Introduction
The Beltana deposit, near Puttapa, is the largest of several willemite ore bodies discovered in the Flinders Ranges of South Australia in the late 1960s. Geochemical stream sediment sampling within Lower Cambrian sediments detected anomalies for lead (40 ppm to 400 ppm) and zinc (100 to 1300 ppm). Subsequent rock sampling and detailed mapping led to the discovery of outcrops of willemite at Beltana, Aroona and Third Plain (Johns, Reference Johns1972). Diamond drilling at Beltana located one major and several minor orebodies. Mining by opencut methods commenced in February 1974 and continued intermittently until 2003 (Emselle, Reference Emselle, McPhail and Welch2005). From a mineralogical point of view, the Beltana deposit is the most interesting. The willemite orebody is associated with hematite alteration, is characterised by the total absence of sulfides and contains high levels of arsenic (0.5 wt.%), lead (0.4–2.5 wt.%) and manganese (Groves et al., Reference Groves, Carman and Dunlap2003). Most of the arsenic and lead in the willemite ore are hosted in the mineral hedyphane, Ca2Pb3(AsO4)3Cl (Brugger et al., Reference Brugger, McPhail, Wallace and Waters2003). Supergene alteration has resulted in the formation of a suite of arsenate and vanadate minerals, such as adamite, austinite, mimetite, tsumcorite, descloizite (Elliott et al., Reference Elliott, Pring and Birch1988; Elliott, Reference Elliott1991) and puttapaite, the last of which is the subject of this paper.
Puttapaite was collected from ore stockpiles in the 1990s and was at the time recognised as a probable new mineral species based on the unique chemistry. The size and nature of the crystals, which occur as rosette-like aggregates composed of many individuals, has made structural characterisation difficult. The new mineral is named for the locality. The mineral and its name (symbol Ptp) have been approved by the Commission on New Minerals, Nomenclature and Classification of the International Mineralogical Association (IMA2020-025, Elliott and Kampf, Reference Elliott and Kampf2020). The holotype specimen has been deposited in the South Australian Museum, Adelaide, South Australia (Registration number G34869).
Occurrence
The Beltana deposit is a high-grade hypogene willemite deposit hosted in Lower Cambrian carbonate rocks in the northern Flinders Ranges (Groves et al., Reference Groves, Carman and Dunlap2003; Brugger et al., Reference Brugger, McPhail, Wallace and Waters2003). The willemite occurs as a replacement of dolomitised and hematitised Ajax Limestone of Lower Cambrian age. Mineralisation is structurally controlled and associated with brecciation and extensive hematite-rich hydrothermal zincian dolomitisation. The texture of the willemite is heterogeneous, resulting from various depositional mechanisms: direct replacement of carbonate host rock, open-space filling, internal sedimentation and brecciation. Acidic ore fluids corroded the host carbonate units and created open space by means of hydrothermal karsting and subsequent deposition of internal willemite sediment. Late-stage gangue minerals include Mn-rich calcite, dolomite and minor quartz. On the periphery of the deposit, smithsonite formed by weathering of willemite. Using numerical geochemical modelling, Brugger et al. (Reference Brugger, McPhail, Wallace and Waters2003) were able to show that willemite will precipitate at temperatures above 120°C as a result of water–rock interaction and fluid mixing processes. The mineralising fluids carried large quantities of oxidised arsenic, as demonstrated by the large amounts of hedyphane in the deposit. The presence of arsenate in the hydrothermal fluids is likely to have inhibited the oxidation of sulfate to sulfide and resulted in the stabilisation and precipitation of willemite rather than sphalerite and galena. Secondary arsenate minerals have formed in cavities in the willemite as a result of supergene alteration. A detailed description of the mineralogy is given by Elliott (Reference Elliott1991). The new mineral occurs in vugs in a matrix composed of willemite and hematite. Associated minerals are rhodochrosite, hedyphane and adamite.
