Introduction
The global treatment gap for anxiety disorders is well established (Alonso et al., Reference Alonso, Liu, Evans-Lacko, Sadikova, Sampson and Chatterji2018; Roberge, Fournier, Duhoux, Nguyen, & Smolders, Reference Roberge, Fournier, Duhoux, Nguyen and Smolders2011; Stein et al., Reference Stein, Roy-Byrne, Craske, Campbell-Sills, Lang, Golinelli and Sherbourne2011), and the benefits that would derive from the scaling-up of evidence-based treatments are only starting to gain traction in a population health perspective (Chisholm et al., Reference Chisholm, Sweeny, Sheehan, Rasmussen, Smit, Cuijpers and Saxena2016). As the sixth leading cause of years of life lived with disability in both high- and low-income countries, the burden of anxiety disorders can be explained by high prevalence rates, compounded with an early age of onset, comorbidity, and recurrent or chronic course (Baxter, Vos, Scott, Ferrari, & Whiteford, Reference Baxter, Vos, Scott, Ferrari and Whiteford2014). Anxiety disorders account for reduced health-related quality of life, work participation and productivity, and increased social welfare and health service expenditures (Chisholm et al., Reference Chisholm, Sweeny, Sheehan, Rasmussen, Smit, Cuijpers and Saxena2016; Olatunji, Cisler, & Tolin, Reference Olatunji, Cisler and Tolin2007; Sherbourne et al., Reference Sherbourne, Sullivan, Craske, Roy-Byrne, Golinelli, Rose and Stein2010).
Cognitive-behavior therapy (CBT) is the most research-supported psychotherapy for the treatment of anxiety disorders (Butler, Chapman, Forman, & Beck, Reference Butler, Chapman, Forman and Beck2006; Craske & Stein, Reference Craske and Stein2016; Cuijpers, Cristea, Karyotaki, Reijnders, & Huibers, Reference Cuijpers, Cristea, Karyotaki, Reijnders and Huibers2016; Katzman et al., Reference Katzman, Bleau, Blier, Chokka, Kjernisted, Van Ameringen and Walker2014; Norton & Price, Reference Norton and Price2007), and patients report a preference for psychotherapy (McHugh, Whitton, Peckham, Welge, & Otto, Reference McHugh, Whitton, Peckham, Welge and Otto2013). A key issue in scaling-up treatments is to improve the dissemination and uptake of CBT (Shafran et al., Reference Shafran, Clark, Fairburn, Arntz, Barlow, Ehlers and Wilson2009). To overcome challenges in providing access to a high-intensity psychotherapy with a therapist competent in diagnosis-specific CBT, transdiagnostic approaches to CBT (tCBT) that can be applied to diagnostically heterogeneous groups of patients with anxiety disorders could be more conducive to widespread dissemination and implementation (Barlow et al., Reference Barlow, Farchione, Bullis, Gallagher, Murray-Latin, Sauer-Zavala and Cassiello-Robbins2017; Craske, Reference Craske2012; Gunter & Whittal, Reference Gunter and Whittal2010; Norton & Roberge, Reference Norton and Roberge2017). Trials of tCBT interventions delivered in Internet-based, face-to-face individual, or group modalities have yielded similar efficacy data to those reported for diagnosis-specific CBT protocols (Barlow et al., Reference Barlow, Farchione, Bullis, Gallagher, Murray-Latin, Sauer-Zavala and Cassiello-Robbins2017; Newby, McKinnon, Kuyken, Gilbody, & Dalgleish, Reference Newby, McKinnon, Kuyken, Gilbody and Dalgleish2015; Norton & Philipp, Reference Norton and Philipp2008; Pearl & Norton, Reference Pearl and Norton2017; Reinholt & Krogh, Reference Reinholt and Krogh2014).
In community-based mental health care, a group tCBT modality could be of interest to facilitate group formation for mixed anxiety disorders, address psychiatric comorbidity, promote efficient use of professional resources, and abridge therapist training (Norton & Roberge, Reference Norton and Roberge2017). The efficacy of group tCBT has been established in multiple open, benchmarking, and randomized controlled trials (RCT), demonstrating equivalent efficacy across anxiety diagnoses, equivalent outcomes for principal anxiety disorder diagnosis in comparison to diagnosis-specific CBT, and superior outcomes to diagnosis-specific CBT for comorbid anxiety disorders (Erickson, Janeck, & Tallman, Reference Erickson, Janeck and Tallman2007; Norton, Reference Norton2008, Reference Norton2012a; Norton et al., Reference Norton, Barrera, Mathew, Chamberlain, Szafranski, Reddy and Smith2013; Norton & Barrera, Reference Norton and Barrera2012; Norton & Hope, Reference Norton and Hope2005; Schmidt et al., Reference Schmidt, Buckner, Pusser, Woolaway-Bickel, Preston and Norr2012; Wuthrich & Rapee, Reference Wuthrich and Rapee2013). Among the RCTs published on group tCBT, most were explanatory trials designed to examine efficacy in specialized clinical settings. Only one study of group tCBT was reported as pragmatic, and showed a medium effect size compared with a waitlist condition (Erickson et al., Reference Erickson, Janeck and Tallman2007). In recent years, there has been a call for more pragmatic trials with a focus on effectiveness in routine clinical practice conditions (Loudon et al., Reference Loudon, Treweek, Sullivan, Donnan, Thorpe and Zwarenstein2015; Zwarenstein et al., Reference Zwarenstein, Treweek, Gagnier, Altman, Tunis, Haynes and Moher2008). To our knowledge, no controlled effectiveness trial of group tCBT has been published examining outcomes in real-world settings with treatment-as-usual (TAU) as a comparison to inform policy and clinical decision making.
