Introduction
Physical dormancy (PY) is a form of seed dormancy imposed by a water-impermeable seed or fruit coat, typically resulting from a specialized palisade cell layer that restricts water uptake, thereby preventing germination even under otherwise favourable conditions (Baskin and Baskin, Reference Baskin and Baskin2021). PY has been documented thus far in several genera of 19 angiosperm families, reflecting its broad ecological significance and adaptive value in delaying germination until environmental conditions are suitable for seedling establishment (Meisert, Reference Meisert2002; Turner et al., Reference Turner, Merritt, Baskin, Dixon and Baskin2005; Jaganathan, Reference Jaganathan2025b). PY is broken when environmental cues such as temperature fluctuations (Jayasuriya et al., Reference Jayasuriya, Athugala, Wijayasinghe, Baskin, Baskin and Mahadevan2015; Cuena Lombraña et al., Reference Cuena Lombraña, Dessì, Podda, Fois, Luna, Porceddu and Bacchetta2024), fire (Pausas and Lamont, Reference Pausas and Lamont2022), or gut passage (Long et al., Reference Long, Gorecki, Renton, Scott, Colville, Goggin, Commander, Westcott, Cherry and Finch‐Savage2015; Jaganathan et al., Reference Jaganathan, Yule and Liu2016) induce structural changes in the seed coat, particularly at the water gap – a specialized region that functions as an environmental sensor and regulates water entry (Gama-Arachchige et al., Reference Gama-Arachchige, Baskin, Geneve and Baskin2013; Geneve et al., Reference Geneve, Baskin, Baskin, Jayasuriya and Gama-Arachchige2018). Interestingly, empirical treatments such as hot water or wet heat effectively mimic natural cues and promote water gap opening (Baskin and Baskin, Reference Baskin and Baskin2014). In contrast, acid scarification both inside the gut and empirical application, breaks dormancy by presumably altering other regions of the seed coat without necessarily opening the water gap (Jaganathan et al., Reference Jaganathan, Li, Biddick, Han, Song, Yang, Han and Liu2019). Once a pathway for entry is created either through water gap or other cracks in the seed coat, water rapidly enters the seed, triggering metabolic activation and proceeding to the onset of germination (Jaganathan, Reference Jaganathan2025a).
Water gaps are classified into three types based on their morphology and mechanism of opening: Type I, where surface cells separate to form a water entry point; Type II, involving the lifting or displacement of a lid-like structure; and Type III, characterized by the dislodgement of a plug-like structure known as an oculus (Gama-Arachchige et al., Reference Gama-Arachchige, Baskin, Geneve and Baskin2013; Geneve et al., Reference Geneve, Baskin, Baskin, Jayasuriya and Gama-Arachchige2018). Water gaps may occur as simple structures (a single entry site) or as part of a compound complex involving two, coordinated openings. Water gap structure has been thoroughly examined in several species, such as Fabaceae, Malvaceae, Sapindaceae, and their ecological importance has been understood to some extent (Baskin, Reference Baskin2003; Gama-Arachchige et al., Reference Gama-Arachchige, Baskin, Geneve and Baskin2013; Geneve et al., Reference Geneve, Baskin, Baskin, Jayasuriya and Gama-Arachchige2018). In Dodonaea petiolaris F. Muell. (Sapindaceae), Turner et al. (Reference Turner, Cook, Baskin, Baskin, Tuckett, Steadman and Dixon2009) identified that a small plug-like structure positioned near the hilum (Type II) and opposite the radicle acts as the water gap. This plug, composed of palisade and subpalisade layers, is displaced in response to moist heat or boiling water, enabling water imbibition and subsequently triggering germination. The water gap in Sapindus species has been documented as the hilar slit (Type I), while micropylar slit is the water gap in Cardiospermum halicacabum L. (Gama-Arachchige et al., Reference Gama-Arachchige, Baskin, Geneve and Baskin2013; Geneve et al., Reference Geneve, Baskin, Baskin, Jayasuriya and Gama-Arachchige2018). On the other hand, Koelreuteria paniculata Laxm. has a Type III water gap in the form of a hilar oculus – a circular, plug-like structure that is dislodged under moist heat, with additional blister-like structures potentially forming under dry heat (Geneve et al., Reference Geneve, Baskin, Baskin, Jayasuriya and Gama-Arachchige2018). The water gap has been extensively studied at the genus level in Fabaceae, whereas in Sapindaceae it has been identified in only one species within a genus, thereby limiting knowledge of water gap at the genus level.
