Introduction
Dementia refers to a group of progressive brain disorders that result in multiple cognitive and behavioural impairments (Ballard et al. Reference Ballard, Gauthier, Corbett, Brayne, Aarsland and Jones2011). Enabling people with dementia to ‘live well’ with the condition is a priority for policy and practice (Department of Health, 2009). Living well with chronic illness and disability is viewed by the Institute of Medicine (2012) as ‘the best achievable state of health that encompasses all dimensions of physical, mental and social well-being’ such that ‘to live well takes on a unique and equally important personal meaning, which is defined by a self-perceived level of comfort, function and contentment with life’ (p. 32). It is important to gain a clear understanding of the extensive range of factors that may influence ability to live well with dementia in order to identify potential targets for intervention.
In instances where it has been discussed explicitly (e.g. Small, Reference Small2007), the concept of ‘living well’ with dementia has been equated with experiencing a good quality of life (QoL). Living well can however mean more to an individual than current QoL. A sense of subjective well-being can be also associated with living well. Likewise, living well can encompass the experience of a life that is and has been lived well – life satisfaction. The constructs of QoL, subjective well-being and life satisfaction are inter-related but encompass distinct elements (Clare et al. Reference Clare, Nelis, Quinn, Martyr, Henderson and Hindle2014a).
Subjective well-being refers to both a positive cognitive appraisal of one's current situation and the experience of an appropriate balance of positive and negative emotions (Diener & Chan, Reference Diener and Chan2011). In line with this definition, Kitwood's model of personhood (Reference Kitwood1997) emphasises the way in which interactions and environments contribute to enhancing or detracting from well-being for people with dementia. Subjective perceptions of satisfaction with life reflect the experience of meaning and purpose, the ability to experience continued personal growth, a sense of being in control of one's life, active social participation and happiness (St. John & Montgomery, Reference St. John and Montgomery2010).
QoL is sometimes conceptualised specifically in relation to health (health-related QoL, HRQoL) (Albert et al. Reference Albert, Jacobs, Sano, Marder, Bell and Devanand2001). This is a narrow formulation, however, and given the pervasive effects of dementia, there is likely to be considerable overlap between specific HRQoL and more generic elements of QoL (Wilson & Cleary, Reference Wilson and Cleary1995). The World Health Organisation (WHO) defines QoL much more broadly as ‘an individual's perceptions of their position in life in the context of the culture and value systems in which they live and in relation to their goals, expectations, standards and concerns. It is a broad ranging concept affected in a complex way by a person's physical health, psychological state, level of independence social relationships and their relationship to salient features of their environment’ (p. 1) (World Health Organization, 1995). There is no single accepted theoretical model underlying the conceptualisation and assessment of QoL in dementia, but the most influential formulation is Lawton's dementia-specific framework (Reference Lawton1994), which recognises that adequate measurement of QoL requires evaluation of multiple dimensions of a person's life, presenting a broad conceptualisation that accords well with the WHO definition.
Life satisfaction and subjective well-being have not been studied in depth in people with dementia, with a few important exceptions (Zank & Leipold, Reference Zank and Leipold2001). QoL has been investigated more substantially, but it has been argued that nevertheless little is known about the QoL of people with dementia, particularly those with more severe conditions (Kane et al. Reference Kane, Kling, Bershadsky, Kane, Giles and Degenholtz2003; Banerjee et al. Reference Banerjee, Samsi, Petrie, Alvir, Treglia and Schwam2009; Cordner et al. Reference Cordner, Blass, Rabins and Black2010), or about whether QoL changes over time as dementia severity increases. In contrast, QoL can be assessed using either generic or dementia-specific measures, of which there are several available (Ready & Ott, Reference Ready and Ott2003; Ettema et al. Reference Ettema, Dröes, de Lange, Mellenbergh and Ribbe2005; Bowling et al. Reference Bowling, Rowe, Adams, Sands, Samsi and Crane2015; Algar et al. Reference Algar, Woods and Windle2016; Missotten et al. Reference Missotten, Dupuis and Adam2016). Although many of these measures share content themes and conceptual underpinnings (Missotten et al. Reference Missotten, Dupuis and Adam2016), different measures may yield different results. These methodological limitations and complexities make it difficult to draw clear conclusions about factors associated with aspects of living well with dementia.
While life satisfaction is typically assessed by self-report and well-being is typically assessed by informant ratings, QoL in contrast may be assessed either directly by self-report or by means of informant or proxy ratings made by family or health care professionals. Self-report by people with mild (Woods et al. Reference Woods, Nelis, Martyr, Roberts, Whitaker and Markova2014) and moderate-to-severe dementia (Hoe et al. Reference Hoe, Katona, Roch and Livingston2005) has been shown to be reliable, though reliability may depend on which measure is used (Ready & Ott, Reference Ready and Ott2003) and dementia severity. Comparison of self- and informant ratings in situations where both can be obtained indicates that informant ratings are typically more negative than self-ratings (Beer et al. Reference Beer, Flicker, Horner, Bretland, Scherer and Lautenschlager2010; Moyle & O'Dwyer, Reference Moyle and O'Dwyer2012; Crespo et al. Reference Crespo, Hornillos and Bernaldo de Quiros2013), and this has implications for how we evaluate informant reports of QoL in people with very severe dementia where only informant or proxy ratings are possible. Furthermore, different informants may have different perspectives; for example, ratings made by family carers and paid/professional carers appear to be associated with different factors (Clare et al. Reference Clare, Quinn, Hoare, Whitaker and Woods2014b; Edelman et al. Reference Edelman, Fulton, Kuhn and Chang2005).
