In both humans and chimpanzees (Pan troglodytes), a converging literature suggests the existence of a basic set of dispositional traits (i.e., personality) that can be organized into a coherent hierarchy such that broader traits can be differentiated into more fine-grained traits. Specifically, there is increasing evidence that various models of personality (i.e., two-, three-, four-, and five-factor models [FFM]) are hierarchically related among human adults (e.g., Digman, Reference Digman1997; Markon, Krueger, & Watson, Reference Markon, Krueger and Watson2005), human youth (e.g., Tackett, Krueger, Iacono, & McGue, Reference Tackett, Krueger, Iacono and McGue2008; Tackett et al., Reference Tackett, Slobodskaya, Mar, Deal, Halverson, Baker and … Besevegis2012), and chimpanzees (e.g., Latzman, Freeman, Schapiro, & Hopkins, Reference Latzman, Freeman, Schapiro and Hopkins2015; Latzman, Hopkins, Keebaugh, & Young, Reference Latzman, Hopkins, Keebaugh and Young2014). Furthermore, largely consistent with findings in humans (e.g., Allen & DeYoung, Reference Allen and DeYoung2017; Bouchard, Reference Bouchard2004; Bouchard & McGue, Reference Bouchard and McGue2003; DeYoung, Reference DeYoung2010; Power & Pluess, Reference Power and Pluess2015) personality traits in chimpanzees appear to have significant genetic (e.g., Latzman, Freeman et al., Reference Latzman, Freeman, Schapiro and Hopkins2015; Weiss, King, & Figueredo, Reference Weiss, King and Figueredo2000) and neuroanatomical contributions (Blatchley & Hopkins, Reference Blatchley and Hopkins2010; Latzman, Freeman et al., Reference Latzman, Freeman, Schapiro and Hopkins2015). Whereas widely studied in humans, the limbic system has yet to be examined in chimpanzees with regard to associations with variation in personality. Given the importance of the limbic system in the explanation of variability in emotion (e.g., Hariri, Bookheimer, & Mazziotta, Reference Hariri, Bookheimer and Mazziotta2000; MacLean, Reference MacLean1955), the current study thus aimed to assess the association between amygdala and hippocampal volume and personality dimensions across levels of the personality hierarchy in a large sample of chimpanzees. Through the use of an unparalleled animal model (i.e., chimpanzees), the current study will provide critical insight into the neuroanatomical foundations of hierarchical personality dimensions and suggest avenues for future neurobiological investigations of personality in both human and nonhuman samples.
Research indicates that innate individual differences form the basis of personality traits and these traits can be organized into higher-order personality dimensions with two “metatraits,” Alpha and Beta, representing the most fundamental organization (DeYoung, Reference DeYoung2010; Digman, Reference Digman1990; Markon, Krueger, & Watson, Reference Markon, Krueger and Watson2005). Adult personality traits emerge through differentiation of three temperament dimensions, often referred to as the “Big Three”(Clark, Reference Clark2005). Two of these dimensions are affective—Negative Emotionality (NEM) and Positive Emotionality (PEM). The third dimension, Disinhibition (DIS), plays a role in the regulation of perception and interpretation of incoming stimuli (Clark & Watson, Reference Clark and Watson2008; Tellegen, Reference Tellegen1985). Converging evidence reveals hierarchical associations between these three temperament traits and dimensions of the widely studied FFM of personality (Markon, Reference Markon2009). The FFM includes: Neuroticism, which reflects individual differences in sensitivity to threat, punishment, and negative affect; Extraversion, which is thought to reflect sensitivity to reward and positive affect; Openness, which reflects the ability and tendency to detect, explore, and utilize abstract, sensory information; Agreeableness, thought to be the tendency toward altruism and cooperation; and Conscientiousness, which reflects top-down control of impulses and behavior toward compliance with rules and the pursuit of nonimmediate (McCrae & Costa, Reference McCrae and Costa2008). As one moves from this five-factor level up the hierarchy, Neuroticism and parts of low Agreeableness, together, form NEM; Extraversion and Openness, together, form PEM; low Agreeableness and low Conscientiousness, together, form DIS. In addition, both the FFM and the Big Three can be subsumed within the two broader, higher-order factors, or “metatraits” described earlier. Here, low DIS (FFM Agreeableness and Conscientiousness) as well as low NEM (FFM Neuroticism) can be captured by Alpha, whereas PEM (FFM Extraversion) can be captured by Beta (DeYoung, Reference DeYoung2006; Digman, Reference Digman1990; Markon, Krueger, & Watson, Reference Markon, Krueger and Watson2005). In an effort to elucidate this hierarchical organization of personality in nonhuman primates, recent studies in chimpanzees have found two largely similar, fundamental metatraits. These metatraits were then successfully differentiated into three factors similar to the Big Three model, and then further differentiated into five lower-order factors similar to the FFM (Latzman, Freeman et al., Reference Latzman, Freeman, Schapiro and Hopkins2015; Latzman et al., Reference Latzman, Hopkins, Keebaugh and Young2014). While the hierarchical structure of personality is largely clear and understood, the neurobiological foundations of these dimensions is less well understood and has been largely understudied.
Recent human neuroimaging research has illustrated the significance of various brain regions in the explanation of variation in personality (DeYoung, Reference DeYoung2010). Specifically, a large body of theoretical and empirical research underscores the importance of limbic structures in the explanation of emotion processes broadly and personality more specifically. Indeed, both the amygdala and the hippocampus have been found to associate with variation in negative and positive emotion. In particular, a positive association has emerged between amygdala volume and extraversion (Cremers et al., Reference Cremers, van Tol, Roelofs, Aleman, Zitman, van Buchem and … van der Wee2011; Omura, Constable, & Canli, Reference Omura, Constable and Canli2005); whereas smaller amygdala and mesial orbital frontal cortex (FC) volumes have been associated with higher levels of neuroticism/NEM (Lewis et al., Reference Lewis, Panizzon, Eyler, Fennema-Notestine, Chen, Neale and … Kremen2014). The amygdala has also been implicated in the judgment of negative emotion in facial expressions and in social judgments of trustworthiness and approachability (Adolphs, Tranel, & Damasio, Reference Adolphs, Tranel and Damasio1998; Adolphs, Tranel, Damasio, & Damasio, Reference Adolphs, Tranel, Damasio and Damasio1994). Recent work has also expanded our understanding of the amygdala’s function to include a motivational salience hypothesis by which the amygdala is sensitive to motivational relevance (Cunningham, Van Bavel, & Johnsen, Reference Cunningham, Van Bavel and Johnsen2008).This hypothesis may be related to the motivational systems proposed by Gray and McNaughton (Reference Gray and McNaughton2000), which described a “conceptual nervous system” theory of personality, including a behavioral approach system, which responds to cues of reward, and a behavioral inhibition system (BIS) as well as a fight–flight–freeze system (FFFS) that responds to cues of threats. Here, the dopaminergic system (which runs through the mesolimbic pathway) is linked to the behavioral approach system, the hippocampus and amygdala are implicated in BIS functioning, and the amygdala, hypothalamus, and periaqueductal gray are associated with FFFS functioning (Gray & McNaughton, Reference Gray and McNaughton2000). Thus, both BIS and FFFS functioning are thought to be associated with the hippocampus and amygdala.
