Volume 78 - Issue 7 - December 2014
Research Article
The crystal structure of kiddcreekite solved using micro X-ray diffraction and the EPCryst program
- Liu Wenyuan, Dong Cheng, Gu Xiangping, Liu Yu, Qiu Xiaoping, Chen Yuchuan
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 1517-1525
-
- Article
- Export citation
-
Abundant kiddcreekite grains were identified in the Zijinshan Cu-Au epithermal deposit in Fujian Province, China. The mineral occurs as polycrystalline grains, 5–300 μm in size, associated with colusite, enargite, stannoidite, mawsonite, vinciennite, hemusite, tennantite and wolframite in a predominantly covellite ore. Based on electron microprobe analysis, the empirical formula of the kiddcreekite is Cu6.2Sn0.97W0.95S7.83, without significant Se or Te contents. The crystal structure of kiddcreekite was solved using the direct-space method (EPCryst) from laboratory micro X-ray diffraction (μXRD) data and refined by the Rietveld method. The R values of the final Rietveld refinement were Rp = 9.06%, Rwp = 8.31%, RB = 3.16 and RF = 2.17%. Kiddcreekite has a cubic structure, space group F
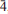 3m and lattice parameter a = 10.8178(3) Å (Z = 4, V = 1265.95(6) Å3). In the unit cell, W, Sn and Cu atoms occupy the 4a, 4c and 24f Wyckoff positions, respectively, and S atoms occupy two sets of 16e Wyckoff positions. The structure of kiddcreekite consists of stacked double MeS4 layers (giving a W–Sn–Cu tier and a Cu–vacancy tier) as in the sphalerite substructure. This study also demonstrates the possibility of using laboratory µXRD data coupled with the direct-space method to solve inorganic structures in cases where samples are too small for conventional powder and single-crystal diffraction.
3m and lattice parameter a = 10.8178(3) Å (Z = 4, V = 1265.95(6) Å3). In the unit cell, W, Sn and Cu atoms occupy the 4a, 4c and 24f Wyckoff positions, respectively, and S atoms occupy two sets of 16e Wyckoff positions. The structure of kiddcreekite consists of stacked double MeS4 layers (giving a W–Sn–Cu tier and a Cu–vacancy tier) as in the sphalerite substructure. This study also demonstrates the possibility of using laboratory µXRD data coupled with the direct-space method to solve inorganic structures in cases where samples are too small for conventional powder and single-crystal diffraction.
The crystal structure of camerolaite and structural variation in the cyanotrichite family of merotypes
- S. J. Mills, A. G. Christy, C. Schnyder, G. Favreau, J. R. Price
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 1527-1552
-
- Article
- Export citation
-
We present Raman data for camerolaite, cyanotrichite and carbonatecyanotrichite, and using synchrotron single-crystal X-ray diffraction have solved the structure of camerolaite from the Tistoulet Mine, Padern, Aude Department, France. Camerolaite crystallizes in space group P1 with the unit-cell parameters: a = 6.3310(13) Å, b = 2.9130(6) Å, c = 10.727(2) Å, α = 93.77(3)°, β = 96.34(3)°, γ =79.03(3)º, V = 192.82(7) Å3 and Z = ⅓, with respect to the ideal formula from the refinement, Cu6Al3(OH)18(H2O)2[Sb(OH)6](SO4). The crystal structure was solved to R1 = 0.0890 for all 1875 observed reflections [Fo > 4σFo] and 0.0946 for all 2019 unique reflections. The P cell has been transformed into a C-centred cell that aids comparison with that of the structurally related khaidarkanite by aC = 2aP – bP, giving parameters a = 12.441(3), b = 2.9130(6), c = 10.727(2) Å, α = 93.77(3), β = 95.57(3), γ = 92.32(3)º and Z = ⅔ in C1. Edge-sharing octahedral ribbons Cu2Al(O,OH,H2O)8 form hydrogen-bonded layers || (001), as in khaidarkanite. The partially occupied interlayer Sb and S sites of the average structure are in octahedral and tetrahedral coordination by oxygen, respectively. They cannot be occupied simultaneously, which leads to regular alternation of [Sb(OH)6]– and SO42– groups in rods || y, resulting in local tripling of the periodicity along y for the Sb(OH)6–SO4 rods. Thus, camerolaite has a ‘host–guest’ structure in which an invariant host module (layers of Cu–Al ribbons) has embedded rod-like guest modules with a longer periodicity. Coupling between the phases of these rods is only short-range, resulting in diffuse X-ray scattering rather than sharp superstructure reflections. Similar disorder is known for parnauite, and is deduced for other members of the cyanotrichite group (cyanotrichite, carbonatecyanotrichite and khaidarkanite). Group members all share the Cu–Al ribbon module but have interlayer rods of different compositions and topologies; thus, they form a merotypic family. The low symmetry of the camerolaite average structure suggests other possibilities for structure variation in the group, which are discussed.