Previous reviews have focused on specific aspects of QoL in dementia, including dementia-specific HRQoL (Banerjee et al. Reference Banerjee, Samsi, Petrie, Alvir, Treglia and Schwam2009), measurement (Ready & Ott, Reference Ready and Ott2003; Ettema et al. Reference Ettema, Dröes, de Lange, Mellenbergh and Ribbe2005; Bowling et al. Reference Bowling, Rowe, Adams, Sands, Samsi and Crane2015; Algar et al. Reference Algar, Woods and Windle2016; Missotten et al. Reference Missotten, Dupuis and Adam2016), residential care (Lawrence et al. Reference Lawrence, Fossey, Ballard, Moniz-Cook and Murray2012; Moyle & O'Dwyer, Reference Moyle and O'Dwyer2012; Beerens et al. Reference Beerens, Zwakhalen, Verbeek, Ruwaard and Hamers2013; Robertson et al. Reference Robertson, Cooper, Hoe, Hamilton, Stringer and Livingston2017), qualitative accounts (O'Rourke et al. Reference O'Rourke, Duggleby, Fraser and Jerke2015a), and a recent narrative overview (Jing et al. Reference Jing, Willis and Feng2016). To date, however, there has been no comprehensive systematic review and meta-analysis of factors associated with QoL, well-being and life satisfaction for people with dementia. A careful and comprehensive review is needed to summarise the evidence. In this systematic review and meta-analysis, we examined evidence from quantitative observational studies in order to identify factors associated with QoL, well-being and life satisfaction in people with dementia.
Method
Search strategy
We searched PubMed, CINAHL, Web of Science, PsycNET, AgeInfo, Zetoc, Social Care Online and OpenGrey for English-language publications until 7 January 2016. The search comprised two strings. The first string comprised: (well-being OR wellbeing OR life satisfaction OR quality of life OR *QoL* OR Health Status OR ADRQL OR Apparent Emotion Scale OR BASQID OR CDQLP OR Discomfort Scale OR Duke Health Profile OR DHP OR EQ-5D OR Health Utilities Index OR HUI* OR Nottingham Health Profile OR NHP OR Pleasant Events Schedule-AD OR Progressive Deterioration Scale OR PWB-CIP OR SF-12 OR SF-36 OR ICECAP OR QUALIDEM OR QUALID) AND (dementia OR Alzheimer* OR Parkinson* OR Lewy OR Fronto). The second search string was designed to identify articles in residential care settings that may not mention dementia in the title or abstract and comprised: (well-being OR wellbeing OR life satisfaction OR quality of life OR *QoL* OR health status) AND (nursing home OR long term care OR assisted living OR residential living OR care home OR respite OR day care). Names of specific measures used in the searches were derived from two previous reviews (Ready & Ott, Reference Ready and Ott2003; Ettema et al. Reference Ettema, Dröes, de Lange, Mellenbergh and Ribbe2005). No date restrictions were imposed. We examined reference lists of review articles identified in the searches and searched online for additional publications by first authors of included studies. The review protocol was registered with PROSPERO: CRD42014013633.
Inclusion and exclusion criteria
The population of interest was people with a diagnosis of dementia, irrespective of type, severity, age or living situation. We included observational studies reporting cross-sectional and/or longitudinal associations between specific variables and scores on a standardised questionnaires or rating scales measuring QoL, life satisfaction or well-being. We excluded studies where fewer than 75% of participants had dementia, and intervention studies that did not provide baseline data.
Procedure
Figure 1 shows the article selection process. Title, abstract and full-text screening were conducted by two independent reviewers using a structured proforma, and any disagreements were referred to a third individual for resolution. Online Supplementary Table S1 provides a list of studies excluded at the full-text screening stage, with reasons. Study quality was assessed by two independent reviewers using a checklist adapted from existing measures (DuRant, Reference DuRant1994; Downs & Black, Reference Downs and Black1998; Zaza et al. Reference Zaza, Wright-De Agüero, Briss, Truman, Hopkins and Hennessy2000; Sanderson et al. Reference Sanderson, Tatt and Higgins2007); see online Supplementary Table S2. Scores from the quality assessment of each article were summed; articles that scored 19 or lower were judged to be of poor quality, articles that scored between 20 and 25 were judged to be of satisfactory quality and articles that scored 26 or greater were judged to be of good quality. A structured proforma was used for data extraction.