PY seeds are unequivocally desiccation tolerant, as the onset of seed coat impermeability in species known to exhibit PY typically occurs only after moisture content falls below a critical threshold, generally ranging from 14% to 7% on a fresh weight basis (Jaganathan, Reference Jaganathan2022). PY seeds are capable of surviving for millennia under ambient room temperature conditions (Shen-Miller, Reference Shen-Miller2002), indicating that low-temperature storage may be unnecessary to ensure long-term storage (Shen‐Miller et al., Reference Shen‐Miller, Mudgett, Schopf, Clarke and Berger1995). Interestingly, exposing species with impermeable seed coat to liquid nitrogen has been shown to alleviate dormancy (Fernández et al., Reference Fernández, Marrero and Montero2021; Gong et al., Reference Gong, Zhang, Lv, Lv, Zhao, Sha, Zhou and Sun2025). For instance, seeds of Trifolium arvense improved germination following liquid nitrogen (LN2) storage (Pritchard et al., Reference Pritchard, Manger and Prendergast1988). In Acacia seyal Delile (Fabaceae) and Delonix regia (Bojer ex Hook.) Raf. (Fabaceae), brief LN2 exposure followed by warm water treatment has been shown to markedly increase germination rates (Hassan and Hassan, Reference Hassan and Hassan2019). Similarly, seeds of Tilia miqueliana Maxim. (Malvaceae), LN2 treatment alters the ultrastructure of the pericarp – reducing cellulose and hemicellulose content, weakening tissue integrity, and producing intercellular gaps – which accelerates seed coat removal while preserving viability (Wu et al., Reference Wu, Sun, Peng, Shen, Visscher, Pritchard, Wang and Deng2023). These results demonstrate the efficacy of cryogenic scarification as a viable alternative to acid-based methods, albeit this method has not gained attention in breaking PY. The majority of previous studies on the effect of LN2 on PY break have been conducted in Fabaceae species; therefore, and we explored this in Sapindacea.
Dodonaea viscosa commonly known as broadleaf hopbush, is a widely distributed evergreen shrub or small tree belonging to the family Sapindaceae. The species exhibits remarkable ecological versatility, occupying a broad range of habitats across tropical, subtropical, and warm temperate regions, including parts of Africa, the Americas, southern Asia, and Australasia. Previously, the species was considered non-dormant, but several studies across different locations clearly show the seeds have PY (Baskin et al., Reference Baskin, Davis, Baskin, Gleason and Cordell2004; Phartyal et al., Reference Phartyal, Baskin, Baskin and Thapliyal2005; Jaganathan and Liu, Reference Jaganathan and Liu2014). Thus, we posed the following question: 1) What is the anatomical location and structural nature of the water gap in seeds of D. viscosa? 2) How does exposure to liquid nitrogen affect seed viability and the integrity of seed coat structures in D. viscosa?
Materials and methods
Seed material
The seeds of D. viscosa were collected from 23 trees growing within 2 km radius, in the autumn of 2023 from Dali Bai Autonomous Prefecture, Yunnan Province, China (99°09′ ∼ 102°19′E, 22°02′ ∼ 24°50′N). The collected seeds were transported to the laboratory and stored in sealed bags at room temperature (25°C) with a relative humidity of 50% for three months before the onset of the experiment.
Seed mass and moisture content determination
Three replicates of 100 randomly selected seeds were weighed using a digital balance with a precision of 0.0001 g (QUINTIX35-1CN, Sartorius) to determine the average seed mass. Seed moisture content was assessed following the standard protocol recommended by the International Seed Testing Association (ISTA, 2024). Three replicates of 15 seeds were oven-dried at 103 °C for 17 hours, and MC was calculated as the percentage reduction in mass from fresh to dry weight, expressed on a fresh weight basis (%).
Seed imbibition and germination
Four replicates of 25 D. viscosa seeds were imbibed on moist filter paper in 90 mm Petri dishes and incubated in a growth chamber with a 12 h photoperiod at 25 °C and 12 h darkness at 20 °C. This temperature range reflects the spring conditions in Dali, Yunnan. To monitor imbibition, seeds were removed daily, surface water dried by placing them in between soft paper towels, weighed, and returned to the dishes. Seed water uptake was expressed as a percentage increase relative to initial seed mass (Baskin et al., Reference Baskin, Davis, Baskin, Gleason and Cordell2004). Germination was assessed using four replicates of 25 seeds under identical incubation conditions, with seeds examined every two days over 30 days. Seeds with emerged radicles ≥ 2 mm were scored as germinated.
Following this initial assessment, all seeds were soaked in water, and permeable seeds were identified and removed upon swelling, thereby separating them from the impermeable seeds. In all subsequent experiments, only impermeable seeds were used, referred to as untreated seeds.
