Published online by Cambridge University Press: 14 April 2009
1. Nude is a new recessive gene causing hairlessness in the mouse. It is linked to rex and trembler in linkage group VII. The order of the three loci and the recombination frequencies are as follows:
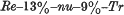
2. In addition to hairlessness the new gene causes reduced body growth rate, very low fertility and a liver disease causing death. Nude mice may be classified at birth by the absence of vibrissae.
3. The hairlessness is due to abnormal keratinization of hair in the follicles. The skin histology resembles that of naked mice. The hair follicles were found to be deficient in free sulphydryl groups.
4. The majority of nude mice die of general body weakness within 2 weeks of birth. The survivors grow slowly and may live for a considerable period. But all nude mice eventually die, usually between 3 and 14 weeks of age.
5. The livers of dead or moribund nude mice are covered with lesions and scars. The defect has been traced histologically to its initial stage, namely, necrosis of small areas of tissue.
6. Attempts to relate the deficiency of sulphydryl groups in the hair follicles to abnormal sulphur metabolism in the liver were unsuccessful.
7. Pseudo-cysts of a parasitic protozoan, Toxoplasma gondii, were identified in the liver and brain of nude mice. In one case the free form of the organism was found.
8. The possible relationship between the liver disease and the pathogenic organism is discussed.