In recent years the placebo effect has returned to the limelight. The reasons for this are complex, but they probably include the increased use of neuroimaging, the clinical improvement noted in placebo groups in trials of antidepressants, and the growing public and research interest in alternative medicine. Yet placebos have been an enigmatic phenomenon of medical science for the past 50 years. Lauded as the gold standard against which all therapies should be compared, their effects dismissed as the murky non-specific noise that muddies our objective understanding of therapeutic processes, they are both champion and pariah. Placebos are paradoxical, able to be simultaneously the most tested intervention in randomised controlled trials and yet the least understood of all therapies; the most quoted treatment in medical science and yet notoriously elusive to definition. This article explores the placebo effect’s history, definition, efficacy and purported mechanisms, particularly in relation to psychiatry.
Methodology
I gathered information from a methodological and comprehensive search of several databases (over 40 000 titles or abstracts were scanned), a hand-search of bibliographies, personal contacts with experts, and a number of books (Reference BrodyBrody, 1980; Reference White, Tursky and SchwartzWhite et al, 1985; Reference HarringtonHarrington, 1999; Reference BrodyBrody, 2000; Reference PetersPeters, 2001; Reference Guess, Kleinman and KusekGuess et al, 2002; Reference MoermanMoerman, 2002; Reference EvansEvans, 2003). In keeping with the nature of an APT article, I have not exhaustively referenced every result discussed here. Instead, I have cited only key studies or reviews. A full list of references is available from me on request.
Definitions
Although placebos are routinely referred to, they and their effects are elusively hard to define. However, for research and clinical purposes, a consensus definition of the placebo effect is important.
Some argue, pragmatically, that the placebo effect is the ‘therapeutic effect of a placebo administration’ (Reference Kienle, Kiene and PetersKienle & Kiene, 2001). However, this leads to a tautological quagmire. Another pragmatic definition is the ‘difference in outcome between a placebo treated group and an untreated control group in an unbiased experiment’ (Reference Ernst and PetersErnst, 2001). Although this is a powerful definition for research paradigms, it arguably lacks much explanatory quality and feels like a scientific cop-out.
Reference WolfWolf (1959) described the placebo effect as ’any effect attributable to a pill, potion, or procedure, but not to its pharmacodynamic or specific properties’. However, as Reference Kienle, Kiene and PetersKienle & Kiene (2001) argue, the ‘non-specific’ cannot be determined positively, but only negatively, by exclusion of what is specific in the case in question. To define something usually involves reference to its specific properties and qualities; but this definition of placebo effect aims to define what it is not. It is thus conceivable that, should the mechanisms of the placebo effect be elucidated, the term itself would become redundant.
Some define the placebo effect by reference to its purported underlying mechanisms, such as expectation, transference and conditioning. Reference BrodyBrody (1980) expands the definition to include processes such as the influence of doctor attitude and enthusiasm on the effect of active medications. Likewise, Reference MoermanMoerman (2002) equates the placebo effect to a ‘meaning response’, referring to the wider ‘psychological and physiological effects of meaning in the treatment of illness’. But as Reference Shapiro and ShapiroShapiro & Shapiro (1997) observe, ‘none of the mechanisms has been experimentally verified as the major variable. An operational definition is therefore premature’.
The term ‘placebo’
Reference EvansEvans (2003) notes that the term originated from Psalm 116:9 of the 14th-century Latin bible, meaning ‘I will please’. Ironically, this is based on a typological error: the original Hebrew ethhallech, meaning ‘I shall walk [with the Lord in the land of the living]’, was mistranslated as euarestaso (‘I shall please … ’) in the Greek and thus to ‘placebo’ in the Latin (Reference MoermanMoerman, 2002). The psalm is used in Catholic vespers for the dead. In the past, some felt the priests’ fees for singing these prayers were exorbitant and their motives dubious; the word thus came to stand for words that were insincere but consoling.
