Violent or aggressive behaviour is a common reason for emergency psychiatric presentations, with assaultive behaviour seen in 3–10% of psychiatric patients (Reference Tardif and SweillamTardiff & Sweillam, 1982; Reference Tardiff and KoenigsbergTardiff & Koenigsberg, 1985). A haloperidol–promethazine mix is commonly used for rapid tranquillisation of agitated or violent patients in India and Brazil (Reference Huf, Coutinho and FagundesHuf et al, 2002a ). Promethazine is an antihistamine that adds to the sedative effect of haloperidol and prevents acute dystonic reactions otherwise common with the intramuscular use of haloperidol (Reference Van Harten, Hoel and KahnVan Harten et al, 1999). As a haloperidol–promethazine mix had not been evaluated previously in the context of a randomised trial, two pragmatic trials were designed by the Tranquilizǎcção Rápida-Ensaio Clínic TREC Collaborative Group to assess this combination against intramuscular benzodiazepines. One trial was conducted in Rio de Janeiro (TREC–Rio) and the other in India (TREC–India). Here we present results of TREC–India and comment briefly on those of TREC–Rio. Intramuscular lorazepam is used increasingly to handle psychiatric emergencies in India, costs the same as the haloperidol–promethazine combination (£0.17) and offers the advantage of producing no dystonic or extrapyramidal adverse effects; however, its efficacy in relation to the antipsychotic–antihistamine combination is unclear. TREC–India was a pragmatic randomised trial undertaken in real-world conditions comparing an intra-muscular combination of haloperidol plus promethazine v. intramuscular lorazepam.
METHOD
Setting
This pragmatic randomised controlled trial, designed to include patients typical of those presenting to emergency services and to interfere little with routine practice, was conducted in the emergency services of the Department of Psychiatry at the Christian Medical College, in Vellore in the southern Indian state of Tamil Nadu. The majority of patients presenting to the psychiatric emergency services of this 1800-bed teaching hospital were accompanied by family members and were either brought directly, or were referred by general practitioners in the town or adjoining towns and villages and from emergency services of this and other hospitals.
Patient selection
Consecutive patients were assessed and were eligible for trial entry if the attending physician felt that intramuscular sedation was clearly indicated because of agitation, aggression or violent behaviour, and if the physician did not feel that either one of the interventions posed an additional risk for the patient. In keeping with prevailing clinical practice in this country, consent was obtained from a responsible relative if patients refused, or lacked capacity to consent to treatment by virtue of severe mental illness. For this trial relatives were fully informed and their written consent obtained; patients without a responsible relative were excluded. This trial compared two low-risk interventions in common use, the relative benefits of which are unknown. The institutional research and ethics committee approved the trial design, the consent procedure and the form used.
Sample size
From the existing literature, with tranquillisation of 73% of people given benzodiazepines and 57% given typical antipsychotics (Reference Battaglia, Moss and RushBattaglia et al, 1997; Reference Joy, Adams and LawrieJoy et al, 2003), with a power of 80% at 95% confidence intervals and an expected precision of 20%, the minimum sample size required was 90 people per arm.
Randomisation and interventions
Eligible patients were randomised to receive either intramuscular haloperidol (10 mg) and promethazine (25 or 50 mg) mixed in the same syringe, or intramuscular lorazepam (4 mg). All doses were at the discretion of the attending doctor, although the recommended dose was 10 mg haloperidol plus 50 mg promethazine, or 4 mg lorazepam. These doses were arrived at by prevailing clinical practice and a pilot study that showed that at least 4 mg lorazepam was required to achieve a similar degree of tranquillisation as the haloperidol–promethazine mix.
Randomisation was according to a computer-generated random numbers list in varying sized blocks of less than 10 prepared by the UK collaborator. This collaborator worked with a member of the TREC–India team who had no clinical responsibilities in conducting the trial. The team member and a pharmacist prepared consecutively numbered opaque cardboard boxes, identical in appearance and weight, on the outside of which was a form with questions to be completed by the attending doctor while ‘blind’ to the contents of the box. The boxes contained haloperidol (5 mg × 2 ampoules) plus promethazine (50 mg × 1 ampoules) or lorazepam (4 mg × 1 ampoule), as determined by the randomisation list, one disposable syringe and needle and study follow-up forms. All those involved clinically in the study had no indication of what medicines were in the boxes until they were opened.
