1. Introduction
The cambium is a major meristem in terrestrial ecosystems (Larson, Reference Larson1994), leading to the formation of wood, which constitutes one of the main terrestrial carbon pools (Cuny et al., Reference Cuny, Rathgeber, Frank, Fonti and Mäkinen2015). Climate warming is intensifying aridification, thus reducing tree growth worldwide (Babst et al., Reference Babst, Bouriaud, Poulter, Trouet, Girardin and Frank2019), which limits the potential to mitigate CO2 emissions of seasonally dry forests. This is the case of Mediterranean forests, where trees respond to drought through bimodal radial growth with peaks during warm-wet seasons, i.e. spring and autumn (Camarero et al., Reference Camarero, Olano and Parras2010; Cherubini et al., Reference Cherubini, Gartner, Tognetti, Bräker, Schoch and Innes2003). However, we lack a mechanistic understanding of how bimodal growth responds to carbon availability and drought stress, which is required to assess long-term changes of carbon uptake in seasonally dry forest ecosystems.
Bimodal growth patterns reflect the sensitivity of radial growth to seasonal climate conditions and have been mainly reported for conifers (but see Camarero et al., Reference Camarero, Serra-Maluquer, Colangelo, Gazol and Pizarro2024), including isohydric (‘water saver’) pines (e.g., Pinus halepensis Mill., Pinus pinaster Ait.) and also anisohydric (‘water spender’) junipers (e.g., Juniperus phoenicea L., Juniperus thurifera L.) (Liphschitz et al., Reference Liphschitz, Lev-Yadun, Rosen and Waisel1984; Valeriano et al., Reference Valeriano, Gutiérrez, Colangelo, Gazol, Sánchez-Salguero, Tumajer, Shishov, Bonet, Martínez de Aragón, Ibáñez, Valerio and Camarero2023). Bimodality may be a facultative phenomenon driven by site climate conditions (e.g., aridity, continentality), species-specific xylogenesis or year-to-year shifts in spring and autumn precipitation (Campelo et al., Reference Campelo, Ribas and Gutiérrez2021; Pacheco et al., Reference Pacheco, Camarero and Carrer2016, Reference Pacheco, Camarero, Ribas, Gazol, Gutierrez and Carrer2018; Tumajer et al., Reference Tumajer, Shishov, Ilyin and Camarero2021; Valeriano et al., Reference Valeriano, Gutiérrez, Colangelo, Gazol, Sánchez-Salguero, Tumajer, Shishov, Bonet, Martínez de Aragón, Ibáñez, Valerio and Camarero2023). In many of these studies, bimodality is related to the formation of latewood intra-annual wood density fluctuations (IADFs), characterized by earlywood-like tracheids with wide lumens and thin cell walls formed within the latewood (Camarero et al., Reference Camarero, Gazol, Valeriano, Pizarro and González de Andrés2023; De Micco et al., Reference De Micco, Campelo, De Luis, Bräuning, Grabner, Battipaglia and Cherubini2016; Olano et al., Reference Olano, García-Cervigón, Arzac and Rozas2015; Vieira et al., Reference Vieira, Campelo and Nabais2009).
Bimodal growth and latewood IADFs are triggered by wet-cool conditions in late summer and early autumn, resuming cambial activity (carbon sink) (Collado et al., Reference Collado, Camarero, Martínez de Aragón, Pemán, Bonet and De-Miguel2018; Rozas et al., Reference Rozas, García-González and Zas2011), or by manipulating carbon allocation through girdling (Oberhuber et al., Reference Oberhuber, Gruber, Lethaus, Winkler and Wieser2017; Reference Oberhuber, Landlinger-Weilbold and Schröter2021). Because carbon sinks and sources are modulated by different cues, bimodality and IADF formation are not necessarily linked (Rigling et al., Reference Rigling, Bräker, Schneiter and Schweingruber2002; Wimmer et al., Reference Wimmer, Strumia and Holawe2000). For instance, Oberhuber et al. (Reference Oberhuber, Landlinger-Weilbold and Schröter2021) induced bimodality in a mountain Scots pine (Pinus sylvestris L.) stand, usually showing a spring growth peak (unimodal growth), by blocking phloem transport, thus shifting carbon availability and leading to the resumption of cambial activity above the girdle after about 2 weeks of treatment application. Thus, facultative bimodality offers an interesting setting to assess the relative roles played by carbon sinks and sources on radial growth in seasonally dry areas by manipulating carbon availability through girdling.
Girdling is widely used to study the allocation of photoassimilates and trade-offs between carbon sources and sinks (Rademacher et al., Reference Rademacher, Basler, Eckes-Shephard, Fonti, Friend, Le Moine and Richardson2019). Girdling involves the removal of bark, phloem and cambium, resulting in an abrupt shut-down of the carbon supply of new assimilates from the canopy to organs downstream of the girdle (Maunoury-Danger et al., Reference Maunoury-Danger, Fresneau, Eglin, Berveiller, François, Lelarge-Trouverie and Damesin2010). Girdling also induces the accumulation of non-structural carbohydrates (NSCs) above the girdle (Jordan & Habib, Reference Jordan and Habib1996), with this response weakening with the distance from the girdle (Goren et al., Reference Goren, Huberman, Goldschmidt and Janick2010). Girdling does not uniquely alter plant carbon function, as it also dries out the outer xylem rings, leading to a loss of hydraulic conductance and overall tree transpiration (De Schepper et al., Reference De Schepper, Steppe, Van Labeke and Lemeur2010; Domec & Pruyn, Reference Domec and Pruyn2008). This explains anatomical changes observed in conifers, including suppressed xylem production, enhanced phloem development, enlarged axial parenchyma cells and reduced size of conducting elements (Serkova et al., Reference Serkova, Tarelkina, Ivanova, Semenova and Novitskaya2024). The post-girdling increase in cell-wall thickness may be due to an increase in NSCs by accumulation of photosynthates above the girdle (Jordan & Habib, Reference Jordan and Habib1996; Winkler & Oberhuber, Reference Winkler and Oberhuber2017).
Here, we combine the long-term (2000–2022) assessment of IADF formation and its climate drivers with a short-term (one growing season) girdling experiment carried out on branches of Spanish juniper (J. thurifera L.) individuals inhabiting a semi-arid site. To monitor the impacts of girdling on carbon availability and radial growth dynamics, we analyzed NSC concentrations in sapwood and leaves and used automatic point dendrometers, respectively. Our aims were: (i) to monitor the impacts of girdling on branch diameter and NSC concentrations; and (ii) to test whether girdling triggered bimodality and latewood IADF production. We hypothesized that latewood IADF production depends on sufficient late-summer moisture availability (cf. Camarero et al., Reference Camarero, Gazol, Valeriano, Pizarro and González de Andrés2023). By focusing on seasonally dry forests and a tree species showing facultative bimodal growth and IADF formation, our general purpose is to contribute knowledge to disentangle the roles played by carbon sinks and sources on radial growth.