Aim of the study
We designed this pragmatic trial to examine the effectiveness of group tCBT in community-based care to reduce anxiety symptoms of adults with anxiety disorders, as a complement to TAU. Primary question: When group tCBT is added to TAU in primary care for a mixed sample of patients with panic disorder, agoraphobia, social anxiety disorder, and/or generalized anxiety disorder, is it more effective in reducing self-reported and clinician-rated anxiety severity than TAU alone? The main hypothesis is that group tCBT + TAU will be more effective than TAU alone, showing the superiority of the tCBT intervention with usual care.
Method
Study design
This was a multisite, single-blind, patient-level, two-arm pragmatic superiority randomized trial in community-based care settings within three regions in Québec, Canada. The protocol was published (Roberge et al., Reference Roberge, Provencher, Gosselin, Vasiliadis, Gaboury, Benoit and Norton2018).
Study setting
The study was conducted in three integrated health and social services centers purposefully selected based on diversity (e.g. population size, university teaching hospital) to support the generalizability of findings: Quebec City and adjacent areas (i.e. urban and rural population of 737 000 inhabitants); Estrie region (i.e. urban and rural population of 474 000 inhabitants); Laval City (i.e. predominantly urban population of 435 000 inhabitants). The universal health insurance system in Québec (Canada) provides health care and social services coverage for the population, but there is heterogeneity in mental health care delivery in both the public and private sectors. In each region, the study was conducted in collaboration with community-based primary mental health care teams that act as a gateway to specialized mental health care. They contributed in-kind support to the study, including public sector psychologists and psychotherapists as co-therapists and office space for assessment and treatment delivery.
Participants
Eligibility criteria were broad for anxiety severity, psychiatric comorbidity, diagnosis status, and ongoing treatments to include typical community-based care patients. Potential participants were required to meet the following inclusion criteria: (1) aged 18–65 (upper age limit added 18 November 2016); (2) fluent in spoken and written French; (3) meeting Diagnostic and Statistical Manual of Mental Disorders (DSM-5) diagnostic criteria for at least one of the following principal anxiety disorders (American Psychiatric Association, 2013): panic disorder, agoraphobia, social anxiety disorder and generalized anxiety disorder according to a clinical severity rating (CSR) ⩾4 for the Anxiety and Related Disorders Interview Schedule for DSM-5 (ADIS-5) (Brown & Barlow, Reference Brown and Barlow2014). We excluded patients with active suicidal intent, psychosis, bipolar disorder, substance-related and addictive disorders, cognitive impairment, and consultation with a psychiatrist in the past 12 months. Exclusion criteria were based on the assumption that patients with these clinical profiles would not typically be referred to group tCBT in routine care. In the Quebec health care system, patients consulting a psychiatrist for common mental disorders are accessing specialized mental health care and would not be likely candidates for group tCBT provided in community-based care.
Participants were recruited through advertisements in newspapers, geo-targeted Facebook and Google AdWords, and bulletin boards in the community. The recruitment strategy aimed at recruiting typical participants that would seek treatment in community-based care for anxiety symptoms, with heterogeneity in diagnosis, treatment status, and social determinants of health (e.g. employment, education, access to health services). As one out of five primary care patients with anxiety disorders does not have a family physician (Roberge, Fournier, Menear, & Duhoux, Reference Roberge, Fournier, Menear and Duhoux2014), recruitment was not linked to clinician follow-up in medical clinics. We proceeded with a three-stage recruitment process. Filter 1: Self-referred individuals completed an online screening survey that included brief eligibility questions (e.g. age, region), mental disorder diagnosis by a professional, screening questions for common mental disorders, as well as the Generalized Anxiety Disorder-7 (GAD-7; cut point value ⩾8) (Spitzer, Kroenke, Williams, & Löwe, Reference Spitzer, Kroenke, Williams and Löwe2006), the two-item Patient Health Questionnaire (PHQ-2; Kroenke, Spitzer, & Williams, Reference Kroenke, Spitzer and Williams2003) and the CAGE (Ewing, Reference Ewing1984) for the presence of possible alcohol abuse and dependence. Filter 2: Individuals meeting basic eligibility criteria were contacted by clinical evaluators (trained Ph.D. students in psychology) for a brief telephone-screening interview. Filter 3: Face-to-face baseline assessment (T 0) interviews were conducted with potential candidates to assess their eligibility based on the ADIS-5 (Brown & Barlow, Reference Brown and Barlow2014). Patients meeting eligibility criteria were given self-reported questionnaires to complete.
Randomization and masking
Participants meeting eligibility criteria were randomly assigned to group tCBT + TAU or TAU. Randomization at the patient level was stratified by site (Estrie, Québec, Laval), with blocks of four, and carried out using a code generated by statistical software with a ratio of 1:1. At each of the three sites, participants were sequentially enrolled into the trial and tCBT groups began as soon as at least eight participants were randomized into the tCBT condition. Allocation concealment was ensured by an online computer tool for clinical trials that only released the randomization code to the principal investigator (PR) and was then conveyed to the research coordinator who informed participants by telephone. Although blinding of participants and therapists was not possible, concealment of treatment allocation was maintained for the clinical evaluators, research team, and data analyst. Participants were systematically asked not to discuss group participation with clinical evaluators.
