present in the interlayer. The [Fe2+O(H2O)5] octahedron is linked directly to the sheet via the uranyl oxygen atom. Adjacent sheets are linked by hydrogen bonds only. The sheet topology and geometrical isomerism is discussed and a comparison of the composition obtained from electron-probe microanalysis, powder-diffraction data, Raman and infrared spectra of deliensite samples from Mas d'Alary, Lodève, France; L'Ecarpière mine, Gétigné, France; and several localities at Jáchymov, Western Bohemia, Czech Republic is made.
Štěpite, U(AsO3OH)2·4H2O, from Jáchymov, Czech Republic: the first natural arsenate of tetravalent uranium
- J. Plášil, K. Fejfarová, J. Hloušek, R. Škoda, M. Novák, J. Sejkora, J. Čejka, M. Dušek, F. Veselovský, P. Ondruš, J. Majzlan, Z. Mrázek
-
- Journal:
- Mineralogical Magazine / Volume 77 / Issue 1 / February 2013
- Published online by Cambridge University Press:
- 05 July 2018, pp. 137-152
-
- Article
- Export citation
-
Štěpite, tetragonal U(AsO3 OH)2(H2O)4 (IMA 2012-006), is the first natural arsenate of tetravalent uranium. It occurs in the Geschieber vein, Jáchymov ore district, Western Bohemia, Czech Republic, as emerald-green crystalline crusts on altered arsenic. Associated minerals include arsenolite, běhounekite, claudetite, gypsum, kaatialaite, the new mineral vysokýite (IMA 2012-067) and a partially characterized phase with the formula (H3O)+2(UO2)2(AsO4)2˙6H2O. Štěpite typically forms tabular crystals with prominent {001} and {010} faces, up to 0.6 mm in size. The crystals have a vitreous lustre and a grey to greenish grey streak. They are brittle with an uneven fracture and a very good cleavage on (001). Their Mohs hardness is about 2. Štěpite is not fluorescent in either short-wave or long-wave ultraviolet light. It is biaxial (–) with refractive indices (at 590 nm) of α = 1.636(2), β = 1.667(3), γ = 1.672(2) and 2Vobs < ~5°, anomalous greyish to pale yellow interference colours, and no pleochroism. The composition is as follows: 0.12Na2O, 50.19 UO2, 0.04SiO4, 0.09 P2O5, 0.93 As2O5, 1.95 SO3, 16.41 H2O; total 107.90 wt.%, yielding an empirical formula (based on 12 O a. p. f. u.) of (U1.01Na0.02)Σ1.03[(AsO3OH)1.82 (PO3OH)0.04(SO4)0.13(SiO4)0.01]Σ 2.00˙4H2O. Štěpite is tetragonal, crystallizing in space group I41/acd, with a = 10.9894(1), c = 32.9109(6) Å, V = 3974.5(1) Å3, Z = 16 and Dcalc = 3.90 g cm-3. The six strongest peaks in the X-ray powder-diffraction pattern [dobs in Å (I) (hkl)] are as follows: 8.190(100)(004), 7.008(43)(112), 5.475(18)(200), 4.111(16)(008), 3.395(20)(312,217), 2.1543(25)(419). The crystal structure of šteěpite has been solved from singlecrystal X-ray diffraction data by the charge-flipping method and refined to R1 = 0.0353 based on 1434 unique observed reflections, and to wR2 = 0.1488 for all 1523 unique reflections. The crystal structure of štšpite consists of sheets perpendicular to [001], made up of eight-coordinate uranium atoms and hydroxyarsenate 'tetrahedra'. The ligands surrounding the uranium atom consist of six oxygen atoms which belong to the hydroxyarsenate groups and two oxygen atoms which belong to interlayer H2 O molecules. Each UO8 polyhedron is connected to five other U polyhedra via six AsO3OH groups. Adjacent electroneutral sheets, of composition [U4+(AsO3OH)22-]0, are linked by hydrogen bonds involving H2 O molecules in the interlayers and OH groups in the sheets. The new mineral is named in honour of Josef Štěp (1863–1926), a Czech mining engineer and 'father' of the world's first radioactive spa at Jáchymov.
Švenekite, Ca[AsO2(OH)2]2, a new mineral from Jáchymov, Czech Republic
- P. Ondruš, R. Skála, J. Plášil, J. Sejkora, F. Veselovský, J. Čejka, A. Kallistová, J. Hloušek, K. Fejfarová, R. Škoda, M. Dušek, A. Gabašová, V. Machovič, L. Lapčák
-
- Journal:
- Mineralogical Magazine / Volume 77 / Issue 6 / August 2013
- Published online by Cambridge University Press:
- 05 July 2018, pp. 2711-2724
-
- Article
- Export citation
-
Švenekite (IMA 99-007), Ca[AsO2(OH)2]2, is a rare supergene arsenate mineral occurring in the Geschieber vein, Jáchymov ore district, Western Bohemia, Czech Republic. It grows directly on the granite rocks and occurs isolated from other arsenate minerals otherwise common in Jáchymov. Švenekite usually forms clear transparent coatings composed of indistinct radiating to rosette-shaped aggregates up to 3 mm across. They are composed of thin lens- or bladed-shaped crystals, usually 100 – 150 μm long. Švenekite is transparent to translucent and has a white streak and a vitreous lustre; it does not fluoresce under ultraviolet light. Cleavage is very good on {010}. The Mohs hardness is ∼2. Švenekite is biaxial, non-pleochroic. The refractive indices are α' = 1.602(2), γ' = 1.658(2). The empirical formula of švenekite (based on As + P + S = 2 a.p.f.u., an average of 10 spot analyses) is (Ca1.00Mg0.01)Σ1.01[AsO2(OH)2]1.96[PO2(OH)2]0.03(SO4)0.01. The simplified formula is Ca[AsO2(OH)2]2 and requires CaO 17.42, As2O571.39, H2O 11.19, total 100.00 wt.%. Raman and infrared spectroscopy exhibit dominance of O – H vibrations and vibration modes of distorted tetrahedral AsO2(OH)2 units. Švenekite is triclinic, space group P
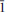 , with a = 8.5606(5), b = 7.6926(6), c = 5.7206(4) Å, α = 92.605(6), β = 109.9002(6), γ = 109.9017(6)º, and V = 327.48(4) Å3, Z = 2, Dcalc = 3.26 g·cm–3. The a:b:c ratio is 0.7436:1:1.1082 (for single-crystal data). The six strongest diffraction peaks in the X-ray powder diffraction pattern are [d (Å)/I(%)/(hkl)]: 3.968(33)(2
, with a = 8.5606(5), b = 7.6926(6), c = 5.7206(4) Å, α = 92.605(6), β = 109.9002(6), γ = 109.9017(6)º, and V = 327.48(4) Å3, Z = 2, Dcalc = 3.26 g·cm–3. The a:b:c ratio is 0.7436:1:1.1082 (for single-crystal data). The six strongest diffraction peaks in the X-ray powder diffraction pattern are [d (Å)/I(%)/(hkl)]: 3.968(33)(2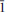 0); 3.766(35)(2
0); 3.766(35)(2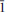
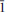 ); 3.697(49)(101); 3.554(100)(020); 3.259(33)(2
); 3.697(49)(101); 3.554(100)(020); 3.259(33)(2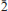 0); 3.097(49)(1
0); 3.097(49)(1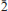 1). The crystal structure of švenekite was refined from single-crystal X-ray diffraction data to R1 = 0.0250 based on 1309 unique observed, and to wR2 = 0.0588, for all 1588 unique reflections (with GOFall = 1.20). The structure of švenekite consists of sheets of corner-sharing CaO8 polyhedra and AsO2 OH2 groups, stacked parallel to (001). Adjacent sheets are linked by hydrogen bonds. The švenekite structure possesses very short symmetrical hydrogen bonds (with the D–H lengths ∼1.22 Å). The mineral is named to honour Jaroslav Švenek, the former curator of the mineralogical collection of the National Museum in Prague, Czech Republic.
