Premenstrual dysphoric disorder (PMDD) is a severe affective disorder, characterised by cyclic affective and physical symptoms that emerge during the luteal phase of the menstrual cycle and remit following menstruation. Reference Reilly, Patel, Unachukwu, Knox, Wilson and Craig1 While the exact pathophysiology remains unclear, accumulating evidence suggests dysregulation of the hypothalamic–pituitary–adrenal (HPA) axis Reference Hantsoo and Epperson2 and altered serotonergic functioning Reference Sacher, Zsido, Barth, Zientek, Rullmann and Luthardt3 as the key mechanisms underlying PMDD. Given that the HPA axis plays a crucial role in coordinating stress responses Reference Herman, McKlveen, Ghosal, Kopp, Wulsin and Makinson4 and interacts closely with the serotonergic system, Reference Lowry5 disruptions in these systems may contribute to the heightened affective and physiological sensitivity observed in PMDD. Reference Gordon, Girdler, Meltzer-Brody, Stika, Thurston and Clark6
One widely studied marker of HPA axis activity is the cortisol awakening response (CAR), a rapid rise in cortisol levels occurring within the first 30–45 min after waking. Reference Clow, Thorn, Evans and Hucklebridge7 CAR is influenced by both endogenous and exogenous factors, including stress, Reference Giglberger, Peter, Kraus, Kreuzpointner, Zänkert and Henze8 vulnerability to affective disorders Reference van Santen, Vreeburg, Van der Does, Spinhoven, Zitman and Penninx9 and neuroendocrine regulation. Reference Clow, Hucklebridge, Stalder, Evans and Thorn10 While females display an increased CAR compared with males, Reference Pruessner, Wolf, Hellhammer, Buske-Kirschbaum, von Auer and Jobst11 no CAR differences have yet been reported between the follicular and luteal phases in healthy, naturally cycling females, except for a blunted CAR during menses Reference Ozgocer, Ucar and Yildiz12 and an increased CAR during ovulation. Reference Wolfram, Bellingrath and Kudielka13 However, in the only study directly comparing patients with PMDD and healthy controls, a delayed CAR peak and flattened diurnal cortisol slope were observed in patients with PMDD, Reference Beddig, Reinhard and Kuehner14 suggesting dysregulated HPA axis activity in this population.
One possible mechanism underlying this dysregulation is serotonergic dysfunction, which has long been implicated in PMDD. Reference Parry15 Given that selective serotonin reuptake inhibitors exert their therapeutic effects by binding to the serotonin transporter (5-HTT), and pose an effective pharmacological treatment for PMDD, Reference Rapkin and Lewis16 alterations in 5-HTT expression or function may contribute to the abnormal CAR observed in PMDD. However, no study has yet examined the interaction between CAR and 5-HTT in PMDD. This represents a critical gap in understanding how stress regulation and serotonin function interact in this disorder.
The present study explores the interactions among CAR, 5-HTT binding levels and depressive symptoms. Specifically, we aim to explore whether patients with PMDD exhibit a dysregulated CAR, differing from controls depending on the menstrual cycle phase. Due to the role of oestradiol in stress Reference Jacobs, Holsen, Lancaster, Makris, Whitfield-Gabrieli and Remington17 and emotion regulation, Reference Rehbein, Kogler, Hornung, Morawetz, Bayer and Krylova18 the periovulatory phase, characterised by an oestradiol peak, is contrasted with the premenstrual phase, characterised by declining oestradiol levels and PMDD symptoms. We further explore the association between 5-HTT binding levels and CAR, given that altered serotonergic function could contribute to HPA axis dysregulation in PMDD. Finally, we explore the relationship between CAR and affective symptoms that may vary across the cycle, possibly reflecting heightened sensitivity to hormonal fluctuations in PMDD.
By integrating HPA axis function, serotonergic regulation and menstrual cycle-dependent mood changes, this study provides a novel perspective on the neurobiological underpinnings of PMDD. Understanding these mechanisms may inform individualised treatment strategies, particularly regarding the serotonergic modulation of stress responses in PMDD.
Method
Participants
For this longitudinal study, participants were either healthy (n = 29) or diagnosed with PMDD (n = 30). Participants were all female, between 19 and 34 years of age and reported having regular menstrual cycles (length of 23–35 days in the past 6 months). Participants had not used hormonal contraceptives within 1 year of study participation, were not pregnant, postpartum (within 1 year after delivery) or (peri)menopausal and had not recently undergone abortion (within 1 year). Eligible participants were non-smokers in good physical health with no lifetime history of, or comorbid, Axis I or Axis II disorder Reference Beesdo-Baum, Zaudig and Wittchen19,Reference Beesdo-Baum, Zaudig and Wittchen20 (for controls and patients with PMDD, respectively); no current or lifetime antidepressant use; no use of prescription medications or herbal supplements within the past 2 months; and no use of over-the-counter medications within the past 2 weeks. For the PMDD group, the same inclusion criteria applied, but participants additionally reported a history of premenstrual symptoms (within the past 6 months). PMDD diagnosis was based on the Structured Clinical Interview for DSM-5 (SCID-5) Reference Beesdo-Baum, Zaudig and Wittchen19 performed by a licensed psychiatrist (J.S.), and participants further met the PMDD criteria of the Premenstrual Symptoms Screening Tool (PSST). Reference Steiner, Macdougall and Brown21 The final group allocation was assessed by agreement between two independent researchers.