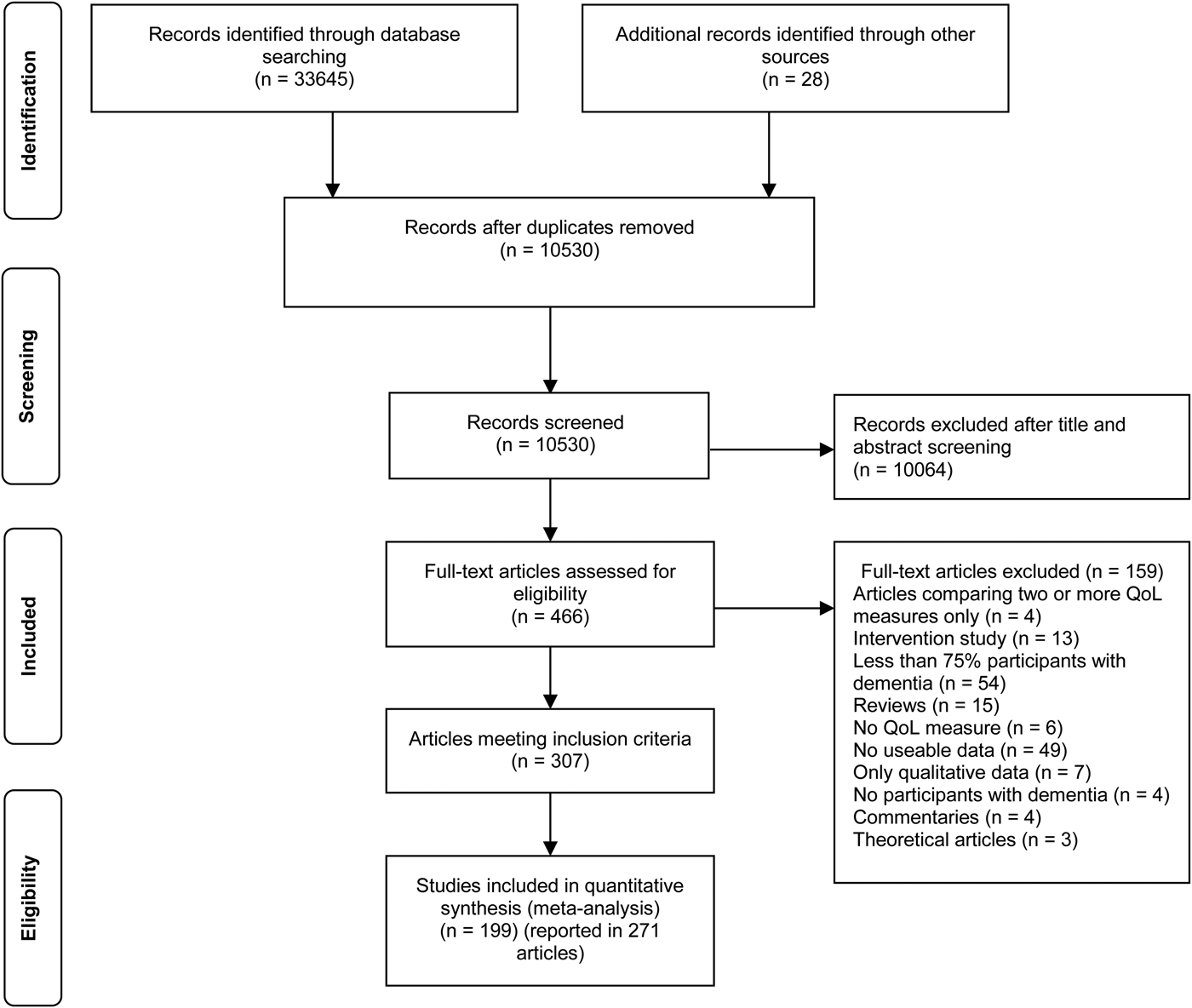
Fig. 1. Flow diagram showing process of study selection.
A description of each outcome measure used in the included studies was taken from either the relevant measure development article or, if this was unavailable, from the most comprehensive description of the measure provided in an included article. Based on this description, each measure was classified as assessing either ‘general QoL’, ‘general HRQoL’, ‘dementia-specific QoL/HRQoL’, ‘life satisfaction’ or ‘subjective well-being’. This process was conducted by three authors working independently, with any disagreements being referred to a fourth individual for resolution.
Where multiple articles reported data from the same study, details for each factor were taken from the article reporting the largest sample size. If an article included data from two sources [e.g. two different datasets were analysed separately (e.g. Trigg et al. Reference Trigg, Skevington and Jones2007; Li et al. Reference Li, Suishu, Hattori, Liang, Gao and Feng2013) or data were analysed separately for mild and moderate dementia (e.g. Novelli & Caramelli, Reference Novelli and Caramelli2010)], these were classed as separate studies for the purposes of the analysis. We extracted correlational data provided in study reports or regression data, standardised β (Peterson & Brown, Reference Peterson and Brown2005), odds ratios, p values, or t or F statistics converted to correlations, adopting a standardised correlation direction to facilitate comparison (Borenstein et al. Reference Borenstein, Hedges, Higgins and Rothstein2009).
Statistical analysis
Meta-analysis for each factor was undertaken where comparable data were available from at least five studies. All computations were based on Fisher's z transformations and were conducted using Comprehensive Meta-Analysis 2 (Borenstein et al. Reference Borenstein, Hedges, Higgins and Rothstein2005) software which calculated average z scores and p values, weighted effect r values and 95% confidence intervals. A two-sided 5% significance level was applied. Multiple within-study correlations were averaged to correct for violations of independence, so that all available data could be included in the analysis. Between-study heterogeneity for each measure was assessed using the I 2 index (Higgins et al. Reference Higgins, Thompson, Deeks and Altman2003). Estimated effect sizes (subsequently ‘effect sizes’) ⩽0.09 were considered negligible, 0.10–0.29 small, 0.30–0.49 moderate and ⩾0.50 large (Cohen, Reference Cohen1988). A random-effects model (DerSimonian & Laird, Reference DerSimonian and Laird1986) was employed in view of anticipated high heterogeneity across studies.
Where comparisons were described simply as non-significant, the correlation was set at zero. Where p values were given as a range, the value used to calculate the correlation was set at the upper limit of the range (e.g. for p < 0.05 the value was set at p = 0.049).
Separate analyses were conducted for each of the rating methods used in cross-sectional studies: self-rating by the person with dementia, informant rating by a family carer or health care professional (reflecting the informant's appraisal of the person with dementia), proxy rating by a family carer or health care professional (reflecting the appraisal that the proxy thinks the person with dementia would make), and differences between parallel self- and informant ratings. Longitudinal studies were examined separately to identify baseline predictors at follow-up.
Pre-specified sub-group analyses examined whether associations of relevant factors with living well outcomes differed according to the type of rating, type of dementia, living situation of the person with dementia, relationship with the carer or type of measure.
Random-effects meta-regression analyses were used to investigate pre-specified moderating effects of age and cognitive status, indicated by Mini-Mental State Examination (MMSE) scores (Folstein et al. Reference Folstein, Folstein and McHugh1975), on moderate associations where 10 (Borenstein et al. Reference Borenstein, Hedges, Higgins and Rothstein2009) or more studies were available.
