Refine listing
Actions for selected content:
Volume 77 - Issue 6 - August 2013
Contents
CNMNC Newsletter 16
New minerals and nomenclature modifications approved in 2013
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 2695-2709
-
- Article
- Export citation
Research Article
Švenekite, Ca[AsO2(OH)2]2, a new mineral from Jáchymov, Czech Republic
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 2711-2724
-
- Article
- Export citation
Pseudomorphic transformation of Ca/Mg carbonates into phosphates with focus on dolomite conversion
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 2725-2737
-
- Article
- Export citation
Vanadoallanite-(La): a new epidote-supergroup mineral from Ise, Mie Prefecture, Japan
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 2739-2752
-
- Article
- Export citation
From structure topology to chemical composition. XIV. Titanium silicates: refinement of the crystal structure and revision of the chemical formula of mosandrite, (Ca3 REE)[(H2O)2Ca0.5□0.5]Ti(Si2O7)2(OH)2(H2O)2, a Group-I mineral from the Saga mine, Morje, Porsgrunn, Norway
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 2753-2771
-
- Article
- Export citation
Oxo-magnesio-hastingsite, NaCa2(Mg2Fe3+3 )(Al2Si6)O22O2, a new anhydrous amphibole from the Deeti volcanic cone, Gregory rift, northern Tanzania
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 2773-2792
-
- Article
- Export citation
Mineralogy of atmospheric dust impacting the Rio Tinto mining area (Spain) during episodes of high metal deposition
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 2793-2810
-
- Article
- Export citation
Joteite, Ca2CuAl[AsO4][AsO3(OH)]2(OH)2·5H2O, a new arsenate with a sheet structure and unconnected acid arsenate groups
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 2811-2823
-
- Article
- Export citation
The dumortierite supergroup. I. A new nomenclature for the dumortierite and holtite groups
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 2825-2839
-
- Article
- Export citation
The dumortierite supergroup. II. Three new minerals from the Szklary pegmatite, SW Poland: Nioboholtite, (Nb0.6□0.4)Al6BSi3O18, titanoholtite, (Ti0.75□0.25)Al6BSi3O18, and szklaryite, □Al6BAs3+ 3O15
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 2841-2856
-
- Article
- Export citation
Irinarassite Ca3Sn2SiAl2O12 – new garnet from the Upper Chegem Caldera, Northern Caucasus, Kabardino-Balkaria, Russia
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 2857-2866
-
- Article
- Export citation
Alpha particle damage in biotite characterized by microfocus X-ray diffraction and Fe K-edge X-ray absorption spectroscopy
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 2867-2882
-
- Article
-
- You have access
- Open access
- Export citation
Obituary
Professor Neil F.C. Hudson: 1947-2012
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 2883-2885
-
- Article
-
- You have access
- Export citation
Front matter
MGM volume 77 issue 6 Cover and Front matter
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. f1-f6
-
- Article
-
- You have access
- Export citation
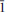 , with
, with 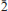 0); 3.097(49)(1
0); 3.097(49)(1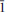 0 0, 0
0 0, 0  ), 2.349 (29) (
), 2.349 (29) (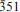 ) and 2.162 (36) (261). The isotopic composition of H and O, as well as the concentration of trace elements in oxo-magnesio-hastingsite suggest its formation from a meltoriginated from a mantle source metasomatized by slab-derived fluids.
) and 2.162 (36) (261). The isotopic composition of H and O, as well as the concentration of trace elements in oxo-magnesio-hastingsite suggest its formation from a meltoriginated from a mantle source metasomatized by slab-derived fluids.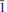 0}, {2
0}, {2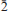 2,
2,