Early Development of the Placenta
Development of the placenta begins with cavitation of the morula, the compact sphere of cells, and transitions to the blastocyst stage of embryonic development. At this stage, the first lineage segregation takes place, with a single layer of cells on the outer part of the blastocyst forming the trophectoderm (TE), the precursor to trophoblast, the epithelial cells of the placenta[Reference Veeck and Zaninovic1]. Cells remaining in the middle of the blastocyst are referred to as the inner cell mass (ICM), which gives rise to all embryonic structures, as well as some extraembryonic structures (see below). TE cells have tight junctions, which are required for fluid accumulation within the blastocoel cavity. Unlike cells in the ICM, TE cells are also characterized by apico-basal polarity, a process that sets into motion signals leading to trophoblast lineage specification[Reference Veeck and Zaninovic1].
Once formed, the polar TE (cells nearest to the ICM) is responsible for attachment to the endometrium. Attachment is rapidly followed by invasion of the blastocyst into and below the uterine lining. Once embedded within the endometrium, the TE rapidly expands, giving rise to two cell populations: a mononuclear cytotrophoblast (CTB) and a multinucleated syncytium. The latter secretes enzymes that degrade the surrounding tissue and also tap into maternal sinusoids, finally giving rise to blood-filled lacunae that are lined by this syncytium. In the meantime, the mononuclear CTBs proliferate, forming a shell around the implanted blastocyst[Reference James, Carter and Chamley2] (Figure 1.1a). Invaginations of CTB within this shell begin the formation of primary villi, while the invasion of these structures with mesenchymal cells leads to the formation of secondary villi (Figure 1.1b).

Figure 1.1 Cross-section through a 4-week gestational age embryo. (a) Placenta (the cytotrophoblastic shell, or CTB) makes up a much greater proportion of tissue compared to the embryo-proper (E). (b) Secondary villi containing mesenchyme (M) surround lacunae (L) containing maternal blood.
Cytotrophoblast as the Trophoblast Stem Cell
Akin to stem cells in other stratified epithelia, such as skin, the cytotrophoblast (CTB) reside adjacent to villous stroma and sit atop a basement membrane. Similar to stem cells in these other organs, CTB express the N-terminal truncated isoform of p63 (∆Np63), an oncogene and member of the p53 nuclear protein family, which is lost quickly upon differentiation of CTB, as they move away from the underlying villous stroma[Reference Lee, Kim and McKeon3]. Other markers expressed in CTB include membrane proteins E-Cadherin, EGFR, and integrins alpha-6 and beta-4; as they are proliferative cells, they also express Ki67[Reference Benirschke, Burton and Baergen4].
Whether the proliferating CTB layer, or the TE from which it arose, contains true trophoblast stem cells (TSC) – cells which can give rise to all trophoblast subtypes – is not known. Unlike mouse TSC, which have been derived from both pre-implantation blastocysts and post-implantation extraembryonic tissues[Reference Tanaka, Kunath and Hadjantonakis5], human TSC have yet to be characterized or derived[Reference Chang and Parast6]. While there is not a clear consensus, data point to the early post-implantation villous placenta as the most likely niche for human TSC[Reference Chang and Parast6]. In addition to p63, at least a subset of CTB in 5–8 week gestation placentae also express CDX2, a homeobox-domain containing transcription factor, which defines TSC in mice; this CTB subset greatly diminishes by the end of the first trimester[Reference Horii, Li and Wakeland7]. Nevertheless, isolation and further characterization of this subpopulation, including its capacity to differentiate into all trophoblast subtypes (“multipotency”), have yet to be evaluated.