Dormancy-breaking treatments
Hot-water treatment
One hundred untreated seeds for each group were enclosed in mesh bags and immersed in boiling water (100 ± 2°C) for 5, 10, 20, or 30 seconds. Subsequently, seeds were removed and cooled at ambient temperature for 15 minutes before being placed on moist filter paper in Petri dishes to assess imbibition, under the conditions described above. The percentage increase in seed mass was calculated until germination began. Following this, we recorded germination until either all seeds germinated or no germination occurred for 30 days.
Liquid nitrogen (LN2) freeze-thaw cycle treatment
Five replicates of 100 untreated seeds each were placed in separate non-woven bags and immersed in a dewar flask containing LN₂. After 24 hours of immersion in LN₂, seeds were retrieved and rewarmed at room temperature for 2 hours, constituting one freeze–thaw cycle. One group was used for germination assessment, while the remaining groups were returned to LN₂. Subsequently, the other four groups underwent 2, 3, 4, and 5 freeze–thaw cycles, respectively.
Morphological anatomy
Photomicrographs
A Canon EOS 60D digital camera was used to photograph intact seeds, seeds after one day of imbibition, and their cross sections; separate photographic sessions with appropriate adjustments were conducted to document impermeable seeds before and after liquid nitrogen (LN₂) treatment to ensure optimal image quality at each stage.
Light microscopy
Untreated impermeable seeds, those subjected to boiling water and LN₂ treatments were mounted on glass slides and examined using a stereo optical microscope (PXS6555T-J3, CEWEI) at appropriate magnifications. Images were captured with a digital camera integrated into the microscope to document morphological features and treatment effects. To ensure high-quality image acquisition for comparative analysis, lighting and focus were kept similar for all images.
Blocking the water gap
One hundred impermeable seeds were immersed in boiling water for 10 seconds and subsequently air-dried. The hilum region of each seed was examined individually under a stereo microscope and sealed with a hydrophobic layer of Vaseline applied using a sharpened toothpick. The sealed seeds were then randomly assigned to four groups and placed in Petri dishes containing wet filter paper to assess imbibition, which was quantified as the percentage increase in seed mass.
Scanning electron microscopy (SEM)
Three untreated impermeable and four impermeable seeds treated with boiling water for 10 seconds were analysed using atmospheric electron microscope. Seeds were mounted on sample stubs using carbon paint and sputter-coated with a 5 nm gold layer using a Leica EM ACE600 sputter coater. The coated samples were examined with a JSM-IT500HR field emission scanning electron microscope operated at an accelerating voltage of 3 kV to assess alterations in the hilum following hot water treatment.
Analysis of data
Linear mixed-effects models were used to analyse germination responses to hot-water duration and liquid nitrogen (LN₂) freeze–thaw cycles. Germination data were aggregated at the replicate level (number germinated out of 25 seeds), and proportions were slightly adjusted to avoid boundary values of 0 and 1 using ![]() $\left( {g + 0.5)/(n + 1} \right)$ before logit transformation. The resulting logit-transformed germination proportion was treated as the response variable, with treatment (hot-water duration or LN₂ cycle number) included as a fixed factor and replicate group fitted as a random intercept to account for among-group variability.
$\left( {g + 0.5)/(n + 1} \right)$ before logit transformation. The resulting logit-transformed germination proportion was treated as the response variable, with treatment (hot-water duration or LN₂ cycle number) included as a fixed factor and replicate group fitted as a random intercept to account for among-group variability.
All analyses were conducted in R using the package lme4 for mixed-effects modelling and emmeans for post-hoc contrasts (Brown, Reference Brown2021; Team, 2025). For each experiment, Wald z-statistics with associated p-values were extracted from model summaries to assess fixed effects. Estimated marginal means on the logit scale were obtained with emmeans, back-transformed to probabilities with 95% confidence intervals, and Tukey-adjusted pairwise comparisons among treatments were used to identify significant differences while controlling for multiple testing. Pairwise comparisons are reported as Tukey-adjusted tests on estimated marginal means from the mixed-effects models.
Results
Seed mass, imbibition and germination
The mass of one hundred seeds was 1.05 ± 0.02 g. The germination percentage of fresh seeds was 11.00 ± 0.83%, and the proportion of impermeable seeds was 47.00 ± 0.83%. The moisture content of the seed batch, comprising both permeable and impermeable seeds upon arrival at the laboratory, was 5.68 ± 0.33%.