Nonetheless, although implicated, in part, in BIS functioning in Gray’s (Gray & McNaughton, Reference Gray and McNaughton2000) model as described above, particularly with regard to information processing as well as in certain processes crucial for anxiety maintenance, the role of the hippocampus in emotion functioning is relatively less well understood than that of its neighbor, the amygdala. Although commonly thought of with regard to memory processes, the hippocampus has also been described as an inhibitory organ, particularly with regard to approach and avoid behavior. Stimuli that create an approach–avoidance conflict activate the BIS (DeYoung & Gray, Reference DeYoung and Gray2009; McNaughton & Corr, Reference McNaughton and Corr2014), thereby suggesting the role of the hippocampus in such processes. Indeed, the septo-hippocampal system (comprising a pathway from the septum to the hippocampus) is a key element in the BIS (Gray & McNaughton, Reference Gray and McNaughton2000). In addition, Phillips, Ladouceur, and Drevets (Reference Phillips, Ladouceur and Drevets2008) described a neural model of voluntary and automatic regulatory subprocesses of emotion that center around regions of the prefrontal cortex, hippocampus, and parahippocampus. In sum, the existing literature suggests that the hippocampus is linked to information processing and behavioral regulation while the amygdala corresponds more to affective associations to sensory information. Nonetheless, it is important note that effect sizes of associations between personality and structural variation are quite small. Indeed, although reliable, neuroticism, for example, has been found to associate with amygdala volume in very large samples only weakly (r=.10; Holmes et al., Reference Holmes, Lee, Hollinshead, Bakst, Roffman, Smoller and Buckner2012).
Largely consistent with the human research, nonhuman primate studies involving rhesus macaque monkeys (Macaca mulatta) have shown similar associations between the amygdala and sophisticated social judgments and emotional processing to those found in humans (Amaral, Reference Amaral2002; Prather et al., Reference Prather, Lavenex, Mauldin-Jourdain, Mason, Capitanio, Mendoza and Amaral2001). For example, amygdala lesions have been found to associate with fear mediation of social behavior but not fear mediation of inanimate objects (Prather et al., Reference Prather, Lavenex, Mauldin-Jourdain, Mason, Capitanio, Mendoza and Amaral2001), as well as an evaluative process of potential threat (Amaral, Reference Amaral2002). These findings provide support for the motivational relevance processes associated with the amygdala. Animals with amygdala, but not hippocampal, lesions have also shown an inability to acquire new potentiated startle reflexes after surgery, suggesting that the amygdala is essential for the fear-potentiated startle acquisition (Antoniadis, Winslow, Davis, & Amaral, Reference Antoniadis, Winslow, Davis and Amaral2007), and further implicating the amygdala’s involvement in initial emotion association. Research investigating the role of the nonhuman primate hippocampus has yielded similar results to those seen with humans. For example, in one study, rhesus macaques with hippocampal lesions demonstrated decreased emotional responsiveness to staring humans, a threatening stimulus (Machado & Bachevalier, Reference Machado and Bachevalier2008). In another study, increased excitability and exploration, as well as reduced responses to affiliative signals were associated with similar lesions (Machado & Bachevalier, Reference Machado and Bachevalier2006). Both studies demonstrate a decrease of emotional reactivity in response to threatening stimuli in animals whose hippocampus was lesioned. All told, these results further support the emotion eliciting role of the amygdala and the regulatory role of the hippocampus.
Although, similar to in humans, associations between emotional reactivity and amygdala and hippocampal structural variation are present in monkeys, research to date has yet to investigate these relations in our closest living nonhuman relative, chimpanzees. In addition to a large set of commonly shared genes (The Chimpanzee Sequencing and Analysis Consortium, 2005), considerable parallels exist between the brains of chimpanzees and humans (Freeman, Cantalupo, & Hopkins, Reference Freeman, Cantalupo and Hopkins2004; Hopkins, Li, Crow, & Roberts, Reference Hopkins, Li, Crow and Roberts2017). Furthermore, chimpanzees and humans share a myriad of emotional processes making chimpanzees an ideal model of human emotion (Phillips et al., Reference Phillips, Bales, Capitanio, Conley, Czoty, t Hart and … Nader2014). Evolutionarily, chimpanzees share a great deal of history with humans, including living in complex social environments requiring advanced social cognition for alliance formation and recognition of emotional displays (Nelson & Winslow, Reference Nelson and Winslow2009).
Importantly, as described above, a consistent recent literature has provided strong empirical support for the existence of not only a set of heritable converging personality dimensions in humans and chimpanzees (Freeman et al., Reference Freeman, Brosnan, Hopper, Lambeth, Schapiro and Gosling2013; Freeman & Gosling, Reference Freeman and Gosling2010; Latzman, Sauvigné, & Hopkins, Reference Latzman, Sauvigné and Hopkins2016), but support for a hierarchical organization of these traits largely consistent with that found in humans (Latzman, Freeman et al., Reference Latzman, Freeman, Schapiro and Hopkins2015; Latzman et al., Reference Latzman, Hopkins, Keebaugh and Young2014).The paralleled existence of basic traits in both humans and chimpanzees lends itself to valuable comparative models of human personality, largely void of the confounding factors associated with the typical socio-cultural pressures present in human studies. Given the relatively smaller array of confounding variables, dispositional variability is therefore likely more strongly rooted within biology. In sum, chimpanzees are an ideal model for understanding the neurobiological underpinnings of personality and emotion processes (for a review, see Latzman, Green, & Fernandes, Reference Latzman, Green and Fernandes2017).
