Intimate partner violence (IPV), seen as a violation of human rights, remains a prevalent global public health issue affecting as many as one in three women.Reference Garcia-Moreno, Jansen, Ellsberg, Heise and Watts1,2 Being a survivor of IPV is associated with a wide range of poor health outcomes.Reference Black, Basile, Breidling, Smith, Walters and Merrick3,Reference Bacchus, Ranganathan, Watts and Devries4 For example, survivors of IPV undertake more harmful lifestyle choices such as smokingReference Crane, Hawes and Weinberger5 and excessive alcohol use.Reference La Flair, Bradshaw, Storr, Green, Alvanzo and Crum6 There is a strong relationship between being a survivor of IPV and poor mental health outcomes. The pathway that leads to this association is complex, with there likely being biochemical, psychological and environmental risk factors that predispose survivors of abuse to go on to develop poor mental health outcomes.Reference Delara7 Previous systematic reviews of observational studies have identified associations between being a survivor of IPV with depression, suicide, post-traumatic stress disorder and prenatal depressive symptoms.Reference Reingle Gonzalez, Jetelina, Olague and Wondrack8–Reference Trevillion, Oram, Feder and Howard12 In a recent review of all cohorts exploring the impact of IPV on female survivors' physical and mental health,Reference Bacchus, Ranganathan, Watts and Devries4 13 of these studies explored the relationship between IPV and depression. None of these 13 studies were set in the UK. Although there are challenges in discerning the temporality of this relationship, this association appeared to be bidirectional. When depression was explored as both a dependent and independent variable, a positive correlation remained. Only one cohort study set in the USA ascertained a positive relationship between IPV and subsequent diagnosis of generalised anxiety disorder.Reference Suglia, Duarte and Sandel13 We identified no cohort studies that investigated the relationship between IPV and subsequent diagnosis of serious mental illness (SMI), including schizophrenia, bipolar disorder, mania and other forms of non-affective psychoses. An alternative reviewReference Trevillion, Oram, Feder and Howard12 compiling case–control and cross-sectional data highlighted just one study investigating the relationship between IPV and bipolar disorder, which appear to be linked.Reference Pietrzak, Goldstein, Southwick and Grant14 A separate more recent reviewReference Khalifeh, Oram, Osborn, Howard and Johnson15 exploring the prevalence of experiencing domestic violence within the past year in groups of patients with severe mental illness, reported a prevalence of domestic violence exposure between 15 and 22%.
Because of the prevalence of IPV, there appears to be an association between mental health burden and exposure to IPV. So far there has not been a UK-based cohort study assessing the relationship between IPV and anxiety, depression and SMI. As well as shedding new light on the relationship between IPV and anxiety/SMI, it is also important to quantify the extent of this burden to enable planning of targeted mental health services in the UK for this group at risk. Therefore, we aimed to explore this association by using primary care records derived from The Health Improvement Network (THIN) database.
Method
Study design and data source
This study was a population-based, retrospective, open cohort study using the THIN database, comparing female patients coded with previous exposure of IPV with female patients not coded to have experienced IPV. The THIN database consists of UK electronic medical records derived from over 750 general practices (family practices), comprising approximately 3.6 million patients at the time of this study. THIN is deemed demographically representative of the UK population.Reference Blak, Thompson, Dattani and Bourke16 Information regarding patients' symptoms and diagnoses are recorded using the Read code hierarchy system.17,Reference Booth18 To reduce under-recording of events, general practices were included 12 months following their instalment of electronic practice records or from the practice's acceptable mortality recording date.
Study population
The study period was set between 1 January 1995 and 1 December 2017. During this period, women over the age of 18 years who had documented exposure to IPV, noted through Read codes by their General Practitioner (GP), were deemed to be our exposed group. The index date for individuals in the exposed group was taken to be the first inserted Read code relating to IPV exposure once a patient was eligible to take part in the study or, alternatively, the study start date for patients with a previous record of IPV (prevalent cases). To mitigate immortality time bias,Reference Lévesque, Hanley, Kezouh and Suissa19 the same index date was assigned to the corresponding unexposed patient. Each exposed survivor of IPV was matched with up to four control patients, who had not been documented to have a Read code relating to IPV exposure, from general practices within the database forming the unexposed group. Controls from the unexposed group were matched individually to cases based on age at index date (±1 year), and gender.
