Summations
This scoping review synthesises evidence from 30 studies examining the Emotional Go/No-Go task (EGNG), Delay Discounting Task (DDT), and Balloon Analogue Risk Task (BART) among adolescents evaluated for depression and anxiety. Across tasks, behavioural associations with internalising symptoms were generally small, inconsistent, and context dependent. EGNG and BART paradigms most often revealed selective emotional-context effects rather than global deficits in inhibitory control or risk-taking. Evidence for DDT was limited but suggested that associations with depression and anxiety may vary across developmental, cultural, and socioeconomic contexts. The literature remains concentrated in high-income, English-speaking settings, with relatively few longitudinal or intervention studies.
Considerations
Several limitations of the current evidence base should be noted. First, the predominance of cross-sectional designs limits inference about developmental trajectories and causal relationships between self-regulation processes and internalising symptoms. Second, substantial heterogeneity in task implementation, outcome metrics, and reporting practices reduces comparability across studies. Third, the geographic concentration of studies in high-income countries constrains generalisability, particularly given the importance of cultural and contextual factors in neuropsychological task performance. Finally, the small number of intervention and longitudinal studies limits conclusions about clinical utility and sensitivity to change. Future research should prioritise harmonised protocols, culturally adapted task implementations, and longitudinal and intervention-based designs to clarify when neuropsychological measures of self-regulation are most informative for adolescent mental health research.
OSF registration number: https://doi.org/10.17605/OSF.IO/P4RHB.
Introduction
Adolescence is a sensitive developmental period for the maturation of cognitive control, emotion regulation, and value-based decision-making. These are core components of self-regulation that support goal-directed behaviour in increasingly complex social environments (Steinberg, Reference Steinberg2008; Crone & Dahl, Reference Crone and Dahl2012). Self-regulation broadly refers to the ability to modulate thoughts, emotions, and behaviours in response to situational demands, drawing on inhibitory control, attentional selection, and affective modulation (Diamond, Reference Diamond2013). These capacities rely on coordinated development within fronto-limbic circuitry, including protracted maturation of the prefrontal cortex and its connectivity with subcortical regions implicated in salience and affect (Casey et al., Reference Casey, Jones and Hare2008). This neurodevelopmental trajectory creates a window of heightened plasticity supporting rapid gains in regulatory skills while simultaneously increasing vulnerability to psychopathology when regulatory demands outpace regulatory capacity.
Depressive and anxiety disorders often emerge during adolescence (Racine et al., Reference Racine, McArthur, Cooke, Eirich, Zhu and Madigan2021; Kieling et al., Reference Kieling, Buchweitz, Caye, Silvani, Ameis, Brunoni, Cost, Courtney, Georgiades, Merikangas, Henderson, Polanczyk, Rohde, Salum and Szatmari2024) and are linked to alterations in self-regulatory processes (Li et al., Reference Li, Xia, Shen, Dong, Xu and Yang2024) and other executive functions (Fontaine et al., Reference Fontaine, Blake, Koen, Stein, Hammar and Groenewold2025). At a cognitive level, depression has been associated with executive dysfunction, slowed processing, working memory difficulties, and a bias toward negative information processing, while anxiety is frequently characterised by heightened threat sensitivity, attentional capture by salient cues, and difficulties disengaging from negative or uncertain information (Davidson et al., Reference Davidson, Pizzagalli, Nitschke and Putnam2002; Gotlib & Joormann, Reference Gotlib and Joormann2010; Hasselbalch et al., Reference Hasselbalch, Knorr and Kessing2011; Thibaut et al., Reference Thibaut, Mwamuka, Nyamayaro, Rubin, Nakasujja, Langenecker and Abas2023).
These profiles have motivated growing interest in objective, performance-based measures that can complement symptom scales and clarify candidate mechanisms relevant for prevention and intervention. Neuropsychological approaches are particularly attractive in this context because they provide standardised task environments in which inhibitory control, reward valuation, and risk-related decision-making can be quantified in ways that may be less affected by self-report biases and may map more directly onto theoretical models of affective and cognitive control (Kaltenboeck & Harmer, Reference Kaltenboeck and Harmer2018).
Among the wide range of paradigms used to assess self-regulation, three task families appear in adolescent internalising research: the Emotional Go/No-Go (EGNG) task, the Delay Discounting Task (DDT) and the Balloon Analogue risk task (BART). The EGNG assesses affective inhibitory control by requiring participants to execute or withhold a prepotent response in the presence of emotionally salient cues (often emotion words or facial emotional expressions), thereby capturing the interaction between cognitive control and affective interference (Hare et al., Reference Hare, Tottenham, Davidson, Glover and Casey2005; Schulz et al., Reference Schulz, Fan, Magidina, Marks, Hahn and Halperin2007). The DDT measures intertemporal choice by asking individuals to choose between smaller, immediate rewards and larger, delayed rewards; steeper discounting reflects a stronger preference for immediate outcomes and is commonly interpreted as reduced capacity to tolerate delay or to value future consequences (Mazur, Reference Mazur, Commons, Mazur, Nevin and Rachlin1987; Kirby et al., Reference Kirby, Petry and Bickel1999). The BART evaluates risk-taking behaviour by having participants incrementally inflate virtual balloons to accrue potential gains while facing an increasing probability of loss, offering a behavioural proxy for risk–reward trade-offs under uncertainty (Lejuez et al., Reference Lejuez, Read, Kahler, Richards, Ramsey, Stuart, Strong and Brown2002; Lejuez et al., Reference Lejuez, Aklin, Daughters, Zvolensky, Kahler and Gwadz2007). Together, these paradigms provide complementary windows into inhibitory control under emotional load, valuation of delayed rewards, and risk-taking under uncertainty domains relevant to both depression and anxiety.