Although promising, little research to date has explicitly examined the neural foundation of personality in chimpanzees. One recent study examined associations between FFM personality traits and the FC, and revealed evidence of an association between FC variation and personality. Specifically, FC grey matter (GM) volume was found to associate with Extraversion and Dominance (a trait that parallels reversed FFM Neuroticism; Latzman, Sauvigné, & Hopkins, Reference Latzman, Sauvigné and Hopkins2016), the two traits considered to be most affectively based (Latzman, Hecht, Freeman, Schapiro, & Hopkins, Reference Latzman, Hecht, Freeman, Schapiro and Hopkins2015; Watson & Clark, Reference Watson and Clark1997). Similarly, in male chimpanzees, individual variation in the subgenual cingulate cortex is associated with Dominance and Conscientiousness (Blatchley & Hopkins, Reference Blatchley and Hopkins2010). Despite clear associations with emotional processes in both humans and nonhuman (i.e., monkeys) primates, there is a notable gap in the literature assessing the relationship between personality and limbic structures (i.e., amygdala and hippocampus) in chimpanzees.
1. Current study
Within a relatively large sample of captive, socially housed chimpanzees, the overarching goal of the current study was to investigate associations between individual variability in limbic structures and dimensions of personality across levels of the personality hierarchy. Given the converging evidence of amygdala associations with extraversion (Canli, Sivers, Whitfield, Gotlib, & Gabrieli, Reference Canli, Sivers, Whitfield, Gotlib and Gabrieli2002; Cremers et al., Reference Cremers, van Tol, Roelofs, Aleman, Zitman, van Buchem and … van der Wee2011; Omura, Constable, & Canli, Reference Omura, Constable and Canli2005) and its negative associations with neuroticism/NEM (Aghajani et al., Reference Aghajani, Veer, Van Tol, Aleman, Van Buchem, Veltman and … van der Wee2014; Haas, Omura, Constable, & Canli, Reference Haas, Omura, Constable and Canli2007; Omura, Constable, & Canli, Reference Omura, Constable and Canli2005), we expected amygdala GM volumes to be associated with PEM, NEM, and related traits. Further, with prior evidence of the hippocampus being linked to information processing and behavioral regulation of emotion (Barros-Loscertales et al., Reference Barros-Loscertales, Meseguer, Sanjuan, Belloch, Parcet, Torrubia and Avila2006; Davidson, Jackson, & Kalin, Reference Davidson, Jackson and Kalin2000; McDonald & White, Reference McDonald and White1994), we expected hippocampal GM volumes to be most highly associated with DIS-related dimensions including Alpha at the two-factor level of the hierarchy and Impulsivity at the four- and five-factor levels. Given the hypothesis-driven nature of the current study, and as described previously (i.e., Ecker et al., Reference Ecker, Rocha-Rego, Johnston, Mourao-Miranda, Marquand and Daly2010; Pearlson, Reference Pearlson2008), we chose to adopt a region of interest (ROI) approach to analyses.
2. Method
2.1. Subjects
Chimpanzees were members of two colonies of apes housed at the Yerkes National Primate Research Center (YNPRC) in Atlanta, Georgia and the National Center for Chimpanzee Care (NCCC) at the University of Texas MD Anderson Cancer Center in Bastrop, Texas. Imaging data and personality ratings were available for 75 adult and subadult chimpanzees at YNPRC, including 51 females and 24 males with apes ranging in age from 9 to 53 years (M age=24.15, SD=11.34). Ratings were available for 116 adult and subadult chimpanzees at the NCCC, including 59 females and 57 males with apes ranging in age from 8 to 51 years (M age=28.16, SD=10.68). All apes were combined into a single sample for analyses, resulting in a final sample of 191 chimpanzees.
All aspects of the research complied with the American Psychological Association’s Guidelines for Ethical Conduct in the Care and Use of Nonhuman Animals in Research (APA, 2012), followed the Institute of Medicine guidelines for research with chimpanzees, and was done with the approval of the local Institutional Care and Use Committees. All the chimpanzees were housed in social groups ranging from 2 to 16 individuals. The chimpanzees were housed in indoor–outdoor compounds and had access to both portions of their enclosures 24 hr a day. During the winter seasons, the indoor facilities were heated, while air conditioning or fans and misters are provided in the hotter summer months. Lighting in the outdoor facility follows the typical seasonal cyclic change in sunrise and sunset. Standard tungsten lighting is provided in the indoor facility and the lights are on a 12 h on-off cycle. The chimpanzees are fed two to five times per day with a diet that consists of fruits, vegetables, and commercially produced primate chow. In addition, they receive a number of foraging opportunities each day. Environmental enrichment, such as simulated tool use tasks or other nonnutritive substrates, were provided to the chimpanzees on a daily basis. At no time were the subjects ever food or water deprived.
2.2. Assessment of personality
As described previously (i.e., Latzman, Freeman et al., Reference Latzman, Freeman, Schapiro and Hopkins2015) personality ratings were collected using a 41-item personality questionnaire developed through the consideration of both existing human personality literature as well as those traits that may be specific to chimpanzees (Freeman et al., Reference Freeman, Brosnan, Hopper, Lambeth, Schapiro and Gosling2013). Each of the 41 items consists of a single trait accompanied by a behavioral definition and a Likert-type scale ranging from 1=“least descriptive of the chimpanzee,” to 7=“most descriptive of the chimpanzee”. Strong evidence has been reported for five factors: Reactivity/unpredictability, Dominance, Extraversion, Openness, and Agreeableness. These scales have been found to evidence strong convergent and discriminant validity with various in vivo behavior and have demonstrated strong criterion validity with other validated personality scales (Freeman et al., Reference Freeman, Brosnan, Hopper, Lambeth, Schapiro and Gosling2013) and have also been shown to demonstrate clear translational value to personality traits studied in humans (Latzman, Sauvigné, & Hopkins, Reference Latzman, Sauvigné and Hopkins2016). Further, reliability has been shown to be adequate, both in terms of interrater reliability, and internal consistency (Freeman et al., Reference Freeman, Brosnan, Hopper, Lambeth, Schapiro and Gosling2013; Hopper et al., Reference Hopper, Price, Freeman, Lambeth, Schapiro and Kendal2014; Reamer et al., Reference Reamer, Haller, Thiele, Freeman, Lambeth and Schapiro2014). Using this instrument, chimpanzees were rated by colony staff members that worked with the animals for an extended period of time and “feel that they have enough experience for an accurate rating.” YNPRC chimpanzees were rated by five staff members and NCCC chimpanzees were rated by 17 staff members. The vast majority of apes were rated by at least two raters. Previous studies that have included data from these apes have demonstrated adequate interrater reliabilities for personality ratings (Freeman et al., Reference Freeman, Brosnan, Hopper, Lambeth, Schapiro and Gosling2013; Latzman, Freeman et al., Reference Latzman, Freeman, Schapiro and Hopkins2015). The average rating from all raters were computed and used for all analyses.
