Summations
-
• Glucagon-like peptide-1 receptor agonists (liraglutide, semaglutide, and exendin-4) were found to restore autophagic flux in both cell and animal models of neurologic disorders.
-
• Some discrepancies in autophagy-specific marker levels were detected between studies in response to disease induction and intervention administration, potentially due to a lack of assessing dynamic autophagy.
Considerations
-
• No human studies were included, limiting translatability to human populations.
-
• The included preclinical studies had varying models, intervention type (liraglutide, semaglutide, or exendin-4), dose, and end points, making direct comparisons challenging.
Introduction
Alzheimer’s disease (AD) and Parkinson’s disease (PD) are progressive neurodegenerative disorders that negatively impact the lives of those afflicted (Caligiore et al., Reference Caligiore, Giocondo and Silvetti2022). AD is the most common form of dementia, with an estimated prevalence of 416 million persons worldwide from 2010 to 2020 and growing incidence rates (increasing by 147.95% from 1990 to 2019) (Gustavsson et al., Reference Gustavsson, Norton, Fast, Frölich, Georges, Holzapfel, Kirabali, Krolak-Salmon, Rossini, Ferretti, Lanman, Chadha and van der Flier2023; Li et al., Reference Li, Feng, Sun, Hou, Han and Liu2022). AD is primarily characterised by memory impairment and cognitive decline (Alzheimer’s disease facts and figures, 2024). Additionally, hallmark neuropathological features include the presence of extracellular amyloid-beta (aβ) plaques and intracellular neurofibrillary tangles (NFTs) (DeTure and Dickson, Reference DeTure and Dickson2019). The accumulation and aggregation of the aforementioned features is associated with impaired synaptic communication, dysfunctional mitochondria, neuroinflammation, and neuronal death, among other complications (Bloom, Reference Bloom2014).
Meanwhile, the global incidence cases of PD in 2021 were approximately 1.34 million, with a prevalence of roughly 11.77 million persons worldwide (a 273.76% increase from 1990) (Luo et al., Reference Luo, Qiao, Li, Wen, Zhang and Li2024; Li et al., Reference Li, Ye, Huang, Ye and Chen2025). PD is primarily characterised by motor manifestations, including bradykinesia, resting tremor, and muscular rigidity. However, a wide spectrum of non-motor features, such as affective disturbances and cognitive impairments, also constitute core components of the disorder and may exert an equal or even greater impact on patients’ quality of life (Hayes, Reference Hayes2019). Typical pathological characteristics of PD include the loss of dopaminergic neurons in the substantia nigra (SN) and the misfolding and clumping of alpha-synuclein (α-syn), which eventually forms Lewy bodies. The aggregation and accumulation of these toxic proteins are thought to contribute to neuroinflammation, disrupted synaptic function, mitochondrial impairment, and cell death (Kim et al., Reference Kim, Kågedal and Halliday2014; Reich and Savitt, Reference Reich and Savitt2019).
In addition to neurodegenerative disorders, chronic mental disorders [e.g., Major Depressive Disorder (MDD) and Bipolar Disorder (BD)] are also associated with substantial morbidity and mortality (Maj et al., Reference Maj, Stein, Parker, Zimmerman, Fava, De Hert, Demyttenaere, McIntyre, Widiger and Wittchen2020; McIntyre et al., Reference McIntyre, Berk, Brietzke, Goldstein, López-Jaramillo, Kessing, Malhi, Nierenberg, Rosenblat, Majeed, Vieta, Vinberg, Young and Mansur2020, Reference McIntyre, Alsuwaidan, Baune, Berk, Demyttenaere, Goldberg, Gorwood, Ho, Kasper, Kennedy, Ly-Uson, Mansur, McAllister-Williams, Murrough, Nemeroff, Nierenberg, Rosenblat, Sanacora, Schatzberg, Shelton, Stahl, Trivedi, Vieta, Vinberg, Williams, Young and Maj2023).
Notably, several studies have demonstrated that the pathophysiology of the aforementioned conditions includes alterations in cellular autophagy (Uddin et al., Reference Uddin, Stachowiak, Mamun, Tzvetkov, Takeda, Atanasov, Bergantin, Abdel-Daim and Stankiewicz2018; Nechushtai et al., Reference Nechushtai, Frenkel and Pinkas-Kramarski2023). Autophagy is the regulated process by which intracellular components are delivered to the lysosome to either be degraded or recycled.
There are three types of autophagy, which are primarily characterised by their method of cargo delivery: macro-autophagy, micro-autophagy, and chaperone-mediated autophagy (CMA) (Levine and Kroemer, Reference Levine and Kroemer2008). Macro-autophagy has received disproportionality greater emphasis in research. Briefly, macro-autophagy (hereafter referred to as autophagy) involves autophagosomes, double-membraned vesicles that are newly created in the cytosol, which ultimately seal cargo inside to be transported and degraded in the lysosome via vesicular fusion. In micro-autophagy, cytoplasmic components are directly internalised via invagination of the lysosomal membrane, forming intraluminal vesicles for breakdown. In contrast, CMA is highly selective: chaperone proteins recognise cytosolic proteins with a targeting motif and translocate them across the lysosomal membrane for degradation (Parzych and Klionsky, Reference Parzych and Klionsky2014; Kaushik and Cuervo, Reference Kaushik and Cuervo2018).
Autophagy is triggered by numerous cellular stress stimuli, including but not limited to redox stress, endoplasmic reticulum (ER) stress, mitochondrial damage, nutrient deprivation, hypoxia, and immune signals, resulting in the activation of various pathways coordinated by several autophagy-related gene (ATG) proteins (Kroemer et al., Reference Kroemer, Mariño and Levine2010; Shibutani et al., Reference Shibutani, Saitoh, Nowag, Münz and Yoshimori2015; Raudenska et al., Reference Raudenska, Balvan and Masarik2021). Any defects in autophagy disrupt cellular homeostasis and can lead to serious consequences potentially linked to neurodegenerative and other mental disorders (Wong and Cuervo, Reference Wong and Cuervo2010).