The existing literature nevertheless raises important questions about consistency and interpretability. Emotion-laden inhibitory-control tasks, for example, can yield different conclusions depending on stimulus type (faces versus words), task structure (go/no-go ratios, timing parameters), and outcome definitions (commission errors, omission errors, reaction time indices, or composite bias scores). Related evidence indicates that early adversity and chronic stress may further alter emotion–control interactions, which may in turn influence performance patterns on tasks such as the EGNG (Tottenham et al., Reference Tottenham, Hare and Casey2011).
Parallel concerns apply to decision-making paradigms. Delay discounting has been proposed as a transdiagnostic marker relevant to affective symptoms and self-regulatory difficulties, including problems with delay of gratification, planning, and impulse control (Amlung et al., Reference Amlung, Marsden, Holshausen, Morris, Patel and MacKillop2019). Some studies suggest steeper discounting among adolescents with mood symptoms, while others report weak or inconsistent associations after comorbidities, socioeconomic context, and task parameters are considered (Peters & Büchel, Reference Peters and Büchel2011).
Risk-taking paradigms also show variability in relation to internalising symptoms. Anxiety is sometimes linked to risk avoidance in safe contexts, yet under social-evaluative stress or heightened arousal, risk behaviour can shift in the opposite direction; depressive symptoms have also been linked to altered sensitivity to reward and loss, though findings are not consistent across samples and designs (Hankin et al., Reference Hankin, Snyder, Gulley, Schweizer, Bijttebier, Nelis and Vasey2016). These patterns suggest that ‘task effects’ may be conditional depending on affective context, stress exposure, sample characteristics, and how outcomes are operationalised underscoring the need to map how tasks are implemented and interpreted across studies.
Beyond methodological inconsistency, the global relevance of this evidence base remains a central concern. The feasibility and validity of neuropsychological tasks depend on cultural context, language, stimulus familiarity, and the extent to which tasks can be adapted without changing the construct being measured. This issue is particularly salient because most adolescents live in low- and middle-income countries, where the burden of adolescent mental health needs is substantial and where scalable, low-cost assessment tools are of high practical value.
Yet cross-cultural cognitive research has cautioned that task performance is shaped by educational context, cultural norms, and test familiarity, emphasising the importance of careful adaptation and transparent reporting when paradigms are transferred across settings (Ardila, Reference Ardila2005). For self-regulation tasks in particular, adaptation may involve changes to facial-stimulus sets, reward magnitudes and currencies, instructions, and response devices each of which can influence performance metrics and comparability across studies. Mapping where and how these tasks have been used, in which languages, and with what types of adaptations is therefore essential for assessing the generalisability of conclusions and for identifying priorities for future research.
Taken together, these considerations point to a clear need for a structured synthesis that goes beyond whether tasks have been used and instead examines how they have been operationalised, which populations have been studied, and what kinds of designs have been employed. We therefore conducted a scoping review to address three questions: (1) How have the EGNG, DDT, and BART paradigms been applied in studies of adolescent depression and anxiety? (2) What populations, languages, and country contexts have been represented, and where do major geographic gaps remain? (3) To what extent have these tasks been used in longitudinal and intervention research, and how consistently have outcomes been defined and reported? By mapping the evidence base across these domains, the review aims to clarify the current state of task-based self-regulation research in adolescent internalising psychopathology and to identify methodological and geographic priorities for strengthening comparability, interpretability, and global relevance.
In this review, we use ‘self-regulation’ as an umbrella term for separable component processes that are operationalised differently across task families. EGNG paradigms primarily index affective interference control/response inhibition under emotional distraction, DDT tasks index temporal choice and valuation (preference for delayed versus immediate rewards), and BART tasks index risk-taking under uncertainty, which can be sensitive to arousal and social-evaluative context. Convergence across tasks would therefore be reflected not by uniform behavioural deficits, but by consistent symptom-related patterns within these domains (e.g., greater emotion-driven interference in EGNG, more present-focused choice in DDT, or context-dependent risk modulation in BART).
Methods
Search strategy and study selection
We searched MEDLINE (via PubMed), PsycINFO, and Scopus from database inception to 15 December 2025 (initial search: 1 December 2023; updated: 15 December 2025). The strategy combined controlled vocabulary (e.g., MeSH/thesaurus terms) and free-text keywords across three concept blocks: (i) adolescents (10–19 years), (ii) depression and/or anxiety, and (iii) neuropsychological self-regulation tasks (EGNG, DDT, BART and variants). Searches were adapted to each database using appropriate Boolean operators, field tags, and (where applicable) limits/filters. Records were imported into EndNote and de-duplicated. Prior to screening, a calibration exercise on 20 records was conducted to harmonise eligibility criteria and task definitions. Titles/abstracts and full texts were screened independently in duplicate, with disagreements resolved by consensus; inter-rater agreement across screening stages was high (κ = 0.87). OSF registration number: https://doi.org/10.17605/OSF.IO/P4RHB.