New arsenate minerals from the Arsenatnaya fumarole, Tolbachik volcano, Kamchatka, Russia. II. Ericlaxmanite and kozyrevskite, two natural modifications of Cu4O(AsO4)2
- I. V. Pekov, N. V. Zubkova, V. O. Yapaskurt, D. I. Belakovskiy, M. F. Vigasina, E. G. Sidorov, D. Yu. Pushcharovsky
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 1553-1569
-
- Article
- Export citation
-
Two new minerals, ericlaxmanite and kozyrevskite, dimorphs of Cu4O(AsO4)2, were found in sublimates of the Arsenatnaya fumarole at the Second scoria cone of the Northern Breakthrough of the Great Tolbachik Fissure Eruption, Tolbachik volcano, Kamchatka, Russia. They are associated with each other and with urusovite, lammerite, lammerite-b, popovite, alarsite, tenorite, hematite, aphthitalite, langbeinite, As-bearing orthoclase, etc. Ericlaxmanite occurs as tabular, lamellar, equant or short prismatic crystals up to 0.1 mm in size, their clusters and pseudomorphs after urusovite crystal crusts up to 1.5 cm × 2 cm in area. Kozyrevskite occurs as prismatic crystals up to 0.3 mm long in clusters and as individual crystals. Both minerals are transparent with a vitreous lustre. They are brittle, with Mohs’ hardness ~3–. Ericlaxmanite is green to dark green. Kozyrevskite is bright grass green to light yellowish green; Dcalc is 5.036 (ericlaxmanite) and 4.934 (kozyrevskite) g cm–3. Both minerals are optically biaxial (–); ericlaxmanite: α = 1.870(10), β = 1.900(10), γ = 1.915(10), 2Vmeas = 60(15)º; kozyrevskite: α = 1.885(8), β = 1.895(8), γ = 1.900(8), 2Vmeas. = 75(10)º. The Raman spectra are given. Chemical data (wt.%, electron microprobe; the first value is for ericlaxmanite, the second for kozyrevskite): CuO 57.55, 58.06; ZnO 0.90, 1.04; Fe2O3 0.26, 0.12; SiO2 n.d., 0.12; P2O5 0.23, 1.23; V2O5 0.14, 0.37; As2O5 40.57, 38.78; SO3 0.17, 0.43; total 99.82, 100.15. The empirical formulae, based on 9 O a.p.f.u., are: ericlaxmanite: (Cu3.97Zn0.06Fe0.02)Σ4.05(As1.94P0.02V0.01S0.01)Σ1.98O9 and kozyrevskite: (Cu3.95Zn0.07Fe0.01)Σ4.03(As1.83P0.09S0.03V0.02Si0.01)Σ1.98O9. Ericlaxmanite is triclinic, P
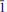 , a = 6.4271(4), b = 7.6585(4), c = 8.2249(3) Å , α = 98.396(4), β = 112.420(5), γ = 98.397(5)º, V = 361.11(3) Å3 and Z = 2. Kozyrevskite is orthorhombic, Pnma, a = 8.2581(4), b = 6.4026(4), c = 13.8047(12) Å , V = 729.90(9) Å3 and Z = 4. The strongest reflections in the X-ray powder patterns [d Å (I)(hkl)] are: ericlaxmanite: 3.868(46)(101), 3.685(100)(020), 3.063(71)(012), 2.957(58)(0
, a = 6.4271(4), b = 7.6585(4), c = 8.2249(3) Å , α = 98.396(4), β = 112.420(5), γ = 98.397(5)º, V = 361.11(3) Å3 and Z = 2. Kozyrevskite is orthorhombic, Pnma, a = 8.2581(4), b = 6.4026(4), c = 13.8047(12) Å , V = 729.90(9) Å3 and Z = 4. The strongest reflections in the X-ray powder patterns [d Å (I)(hkl)] are: ericlaxmanite: 3.868(46)(101), 3.685(100)(020), 3.063(71)(012), 2.957(58)(0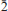 2), 2.777(98)(
2), 2.777(98)(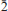 12,
12, 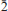
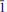 1), 2.698(46)(
1), 2.698(46)(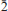
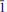 2) and 2.201(51)(013, 031); kozyrevskite: 3.455(100)(004), 3.194(72)(020, 104), 2.910(69)(022), 2.732(82)(122), 2.712(87)(301) and 2.509(92)(123). Their crystal structures, solved from single-crystal X-ray diffraction data [R = 0.0358 (ericlaxmanite) and 0.1049 (kozyrevskite)], are quite different. The ericlaxmanite structure is based on an interrupted framework built by edge- and corner-sharing Cu-centred, distorted tetragonal pyramids, trigonal bipyramids and octahedra. The kozyrevskite structure is based on complicated ribbons of Cu-centred distorted tetragonal pyramids and trigonal bipyramids. Ericlaxmanite is named in honour of the Russian mineralogist, geologist, geographer, biologist and chemist Eric Laxman (1737–1796). Kozyrevskite is named in honour of the Russian geographer, traveller and military man Ivan Petrovich Kozyrevskiy (1680–1734), one of the first researchers of Kamchatka.
2) and 2.201(51)(013, 031); kozyrevskite: 3.455(100)(004), 3.194(72)(020, 104), 2.910(69)(022), 2.732(82)(122), 2.712(87)(301) and 2.509(92)(123). Their crystal structures, solved from single-crystal X-ray diffraction data [R = 0.0358 (ericlaxmanite) and 0.1049 (kozyrevskite)], are quite different. The ericlaxmanite structure is based on an interrupted framework built by edge- and corner-sharing Cu-centred, distorted tetragonal pyramids, trigonal bipyramids and octahedra. The kozyrevskite structure is based on complicated ribbons of Cu-centred distorted tetragonal pyramids and trigonal bipyramids. Ericlaxmanite is named in honour of the Russian mineralogist, geologist, geographer, biologist and chemist Eric Laxman (1737–1796). Kozyrevskite is named in honour of the Russian geographer, traveller and military man Ivan Petrovich Kozyrevskiy (1680–1734), one of the first researchers of Kamchatka.
The Piteiras emerald mine, Minas Gerais, Brazil: fluid-inclusion and gemmological perspectives
- E. P. Lynch, A. Costanzo, M. Feely, N. J. F. Blamey, J. Pironon, P. Lavin
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 1571-1587
-
- Article
- Export citation
-
New fluid inclusion analyses using a range of analytical techniques including quadropole mass spectrometric analyses coupled with gemmological investigations were conducted on rough and gemquality faceted emeralds from the Piteiras Mine, Minas Gerais, Brazil. These data complement those of Rondeau et al. (2003) who also presented analyses of the Piteiras emeralds. Emeralds are found typically as euhedral-to-anhedral crystals disseminated in biotite-phlogopite schist and range from 0.5 to 50 mm long. Emerald mineralization is associated closely with alkaline metasomatized pegmatite bodies, quartz boudin and veins, and talc-chlorite bands. Four types of fluid inclusions are recognized in the emeralds. These include aqueous brine and aqueous carbonic varieties containing one or two liquid phases, along with gas bubbles and/or solid crystals (e.g. carbonates). Primary fluid inclusions in emeralds record salinities of ~4–24 eq. wt.% NaCl and minimum trapping temperatures from ~350 to 480ºC. Combined microthermometry, Raman spectroscopy and crush-leach gas analyses indicate that the mineralizing fluid was an aqueous carbonic brine enriched in reduced volatile species such as CH4, N2, H2S and alkanes. With respect to their optical properties (RI ε = 1.573–1.580; RI ω = 1.580–1.588; birefringence = 0.006–0.008) and specific gravity (2.65–2.78), the Piteiras emeralds fall within the expected range for metasomatic, schist-hosted emeralds.