2.3. Image collection and procedure
All chimpanzees were scanned during one of their annual physical examinations that occurred within 2-years of their personality ratings. Magnetic resonance image (MRI) scans followed standard procedures at the YNPRC and NCCC and guidelines of the Institute of Medicine on the use of chimpanzees in research and were designed to minimize stress. Thus, the animals were first sedated with ketamine (10 mg/kg) or telazol (3–5 mg/kg) and were subsequently anaesthetized with propofol (40–60 mg/(kg/hr)). They were then transported to the MRI scanning facility and placed in a supine position in the scanner with their head in a human-head coil. Upon completion of the MRI, chimpanzees were singly housed for 2–24 hr to permit close monitoring and safe recovery from the anesthesia before being returned to their home social group. All procedures were approved by the Institutional Animal Care and Use Committees at YNPRC and NCCC. 120 chimpanzees were scanned using a 3.0 Tesla scanner (Siemens Trio; Siemens Medical Solutions USA Inc., Malvern, PA, USA). T1-weighted images were collected using a three-dimensional gradient echo sequence (pulse repetition=2,300 ms, echo time=4.4 ms, number of signals averaged=3, matrix size=320×320, with 0.6×0.6×0.6 resolution). The remaining 71 chimpanzees were scanned using a 1.5T G.E. echo-speed Horizon LX MR scanner (GE Medical Systems, Milwaukee, WI, USA). T1-weighted images were collected in the transverse plane using a gradient echo protocol (pulse repetition=19.0 ms, echo time=8.5 ms, number of signals averaged=8, matrix size=256×256, with 0.7×0.7×1.2 resolution).
Initially, each T1-weighted MRI scan was skull stripped and the volume segmented into GM, white matter (WM) and cerebrospinal fluid using FSL (Analysis Group, FMRIB, Oxford, UK) (Smith et al., Reference Smith, Jenkinson, Woolrich, Beckmann, Behrens, Johansen-Berg and … Flitney2004; Zhang, Brady, & Smith, Reference Zhang, Brady and Smith2001). Next, each individual T-1 MRI scan was linearly registered to a previously constructed symmetrical template of the chimpanzee brain using procedures that have been described elsewhere (Li et al., Reference Li, Preuss, Rilling, Hopkins, Glasser, Kumar and … Hu2010). The matrix derived from the linear registration was saved for later use. Before tracing, images were aligned in the axial, coronal, and sagittal planes along the anterior commisure - posterior commisure line and virtually sliced into 1 mm sections.
2.4. ROI tracing
2.4.1. Amygdala
Tracing of the amygdala was primarily done in the coronal plane using the mouse-driven trace tool within Analyze 10.0 software. The slice where the optic chiasm fuses was the most anterior slice where the amygdala was present, and served as the anterior border. The tentorial indentation or WM tract was used as the inferior border on most scans, depending on which was most prominent. An imaginary line stretching from the endorhinal sulcus to the fundus of the inferior portion of the circular sulcus of the insula was used as the superior border (see Figure 1A). The WM was used as the lateral border and the 3rd ventricle as the medial border; the GM within these boundaries in both hemispheres was assumed to be part of the amygdala (see Figure 1A). Moving posteriorly, when the horns of the lateral ventricles appeared, the tentorial indentation (or WM track) was no longer used as the inferior border. Instead, the lateral horn was used as the inferior border, and any tissue below this landmark was considered part of the hippocampus (see Figure 1B). The most posterior slice was defined as the first slice within which the mammillary bodies appeared in the image.
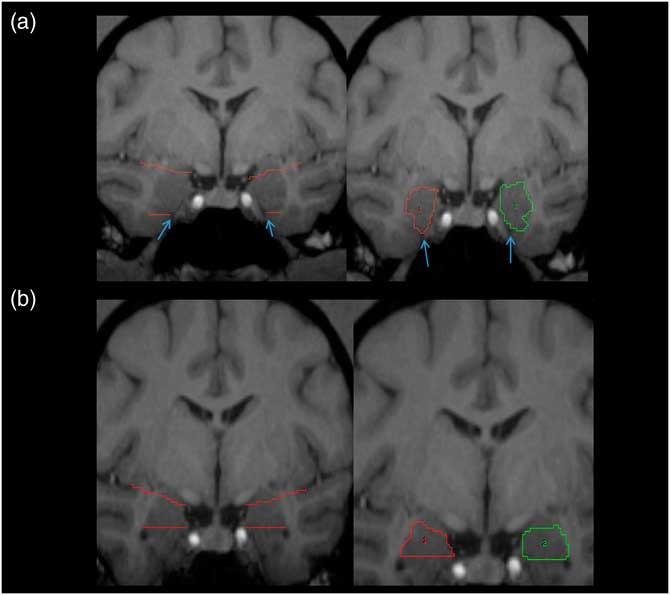
Figure 1 Pictorial representation of amygdala. (A) Left panel: the white matter tracts and/or the tentorial indentation (blue arrows) are used as the inferior border. (B) Left panel: upper angled red lines represent the imaginary line from the endorhinal sulcus to the inferior portion of the circular sulcus of the insula, which was used as the superior border of the amygdala. Right panels: traced amygdala on most anterior (A) and posterior (B) slices, with landmarks removed. ROIs for right (1) and left (2) shown in red and green, respectively. For more details, see amygdala region of interest tracing methods text.
2.4.2. Hippocampus
We quantified the hippocampus following procedures previously employed with humans and chimpanzees (De Bellis et al., Reference De Bellis, Keshavan, Clark, Casey, Giedd, Boring and … Ryan1999; Freeman, Cantalupo, & Hopkins, Reference Freeman, Cantalupo and Hopkins2004; Gosche, Mortimer, Smith, Markesbery, & Snowdon, Reference Gosche, Mortimer, Smith, Markesbery and Snowdon2001; Hopkins, Lyn, & Cantalupo, Reference Hopkins, Lyn and Cantalupo2009; Pegues, Rogers, Amend, Vinogradov, & Deicken, Reference Pegues, Rogers, Amend, Vinogradov and Deicken2003; Sherwood et al., Reference Sherwood, Cranfield, Mehlman, Lilly, Garbe, Whittier and … Holloway2004, Reference Sherwood, Gordon, Allen, Phillips, Erwin, Hof and Hopkins2011). The hippocampus was primarily traced in the coronal plane, but multiplanar views were used when it was difficult to distinguish landmarks in one plane of view. The anterior border of the hippocampus was defined as the first slice in which the inferior horn of the lateral ventricle was visible. The posterior border was the slice anterior to the crus of the fornix. This excluded the isthmus of the cingulate gyrus and the parahippocampal gyrus. The medial border of the hippocampus was defined by WM represented by the alveus, or the ambient cistern if the alveus could not be seen. The ambient cistern and alveus were often difficult to distinguish from the hippocampus. In such cases, several tactics were used, including enlarging the image of the slice to distinguish WM, and also comparing anterior and posterior slices to determine if WM was present. Laterally, the hippocampal border was the temporal horn of the lateral ventricle and the WM of the parahippocampal gyrus was used as the inferior border. The choroid fissure was used as the superior border of the hippocampus. The definition of the hippocampus included the cornus ammonis, dentate gyrus, subiculum, subicular complex, and hippocampus, proper. However, the fimbria and alveus were not included in the measurement of the hippocampus. As the delineation of the amygdala from the hippocampus was only based on the inferior horn of the lateral ventricle, rather than the alveus, a small portion of the amygdala may have been included in hippocampus measurements (Figure 2).