Procedure
Once eligibility of a patient was ensured, the next consecutive box was taken from the emergency cupboard and this constituted randomisation. The duty doctor recorded the severity of the episode and the initial diagnosis on the form stuck to the outside of the sealed intervention pack. The box was then opened and the intervention administered. The patient was then followed up at 15, 30, 60 and 120 min by the treatment team and at 240 min and at 2 weeks by the study coordinators. Data were also obtained from the case notes as well as from interviews with relatives and the treatment team.
Blinding
The study was blind until the point of treatment assignment, which minimised selection bias. After assignment, rating for the first 2 h was not blind as the management team had to know the prescribed medications. In any event, TREC–India evaluated real-world interventions that are not given blind. The study coordinators, however, who were blind to interventions given, undertook ratings at 240 min. At this time, they also guessed the allocated intervention, to assess their blinding.
Outcomes
Patients were rated at each assessment point on whether they were tranquil or asleep; in addition, the time of onset of tranquillisation and sleep were noted. Participants were considered to be tranquil when they were calm and not exhibiting agitated, aggressive or dangerous behaviour. They were considered to be asleep if, on inspection, they appeared to be sound asleep and were not aroused by ambient disturbances; the depth of this apparent slumber was not assessed further. They were also rated on the Clinical Global Impression – Severity (CGI–S) scale at entry, and the CGI–Improvement (CGI–I) scale (Reference GuyGuy, 1976) with respect to aggression and violence, the Simpson–Angus extrapyramidal side-effects rating scale (Reference Simpson and AngusSimpson & Angus, 1970) and the Barnes Akathisia Scale (Reference BarnesBarnes, 1989) at each assessment point; any other clinically important adverse effect, especially dystonia, was also noted. These assessments were conducted only on participants who were awake, as extrapyramidal symptoms are usually not apparent during sleep or, in the case of dystonia or akathisia, are likely to prevent sleep. Other outcomes within the first 4 h were the use of additional medication for control of agitated or aggressive behaviour, the use of physical restraints, the need for further medical attention and numbers absconding. Participants were also followed up 2 weeks later to check for adverse effects or adverse outcomes and compliance with oral medication. The primary outcome was ‘tranquil or asleep by 4 h’.
Data analyses
We used double data entry and analysed data using the Statistical Package for Social Sciences (SPSS) version 9.0 for Windows. We assessed the adequacy of randomisation by comparing participants' baseline socio-demographic and clinical characteristics. We compared proportions tranquillised, asleep, improved (CGI much and very much improved, stipulated in the trial protocol), requiring restraints, requiring the doctor to be recalled and requiring additional sedation, all using the chi-squared test, with a continuity correction, or Fisher's exact test, as appropriate. We calculated relative risks and an absolute measure, the number-needed-to-treat (NNT), and their 95% confidence intervals (Reference AltmanAltman, 1998) using intention-to-treat analysis. We also used repeated measures analysis of variance to compare mean CGI–I scores between groups across various time points, with being asleep at the follow-up points entered as a covariate. We used the Mann–Whitney U-test to compare mean times to tranquillisation and sedation in the two groups, as the data did not have a normal distribution. The kappa statistic was used to evaluate agreement between the blinded guesses of the coordinators regarding treatment allocation.
RESULTS
A total of 221 patients presented to the emergency services with disruptive behaviour over a 5-month period in 2002. Twenty-one people could not be included in the trial for reasons outlined in the CONSORT diagram (Fig. 1). The remaining 200 patients were randomised. The follow-up rate for the primary outcome at 4 h was 100% for those given lorazepam and 99% for the antipsychotic–antihistamine combination. Follow-up rates for secondary outcomes and at 2 weeks were 92% for those allocated to lorazepam and 90% for people given the antipsychotic–antihistamine combination.