Synthetic norsethite, BaMg(CO3)2: revised crystal structure, thermal behaviour and displacive phase transition
- H. Effenberger, T. Pippinger, E. Libowitzky, C. L. Lengauer, R. Miletich
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 1589-1611
-
- Article
- Export citation
-
The crystal structure of synthetic BaMg(CO3)2 whose mineral name is norsethite was re-investigated by single-crystal X-ray diffraction. Complementary in situ high- and low-temperature studies by means of vibrational spectroscopy (Raman, IR), powder X-ray diffraction techniques and thermal analyses were performed. Diffraction images (298 K) revealed weak superstructure reflections caused by the displacement of the O atoms in the earlier considered R
 m structure model (a = 5.0212(9), cnew = 2 cold = 33.581(6) Å , R
m structure model (a = 5.0212(9), cnew = 2 cold = 33.581(6) Å , R c, Z = 6, R1 = 0.011, sinθ/λ < 0.99 Å –1). Thermal analyses reveal decarbonatization in two decomposition steps above 750 K, and the heat-flow curves (difference scanning calorimetry) give clear evidence of a weak and reversible endothermal change at 343±1 K. This agrees with a discontinuity in the IR and single-crystal Raman spectra. The changing trend of the c/a ratio supports this discontinuity indicating a temperature-induced structural transition in the range between 343 and 373 K. As the change of the unit-cell volume is almost linear, the character of the transition is apparently second order and matches the mechanism of a subtle displacement of the oxygen atom position. The apparent instability of the R
c, Z = 6, R1 = 0.011, sinθ/λ < 0.99 Å –1). Thermal analyses reveal decarbonatization in two decomposition steps above 750 K, and the heat-flow curves (difference scanning calorimetry) give clear evidence of a weak and reversible endothermal change at 343±1 K. This agrees with a discontinuity in the IR and single-crystal Raman spectra. The changing trend of the c/a ratio supports this discontinuity indicating a temperature-induced structural transition in the range between 343 and 373 K. As the change of the unit-cell volume is almost linear, the character of the transition is apparently second order and matches the mechanism of a subtle displacement of the oxygen atom position. The apparent instability of the R c structure is also evidenced by the remarkably larger anisotropic displacement of the oxygen atom.
c structure is also evidenced by the remarkably larger anisotropic displacement of the oxygen atom.
Innsbruckite, Mn33(Si2O5)14(OH)38 – a new mineral from the Tyrol, Austria
- Hannes Krüger, Peter Tropper, Udo Haefeker, Reinhard Kaindl, Martina Tribus, Volker Kahlenberg, Christoph Wikete, Martin R. Fuchs, Vincent Olieric
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 1613-1627
-
- Article
- Export citation
-
A description of the new mineral innsbruckite, Mn33(Si2O5)14(OH)38, a hydrous manganese phyllosilicate found in Tyrol, Austria is given. The crystal structure was determined by singlecrystal synchrotron radiation diffraction experiments at the X06DA beamline at the Swiss Light Source (Paul Scherrer Institute, Villigen, Switzerland). The space group is Cm and lattice parameters are a = 17.2760(19), b = 35.957(5), c = 7.2560(8) Å , β = 91.359(7)º, V = 4506.1(10) Å3, Z = 2. Innsbruckite belongs to the group of modulated 1:1 layer silicates and is chemically and structurally quite closely related to bementite, Mn7(Si2O5)3(OH)8. The chemical analysis revealed a close to ideal composition with only minor amounts of Al, Fe and Mg. Using Liebau’s nomenclature for silicate classification the silicate anion can be described as an unbranched siebener single layer. Innsbruckite shows a complex topology of the silicate sheet, exhibiting 4-, 5-, 6- and 8-membered rings. The silicate sheet is fully characterized using vertex symbols, and its topology is compared to those in other complex sheet silicates. Furthermore, the structural investigation is complemented with Raman spectroscopic studies.
Leguernite, Bi12.67O14(SO4)5, a new Bi oxysulfate from the fumarole deposit of La Fossa crater, Vulcano, Aeolian Islands, Italy
- Anna Garavelli, Daniela Pinto, Donatella Mitolo, Luca Bindi
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 1629-1645
-
- Article
- Export citation
-
Leguernite, ideally Bi12.67O14(SO4)5, is a new mineral found in high-temperature fumarolic assemblages at La Fossa crater, Vulcano, Aeolian Islands, Italy. It occurs as aggregates of needleshaped crystals associated strictly with anglesite, balićžunićite and an unknown Bi sulfate. Leguernite is colourless to white, transparent, non-fluorescent, has a sub-adamantine lustre and a white streak. Electron microprobe data led to the chemical formula (on the basis of 34 anions p.f.u.) (Bi12.40Pb0.15)Σ=12.55S5.08O34. The calculated density is 7.375 g cm–3. A Raman spectrum collected on a single crystal of leguernite confirmed the anhydrous nature of the mineral.
Leguernite is monoclinic, space group P2, with a = 11.2486(11), b = 5.6568(6), c = 11.9139(10) Å , β = 99.177(7)º, V = 748.39(12) Å3 and Z = 1. The crystal structure is built up of Bi–O blocks of a fluorite-like structure with Bi12O14 composition separated by a single sulfate ion along [100] and by Bi(SO4)45– groups along [101]. It can also be described as composed of (001) layers with composition [Bi12O14(SO4)6+]n alternating with layers of composition [Bi(SO4)4]n5– along [001]. Leguernite shows significant similarities with the synthetic Bi14O16(SO4)5 compound.
The eight strongest reflections in the powder X-ray diffraction data [d in Å (I) (hkl)] are: 3.220 (100) (013), 3.100 (95) (
 11), 2.83 (30) (020), 2.931 (25) (302), 2.502 (25) (
11), 2.83 (30) (020), 2.931 (25) (302), 2.502 (25) ( 04), 2.035 (20) (322), 1.875 (20) (
04), 2.035 (20) (322), 1.875 (20) ( 24) and 5.040 (15) (110).