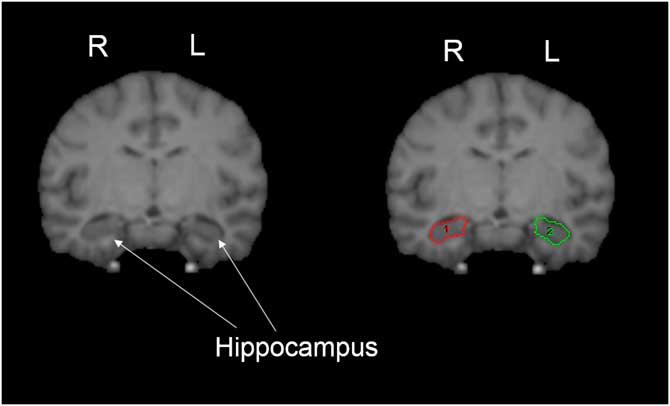
Figure 2 Pictorial representation of hippocampus. The white matter of the parahippocampal gyrus was used as the inferior border, and the choroid fissure was used as the superior border of the hippocampus. The definition of the hippocampus included the cornus ammonis, dentate gyrus, subiculum, subicular complex, and hippocampus, proper (shown on right image). ROIs for right (1) and left (2) shown in red and green, respectively. For more details, see hippocampus region of interest tracing methods text.
Once traced, the individual area measures were summed across all slices within each hemisphere to create a total amygdala and hippocampus volume for each subject. In order to adjust for individual variation in brain size, the volume of the left and right amygdalae and hippocampi were divided by the total volume of the left and right hemisphere (Amyg_Ratio, Hipp_Ratio). Total hemispheric volume included all gray and WM within each hemisphere and excluded cerebrospinal fluid, the cerebellum, and all brain stem structures. To account for potential differences in the regions based on image resolution related to the strength of the scanner magnet, the Amyg_Ratio and Hipp_Ratio values were converted to standardized z-scores within the sample of brains scanned at 3.0 T or 1.5 T. These z-scores were used in all subsequent analyses in the explanation of variation in personality.
2.5. Data analysis
Hierarchical personality dimensions were extracted through a series of oblique geomin rotated maximum likelihood (ML) exploratory factor analyses (EFA). Given previous findings in both humans (e.g., Markon, Krueger, & Watson, Reference Markon, Krueger and Watson2005) and chimpanzees (e.g., Latzman, Freeman et al., Reference Latzman, Freeman, Schapiro and Hopkins2015) of a hierarchical structure with five factors providing the best fit to the data at the most nuanced level, four models extracting two to five factors were run and ML estimated factor scores were saved for use in subsequent path analyses. Before fitting models, however, an eigenvalue Monte Carlo p-values (i.e., parallel analysis; Horn, Reference Horn1965) was derived to confirm the appropriateness of extracting up to five factors.
To assess associations between standardized amygdala and hippocampal GM volume and personality dimensions across levels of the hierarchy, a series of path analyses were fitted. Specifically, we investigated the extent to which amygdala and hippocampal structural variation explained personality traits across levels of the hierarchy by regressing personality (two- to five-factor solutions) on GM volumes in four path models. Both amydala and hippocampal volume were included simultaneously in all models as a means of reducing Type II error. Consistent with previous studies (e.g., Latzman, Hecht et al., Reference Latzman, Hecht, Freeman, Schapiro and Hopkins2015), age and sex were included as covariates in all path models. Both EFA and path analyses were conducted using Mplus Version 7.4 (Muthén & Muthén, 1998–2015).
3. Results
3.1. Hierarchical personality dimensions
As described above, before structural analyses, a parallel analysis was conducted. As shown in Supplemental Figure S1, consistent with previous principal components analysis (PCA) findings (Latzman, Freeman et al., Reference Latzman, Freeman, Schapiro and Hopkins2015; Latzman et al., Reference Latzman, Hopkins, Keebaugh and Young2014), these analyses suggested that a five-factor solution was most appropriate at the most nuanced level. We thus extracted up to five factors. Results from two- to five-factor EFA solutions are provided in the Supplemental Materials (Tables S2–S5). In short, the two-factor model appeared to distinguished Alpha, anchored by mischievous, impulsive, bold, and aggressive from Beta, anchored by affectionate, intelligent, reverse-keyed autistic, and affiliative. In the three-factor model, the Alpha factor bifurcated into a low NEM factor anchored by bold, reverse-keyed timid, dominant, and reverse-keyed fearful, and an Aroused DIS factor anchored by excitable, reverse-keyed relaxed, reverse-keyed calm¸ and anxious, whereas the Beta dimension emerged as PEM, anchored by inquisitive, inventive, affiliative, and intelligent. The four-factor solution included an Agreeableness dimension anchored by affiliative, affectionate, reverse-keyed autistic, and reverse-keyed solitary. Further, emerging from the broader NEM factor at the three-factor level was an Impulsivity factor, anchored by deceptive, temperamental, irritable, and impulsive, and a Dominance factor anchored by reverse-keyed fearfulness, timid, and anxious, and bold. Lastly within the four-factor solution an Extraversion factor emerged primarily from three-factor PEM anchored by playful, active, human-oriented, and excitable. Finally, at the five-factor level, a structure resembling the FFM emerged. In addition to factors similar to the four from the four-factor model, albeit with Dominance reverse-keyed, a fifth factor termed Intellect emerged anchored by human-oriented, persistent, jealous, and inventive.