The word has cropped up in English literature. In The Parson’s Tale, Chaucer cites it in the line ‘flatterers are the devil’s chatelaines for ever singing placebo’ (Reference EvansEvans, 2003). In his Essays, Civil and Moral: Of Counsel, Bacon wrote: ‘let [a king] beware how he opens his own inclination too much … for else counsellors will but take wind of him, and … sing him a song of placebo’ (Reference EvansEvans, 2003). It entered the medical lexicon in Hooper’s Medical Dictionary in 1811, which derided placebo as ’an epithet given to any medicine adopted to please rather than to benefit the patient’ (Reference BrodyBrody, 1980). (It is worth noting that few medicines then would have had any specific benefit, and many were downright toxic.)
Effectiveness of placebos: evidence from history to the modern era
One argument for the effectiveness of placebos is that history is replete with treatments that modern science now deems ineffective. Table 1 illustrates several examples from history (Reference Shapiro, Shapiro and MatarazzoShapiro & Shapiro, 1984; Reference HarringtonHarrington, 1999). A 1950s’ British Medical Journal editorial cited that placebos were given to 40% of patients in general practice (Anonymous, 1952).
Table 1 Psychiatric interventions from the past

| Intervention | Condition |
|---|---|
| Throwing patients into the sea for a ‘surprise bath’ | Melancholia and insanity |
| Mechanical machines that caused vigorous spinning | Insomnia, insanity and unruly behaviour |
| Hellebore, alcohol, opium, and quinine sulphate | Melancholia |
| Carbon dioxide inhalation | Psychosis and neurosis |
| Testicular vein blood serum, diencephalic radiation | Manic depression |
The placebo effect was first scientifically documented by Reference BeecherBeecher (1955), who found that soldiers in the Second World War experienced an analgesic effect with saline (given because of depleted morphine stocks). His clinical review of generally uncontrolled studies on placebo analgesia found that 30% of clinical effect could be attributable to the placebo effect.
Various studies have since sought to quantify the effect. Overall, it is lowest in double-blind studies, higher in single-blind and highest in uncontrolled studies (Reference Shapiro, Shapiro and MatarazzoShapiro & Shapiro, 1984). However, there is little consensus on the magnitude of the effect: one review found a positive placebo response in half of patients (Reference Shapiro and ShapiroShapiro & Shapiro, 1997), whereas another found no significant therapeutic placebo effect (Reference Kienle and KieneKienle & Kiene, 1996, Reference Kienle, Kiene and Peters2001).
Ultimately, the placebo effect can only be truly quantified when comparing placebo groups with no-treatment groups, thereby allowing for processes such as the Hawthorne effect, regression to the mean and the natural course of illness. Reference Hrobjartsson and GotzscheHrobjartsson & Gotzsche’s (2001) meta-analysis of 130 trials that included no-treatment groups concluded that there was little evidence of the placebo effect. However, the authors’ conclusions were based on averaging results from over 40 conditions, many of which one would not expect to be placebo responsive. The authors themselves point to various methodological confounders, and some of their data actually supported the presence of placebo effects.
In psychiatry, Reference Walsh, Seiddman and SyskoWalsh et al’s (2002) systematic review of 75 randomised controlled trials of anti-depressants demonstrated that up to half of patients on placebo improved significantly. Reference Kirsch and SapirsteinKirsch & Sapirstein (1998) analysed data from no-treatment groups in psychotherapy studies and compared them with placebo groups in antidepressant studies. Drug groups showed 33% more improvement over placebo, but placebo groups showed 200% improvement over no-treatment groups. They calculated that up to half of the drug effect may be attributable to placebo effect. Other studies have noted a 25–33% improvement in people with agoraphobia or panic disorder (Reference Coryell and NoyesCoryell & Noyes, 1988; Reference Rosenberg, Mellegard and RosenbergRosenberg et al, 1991).
What influences the placebo effect?
Placebo reactors
In the 1960s, research indicated that certain personality types and characteristics were more frequent in placebo responders: anxious, emotionally labile, suggestible, dependent on and dominated by others, and church-going (Reference JospeJospe, 1978). Overall, however, the results were a hotchpotch of inconsistencies: extroverted and introverted, outgoing and less socially confident, low IQ and more verbally skilled, well adjusted and submissive. Research was also blighted by small sample sizes and the use of healthy volunteers rather than ill patients. Methodological weaknesses included lack of baseline assessments, double-blinding and validated tools.