24) and 5.040 (15) (110).The name is in honour of Franc¸ois “Fanfan” Le Guern (1942–2011), who was a very active volcanologist and specialist in volcanic gases and sublimates. Both the mineral and the mineral name have been approved by the IMA-CNMNC (2013–051).
Structure of Sr-Zr-bearing perrierite-(Ce) from the Burpala Massif, Russia
- Marcin Stachowicz, Bogusław Bagiński, Ray Macdonald, Pavel M. Kartashov, Artur OzięBło, Krzysztof Wożniak
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 1647-1659
-
- Article
- Export citation
-
Sr- and Zr-bearing perrierite-(Ce) occurring in aegirinized syenite pegmatites of the Burpala massif, Russia, is compositionally intermediate between perrierite-(Ce) and hezuolinite and occupies a compositional gap in minerals of the chevkinite group. Its crystal structure has been determined using a single-crystal diffractometer fitted with a CCD detector and MoKα X-ray radiation. The mineral is monoclinic; a = 13.815(1), b = 5.668(1), c = 11.842(1) Å , β = 113.843(3)º, V = 848.18(4) Å3, space group C2/m, Z = 2. The crystal structure was refined with the occupancies [(Ce1.2La1.0Nd0.15) (Sr1.0Ca0.5Na0.15)]4(Zr0.5Fe0.3Mn0.2)(Ti1.3Fe0.7)2Ti2(Si2O7)2O8 on the basis of chemical composition although the allocation of cations to particular sites was performed on the basis of the number of refined electrons in each unique site. The dominance of Zr in the B site links the Burpala perrierite-(Ce) to more Sr-Zr-rich members of the chevkinite group, such as hezuolinite and rengeite. As in all of the perrierite members, there is a distortion of the D site octahedra, which is interpreted as due to the packing of the REE ions.
High-pressure Raman study of CH4 in melanophlogite (type I clathrate)
- G. D. Gatta, D. Bersani, P. P. Lottici, M. Tribaudino
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 1661-1669
-
- Article
- Export citation
-
The evolution with pressure of the CH4 vibrational modes related to C–H stretching has been followed by in situ Raman spectroscopy on a single crystal of cubic melanophlogite from Varano Marchesi (Parma, Italy) containing only CH4 in the cages. The sample was compressed hydrostatically in a diamond anvil cell up to 6.5 GPa, using a non-penetrating P-transmitting medium. Two modes at 2900 and 2910 cm–1 (ν1 and ν2) were followed in response to the applied pressure, corresponding to C–H stretching of CH4 enclathrated in the 51262 and 512 cages, respectively. A change in slope of the frequency vs. P linear trend of the sharper peak at 2900 cm–1, observed between 1 and 1.5 GPa, is interpreted as evidence of the P-induced cubic to tetragonal transition, previously observed by X-ray diffraction at P ≥ 1.2 GPa. At pressures below the transition, the shift with P of the CH4 modes is comparable to that observed in methane ice hydrate, which becomes hexagonal at P ≥ 0.9 GPa however. The ratio of the integrated areas of the two C–H stretching Raman peaks does not change significantly with pressure across the transition. At P ≥ 5.1 GPa, a shoulder appears, close to the n1 peak, along with a slight change in slope of peak shift and peak broadening. The shoulder is maintained in decompression down to P = 4.4 GPa, showing slight hysteresis. At the same pressures however, X-ray diffraction shows no evidence of a phase transition, suggesting that a rearrangement of CH4 configuration occurs, without any effect on the tetrahedral framework.
The stability of onoratoite, Sb8O11Cl2, in the supergene environment
- Adam J. Roper, Peter Leverett, Timothy D. Murphy, Peter A. Williams
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 1671-1675
-
- Article
- Export citation
-
Synthesis and solubility studies of onoratoite have been undertaken to determine the role of this rare secondary phase in the immobilization of Sb and the conditions responsible for its formation in the supergene zone. Solubility studies were undertaken at 298.15 K. A value of ΔGfθ (Sb8O11Cl2, s, 298.15 K) = –2576 ±12 kJ mol–1 was derived. Calculations involving sénarmontite, Sb2O3, klebelsbergite, Sb4O4SO4(OH)2 and schafarzikite, FeSb2O4, show that onoratoite is a thermodynamically stable phase only at negligible activities of SO42–(aq) and low activities of Fe2+(aq), at low pH and very high activities of Cl–(aq). This explains why onoratoite is such a rare secondary phase and why it cannot exert any significant influence on the dispersion of Sb in the supergene environment.
High-pressure synthesis and application of a 13C diamond pressure sensor for experiments in a hydrothermal diamond anvil cell
- Nadezda Chertkova, Shigeru Yamashita, Eiji Ito, Akira Shimojuku
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 1677-1685
-
- Article
- Export citation
-
Polycrystalline, cubic 13C diamond was synthesized from amorphous carbon in the Kawai-type multianvil apparatus at 21 GPa and at a temperature greater than 2350ºC. The polycrystalline diamond was homogeneous with a small grain size (10–20 μm) and a sharp Raman peak, and thereby was suitable as a pressure sensor for the experiments in a hydrothermal diamond anvil cell. Pressure- and temperature-dependence of the Raman shift of the synthesized 13C diamond was investigated in situ at simultaneous high pressures and high temperatures in the hydrothermal diamond anvil cell, using the ruby fluorescence line, quartz Raman shift and H2O phase transitions as pressure references. It was observed that the frequency shift with pressure is independent of temperature and vice versa up to 500ºC and 4.2 GPa. The present study indicates that the 13C diamond Raman shift can be used for pressure determination with an accuracy better than ±0.3 GPa under the conditions examined.