Procedures
The group tCBT program (Norton, Reference Norton2012b) is based on extensive theoretical and empirical evidence highlighting common transdiagnostic mechanisms, processes, and maintenance factors across anxiety disorders (Barlow, Allen, & Choate, Reference Barlow, Allen and Choate2004; Norton & Paulus, Reference Norton and Paulus2017). The treatment protocol for therapists included four components: education and self-monitoring, specific cognitive restructuring (i.e. identify and challenge anxiety thoughts), graduated exposure and response prevention, and generalized cognitive restructuring (i.e. focus on more general neurotic style). Groups encompassed weekly 2 h sessions with 8–10 patients during a 12-week period. Patients received a structured workbook at the first session, comprising psychoeducation material of the nature of anxiety and treatment components, monitoring and exercise forms, as well as weekly homework. A telephone individual contact was established with each participant by one of the two therapists prior to the first session to introduce the treatment. During that initial contact, a preliminary exposure hierarchy was elaborated with each participant and written down on a specific form. Prior to conducting the first exposure session (session 4), this form was revised and updated for each participant. Principal therapists were Ph.D. level psychologists with at least 2 years of clinical experience with CBT to ensure basic competence for treatment fidelity. Co-therapists were registered psychologists or psychotherapists selected by the health care managers at each study site to reflect the range of clinical backgrounds in the public sector. They shared the responsibility of leading therapists for treatment delivery. In case of feasibility issues preventing the contribution of public sector therapists, Ph.D. students in psychology would act as a substitute for group treatment delivery.
Initial training was provided with a 2-day workshop with the tCBT protocol developer (PJN), and tailored individual training was provided to therapists that joined the project after the workshop (MDP). Supervision (MDP, PG), in the form of case-discussion via telephone, was provided at predefined times during the delivery of the intervention (pre-treatment, between sessions 3–4, 6–7, 10–11, post-treatment). Case discussion material was brought-up by the clinicians themselves. Treatment integrity data were not accessed by supervisors during the trial to provide remediation strategies to therapists, a distinction from research-grade supervision in explanatory trials. Therapeutic treatment adherence and competence was only assessed at the end of the trial by a random review of 33% of audio recordings of treatment sessions with an integrity rating scale (Norton & Barrera, Reference Norton and Barrera2012). Based on a five-point scale tailored to each component of the protocol, a mean score of 3 or below on a given session was considered ‘out of protocol’, with scores on specific items varying between ‘no or ineffective discussion’ (score of 1) to ‘extremely effective discussion’ (score of 5).
No restrictions were imposed regarding TAU as the trial prioritized the assessment of the added value of tCBT to real-world conditions. Therefore, no restrictions were introduced following enrolment regarding healthcare utilization or treatment of mental disorders (e.g. starting a new medication, dosage change, psychotherapy). Data on service utilization and psychotropic medication were collected throughout the trial. Due to variations in the medical follow-up of participants with our recruitment strategy, clinicians were not informed of study participation by the research team.
Participants randomly assigned to the TAU-only condition were offered tCBT on completion of the study (12-months delay following baseline).
Outcomes
Assessment of participants was conducted at 4 months (T 1; post-treatment, in person), 8 months (T 2; telephone) and 12 months (T 3; in person) with both patient- and clinician-reported measures. They comprised the self-reported Beck Anxiety Inventory (BAI) (Beck, Epstein, Brown, & Steer, Reference Beck, Epstein, Brown and Steer1988) and the CSR of the ADIS-5 (Brown & Barlow, Reference Brown and Barlow2014) for the principal anxiety disorder, ranging on a scale from 0 (no symptoms) to 8 (extremely severe symptoms) (added 12 December 2017). Sociodemographic and health care utilization data were also collected, as well as diagnosis-specific measures (e.g. PHQ-9; Kroenke, Spitzer, & Williams, Reference Kroenke, Spitzer and Williams2001), functioning measures (e.g. Sheehan Disability Scale; Sheehan, Harnett-Sheehan, & Raj, Reference Sheehan, Harnett-Sheehan and Raj1996), and therapy process measures (described in the protocol; Roberge et al., Reference Roberge, Provencher, Gosselin, Vasiliadis, Gaboury, Benoit and Norton2018). Participants received a $20 compensation for in-person assessments. Data collection was managed independently of treatment assignment by clinical evaluators who were blind to the participant's treatment assignment.
Statistical analysis
Because no prior pragmatic trials of group tCBT compared to TAU have been conducted, we conservatively estimated sample size by assuming an estimated effect size of the intervention (Cohen's d) of 0.45 at post-treatment, with a power of 0.8, an adjusted α value of 0.03 to account for two primary outcomes (James, Reference James1991) (estimated correlation of 0.7 based on pilot data), and a two-tail independent univariate comparison. The estimated sample size was 182 individuals and, with an estimated 15% rate of loss at follow-up factored in, the proposed final sample size was thus 215 patients (107 and 108 for each arm, respectively).