The primary outcome explored in this study was the development of mental illness, which comprised depression, anxiety, SMI and a combination of these three. If individuals in either group had a diagnosis of one of the primary outcome diagnoses before the study start date, they were excluded. However, as much of the exposed population experienced mental illness at baseline, we have also described the risk of mental illness at entry to the study.
Covariates that affect the development of mental illness were included in the baseline data for this population. These included body mass index, deprivation assessed by the Townsend Deprivation Index score,Reference Townsend, Phillimore and Beattie20 smoking status and alcohol use.
Read code lists for the exposure and outcomes are provided in the supplementary material (Supplementary File 1 available at https://doi.org/10.1192/bjp.2019.124).
Statistical analysis
Categorical baseline data were described by proportions. Continuous data were described by means and s.d. Missing data is highlighted in Table 1. As much of the exposed population had mental illness at baseline, we have also described the odds of having mental illness at baseline between the exposed and unexposed groups. This has been described by a logistic regression, which provides an unadjusted odds ratio and adjusted odds ratio (aOR), factoring in the covariates of interest above. Odds ratios were calculated with 95% CIs and statistical significance was set at P < 0.05. Where there was missing data in our covariates, they were treated as a separate missing category and included in the regression analysis.
Table 1 Baseline characteristics
After patients with the mental illness of interest at baseline were excluded, Poisson regression was used to calculate an incidence rate ratio (IRR) for each outcome of interest during the study period. Following adjustment for important documented covariates that may independently affect the outcome of interest, we have calculated and presented an adjusted incidence rate ratio (aIRR). IRRs were calculated with 95% CIs and statistical significance was set at P < 0.05. These are presented for each of the outcomes of interest: depression, anxiety, SMI and a composite of the three.
To account for survival bias, a sensitivity analysis was conducted, excluding prevalent cases of IPV and thereby including only incident cases of IPV with their respective controls. To account for possible misclassification of mental illness coding, we have conducted a second sensitivity analysis whereby we have explored the incidence of a new starting prescription of anxiolytic, antidepressant and antipsychotic medications (drug codes taken from relevant British National Formulary (https://bnf.nice.org.uk/) chapters are presented in Supplementary File 1), for the main results. Stata version 14.2 for MacOS software was used to conduct all analysis throughout the study.
Ethical approval and data accessibility
Anonymised data was used throughout the study, provided by the data provider IQVIA to University of Birmingham. Studies using the THIN database have had initial ethical approval from the National Health Service South-East Multicentre Research Ethics Committee, subject to prior independent scientific review. The Scientific Review Committee (IMS Health) approved the study protocol (reference number SRC18THIN034) before its undertaking. The full data-set and statistical analysis code are available from author K.N. (k.nirantharan@bham.ac.uk).
Results
Baseline characteristics
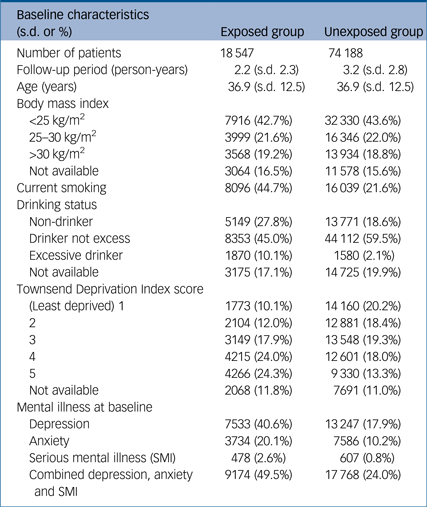
A total of 18 547 women who had been documented to have experienced IPV were matched to 74 188 controls by age and gender. The mean length of follow-up in the exposed group was shorter compared with the unexposed group. Mean age in both groups was similar. Obesity, prevalence of smoking and number of women who were excessively drinking at baseline were significantly higher in the exposed group compared with the unexposed. The exposed group were also more socioeconomically deprived at baseline. At baseline, a high proportion of individuals who were exposed to IPV compared with the unexposed group had experienced mental illness in the form of depression (40.6%), anxiety (20.1%) or SMI (2.6%) compared with the unexposed group at 17.9%, 10.2% and 0.8%, respectively. Characteristics of both populations are described in detail in Table 1.