Fig. 1 CONSORT diagram for TREC–India.
All patients allocated to the lorazepam arm received 4 mg of the drug and everyone randomised to haloperidol–promethazine received 10 mg of haloperidol combined with 50 mg (96/100) or 25 mg (4/100) promethazine.
Most patients were male, were diagnosed to have mania (ICD–10; World Health Organization, 1992) and were rated as markedly or severely ill (Table 1). Nineteen patients with severe depression with agitation, psychotic or suicidal behaviour were judged to require parenteral medication to prevent harm to themselves or others. Groups were evenly balanced on the numbers on psychotropic medication, mean age and CGI mean scores.
Table 1 Baseline demographic and clinical characteristics
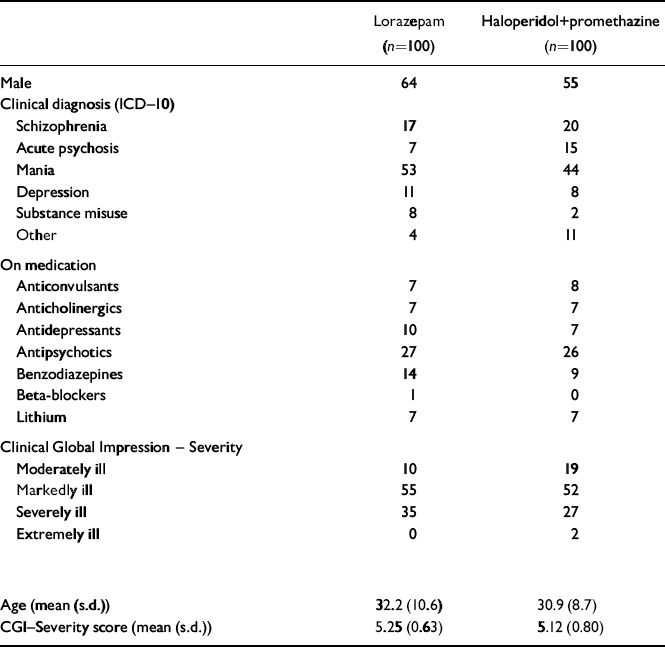
| Lorazepam | Haloperidol+promethazine | |
|---|---|---|
| (n=100) | (n=100) | |
| Male | 64 | 55 |
| Clinical diagnosis (ICD–10) | ||
| Schizophrenia | 17 | 20 |
| Acute psychosis | 7 | 15 |
| Mania | 53 | 44 |
| Depression | 11 | 8 |
| Substance misuse | 8 | 2 |
| Other | 4 | 11 |
| On medication | ||
| Anticonvulsants | 7 | 8 |
| Anticholinergics | 7 | 7 |
| Antidepressants | 10 | 7 |
| Antipsychotics | 27 | 26 |
| Benzodiazepines | 14 | 9 |
| Beta-blockers | 1 | 0 |
| Lithium | 7 | 7 |
| Clinical Global Impression – Severity | ||
| Moderately ill | 10 | 19 |
| Markedly ill | 55 | 52 |
| Severely ill | 35 | 27 |
| Extremely ill | 0 | 2 |
| Age (mean (s.d.)) | 32.2 (10.6) | 30.9 (8.7) |
| CGI–Severity score (mean (s.d.)) | 5.25 (0.63) | 5.12 (0.80) |
The study coordinators accurately guessed allocation for 58% of those given lorazepam and 33% of those given the antipsychotic–antihistamine combination (k–0.68).
Equal numbers of people (96%) were ‘tranquil or asleep’ by 4 h. The combination treatment, however, resulted in more people being tranquil/asleep by 15 min, 30 min, 1 h and 2 h (Table 2). Haloperidol plus promethazine was also superior to lorazepam in inducing sleep. The 40% difference in favour of the antipsychotic–antihistamine mix at 15 min increased to 47% by 30 min but receded to 31% by 4 h. That the combination treatment produced faster onset of tranquillisation/sedation was additionally evident when mean time to onset of tranquillisation/sleep and mean time to sleep were compared (Table 3). Four people given lorazepam were never tranquil, compared with one allocated to the haloperidol–promethazine mix. Twenty-three people given lorazepam failed to sleep at all during the 4-h follow-up compared with only eight in the combination group.