Intention-to-treat analyses were computed with all available data, by randomization condition, and without considering adherence. Linear mixed regression models (variance components correlation matrix) were constructed with maximum-likelihood estimation techniques to evaluate the effect of the intervention over time on primary outcomes. To control for intra-group and intra-site variability of therapy, a random effect was added on the participants nested in the therapy groups, nested in the three sites. Another random effect on the therapy groups nested in the three sites was added to control for the variability between groups. The effects of intervention were adjusted at baseline for comorbid depressive symptoms (PHQ-9 score), psychotropic medication (yes/no), and principal anxiety disorder (ADIS-5). Estimates and confidence intervals were computed with Bootstrap resampling (R = 1000) to account for disparity from normal distribution of the model residuals. All analyses were conducted with all available data without imputation, as estimation of parameters using the maximum-likelihood method is considered adequate to address missing data at post-treatment (T 1) in multilevel models (Little & Rubin, Reference Little and Rubin2019; van Buuren, Reference van Buuren2012). Cohen's d effect sizes were generated within (pre-post) and between conditions for both BAI and principal diagnosis ADIS CSR (Kadel & Kip, Reference Kadel and Kip2012). Treatment response and high end-state functioning status were compared between conditions using χ2 tests. A sensitivity analysis was conducted ‘per protocol’. A linear mixed regression model from baseline (T 0) to 12 months (T 3) was also conducted to examine the maintenance of gains over time. Statistical analyses were conducted with SAS version 9.4 (PROC MIXED procedure for regression models).
Results
Sample characteristics
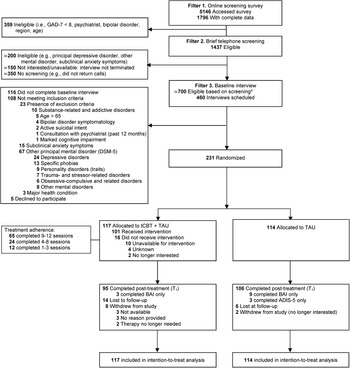
Between 12 September 2016 and 16 February 2018, 344 patients completed a baseline interview, and 231 were randomly assigned to tCBT + TAU or TAU (Site 1, n = 53; Site 2, n = 96; Site 3, n = 82) (Fig. 1). Post-treatment of the last participant recruited was completed on 15 June 2018. The most common principal diagnosis was generalized anxiety disorder (52.8%), followed by social anxiety disorder (29.4%), panic disorder (14.3%), and agoraphobia (3.5%). The majority of participants (82.7%) had at least one comorbid diagnosis, with a median of 2 (range 0–6), including anxiety disorders (74.9%) and major or persistent depressive disorders (28.1%). The interrater agreement for ADIS-5 principal diagnosis was 83.3% (CSR ± 1 criterion). Participant characteristics included a majority of women (85.7%), an average age of 37 years, a university diploma (42.2%), full-time employment (60.6%), and satisfactory economic situation (77.3%). In the past 12 months, 63.6% consulted a general practitioner, 45.5% consulted a psychologist or psychotherapist, and 21.2% other health professionals (e.g. social worker, nurse) for mental health reasons. The majority of patients reported taking a psychotropic medication in the past 12 months (71.9%), including selective serotonin reuptake inhibitors/serotonin–norepinephrine reuptake inhibitors (53.7%) and benzodiazepines (32.5%). Socio-demographic characteristics, clinical severity, service use, and medication were well balanced between conditions at baseline (Table 1), with the exception of a higher proportion of cases with principal generalized anxiety disorder, and a lower proportion of cases with social anxiety disorder or panic disorder in the tCBT + TAU condition. Online Supplementary Table S1 shows baseline data by condition across the three sites.

Fig. 1. Flow of participants through the trial. aFollowing telephone screening, 240 of the ≃700 eligible people did not participate in baseline interview, generally due to lack of availability for upcoming group treatments.
Table 1. Baseline characteristics
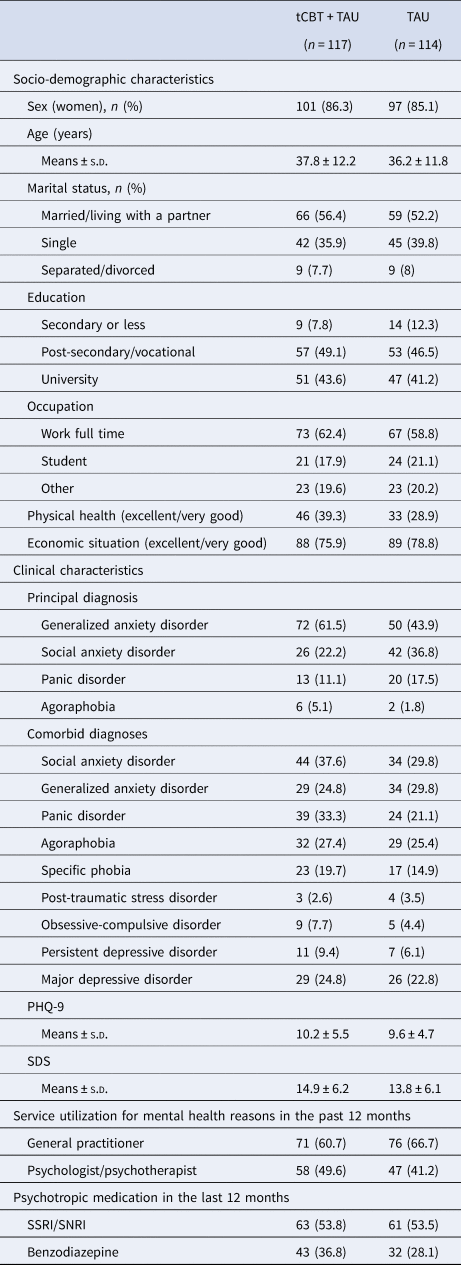
tCBT, transdiagnostic group CBT; TAU, treatment-as-usual; s.d., standard deviation; PHQ-9, Patient Health Questionnaire – depression scale; SDS, Sheehan Disability Scale; SSRI, selective serotonin reuptake inhibitors; SNRI, serotonin–norepinephrine reuptake inhibitors.