2. Materials and methods
2.1. Study site, tree species and climate data
We performed the experiment in the ‘Vedado de Peñaflor’ (41.7834° N, 0.7217° W, 374 m a.s.l.), a well-preserved mixed pine-juniper forest subjected to semi-arid climate conditions and located near Zaragoza city (north-eastern Spain). The dominant tree species is Aleppo pine (Pinus halepensis Mill.), with a mean basal area of 8.0 m2 ha-1, whereas arboreal (Spanish juniper, J. thurifera L.) or shrubby junipers (Phoenicean juniper, Juniperus phoenicea L.) are also frequent. Other shrub and sub-shrub species include: Pistacia lentiscus L., Rhamnus lycioides L., Salvia rosmarinus Spenn., Thymus spp., Genista scorpius (L.) DC., Cistus clusii Dunal, Globularia alypum L. and Ephedra nebrodensis Tineo.
According to data from the nearby Zaragoza-airport station (41.6600° N, 1.0044° W, 249 m a.s.l., period 1941–2024), the mean annual temperature and total annual precipitation are 14.8°C and 358 mm, respectively. The period with negative water balance goes from May to October, with a mean soil moisture content (measured at 10–15 cm depth, period 2007–2011) of 10.8% (Camarero et al., Reference Camarero, Olano and Parras2010, Reference Camarero, Rubio-Cuadrado and Gazol2021). Soil moisture content peaks from February to May (30–35%) and reaches its minimum values from July to September (5–10%). Soils are basic, with gypsum and marl forming the parent rock material. No anthropogenic disturbance (wildfire, thinning or logging) has affected the study site at least since the 1970s. The year 2022 was extremely dry and warm, with a total annual precipitation of only 222 mm (Supplementary Figure S1). In that year, winter precipitation, which is critical for tree radial growth in the study area (cf. Pasho et al., Reference Pasho, Camarero and Vicente-Serrano2012), was only 30% (20 mm) of the long-term average (68 mm).
Monthly climate data were obtained from the 4-km Terraclimate dataset (Abatzoglou et al., Reference Abatzoglou, Dobrowski, Parks and Hegewisch2018), which provides homogeneous, checked series for several variables such as mean maximum temperature (T max), mean minimum temperature (T min), precipitation (Prec.), soil moisture (SM), climate water deficit (CWD, as difference between precipitation and potential evapotranspiration) and vapor pressure deficit (VPD). Monthly reference evapotranspiration was calculated using the Penman Montieth equation (Allen et al., Reference Allen, Pereira, Raes and Smith1998). These data were highly representative of the study location and showed good correlations with local climate series. For instance, the series of August T max from Terraclimate and Zaragoza airport were highly correlated (r = 0.934, p < 0.0001, period 2000–2022). Local climate data recorded every hour (air temperature, precipitation) were obtained from an automated weather station (height = 1.5 m) situated in an open area in the study site (Campbell CR-1000, Campbell Sci., Logan, USA).
To assess drought severity, we obtained weekly and monthly values of the Standardized Precipitation-Evapotranspiration Index (SPEI), which accounts for the cumulative effects of climatic water balance. We used the 1-km2 Spanish SPEI dataset (period 1960-2023) available at the webpage https://monitordesequia.csic.es/ (Vicente-Serrano et al., Reference Vicente-Serrano, Tomas-Burguera, Beguería, Reig, Latorre, Peña-Gallardo, Luna, Morata and González-Hidalgo2017). The summer of 2022 was exceptionally dry since 1960, as indicated by the very low SPEI values (Supplementary Figure S1). The CWD values of the hydrological year (October 1 to September 30) were −843 mm in 2020–2021 and −1031 mm in 2021–2022, whereas the 2020–2025 average was −715 mm.
2.2. Experimental design
We selected five dominant, mature and apparently healthy J. thurifera individuals growing at the study site and showing similar diameters at 1.3 m (14–22 cm) and canopy height (5.1–5.5 m). On 12 June 2021, after the spring growth peak, we selected two branches located at mid canopy with a diameter ranging from 7 to 15 cm in each individual (Figure 1a). Branches were selected for girdling because we assumed they were autonomous units for carbon transfer and storage, and because the juniper species is protected in the study area, which precluded girdling the main stem due to protection measures. Branches corresponded to girdled and non-girdled (control) branches. One branch per individual was girdled, and the other remained as a control for a total of 10 branches (5*2). Branches were collected in early December 2022, i.e. 17 months after girdling, to analyze IADFs and the concentration of NSCs during 2022.
2.3. Dendrometer records and processing
Automatic point dendrometers (TOMST, Prague, Czech Republic) were used to monitor changes in branch diameter (‘branch ΔD’ hereafter) at a high temporal (15 min) and spatial (0.27 μm) resolution from April until November 2022. Onset of radial growth of J. thurifera occurs from mid to late April in the study area, according to xylogenesis data and dendrometer records (Camarero et al., Reference Camarero, Olano and Parras2010, Reference Camarero, Rubio-Cuadrado and Gazol2021). Dendrometers additionally registered air temperature, which was used in subsequent analyses. They were placed above the girdle and on the living phloem after carefully removing the dead bark to avoid hygroscopic effects related to bark presence. The sensor tip was placed at 11–12 cm from the girdle (Figure 1a). In addition, we placed band dendrometers (EMS DRS26, Brno, Czech Republic) at 1.3 m in the stems of five junipers to record changes in stem diameter with hourly resolution during a wet year (2020) and to compare them with those obtained for the 2022 dry year. Means were obtained for the period from January to October, encompassing the whole growing season.