1). The crystal structure of švenekite was refined from single-crystal X-ray diffraction data to R1 = 0.0250 based on 1309 unique observed, and to wR2 = 0.0588, for all 1588 unique reflections (with GOFall = 1.20). The structure of švenekite consists of sheets of corner-sharing CaO8 polyhedra and AsO2 OH2 groups, stacked parallel to (001). Adjacent sheets are linked by hydrogen bonds. The švenekite structure possesses very short symmetrical hydrogen bonds (with the D–H lengths ∼1.22 Å). The mineral is named to honour Jaroslav Švenek, the former curator of the mineralogical collection of the National Museum in Prague, Czech Republic.
Hydroniumjarosite, (H3O)+Fe3(SO4)2(OH)6, from Cerros Pintados, Chile: Single-crystal X-ray diffraction and vibrational spectroscopic study
- J. Plášil, R. Škoda, K. Fejfarová, J. Čejka, A. V. Kasatkin, M. Dušek, D. Talla, L. Lapčák, V. Machovič, M. Dini
-
- Journal:
- Mineralogical Magazine / Volume 78 / Issue 3 / June 2014
- Published online by Cambridge University Press:
- 05 July 2018, pp. 535-547
-
- Article
- Export citation
-
The natural hydroniumjarosite sample from Cerros Pintados (Chile) was investigated by electron microprobe, single-crystal X-ray diffraction and vibrational spectroscopy (Infrared and Raman). The chemical composition of studied specimens (wt.%, mean of seven analyses) obtained from electron microprobe (in wt.%): Na2O 1.30, K2O 0.23, CaO 0.04, Fe2O3 50.49, Al2O3 0.37, SiO2 0.33, SO3 33.88, H2O (calculated on the basis of Σ(OH–+H3O+) deduced from the charge balance) 13.32, total 99.98, corresponds to the empirical formula (H3O)0.77+(Na0.20K0.02)∑0.22(Fe2.95Al0.03)∑2.98 (OH)6.12[(SO4)1.97(SiO4)0.03]∑2.00 (calculated on the basis of S + Si = 2 a.p.f.u. (atoms per formula unit)). The studied hydroniumjarosite is trigonal, with space group R
 m, with a = 7.3408(2), c = 17.0451(6) Å and V = 795.46(4) Å3. The refined structure architecture is consistent with known jarosite-series minerals, including synthetic hydroniumjarosite. However, in the current study the presence of H3O+ is well documented in difference Fourier maps, where characteristic positive difference Fourier maxima, with apparent trigonal symmetry, were localized in the vicinity of the O4 atom in the channel-voids of the structure. The structure of natural hydroniumjarosite, including the H atoms, was refined to R1 = 0.0166 for 2113 unique observed reflections, with Iobs>3σ(I). The present structure model, which includes the position of the H atom within the hydronium ion, is discussed with regard to the vibration spectroscopy results and earlier published density-functional theory (DFT) calculations for the alunite-like structure containing H3O+.
m, with a = 7.3408(2), c = 17.0451(6) Å and V = 795.46(4) Å3. The refined structure architecture is consistent with known jarosite-series minerals, including synthetic hydroniumjarosite. However, in the current study the presence of H3O+ is well documented in difference Fourier maps, where characteristic positive difference Fourier maxima, with apparent trigonal symmetry, were localized in the vicinity of the O4 atom in the channel-voids of the structure. The structure of natural hydroniumjarosite, including the H atoms, was refined to R1 = 0.0166 for 2113 unique observed reflections, with Iobs>3σ(I). The present structure model, which includes the position of the H atom within the hydronium ion, is discussed with regard to the vibration spectroscopy results and earlier published density-functional theory (DFT) calculations for the alunite-like structure containing H3O+.
Bêhounekite, U(SO4)2(H2O)4, from Jáchymov (St Joachimsthal), Czech Republic: the first natural U4+ sulphate
- J. Plášil, K. Fejfarová, M. Novák, M. Dušek, R. Škoda, J. Hloušek, J. Čejka, J. Majzlan, J. Sejkora, V. Machovic, D. Talla
-
- Journal:
- Mineralogical Magazine / Volume 75 / Issue 6 / December 2011
- Published online by Cambridge University Press:
- 05 July 2018, pp. 2739-2753
-
- Article
- Export citation
-
Bêhounekite, orthorhombic U(SO4)2(H2O)4, is the first natural sulphate of U4+. It was found in the Geschieber vein, Jáchymov (St Joachimsthal) ore district, Western Bohemia, Czech Republic, crystallized on the altered surface of arsenic and associated with kaatialaite, arsenolite, claudetite, unnamed phase UM1997-20-AsO:HU and gypsum. Bêhounekite most commonly forms short-prismatic to tabular green crystals, rarely up to 0.5 mm long. The crystals have a strong vitreous lustre and a grey to greenish grey streak. They are brittle with an uneven fracture and have very good cleavage along ﹛100﹜. The Mohs hardness is about 2. The mineral is not fluorescent either in short- or long-wavelength UV radiation. Bêhounekite is moderately pleochroic, α∼β is pale emerald green and γ is emerald green, and is optically biaxial (+) with α = 1.590(2), β = 1.618(4), γ = 1.659(2) (590 nm), 2V (calc.) = 81°, birefringence 0.069. The empirical formula of bêhounekite (based on 12 O atoms, from an average of five point analyses) is (U0.99Y0.03)Σ1.02(SO4)1.97(H2O)4. The simplified formula is U(SO4)2(H2O)4, which requires UO2 53.77. SO3 31.88, H2O 14.35, total 100.00 wt.%. Bêhounekite is orthorhombic, space group Pnma, a = 14.6464(3), b = 11.0786(3), c = 5.6910(14) Å, V = 923.43(4) Å3, Z = 4, Dcalc = 3.62 g cm–3. The seven strongest diffraction peaks in the X-ray powder diffraction pattern are [dobs in Å (I) (hid)]: 7.330 (100) (200), 6.112 (54) (210), 5.538 (21) (020), 4.787 (42) (111), 3.663 (17) (400), 3.478 (20) (410), 3.080 (41) (321). The crystal structure of bêhounekite has been solved by the charge-flipping method from single-crystal X-ray diffraction data and refined to R1 = 2.10 % with a GOF = 1.51, based on 912 unique observed diffractions. The crystal structure consists of layers built up from [8]-coordinate uranium atoms and sulphate tetrahedra. The eight ligands include four oxygen atoms from the sulphate groups and four oxygen atoms from the H2O molecules. Each uranium coordination polyhedron is connected via sulphate tetrahedra with other uranium polyhedra and through hydrogen bonds to the apices of sulphate tetrahedra. The dominant features of the Raman and infrared spectra of bêhounekite are related to stretching vibrations of SO4 tetrahedra (∼1200–950 cm–1), O-H stretching modes (∼3400–3000 cm–1) and H—O—H bending modes (∼1650 cm–1). The mineral is named in honour of František Bêhounek, a well known Czech nuclear physicist.