By the 1970s, the consensus was that no consistent ‘placebo responder’ was out there (Reference HarringtonHarrington, 1999). This view is furthered by the demonstration that placebo ‘run-in’ groups in antidepressant trials, whose ‘placebo reactors’ are eliminated before the trial commences, do not actually lower the placebo response rate or increase the drug–placebo difference (e.g. Reference Quitkin, McGrath and StewartQuitkin et al, 1998; Reference Lee, Walker and JakulLee et al, 2004). As Reference BrodyBrody (1980) remarked, ‘in more cases than not, an individual who responds to placebo in one set of circumstances will fail to respond in other circumstances’. However, a recent study has stoked the fires again. Reference Geers, Helfer and KosbabGeers et al(2005) evaluated placebo response in optimists compared with pessimists in an experimental paradigm using healthy volunteers, and found that both personality and situation affected response.
Practitioner qualities
Research assessing the practitioner characteristics that could influence the placebo effect also depicts a mixed picture, although enthusiasm for treatment, confidence, authority, empathy and warmth appear to increase the effect (Reference Crow, Gage and HampsonCrow et al, 1999; Reference HarringtonHarrington, 1999; Reference Ernst and PetersErnst, 2001). Most studies were conducted on placebo analgesia.
Nature of administration
In Reference Ernst and PetersErnst’s (2001) review, invasive, uncomfortable, sophisticated or painful interventions tended to enhance the placebo effect. In psychiatry, chlordiazepoxide administered in capsules was more efficacious than the same dose in tablet form for treating anxiety, phobias and insomnia (Reference Hussain and AhadHussain & Ahad, 1970).
Reference de Craen, Roos and de VriesDe Craen et al(1996) reviewed the literature on people’s perceptions on the influence of drug colour and the degree of influence colour actually had on clinical effect. Overall, four studies showed that people perceived blue as ‘depressant’ and orange and red as ‘stimulant’. Reference Cattaneo, Lucchelli and FilippuciCattaneo et al(1970) showed that men preferred orange placebos, but women blue, for pre-operative tranquillisers. Their later study showed that blue had a sedative effect on Italian women, but caused insomnia in Italian men (Reference Lucchelli, Cattaneo and ZattoniLucchelli et al, 1978). The authors speculated that Italian women may associate blue with the comforting vision of the Virgin Mary, whereas Italian men perhaps associated the same colour with the excitement of the Azzurri football team (the Sky Blues).
It is important to note that the studies above assessed the influence of colour on active medication, not placebos. However, an extrapolation to placebos serves to demonstrate that whatever processes underlie the placebo effect, they are not limited solely to placebos.
Drug names
One study compared four groups treated for headache, and found that the order of therapeutic effect was: placebo < ‘branded’ placebo < aspirin < branded aspirin (Reference Braithwaite and CooperBraithwaite & Cooper, 1981). That branding accentuated the clinical effect suggests that drug name holds particular meaning for patients (by virtue of the linguistic name itself, or via the patient’s experience of the drug name), and that ‘meaning’ itself can have a clinical impact.
The linguistic aspect of drug names was further explored by Reference SchonauerSchonauer (1994). Medical students and doctors evaluated various drug names (some fake) using a Likert-style questionnaire. The study concluded that two aspects of a drug’s name affected its perceived clinical effect: phonetic and semantic qualities. To illustrate with ‘Viagra’, one can suggest that its juxtaposition of hard-sounding syllables with the letter ‘a’ (phonetic quality) and its similarity with words such as ‘vigour’ and ‘Niagara’ (semantic quality) may act to enhance its meaning for an individual with impotency. Despite the fascinating speculation, Schonauer did not conduct research on whether this actually has any impact on clinical effect.
Mechanisms
By understanding the mechanisms that underlie the placebo effect (Box 1), a rationalised therapeutic approach can be developed for each condition to maximise the clinical effect.
Box 1 Purported mechanisms underlying the placebo effect
-
• Anxiety reduction
-
• Expectation
-
• Transference
-
• ‘Meaning effects’
-
• Conditioning
Anxiety reduction
The anxiety-ameliorating effect of a therapeutic encounter has been suggested as the primary mechanism underlying the placebo effect. Support comes from studies showing that saline placebo reduced affective but not sensory ratings of experimental pain (Reference Gracely, Bonica, Liebeskind and Albe-FessardGracely, 1979). Furthermore, recent neuroimaging research suggests that placebo analgesia may operate via anxiety relief, which is itself influenced by underlying cognitions (Reference Petrovic, Dietrich and FranssonPetrovic et al, 2005).