Unique thallium mineralization in the fumaroles of Tolbachik volcano, Kamchatka Peninsula, Russia. I. Markhininite, TlBi(SO4)2
- Oleg I. Siidra, Lidiya P. Vergasova, Sergey V. Krivovichev, Yuri L. Kretser, Anatoly N. Zaitsev, Stanislav K. Filatov
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 1687-1698
-
- Article
- Export citation
-
Markhininite, ideally TlBi(SO4)2, was found in a fumarole of the 1st cinder cone of the North Breach of the Great Fissure Tolbachik volcano eruption (1975–1976), Kamchatka Peninsula, Russia. Markhininite occurs as white pseudohexagonal plates associated with shcherbinaite, pauflerite, bobjonesite, karpovite, evdokimovite and microcrystalline Mg, Al, Fe and Na sulfates. Markhininite is triclinic, P
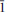 , a = 7.378(3), b = 10.657(3), c = 10.657(3) Å , α = 61.31(3), β = 70.964(7), γ = 70.964(7)º, V = 680.2(4) Å3, Z = 4 (from single-crystal diffraction data). The eight strongest lines of the X-ray powder diffraction pattern are (I/d/hkl): 68/4.264/111, 100/3.441/113, 35/3.350/222, 24/3.125/122, 23/3.054/202, 45/2.717/022, 20/2.217/331, 34/2.114/204. Chemical composition determined by electron microprobe analysis is (wt.%): Tl2O 35.41, Bi2O3 38.91, SO3 25.19, total 99.51. The empirical formula based on 8 O a.p.f.u. is Tl1.04Bi1.05S1.97O8. The simplified formula is TlBi(SO4)2, which requires Tl2O 35.08, Bi2O3 38.48, SO3 26.44, total 100.00 wt.%. The crystal structure was solved by direct methods and refined to R1 = 0.055 on the basis of 1425 independent observed reflections. The structure contains four Tl+ and two Bi3+ sites in holodirected symmetrical coordination. BiO8 tetragonal antiprisms and SO4 tetrahedra in markhininite share common O atoms to produce [Bi(SO4)2]– layers of the yavapaiite type. The layers are parallel to (111) and linked together through interlayer Tl+ cations. The mineral is named in honour of Professor Yevgeniy Konstantinovich Markhinin (b. 1926), Institute of Volcanology, Russian Academy of Sciences, Kamchatka peninsula, Russia, in recognition of his contributions to volcanology. Markhininite is the first oxysalt compound that contains both Tl and Bi in an ordered crystal structure.
, a = 7.378(3), b = 10.657(3), c = 10.657(3) Å , α = 61.31(3), β = 70.964(7), γ = 70.964(7)º, V = 680.2(4) Å3, Z = 4 (from single-crystal diffraction data). The eight strongest lines of the X-ray powder diffraction pattern are (I/d/hkl): 68/4.264/111, 100/3.441/113, 35/3.350/222, 24/3.125/122, 23/3.054/202, 45/2.717/022, 20/2.217/331, 34/2.114/204. Chemical composition determined by electron microprobe analysis is (wt.%): Tl2O 35.41, Bi2O3 38.91, SO3 25.19, total 99.51. The empirical formula based on 8 O a.p.f.u. is Tl1.04Bi1.05S1.97O8. The simplified formula is TlBi(SO4)2, which requires Tl2O 35.08, Bi2O3 38.48, SO3 26.44, total 100.00 wt.%. The crystal structure was solved by direct methods and refined to R1 = 0.055 on the basis of 1425 independent observed reflections. The structure contains four Tl+ and two Bi3+ sites in holodirected symmetrical coordination. BiO8 tetragonal antiprisms and SO4 tetrahedra in markhininite share common O atoms to produce [Bi(SO4)2]– layers of the yavapaiite type. The layers are parallel to (111) and linked together through interlayer Tl+ cations. The mineral is named in honour of Professor Yevgeniy Konstantinovich Markhinin (b. 1926), Institute of Volcanology, Russian Academy of Sciences, Kamchatka peninsula, Russia, in recognition of his contributions to volcanology. Markhininite is the first oxysalt compound that contains both Tl and Bi in an ordered crystal structure.
Unique thallium mineralization in the fumaroles of the Tolbachik volcano, Kamchatka Peninsula, Russia. II. Karpovite, Tl2VO(SO4)2(H2O)
- Oleg I. Siidra, Lidiya P. Vergasova, Yuri L. Kretser, Yuri S. Polekhovsky, Stanislav K. Filatov, Sergey V. Krivovichev
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 1699-1709
-
- Article
- Export citation
-
Karpovite, ideally Tl2VO(SO4)2(H2O), was found in a fumarole of the 1st cinder cone of the North Breach of the Great Fissure Tolbachik volcano eruption (1975–1976), Kamchatka Peninsula, Russia. Karpovite occurs as bundles of white, needle-like crystals associated with shcherbinaite, pauflerite, bobjonesite, markhininite, evdokimovite and microcrystalline Mg, Al, Fe and Na sulfates. Karpovite is monoclinic, P21, a = 4.6524(4), b = 11.0757(9), c = 9.3876(7) Å , β = 98.353(2)º, V = 478.60(7) Å3, Z = 2 (from single-crystal diffraction data). The eight strongest lines of the X-ray powder diffraction pattern are (I/d/hkl): 64/4.289/012, 81/4.253/110, 38/3.683/111, 47/3.557/022, 100/3.438/
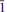 21, 52/2.982/013, 59/2.945/112, 54/2.354/132. The chemical composition determined by the electron microprobe analysis is (wt.%) Tl2O 61.43, VO2 11.53, SO3 23.55, H2O 2.61, total 99.12. The empirical formula (calculated on the basis of 10 O a.p.f.u.) is Tl2.00V0.96S2.03O9(H2O). The simplified formula of karpovite is Tl2VO(SO4)2(H2O), which requires Tl2O 61.93, VO2 12.09, SO3 23.34, H2O 2.62 total 100.00 wt.%. The crystal structure was solved by direct methods and refined to R1 = 0.026 for 4196 independent observed reflections. The structure contains two symmetrically independent Tl+ sites, one V4+ site and two S6+ sites. VO5H2O octahedra and SO4 tetrahedra link together by sharing corners to form kröhnkite-type stripes parallel to the a axis with their planes oriented parallel to (021) and (02
21, 52/2.982/013, 59/2.945/112, 54/2.354/132. The chemical composition determined by the electron microprobe analysis is (wt.%) Tl2O 61.43, VO2 11.53, SO3 23.55, H2O 2.61, total 99.12. The empirical formula (calculated on the basis of 10 O a.p.f.u.) is Tl2.00V0.96S2.03O9(H2O). The simplified formula of karpovite is Tl2VO(SO4)2(H2O), which requires Tl2O 61.93, VO2 12.09, SO3 23.34, H2O 2.62 total 100.00 wt.%. The crystal structure was solved by direct methods and refined to R1 = 0.026 for 4196 independent observed reflections. The structure contains two symmetrically independent Tl+ sites, one V4+ site and two S6+ sites. VO5H2O octahedra and SO4 tetrahedra link together by sharing corners to form kröhnkite-type stripes parallel to the a axis with their planes oriented parallel to (021) and (02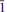 ). Tl+ cations are located between the chains, linked into a three-dimensional structure. The new mineral is named in honour of Professor Gennadii Alexandrovich Karpov (b. 1938), volcanologist at the Institute of Volcanology, Russian Academy of Sciences, Petropavlovsk-Kamchatskii, Kamchatka Peninsula, Russia.