Leydetite, Fe(UO2)(SO4)2(H2O)11, a new uranyl sulfate mineral from Mas d’Alary, Lodève, France
- J. Plášil, A. V. Kasatkin, R. Škoda, M. Novák, A. Kallistová, M. Dušek, R. Skála, K. Fejfarová, J. Čejka, N. Meisser, H. Goethals, V. Machovič, L. Lapčák
-
- Journal:
- Mineralogical Magazine / Volume 77 / Issue 4 / June 2013
- Published online by Cambridge University Press:
- 05 July 2018, pp. 429-441
-
- Article
- Export citation
-
Leydetite, monoclinic Fe(UO2)(SO4)2(H2O)11(IMA 2012–065), is a new supergene uranyl sulfate from Mas d'Alary, Lodève, Hérault, France. It forms yellow to greenish, tabular, transparent to translucent crystals up to 2 mm in size. Crystals have a vitreous lustre. Leydetite has a perfect cleavage on (001). The streak is yellowish white. Mohs hardness is ∼2. The mineral does not fluoresce under long- or short-wavelength UV radiation. Leydetite is colourless in transmitted light, non-pleochroic, biaxial, with α = 1.513(2), γ = 1.522(2) (further optical properties could not be measured). The measured chemical composition of leydetite, FeO 9.28, MgO 0.37, Al2O30.26, CuO 0.14, UO340.19, SO321.91, SiO20.18, H2O 27.67, total 100 wt.%, leads to the empirical formula (based on 21 O a.p.f.u.), (Fe0.93Mg0.07Al0.04Cu0.01)Σ1.05(U1.01O2)(S1.96Si0.02)Σ1.98O8(H2O)11. Leydetite is monoclinic, space group C2/c, with a = 11.3203(3), b = 7.7293(2), c = 21.8145(8) Å, β = 102.402(3)°, V = 1864.18(10) Å3, Z = 4, and Dcalc = 2.55 g cm–3. The six strongest reflections in the X-ray powder diffraction pattern are [dobs in Å (I) (hkl)]: 10.625 (100) (002), 6.277 (1) (
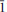 11), 5.321 (66) (004), 3.549 (5) (006), 2.663 (4) (008), 2.131 (2) (0 0 10). The crystal structure has been refined from single-crystal X-ray diffraction data to R1 = 0.0224 for 5211 observed reflections with [I > 3σ(I)]. Leydetite possesses a sheet structure based upon the protasite anion topology. The sheet consists of UO7 bipyramids, which share four of their equatorial vertices with SO4 tetrahedra. Each SO4 tetrahedron, in turn, shares two of its vertices with UO7 bipyramids. The remaining unshared equatorial vertex of the bipyramid is occupied by H2O, which extends hydrogen bonds within the sheet to one of a free vertex of the SO4 tetrahedron. Sheets are stacked perpendicular to the c direction. In the interlayer, Fe2+ ions and H2O groups link to the sheets on either side via a network of hydrogen bonds. Leydetite is isostructural with the synthetic compound Mg(UO2)(SO4)2(H2O)11. The name of the new mineral honours Jean Claude Leydet (born 1961), an amateur mineralogist from Brest (France), who discovered the new mineral.
11), 5.321 (66) (004), 3.549 (5) (006), 2.663 (4) (008), 2.131 (2) (0 0 10). The crystal structure has been refined from single-crystal X-ray diffraction data to R1 = 0.0224 for 5211 observed reflections with [I > 3σ(I)]. Leydetite possesses a sheet structure based upon the protasite anion topology. The sheet consists of UO7 bipyramids, which share four of their equatorial vertices with SO4 tetrahedra. Each SO4 tetrahedron, in turn, shares two of its vertices with UO7 bipyramids. The remaining unshared equatorial vertex of the bipyramid is occupied by H2O, which extends hydrogen bonds within the sheet to one of a free vertex of the SO4 tetrahedron. Sheets are stacked perpendicular to the c direction. In the interlayer, Fe2+ ions and H2O groups link to the sheets on either side via a network of hydrogen bonds. Leydetite is isostructural with the synthetic compound Mg(UO2)(SO4)2(H2O)11. The name of the new mineral honours Jean Claude Leydet (born 1961), an amateur mineralogist from Brest (France), who discovered the new mineral.
Meisserite, Na5(UO2)(SO4)3(SO3OH)(H2O), a new uranyl sulfate mineral from the Blue Lizard mine, San Juan County, Utah, USA
- J. Plášil, A. R. Kampf, A. V. Kasatkin, J. Marty, R. Škoda, S. Silva, J. Čejka
-
- Journal:
- Mineralogical Magazine / Volume 77 / Issue 7 / October 2013
- Published online by Cambridge University Press:
- 05 July 2018, pp. 2975-2988
-
- Article
- Export citation
-
Meisserite (IMA2013-039), Na5(UO2)(SO4)3(SO3OH)(H2O), is a new uranyl sulfate mineral from the Blue Lizard mine, San Juan County, Utah (USA). It is named in honour of the prominent Swiss mineralogist Nicolas Meisser. The new mineral was found in a sandstone matrix and is associated with chalcanthite, copiapite, ferrinatrite, gypsum, johannite and another new Na-bearing uranyl sulfate, belakovskiite (IMA2013-075). Meisserite is a secondary mineral formed by the post-mining weathering of uraninite. The mineral is triclinic, P
 , a = 5.32317(10), b = 11.5105(2), c = 13.5562(10) Å, α = 102.864(7)°, β = 97.414(7)°, γ = 91.461(6)°, V = 801.74(6) Å3, and Z = 2. Crystals are prisms elongated on [100], up to 0.3 mm long, exhibiting the forms {010} and {001}. Meisserite is pale green to yellowish green, translucent to transparent and has a very pale yellow streak. It is brittle, with fair cleavage on {100} and {001}, and uneven fracture. The Mohs hardness is estimated at 2. Meisserite is somewhat hygroscopic and easily soluble in water. The calculated density based on the empirical formula is 3.208 g/cm3. Meisserite exhibits bright yellow green fluorescence under both long- and shortwave UV radiation. The mineral is optically biaxial (–), with α = 1.514(1), β = 1.546(1), γ = 1.557(1) (measured in white light). The measured 2V is 60(2)° and the calculated 2V is 60°. Dispersion is r > v, perceptible, and the optical orientation is X ≈ a, Z ≈ c*. The mineral is pleochroic, with X (colourless) < Y (pale yellow) ≈ Z (pale greenish yellow). The empirical formula of meisserite (based on 19 O a.p.f.u.) is Na5.05(U0.94O2)(SO4)3[SO2.69(OH)1.31](H2O). The Raman spectrum is dominated by the symmetric stretching vibrations of UO22+, SO42– and also weaker O–H stretching vibrations. The eight strongest powder X-ray diffraction lines are [dobs in Å (hkl)Irel]: 13.15 (001) 81, 6.33 (0