Inclusion and exclusion criteria
Studies were eligible if they: (a) included adolescents aged 10–19 years; (b) assessed depressive and/or anxiety symptoms using a clinical diagnosis or validated self-report instrument; and (c) reported behavioural outcomes on at least one of the following paradigms: EGNG, DDT, or BART or BART-Y. Studies using the BART with monetary rewards were included alongside youth-adapted versions BART-Y because both paradigms share the same task structure sequential risk-taking under increasing uncertainty with incremental gains and probabilistic loss. Differences across studies primarily reflect reward framing rather than task mechanics. Accordingly, these tasks are treated as indexing comparable processes relevant to risk-taking in children and adolescents for assessing depression or anxiety. Eligible studies were peer-reviewed journal articles published in English and reported sufficient statistical information for data charting. Studies were excluded if they included only adults; did not report extractable task outcomes; used non-standard task variants that precluded comparison (e.g., auditory Go/No-Go paradigms); or otherwise lacked the information needed to map task implementation and findings.
Critical appraisal
Methodological quality was appraised using the Joanna Briggs Institute critical appraisal checklists for observational and experimental study designs (JBI, 2020). Appraisal results were used to support interpretation of the evidence base. Consistent with the purpose of a scoping review, results were synthesised narratively. Although some studies reported means and standard deviations, outcome metrics were too heterogeneous for quantitative meta-analysis. This limitation was acknowledged and the OSF preregistration was updated to reflect a purely narrative synthesis.
Results
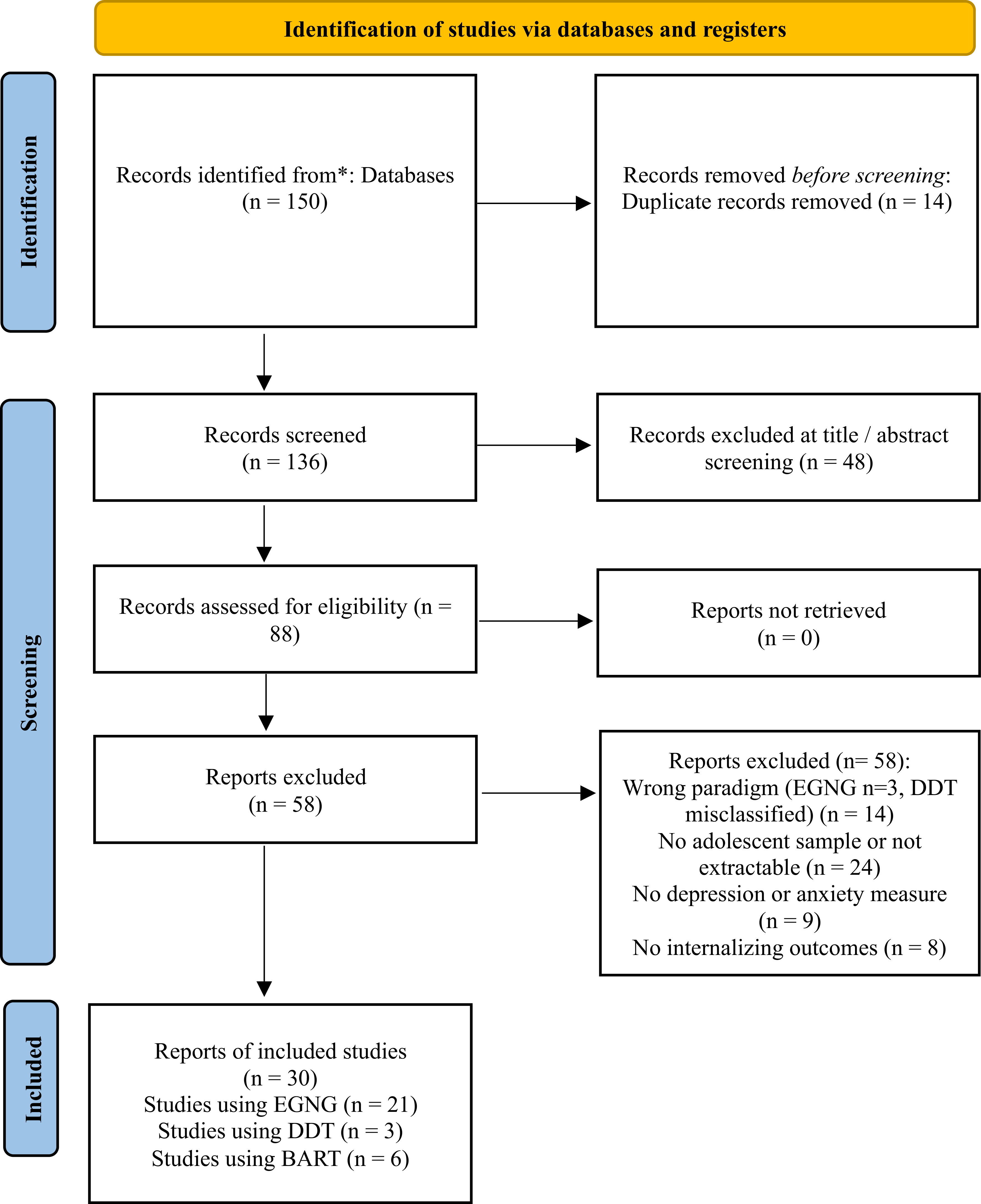
The literature search process followed a structured and systematic approach to identify relevant studies for inclusion in this review. The initial search across three major databases yielded a total of 150 records. Following the removal of 14 duplicate entries, 136 records remained for title and abstract screening. Of these, 48 records were excluded for failing to meet the basic inclusion criteria, leaving 88 reports for full-text assessment. No reports were unretrievable.