For the parent study, Reference Sacher, Zsido, Barth, Zientek, Rullmann and Luthardt3 which assessed 5-HTT binding across the menstrual cycle in PMDD and health, an a priori power analysis performed with G*Power 2019 for Windows (Heinrich Heine Universität Düsseldorf, Düsseldorf, Germany; see https://www.psychologie.hhu.de/arbeitsgruppen/allgemeine-psychologie-und-arbeitspsychologie/gpower) yielded a required sample of N = 52 to detect a moderate effect size (0.4), at a power of 0.8 and an α-level of 0.05 (https://osf.io/fvghx). The current exploratory study was pre-registered as an amendment of this parent study (https://osf.io/ygfzs). Written informed consent was obtained from all participants. The authors assert that all procedures contributing to this work comply with the ethical standards of the relevant national and institutional committees on human experimentation, and with the Helsinki Declaration of 1975 as revised in 2013. All procedures involving human participants were approved by the Ethics Committee of the Faculty of Medicine, University of Leipzig (approval no. 246-2009-ff).
Procedure
Participants underwent two assessments: the first within 24 h of their estimated ovulation (periovulatory phase), the second 13–14 days after ovulation/within 3 days of onset of the next menses (premenstrual phase). The order of testing sessions was randomised between participants. Testing sessions included fasting blood samples for endocrine assessment, a series of questionnaires, a neuropsychological test battery and [11C]DASB positron emission tomography (PET) and magnetic resonance imaging (MRI) scans, matched for season and time of day (Fig. 1(a)). Participants were instructed to self-collect saliva samples in the morning of, and across, each testing day (Fig. 1(b)).
(a) Study design. All participants were tested during the periovulatory and the premenstrual phase. Menstrual cycle phases were determined using cycle tracking, folliculometries and urine-based ovulation tests. Additionally, blood samples were drawn at each testing day to verify cycle phases by ovarian hormone levels. Participants performed positron emission tomography (PET) and magnetic resonance imaging (MRI) scans, undertook a neurocognitive test battery, filled in a series of questionnaires and collected saliva samples at each testing day. (b) Saliva sampling. Participants self-collected eight saliva samples (S1–8) across each testing day. The cortisol peak refers to the maximum cortisol concentration within the second and third saliva sample. E2, estradiol; P4, progesterone.

Menstrual cycle monitoring
For accurate determination of the two cycle phases, participants were asked to track their basal body temperature, possible bleeding and disruptive factors for at least one menstrual cycle before study participation, using a smartphone app (myNFP Web Classic v2.0.0., myNFP GmbH, Tübingen, Germany; see https://www.mynfp.de/). Folliculometries were performed by a gynaecologist to determine the precise time of ovulation. Participants used urine ovulation tests (Diagnostik Nord GmbH hLK-K20 hLH Kassettentest) to detect the luteinising hormone surge preceding ovulation. Additionally, fasting blood serum samples were collected using S-Monovettes® (Sarstedt, Germany) to measure ovarian hormones at each testing session. Oestradiol and progesterone concentrations were determined using liquid chromatography–tandem mass spectrometry (LC–MS/MS). Follicle-stimulating hormone (FSH) and luteinising hormone concentrations were determined using electrochemiluminescence immunoassay (ECLIA, Roche).
Cortisol sampling
On each day of testing, participants self-collected cortisol saliva samples with Salivettes® (Sarstedt, Germany) at home immediately following awakening, as well as 30 and 60 min post-awakening. Participants were instructed not to brush their teeth or eat or drink anything other than water within 10 min prior to saliva sampling. Additional saliva samples were provided at 09.00, 12.00, 15.00, 18.00, and 21.00 h. Participants were instructed to store saliva samples in the refrigerator until their delivery to the laboratory, where they were then stored at −80°C. Cortisol saliva samples were analysed using a time-resolved fluorescence immunoassay Reference Dressendörfer, Kirschbaum, Rohde, Stahl and Strasburger22 at the biochemical laboratory of the University of Trier, with intra- and inter-assay coefficients of variation of 4.0−6.7 and 7.1−9.0%, respectively.
To quantify CAR, multiple indices have been proposed. Reference Fekedulegn, Andrew, Burchfiel, Violanti, Hartley and Charles23 Informed by CAR research showing delayed cortisol peaks in patients with PMDD, Reference Beddig, Reinhard and Kuehner14 here we determined the cortisol peak (maximal cortisol concentration) within the second and third saliva samples (30 and 60 min post-awakening) per cycle phase. Other pre-registered outcomes (i.e. area under the curve with respect to increase, delta) have been included as supplementary analyses (Supplementary Tables 1 and 2 available at https://doi.org/10.1192/bjp.2025.10432).