Notably, replicated evidence suggests that glucagon-like peptide-1 receptor agonists (GLP-1RAs), originally developed for type 2 diabetes (T2D), may be effective in the treatment and prevention of neurodegenerative and other mental disorders (Au et al., Reference Au, Lam, Lim and McIntyre2025b; Lee et al., Reference Lee, Yin, Xiao, Rhee, Lo, Wong, Fox, Teopiz, Lam, Zheng, Le, Mansur, Rosenblat and McIntyre2025; McIntyre et al., Reference McIntyre, Rasgon, Goldberg, Wong, Le, Mansur, Rosenblat, Teopiz and Stahl2025; Hölscher, Reference Hölscher2024). Their neuroprotective effect may partly be due to the essential role of brain insulin signalling in synaptic plasticity and neuronal survival (Wong et al., Reference Wong, Le, Dri, Teopiz and McIntyre2025; Zheng et al., Reference Zheng, Soegiharto, Au, Valentino, Le, Wong, Teopiz, Rhee, Guillen-Burgos, Cao and McIntyre2025). Since brain insulin resistance is commonly found in neurodegenerative disorders, it is plausible that the insulin-sensitising effect of GLP-1RAs could ameliorate this and restore cognitive function (Watson et al., Reference Watson, Wroolie, Tong, Foland-Ross, Frangou, Singh, McIntyre, Roat-Shumway, Myoraku, Reiss and Rasgon2019; Hong et al., Reference Hong, Chen and Hu2024; Sun and Mi, Reference Sun and Mi2025). However, beyond this role, GLP-1RAs’ mechanistic basis in the treatment of psychiatric disorders implicates several other systems, with accumulating evidence suggesting direct influence on autophagy (Zhang et al., Reference Zhang, Yan, Yu, Cheng, Yi, Guo, Liu, Shang, Wang, Hu and Chen2021b; Hong et al., Reference Hong, Chen and Hu2024). The overarching objective herein is to synthesise results of studies that have evaluated the effects of GLP-1RAs on molecular and cellular aspects of autophagy to assess the therapeutic potential of these agents in neurodegenerative disorders. investigate the effect of GLP-1RAs on autophagy and the implications of repurposing these agents as a potential prevention and treatment of neurodegenerative disorders, such as AD and PD.
Methods
Search strategy
This study was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) (Page et al., Reference Page, McKenzie, Bossuyt, Boutron, Hoffmann, Mulrow, Shamseer, Tetzlaff, Akl, Brennan, Chou, Glanville, Grimshaw, Hróbjartsson, Lalu, Li, Loder, Mayo-Wilson, McDonald, McGuinness, Stewart, Thomas, Tricco, Welch, Whiting and Moher2021). A literature search was performed for studies that investigated the effect of GLP-1RAs on autophagy in models of neurodegenerative disorders, specifically AD and PD.
A systematic search was conducted on PubMed, Web of Science, and OVID (including Medline, Embase, and APA PsycInfo databases) from inception to June 17, 2025 for English-language articles using the following search string: ((‘GLP-1’ OR ‘Glucagon-Like Peptide-1’ OR ‘GLP-1 agonist’ OR ‘GLP-1 receptor agonist’ OR ‘Glucagon-Like Peptide-1 agonist’ OR ‘Glucagon-Like Peptide-1 receptor agonist’ OR ‘Glucagon like peptide-1’ OR ‘Semaglutide’ OR ‘Ozempic’ OR ‘Wegovy’ OR ‘Rybelsus’ OR ‘Dulaglutide’ OR ‘Trulicity’ OR ‘Liraglutide’ OR ‘Saxenda’ OR ‘Victoza’ OR ‘Tirzepatide’ OR ‘Mounjaro’ OR ‘Zepbound’ OR ‘Exenatide’ OR ‘Byetta’ OR ‘Bydureon’ OR ‘Bydureon BCise’) AND (Autophagy) AND (‘Alzheimer’s’ OR ‘Alzheimer’s Disease’ OR ‘Neurodegenerative Disorder’ OR ‘Dementia’ OR’ Parkinson’s’ OR ‘Parkinson’s Disease’ OR ‘Neurological Disorder’ OR ‘Memory Disorder’)).
Subsequently, two reviewers (M.C.S. and I.H.) independently screened the studies on Covidence to prevent any influence on decisions from either of the reviewers and to enable transparency with the screening process. After duplicates were removed, articles were assessed based on their title, abstract, and full text according to the eligibility inclusion/exclusion criteria. Conflicts on screening were resolved via a discussion and a consensus decision.
Eligibility criteria
Any experimental studies using in vitro or in vivo models to investigate AD or PD were included. The presence of AD in these models was assessed based on the detection of two core biomarkers: (1) amyloid-beta (aβ) plaques and (2) NFTs of abnormally phosphorylated tau proteins (Jack et al., Reference Jack, Andrews, Beach, Buracchio, Dunn, Graf, Hansson, Ho, Jagust, McDade, Molinuevo, Okonkwo, Pani, Rafii, Scheltens, Siemers, Snyder, Sperling, Teunissen and Carrillo2024). Meanwhile, PD models were included based on the presence of key pathological features, including α-syn aggregation and the progressive loss of dopaminergic neurons (Ioghen et al., Reference Ioghen, Ceafalan and Popescu2023; Pandey et al., Reference Pandey, Karmakar, Majie, Dwivedi, Md and Gorain2024). Furthermore, the presence of key clinical motor symptoms in in vivo samples must be observed: bradykinesia in combination with rigidity or resting tremors, or both (Postuma et al., Reference Postuma, Berg, Stern, Poewe, Olanow, Oertel, Obeso, Marek, Litvan, Lang, Halliday, Goetz, Gasser, Dubois, Chan, Bloem, Adler and Deuschl2015).
Studies were included if they investigated the effect of FDA-approved GLP-1RAs in comparison to controls who received either a placebo or no intervention. Additionally, studies that reported any changes in autophagy along with improvements in core symptoms of AD or PD were included.
Studies were excluded if they: (1) did not have participants with AD or PD (i.e., had any other neurodegenerative condition); (2) used non-FDA-approved GLP-1RAs or any form of dipeptidyl peptidase-4 inhibitors due to any potential differences in mechanism from GLP-1RAs; (3) did not assess autophagy as an outcome; (4) had any other study design except an experimental/preclinical or randomised controlled trial (RCT).
Data extraction and analysis
Data extraction was performed independently by two reviewers (M.C.S. and I.H.) on Google Spreadsheets. The studies were split into two sections: an in vitro section and an in vivo section. Once in their respective sections, the studies were organised and analysed through several components: (1) author; (2) disease model (AD or PD); (3) study design (experimental vs RCT); (4) cell type used (for in vitro studies) or animal model characteristics (for in vivo studies); (5) stressor used; (6) sample size; (7) type of GLP-1RA used and its dosage; (8) groups (control(s) and experimental); (9) main findings pertaining to autophagy. To enable some level of comparability between all the studies, a focus on extracting data on autophagy-specific markers such as beclin-1, ATG7, ATG3, microtubule-associated protein light chain 3 (LC3-II/LC3-I), lysosomal-associated membrane protein 1 (LAMP1), and p62 [also commonly known as sequestosome 1 (SQSTM1)] was done. The extracted data were assessed for inclusion by both reviewers.