Appearance and physical properties
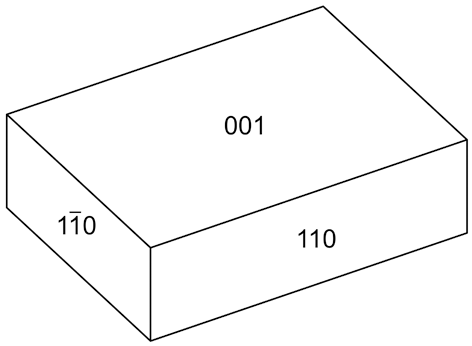
Puttapaite occurs as diamond-shaped tablets in rosette-like aggregates to 50 μm across (Figs 1 and 2). Individual tablets are up to 45 μm in length and 5 μm in thickness. Crystals are flattened on {001} and the observed forms are {001} and {110} (Fig. 3). The colour is pale green with a pale-green streak and a vitreous lustre. The tenacity is brittle, no cleavage was observed, and the fracture is splintery. Due to the small size of the crystals, the Mohs hardness could not be measured. A density of 3.562 g/cm–3 was calculated using the empirical chemical formula and unit-cell parameters from single-crystal data. Puttapaite is optically biaxial (–) with α = 1.700(5), β = 1.720(5) and γ = 1.730(5) (measured in white light). The 2V, measured on a spindle stage, using extinction data analysed with the program EXCALIBR (Gunter et al., Reference Gunter, Bandli, Bloss, Evans, Su and Weaver2004) is 67(1)°; the calculated 2V is 69.8°. Dispersion could not be observed. Crystals are pleochroic with X light blue grey, Y colourless and Z not observed; X > Y. The Gladstone–Dale compatibility, 1 – (K P/K C), (Mandarino, Reference Mandarino2007) is 0.047 (good) using the empirical formula and the unit-cell parameters determined from single-crystal data.
Pale-green crystals of puttapaite associated with smithsonite (orange brown) and willemite (white). The scale bar is 100 mm (specimen in private collection).

Scanning electron microscope photomicrograph showing crystals of puttapaite. The field of view is 0.3 mm (specimen in private collection).

Crystal drawing of puttapaite; clinographic projection in standard orientation.
Chemical composition