Table 2 Main outcomes
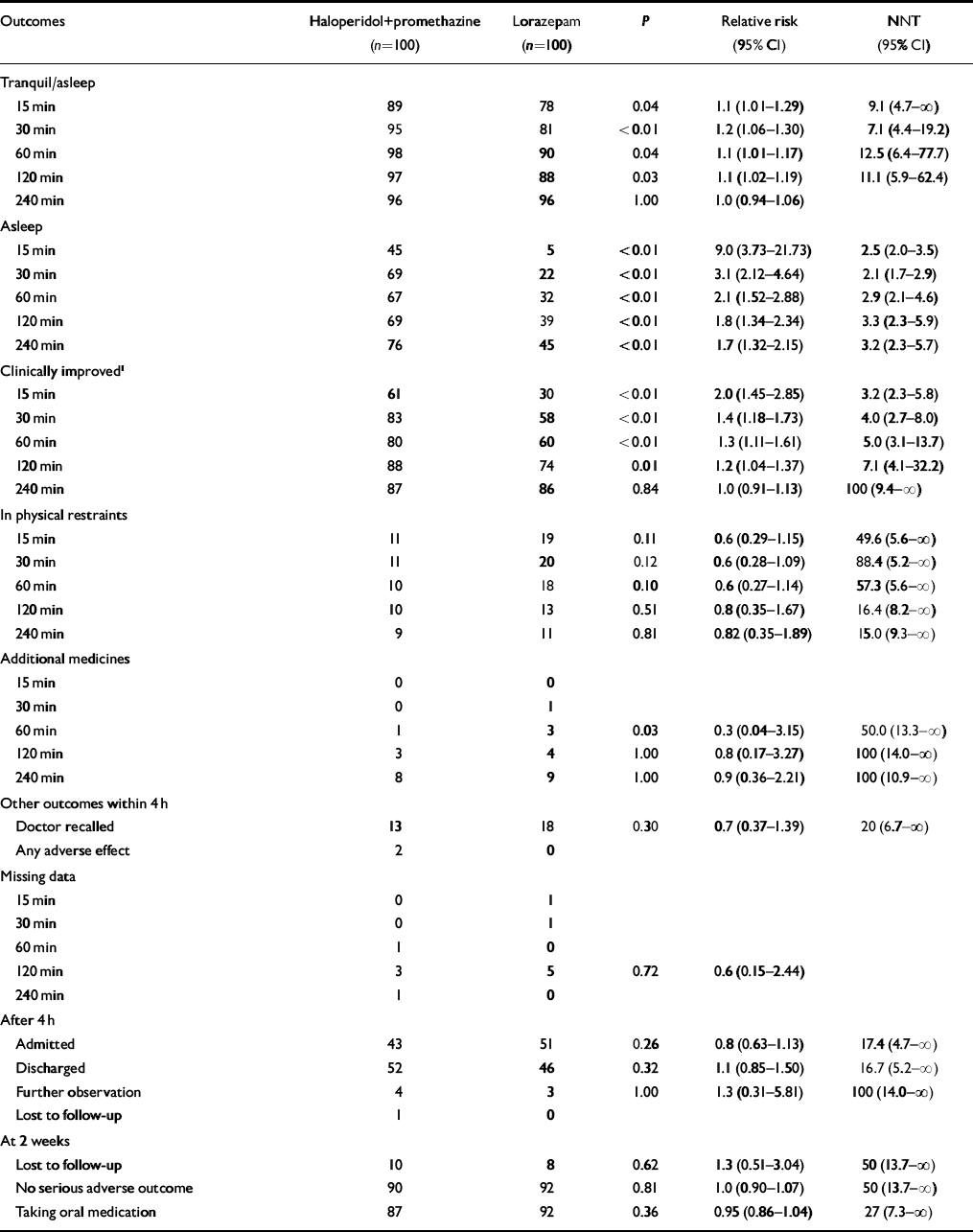
| Outcomes | Haloperidol+promethazine | Lorazepam | P | Relative risk | NNT |
|---|---|---|---|---|---|
| (n=100) | (n=100) | (95% CI) | (95% CI) | ||
| Tranquil/asleep | |||||
| 15 min | 89 | 78 | 0.04 | 1.1 (1.01-1.29) | 9.1 (4.7-∞) |
| 30 min | 95 | 81 | <0.01 | 1.2 (1.06-1.30) | 7.1 (4.4-19.2) |
| 60 min | 98 | 90 | 0.04 | 1.1 (1.01-1.17) | 12.5 (6.4-77.7) |
| 120 min | 97 | 88 | 0.03 | 1.1 (1.02-1.19) | 11.1 (5.9-62.4) |
| 240 min | 96 | 96 | 1.00 | 1.0 (0.94-1.06) | |
| Asleep | |||||
| 15 min | 45 | 5 | <0.01 | 9.0 (3.73-21.73) | 2.5 (2.0-3.5) |
| 30 min | 69 | 22 | <0.01 | 3.1 (2.12-4.64) | 2.1 (1.7-2.9) |
| 60 min | 67 | 32 | <0.01 | 2.1 (1.52-2.88) | 2.9 (2.1-4.6) |
| 120 min | 69 | 39 | <0.01 | 1.8 (1.34-2.34) | 3.3 (2.3-5.9) |
| 240 min | 76 | 45 | <0.01 | 1.7 (1.32-2.15) | 3.2 (2.3-5.7) |
| Clinically improved1 | |||||
| 15 min | 61 | 30 | <0.01 | 2.0 (1.45-2.85) | 3.2 (2.3-5.8) |
| 30 min | 83 | 58 | <0.01 | 1.4 (1.18-1.73) | 4.0 (2.7-8.0) |
| 60 min | 80 | 60 | <0.01 | 1.3 (1.11-1.61) | 5.0 (3.1-13.7) |
| 120 min | 88 | 74 | 0.01 | 1.2 (1.04-1.37) | 7.1 (4.1-32.2) |
| 240 min | 87 | 86 | 0.84 | 1.0 (0.91-1.13) | 100 (9.4-∞) |
| In physical restraints | |||||
| 15 min | 11 | 19 | 0.11 | 0.6 (0.29-1.15) | 49.6 (5.6-∞) |
| 30 min | 11 | 20 | 0.12 | 0.6 (0.28-1.09) | 88.4 (5.2-∞) |
| 60 min | 10 | 18 | 0.10 | 0.6 (0.27-1.14) | 57.3 (5.6-∞) |
| 120 min | 10 | 13 | 0.51 | 0.8 (0.35-1.67) | 16.4 (8.2-∞) |
| 240 min | 9 | 11 | 0.81 | 0.82 (0.35-1.89) | 15.0 (9.3-∞) |
| Additional medicines | |||||
| 15 min | 0 | 0 | |||
| 30 min | 0 | 1 | |||
| 60 min | 1 | 3 | 0.03 | 0.3 (0.04-3.15) | 50.0 (13.3-∞) |
| 120 min | 3 | 4 | 1.00 | 0.8 (0.17-3.27) | 100 (14.0-∞) |
| 240 min | 8 | 9 | 1.00 | 0.9 (0.36-2.21) | 100 (10.9-∞) |