However, Reference Montgomery and KirschMontgomery & Kirsch (1996) administered placebo local anaesthetic to various specific body parts, and applied pain to these and non-treated parts. There was a difference in reported pain between treated and non-treated parts that could not have been due to the global changes in affect associated with anxiety reduction. Other inconsistencies also go against anxiety reduction as a mechanism: for example, anxiety can actually alleviate pain (in stress-induced analgesia) (Reference Shapiro, Shapiro and HarringtonShapiro & Shapiro, 1999).
Expectation
The cognitive–behavioural perspective of anxiety indicates that cognitive components are integral. The two studies described above reflect the role of expectation in anxiety reduction (Reference Montgomery and KirschMontgomery & Kirsch, 1996; Reference Petrovic, Dietrich and FranssonPetrovic et al, 2005). That the placebo effect generally corresponds with the person’s prior beliefs about the intervention suggests that expectation may also underlie the effect. Furthermore, neuroimaging research associates the placebo response with widespread frontal and prefrontal activation, consistent with the processing of expectation and executive function (Reference Benedetti, Mayberg and WagerBenedetti et al, 2005). Prior and current beliefs can be influenced by various factors (Box 2).
Box 2 Some factors influencing expectation
Past influences
-
• Direct experience
-
• of the intervention
-
• of the practitioner
-
• of the setting
-
-
• Experience of others’ accounts
-
• Media influences
-
• Culture
Current influences
-
• Logic
-
• Verbal information
-
• Non-verbal cues
-
• Attitude towards
-
• the intervention
-
• the practitioner
-
• the setting
-
-
• The practitioner’s
-
• attitude
-
• personality
-
• temperament
-
• experience
-
-
• Knowledge
Reference Crow, Gage and HampsonCrow et al(1999) systematically reviewed the literature on the influence of both ‘expectations of outcome’ and ‘self-efficacy expectation’ (i.e. the patient’s expectation that they themselves can influence outcome). Increasing self-efficacy expectations (e.g. skill training, relaxation techniques, providing information) before medical procedures or in managing chronic conditions can improve outcome. Moreover, optimistic outcome expectancies are more effective than cautious or sceptical expectancies at enhancing clinical outcome, especially on subjective self-report measures. However, most studies did not examine the direct relationship between expectancy and outcome, and few studies assessed pre-existing expectancies. Studies demonstrating the attenuation of post-operative pain and anxiety by relaxation techniques do not necessarily show that the placebo effect is mediated by expectation; many results cited could be explained by other mechanisms.
Reference Buckman and SabbaghBuckman & Sabbagh (1993) found that pregnant women believing they were receiving an anti-emetic for morning sickness felt less nauseous and their stomachs showed an objective decrease in nausea-associated movement. However, the participants had actually been given an emetic: the placebo effect, mediated by expectation, had reversed the pharmacological effects of the drug.
In Reference Smith and McDanielSmith & McDaniel’s study (1983), tuberculin from a green vial was applied monthly for 6 months to patients’ arms, causing inflammation from delayed hypersensitivity response; simultaneous administration of saline from a red vial to the other arm did not cause inflammation. The contents of the vials were then secretly reversed. However, tuberculin from the red vial produced less inflammation than when it was administered from the green vial. Saline from the green vial did not produce inflammation. The authors argued that expectation (that the red vial’s contents do not cause inflammation) had influenced the immune system.
In the case of depressive disorder, Reference Kirsch and HarringtonKirsch (1999) argues that the meta-cognitions experienced in depression are the most depressing aspect of depression, and that the placebo effect may operate by instilling the belief that the depression may subside. Furthermore, the efficacy gap in trials between ‘active placebos’, which mimic side-effects, and antidepressants is particularly small (Reference Moncrieff, Wessely and HardyMoncrieff et al, 2004). This implies that the patient’s very belief that the medication is active enhances clinical outcome.