Sensitivity analysis
To examine whether inserting a correlation of 0 for ‘non-significant’ p values influenced the findings, we removed these insertions and repeated the meta-analysis. We also investigated the impact of poor-quality articles on the results. Fourteen articles judged to be of poor quality (seven of which were conference abstracts) were removed from the analysis.
Results
We identified 307 articles for inclusion: 282 journal papers, 16 conference abstracts, three Health Technology Assessment reports, one book chapter and five PhD theses (Fig. 1). These reported findings from 213 individual studies from 36 countries mostly in Europe or North America; online Supplementary Tables S3–S9 provide details and quality ratings. Data on QoL were reported in 205 studies, well-being in five and life satisfaction in three.
Measures of QoL, well-being and life satisfaction
The studies utilised 45 measures of QoL, seven measures of well-being and four measures of life satisfaction (see online Supplementary Table S10). The QoL-AD (Logsdon et al. Reference Logsdon, Gibbons, McCurry, Teri, Albert and Logsdon2000) was the most frequently employed measure of QoL in studies where the focus was self-rated or informant-rated QoL or the difference between self-rated and informant-rated QoL, including longitudinal studies. DEMQOL-Proxy (Smith et al. Reference Smith, Lamping, Banerjee, Harwood, Foley and Smith2005) was the most frequently employed measure of proxy-rated QoL. Distributions of scores on QoL measures are shown in online Supplementary Tables S11–S12c. Self-ratings by people with dementia on the QoL-AD and EQ-5D were higher than the corresponding informant ratings. For all other measures, insufficient data were available to allow a comparison of different rating types.
Studies included in the meta-analysis
In studies of well-being and life satisfaction, there was no overlap in the factors for which associations were examined; each study examined associations with a different set of factors, with the exception of depression which was included in two separate studies of well-being. Consequently it was not possible to conduct meta-analyses of factors associated with well-being and life satisfaction, and meta-analyses were conducted only for studies examining QoL. Data from 37639 people with dementia in 198 studies reported in 272 articles were included (seven studies were excluded due to lack of any data that met criteria for inclusion in the meta-analysis). These studies examined associations of QoL with 159 individual factors relating to the person with dementia and 69 factors relating to the carer. Data on 33 factors relating to the person with dementia and 10 factors relating to the carer were included in the meta-analysis (online Supplementary Tables S13a–S17).
Self-rated QoL
Self-ratings of QoL by the person with dementia were examined in 130 studies reporting associations with 110 factors relating to the person with dementia and 34 factors relating to the carer, of which 28 and eight, respectively, were included in the meta-analysis, based on 129 studies (Fig. 2 and online Supplementary Tables S13a–b).
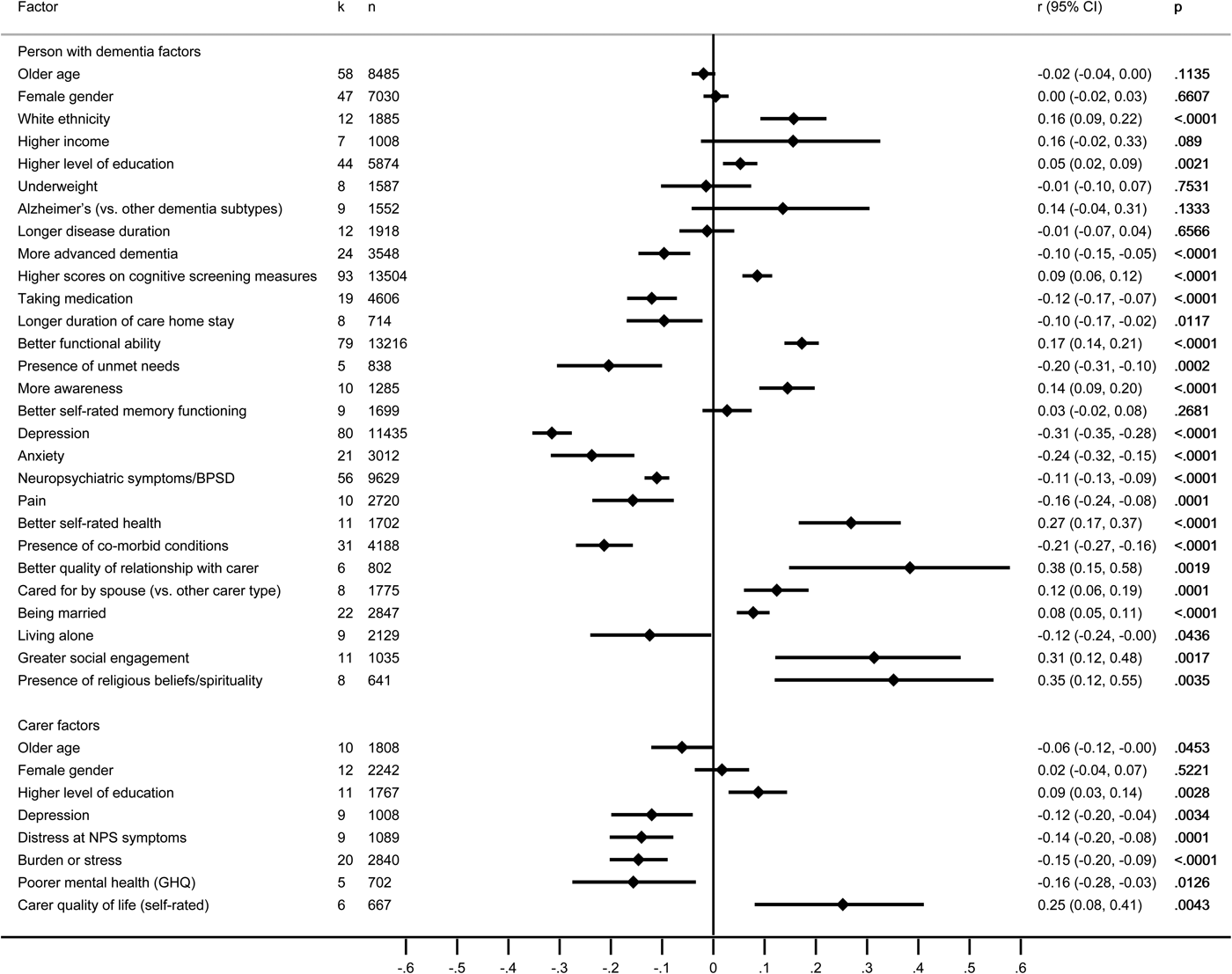
Fig. 2. Forest plot for factors associated with the quality of life self-ratings made by people with dementia. Note: negative scores indicate that factors were related to poorer QoL, and positive scores indicate that factors were related to better QoL.