). Tl+ cations are located between the chains, linked into a three-dimensional structure. The new mineral is named in honour of Professor Gennadii Alexandrovich Karpov (b. 1938), volcanologist at the Institute of Volcanology, Russian Academy of Sciences, Petropavlovsk-Kamchatskii, Kamchatka Peninsula, Russia.
Unique thallium mineralization in the fumaroles of Tolbachik volcano, Kamchatka Peninsula, Russia. III. Evdokimovite, Tl4(VO)3(SO4)5(H2O)5
- Oleg I. Siidra, Lidiya P. Vergasova, Yuri L. Kretser, Yuri S. Polekhovsky, Stanislav K. Filatov, Sergey V. Krivovichev
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 1711-1724
-
- Article
- Export citation
-
Evdokimovite, ideally Tl4(VO)3(SO4)5(H2O)5, was found in a fumarole of the 1st cinder cone of the North Breach of the Great Fissure Tolbachik volcano eruption of 1975–1976, Kamchatka Peninsula, Russia. Evdokimovite occurs as thin, colourless needles up to 0.09 mm long associated with shcherbinaite, pauflerite, bobjonesite, markhininite, karpovite and microcrystalline Mg, Al, Fe and Na sulfates. Evdokimovite is monoclinic, P21/n, a = 6.2958(14), b = 10.110(2), c = 39.426(11) Å , β = 90.347(6)º, V = 2509.4(10) Å3 and Z = 4 (from single-crystal diffraction data). The eight strongest lines of the powder X-ray diffraction pattern are (I/d/hkl): 57/9.793/011, 100/8.014/013, 26/6.580/006, 19/ 4.011/026, 29/3.621/118, 44/3.522/125, 19/3.010/036, 21/2.974/212. Chemical composition determined by the electron microprobe analysis is (wt.%): Tl2O 55.40, VO2 14.92, SO3 25.83, H2O 5.75, total 101.90. The empirical formula for evdokimovite calculated on the basis of (Tl + V + S) = 12 a.p.f.u. is Tl4.10V2.83S5.07H10.00O27.94. The simplified formula is Tl4(VO)3(SO4)5(H2O)5. The crystal structure was solved by direct methods and refined to R1 = 0.11 on the basis of 3660 independent observed reflections. V4+O6 octahedra and SO4 tetrahedra share common corners to form two types of vanadyl-sulfate chains, [(VO)(H2O)2(SO4)2]2– and [(VO)2(H2O)3(SO4)3]2–. Thallium atoms are located in between the chains. The structure can be described as a stacking of layers of two types, A and B. The A layer contains [(VO)2(H2O)3(SO4)3]2– chains and the Tl2 and Tl3 atoms, whereas the B layer contains [(VO)(H2O)2(SO4)2]2– chains and the Tl1 atoms. Stacking of the layers can be described as ...A’*BAA’B*A*..., where A and A’ denote A layers with opposite orientations of the [(VO)2(H2O)3(SO4)3]2– chains, and the A* and B* layers are rotated by 180º relative to the A and B layers, respectively. [(VO)2(H2O)3(SO4)3]2– chains are modulated and are arranged to form elliptical tunnels hosting disordered Tl(4), Tl(4A) and Tl(4B) sites. The new mineral is named in honour of Professor Mikhail Dmitrievich Evdokimov (1940–2010), formerly of the Department of Mineralogy, St Petersburg State University, for his contributions to mineralogy and petrology, and especially for teaching mineralogy to several generations of students at the University. Evdokimovite is the most complex V4+ sulfate known to date with structural information amounting to 1130 bits per unit cell, which places evdokimovite among minerals with the complexity of the vesuvianite group.
Functional twin boundaries and tweed microstructures: a comparison between minerals and device materials
- Oktay Aktas, Ekhard K. H. Salje
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 1725-1741
-
- Article
- Export citation
-
In ferroelastic materials, the existence of degenerate strain states leads to the formation of nanoscale microstructures, such as domain boundaries (twin walls) and tweed. As the symmetry properties of microstructures differ from those of the bulk, they may dramatically change the macroscopic properties of a crystal. In addition, they are likely to have functional properties (ferroelecricity, piezoelectricity, magnetism, conductivity and rapid chemical transport) that are absent in the bulk. The existence of functional properties of twin walls, along with the advances in nano-scale characterization, has opened the door to domain boundary engineering, which aims to use domain boundaries as active elements in device materials. Hence, this relatively new field puts ferroelastic twin walls and possibly tweed at the heart of future electronic devices. Ferroelasticity is very common among minerals. Similar to manmade materials, the same crystallographic principles apply, which means that there are many minerals that await discovery for their functional properties. Thus, this review aims to raise attention to the discovery of minerals with functional microstructures. The current development of functional twin boundaries and tweed structures in physics and materials sciences is compared with the traditional observation of such structures in minerals. With an emphasis on chemical transport and piezoelectric/ ferroelectric behaviour, examples of functional microstructures are given from both man-made materials and minerals in addition to a discussion of the origin of polar twin walls and the introduction of a recent experimental technique, resonant piezoelectric spectroscopy (RPS), for their discovery.