The odds of having depression, anxiety and SMI at baseline
This odds of having mental illness in the population being studied is described in Table 2 and demonstrated in Fig. 1. As described above, at baseline there was a significantly higher burden of mental illness. At study start date, 49.5% (n = 9174/18 457) of the exposed group had experienced any type of mental health outcome compared with 24.0% (n = 17 768/74 188) of the unexposed group. This translated to an increased odds ratio of 3.11 (95% CI 3.01–3.21). Following adjustment for covariates, this translated to an aOR of 2.62 (95% CI 2.52–2.72). When subcategorised by outcomes, the exposed group experienced a higher risk of having depression, anxiety and SMI following adjustment, at the study start date compared with the unexposed group. The aORs were 2.61 (95% CI 2.51–2.71), 1.91 (95% CI 1.82–2.01) and 2.13 (95% CI 1.86–2.43), respectively.
Table 2 Mental illness at baseline

a. Unadjusted odds ratio.
b. Adjusted odds ratio: adjusted for body mass index, age, smoking status, drinking status and Townsend Deprivation Index score at baseline.
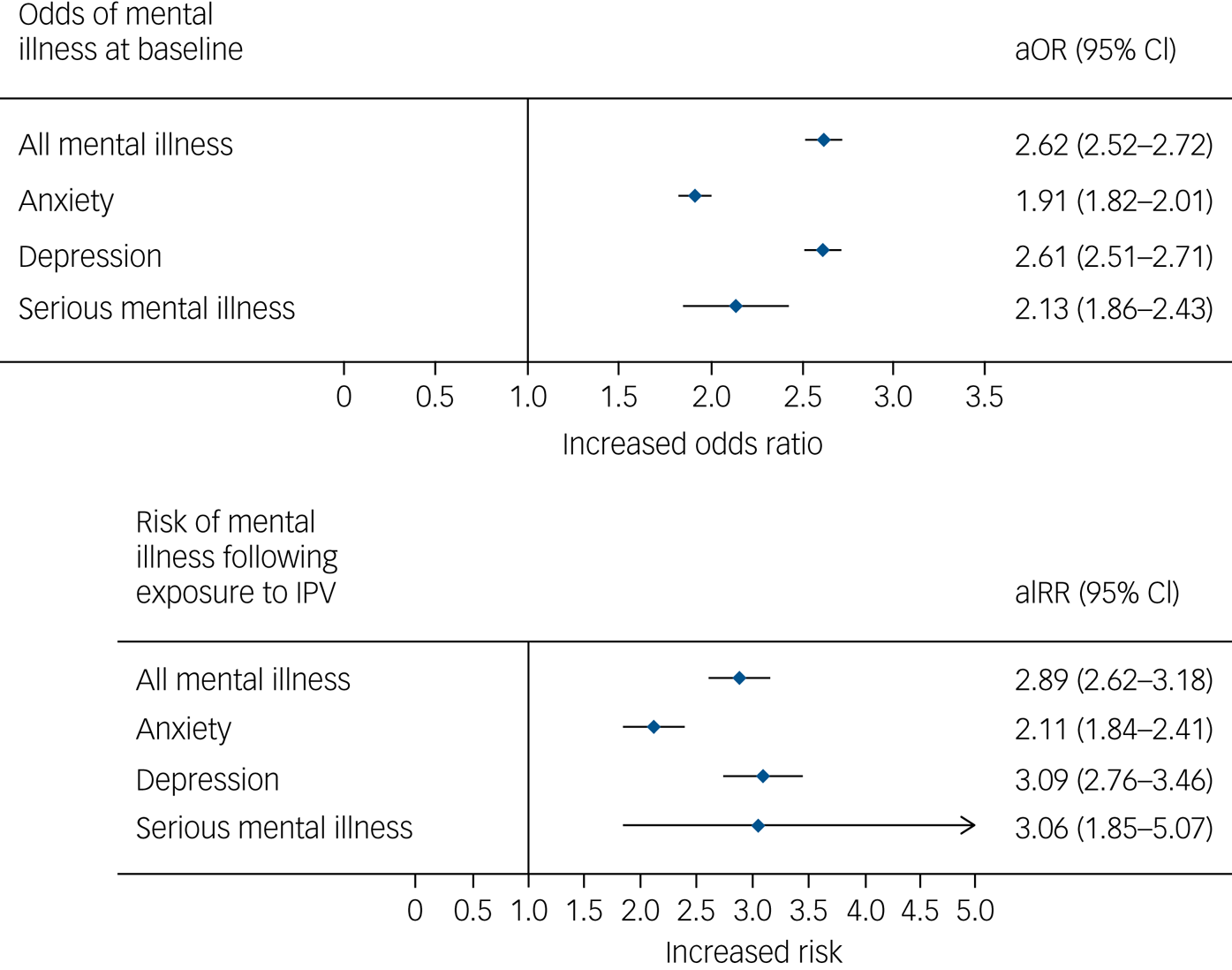
Fig. 1 Risk of mental illness at baseline and following exposure.