During full-text review, 58 reports were excluded. The most common reasons for exclusion were the absence of an adolescent sample or non-extractable adolescent data (n = 24), use of an incorrect paradigm (EGNG n = 3, DDT misclassified n = 14), and the absence of a depression or anxiety measure (n = 9). A further 8 reports were excluded because they did not report internalising outcomes.
Thirty reports met all inclusion criteria and were included in the final synthesis. The full study selection process, from identification through inclusion, is summarised in the PRISMA flow diagram (Figure 1). Twenty-six of the included reports were conducted in high-income settings, most commonly in the United States (k = 14; plus one multi-country sample involving the USA: USA–Lebanon, k = 1), with additional studies from the United Kingdom (k = 4), Canada (k = 3), Australia (k = 2), Germany (k = 1), and the Netherlands (k = 1). Three reports were conducted in an upper-middle-income setting (mainland China; k = 3), and one report was conducted in a lower-middle-income setting (Vietnam; k = 1). Task administration was predominantly in English (k = 24), with additional reports in Mandarin/Chinese (k = 3), German (k = 1), Dutch (k = 1), and Vietnamese (k = 1). Most studies used cross-sectional/case–control designs (k = 22), with fewer longitudinal/prospective studies (k = 5; EGNG k = 3, DDT k = 1, BART-Y k = 1); one longitudinal EGNG study used a pre–post CBT design. No intervention study using a validated DDT paradigm in adolescents with depression/anxiety was identified.
Diagram of study selection and inclusion.

Associational findings of the Emotional Go/No-Go task with anxiety and depression
Table 1 summarizes the findings by task and study. Across 21 EGNG-related reports in the included literature spanning emotional/affective Go/No-Go paradigms and closely related affective control tasks (Kyte et al., Reference Kyte, Goodyer and Sahakian2005; Ladouceur et al., Reference Ladouceur, Dahl, Williamson, Birmaher, Axelson, Ryan and Casey2006; Hare et al., Reference Hare, Tottenham, Galvan, Voss, Glover and Casey2008; Waters & Valvoi, Reference Waters and Valvoi2009; Pan et al., Reference Pan, Batezati-Alves, Almeida, Segreti, Akkal, Hassel, Brent and Phillips2011; Hum, Reference Hum2012; Han et al., Reference Han, Klimes-Dougan, Jepsen, Ballard, Nelson, Houri, Kumra and Cullen2012; Maalouf et al., Reference Maalouf, Clark, Tavitian, Sahakian, Brent and Phillips2012; Cohen-Gilbert & Thomas, Reference Cohen-Gilbert and Thomas2013; Cohen Kadosh et al., Reference Cohen Kadosh, Heathcote and Lau2014; Trinkl et al., Reference Trinkl, Greimel, Bartling, Grünewald, Schulte‐Körne and Grossheinrich2015; Colich et al., Reference Colich, Foland-Ross, Eggleston, Singh and Gotlib2016; Zhang et al., Reference Zhang, Feng and Mai2016; Chuang et al., Reference Chuang, Schwartz and Casey2017; Stoycos et al., Reference Stoycos, Del Piero, Margolin, Kaplan and Saxbe2017; Ho et al., Reference Ho, Dai, Mak and Liu2018; Connell et al., Reference Connell, Danzo, Magee and Dawson2020; Salem et al., Reference Salem, Fristad, Arnold, Taylor, Frazier, Horwitz and Findling2020; Porteous et al., Reference Porteous, Tavakoli, Campbell, Dale, Boafo and Robillard2023; Thomas et al., Reference Thomas, Jones, Williams and Vanderwert2025; Chen et al., Reference Chen, Chen, Wu, Luo, Cai, Yu and Wang2025), the evidence base was dominated by cross-sectional/case–control designs (k = 18), with three longitudinal studies including one pre–post CBT study (Hum, Reference Hum2012; Ho et al., Reference Ho, Dai, Mak and Liu2018; Salem et al., Reference Salem, Fristad, Arnold, Taylor, Frazier, Horwitz and Findling2020).
Eight of the 21 studies reported at least one statistically significant behavioural association or case–control difference between task performance and depression or anxiety, whereas 13 studies reported null behavioural differences or associations. Of the three longitudinal studies, none demonstrated an association: the pre–post CBT study in clinically anxious youth, reported no behavioural differences by emotion or trial type (Hum, Reference Hum2012), a prospective concussion cohort (Ho et al., Reference Ho, Dai, Mak and Liu2018) and another cohort (Salem et al., Reference Salem, Fristad, Arnold, Taylor, Frazier, Horwitz and Findling2020) similarly reported no consistent behavioural performance differences or associations over time.
When significant associations were observed, they tended to be emotion- or context-specific, rather than reflecting generalised inhibitory-control deficits. For depression outcomes, studies most consistently identified mood-congruent negative bias or affective capture. Kyte et al. (Reference Kyte, Goodyer and Sahakian2005) reported that adolescents with a recent first episode of major depressive disorder differed from controls on measures of emotional processing and decision-making, but not attentional set-shifting. Specifically, the depressed group demonstrated a negative emotional bias on the Affective Go/No-Go task, with fewer errors for sad targets and a significant valence × group interaction, alongside altered decision-making characterised by faster responding at highly favourable odds and increased betting at less favourable ratios. Task performance was not significantly related to depressive symptom severity or episode severity, indicating that observed group effects did not parallel severity levels.