Data were presented with frequency (percentage) unless otherwise indicated.
For primary outcome measures, the conditions were balanced on the BAI score and the CSR score for the ADIS principal diagnosis. Online Supplementary Table S2 shows baseline primary outcome data distributed by region and condition. At least one of the two primary endpoints were completed by 95 patients in tCBT + TAU and 106 patients in TAU only. Participants unavailable for follow-up were significantly younger, with a poorer economic situation, a higher proportion of panic disorder and agoraphobia, and a higher BAI score (online Supplementary Table S3).
Treatment adherence
We conducted 12 tCBT groups, with three to five intervention groups conducted in each region. Group composition varied between 7 and 12 participants [mean 10 (s.d. 1.4)]. Treatment adherence was documented for the 117 participants in the intervention condition, with a median of 9 [IQR 4–11] sessions, and 65 (55.6%) were adherent (9–12 sessions), 24 (20.5%) were moderately adherent (4–8 sessions), 12 (10.3%) were non-adherent (1–3 sessions), and 16 (13.7%) did not initiate tCBT. Groups were facilitated by six Ph.D.-level therapists, paired with eight co-therapists from the public sector and two Ph.D. students in psychology. Of the eight therapists from the public sector, two had doctoral-level degrees in psychology, four had a master's degree in clinical psychology, and two had a bachelor's degree in social work. Most therapists (n = 13; 81.3%) took part in the 2-day tCBT training workshop, and all reported having participated in other CBT training activities. Thirteen (81.3%) had previous experience in group therapy facilitation, and eight (50%) had specific experience in group CBT for anxiety. The treatment adherence and competence rating scale indicated a mean (s.d.) of 3.68 (0.55) over a five-point scale.
Analysis of primary outcomes
Linear mixed regression models suggested that 2.6% and 0.8% of the total variation in the BAI score could be accounted for, respectively, by groups (nested into sites) and sites within which participants were enrolled. Similar results were obtained for the ADIS CSR outcome. Table 2 presents the results of the regression models for BAI and ADIS CSR accounting for the clustering structure of the participants (further details in online Supplementary Table S4). Both condition groups improved significantly over time (p < 0.001 for both conditions, for both outcomes). After adjustment for comorbid depressive symptoms, use of psychotropic medication, and principal anxiety disorder, the decrease in the average BAI score over time was significantly greater for tCBT + TAU (difference between groups of 7.80, 95% CI 4.64–11.36; p < 0.001); similarly for the ADIS CSR outcome (difference between groups of 1.87, 95% CI 1.01–2.30; p < 0.001).
Table 2. Primary outcomes and effect size pre-post treatment and between intervention conditions

ADIS-5, Anxiety and Related Disorders Interview Schedule for DSM-5; CSR, clinical severity rating; CI, confidence interval.
Values at baseline and post-treatment presented with means ± standard deviation. Effect sizes presented with Cohen's d (95% confidence interval). For adjusted analysis, bootstrap estimates and 95% CI are presented.
Analysis of secondary outcomes
Effect sizes
The between-condition effect size (Cohen's d) for BAI at post-treatment was 0.76, and 0.79 for the principal diagnosis CSR of the ADIS-5 (Table 2). The within-condition effect size for BAI was 0.89 for tCBT + TAU and 0.24 for TAU, and 1.16 and 0.70, respectively, for the principal diagnosis CSR of the ADIS-5.
Treatment response and high end-state functioning
At post-treatment, both the treatment responder status (CSR < 4; 54.3% v. 17.5%, p < 0.001) and high end-state functioning (CSR ⩽ 2; 30.4% v. 11.3%, p < 0.001) were significantly higher in tCBT + TAU for principal diagnosis at ADIS-5. For BAI, significant reliable improvement (defined as a decrease ⩾10 at T 1; 47.4% v. 21.4%, p < 0.001) and clinically significant improvement (defined as a decrease ⩾10 and a score ⩽17 at T 1; 43.2% v. 15.5%, p < 0.001) were higher in the intervention condition than TAU (Oei & Boschen, Reference Oei and Boschen2009).
Depressive symptoms and disability measures
We also examined secondary outcome measures at post-treatment with multivariate mixed linear models. The decrease in the average scores over time was significantly greater for tCBT + TAU compared to TAU for PHQ-9 (difference between groups of 3.45, 95% CI 1.75–5.12; p < 0.001) as well as Sheehan Disability Scale (difference between groups of 4.67, 95% CI 2.88–6.46; p < 0.001). A between-condition effect size of 0.62 was observed for both measures.
Per protocol sensitivity analysis
Results of the per protocol linear mixed regression models conducted with completers (9–12 sessions) for BAI and ADIS-CSR (Table 2; online Supplementary Table S4) showed a similar pattern of improvement over time favoring tCBT + TAU as in the primary analysis (p < 0.001 for both outcomes).
Maintenance of gains at 12-month follow-up
The adjusted multivariate linear mixed regression models for BAI and ADIS-CSR encompassing all data available from baseline (T 0) to 12 months (T 3) revealed a significant improvement between 4-month (T 1) and 12-month (T 3) follow-up for both conditions, with a significant difference in improvement favoring tCBT + TAU and a condition × time interaction (Fig. 2).