Association between IPV and depression, anxiety and SMI
The main results are presented in Table 3 and Fig. 1. During our study period 1254 patients (incidence rate 46.62 per 1000 person-years) presented with any type of mental illness in the exposed group compared with 3119 in the unexposed group (incidence rate 14.93 per 1000 person-years). This translated to a significant unadjusted increased IRR of 3.12 (95% CI 2.92–3.33). Following adjustment, this remained significant (aIRR 2.77, 95% CI 2.58–2.97). Anxiety (aIRR 1.99, 95% CI 1.80–2.20), depression (aIRR 3.05, 95% CI 2.81–3.31) and SMI (aIRR 3.08, 95% CI 2.19–4.32) were all positively associated following exposure to IPV.
Table 3 Risk of mental illness development between the exposed and unexposed groups

a. Unadjusted incidence rate ratio.
b. Adjusted incidence rate ratio: adjusted for body mass index, age, smoking status, drinking status and Townsend Deprivation Index score at baseline.
Sensitivity analysis
Our sensitivity analysis, consisting of incident-only cases during the study period (Table 4), also remained congruent with the main results. The baseline characteristics of this group (Supplementary File 2) were similar in nature to the main analysis, with similar differences presenting in average follow-up time, body mass index, smoking, deprivation and alcohol use at baseline. All mental illness in the sensitivity analysis remained strongly associated, with an incidence ratio of 47.29 per 1000 person-years in the exposed group compared with 14.57 per 1000 person-years in the unexposed group, resulting in an aIRR of 2.89 (95% CI 2.62–3.18). Similarly, anxiety (aIRR 2.11, 95% CI 1.84–2.41), depression (aIRR 3.09, 95% CI 2.76–3.46) and SMI (aIRR 3.06 95% CI 1.85–5.07) remained strongly associated with exposure to IPV. In addition, the odds of mental illness at baseline was significantly higher in the exposed group in this sensitivity analysis.
Table 4 Incident-only cases risk of mental illness development between the exposed and unexposed groups

a. Unadjusted incidence rate ratio.
b. Adjusted incidence rate ratio: adjusted for body mass index, age, smoking status, drinking status and Townsend Deprivation Index score at baseline.
The results of our second sensitivity analysis (Supplementary File 3) also support the main findings. At baseline we noted similarly increased odds of having a prescription indicative of mental illness (aOR 3.20, 95% CI 3.08–3.32). When subcategorised by prescription types the aOR for anxiolytics, antidepressants and antipsychotic medication were 2.52 (95% CI 2.36–2.70), 3.25 (95% CI 3.13–3.38) and 1.95 (95% CI 1.82–2.10), respectively. When exploring the association between a new prescription of an agent used for treating mental illness, we also noted a similarly positive effect size (aIRR 2.37, 95% CI 2.24–2.50). This remained positive for each subtype of prescription: anxiolytics (aIRR 1.67, 95% CI 1.55–1.80), antidepressants (aIRR 2.58, 95% CI 2.44–2.73) and antipsychotics (aIRR 1.64, 95% CI 1.52–1.77).