Dormancy-breaking treatments
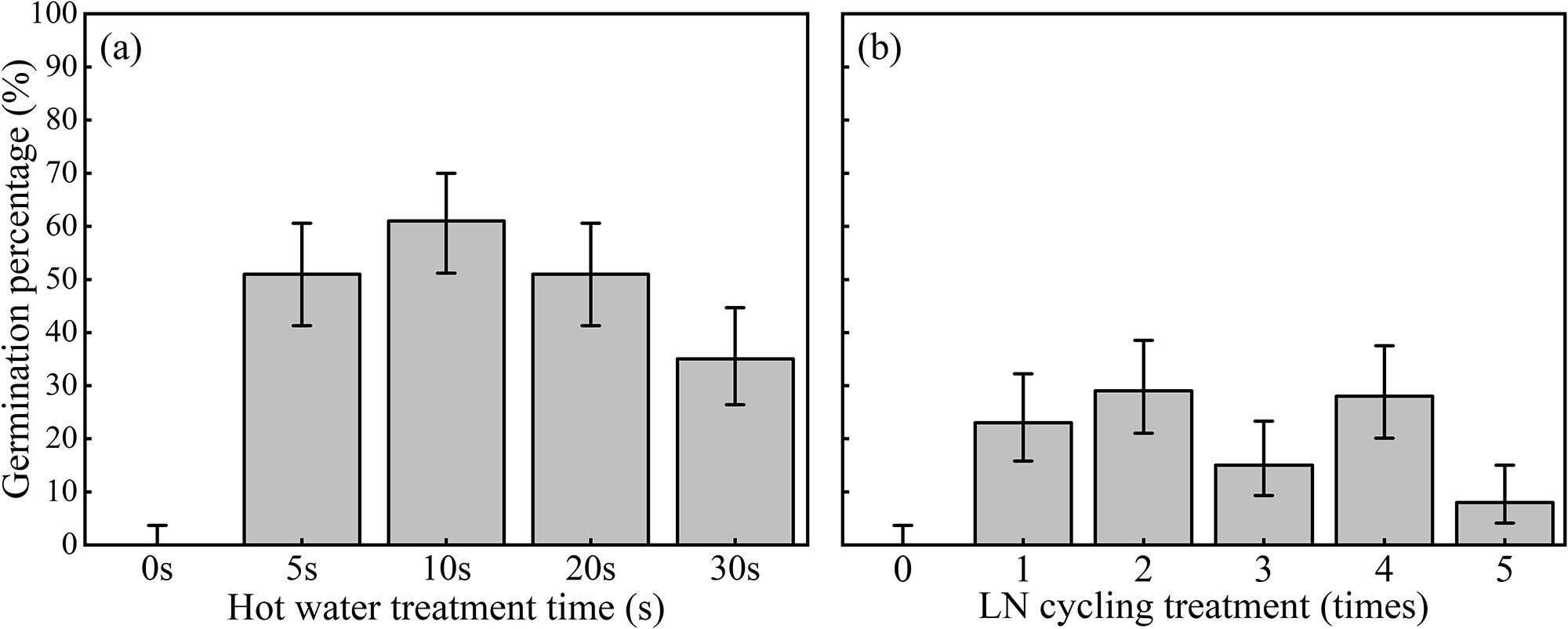
Untreated seeds showed no germination, whereas all hot-water treatments (5–30 s) resulted in significantly higher germination probabilities (Figure 1a). In the corresponding linear mixed-effects model, hot-water exposure yielded strong positive fixed effects relative to the control (5 s: z = 5.06. p < 0.001; 10 s: z = 6.45, p < 0.001; 20 s: z = 5.39, p < 0.001; 30 s: z = 4.43, p < 0.001). The random intercept for group captured within-treatment variation among replicate seed batches and was moderate, yet the treatment effects remained strong and statistically significant after accounting for this variability. Further, differences among germination after different duration in hot-water were comparatively small after controlling for this among-replicate variability, indicating that brief immersion (5–10 s) is sufficient to break dormancy and that longer exposure may be detrimental to the seeds (Figure 1a).
Germination after (a) hot water treatments, and (b) LN2 freeze-thaw cycle treatment. The error bars represent the 95% Wilson CI (%).

Germination responses to liquid nitrogen freeze–thaw cycles were more modest and variable across replicates (Figure 1b). The mixed-effects model revealed no statistically significant differences in germination probability among 2–5 LN₂ cycles relative to one cycle (2 cycles: z = 0.37, p = 0.71; 3 cycles: z = − 0.55, p = 0.58; 4 cycles: z = 0.18, p = 0.85; 5 cycles: z = − 1.26, p = 0.21), and the estimated group-level variance indicated appreciable within-treatment variability among replicate lots. Nevertheless, the negative coefficient estimated for five cycles, even after accounting for this replicate-to-replicate variation, suggests a trend toward reduced germination under the most intense freeze–thaw treatment (Figure 1b).