Chemical data (ten spot analyses) were obtained using a Cameca SXFive electron microprobe (wavelength dispersive spectroscopy mode, an acceleration voltage of 20 kV, a beam current of 20 nA and a 5 μm beam diameter). Raw X-ray intensities were corrected for matrix effects with a φ(ρZ) algorithm (Pouchou and Pichoir, Reference Pouchou, Pichoir, Quantitation, Heinrich and Newbury1991). Both the crystal structure and infrared spectroscopy data confirm the presence of H2O. Analytical data are given in Table 1. The empirical formula for puttapaite, calculated on the basis of 36 oxygen atoms, is Pb1.96(Mn2+1.52Ca0.28Sr0.22)Σ2.02(Zn0.40Mg0.39Cu0.15)Σ0.94(Cr3+2.89Al0.45Fe3+0.40,Mn3+0.26)Σ4.00O2[(AsO4)3.71(Cr6+O4)0.29]Σ4.00(OH)6.13·11.87H2O. The ideal formula is Pb2Mn2+2ZnCr3+4O2(AsO4)4(OH)6·12H2O, which requires PbO 26.20, MnO 8.33, ZnO 4.78, Cr2O3 17.84, As2O5 26.98, H2O 15.87, total 100 wt.%.
Analytical data for puttapaite
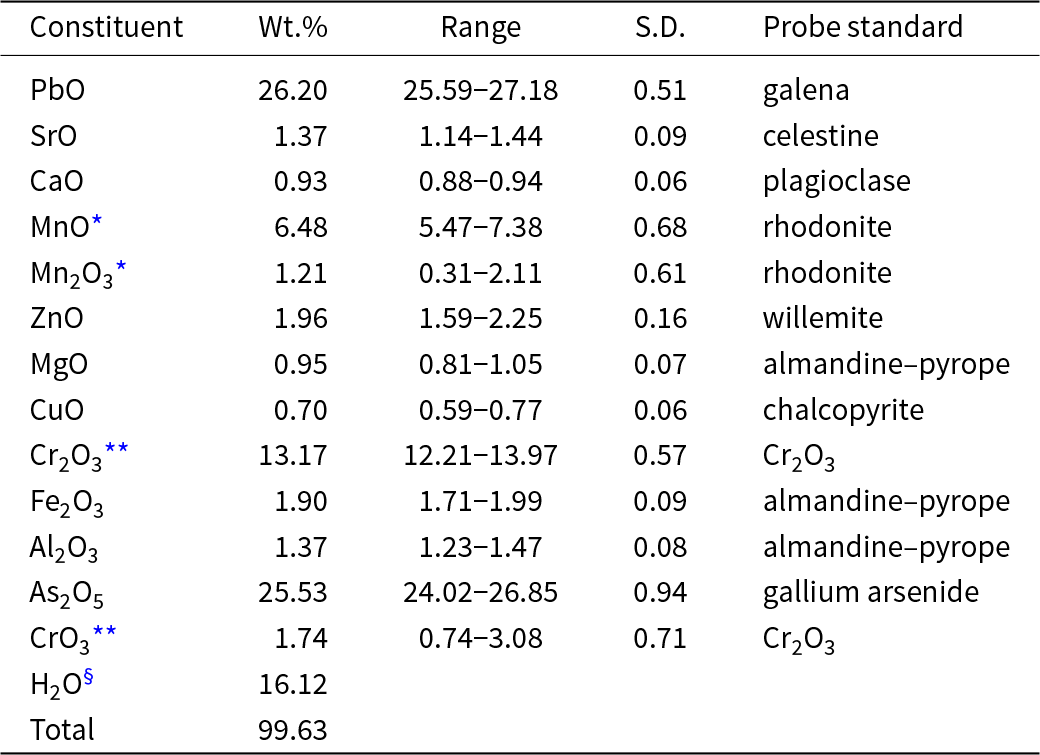
* MnO and Mn2O3 calculated to give Cr3+ + Al + Fe3+ + Mn3+ = 4.00
** Cr2O3 and CrO3 calculated to give AsO4 + Cr6+O4 = 4.00
§ calculated from the refined formula; S.D. – standard deviation
Infrared spectroscopy
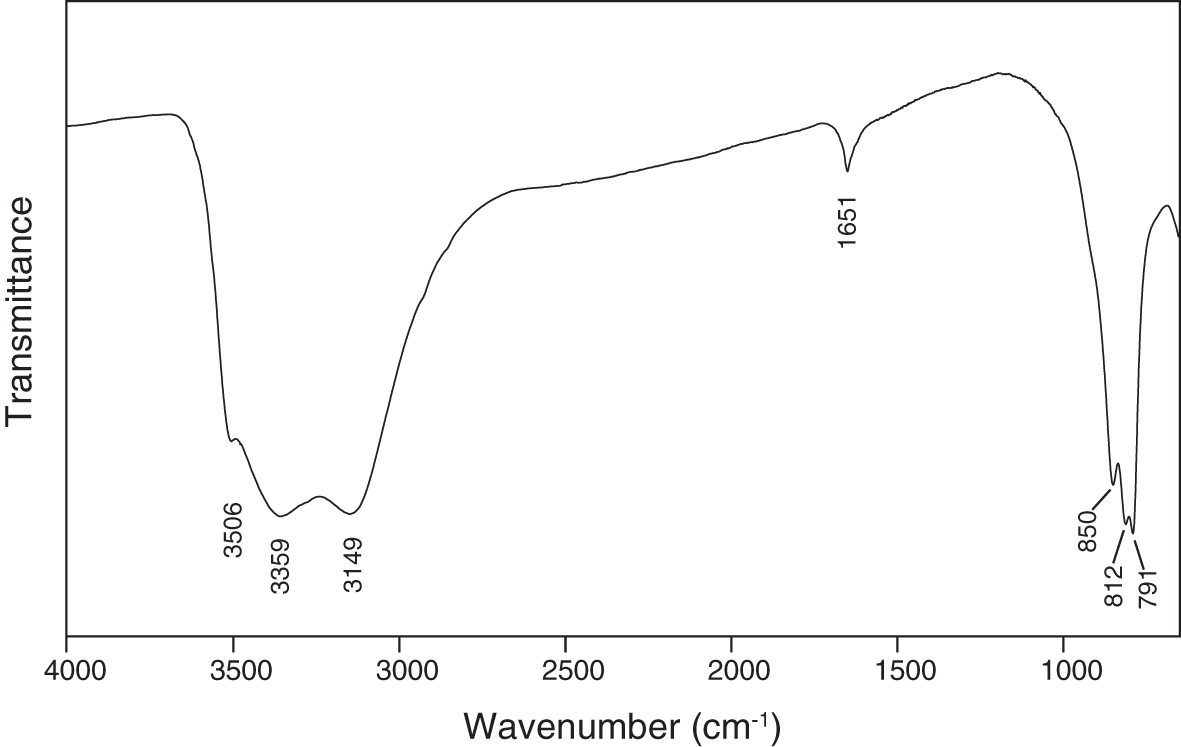
The infrared-absorption spectrum of powdered puttapaite, recorded using a Nicolet 5700 FTIR spectrometer equipped with a Nicolet Continuμm IR microscope and a diamond-anvil cell in the range 650–4000 cm–1, is shown in Fig. 4. A broad band in the O–H stretching region, with three sharp peaks at 3506, 3359 and 3149 cm–1, can be attributed to the presence of H2O and OH groups in the structure. Using the correlation of Libowitzky (Reference Libowitzky1999), the inferred O···O (donor–acceptor) distances are 2.90, 2.77 and 2.69 Å, which correspond to weak- to medium-strength hydrogen bonds. A band at 1651 cm–1 is assigned to the ν2 (δ) H2O bending mode. Bands at 850, 812 and 791cm–1 may be assigned to ν3 vibrations of the AsO4 tetrahedra.