An associated factor is desire; how strongly one wishes for an effect to occur may also influence clinical outcome (Reference Price and BarrellPrice & Barrell, 1984; Reference Jensen and KarolyJensen & Karoly, 1991). Desire and expectation are rarely independent; some argue that their combination amounts to ‘hope’, which in itself could be a potent mediator of the placebo effect: ‘When we have hope, it doesn’t mean that we think things will come out okay, it means that we think things will somehow make sense’ (Reference HarringtonHarrington, 1999).
When considering expectations, does one need to be conscious of such beliefs for them to count? As Reference MoermanMoerman (2002) observes, ‘there are many circumstances where it is plausible to imagine that you know of something in some way, but have no clear sense of it as being “knowledge”; it doesn’t form any particular “expectancy” ’. Furthermore, behavioural and psychoanalytic theories both conjecture that unconscious beliefs can influence our actions.
Transference
Transference is the unconscious projection of feelings, attitudes and wishes, initially formed towards a significant figure in early development, onto another person, such as the practitioner, in the individual’s current life. The placebo effect may operate as the therapeutic encounter invites the patient unconsciously to interact with the doctor in a simulacrum of a trustful parent–child relationship (Reference ForrerForrer, 1964).
It can, however, be argued that the transference mechanism can be subsumed either by expectation (transference equating to unconscious beliefs and attitudes) or by anxiety reduction (transference equating to release of unconscious psychic tension).
Meaning effects
To some, the word ‘expectation’ refers only to conscious beliefs. As cognitive processes may operate in the unconscious mind and perhaps also bear influence on the placebo effect, some researchers prefer a more inclusive terminology that encapsulates both conscious and unconscious processes. Therefore, terms such as ‘meaning’ or ‘context’ effects have been proposed. Reference Helman and PetersHelman (2001) divides such effects into ‘microcontext’ (the setting or physical environment in which the intervention takes place) and ‘macrocontext’ (the wider culture pertaining to the practitioner, patient and setting).
‘The doctor’s office, hospital ward, holy shrine or house of a traditional healer can be compared to a theatre set complete with scenery, props, costumes and script. This script, derived from the culture itself … tells them how to behave, how to experience the event and what to expect from it. It helps to validate the healer, and the power of their methods of healing’ (p. 5).
‘Meaning effects’ are so intrinsic to psychiatry that they are frequently taken for granted. They underlie patients’ clinical histories and presentations, form the root of psychotherapeutic theory and practice, and influence the process of clinical management. Meaning effects are, however, more explicit in transcultural psychiatry, whose essence concerns the very meanings that different cultures attribute to mental health and illness.
Experimental and anecdotal evidence of meaning effects are numerous (Reference MoermanMoerman, 2002). Examples include: surgical patients with a window view of a natural setting, who improved faster than a matched sample with a view of a wall (Reference UlrichUlrich, 1984); Cambodian witnesses to torture who became functionally blinded (Reference Rozee and Van BoemelRozee & Van Boemel, 1989; Reference Drinnan and MarmorDrinnan & Marmor, 1991); and the ‘postponement’ of death until after symbolically meaningful occasions such as festivals (Reference Phillips and SmithPhillips & Smith, 1990).
Many of the studies investigating factors that influence the placebo effect, such as drug name and colour, arguably demonstrate it as a meaning effect. Expectations regarding qualities such as names and colours are rarely explicitly expressed, and such unconscious processes appear more encapsulated by the term ‘meaning effect’ than ‘expectation effect’. Equating placebo effect with meaning effect appears a logical conclusion; after all, humans are beings of meaning. But what if placebo effects can occur in animals?
Conditioning
In an influential study, Reference Ader and CohenAder & Cohen (1975) administered pairings of cyclophosphamide injections and saccharin water in mice. After sufficient numbers of pairings, saccharin water alone could cause immunosuppression when injected into the mice. They argued that the animal’s immune system could be conditioned: the cyclophosphamide was deemed the unconditioned stimulus (UCS) and the saccharin, the conditioned stimulus (CS). Other studies, generally not masked, have also demonstrated conditioned immunosuppression (Reference HerrnsteinHerrnstein, 1962; Reference Moynihan, Koota and BrennerMoynihan et al, 1989).