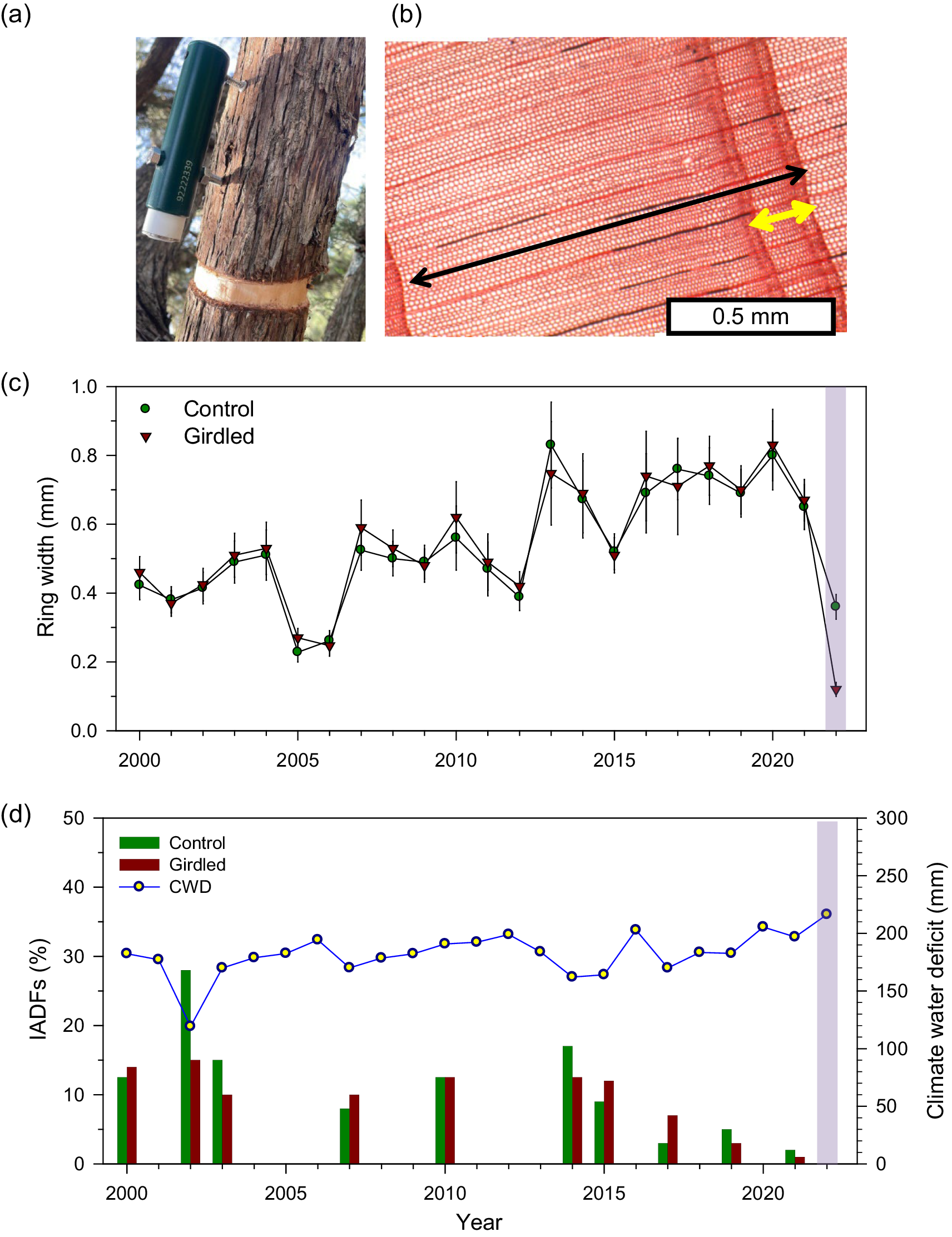
Views of (a) girdled branch with a point dendrometer attached in J. thurifera, (b) latewood IADF (yellow arrow) observed in the 2002 tree ring (black arrow), (c) ring-width data (means ± SD), and (d) frequency of IADF production in control (green bars) and girdled branches (brown bars; the boxes indicates the post-girdling year, 2022). The blue line shows the August climate water deficit (right y axis).

Dendrometer data were processed with the treenetproc package (Haeni et al., Reference Haeni, Knüsel, Wilhelm, Peters and Zweifel2020; Knüsel et al., Reference Knüsel, Peters, Haeni, Wilhelm and Zweifel2021) in R software (version 4.5.0; R Development Core Team, 2025). The package is based on the zero growth concept, which separates growth and water-related components of the dendrometer series assuming that growth begins when the previous maximum diameter is exceeded and ends when shrinkage occurs (Zweifel et al., Reference Zweifel, Zimmermann, Zeugin and Newberry2006, Reference Zweifel, Haeni, Buchmann and Eugster2016). Following a visual quality check, data pre-processing included: (i) excluding one dendrometer time series from controls due to persistent branch shrinkage and loss of contact with the stem surface, resulting in invalid zero readings, (ii) zeroing all time series to May 17 due to a dendrometer malfunction until this date. No further corrections were necessary. From the sub-hourly time series of branch ΔD, two daily transpiration-driven diameter (D) extremes were detected: daily D maxima (D max) were recorded around 6:00 a.m., while daily D minima (D min) were recorded around 3:00 p.m. (Supplementary Figure S4). Then, we calculated the daily time series of (i) branch (nocturnal) swelling as the difference between (midday) D min and the following day (predawn) D max, and (ii) the branch (diurnal) shrinkage as the difference between D max and D min within a day.
2.4. Ring-width data, IADF records and wood density
After sampling, branch cross-sections were air-dried, sanded with different sandpaper grain until rings were visible and scanned at a resolution of 2,400 dpi (Epson Expression 10,000). Two radii of each branch from opposite sides were visually cross-dated under the stereomicroscope from the bark to the pith (Fritts, Reference Fritts1976). The width of the annual rings was measured with 0.001 mm of resolution on scanned images using the CDendro-CooRecorder program (Maxwell & Larsson, Reference Maxwell and Larsson2021). The validation of the visual cross-dating process was undertaken by using the COFECHA software that compares the correlation between the individual series of ring-width and the mean sites series (Holmes, Reference Holmes1983). The IADFs frequency was obtained by counting their presence along the two cross-dated radii of each branch under the stereomicroscope. Only rings with IADF presence in both radii were considered to calculate an annual frequency (%) of IADFs for the common period 2000–2022. This was done by summing the observed IADFs and calculating relative frequencies with respect to the measured branches every year. The IADFs were formed in the mid to late wood (Figure 1b and Supplementary Figure S2). The IADF frequency was standardized and detrended by fitting linear regressions and keeping the resulting residuals.
To quantify the wood density of J. thurifera, we sampled 10 (length 15–20 cm, diameter 5–7 cm) branches from the upper canopy of five individuals (two branches per individual). A similar protocol was followed for the coexisting Juniperus phoenicea to compare both species. Wood density was estimated using the water displacement for volume and oven drying of samples at 70°C for 72 h for mass (Fajardo, Reference Fajardo2025).