, a = 5.32317(10), b = 11.5105(2), c = 13.5562(10) Å, α = 102.864(7)°, β = 97.414(7)°, γ = 91.461(6)°, V = 801.74(6) Å3, and Z = 2. Crystals are prisms elongated on [100], up to 0.3 mm long, exhibiting the forms {010} and {001}. Meisserite is pale green to yellowish green, translucent to transparent and has a very pale yellow streak. It is brittle, with fair cleavage on {100} and {001}, and uneven fracture. The Mohs hardness is estimated at 2. Meisserite is somewhat hygroscopic and easily soluble in water. The calculated density based on the empirical formula is 3.208 g/cm3. Meisserite exhibits bright yellow green fluorescence under both long- and shortwave UV radiation. The mineral is optically biaxial (–), with α = 1.514(1), β = 1.546(1), γ = 1.557(1) (measured in white light). The measured 2V is 60(2)° and the calculated 2V is 60°. Dispersion is r > v, perceptible, and the optical orientation is X ≈ a, Z ≈ c*. The mineral is pleochroic, with X (colourless) < Y (pale yellow) ≈ Z (pale greenish yellow). The empirical formula of meisserite (based on 19 O a.p.f.u.) is Na5.05(U0.94O2)(SO4)3[SO2.69(OH)1.31](H2O). The Raman spectrum is dominated by the symmetric stretching vibrations of UO22+, SO42– and also weaker O–H stretching vibrations. The eight strongest powder X-ray diffraction lines are [dobs in Å (hkl)Irel]: 13.15 (001) 81, 6.33 (0 2) 62, 5.64 (0
2) 62, 5.64 (0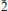 1,020) 52, 5.24 (100,012,
1,020) 52, 5.24 (100,012, 01) 100, 4.67 (101) 68, 3.849 (
01) 100, 4.67 (101) 68, 3.849 (
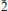 1,102,022) 48, 3.614 (0
1,102,022) 48, 3.614 (0 2,
2,
 3) 41, and 3.293 (
3) 41, and 3.293 ( 13,004) 43. The crystal structure of meisserite (R1 = 0.018 for 3306 reflections with Iobs > 3σI) is topologically unique among known structures of uranyl minerals and inorganic compounds. It contains uranyl pentagonal bipyramids linked by SO4 groups to form chains. Na+ cations bond to O atoms in the chains and to an SO3OH group and an H2>O group between the chains, thereby forming a heteropolyhedral framework.
13,004) 43. The crystal structure of meisserite (R1 = 0.018 for 3306 reflections with Iobs > 3σI) is topologically unique among known structures of uranyl minerals and inorganic compounds. It contains uranyl pentagonal bipyramids linked by SO4 groups to form chains. Na+ cations bond to O atoms in the chains and to an SO3OH group and an H2>O group between the chains, thereby forming a heteropolyhedral framework.
The crystal chemistry of the uranyl carbonate mineral grimselite, (K, Na)3Na[(UO2)(CO3)3](H2O), from Jáchymov, Czech Republic
- J. Plášil, K. Fejfarová, R. Skála, R. Škoda, N. Meisser, J. Hlousek, I. Císařová, M. Dušek, F. Veselovský, J. Čejka, J. Sejkora, P. Ondruš
-
- Journal:
- Mineralogical Magazine / Volume 76 / Issue 3 / June 2012
- Published online by Cambridge University Press:
- 05 July 2018, pp. 443-453
-
- Article
- Export citation
-
Two crystals of the uranyl carbonate mineral grimselite, ideally K3Na[(UO2)(CO3)3](H2O), from Jáchymov in the Czech Republic were studied by single-crystal X-ray diffraction and electron-probe microanalysis. One crystal has considerably more Na than the ideal chemical composition due to substitution of Na into KO8 polyhedra; the composition of the other crystal is nearer to ideal, and similar to synthetic grimselite. The presence of Na atoms in KO8 polyhedra, which are located in channels in the crystal structure, reduces their volume, and as a result the unit-cell volume also decreases. Structure refinement shows that the formula for the sample with the anomalously high Na content is (K2.43Na0.57)Σ3.00Na[(UO2)(CO3)3](H2O). The unit-cell parameters, refined in space group P
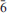 2c, are a = 9.2507(1), c = 8.1788(1) Å, V = 606.14(3) Å3 and Z = 2. The crystal structure was refined to R1 = 0.0082 and wR1 = 0.0185 with a GOF = 1.33, based on 626 observed diffraction peaks [Iobs>3σ(I)].
2c, are a = 9.2507(1), c = 8.1788(1) Å, V = 606.14(3) Å3 and Z = 2. The crystal structure was refined to R1 = 0.0082 and wR1 = 0.0185 with a GOF = 1.33, based on 626 observed diffraction peaks [Iobs>3σ(I)].
Calciodelrioite, Ca(VO3)2(H2O)4, the Ca analogue of delrioite, Sr(VO3)2(H2O)4
- A. R. Kampf, J. Marty, B. P. Nash, J. Plášil, A. V. Kasatkin, R. Škoda
-
- Journal:
- Mineralogical Magazine / Volume 76 / Issue 7 / December 2012
- Published online by Cambridge University Press:
- 05 July 2018, pp. 2803-2817
-
- Article
- Export citation
-
Calciodelrioite, ideally Ca(VO3)2(H2O)4, is a new mineral (IMA 2012-031) from the uraniumvanadium deposits of the eastern Colorado Plateau in the USA. The type locality is the West Sunday mine, Slick Rock district, San Miguel County, Colorado. The new mineral occurs on fracture surfaces in corvusite- and montroseite-impregnated sandstone and forms as a result of the oxidative alteration of these phases. At the West Sunday mine, calciodelrioite is associated with celestine, gypsum, huemulite, metarossite, pascoite and rossite. The mineral occurs as transparent colourless needles, bundles of tan to brown needles and star bursts of nearly black broad blades composed of tightly intergrown needles. Crystals are elongate and striated parallel to [100], exhibiting the prismatic forms {001} and {011} and having terminations possibly composed of the forms {100} and {61
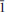 }. The mineral is transparent and has a white streak, subadamantine lustre, Mohs hardness of about 2½, brittle tenacity, irregular to splintery fracture, one perfect cleavage on {001} and possibly one or more additional cleavages parallel to [100]. Calciodelrioite is soluble in water. The calculated density is 2.451 g cm– 3. It is optically biaxial (+) with α = 1.733(3), β = 1.775(3), γ = 1.825(3) (white light), 2Vmeas = 87.3(9)° and 2Vcalc = 87°. The optical orientation is X = b; Z ≈ a. No pleochroism was observed. Electronmicroprobe analyses of two calciodelrioite samples and type delrioite provided the empirical formulae (Ca0.88Sr0.07Na0.04K0.01)Σ1.00(V1.00O3)2(H2.01O)4, (Ca0.76Sr0.21Na0.01)Σ0.98(V1.00O3)2(H2.01O)4 and (Sr0.67Ca0.32)Σ0.99(V1.00O3)2(H2.00O)4, respectively. Calciodelrioite is monoclinic, I2/a, with unit-cell parameters a = 14.6389(10), b = 6.9591(4), c = 17.052(2) Å, β = 102.568(9)°, V = 1695.5(3) Å3 and Z = 8. The seven strongest lines in the X-ray powder diffraction pattern [listed as dobs Å (I)(hkl)] are as follows: 6.450(100)(011); 4.350(16)(013); 3.489(18)(020); 3.215(17)(022); 3.027(50)(multiple); 2.560(28)(