Mood assessment
In all participants, the severity of depressive symptoms experienced within the past week was assessed with the Hamilton Depression Rating Scale (HAM-D, 17 items). Reference Hamilton24 Perceived stress within the past 4 weeks was derived from the Perceived Stress Scale (PSS, 10 items). Reference Cohen, Kamarck and Mermelstein25
Neuroimaging
We applied the radiotracer [11C]-3-amino-4-(2-dimethylaminomethyl-phenylsulfanyl)-benzonitrile ([11C]DASB) Reference Wilson, Ginovart, Schmidt, Meyer, Threlkeld and Houle26 to assess serotonin transporter binding using PET in vivo. Primary outcomes were midbrain Reference Sacher, Zsido, Barth, Zientek, Rullmann and Luthardt3 and prefrontal Reference Frokjaer, Erritzoe, Holst, Jensen, Rasmussen and Fisher27 5-HTT non-displaceable binding potentials (BPND), using the cerebellum as reference region.
Data preparation
Hormone values below the detection threshold were replaced by their respective minimum value. According to expert guidelines, Reference Stalder, Kirschbaum, Kudielka, Adam, Pruessner and Wust28 raw cortisol concentrations were square-root transformed to account for positively skewed distribution, Reference Engert, Efanov, Dedovic, Dagher and Pruessner29 and outliers exceeding mean ± three times the standard deviation were winsorised to the respective maximum or minimum values, Reference Engert, Hoehne and Singer30 before determining cortisol peaks.
Statistical analysis
One linear mixed-effects model was built on cortisol peak, including group (control, PMDD), phase (periovulatory, premenstrual), the interaction between group and phase, and depressive symptoms (HAM-D scores) as fixed effects. A random intercept per participant accounted for inter-individual differences. To control for baseline cortisol concentrations, we added awakening cortisol concentration as a fixed effect, changing the cortisol peak into a proxy for cortisol increase. Similarly, the influence of covariates (oestradiol, progesterone, age, body mass index, PSS scores, time of awakening, hours of sleep, sleep quality) was checked.
Exploratively, cortisol concentrations at each time point (S1–8) and changes in CAR cortisol concentrations (awakening to +30 min, +30 min to +60 min) were assessed using multiple linear mixed-effects models, with the model equation on cortisol peak accounting for awakening cortisol. In addition to the pre-registered analyses, the relationships between cortisol peak and midbrain and prefrontal 5-HTT BPND, as well as HAM-D scores, were assessed using a series of Spearman rank correlations. Lastly, demographics, questionnaire data and ovarian hormone concentrations were compared between groups and phases using independent t-tests and mixed analysis of variance (ANOVA).
Statistical analyses were performed with R version 4.5.0 for Linux (R Core Team, Vienna, Austria; see https://www.R-project.org). Statistical significance was assessed at a critical p-value of 0.05 and, in the case of linear mixed-effects model estimates, verified using model comparisons by means of chi-square (X 2 ) tests and Bayes factors approximated by Bayesian information criterion (BIC), comparing models including and excluding the estimate of interest, if possible. If a significant interaction was observed, post hoc tests were performed to explore simple effects. Bonferroni correction was applied to account for multiple comparisons, if necessary. D (fixed-effect estimate/square root of the sum of variances of random effects) served as an effect size for linear mixed-effects model estimates, and R 2 for Spearman rank correlations.
Results
Sample characteristics and demographics
Sample characteristics and demographics are displayed in Table 1. Patients with PMDD were significantly older than controls, with no group differences in body mass index. Premenstrual PSST scores were significantly higher in patients with PMDD compared with healthy controls. Oestradiol levels were significantly higher in the periovulatory phase compared with the premenstrual (F = 33.39, p < 0.001), while progesterone levels significantly increased across phases (F = 30.68, p < 0.001), with no difference in absolute ovarian hormone levels between groups. Patients with PMDD showed overall higher depressive symptoms (HAM-D scores) than controls, and an increase from the periovulatory phase to the premenstrual. Participants reported higher perceived stress (PSS scores) in the premenstrual phase compared with the periovulatory (F = 11.14, p < 0.01), with no difference between patients with PMDD and controls. No group or cycle phase differences were found in awakening cortisol concentrations.
Sample characteristics and demographics

Sample characteristics and demographic variables between groups and menstrual cycle phases provided as mean (standard deviation). Statistics refer to group differences or group and cycle phase interactions. BMI, body mass index; CTL, control; E2, oestradiol; HAM-D, Hamilton Depression Rating Scale; P4, progesterone; PMDD, premenstrual dysphoric disorder; PSS, Perceived Stress Scale; PSST, Premenstrual Symptom Screening Tool.
a. Bonferroni adjusted p-value controlling for three group comparisons.