Reference Ader and CohenAder & Cohen (1982) expanded on this using a clinical paradigm. Mice, genetically engineered to develop a lupus-like disorder, also underwent the pairings of saccharin (CS) and cyclophosphamide (UCS), the latter now being therapeutic. Conditioned immunosuppression resulted in reduced mortality and proteinuria. They also demonstrated that a 50% schedule, where one group of conditioned mice received cyclophosphamide after only half of the weekly occasions when they received the saccharin solution, reduced the effect, whereas exposure of unconditioned mice to the same number of respective administrations but in an unpaired manner resulted in no placebo effect. Other studies have also demonstrated therapeutic conditioned immunosuppression in animals (Reference Grochowicz, Schedlowski and HusbandGrochowicz et al, 1991) and humans (Reference AderAder, 2003), and another showed that the acquiring of conditioned responses in animals could be prevented by amygdala lesions (Reference Ramirez-Amaya, Alvarez-Borda and Bermudez-RattoniRamirez-Amaya et al, 1998).
Such conditioned responses have been demonstrated in other paradigms (Reference Fields, Price and HarringtonFields & Price, 1999; Reference Clow and PetersClow, 2001). Reference Benedetti, Amanzio and BaldiBenedetti et al(1999) studied responses in 60 surgical patients. The study began by demonstrating that pre-operative intravenous saline placebo had no clinical effect. After surgery, patients received analgesia in the form of daily intravenous opiate injections, which also resulted in measurable respiratory depression. On the fourth day, however, in a masked switch, some patients received a placebo and this also caused respiratory depression. It was found that the conditioned placebo-induced respiratory depression could be antagonised by naloxone.
Some studies have shown that placebo administration following active treatment is therapeutic in, for example, preventing relapse in schizophrenia (Reference Hogarty and GoldbergHogarty & Goldberg, 1973). Placebo caffeine (Reference Flaten and BlumenthalFlaten & Blumenthal, 1999), placebo nicotine (Reference Robinson, Houtsmuller and MoolchanRobinson et al, 2000) and placebo alcohol (Reference NewlinNewlin, 1989) have all been demonstrated to have clinical effects.
Reference Price, Fields and HarringtonPrice & Fields’ review (1999) notes parallels between the placebo response and classical conditioning. Both demonstrate extinction and stimulus generalisation phenomena, and that the conditioned response is smaller than the unconditioned response. Reference Ader and HarringtonAder (1999) argued that such physiological conditioning is more complicated than at first sight, citing evidence demonstrating compensatory conditioned responses, where the conditioned response is a process that actually opposes the drug effect. Occurring when the drug affects the efferent (rather than afferent) arm of the nervous system, such responses take place in anticipation of, and as a homoeostatic means of, attenuating the effects of the unconditioned stimuli.
However, there are many arguments against conditioning. Several experiments argue for conditioning even after only one pairing event; some seem to show that, in therapeutic practice, the first administration of an intervention actually follows no pairing events. Furthermore, do the studies above prove that conditioning is the mediator, as opposed to expectation? Participants responding to ‘placebo’ analgesia, antipsychotic, caffeine, nicotine, or alcohol probably held numerous cognitions about what they were receiving and its expected effects. Several studies found that manipulating these expectations influenced the outcome, overriding any prior conditioning (Reference Fillmore, Mulvihill and Vogel-SprottFillmore et al, 1994; Reference Montgomery and KirschMontgomery & Kirsch, 1997).
As with the link between anxiety reduction and expectation, conditioning and expectatation may also be connected. Reference KirschKirsch (1990) argues that classical conditioning is a means by which expectancies mediating placebo effects are acquired. In fact, words themselves are symbols that become associated (’paired’) with items and concepts in the external world (Reference Siegel, Guess, Kleinman and KusekSiegel, 2002).
One study aimed to elucidate the respective roles of expectation and conditioning on two different groups: healthy volunteers undergoing experimental ischaemic arm pain and patients with Parkinson’s disease (Reference Benedetti, Pollo and LopianoBenedetti et al, 2003). The authors manipulated participants’ expectations of pain experience and motor control respectively. Overall, they found that expectations had no effect on hormonal secretion, but could affect pain experience and motor performance. This implies that unconscious physiological placebo responses such as those affecting the endocrine system may be mediated by conditioning; however, conscious placebo responses such as pain and motor performance may be mediated by expectation, which may ‘trump’ previously conditioned processes.