To confirm links to previous hierarchical personality results as well as the appropriateness of our factor labels, correlations between ML-based factor scores and previously found PCA-based factor scores from Latzman, Freeman et al. (Reference Latzman, Hecht, Freeman, Schapiro and Hopkins2015) were examined. As shown in Supplemental Table S1, a convergent/discriminant pattern of correlations emerged providing clear support for (1) the convergence of the current set of ML-based factor results with previous findings, and (2) the suitability of factor labels described above. Specifically, across all levels of the hierarchy, the highest correlations were between parallel factors. Further, correlations between these factors and other factors within each solution were appreciably lower.
Next, as described above, to investigate associations between personality dimensions at each level of the personality hierarchy and amygdala and hippocampal volume, four path models were run in which personality dimensions (two to five factors) were regressed on hippocampal and amygdala volumes while statistically controlling for age and sex.
3.2. Associations between hierarchical personality dimensions and age and sex
Age evidenced significant negative associations with both Alpha/Stability and Beta/Plasticity (Mdn β=−.31, Mdn t=−4.69, ps<.001) at the two-factor level, PEM and NEM (Mdn β=−.35, Mdn t=−5.45, ps<.001) at the three-factor level, Agreeableness, Impulsivity, and Dominance (Mdn β=−.32, Mdn t=−5.07, ps<.01) at the four-factor level, and Agreeableness, Impulsivity, and Intellect (Mdn β=−.31, Mdn t=−4.75, ps<.001) at the five-factor level; age was further positively associated with low Extraversion at the five-factor level (β=.25, t=3.69, p<.001). While sex was not associated with any of the personality dimensions at the two- or three-factor levels of the hierarchy, males evidenced higher levels of Impulsivity (β=.56, t=4.68, p<.001) and Extraversion (β=.38, t=2.70, p<.01) at the four-factor level and higher levels of Dominance (β=−.44, t=−3.16, p<.01) and Extraversion (β=−.90, t=−7.74, p<.001) at the five-factor level.
3.3. Associations between Hierarchical Personality Dimensions and Amygdala and Hippocampus Volumes
As shown in Table 1, at the most basic level of the personality hierarchy, hippocampal volume was significantly positively associated with Alpha (β=.14, t=2.10, p<.05) and also appeared to be negatively associated with Beta (β=−.11, t=−1.71, p=.09). Amygdala volume was not associated with either personality dimension (βs<|.09|, ts<|1.35|, ps>.10). At the three-factor level, hippocampal volume associated positively with DIS (β=.14, t=2.08, p<.05), a dimension positively linked to Alpha and trended towards a negative association with PEM (β=−.13, t=1.93, p=.05), a dimension positively linked to Beta, and; hippocampal volume did not associate with NEM (β=−.01, t=−0.15, p>.85). Again, amygdala volume did not associate with any of the personality dimensions (βs<|.08|, ts<|1.2|, ps>.20). Next, at the four-factor level of personality hierarchy, whereas none of the associations emerged as significant at the p<.05 level, hippocampal volume emerged as negatively associated with Agreeableness (β=−.13, t=−1.90, p=.06), a trait that breaks off most notably from PEM. Results also suggested a positive association with Dominance (β=.14, t=1.94, p=.05), a DIS-related trait; hippocampal GM volume did not associate with Impulsivity (β=.08, t=1.40, p>.15) or Extraversion (β=.01, t=0.14, p>.85) at the four-factor level. Further, consistent with results for the two previous levels of the hierarchy, amygdala volume did not associate with any of the personality dimensions at the four-factor level (βs<|.11|, ts<|1.55|, ps>.10). Finally, at the most basic FFM level of the personality hierarchy, hippocampal volume was positively associated with the Impulsivity dimension (β=.15, t=2.16, p<.05) as well as the Agreeableness dimension (β=−.11, t=−1.66, p<.10); hippocampal GM volume was not associated with any of the other three dimensions at this level of the hierarchy (βs<|.05|, ts<|0.70|, ps>.50). Consistent with findings across all other levels of the hierarchy, amygdala volume did not emerge as a significant correlate of any of the FFM dimensions of personality (βs<|.10|, ts<|1.60|, ps>.10).
Table 1 Path analyses explaining hierarchical personality dimensions from amygdala and hippocampus grey matter volume

Note. N=191, β=standardized coefficient.
As described in text, age and sex included as covariates in all models.
*p<.05. † p<.10.
4. Discussion
The current study contributes to the growing personality neuroscience literature as well as the nascent empirical literature investigating neurobiological foundations of personality in chimpanzees, our closest nonhuman primate relatives. Research concerning the evolutionary and biological foundations of personality is particularly important considering the critical importance of dispositional variation in the explanation of a range of outcomes including human mental health and well-being (Latzman, Green, & Fernandes, Reference Latzman, Green and Fernandes2017). The current study specifically examined associations between hierarchical personality dimensions and, given previous findings of associations with various emotional processes in both humans and nonhuman primates, GM volume of two core limbic structures, the amygdala and hippocampus. Although hypotheses regarding associations between personality and amygdala volume were not supported, hippocampal volume emerged as an important limbic structure vis-à-vis variation in personality, particularly with regard to traits associated with reduced behavioral control. Taken together, consistent with previous findings in both humans (e.g., Allen & DeYoung, Reference Allen and DeYoung2017) and chimpanzees (e.g., Latzman, Hecht et al., Reference Latzman, Hecht, Freeman, Schapiro and Hopkins2015), results suggest a brain-based explanation for broad personality traits that appears to be largely conserved across both species.
Results of EFA analyses extracting two- to five-factors confirmed the existence of a hierarchical personality structure in chimpanzees. Indeed, the structure that emerged across levels of the hierarchy was consistent with previous structural findings in chimpanzees (i.e., Latzman, Freeman et al., Reference Latzman, Hecht, Freeman, Schapiro and Hopkins2015) despite differing methods employed for factor extraction (ML estimation vs. PCA). Further underscoring the replicability of these results, suggesting a hierarchical structure of personality, were correlational findings of convergence of EFA-derived factors from the current study with PCA-derived factors from previously published work (see Supplemental Table S1).
Specifically, and also consistent with the hierarchical structure of personality repeatedly found in humans (e.g., Digman, Reference Digman1990; Markon, Reference Markon2009; Tackett et al., Reference Tackett, Slobodskaya, Mar, Deal, Halverson, Baker and … Besevegis2012), Alpha (tendency to behave in an undercontrolled and agonistic manner) and Beta factors (tendency to behave in an approach oriented, affiliative manner), emerged at the highest level of hierarchy. These broad dimensions then differentiated most notably into DIS and NEM (Alpha) and PEM (Beta) at the three-factor level. At the next level of the hierarchy, Impulsivity, which can also be thought of as low Conscientiousness within the FFM, Agreeableness, Extraversion, and Dominance emerged from the previous three factors. Lastly, as expected and consistent with previous findings in both humans and chimpanzees, a structure largely parallel to the FFM emerged at the final, most fine-grained level of the hierarchy. In this solution, in addition to the four factors present in the previous solution, a factor labeled Intellect emerged. All told, the series of EFAs resulted in the emergence of a hierarchical structure parallel to what has been previously found in both humans and chimpanzees.