Similarly, Ladouceur et al. (Reference Ladouceur, Dahl, Williamson, Birmaher, Axelson, Ryan and Casey2006) found that emotional context modulated cognitive-control performance differently in anxiety versus depression. While no diagnostic group differences emerged for accuracy or false alarms, depressed youth showed faster responses to sad Go targets under moderate target-probability conditions, whereas anxious youth showed slower responses to neutral Go targets when inhibiting responses to angry No-Go faces under low-probability conditions. Maalouf et al. (Reference Maalouf, Clark, Tavitian, Sahakian, Brent and Phillips2012) also demonstrated a state-dependent negative bias in youth with acute major depressive disorder, who responded faster when shifting toward negative relative to positive targets; this effect was absent in remitted youth and healthy controls and was not explained by symptom severity, illness history, comorbid anxiety, or medication status. One additional case–control study reported broader behavioural differences in a depressed clinical group, including greater reaction-time variability and reduced discriminability on an EGNG task (Chen et al., Reference Chen, Chen, Wu, Luo, Cai, Yu and Wang2025).
In anxiety, significant findings most often reflected threat-context interference or emotional-load effects rather than global inhibitory deficits. Waters and Valvoi (Reference Waters and Valvoi2009) reported gender-specific anxiety-related differences, with anxious girls showing greater interference when neutral Go trials were paired with angry versus happy No-Go distractors, while no comparable anxious–control differences were observed among boys and anxiety severity was not associated with task performance. Trait anxiety was also linked to altered attentional control in non-clinical samples, including associations with No-Go accuracy under emotional distraction (Cohen-Gilbert & Thomas, Reference Cohen-Gilbert and Thomas2013) and faster disengagement from emotional faces interpreted as attentional avoidance (Cohen Kadosh et al., 2014). Finally, one prospective cohort study reported faster Go responding in adolescents with elevated depressive symptoms following concussion, without consistent differences in inhibitory accuracy (Ho et al., Reference Ho, Dai, Mak and Liu2018).
Associational findings of the Delay Discounting Task with anxiety and depression
DDT studies included two cross-sectional (Imhoff et al., Reference Imhoff, Harris, Weiser and Reynolds2014; Anderson & Stanger, Reference Anderson and Stanger2023) and one longitudinal cohort (Ho et al., Reference Ho, Dang and Odum2023). Two studies reported at least one statistically significant association between delay discounting and depression or anxiety (Imhoff et al., Reference Imhoff, Harris, Weiser and Reynolds2014; Ho et al., Reference Ho, Dang and Odum2023). No intervention or treatment study using a validated DDT paradigm in adolescents with depression or anxiety was identified.
Associations for depression or anxiety were reported in terms of steeper delay discounting (i.e., a stronger preference for smaller, immediate rewards). The direction of symptom associations varied by design and context. In a cross-sectional clinical case–control study, Imhoff et al. (Reference Imhoff, Harris, Weiser and Reynolds2014) reported a modest but significant positive association between delay discounting (preference for small, immediate rewards) and depressive symptoms. Depressed adolescents discounted future rewards more steeply than non-depressed peers, and depressed non-smokers showed discounting patterns comparable to both depressed and non-depressed smokers, suggesting that elevated depressive symptoms were associated with impulsive choice behaviour independent of smoking status. At the symptom level, greater delay discounting was correlated with higher depression scores, supporting a link between depressive symptomatology and preference for immediate rewards.
By contrast, findings from the longitudinal school-based cohort in Vietnam of 404 adolescents completing two assessments six months apart suggested a prospective association in the opposite direction (Ho et al., Reference Ho, Dang and Odum2023). Steeper delay discounting (greater preference for small, immediate rewards) at baseline predicted lower depression and anxiety symptoms six months later, even after controlling for baseline symptom levels. In addition, higher baseline depressive symptoms predicted reductions in delay discounting over time, whereas baseline anxiety did not predict later discounting.
In adolescents with type 1 diabetes, Anderson and Stanger (Reference Anderson and Stanger2023) found no direct cross-sectional association between delay discounting and symptoms of anxiety or depression. However, greater delay discounting was associated with poorer glycemic control, and anxiety/depression symptoms moderated this relationship such that the association between discounting and metabolic outcomes was strongest among adolescents with elevated internalising symptoms.
Associational findings of the Balloon Analogue Risk Task with depression and anxiety
Across the six BART reports in the included literature, study designs included three cross-sectional studies (Hulvershorn et al., Reference Hulvershorn, Hummer, Fukunaga, Leibenluft, Finn, Cyders and Brown2015; Suo et al., Reference Suo, Zhang, Yin, Liu and Wang2025; Wang et al., Reference Wang, Evans and Schweizer2025), two longitudinal studies (Tieskens et al., Reference Tieskens, Buil, Koot and van Lier2021; Tran et al., Reference Tran, Rao, Bower, Fuligni and Kuhlman2025), and one within-subject experimental stress manipulation (Reynolds et al., Reference Reynolds, Schreiber, Geisel, MacPherson, Ernst and Lejuez2013). Four studies reported a statistically meaningful association between BART performance and anxiety or depression (Reynolds et al., 2013; Tieskens et al., Reference Tieskens, Buil, Koot and van Lier2021; Suo et al., Reference Suo, Zhang, Yin, Liu and Wang2025; Tran et al., Reference Tran, Rao, Bower, Fuligni and Kuhlman2025).