Assessment of reporting biases
Risk of bias (RoB) was assessed by two independent reviewers (M.C.S. and I.H.). The SYRCLE’s RoB tool for animal studies was utilised for in vivo studies (Hooijmans et al., Reference Hooijmans, Rovers, de Vries, Leenaars, Ritskes-Hoitinga and Langendam2014). Meanwhile, the Modified SYRCLE’s RoB tool was employed for in vitro studies (De la Rosa González et al., Reference De la Rosa González, Guerra-Ojeda, Camacho-Villa, Valls, Alegre, Quintero-Bernal, Martorell, Chenoll, Serna-García, Mauricio and Serna2024). All conflicts were resolved by discussion between the two reviewers.
Results
Search results
The initial literature search yielded 142 studies, of which 64 duplicates were identified on Covidence and 5 were identified manually and removed. Based on title and abstract screening, 42 studies were deemed irrelevant, while the remaining 31 studies progressed onto full-text screening. Seventeen studies were excluded for having either the wrong intervention [e.g., a dual agonist or non-FDA-approved GLP-1RAs (n = 10)], incorrect patient population [e.g., samples were not models of AD or PD but of other conditions (n = 4)], or wrong outcomes [i.e., did not look at autophagy as an outcome (n = 3)]. As a result, 14 articles were included in the review herein and underwent data extraction (Figure 1).
Preferred reporting items for systematic reviews and meta-analyses (PRISMA) flow diagram. The detailed procedure of screening and selecting studies that assess the effect of glucagon-like peptide-1 receptor agonists on autophagy.

Study characteristics
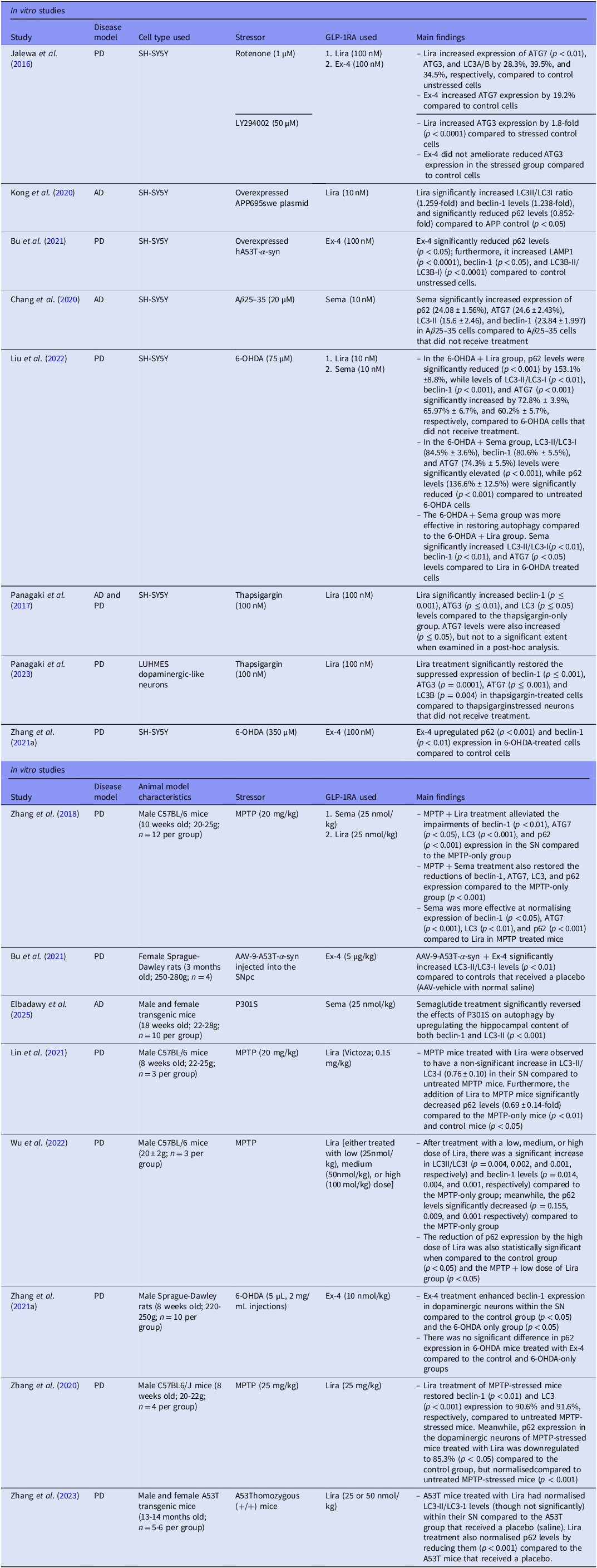
Of the total 14 articles included, two studies (Bu et al., Reference Bu, Liu, Shen, Fan, Yu, Jiang, Tang, Yang, Wu, Zuo, Koprich, Liu, Wu and Wang2021; Zhang et al., Reference Zhang, Jin, Hölscher and Li2021a) incorporated both in vivo and in vitro experiments; therefore, each component was treated as distinct and assessed separately during data extraction. Accordingly, 8 studies were considered in vivo RCTs, while the other 8 were in vitro experimental studies. Of the in vitro cell studies, only 1 used Lund Human Mesencephalic (LUHMES) dopaminergic-like neurons (Panagaki et al., Reference Panagaki, Randi, Szabo and Hölscher2023) while the rest used the SH-SY5Y human neuroblastoma cell line as their model. For their intervention, a total of 3 studies examined liraglutide (Lira) (Panagaki et al., Reference Panagaki, Michael and Hölscher2017; Kong et al., Reference Kong, Wan, Wang, Zhang and Zhang2020; Panagaki et al., Reference Panagaki, Randi, Szabo and Hölscher2023), 2 studies investigated exendin-4 (Ex-4) (Bu et al., Reference Bu, Liu, Shen, Fan, Yu, Jiang, Tang, Yang, Wu, Zuo, Koprich, Liu, Wu and Wang2021; Zhang et al., Reference Zhang, Jin, Hölscher and Li2021a), and 1 study explored semaglutide (Sema) (Chang et al., Reference Chang, Zhang, Hu, Liu and Li2020); furthermore, 2 studies investigated more than 1 GLP-1RA: 1 study examined Lira and Ex-4 (Jalewa et al., Reference Jalewa, Sharma and Hölscher2016), and 1 study looked at Lira and Sema (Liu et al., Reference Liu, Zhao, Wei, Ma and Wu2022).