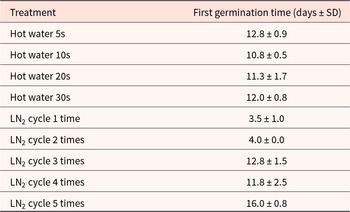
The time to germinate varied markedly across treatments (Table 1). Hot water treatments generally accelerated germination rate compared with multiple LN2 cycles, with the fastest germination observed after LN2 cycle 1 (3.50 ± 1.00 days) and hot water 5 s (12.75 ± 0.96 days) showing intermediate timing, while higher durations and additional LN2 cycles delayed germination (Table 1).
First germination time after different seed treatments for Dodonaea viscosa. Means ± SD (days to germination) are shown for hot-water treatments (5–30 s) and LN2 cycling (1–5 cycles). Shorter times indicate faster germination; longer times indicate slowed germination due to treatment intensity

The first germination time (days) varied markedly across treatments (Table 1). Hot water treatments generally accelerated germination rate compared with multiple LN2 cycles, with the fastest germination observed after LN2 cycle 1 (3.50 ± 1.00 days) and hot water 5 s (12.75 ± 0.96 days) showing intermediate timing, while higher durations and additional LN2 cycles delayed germination (Table 1).
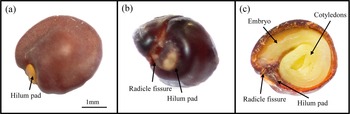
Intact D. viscosa seeds exhibited a clearly defined hilum pad, which was readily visible as a distinct structure on the seed surface (Figure 2a). In seeds dipped in hot-water, after 24 hours of imbibition, seeds displayed the emergence of a radicle fissure adjacent to the hilum pad, indicating initial uptake of water and the commencement of physiological processes leading to germination (Figure 2b). Subsequent cross-sectional examination of imbibed seeds revealed that internal structures, including the embryo and two fleshy cotyledons, had become fully distinguishable (Figure 2c). Notably, the radicle fissure and hilum pad were prominent, and the increased separation in these regions corresponded with tissue hydration and expansion.
Photographs of Dodonaea viscosa seeds. (a) intact seed; (b) seed after imbibing 1 day; (c) cross-section of seed after imbibing 1 day.

Identification of water gap
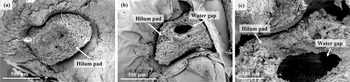
Under stereo optical microscopy, untreated, intact impermeable seeds exhibited a distinct, intact hilum pad, yet showed no visible structural openings (Figure 3a, b). In contrast, seeds subjected to boiling water treatment for 10 seconds displayed pronounced anatomical changes in the hilum pad, which clearly opened up forming the water gap (Figure 3c, d). SEM confirmed impermeable seeds displayed a robust and unbroken hilum pad, with the surrounding seed coat exhibiting no entry route for water (Figure 4a). In contrast, seeds treated with boiling water for 10 seconds revealed the formation of a conspicuous water gap adjoining the hilum pad (Figure 4b, c).
Light micrographs of the surface of water gap regions of Dodonaea viscosa seeds. (a, b) Impermeable seeds; (c, d) After 10s hot water treatment.

Scanning electron micrographs of Dodonaea viscosa seeds. (a-c) Impermeable seeds; (d-f) After 10s hot water treatment.