Consistent with previous findings (e.g., Barros-Loscertales et al., Reference Barros-Loscertales, Meseguer, Sanjuan, Belloch, Parcet, Torrubia and Avila2006; McDonald & White, Reference McDonald and White1994; Walters & Kiehl, Reference Walters and Kiehl2015) and expectations concerning associations between hippocampal GM and personality, results suggest that the hippocampus plays an important role in behavioral and emotional regulation. Indeed, at the most basic two-factor level of the personality hierarchy, Alpha evidenced a positive association with hippocampal GM. The association between hippocampal GM and traits reflecting a general tendency to be undercontrolled and agonistic emerged further down the personality hierarchy as well with hippocampal GM associating positively with DIS at the three-factor level as well as with Dominance at the p<.10 level at the four-factor level and Impulsivity at the five-factor level. Underscoring the association between hippocampal GM and agonistic behavioral tendencies was the negative associations that emerged with Agreeableness, a trait that includes antagonism at the opposite pole, at both the four- and five-factor levels of the personality hierarchy, albeit at the p<.10 level.
In addition to associations between hippocampal GM and DIS-related traits, PEM and the associated more general higher-order dimension of Beta also evidenced associations with hippocampal GM at p<.10, although these associations were negative. These results likely further reflect the importance of the hippocampus for both negative and positive emotions and the behavioral control of these emotions. Whereas the amygdala has garnered the lion’s share of the attention with regard to neurobiological investigations of emotion, the importance of the hippocampus as a mechanism for emotion is not new. Indeed, Gray (Gray & McNaughton, Reference Gray and McNaughton2000) has described the hippocampus’ involvement in inhibitory association formation and the regulation of the weight of affectively negative emotion. Further, in his proposed neurobiological model of emotion, Papez (Reference Papez1937) described a circuit that included the hippocampus. In conjunction with previous findings in chimpanzees regarding associations between neuroanatomical variation and personality (Latzman, Hecht et al., Reference Latzman, Hecht, Freeman, Schapiro and Hopkins2015), particularly the anterior cingulate cortex, results of the current study provide further support for Papez’s model, and other models implicating a hippocampal role in emotional dysregulation (e.g., Gross, Reference Gross1998; Gross & Jazaieri, Reference Gross and Jazaieri2014).
With regard to the amygdala, however, results were not consistent with expectations. Indeed, whereas we expected to find positive associations with both PEM- and NEM-related traits, amygdala GM variation did not associate with any of the personality dimensions across levels of the hierarchy. These results were quite surprising given the plethora of previous findings concerning the clear importance of the amygdala in the explanation of emotion. Nonetheless, there are a number of potential explanations for our unexpected findings. For example, increased GM volume indirectly implies differences in WM volume, and is suggestive of variability in connectivity, however, an ROI approach does not allow for the assessment of connectivity within and between limbic regions (Ge et al., Reference Ge, Grossman, Babb, Rabin, Mannon and Kolson2002). Furthermore, the distinction between structure and function is an important one. The current study focused only on structure (i.e., GM variation) but not function. Whereas there appears to be an important association between the two, they are clearly not interchangeable nor does one explain the other consistently throughout the brain. In agreement with previous assertions (e.g., Gray & McNaughton, Reference Gray and McNaughton2000), it may be that it is hippocampal structure and amygdala function that are most important for variation in personality. This assertion, of course, warrants future research before it can be considered anything more than conjecture. Further, the majority of nonhuman primate studies on the association between limbic structures and personality or emotion have either used functional imaging or have employed lesion methodologies, the latter of which does not allow for differentiation between structure and function. Thus, the relevance of results of these studies to consideration of structural variation is not entirely clear.
A distinction between the structure and function of the areas of interest associated with personality is an important one. It is important to remember that structural associations with personality are not necessarily indicative of the functional relationship between these brain regions and the dimensions of interest. For example, where the amygdala and hippocampus may interact directly due to their reciprocal connections (Gray & McNaughton, Reference Gray and McNaughton2000), each structure might affect a unique process indirectly by its ability to independently increase or decrease arousal independently. Regardless of the explanation for our lack of amygdala findings, additional research is needed. Given the difficulties in discriminating these limbic regions in both humans and chimpanzees, and the converging literature supporting a functional role of the amygdala and hippocampus in emotional regulation, future studies should investigate the potential collective functionality and/or coupled structural variation of these regions. Specifically, it will be important for future research to consider how GM/WM volume within the amygdala and hippocampus relates to connectivity between these, and other limbic regions, and how these regions influence socio-emotional processing and behavioral regulation.
4.1. Implications for understanding human mental health
In addition to implications for understanding the foundation of personality, these results also have important implications for our understanding of the pathophysiology of human mental illness more generally (for a review, see Latzman, Green, & Fernandes, Reference Latzman, Green and Fernandes2017). Indeed, a large literature has demonstrated the importance of variation across personality traits with regard to associations with various mental disorders suggesting the importance of personality research to delineating mechanisms and processes underlying mental disorders (Kotov, Gamez, Schmidt, & Watson, Reference Kotov, Gamez, Schmidt and Watson2010). Further, personality and psychopathology appear to share a common etiology with both determined by innate temperamental traits and further shaped by development and the environment (Caspi et al., Reference Caspi, Houts, Belsky, Goldman-Mellor, Harrington, Israel and … Poulton2014; Clark, Reference Clark2005; Durbin & Hicks, Reference Durbin and Hicks2014). Importantly, the notion of broad, biologically based temperamental traits underlying both personality and psychopathology (i.e., Clark, Reference Clark2005) is consistent with a hierarchical model of personality and also converges with recent hierarchical models of psychopathology (i.e., Hierarchical Taxonomy of Psychopathology (HiTOP); Kotov et al., Reference Kotov, Krueger, Watson, Achenbach, Althoff, Bagby and … Clark2017).