Lukkulaisvaaraite, Pd14Ag2Te9, a new mineral from Lukkulaisvaara intrusion, northern Russian Karelia, Russia
- A. Vymazalová, T. L. Grokhovskaya, F. Laufek, V. A. Rassulov
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 1743-1754
-
- Article
- Export citation
-
Lukkulaisvaaraite, Pd14Ag2Te9, is a new platinum-group mineral discovered in the Lukkulaisvaara intrusion, northern Russian Karelia, Russia. In polished section crystals are ~40 mm across, rimmed by tulameenite and accompanied to varying degrees by telargpalite and Bi-rich kotulskite. Lukkulaisvaaraite is brittle, has a metallic lustre and a grey streak. Values of VHN20 fall between 339 and 371 kg mm–2, with a mean value of 355 kg mm–2, corresponding to a Mohs hardness of ~4. In plane-polarized light, lukkulaisvaaraite is light grey with a brownish tinge, has strong bireflectance, light brownish-grey to greyish-brown pleochroism and distinct to strong anisotropy; it exhibits no internal reflections. Reflectance values of lukkulaisvaaraite in air (R1, R2, in %) are: 40.9, 48.3 at 470 nm, 47.6, 56.4 at 546 nm, 52.1, 61.0 at 589 nm and 57.5, 65.2 at 650 nm. Five electron microprobe analyses of natural lukkulaisvaaraite gave the average composition Pd 52.17, Ag 7.03 and Te 40.36, total 99.61 wt.%, corresponding to the empirical formula Pd14.05Ag1.88Te9.06 based on 25 atoms; the average of nine analyses on synthetic lukkulaisvaaraite is Pd 52.13, Ag 7.31 and Te 40.58, total 100.02 wt.%, corresponding to Pd13.99Ag1.93Te9.08. The mineral is tetragonal, space group I4/m, with a = 8.9599(6), c = 11.822(1) Å , V = 949.1(1) Å3 and Z = 2. The crystal structure was solved and refined from the powder X-ray diffraction (XRD) data of synthetic Pd14Ag2Te9. Lukkulaisvaaraite has a unique structure type and shows similarities to that of sopcheite (Ag4Pd3Te4) and palladseite (Pd17Se15). The strongest lines in the powder XRD pattern of synthetic lukkulaisvaaraite [d(Å),I,hkl] are: 2.8323(58)(130,310), 2.8088(92),(213), 2.5542(66)(312), 2.4312(41)(321,231), 2.1367(57)(411,141), 2.1015(52)(233,323), 2.0449(100)(314), 2.0031(63)(420,240), 1.9700(30)(006), 1.4049(30)(246,426), 1.3187(36)(543,453). The mineral is named for the type locality, the Lukkulaisvaara intrusion in Russian Karelia.
Barlowite, Cu4FBr(OH)6, a new mineral isotructural with claringbullite: description and crystal structure
- Peter Elliott, Mark A. Cooper, Allan Pring
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 1755-1762
-
- Article
- Export citation
-
The new mineral species barlowite, ideally Cu4FBr(OH)6, has been found at the Great Australia mine, Cloncurry, Queensland, Australia. It is the Br and F analogue of claringbullite. Barlowite forms thin blue, platy, hexagonal crystals up to 0.5 mm wide in a cuprite-quartz-goethite matrix associated with gerhardtite and brochantite. Crystals are transparent to translucent with a vitreous lustre. The streak is sky blue. The Mohs hardness is 2–2.5. The tenacity is brittle, the fracture is irregular and there is one perfect cleavage on {001}. Density could not be measured; the mineral sinks in the heaviest liquid available, diluted Clerici solution (D &3.8 g/cm3). The density calculated from the empirical formula is 4.21 g/cm3. Crystals are readily soluble in cold dilute HCl. The mineral is optically non-pleochroic and uniaxial (–). The following optical constants measured in white light vary slightly suggesting a small variation in the proportions of F, Cl and Br: ω 1.840(4)–1.845(4) and ε 1.833(4)–1.840(4). The empirical formula, calculated on the basis of 18 oxygen atoms and H2O calculated to achieve 8 anions and charge balance, is Cu4.00F1.11Br0.95Cl0.09(OH)5.85. Barlowite is hexagonal, space group P63/mmc, a = 6.6786(2), c = 9.2744(3) Å , V = 358.251(19) Å3, Z = 2. The five strongest lines in the powder X-ray diffraction pattern are [d(Å )(I)(hkl)]: 5.790(100)(010); 2.889(40)(020); 2.707(55)(112); 2.452(40)(022); 1.668(30)(220).
Crystal structure and revised chemical formula for burckhardtite, Pb2(Fe3+Te6+)[AlSi3O8]O6: a double-sheet silicate with intercalated phyllotellurate layers
- Andrew G. Christy, Anthony R. Kampf, Stuart J. Mills, Robert M. Housley, Brent Thorne
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 1763-1773
-
- Article
- Export citation
-
The crystal structure of burckhardite from the type locality, Moctezuma, Sonora, Mexico, has been refined to R1 = 0.0362 and wR2 = 0.0370 for 215 reflections with I > 2σ(I). Burckhardtite is trigonal, space group P
 1m, with the unit-cell parameters a = 5.2566(5) Å , c = 13.0221(10) Å , V = 311.62(5) Å3 and Z = 1 for the ideal formula unit Pb2(Fe3+Te6+)[AlSi3O8]O6. There is no long-range order of (Fe3+, Te6+) or (Al3+, Si4+). New microprobe data were used to estimate site scattering factors, and Raman spectroscopic data showed no evidence of O–H stretching bands. Burckhardtite is not closely related to the micas, as supposed previously, but is a double-sheet silicate in which the aluminosilicate anion resembles that of minerals such as cymrite and kampfite. The [(Fe3+Te6+)O6]3– part of the structure is not bonded directly to the aluminosilicate layer, but forms a discrete anionic phyllotellurate layer that alternates with the [AlSi3O8]– double sheets. Similar phyllotellurate layers are known from several synthetic phases. In burckhardtite, Pb2+ cations intercalate between phyllosilicate and phyllotellurate layers, forming a Pb2[FeTeO6] module that is topologically similar to a slab of the structure of rosiaite, Pb[Sb2O6]. The crystal symmetry, structure, classification as a double-sheet silicate and chemical formula, including the determination of the 6+ valence of Te and absence of essential H2O, are all new findings for the mineral.