The Fourier-transform infrared spectrum of powdered puttapaite.
Crystallography
Powder X-ray diffraction
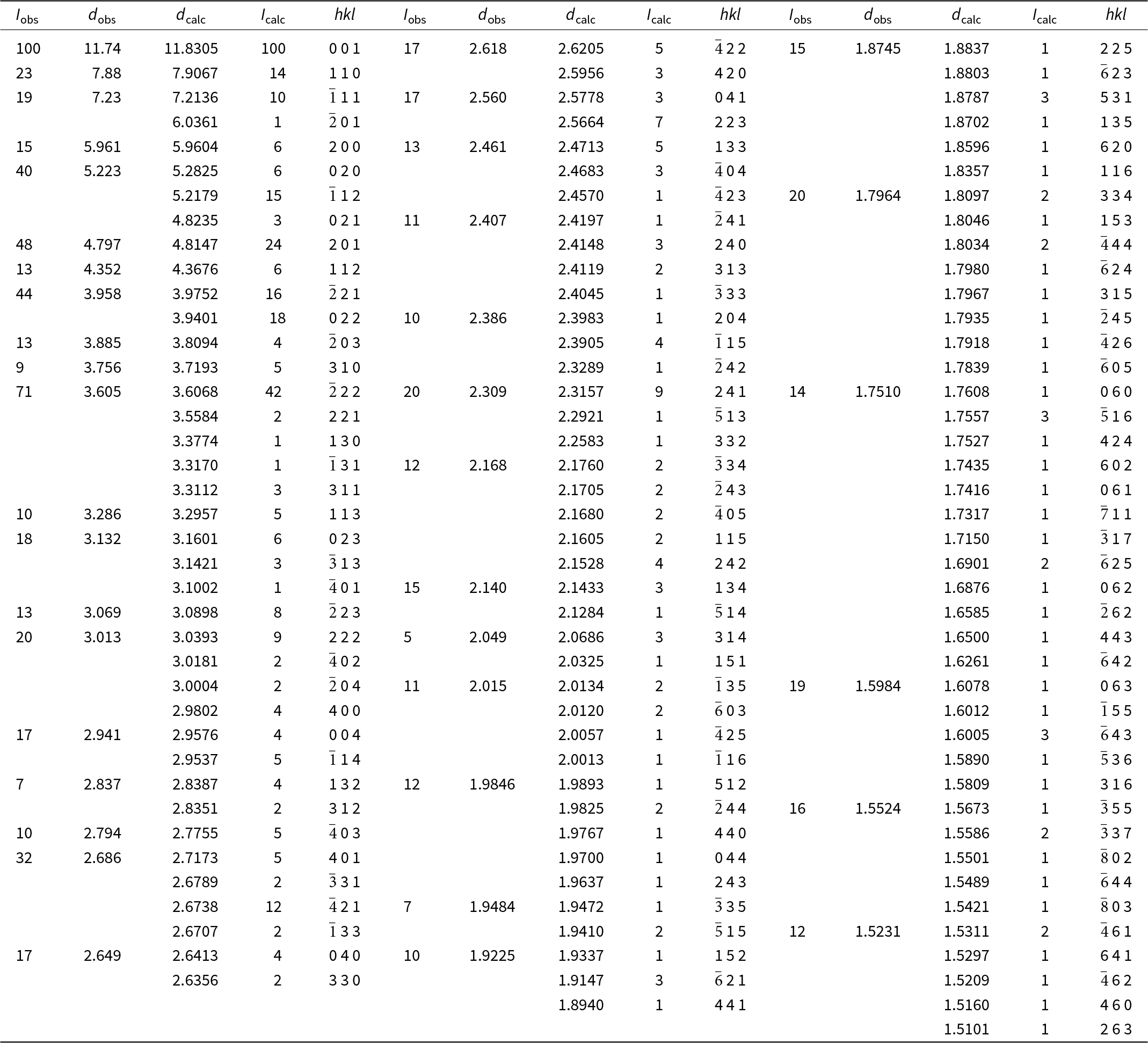
Powder X-ray diffraction data for puttapaite were obtained using a Rigaku R-AXIS Rapid II curved-imaging-plate microdiffractometer, with monochromatised MoKα radiation (50 kV, 40 mA). A Gandolfi-like motion on the φ and ω axes was used to randomise the sample. Observed d values and intensities were derived by profile fitting using JADE Pro software (Materials Data, Inc.). Data (in Å for MoKα) are given in Table 2. Unit-cell parameters refined from the powder data using JADE Pro with whole-pattern fitting are a = 12.480(5), b = 10.588(5), c = 12.297(5) Å, β = 106.434(14)°, V = 1558.5(12) Å3 and Z = 2.