Implantation and Differentiation of Extravillous Trophoblast
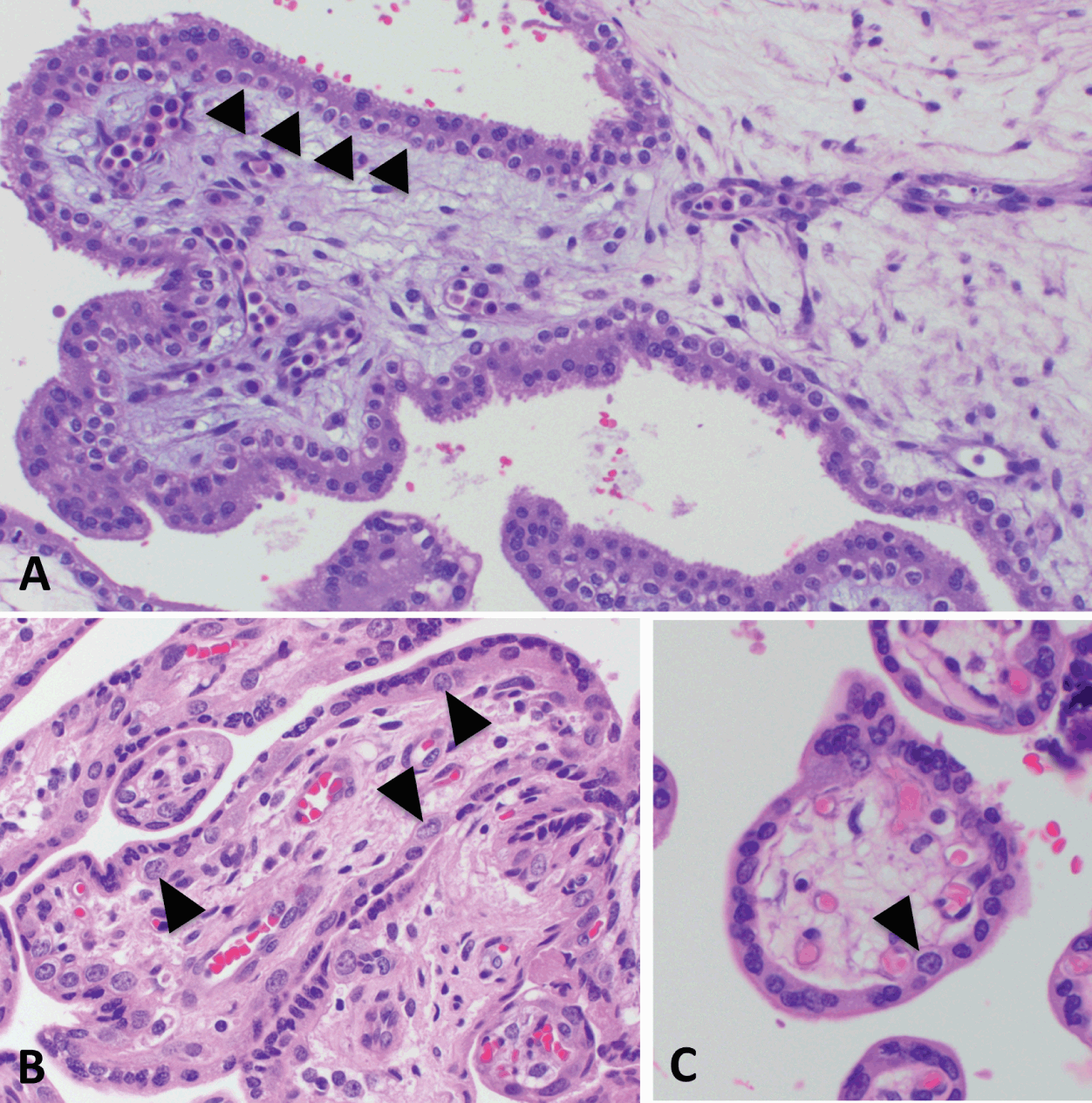
At the basal plate, anchoring chorionic villi extend cell columns towards the uterus, which serve to firmly attach the feto-placental unit to maternal tissues. At the proximal portion of the cell column, trophoblast proliferate, differentiating from CTB into extravillous trophoblast (EVT) as they move further away from the mesenchymal villous core (Figure 1.2). This transition is accompanied by changes in gene expression, including loss of p63, and gain of other transcription factors such as ASCL2. There are also changes in membrane protein expression, with loss of E-Cadherin, EGFR, and integrins alpha-6 and beta-4; and gain of MelCAM, the nonclassical histocompatibility antigens HLA-G, HLA-C, and HLA-E, and integrins alpha-5 and beta-1 (Figure 1.2)[Reference Lee, Kim and McKeon3][Reference Benirschke, Burton and Baergen4]. As the cells approach the uterine wall, they lose their tight epithelial morphology and begin to dissociate from the column, developing more elongated and mesenchymal morphology and gaining invasive potential. Distal cell column and mature EVT at the implantation site are characterized by integrins alpha-1 and beta-1; they retain expression of HLA-G and MelCAM[Reference Benirschke, Burton and Baergen4].

Figure 1.2 Early (8-week gestational age) implantation site. Anchoring villi (V) with trophoblast cell columns (CC) growing toward the uterine wall. The proximal CC (pCC) consists of p63+/Ki67+ cells, which are gradually lost in the distal CC (dCC), containing mature EVT which express HLA-G, among other markers.