Informant-rated QoL
Informant ratings made by the carer were examined in 135 studies reporting associations with 114 factors relating to the person with dementia and 56 factors relating to the carer, of which 26 and 10, respectively, were included in the meta-analysis, based on 130 studies (Fig. 3 and online Supplementary Tables S14a–b).
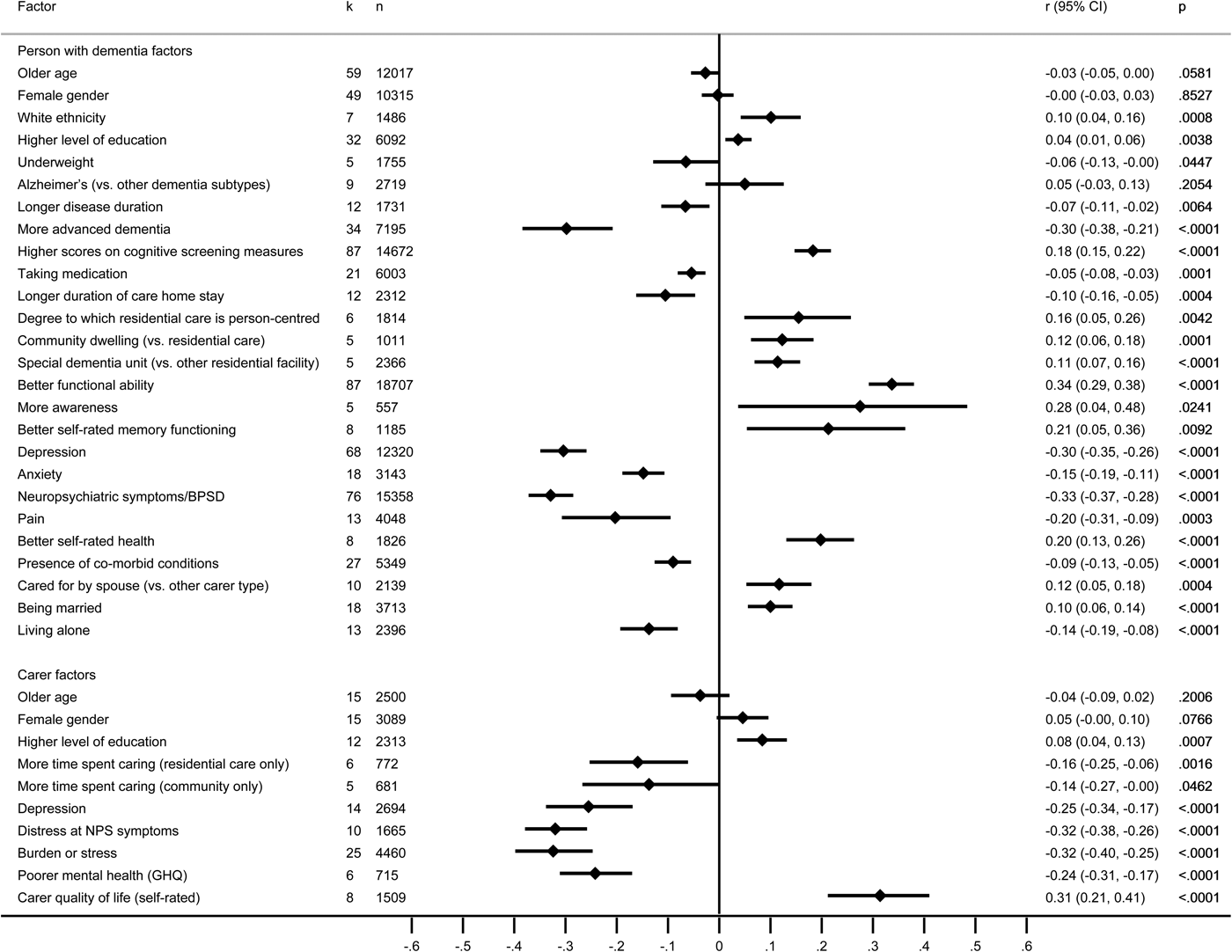
Fig. 3. Forest plot for factors associated with informant ratings of the quality of life of the person with dementia. Note: negative scores indicate that factors were related to poorer QoL, and positive scores indicate that factors were related to better QoL.
Proxy-rated QoL
Proxy ratings were examined in 27 studies reporting associations of 72 factors relating to the person with dementia and 23 factors relating to the carer, of which nine and one, respectively, were included in the meta-analysis, based on 22 studies (Fig. 4 and online Supplementary Tables S15a–b).