}. The mineral is transparent and has a white streak, subadamantine lustre, Mohs hardness of about 2½, brittle tenacity, irregular to splintery fracture, one perfect cleavage on {001} and possibly one or more additional cleavages parallel to [100]. Calciodelrioite is soluble in water. The calculated density is 2.451 g cm– 3. It is optically biaxial (+) with α = 1.733(3), β = 1.775(3), γ = 1.825(3) (white light), 2Vmeas = 87.3(9)° and 2Vcalc = 87°. The optical orientation is X = b; Z ≈ a. No pleochroism was observed. Electronmicroprobe analyses of two calciodelrioite samples and type delrioite provided the empirical formulae (Ca0.88Sr0.07Na0.04K0.01)Σ1.00(V1.00O3)2(H2.01O)4, (Ca0.76Sr0.21Na0.01)Σ0.98(V1.00O3)2(H2.01O)4 and (Sr0.67Ca0.32)Σ0.99(V1.00O3)2(H2.00O)4, respectively. Calciodelrioite is monoclinic, I2/a, with unit-cell parameters a = 14.6389(10), b = 6.9591(4), c = 17.052(2) Å, β = 102.568(9)°, V = 1695.5(3) Å3 and Z = 8. The seven strongest lines in the X-ray powder diffraction pattern [listed as dobs Å (I)(hkl)] are as follows: 6.450(100)(011); 4.350(16)(013); 3.489(18)(020); 3.215(17)(022); 3.027(50)(multiple); 2.560(28)(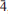 15,413); 1.786(18)(028). In the structure of calciodelrioite (refined to R1 = 3.14% for 1216 Fo > 4σF), V5+O5 polyhedra link by sharing edges to form a zigzag divanadate [VO3] chain along a, similar to that in the structure of rossite. The chains are linked via bonds to Ca atoms, which also bond to H2O groups, yielding CaO3(H2O)6 polyhedra. The Ca polyhedra form a chain along b. Each of the two symmetrically independent VO5 polyhedra has two short vanadyl bonds and three long equatorial bonds. Calciodelrioite and delrioite are isostructural and are the endmembers of the series Ca(VO3)2(H2O)4–Sr(VO3)2(H2O)4. Calciodelrioite is dimorphous with rossite, which has a similar structure; however, the smaller 8-coordinate Ca site in rossite does not accommodate Sr.
15,413); 1.786(18)(028). In the structure of calciodelrioite (refined to R1 = 3.14% for 1216 Fo > 4σF), V5+O5 polyhedra link by sharing edges to form a zigzag divanadate [VO3] chain along a, similar to that in the structure of rossite. The chains are linked via bonds to Ca atoms, which also bond to H2O groups, yielding CaO3(H2O)6 polyhedra. The Ca polyhedra form a chain along b. Each of the two symmetrically independent VO5 polyhedra has two short vanadyl bonds and three long equatorial bonds. Calciodelrioite and delrioite are isostructural and are the endmembers of the series Ca(VO3)2(H2O)4–Sr(VO3)2(H2O)4. Calciodelrioite is dimorphous with rossite, which has a similar structure; however, the smaller 8-coordinate Ca site in rossite does not accommodate Sr.
Vysokýite, U4+[AsO2(OH)2]4·4H2O, a new mineral from Jáchymov, Czech Republic
- J. Plášil, J. Hloušek, R. Škoda, M. Novák, J. Sejkora, J. Čejka, F. Veselovský, J. Majzlan
-
- Journal:
- Mineralogical Magazine / Volume 77 / Issue 8 / December 2013
- Published online by Cambridge University Press:
- 05 July 2018, pp. 3055-3066
-
- Article
- Export citation
-
Vysokýite, U4+[(AsO2(OH)2]4(H2O)4 (IMA 2012–067), was found growing on an altered surface of massive native As in the Geschieber vein, Jáchymov ore district, Western Bohemia, Czech Republic. The new mineral was found in association with běhounekite, štěpite, kaatialaite, arsenolite, claudetite and gypsum. It forms extremely fibrous light-green crystals up to 8 mm long. Crystals have an alabaster lustre and a greenish-white to greyish streak. Vysokýite is brittle with uneven fracture and perfect cleavage along (100) and (001); the Mohs hardness is ∼2. A density of 3.393 g/cm3 was calculated using the empirical formula and unit-cell parameters obtained from a single-crystal diffraction experiment. Vysokýite is non-fluorescent under short or long wavelength UV radiation. It is colourless under the microscope, measured refractive indices are α' = 1.617(3), γ' = 1.654(3); the estimated optical orientation is α' ∼X, γ' ∼Z. The average of five spot wavelength dispersive spectroscopy (WDS) analyses is 29.44 UO2, 1.03 SiO2, 48.95 As2O5, 0.12 SO3, 15.88 H2O (calc.), total 95.42 wt.%. The empirical formula of vysokýite (based on 20 O a.p.f.u.) is U1.00[AsO2(OH)2]3.90(SiO4)0.16 (SO4)0.01·4H2O. The As–O–H and O–H vibrations dominate in the Raman spectrum. Vysokýite is triclinic, space group P
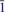 , with a = 10.749(2), b = 5.044(3), c = 19.1778(7) Å, α = 89.872(15)°, β = 121.534(15)°, γ = 76.508(15)°, and V = 852.1(6) Å3, Z = 2 and Dcalc = 3.34 g·cm–3. The strongest diffraction peaks in the X-ray powder diffraction pattern are [dobs in Å (Irel.)(hkl)]: 8.872(100)(100), 8.067(50)(002), 6.399(7)(10
, with a = 10.749(2), b = 5.044(3), c = 19.1778(7) Å, α = 89.872(15)°, β = 121.534(15)°, γ = 76.508(15)°, and V = 852.1(6) Å3, Z = 2 and Dcalc = 3.34 g·cm–3. The strongest diffraction peaks in the X-ray powder diffraction pattern are [dobs in Å (Irel.)(hkl)]: 8.872(100)(100), 8.067(50)(002), 6.399(7)(10 ), 4.773(6)(10
), 4.773(6)(10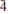 ), 3.411(10)(30
), 3.411(10)(30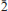 ), 3.197(18)(31
), 3.197(18)(31 ). The crystal structure of vysokýite was solved from single-crystal X-ray diffraction data by the charge-flipping method and refined to R1 = 0.0595 based on 2718 unique observed reflection, and to wR2 = 0.1160 for all 4173 unique reflections. The structure of vysokýite consists of UO8 square antiprisms sharing all of their vertices with 8 As-tetrahedra to form infinite chains parallel to [010]. These chains are linked by hydrogen bonds involving terminal (OH) groups of the double-protonated As-tetrahedra and molecules of H2O located between the chains. The new mineral is named in honour of Arnošt Vysoký (1823–1872), the former chief of the Jáchymov mines and smelters, chemist and metallurgist.