Seed imbibition and blocking the water gap
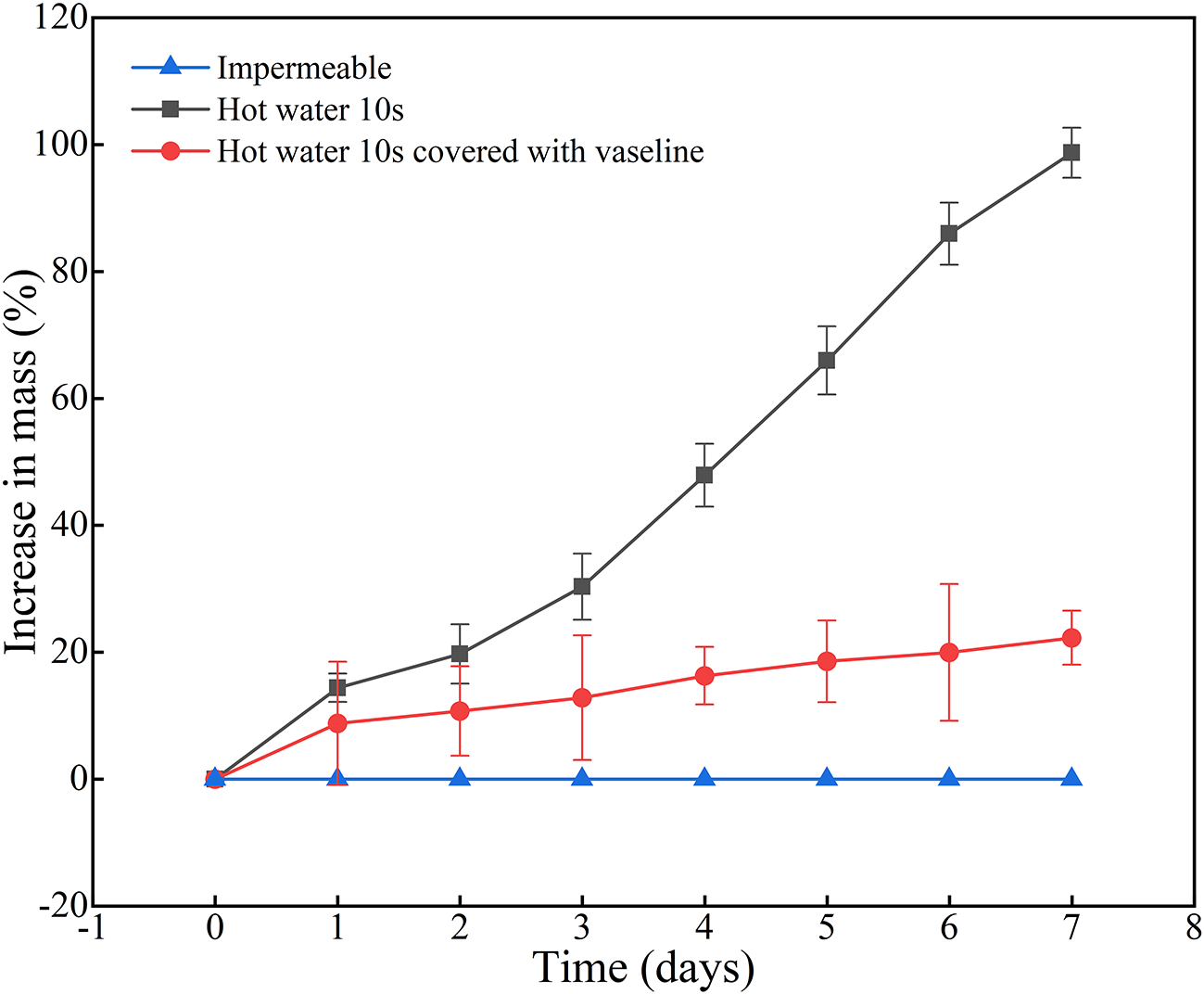
Untreated, intact impermeable did not absorb water and did not increase mass for the 7day (Figure 5) and upto 60 days (data not shown), as we separated impermeable and permeable seeds, and used only impermeable seeds for this experiment. In contrast, seeds treated with hot water for 10 seconds exhibited a rapid and sustained increase in mass, reaching nearly 100% by day 7, by which time 10% of the seeds germinated. When 10s hot-water treated seeds were covered with Vaseline at the hilum, the increase in mass was substantially reduced. Three seeds absorbed water after 30 days, which led to the increase in seed mass percentage.
Percentage increase in mass of impermeable seeds, hot water-treated seeds, hot water-treated seeds covered with Vaseline and measured at 1-day intervals.

Discussion
A substantial proportion of freshly collected D. viscosa seeds failed to absorb water during imbibition tests, confirming the presence of a water-impermeable seed coat and thus PY. This result corroborates earlier reports of seed coat impermeability in D. viscosa and based on consistent observations from multiple geographic regions (Hodgkinson and Oxley, Reference Hodgkinson and Oxley1990; Burrows, Reference Burrows1995; Phartyal et al., Reference Phartyal, Baskin, Baskin and Thapliyal2005; Benítez‐Rodríguez et al., Reference Benítez‐Rodríguez, Gamboa‐deBuen, Sánchez‐Coronado, Alvarado‐López, Soriano, Méndez, Vázquez‐Santana, Carabias‐Lillo, Mendoza and Orozco‐Segovia2014; Jaganathan and Liu, Reference Jaganathan and Liu2014), we conclude PY in this species is definitive trait irrespective of the location. The MC of the seeds collected for this study was 5.68%, which was slightly lower than the 7.8% reported by Jaganathan and Liu (Reference Jaganathan and Liu2014) from south India. However, approximately 10% of seeds from both locations germinated. Nonetheless, in the present study many permeable seeds failed to germinate and often became softened. Because the seeds look healthy during the visual observation at the time of starting experiments, we suspect that the interior seeds had suffered damage from insects or other abiotic factors, as casual cut tests of the ungerminated seeds revealed damaged embryos.