2.5. NSC concentrations
The concentrations of starch and soluble sugars in the branch sapwood and leaves of girdled and non-girdled branches were measured. Although phloem has higher NSC concentrations than xylem, sapwood and leaves are by far the largest tree NSC reservoir due to their greater volume and biomass (Simard et al., Reference Simard, Giovannelli, Treydte, Traversi, King and Frank2013). Wood and leaf samples (ca. 50 g) were placed in a cooler and taken immediately to the laboratory, where they were freeze-dried and milled to a fine powder in a Retsch M400 ball mill (Haan, Germany) prior to analysis. Soluble sugars (SS) were extracted with 80% (v/v) ethanol in a water bath at 60°C, and their concentration was determined colorimetrically using the phenol–sulphuric method (Buysse & Merckx, Reference Buysse and Merckx1993). The starch and complex sugars that remained in the undissolved pellet after the ethanol extraction were enzymatically reduced to glucose using amyloglucosidase (0.5% amyloglucosidase 73.8 U/mg, Fluka 10115) and analyzed following Palacio et al. (Reference Palacio, Maestro and Montserrat-Martí2007). NSCs measured after ethanol extraction are referred to as soluble sugars, and those measured after enzymatic digestion are referred to as starch. The sum of soluble sugars and starch is referred to as total non-structural carbohydrates (NSC).
2.6. Data analyses
The R software (version 4.5.0; R Development Core Team, 2025) was used for data analyses. Mann–Whitney U tests were used to compare inter-annual growth rates, IADF production and NSC concentrations between control and girdled branches, given that the percentage of IADFs did not follow a normal distribution. Spearman correlations were calculated between the annual frequency of IADFs and August climate variables based on previous analyses on xylogenesis and dendrometer data (Camarero et al., Reference Camarero, Olano and Parras2010, Reference Camarero, Rubio-Cuadrado and Gazol2021, Reference Camarero, Gazol, Valeriano, Pizarro and González de Andrés2023).
We assessed the amplitude of daily shrinkage and swelling cycles in branches using linear mixed-effects models (lme4 package; Bates et al., Reference Bates, Mächler, Bolker and Walker2015). To account for the potential lack of normality, dendrometer data were log (x + 1) transformed. The initial model included treatment (girdling vs. control) as a fixed effect, with random intercepts for tree and branch nested within tree. As rain events appeared to influence the daily shrinkage-swelling amplitude, we subsequently fitted models that incorporated the interaction between treatment and rain, considering rain as a qualitative (rainy vs. non-rainy day) or a quantitative (rain amount) fixed effect. We assumed a normal distribution of the model residuals and homoscedasticity, which were verified using diagnostic plots (Santos Nobre & Singer, Reference Santos Nobre and Singer2007). Additionally, we used generalised additive mixed models (GAMMs) to explore the shape of the relationship between rainfall amount and shrinkage-swelling amplitude (mgcv package; Wood, Reference Wood2011). These models allowed for separate, flexible (non-linear) responses for each treatment group, along with random intercepts for tree and branch.
3. Results
3.1. Growth, IADF formation and wood density
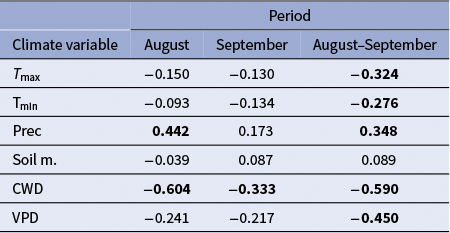
We built a robust ring-width chronology for the common period 2000–2022. The mean ring width was 0.54 ± 0.04 mm (mean ± SE; control branches, 0.53 ± 0.04 mm; girdled branches, 0.55 ± 0.06 mm; U = 242, p = 0.62), and the mean correlation between ring-width series was 0.82 ± 0.11. Growth was strongly reduced in 2022 in girdled branches (Figure 1c), and this was confirmed by stem dendrometer records of the wet and dry 2020 and 2022 years, respectively (Supplementary Figure S3). In 2022, radial growth was significantly lower in girdled than in control branches (0.12 ± 0.01 vs. 0.36 ± 0.04 mm, U = 0.10, p = 0.01; Figure 1c). The annual frequency of latewood IADFs did not significantly differ (U = 260, p = 0.92) between control (mean ± SE = 4.87 ± 1.57 %) and girdled branches (4.22 ± 1.20 %; Figure 1d). Considering the IADF production of all branches across years, correlations with August were higher, in absolute terms, than those with September or August-September climate conditions (Table 1). IADF frequency was negatively correlated with the August water balance deficit (r = −0.604, p = 0.002), i.e. wetter August conditions enhanced IADF production. Regarding wood density, J. thurifera (mean ± SD) showed significantly lower values than J. phoenicea (0.57 ± 0.06 vs. 0.78 ± 0.09 g cm-3; U = 0.01, p = 0.001).
Spearman correlations calculated between the annual frequency of latewood IADFs and August, September and August–September climate variables (T max, mean maximum temperature; T min, mean minimum temperature; Prec, precipitation, Soil m., soil moisture; CWD, climate water deficit; and VPD, vapor pressure deficit).

Bold coefficients indicate significant correlations (p < 0.05) between IADFs and climate variables.