When associations were observed, they were context-, development-, and design-dependent, and effects for anxiety pointed in opposite directions depending on arousal and evaluative demands (Hengen & Alpers, Reference Hengen and Alpers2021). In a longitudinal BART-Y study, higher anxiety symptoms predicted reduced risk-taking over time, indexed by fewer balloon pumps, consistent with increasing risk avoidance across development (Tieskens et al., Reference Tieskens, Buil, Koot and van Lier2021). In contrast, the experimental stress manipulation study demonstrated that adolescents with higher social anxiety exhibited increased risk-taking under acute social-evaluative stress, reflected by more balloon explosions, whereas adolescents with lower social anxiety showed no stress-related change in behaviour (Reynolds et al., 2013).
Additional context-dependent anxiety effects were observed in two experimental studies conducted in China. Suo et al. (Reference Suo, Zhang, Yin, Liu and Wang2025) reported that peer presence increased risk-taking (average number of pumps on the unexploded balloons) in adolescents with low trait anxiety, whereas adolescents with high trait anxiety showed no comparable peer-presence effect. In the study’s second experiment, adolescents with high trait anxiety demonstrated greater risk-taking (more pumps on unexploded balloons) when observed by an unfamiliar peer relative to a familiar peer. These findings indicate that anxiety status moderated the influence of social context on BART performance rather than exerting a uniform main effect.
Evidence for depression-related associations was more limited and indirect. In a prospective longitudinal study, Tran et al. (Reference Tran, Rao, Bower, Fuligni and Kuhlman2025) found that BART-derived reward-seeking moderated the association between early life adversity and later anhedonia, such that greater adversity predicted higher anhedonia only at low or average levels of reward-seeking (i.e., low numbers of pumps on unexploded balloons); no direct main effect of reward-seeking on depressive symptoms was observed. By contrast, depressive symptoms were not directly associated with risk-taking in a cross-sectional social-context BART study (Wang et al., 2025), and a case–control neuroimaging study did not report a direct behavioural association between depression or anxiety symptoms and BART risk-taking indices (Hulvershorn et al., 2015).
Discussion
This scoping review synthesised evidence from 30 reports examining three neuropsychological task families: EGNG and closely related affective inhibitory-control paradigms, DDT, and BART, in relation to adolescent depression and anxiety. The included literature was dominated by studies conducted in high-income settings (26/30; 87%) and by English-language task administration (24/30; 80%), with comparatively limited representation from upper-middle-income settings (3/30; 10%; all from China) and one lower-middle-income setting (1/30; 3%; Vietnam).
Summary of EGNG findings
Across the EGNG-related adolescent literature, behavioural associations with depression and anxiety were inconsistent and generally small, with many studies unable to demonstrate differences in accuracy and reaction-time profiles across depression and anxiety symptom groups or diagnostic categories. Several case–control and neuroimaging/EEG studies reported no behavioural performance differences despite assessing depression or anxiety clinically or dimensionally (e.g., Beesdo et al., Reference Beesdo, Bittner, Pine, Stein, Höfler, Lieb and Wittchen2009; Pan et al., Reference Pan, Batezati-Alves, Almeida, Segreti, Akkal, Hassel, Brent and Phillips2011; Hum, Reference Hum2012; Chuang et al., Reference Chuang, Schwartz and Casey2017; Trinkl et al., 2015). When significant effects were observed, they most often reflected emotion- or context-specific biases rather than generalised inhibitory-control deficits.
In depression, findings consistent with mood-congruent processing included better performance for sad targets in a word-based affective Go/No-Go task (Kyte et al., Reference Kyte, Goodyer and Sahakian2005) and faster responding to sad Go trials in a mixed clinical sample (Ladouceur et al., Reference Ladouceur, Dahl, Williamson, Birmaher, Axelson, Ryan and Casey2006), without corresponding differences in error rates. These patterns suggest selective attentional capture by negative stimuli rather than broad impairments in inhibitory control or response accuracy. Such findings align with broader evidence that negative attentional biases and difficulty disengaging from aversive stimuli are core features of depression and anxiety (Joormann, Reference Joormann2010; Nolen-Hoeksema et al., Reference Nolen-Hoeksema, Wisco and Lyubomirsky2008). State-dependent negative bias was also indicated by faster shifting toward negative versus positive targets in acutely depressed adolescents, an effect absent in remitted youth (Maalouf et al., Reference Maalouf, Clark, Tavitian, Sahakian, Brent and Phillips2012). Importantly, this effect was specific to emotional valence and illness state rather than reflecting uniform deficits across stimulus conditions.
In anxiety, significant effects most consistently reflected threat-context interference, including slower responding on neutral Go trials when angry faces served as No-Go stimuli (Ladouceur et al., Reference Ladouceur, Dahl, Williamson, Birmaher, Axelson, Ryan and Casey2006) and slowed responses to neutral targets embedded within threatening No-Go contexts (Waters & Valvoi, Reference Waters and Valvoi2009), typically without accompanying accuracy differences. These findings suggest that anxiety-related alterations in performance emerge most clearly under conditions of heightened emotional or threat-related load rather than as pervasive control deficits.
More pronounced behavioural impairments were evident primarily in more severe or distinct clinical presentations. For example, adolescents with bipolar disorder during a current depressive episode showed slower and more variable reaction times and increased impulsive errors, highlighting the moderating role of clinical severity and task demands in detecting EGNG-related performance differences (Chen et al., Reference Chen, Chen, Wu, Luo, Cai, Yu and Wang2025). A recurring pattern across EGNG studies was a dissociation between behaviour and biology, with several reports documenting marked neural or psychophysiological differences (e.g., EEG, fMRI) in the absence of behavioural effects (e.g., Trinkl et al., 2015; Chuang et al., Reference Chuang, Schwartz and Casey2017). This pattern is consistent with compensatory recruitment or altered processing efficiency during task performance, although compensatory mechanisms were not directly evaluated in most studies.