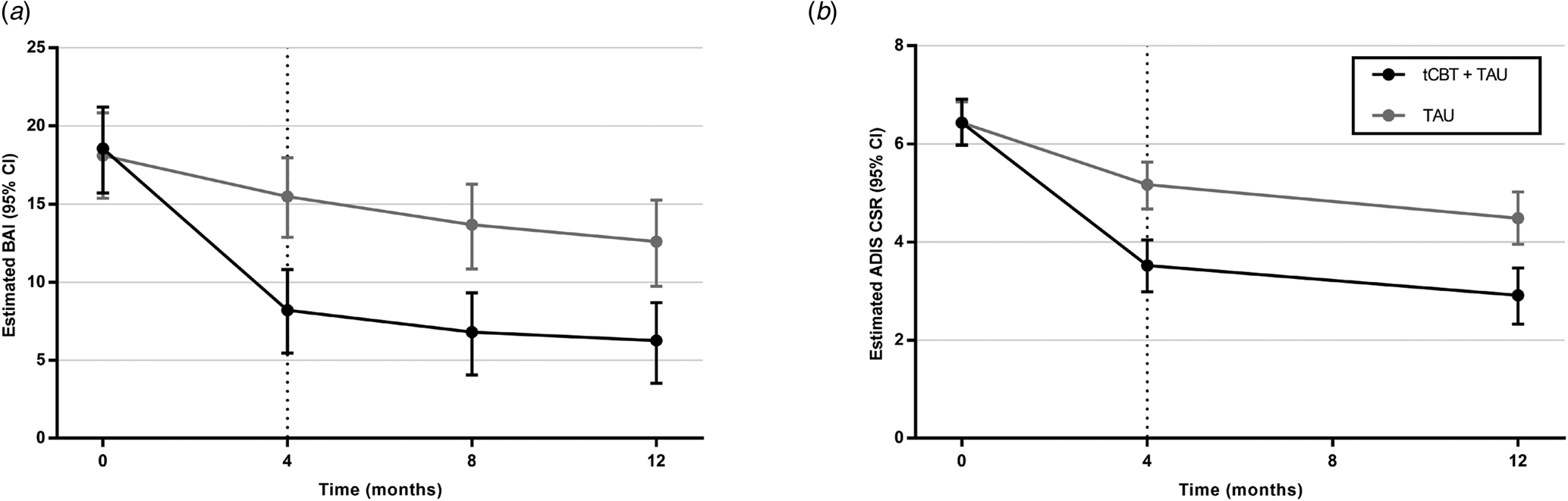
Fig. 2. Estimated scores by baseline treatment assignment. Linear mixed regression models on primary outcomes adjusted for comorbid depressive symptoms (PHQ-9 score), presence (yes/no) of psychotropic medication, and principal anxiety disorder (ADIS-5). Number of patients at baseline, 4, 8, and 12 months were, respectively, 117, 95, 80, and 76 for tCBT + TAU and 114, 103, 90, and 90 for TAU. Bootstrapping was performed to obtain confidence interval estimates. The multivariate linear mixed regression models for BAI and ADIS-CSR found significant improvement over time (p < 0.001), between conditions in favor of tCBT + TAU (p < 0.001) and for interaction (p < 0.001). For BAI and ADIS-CSR, there was a significant difference in each condition between baseline and 4 months (p < 0.001) and a significant difference between 4 and 12 months (p < 0.02). A significant difference between conditions remained at 12 months (p < 0.001).
Mental health service utilization during treatment period
We examined TAU service utilization in both intervention and control conditions during the period between pre-treatment and post-treatment assessments (Table 3). No differences between conditions were revealed regarding the prevalence of service utilization for mental health reasons and psychotropic medication.
Table 3. Health services utilization and medication for mental health reasons during the period between baseline assessment (T 0) and post-treatment assessment (T 1)
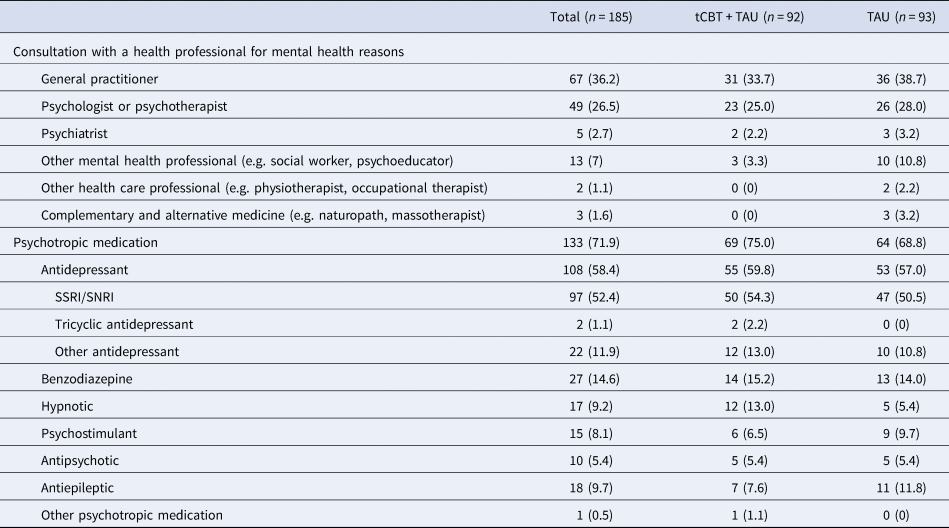
tCBT, transdiagnostic group CBT; TAU, treatment-as-usual; SSRI, selective serotonin reuptake inhibitors; SNRI, serotonin–norepinephrine reuptake inhibitors.