Discussion
Summary of key results
In summary, the results suggest a strong association between exposure to IPV and incident mental illness (aIRR 2.77, 95% CI 2.58–2.97) in this UK primary care data-set. This relationship was significant when assessing the incidence of anxiety, depression and SMI. These relationships remained positive following sensitivity analysis considering only incident cases as well as prescriptions for treatment of mental illness. Another key finding was that the odds of having mental illness at baseline in the IPV group was significantly higher than the unexposed group (aOR 2.62, 95% CI 2.52–2.72). This suggests that there is a higher likelihood of having mental illness before recorded exposure of IPV, but also for those who become exposed, their risk of mental illness continues to increase.
Relationship to current literature
To our knowledge, this is the first cohort study assessing recorded incident depression, anxiety and SMI following exposure to recorded IPV within the UK, using primary care records. Therefore, it is difficult to make comparisons relating to the expected incidence of these outcomes in a UK population. However, this study is consistent with previous work undertaken globally, which suggests a relationship between exposure to IPV and subsequent mental illness. It was noted in a recent meta-analysis,Reference Bacchus, Ranganathan, Watts and Devries4 when depression is considered as a dependent variable as in this case, the pooled odds ratio from previous cohort studies was 1.76 (95% CI 1.26–2.44). Our result is similarly positively associated (aIRR 3.05, 95% CI 2.81–3.31).
Comparatively, our study (aIRR 1.99, 95% CI 1.80–2.20) also supports a link associating anxiety and IPV exposure identified in another cohort study.Reference Suglia, Duarte and Sandel13 However, the main aim of that cohort study was not to ascertain the relationship specifically of IPV exposure to anxiety, but to assess the relationship of multiple factors relating to housing conditions and the development of mental illness, whereas in our study, the development of anxiety following IPV exposure was a primary outcome measure. Aside from cohort studies, there have been several other observational studies (case–control and cohort)Reference Trevillion, Oram, Feder and Howard12 that have identified a positive association between IPV and anxiety (pooled odds ratio, 4.06; 95% CI 2.39–6.97). Our study supports this association in a UK population.
Previously, there has been limited research exploring the relationship between SMI and IPV; however, of the work that has been done,Reference Trevillion, Oram, Feder and Howard12,Reference Pietrzak, Goldstein, Southwick and Grant14,Reference Khalifeh, Oram, Osborn, Howard and Johnson15 there has been a strong positive association, which our study affirms. The results of our study clearly show a strong association between the development of poor mental health outcomes following IPV in a UK setting, which are of importance in psychiatric and primary care settings. It has been shown in a variety of studies that survivors of IPV experience significant barriersReference Hegarty21–Reference Usta and Taleb24 in receiving the necessary healthcare support they often require. One of the key barriers relates to the identification of IPV exposure in women who present to healthcare services. It is clear from this study that there is still significant under-recording of IPV in this database, considering estimates of the prevalence of IPV could be as high as one in three women.2 However, there still remains a strong association with poor mental health outcomes, which does suggest that when women present with depression, anxiety and SMI, a past history of IPV should be explored to aid in management plans. Our study has also shown the increased prevalence of mental illness at baseline in the IPV cohort, suggesting that protective mental health interventions should be introduced early in their treatment plan.