Missing data
Two participants sampled salivary cortisol during only one menstrual cycle phase. Additionally, three cortisol samples were missing at awakening, two at 60 min post-awakening and 12 at later sampling time points. Missing cortisol values were addressed using suitable linear mixed-effects models. One oestradiol and three progesterone concentrations below the detection threshold were replaced by the respective minimal detection values. PSS scores were missing for 10 participants (n = 4 PMDD), while reports of hours and quality of sleep were incomplete for 21 participants (n = 10 PMDD). For 6 awakening cortisol samples (n = 3 PMDD), the time of awakening was unknown.
Cortisol peak
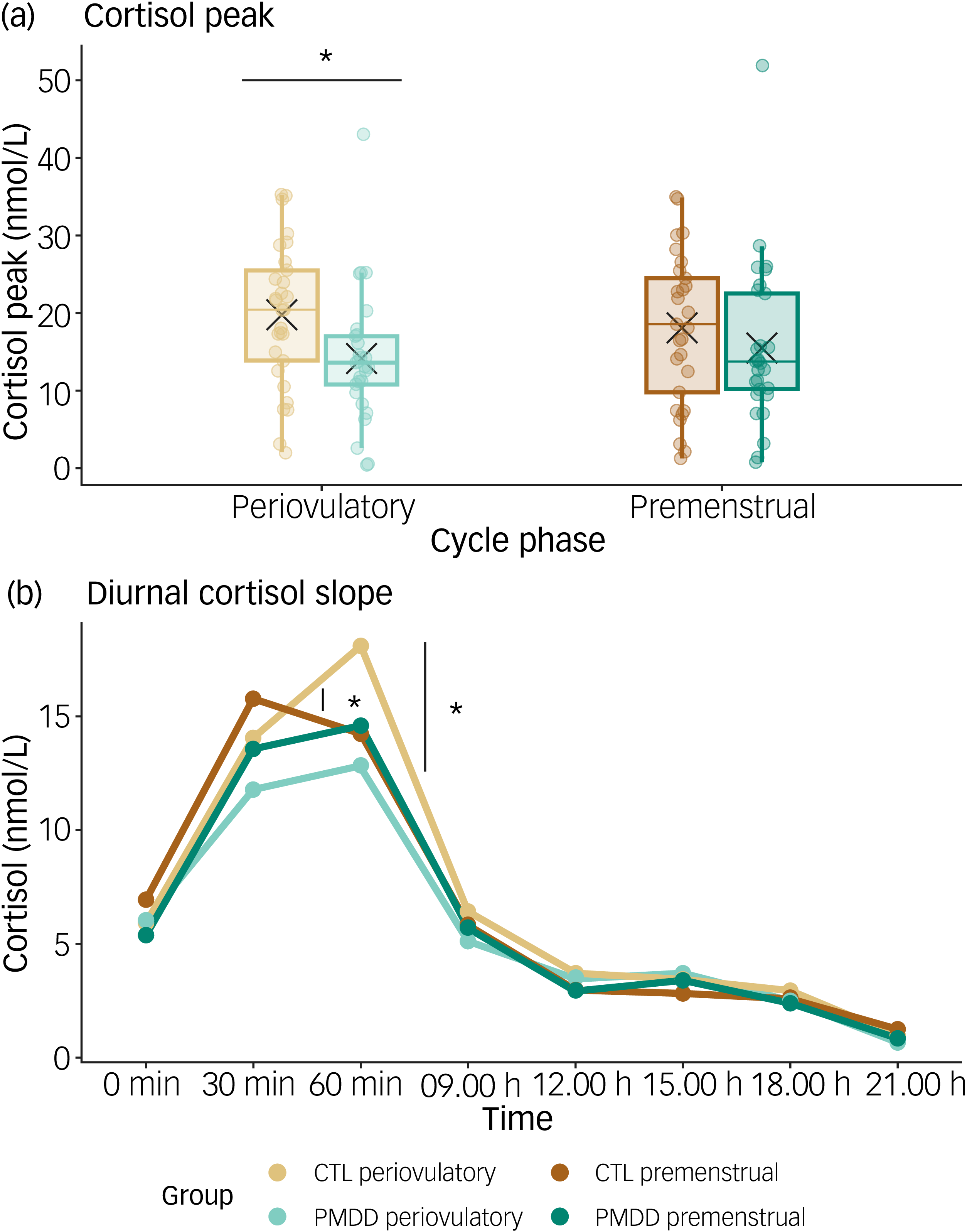
We found a marginally significant interaction between group and phase on cortisol peak (estimate = 0.78, t = 1.96, p = 0.05, d = 0.62; Fig. 2(a)). One model including the interaction improved its fit (X 2 = 3.92, p = 0.05), but a Bayes factor of 0.66 showed anecdotal evidence in favour of the model, excluding the interaction. Disentangling simple effects showed that the interaction was driven by lower cortisol peaks in patients with PMDD compared with controls, specifically during the periovulatory phase.
(a) Cortisol peak concentrations (nmol/L) during the menstrual cycle (periovulatory vs premenstrual) in both groups (patients with premenstrual dysphoric disorder (PMDD) vs healthy controls). Raw cortisol peak concentrations are plotted to facilitate visualisation. Thin horizontal lines indicate median concentrations and crosses indicate mean concentrations per group and phase. Dots represent individuals’ peak cortisol concentrations. Asterisks represent significant levels of p ≤ 0.05. (b) Diurnal cortisol slopes per group and cycle phase. Raw mean cortisol concentrations per time point are plotted to facilitate visualisation. Asterisks represent significant levels of p ≤ 0.05. CTL, control.