Data were presented with frequency (percentage) unless otherwise indicated.
Discussion
This is the first RCT of group tCBT outside of specialized anxiety disorders clinics and, to our knowledge, the first pragmatic trial of group tCBT with TAU as a comparison. The report of this large-scale trial confirms that incorporating group tCBT into usual care can improve outcomes for patients with mixed anxiety disorders in community-based care. We found a significant difference in self-reported and clinician-rated anxiety symptoms in favor of tCBT, with control for comorbid depressive symptoms, psychotropic medication, and specific principal anxiety disorder. Our findings highlight that tCBT can result in significant improvements for anxiety disorders even when delivered without extensive monitoring of treatment fidelity and adherence, and with broad eligibility criteria and recruitment in the community.
The findings of this study are comparable to several meta-analyses of diagnosis-specific CBT and tCBT RCTs for anxiety disorders, although studies were mostly conducted with explanatory designs. First, in a systematic review of diagnosis-specific CBT for major depression and anxiety disorders (Cuijpers et al., Reference Cuijpers, Cristea, Karyotaki, Reijnders and Huibers2016), overall large effect sizes were found for diagnosis-specific CBT of generalized anxiety disorder (g = 0.80), panic disorder (g = 0.81), and social anxiety disorder (g = 0.88). However, only small-to-moderate effect size estimates were obtained when considering only RCTs with TAU control conditions, which provide a better estimate of CBT effect size than a waiting-list condition (Cuijpers et al., Reference Cuijpers, Cristea, Karyotaki, Reijnders and Huibers2016). A meta-analysis of studies for CBT v. TAU found similar results (g = 0.69) for anxiety disorders (Watts, Turnell, Kladnitski, Newby, & Andrews, Reference Watts, Turnell, Kladnitski, Newby and Andrews2015). Therefore, our findings (Cohen's d = 0.76–0.79) are well within range or even superior for effect size to CBT trials with a TAU condition for specific anxiety disorders. Second, the comparison with two recent meta-analyses of tCBT efficacy trials (g = 0.86, 95% CI 0.75–0.96; g = 1.06, 95% CI 0.88–1.24) showed somewhat inferior between-condition effect sizes (Newby et al., Reference Newby, McKinnon, Kuyken, Gilbody and Dalgleish2015; Pearl & Norton, Reference Pearl and Norton2017). However, the ‘per protocol’ between-condition effect sizes analysis (Cohen's d = 0.84–0.93) suggests that group tCBT can be delivered with similar effects in real-world settings for treatment completers (9–12 sessions).
Among other secondary outcomes, we found that patients in tCBT + TAU were more likely to achieve a clinically meaningful improvement, with approximately half of participants meeting treatment responder status for the ADIS principal anxiety disorder (54.3%) or significant reliable improvement (47.4%) for the BAI. This is concordant with the assessment of functional impairment in work/school, social life, and home life/family responsibilities with the Sheehan Disability Scale that showed a moderate improvement for the tCBT + TAU condition. There was also a marked improvement in depressive symptoms, supporting previous findings that tCBT could be advantageous to treat comorbid disorders (Norton et al., Reference Norton, Barrera, Mathew, Chamberlain, Szafranski, Reddy and Smith2013). We also observed maintenance of gains following treatment, as seen in Barlow's unified protocol (Barlow et al., Reference Barlow, Farchione, Bullis, Gallagher, Murray-Latin, Sauer-Zavala and Cassiello-Robbins2017; Bullis, Fortune, Farchione, & Barlow, Reference Bullis, Fortune, Farchione and Barlow2014), and also a significant improvement over time from post-treatment to 12 months for both BAI and ADIS-5 CSR.
The sample was characterized by a heterogeneous population with high rates of psychiatric comorbidity, help-seeking behaviors for mental health reasons, and psychotropic medication use. The broad eligibility criteria did not intend to exclude potentially non-adherent patients due to complex clinical profiles, poor motivation, or psychosocial disadvantage. Rates of mental health care were higher than expected in the 12 months prior to the study, with 45% reporting at least one consultation with a psychologist or psychotherapist and 75% reporting a psychotropic medication. Service utilization at baseline suggests either suboptimal quality of care or a predominance of chronic cases in our sample. There was a higher proportion of participants with a principal generalized anxiety disorder (52.8%) and a lower proportion with agoraphobia (3.5%) in our sample than expected considering prevalence rates of anxiety disorders, which should be considered in the interpretation of the findings. This could be related to the use of the GAD-7 (Spitzer et al., Reference Spitzer, Kroenke, Williams and Löwe2006) as a screening instrument or to differential volunteering patterns for group-based CBT interventions according to clinical profiles (e.g. apprehension in social situations). In terms of sociodemographic characteristics, the sample was predominantly women, and the majority reported a satisfactory economic situation and a post-secondary degree, which could have implications for the generalizability of findings. Replication in samples more consistent with the general population is warranted.