1m, with the unit-cell parameters a = 5.2566(5) Å , c = 13.0221(10) Å , V = 311.62(5) Å3 and Z = 1 for the ideal formula unit Pb2(Fe3+Te6+)[AlSi3O8]O6. There is no long-range order of (Fe3+, Te6+) or (Al3+, Si4+). New microprobe data were used to estimate site scattering factors, and Raman spectroscopic data showed no evidence of O–H stretching bands. Burckhardtite is not closely related to the micas, as supposed previously, but is a double-sheet silicate in which the aluminosilicate anion resembles that of minerals such as cymrite and kampfite. The [(Fe3+Te6+)O6]3– part of the structure is not bonded directly to the aluminosilicate layer, but forms a discrete anionic phyllotellurate layer that alternates with the [AlSi3O8]– double sheets. Similar phyllotellurate layers are known from several synthetic phases. In burckhardtite, Pb2+ cations intercalate between phyllosilicate and phyllotellurate layers, forming a Pb2[FeTeO6] module that is topologically similar to a slab of the structure of rosiaite, Pb[Sb2O6]. The crystal symmetry, structure, classification as a double-sheet silicate and chemical formula, including the determination of the 6+ valence of Te and absence of essential H2O, are all new findings for the mineral.
Lead-antimony sulfosalts from Tuscany (Italy). XVI. Carducciite, (AgSb)Pb6(As,Sb)8S20, a new Sb-rich derivative of rathite from the Pollone mine, Valdicastello Carducci: occurrence and crystal structure
- Cristian Biagioni, Paolo Orlandi, Yves Moëlo, Luca Bindi
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 1775-1793
-
- Article
- Export citation
-
The new mineral species carducciite, (AgSb)Pb6(As,Sb)8S20, has been discovered in the baryte-pyrite- (Pb-Ag-Zn) deposit of the Pollone mine, near Valdicastello Carducci, Apuan Alps, Tuscany, Italy. It occurs as black metallic prismatic crystals, up to 0.5 mm long, associated with pyrite and sterryite. Its Vickers hardness (VHN10) is 61 kg/mm2 (range: 52–66), corresponding to a Mohs hardness of ~2½–3. In reflected light, carducciite is dark grey in colour, moderately bireflectant; internal reflections are very weak and deep red in colour. Reflectance percentages for the four COM wavelengths [Rmin, Rmax (%) (λ)] are: 35.8, 40.8 (471.1 nm), 33.7, 39.0 (548.3 nm), 32.7, 37.6 (586.6 nm) and 30.4, 35.1 (652.3 nm). Electron microprobe analysis gives (wt.% – mean of six analyses): Ag 3.55(12), Tl 0.13(3), Pb 41.90(42), Sb 17.79(19), As 12.41(14), S 22.10(17), total 97.9(6). On the basis of ΣMe = 16 a.p.f.u., the chemical formula is Ag0.96Tl0.02Pb5.91As4.84Sb4.27S20.14. The main diffraction lines, corresponding to multiple hkl indices, are (relative visual intensity): 3.689 (s), 3.416 (s), 3.125 (s), 2.989 (s), 2.894 (s), 2.753 (vs), 2.250 (s). The crystal-structure study gives a monoclinic unit cell, space group P21/c, with a 8.4909(3), b 8.0227(3), c 25.3957(9) Å, β 100.382(2)°, V 1701.63(11) Å3, Z = 2. The crystal structure has been solved and refined to a final R1 = 0.063 on the basis of 4137 observed reflections. It can be described within the framework of the sartorite homologous series, as formed by chemically twinned layers of the dufrénoysite type. The simplified idealized structural formula, based on 20 sulfur atoms, can ideally be written as (AgSb)Pb6(As,Sb)Σ=8S20. Carducciite is an (Ag,Sb)-rich homeotype of dufrénoysite, stabilized by the complete coupled substitution 2 Pb2+ = Ag+ + Sb3+ on a specific site of the crystal structure. Together with barikaite, it belongs to the rathite sub-group of P21/c homeotypes of dufrénoysite, of which the crystal chemistry is discussed. The distribution of Ag, coupled with As or Sb on specific sites, appears to be the main criterion for the distinction between the three species of this sub-group.
Book Reviews
Roy E. Starkey Crystal Mountains: Minerals of the Cairngorms. Self-published. May be ordered online from www.britishmineralogy.com, 2014, 178 pp. ISBN 978-0-9930182-1-3. List price £25 + P&P.
- Ian Parsons
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 1795-1797
-
- Article
- Export citation

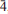 3
3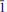 , a = 6.4271(4), b = 7.6585(4), c = 8.2249(3) Å , α = 98.396(4), β = 112.420(5), γ = 98.397(5)º, V = 361.11(3) Å
, a = 6.4271(4), b = 7.6585(4), c = 8.2249(3) Å , α = 98.396(4), β = 112.420(5), γ = 98.397(5)º, V = 361.11(3) Å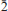 2), 2.777(98)(
2), 2.777(98)(
 11), 2.83 (30) (020), 2.931 (25) (302), 2.502 (25) (
11), 2.83 (30) (020), 2.931 (25) (302), 2.502 (25) (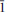 , a = 7.378(3), b = 10.657(3), c = 10.657(3) Å , α = 61.31(3), β = 70.964(7), γ = 70.964(7)º, V = 680.2(4) Å
, a = 7.378(3), b = 10.657(3), c = 10.657(3) Å , α = 61.31(3), β = 70.964(7), γ = 70.964(7)º, V = 680.2(4) Å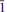 21, 52/2.982/013, 59/2.945/112, 54/2.354/132. The chemical composition determined by the electron microprobe analysis is (wt.%) Tl
21, 52/2.982/013, 59/2.945/112, 54/2.354/132. The chemical composition determined by the electron microprobe analysis is (wt.%) Tl 1m, with the unit-cell parameters a = 5.2566(5) Å , c = 13.0221(10) Å , V = 311.62(5) Å
1m, with the unit-cell parameters a = 5.2566(5) Å , c = 13.0221(10) Å , V = 311.62(5) Å