Correcting for awakening cortisol concentrations strengthened the interaction (estimate = 0.90, t = 2.30, p = 0.02, d = 0.77), with a Bayes factor of 8.39 × 103, supporting this more complex model. Additionally, and as expected, a significant positive main effect of awakening cortisol on cortisol peak concentration was found (estimate = 0.55, t = 4.10, p < 0.01, d = 0.47). When excluding observations from this complex model for which participants reported a delay of ≥10 min in saliva sampling, the interaction was further strengthened (estimate = 0.99, t = 2.23, p = 0.03, d = 0.84). Correcting for self-reported perceived stress levels, time of awakening and self-reported hours and quality of sleep strengthened the interaction further (see Supplementary Table 3 for missing data). The addition of body mass index, age and oestradiol and progesterone concentrations as covariates did not improve the model’s fit.
In sum, patients with PMDD showed attenuated cortisol peaks compared with controls during the periovulatory phase. Controlling for awakening cortisol concentration strengthened this interaction. However, the interaction did not survive correction for testing of three CAR indices.
Cortisol concentration across time
Groups did not differ in regard to their cortisol concentrations at awakening, or at 30 min post-awakening, with similar concentrations across cycle phases. Mirroring the effect found for cortisol peaks, a significant interaction between group and phase was detected at 60 min post-awakening (estimate = 1.14, t = 2.58, p = 0.01, d = 0.87), not surviving correction for comparison of eight cortisol sampling time points. Disentangling simple effects revealed significantly lower cortisol concentrations at 60 min post-awakening in patients with PMDD compared with controls during the periovulatory phase. No group or cycle phase differences were found for later cortisol sampling points (Fig. 2(b)).
Focusing on cortisol change, both groups showed similar increases in cortisol from awakening to 30 min post-awakening across cycle phases. For cortisol change from 30 to 60 min post-awakening, we found a marginally significant interaction between group and phase (estimate = 0.77, t = 1.97, p = 0.05, d = 0.79), not surviving correction for testing of two cortisol changes. Disentangling simple effects revealed higher cortisol increases from 30 to 60 min post-awakening during the periovulatory phase compared with the premenstrual in controls, while patients with PMDD showed similar cortisol increases across cycle phases (Fig. 2(b)).
In sum, explorative analyses propose lower cortisol concentrations at 60 min post-awakening in patients with PMDD compared with controls during the periovulatory phase. Healthy controls displayed a tendency for higher cortisol increases from 30 to 60 min during the periovulatory phase compared with the premenstrual.
Cortisol peak and 5-HTT BPND
Cortisol peaks correlated significantly with midbrain 5-HTT BPND (r = −0.20, p = 0.03, R 2 = 0.04). Splitting by cycle phase revealed that this association was present only in the premenstrual phase (r = −0.34, p < 0.01, R 2 = 0.12) and not in the periovulatory phase (r = −0.04, p = 0.78). In detail, increasing cortisol peaks were linked to decreasing midbrain 5-HTT BPND in the premenstrual cycle phase (Fig. 3(a)), surviving correction for testing binding potentials in two brain regions across two cycle phases, but not for testing three CAR indices. No association was found between cortisol peaks and prefrontal 5-HTT BPND. No associations between 5-HTT binding and other CAR indices were found.
Cortisol peak concentrations (nmol/L) against (a) midbrain 5-HTT BPND per cycle phase, and (b) HAM-D scores per cycle phase. Raw cortisol peak concentrations are plotted to facilitate visualisation. Asterisks indicate significance levels of p < 0.05. Differently shaded dots represent individual data points. Differently shaded lines indicate a significant correlation, with shaded grey areas representing a 95% confidence interval. HAM-D, Hamilton Depression Rating Scale; 5-HTT BPND, non-displaceable serotonin transporter binding potentials.

Cortisol peak and depressive symptoms
Cortisol peaks correlated significantly with HAM-D scores (r = −0.26, p < 0.01, R 2 = 0.07). Splitting by cycle phase revealed that this association was present only in the premenstrual phase (r = −0.30, p = 0.02, R 2 = 0.09) and not in the periovulatory phase (r = −0.21, p = 0.11). In detail, decreasing cortisol peaks were linked to increasing depressive symptoms in the premenstrual phase (Fig. 3(b)), surviving correction for testing this correlation in two cycle phases, but not for testing three CAR indices. No associations between depressive symptoms and other CAR indices were found.
Discussion
Patients with PMDD exhibited attenuated cortisol peaks during the periovulatory phase compared with healthy controls. Additionally, cortisol peaks were negatively associated with midbrain serotonin transporter binding and depressive symptoms during the premenstrual phase. Study findings suggest an altered stress system regulation in PMDD, and highlight the need for critical reflection on CAR guidelines when studying the menstrual cycle and related mood disorders.