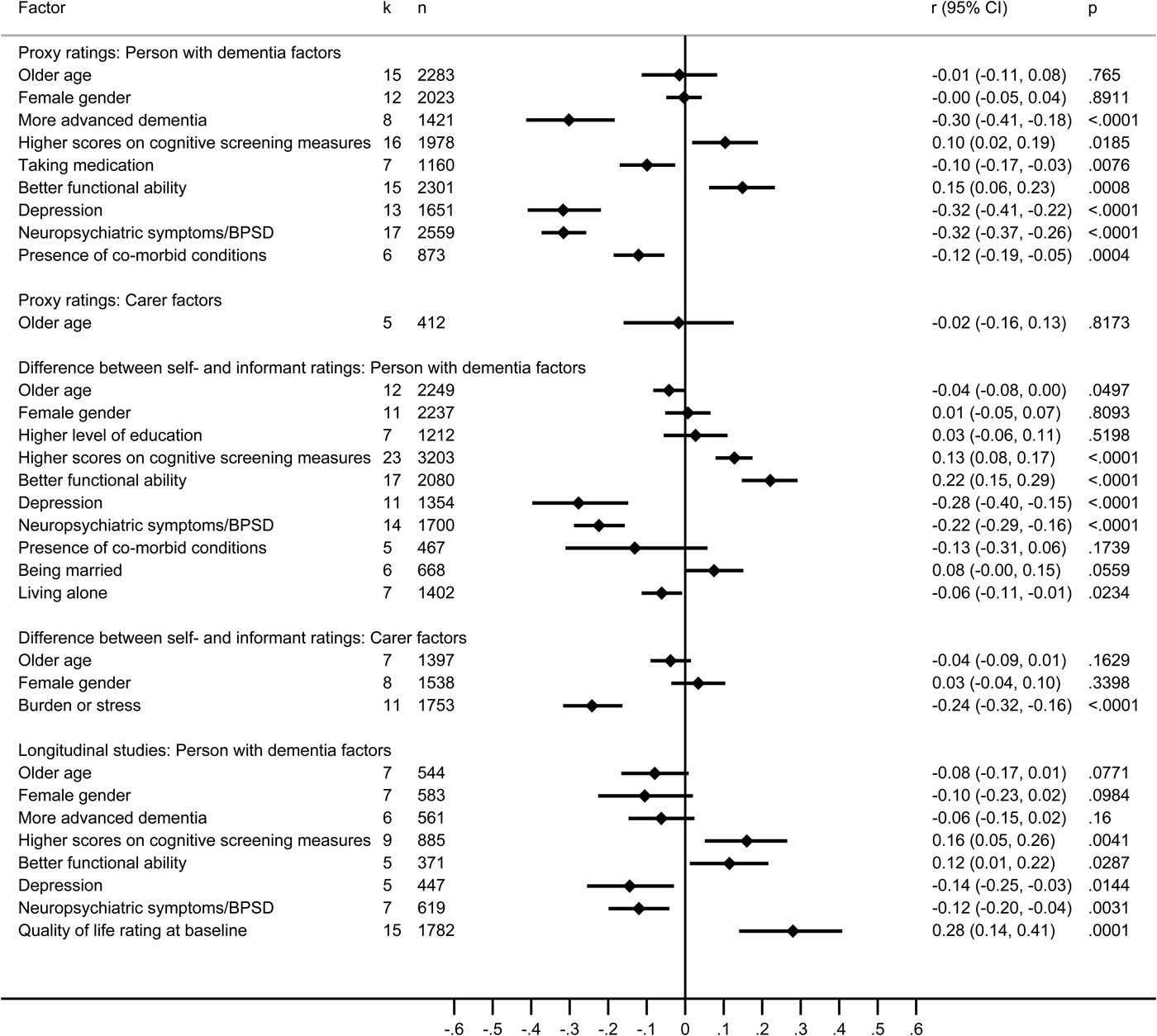
Fig. 4. Forest plots for factors associated with proxy ratings of quality of life, the difference between self- and informant ratings of quality of life, and baseline factors associated with longitudinal studies of quality of life. Note: negative scores indicate that factors were related to poorer QoL, and positive scores indicate that factors were related to better QoL. Longitudinal studies included both self- and informant ratings of QoL.
Difference between self- and informant-rated QoL
Scores reflecting differences between self-rated and informant-rated QoL were examined in 23 studies reporting associations with 30 factors relating to the person with dementia and 13 factors relating to the carer, of which 10 and three, respectively, were included in the meta-analysis, based on 22 studies (Fig. 4 and online Supplementary Tables S16a–b).
Comparing self-rated and informant-rated effect sizes
For variables that were assessed in relation to more than one of the different rating methods of self-, informant and proxy ratings or discrepancies between self- and informant ratings, the direction of association was consistent in all cases, although the strength of association varied. Online Supplementary Table S18a–b reports correlations between effect sizes where factors were examined in relation to both self-rated and informant-rated QoL.
Factors associated with better QoL
Greater social engagement, better quality of current relationship with the carer and religious beliefs/spirituality were moderately associated with better QoL across rating types. Better functional ability, self-rated health and awareness, white ethnicity, having a spouse carer and living in the community, and for people in residential settings, being cared for in a specialist dementia unit and receiving more person-centred care had small or mainly small associations. Small or negligible associations were found for cognitive test scores, self-rated memory functioning and being married. The only carer factor with small or moderate associations with better QoL in the person with dementia was the carer's self-rated QoL.
Factors associated with poorer QoL
Depression and neuropsychiatric symptoms showed mainly moderate associations with poorer QoL across rating types. Greater severity of dementia, anxiety, pain, presence of unmet needs, presence of co-morbid conditions and living alone showed mainly small associations. Use of medication, more advanced dementia and longer duration of stay in residential care showed small or negligible associations. Carer burden/stress and distress at symptoms had moderate or small associations with poorer QoL, while small associations were found for carer depression, mental health and time spent caring.