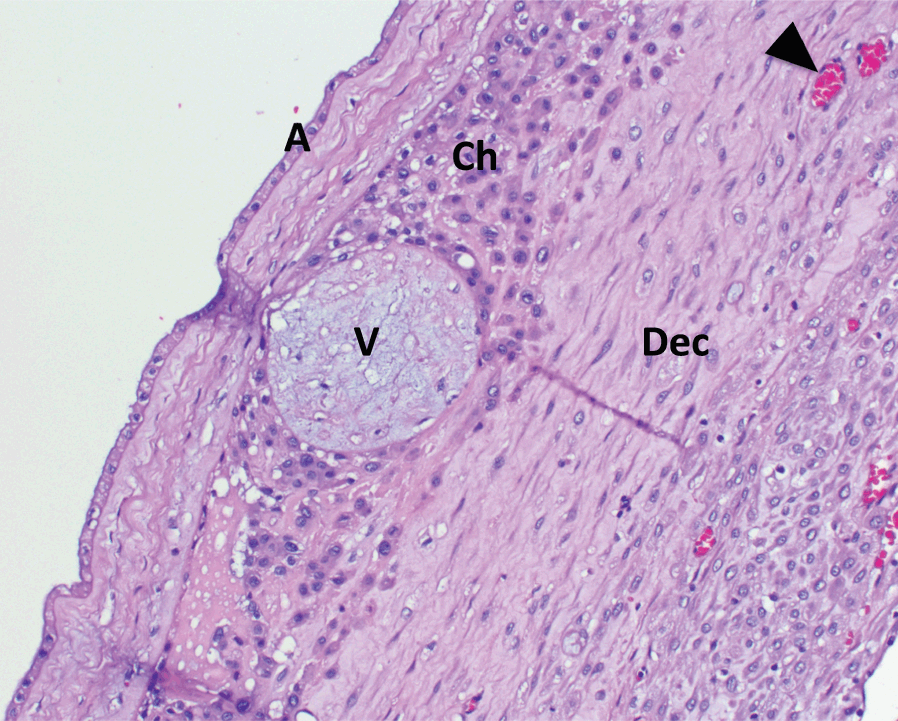
Mature EVT invade the uterine wall, either staying within the decidua or myometrium as interstitial EVT, or remodeling maternal vessels as endovascular EVT. Most mature EVT are mononuclear, though binucleate and multinucleate forms can occur (Figure 1.3a–b). EVT nuclei often appear irregular with prominent nucleoli and are surrounded by abundant eosinophilic-to-amphophilic cytoplasm. As these cells are fully differentiated, mitotic forms are rare. As interstitial EVT, these cells invade the uterine wall, singly or in groups, extending into the inner third of the myometrium. As endovascular EVT, they initially plug maternal spiral arterioles, then migrate and remodel these vessels, replacing maternal endothelium (Figure 1.3c–d). During this process, referred to as “physiologic conversion” or “physiologic change,” maternal spiral arterioles are transformed from high-resistance, low-capacitance vessels to low-resistance, high-capacitance vessels (compare Figure 1.3c to 1.3e–f). As a result of this conversion, these maternal vessels lose the ability to respond to maternal systemic blood pressure and instead continuously deliver a large percentage of maternal cardiac output toward the placenta[Reference Benirschke, Burton and Baergen4]. This process is vital to the continuous growth and development of the placenta (and therefore the fetus). Abnormal or incomplete conversion has been associated with a wide range of pregnancy complications, including preeclampsia, fetal growth restriction (FGR), and recurrent miscarriage. Recent data, however, point to re-endothelialization of the maternal arterioles following physiologic conversion; the consequences of this process are as yet unclear[Reference Benirschke, Burton and Baergen4].

Figure 1.3 EVT at the implantation site. Interstitial EVT can be either (a) mononuclear or (b) multinucleated. Endovascular EVT invade and remodel maternal spiral arterioles: (c) shows arterioles prior to remodeling, (d) an arteriole with an EVT “plug,” and (e) a vessel in process of remodeling, still retaining some smooth muscle in its wall. In (f), the remodeled vessel is lined by endovascular EVT (arrowheads).