In our trial, tCBT was shown effective beyond existing treatments accessible and used by participants in the current health care context. The TAU-only condition experienced a small (BAI; d = 0.24) to moderate improvement in anxiety outcomes (ADIS CSR for principal diagnosis; d = 0.70). With the absence of boundaries on service utilization and medication, in contrast with most CBT trials, an issue to consider is uncertainty regarding differential behavioral changes that may have been prompted by the recruitment process (e.g. motivation, mental health assessment, knowledge about CBT, or waiting for delayed intervention in the controls). Following enrolment, we observed highly varied self-reported service utilization and medication for TAU in both conditions. As there were no differences in the prevalence of co-interventions for psychotherapy, general practitioner, psychiatrists, psychotropic medication, or other mental health care professionals following enrolment, the presence of co-interventions appears favorable to the generalizability of findings. This is important due to increasing trends in the use of psychotropic medication (Kantor, Rehm, Haas, Chan, & Giovannucci, Reference Kantor, Rehm, Haas, Chan and Giovannucci2015).
This trial adopted a flexible stance on the delivery and adherence of the tCBT intervention. The intervention was delivered by co-therapists with varying levels of CBT experience, with a brief training in tCBT and case discussions. Therapeutic integrity was assessed, but there were no remediation strategies to improve fidelity throughout the trial, as the integrity data were analyzed once the trial was over and was not used to inform case discussion material. Overall, therapists were reasonably adherent to the treatment model, as expected for a pragmatic trial. In terms of flexibility in treatment adherence by participants, only 56% were considered adherent (completed >8 sessions). Although these rates of engagement are consistent with those reported in studies of diagnosis-specific group CBT in community settings (Heatherington et al., Reference Heatherington, Harrington, Harrington, Niemeyer, Weinberg and Friedlander2014), improving patient engagement for CBT, particularly in groups, should be a priority in clinical research. These interrelated factors may have caused an underestimation of treatment effects and suggest that optimization of tCBT delivery could be achievable for enhanced patient outcomes. Furthermore, it is unclear why specific variables (e.g. panic disorder, lower age, lower income) were associated with loss to T 1 follow-up, although Issakidis and Andrews (Reference Issakidis and Andrews2004) have noted that studies of treatment dropout in CBT for anxiety disorders tend to show conflicting sociodemographic and clinical predictors.
Among limitations of the study, the randomization appeared successful to balance conditions on most socio-demographic, clinical, and health service utilization baseline data, but it should be noted that there was an imbalance in principal anxiety disorder between conditions. Furthermore, we tried to optimize trial retention with strategies such as minimal data collection for risk of dropout (e.g. BAI only), but this was only partially successful, and attrition is a potential cause for bias. Additionally, clinical evaluators were uninformed of treatment assignment throughout the trial but blinding of participants was not possible, and this may have introduced a performance bias. Another consideration is the choice of primary outcome measures, as current diagnosis-specific and global anxiety assessment tools cannot fully address the transdiagnostic assessment of multiple anxiety disorders (Norton & Roberge, Reference Norton and Roberge2017; Smith, Ratcliff, & Norton, Reference Smith, Ratcliff, Norton, Brown and Clark2014). We tried to alleviate this limitation by using two complementary primary outcome measures.
In conclusion, our study showed that the addition of tCBT was more effective than TAU in a community-based primary mental health care context. The study demonstrates that a single group tCBT protocol could be delivered in real-world community-based settings with positive clinically relevant results, which could facilitate training and group formation in the implementation of evidence-based treatments. Furthermore, our findings were obtained in patients with complex clinical profiles and previous experience of mental health service utilization. Future research should examine the potential for improved treatment effects through enhanced delivery support for treatment integrity and adherence. Our findings suggest that group tCBT might be a valuable evidence-based psychotherapy approach to implement at the population level to improve the outcomes of patients with anxiety disorders.
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1017/S0033291720004316
Acknowledgement
We would like to acknowledge the contribution of the three study sites in Quebec City, Laval and Sherbrooke for the essential support to the conduct of the trial. The three study sites provided in-kind contributions through the involvement of public sector psychologists and psychotherapists as co-therapists in tCBT treatment, the provision of office space in primary care settings for participants' assessment and treatment delivery and administrative support. We are grateful to all the patients that took part in the study. We would like to thank all the clinical evaluators and therapists who contributed to the recruitment and delivery of the intervention to study participants.
Author contributions
Roberge, Benoît, and Carrier had full access to all of the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis. Concept and design: Roberge, Provencher, Norton. Acquisition, analysis, or interpretation of data: Roberge, Provencher, Norton, Carrier, Gaboury, Benoît. Drafting of the manuscript: Roberge, Carrier, Provencher, Norton, Gaboury. Critical revision of the manuscript for important intellectual content: Gosselin, Gaboury, Vasiliadis, Benoît, Antony, Chaillet, Houle, Hudon. Statistical analysis: Carrier. Obtained funding: Roberge, Provencher, Norton, Gosselin, Gaboury, Vasiliadis, Antony, Chaillet, Houle, Hudon. Administrative, technical, or material support: Benoît. Study supervision: Roberge, Provencher.
Financial support
The Canadian Institutes of Health Research funded this study (CIHR grants #334091 and 340410). The funder of the study had no role in study design, data collection, data analysis, data interpretation, or writing of the report. The corresponding author had full access to all the data in the study and had final responsibility for the decision to submit for publication.
Conflict of interest
PJN receives royalties from Guilford Press for sales of ‘Group Cognitive Therapy of Anxiety: A Transdiagnostic Treatment Manual’ (Norton, Reference Norton2012b). All other authors declare no competing interests.
Ethical standards
The protocol was approved by the ethics review boards of the Integrated Health and Social Services Centers in Estrie (#MP-22-2016-570), Québec City (#2017-166), and Laval (#2016-2017-C54). All participants provided written informed consent.