From the included in vivo studies, only 3 used transgenic models (Bu et al., Reference Bu, Liu, Shen, Fan, Yu, Jiang, Tang, Yang, Wu, Zuo, Koprich, Liu, Wu and Wang2021; Zhang et al., Reference Zhang, Shi, Li, Ling, Zhai, Yuan, Ma, Hao, Li, Zhang and Hölscher2023; Elbadawy et al., Reference Elbadawy, Saad, Elfarrash, Ahmed and Abdelkader2025), while the others primarily utilised environmental stressors such as 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP), 6-hydroxydopamine (6-OHDA), or thapsigargin. For their intervention, a total of 4 studies examined Lira (Zhang et al., Reference Zhang, Zhang, Li, Li, Melchiorsen, Rosenkilde and Hölscher2020; Lin et al., Reference Lin, Lin, Lin, Lin, Lan, Wang, Wang, Wang, Tsai, Liou and Chuang2021; Wu et al., Reference Wu, Dong, Chen, Guan, Cao, Zhang, Qi, Guan and Wang2022; Zhang et al., Reference Zhang, Shi, Li, Ling, Zhai, Yuan, Ma, Hao, Li, Zhang and Hölscher2023), 2 studies investigated Ex-4 (Bu et al., Reference Bu, Liu, Shen, Fan, Yu, Jiang, Tang, Yang, Wu, Zuo, Koprich, Liu, Wu and Wang2021; Zhang et al., Reference Zhang, Jin, Hölscher and Li2021a), 1 study explored Sema (Elbadawy et al., Reference Elbadawy, Saad, Elfarrash, Ahmed and Abdelkader2025), and 1 study looked at both Lira and Sema (Zhang et al., Reference Zhang, Zhang, Li and Hölscher2018).
Overall, 5 articles also included dual agonists as another comparison intervention; however, only results pertaining to FDA-approved single GLP-1RAs were included from these studies (Jalewa et al., Reference Jalewa, Sharma and Hölscher2016; Zhang et al., Reference Zhang, Zhang, Li, Li, Melchiorsen, Rosenkilde and Hölscher2020; Zhang et al., Reference Zhang, Jin, Hölscher and Li2021a; Panagaki et al., Reference Panagaki, Randi, Szabo and Hölscher2023; Zhang et al., Reference Zhang, Shi, Li, Ling, Zhai, Yuan, Ma, Hao, Li, Zhang and Hölscher2023). A summary of the study characteristics is exhibited in Table 1.
Study characteristics of in vitro and in vivo studies included

Risk of bias assessment
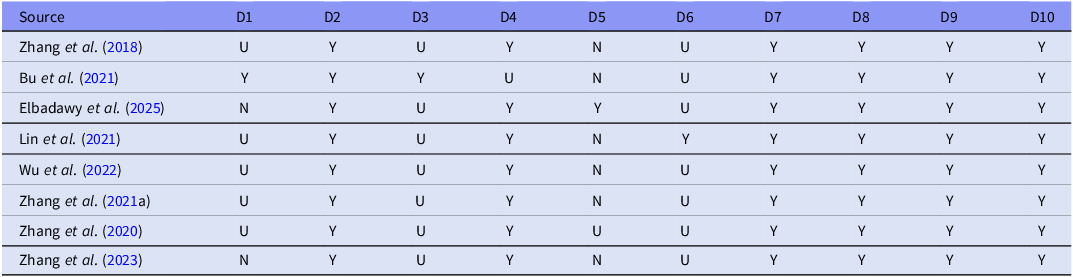
Details of the RoB assessment are presented in Tables 2 and 3 for in vivo and in vitro studies, respectively. In the majority of the in vivo studies, a high risk of selection and performance bias was due to a lack of randomisation and blinding of investigators to intervention groups. Additionally, an unclear RoB was mainly given in circumstances where the criteria of the domain were partially met but not enough to ensure a low RoB. It must be noted that, although all the studies except one (Elbadawy et al., Reference Elbadawy, Saad, Elfarrash, Ahmed and Abdelkader2025) did not have blinded outcome assessors (D7), this was not deemed a serious concern in terms of bias, as the animals were euthanised to measure the outcome related to autophagy. Furthermore, regarding whether the assessment of incomplete data was adequately addressed (D8), only sections related to autophagy were examined for this domain, since only data related to autophagy were extracted.
In vivo risk of bias assessment

Risk of bias domains: D1: Selection bias - Sequence generation; D2: Selection bias - Baseline characteristics; D3: Selection bias - Allocation concealment; D4: Performance bias - Random housing; D5: Performance bias - Blinding; D6: Detection bias - Random outcome assessment; D7: Detection bias - Blinding; D8: Attrition bias - Incomplete outcome data; D9: Reporting bias - Selective outcome reporting; D10: Other - Other sources of bias.
Judgment: Yes (Y) - low risk of bias; Unclear (U) - unclear risk of bias; No (N) - high risk of bias.
In vivo risk of bias assessment

Risk of bias domains: D1: Selection bias - Sample size calculation; D2: Selection bias - Baseline characteristics; D3: Performance bias - Detailed explanation of intervention; D4: Performance bias - Detailed explanation of culture conditions; D5: Detection bias - Details of comparison group; D6: Detection bias - Method of measurement outcome; D7: Detection bias - Blinding; D8: Attrition bias - Incomplete outcome data; D9: Reporting bias - Selective outcome reporting; D10: Other - Other sources of bias.
Judgment: Yes (Y) - low risk of bias; Unclear (U) - unclear risk of bias; No (N) - high risk of bias.
As for the in vitro studies, a high risk of detection bias (D7) was due to a lack of blinding of the outcome assessor; moreover, an unclear RoB for other sources of bias (D10) was given to studies that had potentially concerning conflicts of interest, among other reasons.