Comparison with previous studies
Our cortisol peak finding in PMDD is partially consistent with previous work Reference Beddig, Reinhard and Kuehner14 showing delayed cortisol peaks in patients with PMDD compared with controls across the menstrual cycle. Studies on premenstrual syndrome (PMS), a condition of psychological and somatic symptoms before menses onset below clinical-level thresholds in severity, also report a flat CAR during menses and attenuated CAR at 45 and 60 min post-awakening during the mid-follicular and premenstrual phases. Reference Ozgocer, Ucar and Yildiz12,Reference Hou, Huang and Zhou31 Given the limited research on CAR in PMDD across the menstrual cycle, larger longitudinal studies employing dense-sampling methods and assessments of individualised, multidimensional stress responses are warranted to develop a systematic understanding of CAR signature patterns in PMDD.
Our cortisol peak finding in controls partially aligns with a previous study Reference Wolfram, Bellingrath and Kudielka13 reporting a significantly higher increase in cortisol from awakening to peak levels during ovulation, compared with menses and the follicular and luteal phases. However, most studies do not report differences in CAR across the menstrual cycle. Reference Ozgocer, Ucar and Yildiz32–Reference Kirschbaum, Kudielka, Gaab, Schommer and Hellhammer34 Discrepancies may arise due to study designs that did not confirm ovulation through objective tests Reference Ozgocer, Ucar and Yildiz32 or oestradiol peaks, Reference Haase, Vehlen, Strojny and Domes33 or that omitted the periovulatory phase. Reference Kirschbaum, Kudielka, Gaab, Schommer and Hellhammer34 In contrast, our study included the periovulatory phase, validated through folliculometries, luteinising hormone ovulation tests, serum ovarian hormone levels and digital menstrual cycle tracking, ensuring accurate cycle phase determination.
Research highlights robust gender differences in the HPA axis response to external stressors, including psychosocial stress, Reference Kirschbaum, Kudielka, Gaab, Schommer and Hellhammer34 as well as variations in basal HPA axis activity Reference Klusmann, Schulze, Engel, Bucklein, Daehn and Lozza-Fiacco35 and reactivity, Reference Klusmann, Luecking, Engel, Blecker, Knaevelsrud and Schumacher36 across the menstrual cycle. However, the mechanism underlying the interaction of ovarian hormone fluctuations and HPA axis response remains unclear. The CAR also exhibits gender differences Reference Pruessner, Wolf, Hellhammer, Buske-Kirschbaum, von Auer and Jobst11 and menstrual cycle effects, although for the latter the current evidence is mixed. While some studies link CAR indices to oestradiol and progesterone, Reference Ozgocer, Ucar and Yildiz12,Reference Ozgocer, Ucar and Yildiz32 we found no such association between cortisol peak and ovarian hormone concentrations. Moreover, group differences in cortisol peaks in this study cannot be attributed to differences in absolute oestradiol or progesterone levels, aligning with previous work showing no absolute difference, but rather an altered sensitivity to normal hormonal fluctuations, in patients with PMDD. Reference Schmidt, Nieman, Danaceau, Adams and Rubinow37
In the current study, both controls and patients with PMDD exhibited relatively delayed cortisol peak concentrations. From all cortisol peaks (n = 116), 20.69% occurred at 30 min and 29.31% at 60 min post-awakening in patients with PMDD, compared with 23.28 and 26.72%, respectively, in controls, suggesting a tendency for delayed cortisol peaks in PMDD. Furthermore, excluding 11 observations for which participants reported a delay of ≥10 min in CAR saliva sampling strengthened the interaction effect. Non-responses in CAR (≤2.5 nmol/L cortisol increase) occurred in 24.56% of observations, consistent with other studies, Reference Wust, Wolf, Hellhammer, Federenko, Schommer and Kirschbaum38 indicating that non-compliance and non-responses cannot explain the observed delayed cortisol peaks.
The prominence of delayed peaks in this study may have resulted from the absence of systematic assessment of cortisol peak timing across the menstrual cycle. Previous research has shown that females tend to peak later than males, at around 45 min. Reference Pruessner, Wolf, Hellhammer, Buske-Kirschbaum, von Auer and Jobst11,Reference Wust, Wolf, Hellhammer, Federenko, Schommer and Kirschbaum38 Patients with PMDD have shown delayed cortisol peaks at 45 compared to 30 min in controls. Reference Beddig, Reinhard and Kuehner14 Only a few studies have observed peaks as late as 60 min post-awakening across the menstrual cycle, Reference Ozgocer, Ucar and Yildiz32 and specifically for the follicular and luteal phases. Reference Benz, Meier, Mankin, Unternaehrer and Pruessner39 Our findings support the notion of delayed cortisol peaks at 60 min post-awakening in patients with PMDD across the menstrual cycle, as well as in naturally cycling females during ovulation. This study highlights the need for critical reflection on, and extending the time window of, saliva sampling time points for CAR research across the menstrual cycle. The CAR in patients with PMDD and healthy females remains under-explored.