Factors not significantly associated with QoL
Non-significant or negligible associations with QoL were found across rating types for age, gender or educational level of the person with dementia and carer, income of the person with dementia, type of dementia and disease duration.
Factors predicting longitudinal trajectories of QoL
Baseline predictors of QoL outcomes were examined in 20 longitudinal studies reporting associations with 25 factors relating to the person with dementia and two factors relating to the carer, of which only eight factors pertaining to the person with dementia were included in the meta-analysis, based on 19 studies (Fig. 4 and online Supplementary Table S17). Follow-up ranged from 2 months to 5 years (mean 18.2 months). Non-significant or negligible associations were found for gender, age and more advanced dementia. Depression and neuropsychiatric symptoms in the person with dementia had small associations with worse QoL outcomes, while better baseline QoL ratings, better functional ability and higher scores on cognitive screening tests had small associations with better QoL outcomes. Where comparisons could be made the direction of associations was consistent with that found in cross-sectional studies.
Heterogeneity and sub-group analyses
There was a moderate-to-large degree of between-study heterogeneity for the majority of factors. Sub-group analyses investigated the effects of (a) dementia sub-type by separately analysing studies that focused specifically on people diagnosed with Alzheimer's disease and studies that did not focus specifically on people diagnosed with Alzheimer's disease; (b) living situation of the person with dementia (community and residential care settings); (c) type of carer completing the ratings of QoL (family member and health care professionals); and (d) type of QoL measure used (dementia-specific, health-related and generic). Distinguishing sub-groups in this way had little impact on overall effect sizes or degree of heterogeneity (online Supplementary Tables S19a–S23j).
Moderator variables
Meta-regressions for moderate effect sizes found no moderation effects of age or MMSE score (online Supplementary Tables S24a–S25d). Sensitivity analyses suggested that inserting 0 for non-significant p values and removing articles rated poor in quality had limited impact on the results (online Supplementary Tables S26a–S34).
Discussion
To our knowledge, this is the first comprehensive systematic review exploring factors associated with QoL, well-being and life satisfaction in people with dementia, and the first meta-analysis of factors associated with QoL. We found few studies that have investigated well-being or life satisfaction, possibly because the policy focus on these constructs is relatively recent; therefore, meta-analyses could only be conducted for studies assessing QoL. The majority of studies were cross-sectional, with considerable between-study heterogeneity. Although most factors examined were significantly related to QoL, with the direction of association consistent across rating types, no factors were strongly associated, and most associations were small or negligible. Where moderate associations were found, confidence intervals were typically wide. Demographic and disease characteristics were largely unrelated to QoL, while social, health- and care-related factors were more likely to show associations. Factors reflecting relationships, social engagement and functional ability were associated with better QoL, along with living in the community and having a spouse carer, or receiving specialised and person-centred care in residential settings. Factors indicative of poorer physical and mental health, such as co-morbid conditions, pain, depression, anxiety, neuropsychiatric symptoms, more advanced dementia and for people in residential settings a longer duration of stay, were associated with poorer QoL, as were carer burden and distress. The moderate associations of carer burden and distress with informant-rated QoL suggests that subjective burden could be contributing to lower scores reported in carer-rated as opposed to self-rated QoL. Due to the small number of factors and heterogeneous follow-up periods, longitudinal evidence about predictors of QoL was limited, with self-rated QoL at baseline the strongest predictor of later QoL outcomes.
Our observation that demographic factors are largely unrelated to QoL is consistent with a previous narrative review of HRQoL (Banerjee et al. Reference Banerjee, Samsi, Petrie, Alvir, Treglia and Schwam2009). Aside from this, the overall picture is one of a large number of factors showing occasionally moderate but mainly small or negligible, though statistically significant, associations with QoL. A recent narrative review also reported a complex picture of associations across domains (Jing et al. Reference Jing, Willis and Feng2016). Thus, QoL appears subject to many influences, some inter-related, which may co-vary in differing combinations to influence individual ratings.
In line with the current meta-analysis, the importance of positive relationships and health for QoL has been emphasised in a recent meta-synthesis of qualitative studies (O'Rourke et al. Reference O'Rourke, Duggleby, Fraser and Jerke2015a). However, that review also highlighted the domains of agency and place (feeling settled or unsettled), as key elements of the experience of people with dementia associated with QoL; these factors have not been reflected in quantitative evaluations to date. This serves as a reminder that potential associations examined in quantitative studies may not necessarily reflect all those aspects of life that people with dementia find most relevant. The finding here of poorer physical and mental health being related to QoL is consistent with the emphasis on depression, neuropsychiatric symptoms and functional ability in Lawton's dementia-specific model of QoL (Lawton, Reference Lawton1994), and with evidence from qualitative research showing that people with dementia describe mood and functional ability as important for QoL (O'Rourke et al. Reference O'Rourke, Duggleby, Fraser and Jerke2015a). However, the relevance of other factors included in Lawton's model, such as cognitive competencies, is less well supported by our findings. Our findings further indicate that, beyond the dimensions identified by Lawton, many other factors are associated with QoL, though to a lesser degree.