In adult samples, affective Go/No-Go paradigms similarly demonstrate mood-congruent emotional biases in depression alongside relatively preserved accuracy and selective behavioural effects rather than generalised inhibitory deficits (Murphy et al., Reference Murphy, Sahakian, Rubinsztein, Michael, Rogers, Robbins and Paykel1999). In adult anxiety, threat-related interference and reduced top-down control over threat distractors are also well documented (Bishop et al., Reference Bishop, Duncan, Brett and Lawrence2004), and neuroimaging studies frequently report pronounced neural abnormalities in the context of subtle or absent behavioural differences during implicit emotion regulation (Etkin et al., Reference Etkin, Prater, Hoeft, Menon and Schatzberg2010), consistent with the behaviour–biology dissociation observed here.
Summary of DDT findings
Evidence for delay discounting was based on three reports and showed mixed associations across design and context. In a clinical case–control study, depression was associated with steeper discounting (i.e., a greater preference for smaller, sooner rewards), independent of smoking status (Imhoff et al., Reference Imhoff, Harris, Weiser and Reynolds2014). The sole longitudinal DDT study, conducted in Vietnam, reported an opposite prospective pattern, with steeper discounting predicting lower depression and anxiety symptoms one year later and baseline depressive symptoms predicting reduced discounting over time (Ho et al., Reference Ho, Dang and Odum2023). This finding underscores the importance of developmental, contextual, and cultural moderation and suggests that delay discounting may index adaptive decision-making strategies in certain environments rather than a unitary marker of impulsivity or vulnerability to internalising symptoms. Taken together, the evidence supports interpreting delay discounting as a context-sensitive indicator of self-regulation rather than a uniform correlate of adolescent depression or anxiety.
In adult samples, meta-analytic evidence indicates steeper delay discounting in major depressive disorder relative to controls (Amlung et al., Reference Amlung, Marsden, Holshausen, Morris, Patel and MacKillop2019), broadly aligning with adolescent clinical case–control findings. Associations between discounting and anxiety in adults are more heterogeneous, with reviews emphasising population- and context-specific variability rather than consistent effects, paralleling the mixed adolescent evidence summarised here.
Haushofer and Fehr (Reference Haushofer and Fehr2014), in On the Psychology of Poverty, argued that economic scarcity imposes cognitive and emotional burdens that constrain self-regulatory capacity, promoting present-focused decision-making. This framework offers a useful lens for interpreting divergent adolescent DDT findings, particularly in lower-resource contexts where immediacy preference may reflect adaptive coping rather than dysregulated impulsivity.
Summary of BART findings
Research using BART in relation to adolescent internalising symptoms was sparse (six studies) and did not reveal a consistent association between depression or anxiety and laboratory risk-taking. Four studies reported a statistical association. The two studies reporting null effects were an Australian cross-sectional sample (Wang et al., 2025), and a US case–control neuroimaging study (Hulvershorn et al., 2015).
Where anxiety-related associations were observed, their direction and magnitude varied by context. In a Dutch longitudinal community study using the BART-Y, higher anxiety symptoms predicted reduced risk-taking over time, consistent with increasing risk avoidance across development (Tieskens et al., Reference Tieskens, Buil, Koot and van Lier2021). In contrast, a US within-subject stress manipulation showed that adolescents with higher social anxiety exhibited increased risk-taking under acute social-evaluative stress compared with low-stress conditions (Reynolds et al., 2013). Additional experimental evidence indicated that anxiety moderated the influence of peer context on BART performance, with risk-taking varying as a function of trait anxiety and peer presence or familiarity rather than showing uniform main effects (Suo et al., Reference Suo, Zhang, Yin, Liu and Wang2025). Depression-related associations with BART performance were more limited and indirect (c.f., Tran et al., Reference Tran, Rao, Bower, Fuligni and Kuhlman2025). Collectively, these findings suggest that anxiety-related risk regulation is potentially context-sensitive, whereas depression-related effects on BART performance are not robustly detectable.
In adult samples, BART findings more often indicate reduced risk-taking in depression, consistent with heightened loss sensitivity rather than globally elevated risk-taking (Elliott et al., Reference Elliott, Sahakian, Herrod, Robbins and Paykel1997; Dillahunt et al., Reference Dillahunt, Feldman, Thomas, Farstead, Frandsen, Lee and Westlund Schreiner2022; Follett et al., Reference Follett, Hitchcock, Dalgleish and Stretton2023). Adult anxiety findings also commonly reflect risk avoidance, although stress and task framing can meaningfully modulate behaviour, mirroring the context sensitivity observed in adolescent studies (Maner et al., Reference Maner, Richey, Cromer, Mallott, Lejuez, Joiner and Schmidt2007).
Cross-task synthesis and interpretation
Across task families, two broad patterns emerged. First, EGNG and BART paradigms typically yielded small or context-dependent behavioural associations with depression and anxiety, with significant effects appearing primarily under specific stimulus conditions (EGNG) or contextual and developmental demands (BART). Second, DDT was used in fewer studies but more consistently demonstrated associations, although their direction varied, and the sole longitudinal study suggests potential cultural and contextual influences on prospective relationships. Overall, the evidence does not support consistent replicable of global inhibitory-control or risk-taking deficits in adolescents with internalising symptoms when using the EGNG, BART, or DDT. Instead, task sensitivity appears to depend on population, symptom severity and clinical composition, task parameters (e.g., emotional load, probability structure, stress manipulation), and study design.
