Crossref Citations
This article has been cited by the following publications. This list is generated based on data provided by
Crossref.
Shchipalkina, Nadezhda V.
Pekov, Igor V.
Chukanov, Nikita V.
Zubkova, Natalia V.
Belakovskiy, Dmitry I.
Britvin, Sergey N.
and
Koshlyakova, Natalia N.
2020.
Vittinkiite, MnMn4[Si5O15], a member of the rhodonite group with a long history: definition as a mineral species.
Mineralogical Magazine,
Vol. 84,
Issue. 6,
p.
869.
Day, Maxwell C.
and
Hawthorne, Frank C.
2020.
A structure hierarchy for silicate minerals: chain, ribbon, and tube silicates.
Mineralogical Magazine,
Vol. 84,
Issue. 2,
p.
165.
Bindi, Luca
Nespolo, Massimo
Krivovichev, Sergey V
Chapuis, Gervais
and
Biagioni, Cristian
2020.
Producing highly complicated materials. Nature does it better.
Reports on Progress in Physics,
Vol. 83,
Issue. 10,
p.
106501.
Reinhardt, Nils
Frenzel, Max
Meinert, Lawrence D.
Gutzmer, Jens
Kürschner, Thomas
and
Burisch, Mathias
2021.
Mineralogy and fluid characteristics of the Waschleithe Zn skarn – a distal part of the Schwarzenberg mineral system, Erzgebirge, Germany.
Ore Geology Reviews,
Vol. 131,
Issue. ,
p.
104007.
Brusnitsyn, A. I.
Perova, E. N.
Vereshchagin, O. S.
Britvin, S. N.
Platonova, N. V.
and
Shilovskhikh, V. V.
2021.
The Mineralogy of Iron and Manganese Ores of the Ushkatyn-III Deposit in Central Kazakhstan.
Geology of Ore Deposits,
Vol. 63,
Issue. 8,
p.
772.
Duan, Nan
Cui, Kuixin
Zhu, Chuyu
and
Jin, Shengming
2023.
Study on phase evolution and promoting the pozzolanic activity of electrolytic manganese residue during calcination.
Environmental Research,
Vol. 227,
Issue. ,
p.
115774.
Myšľan, Pavol
Števko, Martin
Mikuš, Tomáš
and
Vrtiška, Luboš
2025.
Mineralogy and genetic considerations of the metamorphosed As-rich manganese ore mineralisation at the Diely occurrence near Poráč, Northern Gemeric Unit, Western Carpathians, Slovakia.
Mineralogical Magazine,
Vol. 89,
Issue. 1,
p.
71.
Myšľan, Pavol
Števko, Martin
Sejkora, Jiří
Ružička, Peter
and
Mikuš, Tomáš
2025.
Metamorphic manganese mineralisation bound to the metacarbonate lenses at the Smolník - Malá Hekerová deposit in the Spišsko-gemerské Rudohorie Mts., Western Carpathians (Slovakia).
Mineralogy and Petrology,
Vol. 119,
Issue. 2,
p.
243.
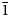 $\bar{1}$) pyroxenoids belonging to the rhodonite structure type, with a focus on their crystal chemistry, is given. These minerals have the general structural formula VIIM(5)VIM(1)VIM(2)VIM(3)VIM(4)[Si5O15]. The following dominant cations at the M sites are known at present: M(5) = Ca or Mn2+, M(1–3) = Mn2+; and M(4) = Mn2+ or Fe2+. In accordance with the nomenclature, the rhodonite group consists of three IMA-approved mineral species having the following the general chemical formulae: M(5)AM(1–3)B3M(4)C[Si5O15], where A = Ca or Mn2+; B = Mn2+; and C = Mn2+ or Fe2+. The end-member formulae of approved rhodonite-group minerals are as follows: rhodonite CaMn3Mn[Si5O15]; ferrorhodonite CaMn3Fe[Si5O15]; and vittinkiite MnMn3Mn[Si5O15].
$\bar{1}$) pyroxenoids belonging to the rhodonite structure type, with a focus on their crystal chemistry, is given. These minerals have the general structural formula VIIM(5)VIM(1)VIM(2)VIM(3)VIM(4)[Si5O15]. The following dominant cations at the M sites are known at present: M(5) = Ca or Mn2+, M(1–3) = Mn2+; and M(4) = Mn2+ or Fe2+. In accordance with the nomenclature, the rhodonite group consists of three IMA-approved mineral species having the following the general chemical formulae: M(5)AM(1–3)B3M(4)C[Si5O15], where A = Ca or Mn2+; B = Mn2+; and C = Mn2+ or Fe2+. The end-member formulae of approved rhodonite-group minerals are as follows: rhodonite CaMn3Mn[Si5O15]; ferrorhodonite CaMn3Fe[Si5O15]; and vittinkiite MnMn3Mn[Si5O15].