The findings of this study are timely in relation to changes over the past 5 years in current practice within the UK and globally, relevant to both the enquiry of IPV and referral to supportive interventions. Previous literature had highlighted failures of UK mental health services in the identification of exposure to IPV in patients utilising their services, and in addition to poor integration of these services with appropriate referral pathways for these survivors.Reference Trevillion, Hughes, Feder, Borschmann, Oram and Howard25 In response to evidence suggesting the negative effects of such a model, the National Institute for Health and Care Excellence introduced the PH50 guidance in 2014, which highlighted the importance for multi-agency staff to enquire routinely about domestic violence and abuse and provide supportive options for referral.26 Within the same year, the World Psychiatric Association (WPA) isolated the importance of identifying gender-based domestic violence in psychiatric consultations, and this has been highlighted within the WPA curriculum for trained mental health professionals.Reference Stewart and Chandra27,28 The result of these changes is hopefully leading to a UK clinical environment that identifies the needs of possible survivors of IPV who may have otherwise been missed. We have since seen the introduction of the Identification and Referral to Improve Safety project) in several sites across the UK,Reference Feder, Davies, Baird, Dunne, Eldridge and Griffiths23 which aims to train and educate GPs in the enquiry of abuse and care pathways of survivors. In addition, another recent project funded by the UK Government is Pathfinder, a 3-year project started in 2017 aiming to establish comprehensive health practice in relation to domestic abuse, to also bridge gaps in provision for the cohort of survivors who may otherwise slip through the net because of lack of identification in clinical settings.29 Although our study does identify that there is perhaps a significant burden of unmet need in a subgroup of women who have experienced IPV, hopefully within the UK we are beginning to see changes within current practice. However, there is still a need to ensure that we are not missing potential opportunities to aid survivors in the disclosure of IPV, and referral to supportive services.
Study limitations
The use of this data-set relies upon the accuracy of imputation of Read codes by GPs. In this study, we were unable to validate the Read codes of IPV and mental illness with participating practices. This is an important future area of research that will help improve the validity of results in future work. However, we were able to conduct a sensitivity analysis using prescriptions relating to mental illness and this show congruent results. An important limitation in this study, is the number of women identified as exposed to IPV appears extremely low compared with previous UK and global estimates of IPV.2,30 Using data derived from the total population during the final year of the study period, we have identified the point prevalence of exposure to IPV in women to be 0.5%, which is low. This highlights another important message of this study, which is to bring to light the need for improved recording of IPV in primary care. Thus, it is possible that members of the unexposed group may actually have experienced IPV but were misclassified, possibly underestimating our effect size. Alternatively, we may have only identified women with severe IPV who chose to present to their GP, overestimating our effect size. In relation to this, because of very low recorded numbers, we were unable to conduct a subgroup analysis of physical, sexual or emotional abuse in the IPV cohort. Therefore, in future work, if coding is improved it will be important to tease out this relationship further. An interesting point of note is that following age and gender matching, there is a shorter follow-up period in the exposed group, which may be representative of the extent of geographical moves women who experience IPV may be making following disclosure of abuse. One of the challenges in this study design is accounting for reverse causality. As discussed in the literature,Reference Bacchus, Ranganathan, Watts and Devries4 there also appears to be a reverse relationship where individuals with mental illness appear to be more likely to become victims of IPV. We attempted to account for this by excluding all survivors with a pre-existing mental health diagnosis. Because of diagnostic delay in identifying mental illnesses,Reference Patel, Shetty, Jackson, Broadbent, Stewart and Boydell31 it is likely that some individuals will have begun to experience symptoms of these conditions before their index date in the study. One fact to note is that at baseline the exposed group had considerably higher odds of having mental illness at baseline (aOR 2.78, 95% CI 2.68–2.89). This could perhaps be owing to a significant delay in presentation of IPV to their GP, meaning that mental illness may precede the recorded exposure to IPV.
In summary, we have undertaken the first cohort study in the UK to use primary care data to ascertain the relationship between recorded IPV and mental illness. In light of the study's limitations, particularly relating to under recording of IPV, we still found an association between IPV exposure, with a twofold increase in the risk of developing anxiety, and a threefold risk increase of developing depression and SMI. Because of the sizeable public health burden posed by IPV, which is mostly under-reported, physicians should continue to pay particular attention to identifying individuals in this group. Early identification of such exposure in women presenting with depression, anxiety and SMI may improve psychological outcomes if a targeted management therapy is used. Further work is needed to explore the dose-dependent relationship between abuse and poor mental health, as well as a greater understanding of the pathway behind this. Also, this question should be explored in other UK cohorts to identify the extent of under-recording and test the effect size we have noted.
Supplementary material
Supplementary material is available online at https://doi.org/10.1192/bjp.2019.124.









eLetters
No eLetters have been published for this article.