Our review found that numerous measures were employed to assess QoL, although the QoL-AD (Logsdon et al. Reference Logsdon, Gibbons, McCurry, Teri, Albert and Logsdon2000) predominated. Several previous reviews have focused on the measurement of QoL (Ready & Ott, Reference Ready and Ott2003; Ettema et al. Reference Ettema, Dröes, de Lange, Mellenbergh and Ribbe2005; Bowling et al. Reference Bowling, Rowe, Adams, Sands, Samsi and Crane2015; Algar et al. Reference Algar, Woods and Windle2016; Missotten et al. Reference Missotten, Dupuis and Adam2016). The absence of a clear shared definition and theoretical model of QoL is reflected in the wide variation in focus and content among available measures. Analysis of domains covered in commonly used measures of dementia-specific QoL (Missotten et al. Reference Missotten, Dupuis and Adam2016), however, indicates that items explore some of the same factors for which associations with QoL are typically examined, particularly affect, but also social interaction, functional ability, neuropsychiatric symptoms, cognition and physical health. This raises concerns about circularity and overlap in measurement. Furthermore, it is unclear whether the quantitative assessment of QoL adequately reflects the perspective of people with dementia (O'Rourke et al. Reference O'Rourke, Fraser and Duggleby2015b). While items in some measures are based on data from qualitative interviews with people with dementia, many measures have been developed with little or no direct input from people with dementia (Bowling et al. Reference Bowling, Rowe, Adams, Sands, Samsi and Crane2015) and few large-scale studies have been conducted that enable people with dementia to say what is important for their QoL (Clare et al. Reference Clare, Nelis, Quinn, Martyr, Henderson and Hindle2014a).
Because of the breadth of included studies, we decided to statistically analyse only factors for which there was data available from five or more studies. Focusing the review in this way allowed us to summarise current understanding and reduce reporting of associations for which there was limited evidence. However, this precluded quantitative analyses for life satisfaction and well-being, and limited the number of factors in the analysis of longitudinal studies. We necessarily examined factors separately, and could not account for likely inter-correlations. A number of studies did not provide exact data for non-significant findings, so a potential limitation resulted from inserting zero correlations from those studies; however, this had a minimal effect on associations or heterogeneity. Most of the studies that did not focus exclusively on those diagnosed with Alzheimer's disease analysed responses from people with different dementia diagnoses as one group, which precluded separate analysis for different dementia sub-types. Methodologically, the widespread misuse of the term ‘proxy’ when researchers mean ‘informant’ created challenges for comparing findings between studies. For example, in DEMQOL-Proxy (Smith et al. Reference Smith, Lamping, Banerjee, Harwood, Foley and Smith2005), instructions allow informants to make either proxy or informant ratings, so ratings from this instrument were difficult to classify correctly. Our review highlights the need for application of clear and consistent terminology and the standardisation of methods (e.g. standard follow-up periods and sample size calculations), and a robust conceptual and methodological framework to guide work in this area.
The findings point to some limitations in how we understand and evaluate living well with dementia. Available measures may not capture what is most meaningful to each individual (Bowling et al. Reference Bowling, Rowe, Adams, Sands, Samsi and Crane2015) or indeed everything that is relevant to living well. In this respect, broadening the perspective to include subjective well-being and satisfaction with life may be informative; these constructs have rarely been assessed, and as yet evidence is limited (Clare et al. Reference Clare, Nelis, Quinn, Martyr, Henderson and Hindle2014a). The pattern we observed of numerous small associations generally supports the idea that QoL is multifaceted, as suggested by Lawton's framework (Reference Lawton1994). It might also reflect the operation of individual differences in what is deemed most important for QoL, so there may be value in considering more personalised approaches to assessment, particularly as no QoL measure includes all of the factors that our review suggests may be important to the QoL of people with dementia. However, developing and implementing personalised approaches would present significant challenges.
The findings raise questions about the use of QoL as an outcome measure in psychosocial intervention trials. First, if QoL is subject to many influences, changes in one area of life may have little effect on the overall picture, so it may not be reasonable to expect interventions that are effective in improving some primary outcomes to influence scores on QoL measures. Second, as noted above, potential circularity in measurement may create a distorted picture. Therefore, the relevance of QoL measures for assessing effectiveness of interventions may need re-evaluation.
Conclusion
Our findings suggest that efforts to improve QoL might focus on supporting relationships, social engagement and everyday functioning, addressing poor physical and mental health, and ensuring high-quality care. However, there is a need for longitudinal evidence that can point to ways of maintaining or improving QoL over time and enable identification of people at risk of declining QoL, so that preventive interventions can be targeted to this group.
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1017/S0033291718000405
Acknowledgements
The authors wish to thank Dr Gill Toms for help with article screening and measure classification; Dr Alexandra Hillman, Isobel Evans and Hannah Scott for help with data quality evaluation; Dr Edward Miller, Dr Daniel Murman and Professor Jean-Luc Novella for providing data for the review; and members of the IDEAL project advisory group and ALWAYs involvement group for helpful comments on previous drafts. The support of the ESRC and NIHR is gratefully acknowledged. The IDEAL study is funded by the Economic and Social Research Council (UK) and the National Institute for Health Research (UK) through grant ES/L001853/2 ‘Improving the experience of dementia and enhancing active life: living well with dementia’ (Investigators: L. Clare, I.R. Jones, C.R. Victor, J.V. Hindle, R.W. Jones, M. Knapp, M. Kopelman, R. Litherland, A. Martyr, F.E. Matthews, R.G. Morris, S.M. Nelis, J.A. Pickett, C. Quinn, J.M. Rusted, J.M. Thom). The funders had no role in the study design, data collection, data analysis and interpretation, writing of the report, or the decision to submit the article for publication.
Author contributions
AM: Developing and piloting search strategy, conducting searches, title screening, abstract screening, full-text screening, data extraction, assessment of study quality, planning and conducting analyses, interpretation of results, drafting review text. LC: Developing the concept for review, drafting protocol, title screening, abstract screening, full-text screening, assessment of study quality, planning analyses, interpretation of results, drafting review text. RC and RL: Title screening, abstract screening, full-text screening, data extraction, assessment of study quality, interpretation of results, commenting on and approving text of review. CH, JH, IRJ, RM, SMN, CQ, JR, JT, CRV, YTW: Abstract screening, interpretation of results, commenting on and approving text of review.
Declaration of Interest
None.