Implications and gaps
This review reveals three key gaps. First, longitudinal evidence is limited overall and unevenly distributed across task families, constraining inference about developmental timing and directionality. Second, intervention and treatment studies are scarce, limiting conclusions about task sensitivity to clinically meaningful change. Third, the concentration of studies in high-income, English-speaking contexts highlights the need for broader evaluation of these paradigms across diverse cultural and socioeconomic settings, alongside clearer reporting of stimulus translation and adaptation, an issue underscored by limited research with non-English administrations and divergent longitudinal patterns observed in Vietnam.
Conclusion
The adolescent literature reviewed here indicates that EGNG and BART paradigms most often yield selective, context-dependent behavioural associations with depression and anxiety, whereas DDT more consistently shows association, with notable contextual differences. Together, these findings support the use of neuropsychological tasks primarily as mechanistic probes often in conjunction with other objective measures rather than as standalone behavioural markers of adolescent depression or anxiety. This review underscores the need for more longitudinal, intervention-focused, and culturally diverse research to clarify when and for whom task-based measures of self-regulation are most informative.
Neuropsychological studies using the Emotional Go/No-Go task (EGNG), the Delay Discounting Task (DDT) or the Balloon Analogue Risk Task (BART) in adolescents assessed for depression or anxiety

CANTAB = Cambridge Neuropsychological Test Automated Battery; ID/ED = Intra-/Extra-Dimensional set-shifting; MDD = Major Depressive Disorder; CBT = Cognitive Behavioural Therapy; RCT = randomised controlled trial; T1D = type 1 diabetes; DSM-IV = Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition; DSM-5 = Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition; ICD-10 = International Classification of Diseases, 10th Revision; K-SADS = Kiddie Schedule for Affective Disorders and Schizophrenia; K-SADS-PL = Schedule for Affective Disorders and Schizophrenia for School-Age Children—Present and Lifetime Version; MFQ = Mood and Feelings Questionnaire; CBCL = Child Behaviour Checklist; SCARED/SCARED-P = Screen for Child Anxiety Related Emotional Disorders (child/parent report); CDI/CDI-2 = Children’s Depression Inventory (2nd edition); BDI/BDI-II = Beck Depression Inventory (II); ADIS-C = Anxiety Disorders Interview Schedule for DSM-IV—Child Version; SCAS-C/SCAS-P = Spence Children’s Anxiety Scale (Child/Parent); MASC = Multidimensional Anxiety Scale for Children; STAI/STAI-C = State–Trait Anxiety Inventory (Child version); TAI = Trait Anxiety Inventory (trait subscale of the STAI); STAIC-S = State–Trait Anxiety Inventory for Children—State scale; YSR = Youth Self-Report; SCL-90 = Symptom Checklist-90; SIQ = Suicidal Ideation Questionnaire; SIS = Suicide Intent Scale; C-CASA = Columbia–Classification Algorithm of Suicide Assessment; SHF = Columbia Suicide History Form/Lethality Rating Scale; SBQ-R = Suicidal Behaviours Questionnaire–Revised; RCADS-25 = Revised Child Anxiety and Depression Scale (25-item); Kinder-DIPS = Diagnostic Interview for Mental Disorders in Children and Adolescents; DIKJ = Depression Inventory for Children and Adolescents; SSS-V = Sensation Seeking Scale—Form V; HAMD-17 = 17-item Hamilton Depression Rating Scale; YMRS = Young Mania Rating Scale; DASS-21 = Depression Anxiety Stress Scales–21; SPAI-23 = Social Phobia and Anxiety Inventory–23; PANAS = Positive and Negative Affect Schedule; UPPS-P-C = UPPS-P Impulsive Behaviour Scale—Child version; SCID = Structured Clinical Interview for DSM-IV; RADS-2 = Reynolds Adolescent Depression Scale—2nd Edition (Anhedonia subscale); ACE = Adverse Childhood Experiences questionnaire.
* Note. Studies using the Balloon Analogue Risk Task (BART) with monetary rewards were included alongside youth-adapted versions (BART-Y) because both paradigms share the same task structure sequential risk-taking under increasing uncertainty with incremental gains and probabilistic loss. Differences across studies primarily reflect reward framing rather than task mechanics. Accordingly, these tasks are treated as indexing comparable processes relevant to risk-taking in children and adolescents for assessing depression or anxiety.
Author contributions
Conceptualization: GE, MS, MG, LK, CL, MJDJ, BAK; Methodology: GE; Analysis: GE; Data curation; GE; Writing – original draft: GE; Writing – review & editing: MS, MG, LK, CL, MJDJ, BAK; Visualization: GE; Supervision: LK, BAK; Project administration: CL, MJDJ; Funding acquisition: CL, MJDJ. All authors have read and approved the final version of the manuscript.
Funding statement
This work was supported by the NIHR (using the UK’s Official Development Assistance Funding) and Wellcome (grant number:221940/Z/20/Z) under the Department of Health and Social Care (DHSC)–Wellcome Partnership for Global Health Research. The views expressed are those of the authors and not necessarily those of the Wellcome Trust, NIHR or the DHSC.
Competing interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Ethical standards
This article does not contain any studies with human participants or animals performed by any of the authors. The authors assert that all procedures contributing to this work comply with the ethical standards of the relevant institutional guidelines and with the Helsinki Declaration of 1975, as revised in 2008.