). The crystal structure of vysokýite was solved from single-crystal X-ray diffraction data by the charge-flipping method and refined to R1 = 0.0595 based on 2718 unique observed reflection, and to wR2 = 0.1160 for all 4173 unique reflections. The structure of vysokýite consists of UO8 square antiprisms sharing all of their vertices with 8 As-tetrahedra to form infinite chains parallel to [010]. These chains are linked by hydrogen bonds involving terminal (OH) groups of the double-protonated As-tetrahedra and molecules of H2O located between the chains. The new mineral is named in honour of Arnošt Vysoký (1823–1872), the former chief of the Jáchymov mines and smelters, chemist and metallurgist.
Hloušekite, (Ni,Co)Cu4(AsO4)2(AsO3OH)2(H2O)9, a new member of the lindackerite supergroup from Jáchymov, Czech Republic
- J. Plášil, J. Sejkora, R. Škoda, M. Novák, A. V. Kasatkin, P. Škácha, F. Veselovský, K. Fejfarová, P. Ondruš
-
- Journal:
- Mineralogical Magazine / Volume 78 / Issue 5 / October 2014
- Published online by Cambridge University Press:
- 05 July 2018, pp. 1341-1353
-
- Article
- Export citation
-
Hloušekite, (Ni,Co)Cu4(AsO4)2(AsO3OH)2(H2O)9, is a new supergene arsenate mineral from the Geister vein (Rovnost mine), Jáchymov (St Joachimsthal), Western Bohemia, Czech Republic. It was found along with veselovský ite, pradetite, lavendulan, arsenolite, babánekite and gypsum on the surface of strongly altered ore fragments containing dominant tennantite and chalcopyrite. Hloušekite forms thin, lath-like crystals, locally elongated reaching up to 3 mm across. It is transparent, has a pale green colour with vitreous lustre, has a greyish-white streak and it is very brittle with an uneven fracture. It does not fluoresce under shortwave or longwave ultraviolet radiation. Cleavage on {010} is perfect; the Mohs hardness is 2–3. The calculated density is 3.295 g cm–3. Hloušekite is optically biaxial with α’ = 1.653(2) and γ’ = 1.73. The estimated optical orientation is γ’ vs. elongation (c) = 14(1)°. In larger grains it is weakly to moderately pleochroic (α = colourless, γ = pale green to green). Hloušekite is triclinic, space group P
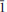 , a = 6.4010(6), b = 8.0041(6), c = 10.3969(14) Å , α = 85.824(8), β = 79.873(9), γ = 84.655(7)° and V = 521.23(10) Å3, with Z = 1, a:b:c = 0.800:1:1.299. The eight strongest lines in the powder X-ray diffraction (XRD) pattern [d in Å (I)(hkl)] are 10.211(100)(001), 7.974(9)(010), 3.984(6)(020), 3.656(5)(1
, a = 6.4010(6), b = 8.0041(6), c = 10.3969(14) Å , α = 85.824(8), β = 79.873(9), γ = 84.655(7)° and V = 521.23(10) Å3, with Z = 1, a:b:c = 0.800:1:1.299. The eight strongest lines in the powder X-ray diffraction (XRD) pattern [d in Å (I)(hkl)] are 10.211(100)(001), 7.974(9)(010), 3.984(6)(020), 3.656(5)(1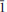 2), 3.631(5)(0
2), 3.631(5)(0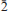 1), 3.241(5)(022), 3.145(5)(200) and 3.006(5)(210). Chemical analysis by electron microprobe yielded MgO0.20, FeO0.10, NiO 5.79, CoO1.80, CuO29.53, ZnO 0.66, Al2O3 0.14, P2O5 0.11, As2O5 45.01, H2O 17.71 (calc.), for a total of 101.05 wt.%. The resulting empirical formula, calculated by stoichiometry (9H2O + 2OH), obtained from the crystal structure, is (Ni0.79Co0.25)Σ1.04(Cu3.78Zn0.08Mg0.05Al0.03Fe0.01)Σ3.95 (AsO4 )1.98(PO4 )0.02(AsO3OH)2.00(H2O)9.00 . The ideal endmember formula , NiCu4(AsO4)2(AsO3OH)2(H2O)9.00, requires NiO7.23, CuO30.81, As2O5 44.51, H2O17.45, total 100.00 wt.%. The crystal structure of hloušekite was solved by charge flipping from single-crystal XRD data and refined to R1 = 0.0599 for 1441 reflections with [Iobs > 3σ(I)]. Hloušekite is a new member of the lindackerite group (also including lindackerite, pradetite and veselovský ite) of the lindackerite supergroup. The ondrušite group of the lindackerite supergroup includes ondrušite, chudobaite, geigerite and klajite. The establishment of these two groups reflects the difference between the crystal structures of their members, mainly in the coordination environment of the Me cations.
1), 3.241(5)(022), 3.145(5)(200) and 3.006(5)(210). Chemical analysis by electron microprobe yielded MgO0.20, FeO0.10, NiO 5.79, CoO1.80, CuO29.53, ZnO 0.66, Al2O3 0.14, P2O5 0.11, As2O5 45.01, H2O 17.71 (calc.), for a total of 101.05 wt.%. The resulting empirical formula, calculated by stoichiometry (9H2O + 2OH), obtained from the crystal structure, is (Ni0.79Co0.25)Σ1.04(Cu3.78Zn0.08Mg0.05Al0.03Fe0.01)Σ3.95 (AsO4 )1.98(PO4 )0.02(AsO3OH)2.00(H2O)9.00 . The ideal endmember formula , NiCu4(AsO4)2(AsO3OH)2(H2O)9.00, requires NiO7.23, CuO30.81, As2O5 44.51, H2O17.45, total 100.00 wt.%. The crystal structure of hloušekite was solved by charge flipping from single-crystal XRD data and refined to R1 = 0.0599 for 1441 reflections with [Iobs > 3σ(I)]. Hloušekite is a new member of the lindackerite group (also including lindackerite, pradetite and veselovský ite) of the lindackerite supergroup. The ondrušite group of the lindackerite supergroup includes ondrušite, chudobaite, geigerite and klajite. The establishment of these two groups reflects the difference between the crystal structures of their members, mainly in the coordination environment of the Me cations.