Results of individual studies
In vivo autophagy-specific biomarkers
Investigations conducted by Zhang et al. (Reference Zhang, Zhang, Li and Hölscher2018); Lin et al. (Reference Lin, Lin, Lin, Lin, Lan, Wang, Wang, Wang, Tsai, Liou and Chuang2021); Wu et al. (Reference Wu, Dong, Chen, Guan, Cao, Zhang, Qi, Guan and Wang2022); and Zhang et al. (Reference Zhang, Zhang, Li, Li, Melchiorsen, Rosenkilde and Hölscher2020) administered MPTP to male C57BL/6 mice to induce PD-like stress.
Zhang et al. (Reference Zhang, Zhang, Li and Hölscher2018) observed a significant suppression of autophagy markers, beclin-1, ATG7, LC3, and p62, in the SN of MPTP-treated mice compared to the control group treated with normal saline (p < 0.001) (n = 12 per group). Administration of Lira to MPTP-treated mice was reported to restore levels of beclin-1 (p < 0.01), ATG7 (p < 0.05), LC3 (p < 0.001), and p62 (p < 0.001), compared to the MPTP-only group. Likewise, Sema treatment also alleviated the levels of these autophagy-specific markers in MPTP-treated mice (p < 0.001). Overall, Sema was more effective at normalising the expression of beclin-1 (p < 0.05), ATG7 (p < 0.001), LC3 (p < 0.01), and p62 (p < 0.001) compared to MPTP mice treated with Lira.
Lin et al. (Reference Lin, Lin, Lin, Lin, Lan, Wang, Wang, Wang, Tsai, Liou and Chuang2021) reported a significant reduction of LC3-II/LC3-I (p < 0.001) and increased p62 accumulation (p < 0.05) in the SN of MPTP-treated mice compared to control mice that received saline (n = 3 per group). Although mice that only received Lira treatment had significantly increased LC3-II/LC3-I (p < 0.05) compared to control mice, MPTP mice treated with Lira were observed to have a non-significant increase in LC3-II/LC3-I compared to MPTP-only treated mice. Mice that only received Lira treatment also had reduced p62 levels compared to control mice (p < 0.01). Furthermore, MPTP-mice treated with Lira had significantly decreased p62 levels compared to control (p < 0.05) and MPTP-only treated mice (p < 0.01).
Wu et al. (Reference Wu, Dong, Chen, Guan, Cao, Zhang, Qi, Guan and Wang2022) found a significant decrease in beclin-1 (p = 0.006) and LC3-II/LC3-I (p = 0.016) in MPTP-treated mice, as well as a significant increase in p62 (p = 0.051), compared to control mice that only received a saline injection (n = 3 per group). Upon treatment with either a low (25 nmol/kg), medium (50 nmol/kg), or high (100 nmol/kg) dose of Lira, beclin-1 (p = 0.014, 0.004, and 0.001, respectively) and LC3-II/LC3-I (p = 0.004, 0.002, and 0.001, respectively) were significantly increased, while p62 levels were significantly reduced (p = 0.155, 0.009, and 0.001, respectively) compared to MPTP-only treated mice. Additionally, the high dose of Lira was found to be more effective at reducing p62 levels compared to the control mice (p < 0.05) and MPTP-treated mice that received the low dose of Lira (p < 0.05).
Zhang et al. (Reference Zhang, Zhang, Li, Li, Melchiorsen, Rosenkilde and Hölscher2020) reported reduced beclin-1 (p < 0.01), LC3 (p < 0.01), and p62 (p < 0.001) levels in the SN of MPTP-treated mice compared to control mice that received saline (n = 4 per group). MPTP mice treated with Lira were observed to have restored expression of beclin-1 (p < 0.01), LC3 (p < 0.001), and p62 (p < 0.001) compared to untreated MPTP mice.
Studies Bu et al. (Reference Bu, Liu, Shen, Fan, Yu, Jiang, Tang, Yang, Wu, Zuo, Koprich, Liu, Wu and Wang2021) and Zhang et al. (Reference Zhang, Shi, Li, Ling, Zhai, Yuan, Ma, Hao, Li, Zhang and Hölscher2023) utilised transgenic mice to model PD, while Elbadawy et al. (Reference Elbadawy, Saad, Elfarrash, Ahmed and Abdelkader2025) used transgenic mice to induce AD-like stress.
Zhang et al. (Reference Zhang, Shi, Li, Ling, Zhai, Yuan, Ma, Hao, Li, Zhang and Hölscher2023) investigated the effects of Lira in male and female A53T homozygous mice (n = 5–6 per group). A53T (+/+) mice overexpressed the human version of α-syn and were compared to control A53T (–/–) mice that received normal saline. A53T (+/+) mice that received normal saline were observed to have significantly lower levels of LC3-II/LC3-I (p < 0.001) and higher levels of p62 (p < 0.05) in the SN compared to control mice. A53T (+/+) mice that received Lira treatment were reported to have restored LC3-II/LC3-I levels, though not significantly, compared to saline-treated A53T (+/+) mice. Lira treatment also normalised p62 protein levels by reducing them compared to A53T (+/+) mice that received normal saline (p < 0.001).
Bu et al. (Reference Bu, Liu, Shen, Fan, Yu, Jiang, Tang, Yang, Wu, Zuo, Koprich, Liu, Wu and Wang2021) injected AAV-9-A53T-α-syn into the right substantia nigra pars compacta (SNpc) of female Sprague-Dawley rats (n = 4). Treatment of these rats with Ex-4 significantly increased the LC3-II/LC3-I ratio (p < 0.01) compared to rats that received a placebo (AAV-vehicle and normal saline).
Elbadawy et al. (Reference Elbadawy, Saad, Elfarrash, Ahmed and Abdelkader2025) utilised male and female P301S mice (n = 20) as a model of taupathy. P301S mice treated with saline (n = 10) were found to have significantly reduced hippocampal levels of beclin-1 and LC3-II compared to control C57BL/6 wild-type mice that received saline (n = 10) (p < 0.001). However, P301S mice treated with Sema (n = 10) had upregulated hippocampal content of both beclin-1 and LC3-II compared to P301S mice that received saline (p < 0.001).
Zhang et al. (Reference Zhang, Jin, Hölscher and Li2021a) induced PD-like stress in male Sprague-Dawley rats (n = 10 per group) via 6-OHDA injections. Treatment of 6-OHDA mice with Ex-4 enhanced beclin-1 expression in dopaminergic neurons within the SN compared to a sham group of mice that received saline injections (p < 0.05) and untreated 6-OHDA mice (p < 0.05). However, there was no significant effect of Ex-4 on p62 expression in 6-OHDA-treated mice compared to the sham and untreated 6-OHDA mice.