Overall, the evidence suggests that ‘expectation effects’ may sometimes utilise conditioning processes, although there are circumstances when conditioning can operate on a physiological level without being recognised consciously or cognitively.
The mind/body gap
Perhaps all these mechanisms may be representations of the same process, focusing on different aspects, particularly in relation to levels of conscious awareness (Fig. 1). For a particular person, place and time, the presence of conscious awareness may mean that it is expectation that it is primarily in operation; for another situation, it may be conditioning.
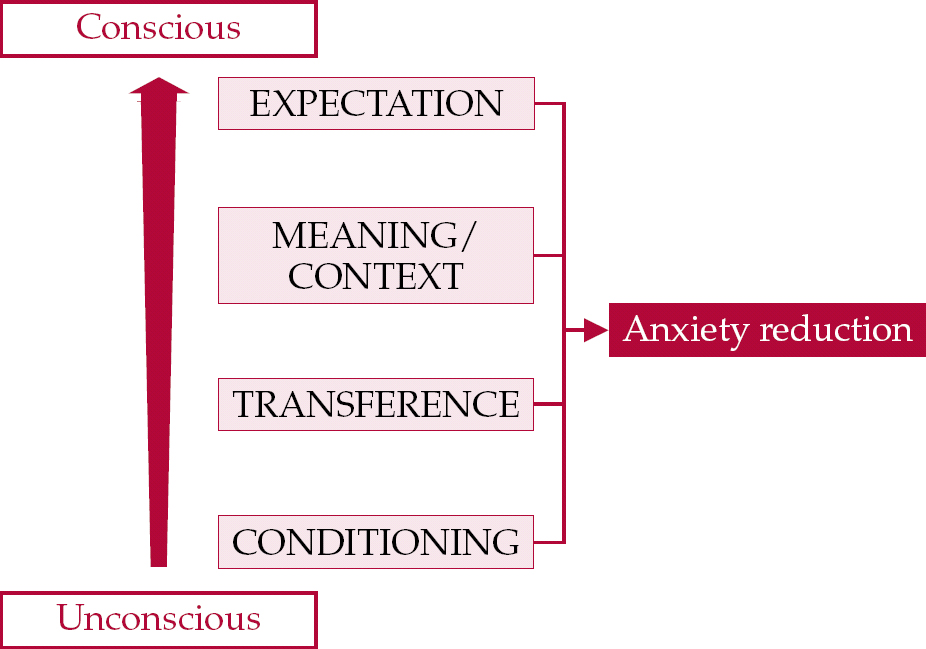
Fig. 1 Mechanisms that underlie the placebo effect. → indicates potential causation.
Some would argue that there are instances where conditioning alone explains purely physiological effects (e.g. conditioned immune responses in mice) and psychological processes alone explain psychological effects (e.g. placebos reduce negative depressive thoughts by instilling hopeful positive cognitions). However, in most cases it is generally necessary to bridge the explanatory gap between the realm of physical biology and the world of psychology and meaning.
Reference Morris and HarringtonMorris (1999) argues for a ‘top-down’ approach:
‘humans activate the neurobiological circuits required for placebo effects through the subtle and diffuse experience of living within the inescapably meaning-rich domain of culture’ (p. 189).
Research, however, has yet to adequately trace the complex web of interacting psychological and physiological processes that underlie the placebo effect.
Evidence shows that the placebo effect can be mediated via peptide release (Reference Brody and HarringtonBrody, 1999) or immunological and endocrinological effects (Reference Clow and PetersClow, 2001; Reference Eskandari, Sternberg, Guess, Kleinman and KusekEskandari & Sternberg, 2002) or, in people with Parkinson’s disease, dopamine release (Reference de la Fuente Fernandez, Ruth and Sosside la Fuente-Fernandez et al, 2001). Evans argues that placebos may operate by suppressing a pathologically occurring inflammatory acute-phase response underlying many conditions, including pain, ischaemic heart disease and perhaps depression (Reference EvansEvans, 2003, Reference Evans2005).