During the implantation process, EVT interact with the maternal immune cells in the decidua, including macrophages and natural killer (NK) cells. Both of these cell populations are distinct from the respective cells at other sites, based on both marker expression and function, and hence are referred to as decidual/uterine macrophages and NK cells. Cross talk between EVT and decidual immune cells leads to secretion of various cytokines, including interferon-gamma from the latter cells, which modulates EVT invasion and regulates the physiologic conversion process[Reference Eastabrook, Hu and von Dadelszen8]. These interactions are vital to implantation, as lack of decidua (and thus the decidual immune cells) leads to invasive placentation (i.e. either in intrauterine pregnancy as in cases of placenta accreta, or extrauterine pregnancy as implantation within the Fallopian tube)[Reference Benirschke, Burton and Baergen4].
More recently, another group of immune cells, namely regulatory T cells, has been found to selectively migrate from peripheral blood into decidua during pregnancy[Reference Tilburgs, Roelen and van der Mast9]. These cells have a dampening effect on the immune response, promoting tolerance of the semi-allogeneic fetus by secretion of cytokines, including TGFb and IL-10[Reference Quinn and Parast10]. That they play a key role in promoting tolerance is demonstrated by a relative increase in their number in cases of fetal-maternal HLA-C mismatch[Reference Tilburgs, Scherjon and van der Mast11]. Additionally, their numbers are decreased at the maternal-fetal interface in pregnancies complicated by severe early-onset preeclampsia, a disease characterized by abnormal EVT differentiation and incomplete physiologic conversion of maternal arterioles[Reference Quinn and Parast10].
Chorionic Villi: Differentiation of Villous Trophoblast and Development of the Fetal Villous Tree
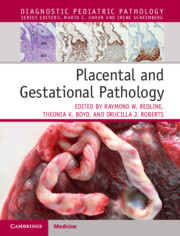
Chorionic villi are the functional units of the placenta. Villous formation begins with invagination of the primitive CTB layer around the blastocyst between 3 and 4 weeks of gestational age. Shortly thereafter, extraembryonic mesoderm (ExM), derived from the ICM, extends into the primitive villi, forming secondary villi (see Figure 1.1b). Between 5 and 6 weeks of gestational age, capillaries form within the villous core from the same ExM, thereby transforming these structure into tertiary villi (Figure 1.4). It is important to note that formation of fetal vessels within chorionic villi begins independently of vascularization within embryonic structures, including the umbilical cord, or cardiac development. Thus even anembryonic gestations can harbor primitive vessels within chorionic villi, though other signs of embryogenesis (such as infiltration of these spaces with nucleated red blood cells) will be absent.
First trimester villi consist of abundant mesenchyme surrounded by two continuous layers of trophoblast, the inner proliferative CTB layer, and the outer syncytiotrophoblast (Figure 1.4). Despite the name, the latter is considered distinct from the primitive syncytium, the layer of multinucleated cells which surround the early embryo and are responsible for initial implantation events. The primitive syncytium is more likely to be similar to implantation site EVT; in fact, some implantation site “giant cells” (see Figure 1.3b) may be remnants of the primitive syncytium. However, syncytiotrophoblast-proper arise by fusion of underlying CTB, a process that is mediated by the human endogenous retroviral gene products syncytins 1 and 2 (ERVW-1 and ERVFRD-1, respectively). Throughout gestation, CTB continue to proliferate, and subsequently fuse with and contribute to the overlying syncytiotrophoblast layer. However, since growth of the underlying stroma outpaces that of the trophoblast, over time, the trophoblast layer becomes thinner overall, and the CTB layer discontinuous (Figure 1.4)[Reference Simpson, Mayhew and Barnes12]. In the meantime, the nuclei of syncytiotrophoblast also undergo a maturation process, transitioning from an open, transcriptionally active euchromatin seen in nuclei of syncytial “sprouts,” common in the first half of pregnancy, to the more condensed chromatin seen in nuclei of true syncytial “knots,” aggregates which show evidence of oxidative damage and are common in post-term placentae[Reference Fogarty, Ferguson-Smith and Burton13]. Syncytiotrophoblast fragments are shed into maternal circulation as part of normal turnover of this tissue, a process that appears to be accelerated in some placental pathologies, including preeclampsia[Reference Mayhew14].
Following villous vascularization and formation of primitive tertiary villi, these structures undergo branching morphogenesis, a process by which chorionic villi develop from a group of relatively uniform structures, to ones where larger stem villi branch into intermediate and then into terminal villi, the primary sites of gas and nutrient exchange[Reference Benirschke, Burton and Baergen4]. Branching morphogenesis is a complex process that requires close interaction and coordination between trophoblast and ExM-derived mesenchyme and fetal vasculature. In trophoblast, this process is mediated by induction of glial cells missing-1 (GCM1), a transcription factor previously identified as the “master” switch for development of the labyrinth, the equivalent of chorionic villi in mice[Reference Cross, Nakano and Natale15]. In the human placenta, GCM1 in expressed in a subset of CTB and coordinates cell-cell fusion and syncytiotrophoblast formation, as well as branching morphogenesis[Reference Huppertz, Burton and Cross16]. Abnormalities in villous branching take many forms: they range from a decrease in properly formed terminal villi and thus gas and nutrient exchange interface in severe forms of preeclampsia and FGR, to the uncoupling of vasculogenesis and villous branching, resulting in excessive vessel formation in diabetic placentas.