Interestingly, the proportion of permeable seed observed in this work is common in several PY species (Baskin and Baskin, Reference Baskin and Baskin2014). This has been suggested as an adaptation and a protection strategy, wherein the permeable proportion of the seeds germinate immediately and the impermeable proportion establish persistent soil seed bank and germinate after fire or seasonal temperature fluctuations could make them permeable (Zalamea et al., Reference Zalamea, Sarmiento, Arnold, Davis and Dalling2015; McCulloch et al., Reference McCulloch, Dalling and Zalamea2024). The percentage of permeable proportion tends to be largely influenced by the environmental conditions during seed development, affecting the moisture content of the seeds (Magalhães et al., Reference Magalhães, Garcia and Oliveira2021) and plausibly the hardness of the seed coat (Visscher et al., Reference Visscher, Gómez Barreiro, Soto Gomez, Carta, Liu, Wu, Muthuthanthirige, Forest, McCabe and Pritchard2025). The permeable proportion of the seeds can subsequently become impermeable when the permeable seeds on soil dry and reach a moisture content that onsets impermeability(Tozer and Ooi, Reference Tozer and Ooi2014; Jaganathan, Reference Jaganathan2022).
Although boiling water treatment and soil temperature fluctuations have been recognized as effective methods for breaking dormancy in D. viscosa (Baskin et al., Reference Baskin, Davis, Baskin, Gleason and Cordell2004; Jaganathan and Liu, Reference Jaganathan and Liu2014), the primary objective of this study was to elucidate the anatomical features of water gap structures within this species (Jaganathan and Liu, Reference Jaganathan and Liu2014; Wang et al., Reference Wang, Yan, Fan, Shi and Liu2018). In the family Sapindaceae, several types of water gap structures have been identified, including the hilar oculus, micropylar slit, gap adjacent to the hilum, and hilar slit (Gama-Arachchige et al., Reference Gama-Arachchige, Baskin, Geneve and Baskin2013). Within the genus Dodonea, previous studies have suggested that the water gap is localized at the gap adjacent to the hilum (Turner et al., Reference Turner, Cook, Baskin, Baskin, Tuckett, Steadman and Dixon2009). However, our SEM and photomicrographic analyses revealed that the hilar slit undergoes opening in response to hot-water treatment. To confirm whether the observed opening of the hilar slit served as the functional water gap, we conducted detailed imbibition studies. Our SEM and photomicrographic analyses results demonstrated that water uptake in D. viscosa seeds primarily occurs through the hilar slit. Because this is a smaller part of the seed coat, the water uptake was slow requiring 10 days for germination to occur (Table 1).
In the present work, the water gap in D. viscosa was identified as the gap adjacent to the hilum with and include them under Type II, characterized by the formation and opening of a hilar slit that permits water entry into the seed. This structure is distinct from several other Sapindaceae species, which possess Type I water gaps such as the hilar oculus or micropylar slit (Gama-Arachchige et al., Reference Gama-Arachchige, Baskin, Geneve and Baskin2013; Geneve et al., Reference Geneve, Baskin, Baskin, Jayasuriya and Gama-Arachchige2018). Unlike Type I gaps which are found in several genera of Sapindaceae (Gama-Arachchige et al., Reference Gama-Arachchige, Baskin, Geneve and Baskin2013), Dodonea shows Type II water gap (Turner et al., Reference Turner, Cook, Baskin, Baskin, Tuckett, Steadman and Dixon2009). No secondary structural openings were observed during dormancy break, and seeds failed to imbibe water when the hilar slit was covered with Vaseline. Therefore, at least in Dodonea, there is no evidence for occurrence of water gap complex. As such, this result indicates intra-genus variation or species-specific adaptations in PY species. Whilst the evolutionary and ecological significance of water gap complex compared with simple water gap remains poorly understood (Geneve et al., Reference Geneve, Baskin, Baskin, Jayasuriya and Gama-Arachchige2018), our results indicate intra-genus variation or species-specific adaptations in PY species. It has been shown before water gap can vary within genus in some Fabaceae. Within the genus Senna, the structure functioning as the water gap varies among species. For instance, Senna alata utilizes only the pleurogram as the water gap, whereas Senna reniformis and Senna silvestris exhibit water entry through both the pleurogram and the hilar region, demonstrating functional diversity within a single genus (Rodrigues-Junior et al., Reference Rodrigues-Junior, Mello, Baskin, Baskin, Oliveira and Garcia2019). We demonstrate such variation within the genus Dodonaea, highlighting that the water gap can be a similar yet distinct trait in some closely related species, providing evidence of the diversity in other genera.