| Other outcomes within 4 h | |||||
| Doctor recalled | 13 | 18 | 0.30 | 0.7 (0.37-1.39) | 20 (6.7-∞) |
| Any adverse effect | 2 | 0 | |||
| Missing data | |||||
| 15 min | 0 | 1 | |||
| 30 min | 0 | 1 | |||
| 60 min | 1 | 0 | |||
| 120 min | 3 | 5 | 0.72 | 0.6 (0.15-2.44) | |
| 240 min | 1 | 0 | |||
| After 4 h | |||||
| Admitted | 43 | 51 | 0.26 | 0.8 (0.63-1.13) | 17.4 (4.7-∞) |
| Discharged | 52 | 46 | 0.32 | 1.1 (0.85-1.50) | 16.7 (5.2-∞) |
| Further observation | 4 | 3 | 1.00 | 1.3 (0.31-5.81) | 100 (14.0-∞) |
| Lost to follow-up | 1 | 0 | |||
| At 2 weeks | |||||
| Lost to follow-up | 10 | 8 | 0.62 | 1.3 (0.51-3.04) | 50 (13.7-∞) |
| No serious adverse outcome | 90 | 92 | 0.81 | 1.0 (0.90-1.07) | 50 (13.7-∞) |
| Taking oral medication | 87 | 92 | 0.36 | 0.95 (0.86-1.04) | 27 (7.3-∞) |
Table 3 Time until onset of tranquillisation and sleep

| Time, min (mean (s.d.)) | Mann–Whitney U | P | ||
|---|---|---|---|---|
| Lorazepam | Haloperidol+promethazine | |||
| Time to tranquillisation | 47.8 (46.7) | 29.7 (35.6) | 327.0 | 0.0001 |
| Time to sleep | 80.6 (64.3) | 37.4 (42.9) | 1893.5 | <0.0001 |
The haloperidol–promethazine combination also resulted in greater numbers of people being rated as clinically improved (Table 2). Compared with lorazepam, the 31% difference at 15 min in favour of the antipsychotic–antihistamine combination receded to 14% at 2 h. By 4 h there was no difference in CGI scores between the two interventions.
The mean scores on the CGI–I scale over the 4 h of follow-up were entered into repeat measures analysis of variance (Table 4). The CGI scores showed significant differences over time as well as between groups. When being asleep was entered as a covariate to control for differential sedative effects between drugs on clinical improvement ratings, CGI scores continued to show differences over time, but the differences in CGI scores between drugs administered was not significant.