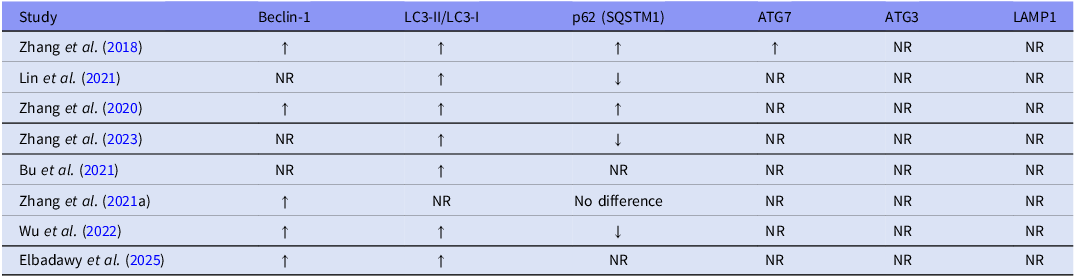
The general influence of the tested GLP-1RAs on autophagy-specific markers, reported in each in vivo study, is summarised in Table 4.
General trends of the tested GLP-1RAs on autophagy-specific markers in each in vivo study

Assessment: ↑, reported increase in expression of autophagy-specific marker by GLP-1RA(s); ↓, reported decrease in expression of autophagy-specific marker by GLP-1RA(s); NR, not reported.
In vitro autophagy-specific biomarkers
Studies Zhang et al. (Reference Zhang, Jin, Hölscher and Li2021a) and Liu et al. (Reference Liu, Zhao, Wei, Ma and Wu2022) treated SH-SY5Y cells with 6-OHDA as a model of PD.
Liu et al. (Reference Liu, Zhao, Wei, Ma and Wu2022) reported a significant reduction in beclin-1, ATG7, and LC3-II/LC3-I, as well as increased p62 levels, in 6-OHDA-treated cells compared to the control group (p < 0.001). Treatment of 6-OHDA cells with Lira was observed to significantly enhance levels of beclin-1 (p < 0.001), ATG7 (p < 0.001), and LC3-II/LC3-I (p < 0.01), while significantly reducing levels of p62 (p < 0.001) compared to cells that only received 6-OHDA. Meanwhile, 6-OHDA cells treated with Sema were found to have elevated levels of beclin-1, ATG7, and LC3-II/LC3-I compared to untreated 6-OHDA cells (p < 0.001); furthermore, Sema treatment decreased levels of p62 compared to the untreated 6-OHDA cells (p < 0.001).
Zhang et al. (Reference Zhang, Jin, Hölscher and Li2021a) reported that 6-OHDA cells treated with Ex-4 had upregulated p62 expression compared to the control group (p < 0.001) and untreated 6-OHDA cells (p < 0.01). Furthermore, Ex-4 treatment was observed to also enhance beclin-1 expression levels compared to the control group (p < 0.01) and untreated 6-OHDA cells (p < 0.05).
Studies Panagaki et al. (Reference Panagaki, Michael and Hölscher2017) and Panagaki et al. (Reference Panagaki, Randi, Szabo and Hölscher2023) utilised a thapsigargin, a sarcoplasmic reticulum ATPase inhibitor to induce stress.
Panagaki et al. (Reference Panagaki, Randi, Szabo and Hölscher2023) used LUMES dopaminergic-like neurons as a model of PD. Neurons treated with thapsigargin had reduced neuronal expression of autophagy-specific markers, beclin-1, ATG3, ATG7, and LC3B, compared to the control (p ≤ 0.001). However, Lira treatment restored the suppressed expression of these markers compared to thapsigargin-only stressed neurons (p ≤ 0.001).
Panagaki et al. (Reference Panagaki, Michael and Hölscher2017) utilised SH-SY5Y cells as a general model of PD and AD. When treated with thapsigargin, the cells had significantly suppressed expression of beclin-1 (p ≤ 0.001), ATG3 (p ≤ 0.01), ATG7 (p ≤ 0.001), and LC3 (p ≤ 0.001) compared to the control group. However, Lira treatment of these cells elevated levels of beclin-1 (p ≤ 0.001), ATG3 (p ≤ 0.01), ATG7, and LC3 (p ≤ 0.05) compared to thapsigargin-only treated cells.
Jalewa et al. (Reference Jalewa, Sharma and Hölscher2016) administered Lira or Ex-4 to SH-SY5Y cells with rotenone-induced autophagy impairment. Upon administration of Lira, a significant increase in ATG7 levels compared to non-stressed SH-SY5Y controls was observed (p < 0.01). Treatment with Ex-4 also increased ATG7 expression, although it was not statistically significant. Moreover, Lira increased the expression of ATG3 and LC3A/B compared to non-stressed SH-SY5Y controls. In another experiment, Jalewa et al. (Reference Jalewa, Sharma and Hölscher2016) exposed SH-SY5Y cells to LY294002, a phosphoinositide 3-kinase (PI3K) inhibitor, followed by either Lira or Ex-4 treatment. Lira treatment enhanced ATG3 expression levels (p < 0.0001), whereas Ex-4 did not ameliorate ATG3 expression compared to the stressed SH-SY5Y controls.
Bu et al. (Reference Bu, Liu, Shen, Fan, Yu, Jiang, Tang, Yang, Wu, Zuo, Koprich, Liu, Wu and Wang2021) overexpressed hA53T-α-syn, a mutated form of α-syn, in SH-SY5Y cells as a model of PD. Treatment with Ex-4 was reported to significantly enhance autophagic flux by increasing expression of LAMP1 (p < 0.0001), beclin-1 (p < 0.05), and LC3B-II/LC3B-I ratio (p < 0.0001), as well as by decreasing p62 levels compared to the control group that received a phosphate-buffered saline pre-treatment.
Chang et al. (Reference Chang, Zhang, Hu, Liu and Li2020) treated SH-SY5Y cells with aβ 25–35 as a model of AD, which was reported to significantly reduce beclin-1, LC3-II, ATG7, and p62 levels compared to the control (p < 0.001). Sema treatment appeared to reverse the damaging effects of aβ 25–35 on autophagy by normalising the expression of beclin-1, LC3-II, ATG7, and p62 compared to untreated aβ 25–35 cells (p < 0.001).
Kong et al. (Reference Kong, Wan, Wang, Zhang and Zhang2020) transfected and overexpressed APP695swe plasmid, a mutated human amyloid precursor protein (APP) known to overproduce aβ proteins, in SH-SY5Y cells as a model of AD. Treatment with Lira significantly increased beclin-1 and LC3-II/LC3-I compared to APP695swe/SH-SY5Y controls (p < 0.05). Lira treatment also significantly reduced expression of p62 compared to APP695swe/SH-SY5Y controls (p < 0.05).