Rare-earth crystal chemistry of thalénite-(Y) from different environments
- Markus B. Raschke, Evan J. D. Anderson, Jason Van Fosson, Julien M. Allaz, Joseph R. Smyth, Radek Škoda, Philip M. Persson, Randy Becker
-
- Journal:
- Mineralogical Magazine / Volume 82 / Issue 2 / April 2018
- Published online by Cambridge University Press:
- 28 February 2018, pp. 313-327
-
- Article
- Export citation
-
Thalénite-(Y), ideally Y3Si3O10F, is a heavy-rare-earth-rich silicate phase occurring in granite pegmatites that may help to illustrate rare-earth element (REE) chemistry and behaviour in natural systems. The crystal structure and mineral chemistry of thalénite-(Y) were analysed by electron microprobe analysis, X-ray diffraction and micro-Raman spectroscopy from a new locality in the peralkaline granite of the Golden Horn batholith, Okanogan County, Washington State, USA, in comparison with new analyses from the White Cloud pegmatite in the Pikes Peak batholith, Colorado, USA. The Golden Horn thalénite-(Y) occurs as late-stage sub-millimetre euhedral bladed transparent crystals in small miarolitic cavities in an arfvedsonite-bearing biotite granite. It exhibits growth zoning with distinct heavy-rare-earth element (HREE) vs. light-rare-earth element (LREE) enriched zones. The White Cloud thalénite-(Y) occurs in two distinct anhedral and botryoidal crystal habits of mostly homogenous composition. In addition, minor secondary thalénite-(Y) is recognized by its distinct Yb-rich composition (up to 0.8 atoms per formula unit (apfu) Yb). Single-crystal X-ray diffraction analysis and structure refinement reveals Y-site ordering with preferential HREE occupation of Y2 vs. Y1 and Y3 REE sites. Chondrite normalization shows continuous enrichment of HREE in White Cloud thalénite-(Y), in contrast to Golden Horn thalénite-(Y) with a slight depletion of the heaviest REE (Tm, Yb and Lu). The results suggest a hydrothermal origin of the Golden Horn miarolitic thalénite-(Y), compared to a combination of both primary magmatic followed by hydrothermal processes responsible for the multiple generations over a range of spatial scales in White Cloud thalénite-(Y).
Alwilkinsite-(Y), a new rare-earth uranyl sulfate mineral from the Blue Lizard mine, San Juan County, Utah, USA
- Anthony R. Kampf, Jakub Plášil, Jiří Čejka, Joe Marty, Radek Škoda, Ladislav Lapčák
-
- Journal:
- Mineralogical Magazine / Volume 81 / Issue 4 / August 2017
- Published online by Cambridge University Press:
- 02 January 2018, pp. 895-907
-
- Article
- Export citation
-
The new mineral alwilkinsite-(Y) (IMA2015-097), Y(H2O)7[(UO2)3(SO4)2O(OH)3]·7H2O, was found in the Blue Lizard mine, San Juan County, Utah, USA, where it occurs as a secondary alteration phase.The mineral is slightly flexible before brittle failure with splintery fracture and perfect cleavage parallel to [010], has Mohs hardness of ∼2–2½, exhibits dull greenish-grey fluorescence and has a calculated density of 3.371 g cm–3. Alwilkinsite-(Y) occursas yellowish-green needles, elongate on [010], with domatic terminations and exhibits the forms {102}, {301} and {124}. It is optically biaxial (+) with α = 1.573(1), β = 1.581(1), γ = 1.601(1) (white light), the measured 2V is 65.3(1)°, the dispersion is r<v (weak), the optical orientation is X = c, Y = a, Z = b and there is no pleochroism. Electron microprobe analyses yielded the empirical formula (Y0.66Dy0.08Gd0.06Er0.05Nd0.03Yb0.03Sm0.02Ce0.01)∑0.94(H2O)7[(UO2)3(S1.01O4)2O(OH)3]·7H2O.The eight strongest powder X-ray diffraction lines are [dobs Å(I)(hkl)]: 9.88(100)(101,002), 7.47(13)(102), 5.621(17)(103,201), 4.483(18)(104), 3.886(14)(130,222), 3.322(46)(multiple), 3.223(13)(multiple) and 3.145(16)(034). Alwilkinsite-(Y) is orthorhombic,P212121, a = 11.6194(5), b = 12.4250(6), c = 19.4495(14) Å, V = 2807.9(3) Å3 and Z = 4. The structure of alwilkinsite-(Y) (R1 = 0.042 for 4244 Fo > 4σF)contains edge-sharing chains of uranyl bipyramids with outlying sulfate tetrahedra that are similar to the chain linkages within the uranyl sulfate sheets of the zippeite structure. Short segments of the uranyl sulfate chains in the alwilkinsite-(Y) structure have the same topology as portionsof the uranyl sulfate linkages in uranopilite. Alwilkinsite-(Y) is named for Alan (Al) J. Wilkins, MD (born 1955), the discoverer of the mineral.
New crystal-chemical data for marécottite
- J. Plášil, R. Škoda
-
- Journal:
- Mineralogical Magazine / Volume 79 / Issue 3 / June 2015
- Published online by Cambridge University Press:
- 02 January 2018, pp. 649-660
-
- Article
- Export citation
-
Marécottite, ideally Mg3[(UO2)4O3(OH)(SO4)2]2(H2O)28, a triclinic, Mg-dominant member of the zippeite group, was described originally from a small uranium deposit at La Creusaz in Wallis (Switzerland). It has recently been found at Jáchymov (Czech Republic), where it forms exceptional crystals, up to 0.3 mm across. According to an electron microprobe study of these crystals, marécottite from Jáchymov is chemically similar to the material from the La Creusaz deposit. However, the Jáchymov crystals exhibit more cation substitution (Zn2+ and Mn2+ for Mg2+). The chemical composition of marécottite from Jáchymov corresponds to the empirical formula [(Na0.05K0.07)Σ0.12(Mg1.83Zn0.41Mn0.41Cu0.15Ni0.08)Σ2.88Al0.07]Σ3.07(UO2)8[(SO4)3.77(SiO4)0.21]Σ3.98O6(OH)1.84·28H2O (the mean of four representative spots; calculated on the basis of eight U atoms and 28 H2O per formula unit and 1.84 OH for charge balance). According to single-crystal X-ray diffraction, marécottite from Jáchymov is triclinic, P1, a = 10.8084(2), b = 11.2519(3), c = 13.8465(3) Å, α = 66.222(2), β = 72.424(2), γ = 70.014(2)o, V = 1421.57(6) Å3 and Z = 1. The crystal structure was refined from a highly redundant dataset (30,491 collected reflections) to R1 = 0.0367 for all 7042 unique reflections. The refined structure confirms the previously determined structure for the crystal from the La Creusaz deposit. An extensive network of hydrogen bonds is an important feature that keeps the whole structure together, but the positions of H atoms had not been determined previously. The H-bond scheme proposed based on a detailed bond-valence analysis and the role of different types of molecular H2O in the structure is discussed.