Powder X-ray diffraction data for puttapaite
Single-crystal X-ray diffraction
A single-crystal data collection was made at the macromolecular beam line MX2 of the Australian Synchrotron (Aragao et al., Reference Aragao, Aishima, Cherukuvada, Clarken, Clift, Cowieson, Ericsson, Gee, Macedo, Mudie, Panjikar, Price, Riboldi-Tunnicliffe, Rostan, Williamson and Caradoc-Davies2018). Data were collected using a Dectris EigerX 16M detector and monochromatic radiation with a wavelength of 0.710760 Å. The data set was processed using XDS (Kabsch, Reference Kabsch2010) without scaling, and with absorption correction and scaling using SADABS (Bruker, Reference Bruker2001). Data collection details are given in Table 3.
Crystal data, data collection and refinement details

† wR2 = Σw(|F o|2–|F c|2) 2/Σw|F o|2)1/2; w = 1/[σ2(F o2) + (0.02P)2]; P = ([max of (0 or F o2)] + 2F c2)/ 3
Structure determination
Structure solution in space group C2/m was carried out using SHELXT (Sheldrick, Reference Sheldrick2015a) and the structure was refined using SHELXL-2018 (Sheldrick, Reference Sheldrick2015b) as implemented in the WinGX suite (Farrugia, Reference Farrugia2012). It was impossible to separate a high-quality individual from the rosette-like intergrowths. The best fragment found exhibited high mosaicity and multiple diffraction spots. As a result, relatively high R int and final R values were obtained, 17.3% and 11.89%, respectively. We were unable to locate the H atom positions in difference-Fourier maps. Final atom coordinates and anisotropic-displacement parameters are listed in Table 4, selected interatomic distances and are given in Table 5, and bond-valence values, calculated using the parameters of Gagné and Hawthorne (Reference Gagné and F.C2015) are given in Table 6. The crystallographic information file has been deposited with the Principal Editor of Mineralogical Magazine and is available as Supplementary material (see below).
Fractional atomic coordinates and displacement parameters (in Å2) for puttapaite