3.2. Stem radial responses to girdling
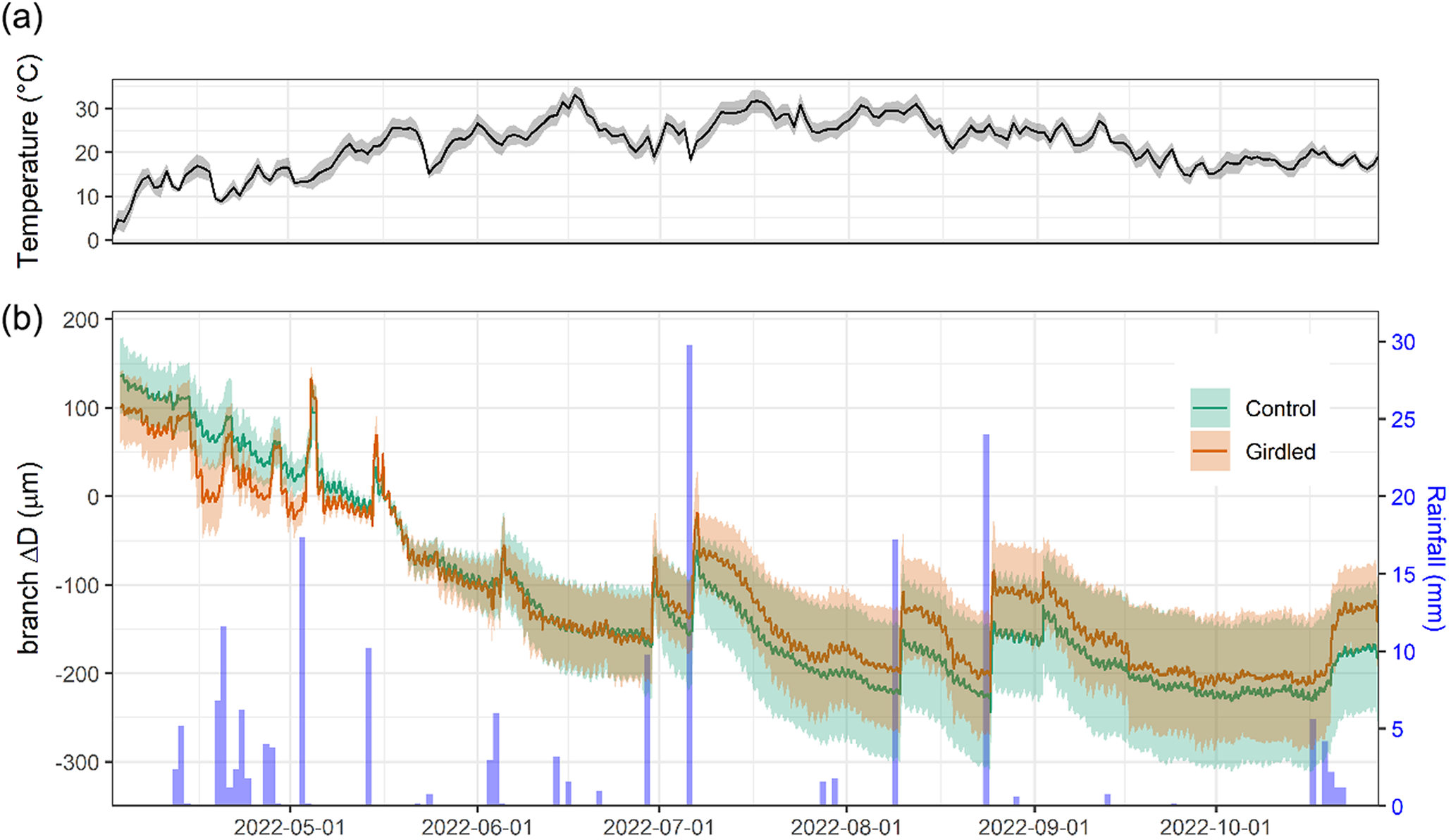
Throughout the period of branch ΔD monitoring in 2022, daily mean temperatures increased progressively, rising from 0°C in April to a peak of 33°C in mid-June, and remained above 20°C until mid-September (Figure 2a). Only 101 mm of rainfall was recorded from June to September (Figure 2b). None of the monitored branches showed net diameter increase, regardless of the girdling treatment (Figure 2b). Instead, we observed a progressive branch shrinkage, consistent with elastic fluctuations related to branch dehydration. Branch diameter reductions were periodically interrupted by sharp increases following rainfall events; however, these transient recoveries did not restore the initial diameter.
(a) Mean daily temperature and (b) variations in diameter measured in girdled (brown line) and control (green line) J. thurifera branches (means ± SEs) and daily rainfall records (blue bars, right y-axis) over the experimental period (from April to November 2022).

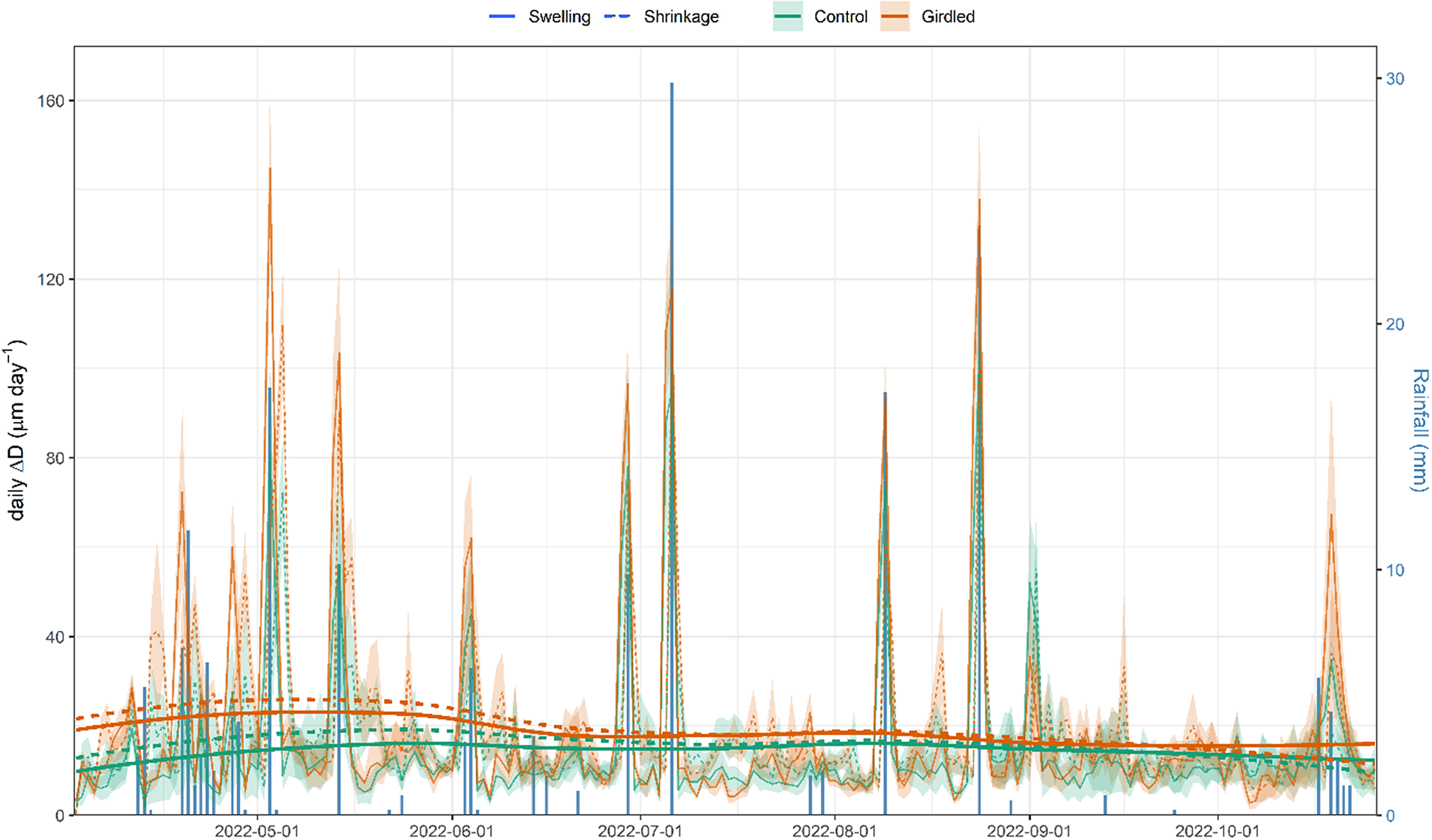
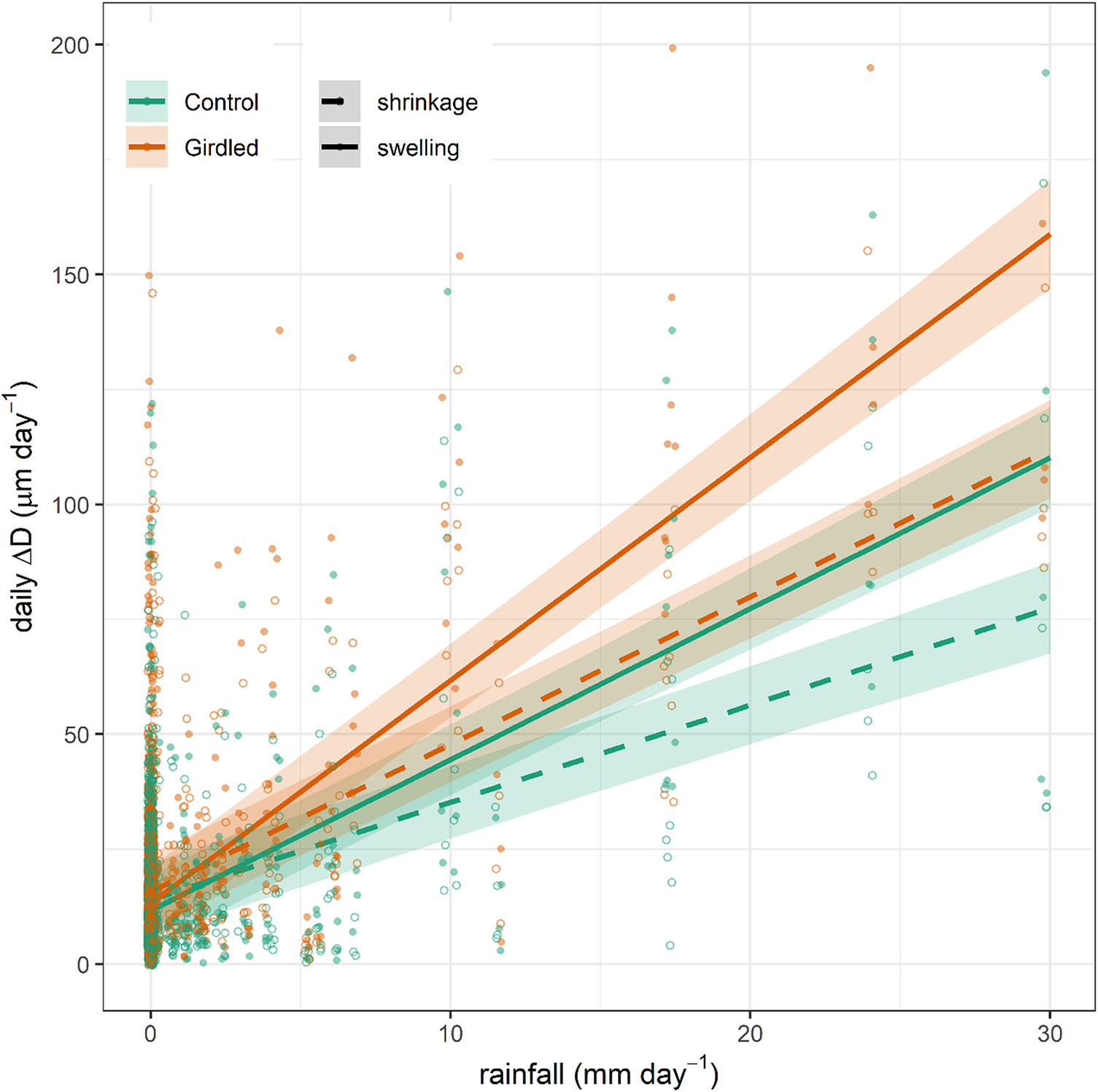
At the daily timescale, diurnal shrinkage tended to exceed nocturnal swelling, regardless of the girdling treatment (Figure 3), consistent with the net reduction in branch ΔD observed over the monitored period. The amplitude of these daily cycles typically remained below 30 μm day-1. However, rainfall events triggered substantial increases in their amplitude, with daily fluctuations reaching up to 140 μm day-1. Across the entire experimental period, treatment (girdling vs. control) did not significantly affect shrinkage (p = 0.34) or swelling (p = 0.25) amplitude. However, models incorporating rainfall revealed significant effects. Both shrinkage and swelling amplitudes were higher on rainy days compared to non-rainy days (p < 0.0001). Furthermore, the amplitude of both swelling and shrinkage increased significantly with the amount of rainfall (p < 0.0001). This rain effect was more pronounced in girdled branches than in control branches, as indicated by a significant treatment:rain interaction (p < 0.0001; Figure 4). The GAMMs confirmed significant effects of rainfall on both shrinkage and swelling (all smooth terms p < 0.0001). Visualization of the treatment effect (girdled–control) across the rainfall gradient revealed a consistently positive influence of girdling on both processes above ca. 5 mm of rain (Supplementary Figure S5). The pattern of this effect was non-linear (parabolic) for swelling, whereas it was more linear for shrinkage.
Daily patterns of branch diameter swelling and shrinkage (continuous and dashed lines) in girdled and control (brown and green colour) J. thurifera branches. Note: Lines and shaded ribbons show mean ± SE. Long-term trends were highlighted using locally estimated scatterplot smoothing (loess). Blue bars show rainfall values (right y-axis).

Linear relationships between rainfall amount and daily diameter swelling and shrinkage (continuous and dashed lines) in girdled and control (brown or green colour) branches of J. thurifera. Lines and shaded ribbons show means ± SE.