Table 4 Clinical Global Improvement (CGI) scale scores over 4h
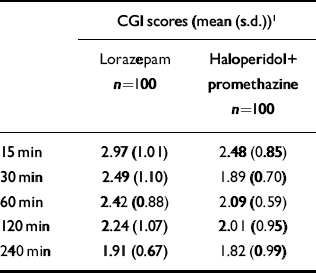
| CGI scores (mean (s.d.))1 | ||
|---|---|---|
| Lorazepam n=100 | Haloperidol+promethazine n=100 | |
| 15 min | 2.97 (1.01) | 2.48 (0.85) |
| 30 min | 2.49 (1.10) | 1.89 (0.70) |
| 60 min | 2.42 (0.88) | 2.09 (0.59) |
| 120 min | 2.24 (1.07) | 2.01 (0.95) |
| 240 min | 1.91 (0.67) | 1.82 (0.99) |
Despite the superiority of the antipsychotic–antihistamine combination in producing sedation, the two interventions did not differ in proportions of people requiring restraint, exhibiting further episodes of agitation or violence and needing additional medication, or in requiring the duty doctor to be recalled (Table 2), nor were there differences for the outcomes of admitted or discharged after 4 h, and lost to follow-up over 4 h and 2 weeks. No differences were evident between interventions in those with different clinical diagnoses, or with respect to age or gender (data available on request). None of those given the combination reported any adverse effects, whereas one person given lorazepam, who had a history of bronchial asthma, complained of moderate worsening of respiratory difficulty and another reported nausea and dizziness following the administration of the benzodiazepine. Two people given lorazepam scored 10 and 18 on the Simpson–Angus scale for extrapyramidal side-effects before the intervention, with no change in scores post-intervention. No other patient scored above zero on the extrapyramidal or akathisia scales before or after the intervention. No patient developed dystonia.
