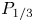153 results
Simulations of radiatively cooled magnetic reconnection driven by pulsed power
-
- Journal:
- Journal of Plasma Physics / Volume 90 / Issue 2 / April 2024
- Published online by Cambridge University Press:
- 19 April 2024, 905900215
-
- Article
- Export citation
Evaluation of critical weed-free period for three sweetpotato (Ipomoea batatas) cultivars
-
- Journal:
- Weed Science , FirstView
- Published online by Cambridge University Press:
- 01 March 2024, pp. 1-8
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Reduced morbidity with early surgical ligation of patent ductus arteriosus in extremely low birth weight infants: a retrospective single-centre study
-
- Journal:
- Cardiology in the Young , First View
- Published online by Cambridge University Press:
- 10 January 2024, pp. 1-6
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Features of immunometabolic depression as predictors of antidepressant treatment outcomes: pooled analysis of four clinical trials
-
- Journal:
- The British Journal of Psychiatry / Volume 224 / Issue 3 / March 2024
- Published online by Cambridge University Press:
- 22 December 2023, pp. 89-97
- Print publication:
- March 2024
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
1 Social Brain Network Connectivity Relates to Social and Adaptive Outcomes Following Pediatric Traumatic Brain Injury
-
- Journal:
- Journal of the International Neuropsychological Society / Volume 29 / Issue s1 / November 2023
- Published online by Cambridge University Press:
- 21 December 2023, pp. 509-510
-
- Article
-
- You have access
- Export citation
What Can State Medical Boards Do to Effectively Address Serious Ethical Violations?
-
- Journal:
- Journal of Law, Medicine & Ethics / Volume 51 / Issue 4 / Winter 2023
- Published online by Cambridge University Press:
- 13 March 2024, pp. 941-953
- Print publication:
- Winter 2023
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
17 - Jazz and the American Songbook
- from Part IV - Literary Genres
-
-
- Book:
- Jazz and American Culture
- Published online:
- 09 November 2023
- Print publication:
- 30 November 2023, pp 262-274
-
- Chapter
- Export citation
Independent and joint contributions of physical disability and chronic pain to incident opioid use disorder and opioid overdose among Medicaid patients
-
- Journal:
- Psychological Medicine / Volume 54 / Issue 7 / May 2024
- Published online by Cambridge University Press:
- 17 November 2023, pp. 1419-1430
-
- Article
- Export citation
Design and implementation of a digital site-less clinical study of serial rapid antigen testing to identify asymptomatic SARS-CoV-2 infection
-
- Journal:
- Journal of Clinical and Translational Science / Volume 7 / Issue 1 / 2023
- Published online by Cambridge University Press:
- 10 May 2023, e120
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Ultra-processed foods in a rural Ecuadorian community: associations with child anthropometry and bone maturation
-
- Journal:
- British Journal of Nutrition / Volume 130 / Issue 9 / 14 November 2023
- Published online by Cambridge University Press:
- 13 March 2023, pp. 1609-1624
- Print publication:
- 14 November 2023
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Impact of the COVID-19 Pandemic on Stroke Subtype Presentation in Patients Without COVID-19 Infection
-
- Journal:
- Canadian Journal of Neurological Sciences / Volume 51 / Issue 1 / January 2024
- Published online by Cambridge University Press:
- 30 January 2023, pp. 78-86
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Chapter 7 - “It’s Complicated”: The Intersect Between Psychiatric Disorders and Infertility
- from II - Therapeutic Approaches
-
-
- Book:
- Fertility Counseling: Clinical Guide
- Published online:
- 24 November 2022
- Print publication:
- 08 December 2022, pp 79-87
-
- Chapter
- Export citation
Chapter 7 - “It’s Complicated”: The Intersect Between Psychiatric Illness and Infertility
- from Part II - Therapeutic Approaches
-
-
- Book:
- Fertility Counseling: Case Studies
- Published online:
- 24 November 2022
- Print publication:
- 08 December 2022, pp 38-43
-
- Chapter
- Export citation
Recent Deaths
-
- Journal:
- Latin American Research Review / Volume 5 / Issue 3 / Fall 1970
- Published online by Cambridge University Press:
- 24 October 2022, p. 182
-
- Article
-
- You have access
- Export citation
A dominance analysis of subjective cognitive complaint comorbidities in former professional football players with and without mild cognitive impairment
-
- Journal:
- Journal of the International Neuropsychological Society / Volume 29 / Issue 6 / July 2023
- Published online by Cambridge University Press:
- 30 August 2022, pp. 582-593
-
- Article
- Export citation
Creation and impact of containment units with high-risk zones during the coronavirus disease 2019 (COVID-19) pandemic
- Part of
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 6 / June 2023
- Published online by Cambridge University Press:
- 17 June 2022, pp. 908-914
- Print publication:
- June 2023
-
- Article
- Export citation
6 - Chronic conditions and multimorbidity: skill-mix innovations for enhanced quality and coordination of care
-
-
- Book:
- Skill-mix Innovation, Effectiveness and Implementation
- Published online:
- 08 April 2022
- Print publication:
- 09 June 2022, pp 152-220
-
- Chapter
-
- You have access
- Open access
- HTML
- Export citation
Comparison of the performance of a clinical classification tree versus clinical gestalt in predicting sepsis with extended-spectrum beta-lactamase–producing gram-negative rods
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue 1 / 2022
- Published online by Cambridge University Press:
- 07 March 2022, e35
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Decisions to attend holiday gatherings during COVID-19 and engagement in key prevention strategies: United States, January 2021
-
- Journal:
- Epidemiology & Infection / Volume 150 / 2022
- Published online by Cambridge University Press:
- 09 February 2022, e32
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Characterisation of age and polarity at onset in bipolar disorder
-
- Journal:
- The British Journal of Psychiatry / Volume 219 / Issue 6 / December 2021
- Published online by Cambridge University Press:
- 25 August 2021, pp. 659-669
- Print publication:
- December 2021
-
- Article
-
- You have access
- Open access
- HTML
- Export citation