3.3. NSC concentrations in sapwood and leaves
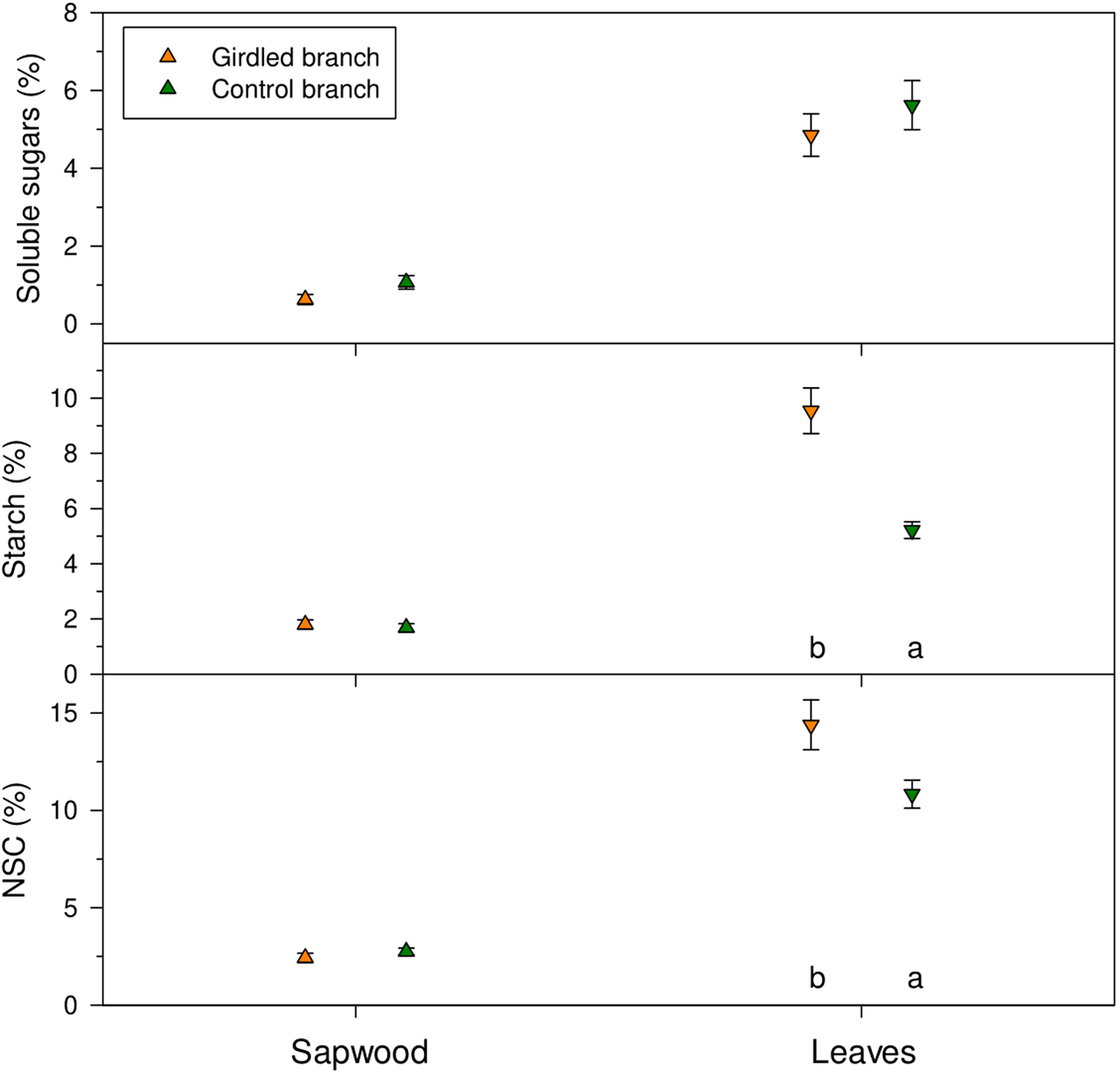
NSC concentrations were higher in leaves (ca. 12 %) than in sapwood (ca. 2 %) (Figure 5). In leaves, we found significantly (p < 0.05) higher starch (U = 1, p = 0.017) and NSC (U = 3, p = 0.037) concentrations in girdled than in control branches. In branch sapwood, we found no differences in soluble sugar (U = 4, p = 0.096) and NSC concentrations (U = 8, p = 0.360) between girdled and control branches.
Concentrations of non-structural carbohydrates measured in sapwood and leaves of girdled (brown symbols) and control (green symbols) J. thurifera branches. Values are means ± SE. Different letters indicate significant (p < 0.05) differences between treatments according to Mann–Whitney tests.

4. Discussion
Girdled and control juniper branches showed permanent and progressive shrinkage after installing the dendrometers in spring (Figure 2). Thus, according to the zero growth concept (Zweifel et al., Reference Zweifel, Haeni, Buchmann and Eugster2016), no radial growth occurred. Following this theoretical framework, branch diameter variations could be attributed to elastic shrinkage-swelling processes due to wood water depletion or replenishment, respectively. Nevertheless, we observed the formation of earlywood in 2022, which reflects limited growth during periods of branch water depletion and shrinkage, but also challenges the zero concept framework. Dendrometers did not record a net stem growth, regardless of girdling, but branches showed continuous shrinkage (Figure 2). This finding suggests that new xylem cells can be produced, even under dry conditions, and these growth processes may not be accurately recorded by point dendrometers, which only monitor a particular place of the stem and may be more sensitive to shrinking/swelling dynamics. We acknowledged the limitation of our data since the growth of the study species starts in April and the dendrometers were zeroed in mid-May. To address this issue, we plan to compare xylogenesis and dendrometer data in the future. The long-term evaluation of latewood IADF production pointed to the importance of late-summer (August) drought as previously reported (Camarero et al., Reference Camarero, Olano and Parras2010, Reference Camarero, Rubio-Cuadrado and Gazol2021, Reference Camarero, Gazol, Valeriano, Pizarro and González de Andrés2023; Salomón & Camarero, Reference Salomón and Camarero2025). The 2022 exceptionally dry conditions probably hampered IADF production regardless of girdling (Figure 1).