DISCUSSION
It is estimated that approximately 15 million people in India suffer from serious mental disorders (schizophrenia 2.7/1000, affective disorders 12.3/1000 and organic psychoses 0.4/1000; Reference Reddy and ChandrasekharReddy & Chandrasekhar, 1998). Although systematically ascertained prevalence data for violence among people with psychiatric disorders in low- and middle-income countries are scant, there is no evidence to suggest that the prevalence of violent or agitated behaviour is likely to be any less in low-income countries such as India than elsewhere. The magnitude of the problem faced by emergency services in India is therefore readily apparent.
Management of agitation and violence in emergency settings: lack of consensus
Drugs commonly used to manage agitation and violence in emergency situations world-wide include antipsychotics, benzodiazepines and antipsychotic and benzodiazepine combinations (Reference AllenAllen, 2002; Reference McAllister-Williams and FerrierMcAllister-Williams & Ferrier, 2002). More recent strategies include longer-acting drugs such as zuclopenthixol acetate (Reference Coutinho, Fenton and AdamsCoutinho et al, 2000) and rapidly acting intra-muscular formulations of the atypical antipsychotics olanzapine (Reference Jones, Taylor and MeehanJones et al, 2001) and ziprasidone (Reference Brook, Lucey and GunnBrook et al, 2000).
The recommendations of guidelines for the management of psychiatric emergencies (Royal College of Psychiatrists, 1998; Expert Consensus Guideline Group, 1999) are not evidence-based (Reference AllenAllen, 2002), or are they followed uniformly (Reference Pilowsky, Ring and ShinePilowsky et al, 1992; Reference CunnaneCunnane, 1994; Reference Binder and McNeilBinder & McNeil, 1999; Reference Huf, Coutinho and FagundesHuf et al, 2002a ). Evidence from randomised trials and systematic reviews is limited and does not indicate the superiority of zuclopenthixol acetate over conventional antipsychotics (Reference Fenton, Coutinho and CampbellFenton et al, 2003), or the commonly used combination of haloperidol and benzodiazepines over haloperidol alone (Reference Battaglia, Moss and RushBattaglia et al, 1997). There is a suggestion that benzodiazepines are superior to typical antipsychotics (Reference Battaglia, Moss and RushBattaglia et al, 1997; Reference AllenAllen, 2002) and that haloperidol is superior to placebo (Reference Joy, Adams and LawrieJoy et al, 2003).
Management of violence in middle- and low-income countries
The management of aggressive or violent psychiatric patients in India includes ‘talking down’ techniques, physical restraint and seclusion, as well as the use of medication. The initial minutes and hours are crucial and drugs that rapidly render people tranquil and/or sedated without producing distressing or dangerous adverse effects are desirable. In low-income countries such as India, the high cost of zuclopenthixol acetate precludes its widespread use; intramuscular atypical antipsychotics are not available and are likely to be prohibitively expensive.
TREC–India and TREC–Rio
TREC–India, the largest and only study for this comparison, randomised violent or agitated patients likely to be seen in everyday clinical practice. It compared two inexpensive, commonly used interventions for clinically relevant outcomes and lost data on only one person (0.5%) for the primary outcome and on 18 people for the 2-week follow-up (9%). Although both interventions are effective for controlling agitated or violent behaviour, with over 75% in each group tranquil/asleep within 15 min of administration and 96% in each group tranquil/asleep by 4 h, 10 mg of intramuscular haloperidol combined with 25–50 mg promethazine is superior to 4 mg intramuscular lorazepam in the speed of onset of sleep and thereby clinical improvement. If lorazepam is used alone a significant proportion of people remain awake for longer, potentially exposing everyone to danger. TREC–India did not find, however, that the reduced ability of lorazepam to sedate compared with the combination was accompanied by a greater need for subsequent intervention or harm to the patient or others.
TREC–Rio (Huf et al, Reference Huf, Coutinho and Fagundes2002a ,Reference Huf, Coutinho and Adams b ; TREC Collaborative Group, 2003) randomised 301 people over 6 months to receive either a haloperidol–promethazine mix or intramuscular midazolam. Though midazolam is available in India, it is five-times as expensive as the haloperidol and promethazine mix and is not in common use in psychiatry.