LN₂ immersion has been explored as a method to break PY in seeds (Patanè and Gresta, Reference Patanè and Gresta2006). When cracks are confined to the seed coat and do not damage critical seed tissues, these cracks can act as the water entry site for germination to occur. Indeed, this crack could be larger than the water gap, explaining quicker germination time after LN₂ treatment (Table 1). Consequently, this method can be effectively applied to a large proportion of seeds (Martin and De la Cuadra, Reference Martin and De la Cuadra2004; Schnadelbach et al., Reference Schnadelbach, Veiga-Barbosa, Ruiz, Pita and Pérez-García2016; Pullman et al., Reference Pullman, Bucalo, Determann and Cruse-Sanders2021; Acosta et al., Reference Acosta, Escobar-Gutiérrez, Ahmed, Cejas, Martínez-Montero, Sánchez, Hajari, Höfer, Lorenzo and Fontes2023). However, our findings demonstrate that LN₂ immersion may induce substantial seed damage rather than effective dormancy release. Following LN₂ freeze–thaw cycles, seeds exhibited pronounced fragmentation of the seed coat and severe cracking of internal tissues, especially the embryo and cotyledons. Such extensive mechanical injury compromises seed integrity, reduces viability (Figure 6), and dramatically impairs the potential for successful germination (Figure 1 (b)).
Photographs of Dodonaea viscosa seeds before and after LN2 treatment. (a, b) seeds before LN2 treatment; (c, d) seeds after LN2 treatment.

LN₂ treatment fails to selectively disrupt the water-impermeable layers and often results in the destruction of critical embryonic structures (Figure 6), which limits its effectiveness as a dormancy breaking method for D. viscosa and presumably for other PY species (Salomao, Reference Salomao1995). However, damage to internal structures, such as the cotyledons or embryo (Figure 6), is not always lethal, and seeds can still germinate despite such damage (Wiesner et al., Reference Wiesner, Laufmann, Stanwood and Wheeler1994). In the present study, we observed only a few seeds germinated (Figure 1b). While cryopreservation is regarded as a means to maintain seed viability over extended periods, seeds dried excessively can excessively damage the seed coat and internal tissues (Stanwood, Reference Stanwood and Kartha1985). The seed MC alone cannot explain this, as this phenomenon is not observed in all species dried to similar MC (Salomão, Reference Salomão2002). Thus, the damage to the seed coat can be attributed to the structural and chemical makeup of the seed coat. For example, the sharp temperature change induced mechanical stress from thermal expansion of the embryo and seed coat, causing damage to the hilum of Glycyrrhiza uralensis Fisch. (Fabaceae) seeds (Kruglikov et al., Reference Kruglikov, Bystrushkin and Belyaev2022). Seeds of PY species have been successfully cryopreserved before. Yet, PY seeds with hard, water-impermeable coats may be particularly susceptible to fissures or fractures upon rapid freezing and thawing. As observed in D. viscosa, LN₂ exposure can result in unintentional damage to the seed coat and embryo, which may compromise post-storage germination and seedling establishment. Therefore, protocols for the cryostorage of easy-to-crack PY seeds should be carefully optimized and preceded by a thorough assessment of potential LN₂-induced injury. Because the cooling rate has been known to affect how seed coat responds to low temperature (Stanwood, Reference Stanwood1980), alternatives that minimize mechanical shock or use slower cooling and rewarming steps may be warranted to preserve both dormancy and viability in such seeds.
In conclusion, this study identifies the hilar slit as the water gap (Type II) in D. viscosa seeds, which differs from D. petiolaris, functioning as a specialized structure that acts as the water entry during dormancy break following hot-water treatment. Our study shows that species within a single genus can exhibit different water gaps within Sapindaceae. Further, we have shown that there are no secondary openings, which confirms the absence of water gap complex. Clearly, exposure to LN₂ freeze–thaw cycles induces irreversible damage to the seed coat, and internal tissues, thereby affecting seed viability and limiting the utility of this method for dormancy alleviation and seed conservation. These findings call for linking precise anatomical and physiological characterization of water gap structures in a wide-range of species and develop effective methods for PY seed conservation.
Author contribution
Xuemin Han, Data curation, Formal analysis, Methodology, Resources, Software, Validation, Visualization, Writing – original draft. Ganesh K. Jaganathan, Conceptualization, Funding acquisition, Investigation, Project administration, Supervision, Writing – review and editing. Baolin Liu, Funding acquisition, Project administration, Supervision.
Declaration of funding
This research was funded by the National Natural Science Foundation of China (NSFC), grant number 32001119.
Conflicts of interest
The authors declare that they have no conflicts of interest.
Data availability
The data used in this research are available from the author upon request.