The current set of findings further intersect with the National Institute of Mental Health’s Research Domain Criteria (RDoC) Initiative which aims to explicate the neurobiological bases of mental illness. RDoC is explicitly interested in dimensional dispositional constructs as phenotypes for neurobiological investigations. The current study’s focus on the neural basis of variation across dispositional processes (i.e., personality), with clear relevance to human mental health, is thus RDoC-relevant. Indeed, along with other ongoing work (Latzman, Young, & Hopkins, Reference Latzman, Young and Hopkins2016), the current research further highlights the importance of nonhuman primate-translational operationalizations of various dispositional domains (Latzman, Sauvigné, & Hopkins, Reference Latzman, Sauvigné and Hopkins2016) and its relevance to various transdiagnostic models of psychopathology (Latzman & Hopkins, Reference Latzman, Sauvigné and Hopkins2016).
4.2. Limitations
The current study is of course not without limitations. The cross-sectional correlational nature of the design does not allow for causal inferences to be made. That is, whether personality factors underlie variation in limbic structure or vice versa cannot be determined from these findings. Further, it is important to note that the imaging techniques utilized in the current study only allow for the investigation of structural variation and its relation to hierarchical personality dimensions. Although our results support Papez’s (Reference Papez1937) original inclusion of the hippocampus in the FC/forebrain—limbic circuit responsible for the control of emotional expressions, as well as Gray’s (Gray & McNaughton, Reference Gray and McNaughton2000) conceptual nervous system of personality, the exact way in which the hippocampus, and other limbic structures, are involved in emotional processing is still not entirely clear. Indeed, although a number of studies have found associations between hippocampal GM and emotion and regulation, the direction of associations is not always in the same direction (e.g., Barros-Loscertales et al., Reference Barros-Loscertales, Meseguer, Sanjuan, Belloch, Parcet, Torrubia and Avila2006; Iidaka et al., Reference Iidaka, Matsumoto, Ozaki, Suzuki, Iwata, Yamamoto and … Sadato2006).
In addition to the independent role the hippocampus appears to play in behavioral and emotional regulation a growing literature in humans highlights an interactive, functional role of the hippocampus and amygdala in emotional processing and regulation (Phelps, Reference Phelps2004; Richardson, Strange, & Dolan, Reference Richardson, Strange and Dolan2004). Unfortunately, given restrictions on functional imaging in chimpanzees (for a discussion of these limitations and the scientific impact, see Hopkins & Latzman, 2017; Latzman & Hopkins, Reference Latzman, Sauvigné and Hopkins2016), an investigation of the functional roles various limbic structures play in emotional processing is currently not possible in this unique model species. Indeed, as described earlier, limbic structure volumes appear to be only weakly correlated with personality variation in humans (e.g., Holmes et al., Reference Holmes, Lee, Hollinshead, Bakst, Roffman, Smoller and Buckner2012), potentially reflecting the fact that gross volumetric measures of structures are likely poor indicators of functional neural processes associated with personality. Thus, even in relatively large sample sizes such as the one used in the current study, concerns with regard to power to detect weak associations may be a concern and represents another potential explanation for the lack of association between amygdala GM variation and personality in the current study.
Although the cytoarchitecture of the amygdala and the hippocampus may be difficult to establish in primates in general, it is substantially more challenging in chimpanzees as compared with humans (Bilir et al., Reference Bilir, Craven, Hugg, Gilliam, Martin, Faught and Kuzniecky1998; Freeman, Cantalupo, & Hopkins, Reference Freeman, Cantalupo and Hopkins2004). In addition, as a consequence to limitations in delineating structural landmarks of the amygdala and hippocampus in chimpanzees, it is possible that some portions of the amygdala have been included in the hippocampus ROI tracing in previous studies (Freeman, Cantalupo, & Hopkins, Reference Freeman, Cantalupo and Hopkins2004). In order to optimize amygdala/hippocampus discrimination, we adjusted landmark barriers accordingly, as described in the ROI tracing methods. Nonetheless, more clearly defined boundaries to distinguish between the amygdala and hippocampus are necessary for future studies investigating structural variation within these limbic regions. It is further important to note that we chose to use a manual tracing, ROI approach to analyses given the hypothesis-driven nature of the current study as well as the more highly powered nature of this approach. Nonetheless, this approach results in lower exploratory power given the restricted focus on specific ROIs. It will be important for future sufficiently powered research to include whole-brain analyses as well to allow for increased exploratory power.
Finally, as described previously (Latzman, Hecht et al., Reference Latzman, Hecht, Freeman, Schapiro and Hopkins2015), the size of the social groups varied in the current study. It will thus be important for future research to examine the effects of differential social housing on both personality and brain structure. It is important to note, though, that the chimpanzees included in the current study have also lived in larger groups at various points in their lives, as the facilities in which the chimpanzees live occasionally change the housing situation of the apes. The personality rating data presented here therefore represent a culmination of raters’ observations and interactions with the apes potentially across potentially different social settings.
5. Conclusions
Limitations notwithstanding, the current study has a number of important implications. Results confirm the hierarchical structure of personality across species highlighting integrative personality frameworks. In conjunction with a burgeoning personality neuroscience literature in humans (e.g., Allen & DeYoung, Reference Allen and DeYoung2017), as well as previous findings in chimpanzees (e.g., Blatchley & Hopkins, Reference Blatchley and Hopkins2010; Latzman, Hecht et al., Reference Latzman, Hecht, Freeman, Schapiro and Hopkins2015) and monkeys (e.g., Amaral, Reference Amaral2002; Fox, Oler, Tromp, Fudge, & Kalin, Reference Fox, Oler, Tromp, Fudge and Kalin2015; Machado & Bachevalier, Reference Machado and Bachevalier2008), this research confirms the importance of neuroscientific approaches to the study of basic dispositions (i.e., personality) and suggest comparative neural foundations in both human and nonhuman primates. Although replication is needed, findings further underscore the importance of the hippocampus in the neurobiological foundation of personality, with support for its regulatory role of emotion. All told, results of the current study contribute to a growing literature underscoring the importance of nonhuman primate personality research to elucidating the biological foundation of personality.
Acknowledgments:
Thanks to Bill Hopkins for overseeing data collection efforts and facilitating access to the National Chimpanzee Brain Resource (NCBR), to Jared Taglialatela for his role in amygdala quantification procedures, and to Sara Skiba for assistance with references, tables and formatting.
Financial Support:
This research was supported in part by National Institutes of Health Grant (grant number MH92923) and by a Georgia State University Brains & Behavior seed grant. The chimpanzees comprising the NCCC are supported by NIH Cooperative Agreement U42 OD-011197.
Conflicts of Interest:
The authors have nothing to disclose.
Supplementary Material:
To view supplementary material for this article, please visit https://doi.org/10.1017/pen.2018.1