Altered stress markers, particularly a flattened diurnal cortisol slope, have been linked to poorer health outcomes. Reference Adam, Quinn, Tavernier, McQuillan, Dahlke and Gilbert40 Similarly, alterations in CAR have been reported for various disorders. For example, while patients with post-traumatic stress disorder exhibit an attenuated CAR, Reference Wessa, Rohleder, Kirschbaum and Flor41 the direction of findings varies for clinical depression. Reference Dedovic and Ngiam42 To reconcile these mixed results, it has been suggested that CAR flexibility, rather than magnitude, may be a more meaningful measure. Reference Mikolajczak, Quoidbach, Vanootighem, Lambert, Lahaye and Fillee43 For example, participants with high self-reported well-being showed greater variability in CAR indices in line with external demands (i.e. weekends versus weekdays), while this variability was less in participants with low self-reported well-being. Reference Mikolajczak, Quoidbach, Vanootighem, Lambert, Lahaye and Fillee43 Applying this to our findings suggests that patients with PMDD, and with more severe depressive symptoms, may lack the flexibility to adapt their cortisol peak across the menstrual cycle, a capacity that healthy females seem to possess. The menstrual cycle, representing a monthly reoccurring phenomenon affecting most females during their reproductive years, imposes physiological demands that potentially require a flexible upregulation of CAR. The concept of CAR flexibility warrants further investigation, Reference Stalder, Oster, Abelson, Huthsteiner, Klucken and Clow44 especially across the menstrual cycle, and may have significant clinical relevance for menstrual cycle-related disorders such as PMDD.
Reimold et al Reference Reimold, Knobel, Rapp, Batra, Wiedemann and Ströhle45 were the first to report an inverse relationship between thalamic 5-HTT binding and increased cortisol response to a combined dexamethasone–corticotrophin test, interpreted as being driven by lower thalamic 5-HTT availabilities in their clinical sample (unmedicated patients with obsessive–compulsive disorder and major depressive disorder) compared with controls. Pioneering work by Frokjaer et al Reference Frokjaer, Erritzoe, Holst, Jensen, Rasmussen and Fisher27,Reference Frokjaer, Erritzoe, Holst, Madsen, Fisher and Madsen46 on the relationship between CAR and the serotonergic system initially showed a positive association between the area under the curve and prefrontal 5-HTT BPND in healthy participants (N = 32, 7 females, 19.7–81.7 years), Reference Frokjaer, Erritzoe, Holst, Jensen, Rasmussen and Fisher27 and in abstinent 3,4-Methylenedioxymethamphetamine (MDMA) users (N = 18 users, 2 females, 20.1–33.6 years). Reference Frokjaer, Erritzoe, Holst, Madsen, Fisher and Madsen46 In a larger follow-up study, this finding could not be replicated (N = 90, 67 females, 18.4–47.7 years). Reference Conradi, Svensson, Larsen, Frokjaer and Yamasue47 No study has yet longitudinally investigated the relationship between CAR indices and 5-HTT availability across the menstrual cycle in both healthy females and patients with PMDD.
In the current study, cortisol peaks were negatively correlated with midbrain 5-HTT BPND during the premenstrual phase, suggesting a facilitatory mechanism between the midbrain serotonergic and stress systems towards the end of the menstrual cycle. We also tested this relationship for the prefrontal cortex given the findings by Frokjaer et al, Reference Frokjaer, Erritzoe, Holst, Jensen, Rasmussen and Fisher27 who emphasised this area as their main region of interest due to the expression of glucocorticoid and mineralocorticoid receptors. The availability of 5-HTT, however, is low in prefrontal cortical areas, Reference Beliveau, Ganz, Feng, Ozenne, Hojgaard and Fisher48 which may partly explain why their results did not replicate in a larger sample. Reference Conradi, Svensson, Larsen, Frokjaer and Yamasue47 The studied samples also differed in demographics, because one contained mostly men of a wider age range Reference Frokjaer, Erritzoe, Holst, Jensen, Rasmussen and Fisher27 while the other contained primarily young women. Reference Conradi, Svensson, Larsen, Frokjaer and Yamasue47 Since we observed no association between prefrontal 5-HTT BPND with cortisol peaks, our results align with the more recent findings. Reference Conradi, Svensson, Larsen, Frokjaer and Yamasue47 Understanding and characterising the relationship between the serotonergic and stress systems in health, but also in stress-related mood disorders, is of great relevance.
Underlying biological mechanisms
The mechanisms linking cortisol dysregulation and depressive symptoms in PMDD may involve a complex interplay between the serotonergic system and HPA axis functioning. Our results, showing a negative correlation between cortisol peaks and depressive symptoms in all participants during the premenstrual phase, align with previous findings linking different CAR indices to mood regulation. Reference Hou, Huang and Zhou31,Reference Ozgocer, Ucar and Yildiz32 Increased 5-HTT binding potential during the premenstrual phase, previously demonstrated in this PMDD sample, Reference Sacher, Zsido, Barth, Zientek, Rullmann and Luthardt3 may reflect a compensatory mechanism stabilising cortisol responses to stress. Genetic factors, such as serotonin transporter genotype, Reference Wust, Kumsta, Treutlein, Frank, Entringer and Schulze49 and epigenetic factors, such as methylation of the serotonin transporter gene, Reference Alexander, Wankerl, Hennig, Miller, Zankert and Steudte-Schmiedgen50 have also been linked to cortisol responses, indicating that serotonergic regulation at the gene level may contribute to HPA axis dysfunction.