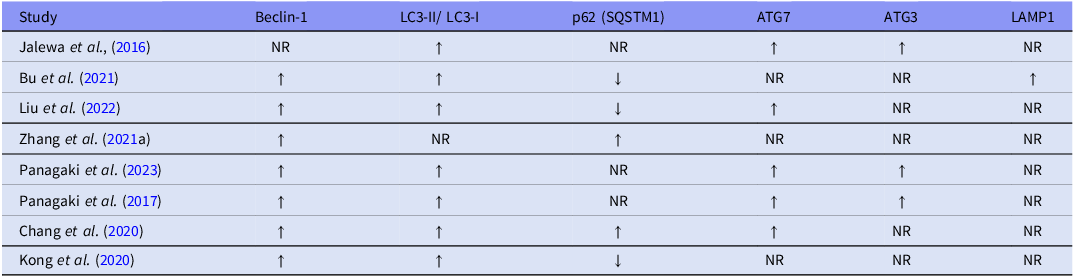
The general influence of the tested GLP-1RAs on autophagy-specific markers, reported in each in vitro study, is summarised in Table 5.
General trends of the tested GLP-1RAs on autophagy-specific markers in each in vitro study

Assessment: ↑, reported increase in expression of autophagy-specific marker by GLP-1RA(s); ↓, reported decrease in expression of autophagy-specific marker by GLP-1RA(s); NR, not reported.
Discussion
This systematic review investigating the effect of GLP-1RAs on molecular and cellular systems salient to autophagy found a consistent and highly replicated restoration and enhancement of autophagy in response to GLP-1RA treatment in cell and animal models of neurological disorders (e.g., AD and PD). Collectively, the in vitro and in vivo studies reported elevated levels of key autophagy-specific markers, such as beclin-1, LC3-II/LC3-I, ATG7, ATG3, and LAMP1, as well as reduced p62 levels, indicating augmented autophagic flux.
Autophagy-specific markers
Among some of its other functions, beclin-1 is a central regulator of autophagy. Beclin-1 serves as a scaffold protein that forms an autophagy-specific complex with multiple other proteins at the ER to create a phagophore, the precursor of an autophagosome, ultimately initiating autophagy (Tran et al., Reference Tran, Fairlie and Lee2021). Deficiency of beclin-1 in response to neurodegenerative-induced stress was consistently observed among the included studies and is supported by other literature (Pickford et al., Reference Pickford, Masliah, Britschgi, Lucin, Narasimhan, Jaeger, Small, Spencer, Rockenstein, Levine and Wyss-Coray2008). Moreover, an elevation of beclin-1 after GLP-1RA administration was a common finding, indicating restored or elevated autophagy.
LC3-I is a cytosolic protein that is conjugated to phosphatidyl-ethanolamine (PE) to form LC3-II. The lipidation of LC3-I to LC3-II enables its insertion into the autophagosome inner and outer membranes, serving as a crucial marker of autophagosomes (Ye et al., Reference Ye, Tyndall, Bui, Bewley, Wang, Hong, Shen, Flanagan, Wang and Tian2023; Runwal et al., Reference Runwal, Stamatakou, Siddiqi, Puri, Zhu and Rubinsztein2019). The included studies reported a notable reduction in the ratio of LC3-II/LC3-I after AD and PD-like stress was induced, indicating impaired autophagy. Following GLP-1RA treatment, LC3-II levels were rescued, implying restored autophagic flux.
ATG7 and ATG3 act as E1-like and E2-like enzymes, respectively. The aforementioned proteins are integral to the process of catalysing the conjugation of LC3-I to PE to form LC3-II after autophagy has been induced (Frudd et al., Reference Frudd, Burgoyne and Burgoyne2018). To our knowledge, Zhang et al. (Reference Zhang, Zhang, Li and Hölscher2018) was the only in vivo study to explore the impact of neurodegenerative stress and GLP-1RA on ATG7, while several other in vitro studies investigated both ATG7 and ATG3 levels. The collective finding was a suppression of both ATG7 and ATG3 levels after disease induction, with an increase of these markers after GLP-1RA treatment.
In addition to its other roles, p62 functions as a receptor that binds specific ubiquitin-tagged proteins and damaged organelles, delivering them to the autophagosome for degradation. Since p62 is degraded along with the cargo, accumulated levels of p62 are thought to be indicative of impaired autophagy (Liu et al., Reference Liu, Ye, Huang, Guo, Xu, Wu, Yang and Liu2016; Kumar et al., Reference Kumar, Mills and Lapierre2022). There were conflicting findings within in vitro and in vivo studies regarding the influence of neurodegenerative-induced stress and GLP-1RAs on p62 levels. Some studies reported elevated p62 in response to disease induction, with GLP-1RAs reducing it. Other articles reported the opposite, where AD and PD lowered p62 levels, and GLP-1RA treatment restored them. Despite this discrepancy, the majority of the included studies, as well as other literature, support the former, where neurodegenerative stress raised p62 levels, with subsequent normalisation of p62 levels following GLP-1RA administration (Runwal et al., Reference Runwal, Stamatakou, Siddiqi, Puri, Zhu and Rubinsztein2019; Bjørkøy et al., Reference Bjørkøy, Lamark, Pankiv, Øvervatn, Brech, Johansen and Klionsky2009).
LAMP1 is a glycosylated transmembrane protein found in lysosomes (Cheng et al., Reference Cheng, Xie, Zhou, Huang, Farfel-Becker and Sheng2018). The literature delineates that disruption of LAMP1 is associated with an accumulation of autophagy vesicles as a result of ineffective clearance inside lysosomes (Eskelinen, Reference Eskelinen2006). Bu et al. (Reference Bu, Liu, Shen, Fan, Yu, Jiang, Tang, Yang, Wu, Zuo, Koprich, Liu, Wu and Wang2021) was the only study to investigate this lysosomal marker and found a notable increase in LAMP1 levels following Ex-4 treatment.
Limitations
While these findings yield valuable insights into GLP-1RAs’ modulation of autophagic processes, there are some limitations to consider, such as the heterogeneity of the included studies. The data reported in this review was collected from preclinical studies that utilised a variety of different cell and animal models, GLP-1RAs (types and dosages), and end points, which made direct comparisons difficult. Moreover, a non-standardised tool was employed to assess the risk of bias for the in vitro studies. None of the studies that we were reviewing were clinical studies, which represents a very significant limitation, and the extent to which the preclinical findings extend to human results is uncertain.