Midazolam consistently induced more rapid tranquillisation and sedation than the haloperidol–promethazine mix. However, the combination treatment in TREC–Rio was less tranquillising/sedating than in TREC–India (Table 5). Similar numbers of people in both trials were male, markedly ill and psychotic. In TREC–India, however, everyone allocated combination treatment received 10 mg haloperidol. In the TREC–Rio haloperidol–promethazine arm, 77/148 (52%) were given 5 mg haloperidol and 71/148 (48%) were given 10 mg. Most people allocated to the combination treatment in both studies were given 50 mg promethazine. Subgroup analysis of the two different doses of haloperidol in TREC–Rio, however, did not suggest differences in numbers tranquil/asleep (Evandro Coutinho, personal communication, 2003). However, evaluation of whether the dose of haloperidol matters will require a direct comparison with an adequately large sample.
Table 5 Proportion asleep if given haloperidol plus promethazine

| TREC–India | TREC–Rio | ||
|---|---|---|---|
| n (%) | n (%) | ||
| 15 min | 45 (45) | 20 min | 43 (29) |
| 30 min | 69 (69) | 40 min | 69 (46) |
| 60 min | 67 (67) | 60 min | 83 (55) |
| 120 min | 88 (88) | 120 min | 95 (63) |
TREC–India did not compare haloperidol alone with a haloperidol–lorazepam combination, but adding promethazine to haloperidol could be superior to adding lorazepam for promoting tranquillisation and sedation and superior to haloperidol alone for preventing extrapyramidal adverse effects such as acute dystonia or akathisia (Reference Salzman, Solomon and MiyawakiSalzman et al, 1991; Reference Battaglia, Moss and RushBattaglia et al, 1997; Reference Brook, Lucey and GunnBrook et al, 2000). No serious adverse effects, particularly those related to the extrapyramidal system, were reported for either treatment, although the moderate worsening of respiratory difficulty reported with lorazepam is in keeping with the known association of benzodiazepines with respiratory depression.
About 15% of people in this trial were physically restrained and less than 10% were given additional medication over the 4 h. This common practice in India and Brazil (Reference Huf, Coutinho and FagundesHuf et al, 2002a ) of physically restraining disruptive patients after administration of a parenteral drug as opposed to administering additional medication requires evaluation.
The two treatment regimens evaluated in this study are inexpensive, effective and available worldwide. Where rapid sedation is needed a combination of intramuscular haloperidol and promethazine is superior to intramuscular lorazepam.
Clinical Implications and Limitations
CLINICAL IMPLICATIONS
-
▪ Intramuscular lorazepam (4 mg) is as effective as haloperidol (10 mg) plus promethazine (25/50 mg) in controlling violence or agitation in people with mental disorders.
-
▪ If rapid sedation is required, the haloperidol–promethazine combination is superior to lorazepam.
-
▪ Pragmatic randomised trials of interventions relevant to low-income countries, with limited funding, clinically meaningful outcomes and low attrition rates, are possible within the field of mental health.
LIMITATIONS
-
▪ Assessments over the first 2 h were not blind and were carried out by multiple raters.
-
▪ The effects of both interventions could be dose-related.
-
▪ Haloperidol alone or in combination with a benzodiazepine was not evaluated.
Acknowledgements
The TREC–India team acknowledges the contribution of Giselle Huf in the design of TREC–Rio, on which this trial is modelled. We thank Evandro Coutinho for sharing unpublished data from TREC–Rio. This trial would not have been possible but for the considerable support and involvement of many doctors, nurses and support staff at the department of psychiatry. We thank Mr N. Ravi for help with preparing the TREC boxes. This trial was funded by intramural research grants from the Fluid Research Fund of the Christian Medical College, Vellore and the Cochrane Schizophrenia Group general fund.









eLetters
No eLetters have been published for this article.