Girdling negatively impacted xylem growth above the girdled zone by blocking phloem transport and reducing cambium dynamics and xylem water flow, at least close to the girdle, as has been previously reported (Serkova et al., Reference Serkova, Tarelkina, Galibina, Moshchenskaya, Sofronova, Ivanova and Semenova2022, Reference Serkova, Tarelkina, Ivanova, Semenova and Novitskaya2024; Tombesi et al., Reference Tombesi, Day, Johnson, Phene and DeJong2014). Starch and NSC concentrations increased in leaves above the girdling, likely due to phloem blockage and accumulation of photosynthates above the girdled part of the branch (Rademacher et al., Reference Rademacher, Basler, Eckes-Shephard, Fonti, Friend, Le Moine and Richardson2019). Importantly, girdled branches were more responsive to rain pulses and showed a higher elastic capacity to rehydrate (and subsequently dehydrate) than control branches (Figure 4), with this effect saturating upon a certain rainfall amount threshold (Supplementary Figure S5). The enhanced amplitude of branch ΔD to rain pulses in girdled branches could not be attributed to higher concentrations of osmolytes (Blumstein et al., Reference Blumstein, Gersony, Martínez-Vilalta and Sala2023), because NSC content in xylem was not significantly different among controls and girdled branches (Figure 5). We speculate that abundant leaf starch reserves of gridled branches may be hydrolyzed into soluble sugars (Richardson et al., Reference Richardson, Carbone, Keenan, Czimczik, Hollinger, Murakami, Schaberg and Xu2013), which are then transported downstream via the phloem, accumulating above the girdled area. Their function as osmolytes could subsequently explain the more elastic rehydration response of girdled branches to rain pulses, as recorded by the dendrometers. Following these NSC allocation patterns, the observed ΔD signal is more likely attributable to swelling and shrinking in the living tissues beyond the cambium (phloem and bark) than those in the sapwood xylem. Notably, junipers showed rapid stem shrinkage after spring rainfall events under mild conditions and also after heavy rainfall events under warmer conditions in summer (Figure 2). These rapid responses could be explained by the hygroscopic nature of the low-density wood of J. thurifera, and its anisohydric (‘water spender’) behavior. However, the high variability in stem swelling/shrinkage dynamics between periods and sites depends on additional traits, in addition to wood density and stomatal closure, including water storage tissue size, elasticity and capacitance (Dietrich et al., Reference Dietrich, Zweifel and Kahmen2018; Salomón et al., Reference Salomón, Limousin, Ourcival, Rodríguez-Calcerrada and Steppe2017), but the underlying mechanisms remain poorly understood. In general, stem or branch shrinkage amplitude increases under dry conditions (elevated evaporative demand, limited soil water supply), but with sufficient internal water storage to allow water release from elastic tissues contributing to transpiration (Steppe et al., Reference Steppe, De Pauw, Lemeur and Vanrolleghem2006; Zweifel et al., Reference Zweifel, Item and Häsler2001). Further research is needed to link drivers such as the compressibility of the elastic storage tissues with functional or physiological traits such as wood density or stomatal conductance rates.
Girdling did not trigger the formation of latewood IADFs in 2022, a pattern which may be attributed to the dry conditions of the year. Inter-annual correlations between climate variables and IADF production indicate that the main trigger of IADF formation was water availability during summer (Figure 1, Table 1). This agrees with previous research on anisohydric species (cf. Voelker et al., Reference Voelker, DeRose, Bekker, Sriladda, Lecksungnoen and Kjelgren2018), including Mediterranean trees such as Aleppo pine (Campelo et al., Reference Campelo, Ribas and Gutiérrez2021; De Micco et al., Reference De Micco, Campelo, De Luis, Bräuning, Grabner, Battipaglia and Cherubini2016; Pacheco et al., Reference Pacheco, Camarero and Carrer2016, Reference Pacheco, Camarero, Ribas, Gazol, Gutierrez and Carrer2018). Therefore, enhanced water availability in late summer and autumn can increase the turgor of cambial derivatives and their radial enlargement rate, leading to the formation of tracheids with a wide lumen within the latewood (Collado et al., Reference Collado, Camarero, Martínez de Aragón, Pemán, Bonet and De-Miguel2018).
The extent to which this climatic signal on IADFs affects the carbon source and sink is still unknown. Previous research showed that girdling induced bimodal growth in Scots pine at a drought-prone Alpine valley, suggesting that belowground organs influence the carbon sink strength and hormonal signalling, albeit wetter climate conditions in this Alpine valley might have also played a role in enabling a second growth peak (Oberhuber et al., Reference Oberhuber, Gruber, Lethaus, Winkler and Wieser2017; Reference Oberhuber, Landlinger-Weilbold and Schröter2021). Our findings do not fully concur with this idea and reinforce the importance of climate stressors as drivers of radial growth, principally the impacts of water availability on major carbon sinks such as meristems (Körner, Reference Körner2015), particularly relevant on seasonally dry Mediterranean regions. At the study site, soil drought was the main constraint of stem radial growth according to previous studies comparing manual and automatic band dendrometer data (see Camarero et al., Reference Camarero, Rubio-Cuadrado and Gazol2021; Salomón & Camarero, Reference Salomón and Camarero2025). Moreover, in valley bottoms of semi-arid sites, where soil moisture and nutrient availability increase, the Spanish juniper grows more, forms more IADFs and reaches larger sizes, emphasizing the importance of water availability for anisohydric species again (Camarero et al., Reference Camarero, Gazol, Valeriano, Pizarro and González de Andrés2023).
To conclude, we assessed the climate drivers of latewood IADF formation and performed a girdling experiment in a facultative bimodal species from a semi-arid site. No IADF formation was found in girdled branches, and late-summer water availability appears as the main trigger of IADF formation. Precluded by the extreme dry summer following treatment application, girdling did not induce bimodal growth or IADF formation. Girdled branches were more elastic in response to rain pulses and presented higher starch concentrations in their leaves. The dependence of radial growth on carbon sinks (cambium) is supported by the presented long-term climate-IADF relationships.
Open peer review
To view the open peer review materials for this article, please visit http://doi.org/10.1017/qpb.2026.10037.
Acknowledgements
The authors thank E. Lahoz for performing the NSC analyses and J. Revilla for the maintenance of meteorological sensors and dendrometers.
Competing interest
The authors declare no competing interests.
Data availability statement
Data associated with this paper will be available on request from the corresponding author.
Author contributions
J.J.C., and A.G. conceived and designed the study; J.J.C., A.G., C.V., E.T. and M.C. conducted data gathering; J.J.C. and R.L.S. analyzed the data; J.J.C. led the writing of the manuscript; A.G., R.L.S., C.V., E.T., A.R.C. and M.C. contributed critically to the drafts. All authors have final approval for publication.
Funding statement
This work was funded by the Spanish Ministry of Science and Innovation (projects PID2021-123675OB-C43 and TED2021-129770B-C21).
Supplementary material
The supplementary material for this article can be found at http://doi.org/10.1017/qpb.2026.10037.



Comments
Zaragoza, 9th October 2025
Prof. Dr. Jesús Julio Camarero
Pyrenean Institute of Ecology
Spanish National Research Council (CSIC)
Avda. Montañana, 1005
50059 Zaragoza, Spain
Charlotte Crofts
Quantitative Plant Biology Editorial Office
quantplantbio@cambridge.org
Dear colleagues, here we submit the manuscript entitled “Girdling increases branch capacity to rehydrate in Juniperus thurifera” by J. Julio Camarero, Roberto L. Salomón, Antonio Gazol, Cristina Valeriano, Elisa Tamudo, Álvaro Rubio-Cuadrado and Michele Colangelo.
The manuscript is submitted as an “Original Research Article” to be included in the issue “TBC: For the ‘Advances in xylem and phloem formation research’ special collection”. In this study, we uncovered the drivers of intra-annual density fluctuations and bimodal radial growth through long-term correlative approaches and a short-term girdling experiment. The findings advance on our understanding about the relationships between carbon sinks and sources as controls of radial growth in trees.
Sincerely,
J.J. Camarero, on behalf of all coauthors