Fetal Membranes
Initially after implantation, villous formation occurs concentrically in the CTB shell around the embryo (see Figure 1.1). However, beginning at 9 to 11 weeks gestation, villi furthest from the umbilical stalk begin to regress, forming the chorion laevae. At the same time, the free amniotic sac fuses with the chorionic mesenchyme. Later, between 18 and 20 weeks of gestation, as the gestational sac expands, these structures fuse with the endometrium opposite from the implantation site, leading to the placenta and the gestational sac occupying the entire uterine cavity[Reference Benirschke, Burton and Baergen4]. Subsequently, at term, fetal membranes are seen to consist of a layer of amnion with a variable amount of underlying amniotic mesenchyme, chorionic trophoblast often containing villus “ghosts,” and underlying decidua capsularis (Figure 1.5). Trophoblast of the chorion laevae (also called chorionic or membranous trophoblast) are considered extravillous trophoblast (EVT), insomuch as they are outgrowths of villi into surrounding matrix, similar to the process that occurs in anchoring villi at the implantation site. The decidua capsularis is often the most enriched source of maternal vessels; although not proven to be representative of maternal vessels in the decidua basalis (underlying the placental disc), vessels in this location are often used to evaluate for presence of lesions associated with hypertensive diseases of pregnancy (decidual arteriopathy and related lesions).
Intraplacental Trophoblast Islands
Another location where EVT arise from villus remnants is in intraplacental trophoblast islands within the placental disc (Figure 1.6). In this location, perivillous deposition of fibrin-type fibrinoid is part of the normal developmental process, which leads to degeneration of the syncytiotrophoblast, exposing the underlying CTB layer to the extracellular matrix. This leads to an induction of EVT differentiation, similar tooa the process in the fetal membranes and at the implantation site.
Extravillous Trophoblast: Terminology and Subtypes
Extravillous trophoblasts are referred to as intermediate trophoblast in a substantial portion of the placenta literature, probably because, when initially described, their morphology appeared to be “intermediate” between cytotrophoblast and syncytiotrophoblast. However, given our current knowledge of their biology – specifically, that they are a distinct lineage from villous syncytiotrophoblast – it is more accurate to refer to them as extravillous trophoblast (EVT).
EVT exist in several locations within the placenta: at the implantation site in the early gestation placenta and basal plate, in fetal membranes, and in intraplacental trophoblast islands. In all these locations, their differentiation is characterized by a transition from proliferative cytotrophoblast to a fully mature EVT. The more immature, “transitional” EVT is characterized by a vacuolated cytoplasm, and retention of p63 and Ki67, while “mature” EVT often has abundant eosinophilic-to-amphophilic cytoplasm, has fully lost expression of p63 and Ki67, and expresses HLA-G and MelCAM, among other markers[Reference Lee, Kim and McKeon3]. In the basal plate, basal plate-type immature (transitional) EVT are located in the vicinity of the anchoring villi and mature as they infiltrate the underlying decidua. In the fetal membranes, membranous-type immature (transitional) EVT are located in the layer of chorion closest to the amniotic mesenchyme, and they mature as they move closer to the decidua. In the intraplacental trophoblast islands, intraplacental type-immature (transitional) EVT are seen adjacent to villus remnants, located toward the center of the islands, with more mature EVT, seen toward the island periphery.
Knowledge Gaps
Large gaps remain in our knowledge of human placental development, including trophoblast differentiation. Both villous and extravillous trophoblast are likely to be much more heterogeneous than currently defined. Evaluation of this heterogeneity is required for identification of additional markers of trophoblast subtypes, which could then lead to a better characterization of differentiation defects in any given placental pathology. Similarly, better in vitro models of human trophoblasts, in co-culture with other cell types, and in 3D, are required to understand the placenta both during normal development and disease. Finally, little is known about development of this organ across gestation; therefore, establishment of noninvasive methods of evaluation, including imaging, with subsequent correlation with clinical and pathologic data, are needed to enhance our knowledge of this vital organ.