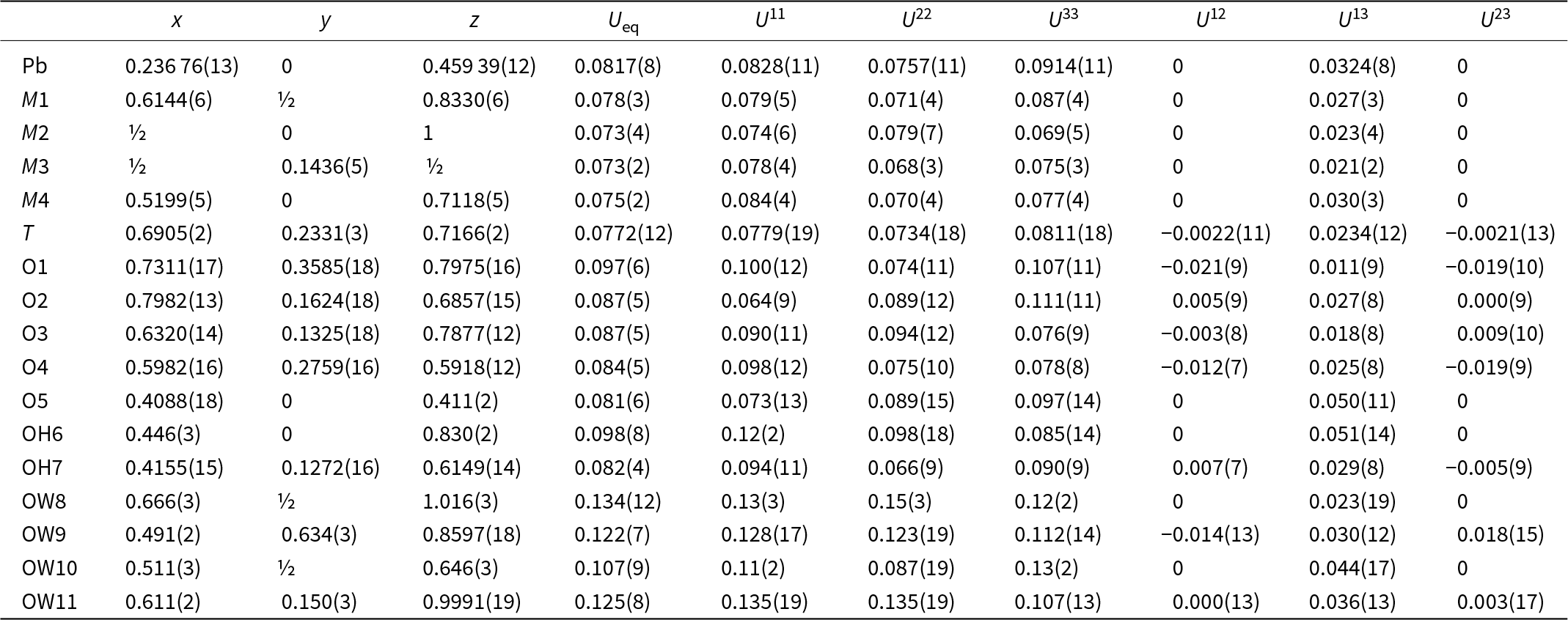
Selected interatomic distances (Å) and possible hydrogen bonds (Å) for puttapaite
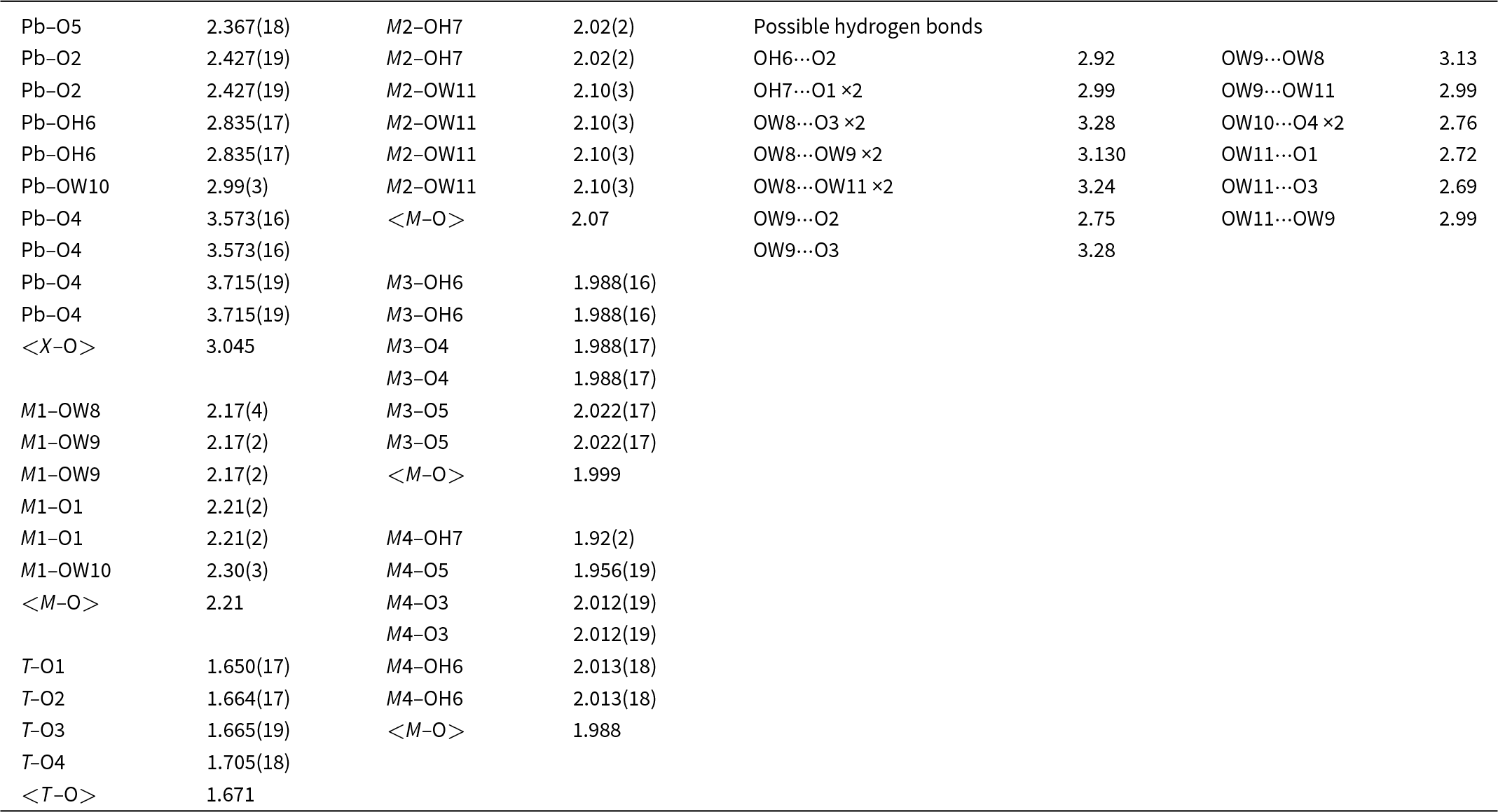
Bond-valence analysis for puttapaite
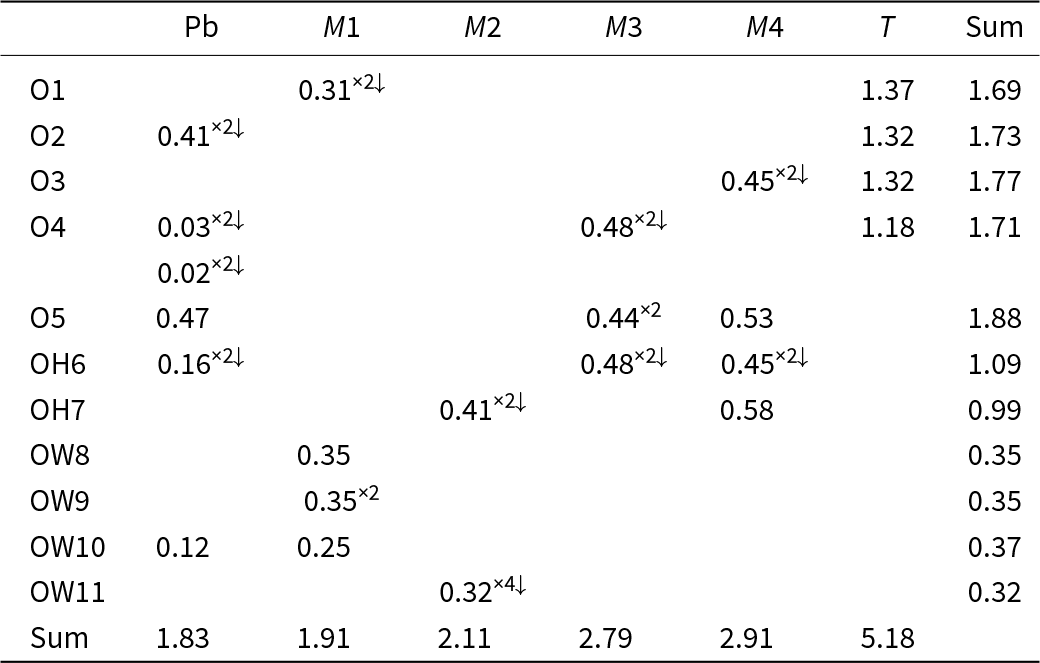
Assignment of cation site-populations to the M sites and T site was completed based on observed mean bond lengths (Table 7). Mn2+ plus minor Ca and Sr were assigned to the larger M1 site, Zn, Mg plus minor Cu were assigned to the medium-sized M2 site and Cr3+ plus minor Al, Fe3+ and Mn3+ were assigned to the smaller M3 and M4 sites. Chemical analysis shows more Cr than is required to fill the M3 and M4 sites and insufficient As to fill the T site, hence 0.29 atoms per formula unit Cr6+ was assigned to the T site. This assignment is supported by the <T–O> distance of 1.671 Å (Table 4), which is less than the distance expected for full occupancy by As5+ of 1.687 Å (Gagné and Hawthorne, Reference Gagné and F.C2018).