Strengths and limitations
The present study is the first to systematically investigate CAR across the menstrual cycle in healthy females and patients with PMDD, exploring its association with 5-HTT availability and depressive symptom severity. Unlike previous research, we employed a longitudinal design with robust measures for menstrual cycle tracking. This thorough approach follows key guidelines for studying the menstrual cycle, Reference Schmalenberger, Tauseef, Barone, Owens, Lieberman and Jarczok51 enhancing the accuracy and reliability of our findings within this complex hormonal framework. At the same time, several limitations should be noted. First, although our design was rigorous, it was conceptualised before publication of the CAR guidelines, Reference Stalder, Kirschbaum, Kudielka, Adam, Pruessner and Wust28 which recommended timing the third CAR measurement at +45 min to best capture the peak. While this may have reduced temporal precision and potentially resulted in smaller effect sizes, our decision to push the third sample to 60 min was, in particular, motivated by previous research related to the menstrual cycle. Reference Wolfram, Bellingrath and Kudielka13 Recent evidence suggests that this sampling protocol can still yield reliable CAR estimates compared with more detailed designs. Reference Nasser, Ozenne, Hogsted, Jensen and Frokjaer52 Second, because we focused on cortisol peaks, it is important to acknowledge that the peak does not represent a fully dynamic CAR index. Although we controlled for awakening cortisol levels, the peak itself reflects a stationary measure. Again, this focus is in line with previous findings of delayed cortisol peaks in relevant populations. Reference Beddig, Reinhard and Kuehner14 Third, our study included a single CAR assessment per menstrual cycle phase, although multiple CAR measurements performed on consecutive days are more reliable. Reference Hellhammer, Fries, Schweisthal, Schlotz, Stone and Hagemann53 However, this single measurement was suitable for exploring CAR during brief time windows, such as periovulatory and premenstrual phases, and the timely association of same-day CAR and 5-HTT measures. To counteract the potential reduced reliability of a single CAR measurement, participants were called at each testing day, most salivary samples were taken under supervision and self-reported delays were statistically controlled for. Fourth, the diagnosis of PMDD was based on the Structured Clinical Interview for DSM-5 Reference Beesdo-Baum, Zaudig and Wittchen19 conducted by a licensed psychiatrist, complemented by PSST Reference Steiner, Macdougall and Brown21 assessments at screening and during the premenstrual phase. While participants prospectively recorded their mood over 2–3 months, these recordings did not capture the full daily spectrum of PMDD symptoms. We acknowledge that daily prospective symptom tracking over multiple cycles would provide stronger diagnostic confirmation. Fifth, due to the exploratory nature of the study, future research on CAR and its relation to the serotonergic system should focus on cortisol peak as outcome measure and confirm the findings of the current study. Sixth, because the study was correlational, we cannot draw definite conclusions about the causal relationships among cortisol dynamics, serotonergic activity and depressive symptoms. While we were able to establish a time course of events due to the longitudinal design, providing important insights into these associations, further interventional studies (e.g. manipulating HPA axis response within the menstrual cycle and/or providing a serotonergic intervention in patients) are needed to establish causality and clarify the underlying mechanisms.
This longitudinal study demonstrates attenuated cortisol peaks in patients with PMDD compared with controls during the periovulatory phase, as well as underlying associations with the serotonergic system and the severity of depressive symptoms during the premenstrual phase. This study characterises PMDD as a disorder showing alterations in endocrine stress system regulation.
Supplementary material
The supplementary material is available online at https://doi.org/10.1192/bjp.2025.10432
Data availability
Codes for statistical analyses are available from the corresponding author upon reasonable request.
Acknowledgements
We thank Anne Krieger for helping with the graphic design of Fig. 1.
Author contributions
Conceptualisation: K.H., R.G.Z., V.E., J.S. Methodology: A.V., S.H., O.S., V.E., J.S. Formal analysis: K.H., R.G.Z., V.E., J.S. Investigation: R.G.Z., J.S. Resources: A.V., S.H., O.S., J.S. Writing (original draft): K.H., J.S. Writing (review and editing): K.H., R.G.Z., A.V., S.W., O.S., V.E., J.S. Visualisation: K.H. Supervision: V.E., J.S. Funding acquisition: J.S.
Funding
Funding was provided by Humboldt-Universität zu Berlin, Berlin School of Mind and Brain (to K.H.), and by DFG (no. 534642099), MPG Brain HATCH Project (Human Cognition Hormones) and University Medical Centre Leipzig. Open access funding provided by Max Planck Society.
Declaration of interest
None.






eLetters
No eLetters have been published for this article.