Furthermore, relatively few studies assessed dynamic autophagic flux. As such, deciphering marker levels in association with improved or impaired autophagy can be misconstrued when the timing and context aren’t considered. This limitation could explain the incongruence in p62 levels after disease induction and GLP-1RA administration, as other articles have reported that p62 levels are not always inversely associated with autophagic flux during cellular stress (Liu et al., Reference Liu, Ye, Huang, Guo, Xu, Wu, Yang and Liu2016). Another instance is the interpretation of LC3-II/LC3-I levels, which can also be somewhat problematic, as LC3-II levels measured at a single time point do not necessarily reflect overall autophagic flux (Mizushima and Yoshimori, Reference Mizushima and Yoshimori2007). For instance, elevations in LC3-II could indicate enhanced autophagosome formation through increased conjugation of LC3-I with PE; however, it could also imply ineffective and substandard lysosomal degradation (leading to an accumulation of autophagosomes with LC3-II) (Sarkar et al., Reference Sarkar, Zhao, Aungst, Sabirzhanov, Faden and Lipinski2014; Sharifi et al., Reference Sharifi, Mowers, Drake and Macleod2015). It remains a possibility that the discrepancies and/or inconsistencies that we have identified in the markers of autophagy that we have evaluated may be related to the inadequate assessment of dynamic autophagy processes as much of the datapoints could be considered static, it would be the case that a more comprehensive and dynamic autophagy approach may result in findings that are discrepant from what we have observed.
An overarching limitation which emanates and surrounds the entire literature review that we have conducted relates to the heterogenous findings. It would be accurate to state that our findings represent a disparate assortment of molecular readouts, most of which are in need of replication especially in human subjects. The degree to which the effects we are observing are specific or pseudospecific cannot be ascertained from the methodologic approach we have taken. Notwithstanding this overarching limitation, a theme throughout the results that is relatively consistent is the observation that molecular changes that are observed relate to processes integral to autophagy.
Future directions
Future research could assess the dynamic process of autophagy, as it may provide a clearer and more integrative understanding of what is occurring, as opposed to static measurements. Additionally, future studies should broaden their investigation to examine the influence of GLP-1RAs on other neurodegenerative conditions and mental disorders. Performing clinical trials would also prove valuable, as they could evaluate whether these findings translate to human populations.
Recent research has investigated microbiota-based therapeutics in ameliorating autophagy through modulating endogenous GLP-1 as a potential method to mediate AD and PD symptomology (Bonfili et al., Reference Bonfili, Cecarini, Berardi, Scarpona, Suchodolski, Nasuti, Fiorini, Boarelli, Rossi and Eleuteri2017; Qi et al., Reference Qi, Xie, Chen, Zhang, Ma, Yu, Yu and Wang2025). Further investigation of this approach could reinforce understanding of these relationships. Another avenue of exploration is the influence of dual agonists on autophagy as opposed to single GLP-1RAs, as the dual agonists have also been reported to have a potentially more robust effect on markers of autophagy (Jalewa et al., Reference Jalewa, Sharma, Gengler and Hölscher2017; Panagaki et al., Reference Panagaki, Gengler and Hölscher2018; Cai et al., Reference Cai, Yang, Qiao, Yang, Wang, Wu, Qi and Hölscher2021).
Conclusion
Collectively, this systematic review found that GLP-1RAs enhance autophagy through augmenting and normalising key autophagy-specific markers, accompanied by improvements in AD and PD symptoms. These findings suggest that GLP-1RAs may exert therapeutic effects in neurologic disorders through autophagy along with other highly replicated impacts on plasticity, trophism, and neuroprotection. Future research should expand these findings to further evaluate the potential of repurposing GLP-1RAs for the prevention and treatment of other neurologic and mental disorders.
Acknowledgements
None.
Author contributions
Maria-Christina Sioufi, Roger S. McIntyre, and Christine E. Dri were responsible for the substantial contributions to the conception or design of the work. Maria-Christina Sioufi and Isabela Heroiu were responsible for the acquisition, analysis, or interpretation of data for the work. Maria-Christina Sioufi was responsible for drafting the work. Maria-Christina Sioufi, Roger S. McIntyre, Sabrina Wong, Kayla M. Teopiz, Christine E. Dri, Gia Han Le, Yang Jing Zheng, Taeho Greg Rhee, Heidi Ka Ying Lo, and Hernan F. Guillen-Burgos were responsible for reviewing the work critically for important intellectual content. All authors were responsible for the final approval of the version to be published.
Disclosures
Dr. Roger S. McIntyre has received research grant support from CIHR/GACD/National Natural Science Foundation of China (NSFC) and the Milken Institute; speaker/consultation fees from Lundbeck, Janssen, Johnson & Johnson, Alkermes, Neumora Therapeutics, Boehringer Ingelheim, Bristol Myers Squibb, Sage, Mitsubishi Tanabe, Purdue, Pfizer, Otsuka, Takeda, MindMed, Neurocrine, Neurawell, Supernus, Bausch Health, Axsome, Novo Nordisk, Kris, Sanofi, Eisai, Intra-Cellular, NewBridge Pharmaceuticals, Viatris, Abbvie, and Atai Life Sciences.
Dr. Taeho Greg Rhee was supported in part by the National Institute on Aging (#R21AG070666; R21AG078972; R01AG088647), National Institute of Mental Health (#R01MH131528), National Institute on Drug Abuse (#R21DA057540), and Health Resources and Services Administration (#R42MC53154-01-00). Dr. Rhee serves as a review committee member for National Institutes of Health (NIH), Patient-Centered Outcomes Research Institute (PCORI) and Substance Abuse and Mental Health Services Administration (SAMHSA) and has received honoraria payments from NIH, PCORI and SAMHSA. Dr. Rhee has also served as a stakeholder/consultant for PCORI and received consulting fees from PCORI. Dr. Rhee serves as an advisory committee member for International Alliance of Mental Health Research Funders (IAMHRF).
Dr. Hernan F. Guillen-Burgos has received research grant support from the Ministry of Science, Technology, and Innovation (MinCiencias) in Colombia, UKRI in the United Kingdom; and speaker fees from Roche, Pfizer, Abbott, GSK, and Synergy R&D.
Kayla M. Teopiz has received fees from Braxia Scientific Corp.