Geschieberite, K2(UO2)(SO4)2(H2O)2, a new uranyl sulfate mineral from Jáchymov
- J. Plášil, J. Hloušek, A. V. Kasatkin, R. Škoda, M. Novák, J. Čejka
-
- Journal:
- Mineralogical Magazine / Volume 79 / Issue 1 / February 2015
- Published online by Cambridge University Press:
- 02 January 2018, pp. 205-216
-
- Article
- Export citation
-
The new mineral geschieberite (IMA2014-006), K2(UO2)(SO4)2(H2O)2, was found in the Svornost mine, Jáchymov, Czech Republic, where it occurs as a secondary alteration phase after uraninite in association with adolfpateraite and gypsum. Geschieberite forms crystalline aggregates of bright green colour (when thick) composed of multiply intergrown prismatic crystals elongated on [001] typically reaching 0.1–0.2 mm across; observable forms are {010} and {001}. Crystals are translucent to transparent with a vitreous lustre. The mineral is brittle, with perfect cleavage on {100} and an uneven fracture. It has a greenish-white streak and a probable Mohs hardness of ∼2. The mineral is slightly soluble in cold H2O. The calculated density is 3.259 g cm–3. The mineral exhibits strong yellowish-green fluorescence under both shortwave and longwave UV radiation. Optically, geschieberite is biaxial (–), with β = 1.596(2) and γ = 1.634(4) (measured at 590 nm), with X = a. Electron-microprobe analyses provided Na2O 0.23, K2O 14.29, MgO 2.05, CaO 0.06, UO3 49.51, SO3 27.74, H2O 6.36 (structure), total 100.24 wt.%, yielding the empirical formula (K1.72Mg0.29Na0.04Ca0.01)Σ2.06(U0.98O2)(S0.98O4)2(H2O)2 based on 12 O atoms per formula unit. The Raman spectrum is dominated by the symmetric stretching vibrations of UO22+, SO42– and weaker O–H stretching vibrations. Geschieberite is orthorhombic, Pna21, with a = 13.7778(3), b = 7.2709(4), c = 11.5488(2) Å, V = 1156.92(7) Å3, Z = 4. The eight strongest powder X-ray diffraction lines are [dobs in Å (hkl) Irel]: 6.882 (200) 100, 5.622 (111) 53, 4.589 (211) 12, 4.428 (202) 16, 3.681 (311) 18, 3.403 (013) 12, 3.304 (401,1̄13) 15 and 3.006 (122) 17. The structure, refined to R = 0.028 for 1882 I > 3σ(I) reflections, contains [(UO2)(SO4)2(H2O)]2– sheets that are based on the protasite anion topology. Sheets are stacked perpendicular to a. Potassium atoms and one H2O molecule are located between these sheets, providing an interlayer linkage. The remaining H2O molecule is localized within the structural unit, at the free vertex of the uranyl pentagonal bipyramid; this vertex does not link to sulfate tetrahedra. The mineral is named for one of the most important ore veins in Jáchymov – the Geschieber vein.
Survey of entomopathogenic nematodes from the families Steinernematidae and Heterorhabditidae (Nematoda: Rhabditida) in Colima, México
- Isaac Zepeda-Jazo, Jaime Molina-Ochoa, Roberto Lezama-Gutiérrez, Steven R. Skoda, John E. Foster
-
- Journal:
- International Journal of Tropical Insect Science / Volume 34 / Issue 1 / March 2014
- Published online by Cambridge University Press:
- 04 February 2014, pp. 53-57
- Print publication:
- March 2014
-
- Article
- Export citation
-
A survey of entomopathogenic nematodes (EPNs) belonging to the families Steinernematidae and Heterorhabditidae was conducted in three municipalities on the Pacific coast of the State of Colima, México, to determine their occurrence and recovery frequency and predominant plant species in cultivated and non-cultivated habitats. Nineteen soil samples were collected: seven from non-cultivated habitats and 12 from habitats or areas cultivated mostly with fruit and grain crops and grasses. Of the 19 soil samples, 14 were positive for EPNs; the total prevalence was 73.7%. From the 14 positive soil samples, 12 steinernematid isolates (85.7%) and two heterorhabditid isolates (14.3%) were recovered. Irrespective of the locations, EPNs from the genus Steinernema were recovered from the three municipalities; EPNs from the genera Steinernema and Heterorhabditis were recovered from Armería and Ixtlahuacán. Only steinernematid isolates were recovered from non-cultivated habitats. Most of the isolates were recovered from cultivated habitats, and our results suggest that there is a higher prevalence of EPNs in cultivated soils.
Introduction
- from Part I - Boundaries and Units
- Edited by Hannah Skoda, Patrick Lantschner, R. L. J. Shaw
-
- Book:
- Contact and Exchange in Later Medieval Europe
- Published by:
- Boydell & Brewer
- Published online:
- 05 February 2013
- Print publication:
- 16 August 2012, pp 27-32
-
- Chapter
- Export citation
-
Summary
It is obvious that processes of exchange must be shaped and often constrained by the boundaries across which they operate, and it is therefore fitting to open this volume with various explorations of the nature of boundaries and units in the later Middle Ages. The relationship between developing boundaries and units, and processes of exchange, is a reciprocal one: the nature of boundaries was itself moulded by the contacts and exchanges which took place across them. This is true of a variety of different types of contact. In the commercial sphere, Spindler's article demonstrates that perceived boundaries between what it was to be considered English or Flemish respectively were concretised by the presence of Flemings in an alien environment. In the social sphere, Dumolyn's article illustrates how social boundaries were shaped by the fear of too much social interchange. Branco and Pépin both consider the ways in which linguistic influences and identities shored up perceptions of boundaries. Keen demonstrates that political exchange across Channel and political borders actually helped to shape the nature of those boundaries, and engendered far more complex configurations of political networks than the French versus English paradigm we might assume.
The relationship between units and boundaries was indeed especially complex, and potentially highly charged: the possibility of their deliberate manipulation in the interests of political expediency meant that exchange across boundaries was a particularly effective way of making points about social and territorial relationships.




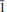 , with
, with 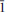 , with
, with 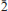 0); 3.097(49)(1
0); 3.097(49)(1
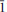 11), 5.321 (66) (004), 3.549 (5) (006), 2.663 (4) (008), 2.131 (2) (0 0 10). The crystal structure has been refined from single-crystal X-ray diffraction data to
11), 5.321 (66) (004), 3.549 (5) (006), 2.663 (4) (008), 2.131 (2) (0 0 10). The crystal structure has been refined from single-crystal X-ray diffraction data to  ,
, 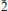 1,020) 52, 5.24 (100,012,
1,020) 52, 5.24 (100,012, 2,
2,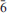 2
2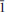 }. The mineral is transparent and has a white streak, subadamantine lustre, Mohs hardness of about 2½, brittle tenacity, irregular to splintery fracture, one perfect cleavage on {001} and possibly one or more additional cleavages parallel to [100]. Calciodelrioite is soluble in water. The calculated density is 2.451 g cm
}. The mineral is transparent and has a white streak, subadamantine lustre, Mohs hardness of about 2½, brittle tenacity, irregular to splintery fracture, one perfect cleavage on {001} and possibly one or more additional cleavages parallel to [100]. Calciodelrioite is soluble in water. The calculated density is 2.451 g cm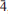 15,413); 1.786(18)(028). In the structure of calciodelrioite (refined to R
15,413); 1.786(18)(028). In the structure of calciodelrioite (refined to R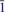 , with
, with  ), 4.773(6)(10
), 4.773(6)(10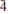 ), 3.411(10)(30
), 3.411(10)(30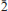 ), 3.197(18)(31
), 3.197(18)(31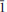 , a = 6.4010(6), b = 8.0041(6), c = 10.3969(14) Å , α = 85.824(8), β = 79.873(9), γ = 84.655(7)° and V = 521.23(10) Å
, a = 6.4010(6), b = 8.0041(6), c = 10.3969(14) Å , α = 85.824(8), β = 79.873(9), γ = 84.655(7)° and V = 521.23(10) Å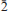 1), 3.241(5)(022), 3.145(5)(200) and 3.006(5)(210). Chemical analysis by electron microprobe yielded MgO
1), 3.241(5)(022), 3.145(5)(200) and 3.006(5)(210). Chemical analysis by electron microprobe yielded MgO