Others argue for a ‘bottom-up’ process, where the physiology is the primary mediator affecting the psychology. Studies using functional neuroimaging or electroencephalograms have demonstrated biological correlates associated with placebo groups (in pain or depression modalities) (Reference Mayberg, Silva and BrannanMayberg et al, 2002; Reference Leuchter, Cook and WitteLeuchter et al, 2002; Reference Benedetti, Mayberg and WagerBenedetti et al, 2005; Reference Kong, Gollub and RosmanKong et al, 2006). These serve to demonstrate that the effect is a real phenomenon and that physical processes are involved, but do not indicate whether the primary agent of causation is the biology, or the psychology or both. Moreover, it can be difficult to discern whether any change in brain activity reflects a pathological process or a reparative homoeostatic response. Furthermore, as in clinical paradigms, most studies do not compare placebo groups with no-treatment groups, thereby making it difficult to discriminate between what are genuine placebo responses and what is natural recovery. Ideally, by determining which brain loci are specifically active during the placebo effect, a neuropsychological model of the effect can be constructed.
Future research
Research into the placebo effect has proved it to be a vastly complicated phenomenon, encompassing complex interactions of conscious and unconscious psychosocial processes. Furthermore, the phenomenon stretches intriguingly across the mind/body gap, a conceptual divide that has plagued scientists and philosophers since Descartes and beyond. Functional neuroimaging may not fully resolve this philosophical conundrum, but it is beginning to unravel some of the placebo effect’s neurobiological components; already several candidate areas and processes are being proposed. Although research so far remains hampered by small samples and lack of replication, it will undoubtedly continue to reveal useful insights.
More studies are needed to elucidate the role of personality and expectation in the placebo effect. The studies from the 1950s and 1960s were generally methodologically weak and their results inconsistent. This paradigm may not be a lost cause, however: perhaps research into personality effects can be revisited, and this time more rigorously, given the improved quality of modern studies. It can be envisaged that such research would take advantage of existing drug trials; standardised attitude and personality questionnaires could be incorporated into the study protocol and its variables then correlated against outcome.
There also remains room for further investigation into how conditioning influences response to therapeutic intervention and also into the biochemical correlates of the placebo effect. Both can be conducted in psychiatry: for example, investigating in patients with depression the relationship between the placebo effect and parameters such as hormonal levels or the dexamethasone suppression test.
The placebo effect may still be maligned or ignored in some quarters, yet recent times have seen a strong movement towards the acknowledgment by the medical community of both its presence and its importance. Such a paradigm shift can only serve to increase public and academic interest and further fuel a research endeavour that is already gathering considerable momentum.
Declaration of interest
None.
MCQs
-
1 The following are mechanisms purported to underlie the placebo effect:
-
a transference
-
b expectation
-
c meaning effects
-
d conditioning
-
e psychoneuroimmunological processes.
-
-
2 Which of the following best represents the ideal research paradigm to investigate the placebo effect?
-
a treatment group v. placebo group
-
b placebo group v. no-treatment group
-
c treatment group v. no-treatment group
-
d placebo group only
-
e treatment group only.
-
-
3 In antidepressant trials, the placebo response rate is increased if:
-
a participants who responded to a placebo in the ‘run-in’ leg of the trial are included
-
b ‘active’ placebos are used
-
c a single-blind design is used
-
d participants with low IQs are recruited
-
e the research is sponsored by a pharmaceutical company.
-
-
4 The following factors have been shown to influence clinical efficacy:
-
a drug colour
-
b nature of administration
-
c drug name
-
d drug content
-
e practitioner enthusiasm.
-
-
5 The following tools may be of benefit in researching the placebo effect:
-
a functional neuroimaging
-
b validated personality questionnaires
-
c clinical trials
-
d no-treatment groups
-
e animal studies.
-
MCQ answers

| 1 | 2 | 3 | 4 | 5 | |||||
|---|---|---|---|---|---|---|---|---|---|
| a | T | a | F | a | F | a | T | a | T |
| b | T | b | T | b | T | b | T | b | T |
| c | T | c | F | c | T | c | T | c | T |
| d | T | d | F | d | F | d | T | d | T |
| e | T | e | F | e | F | e | T | e | T |





eLetters
No eLetters have been published for this article.