Refined site-scattering values (epfu) and assigned site-populations for puttapaite
epfu – electrons per formula unit
Structure description
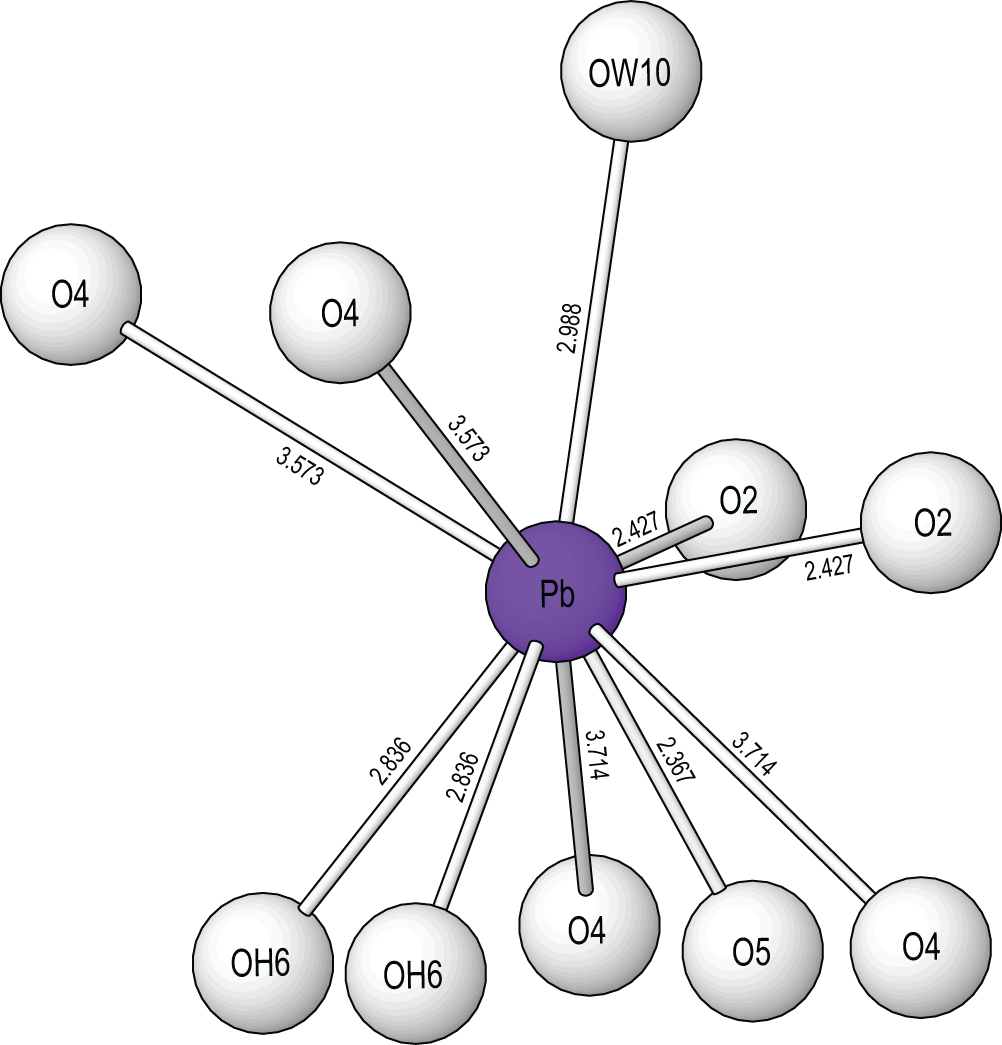
The Pb site is [10]-coordinated (Fig. 5) with three short bonds to O2 and O5, three medium length bonds to OH6 and OW10 and four very long bonds to O4. The site exhibits one-sided coordination typical of Pb2+ with a stereochemically active 6s 2 lone-electron-pair. Chemical analysis shows that the site is fully occupied by Pb. The M sites are each [6]-coordinated by O2– anions, OH groups and H2O groups in regular octahedral arrangements. The As site is coordinated as a regular tetrahedron by four O2– anions and shows only slight distortions [angular range 107.1(8)–111.6(10) Å].
The Pb2+ coordination in puttapaite showing Pb–O bond lengths in angstroms, Å. All crystal structure drawings were done with ATOMS (Dowty, Reference Dowty1999).
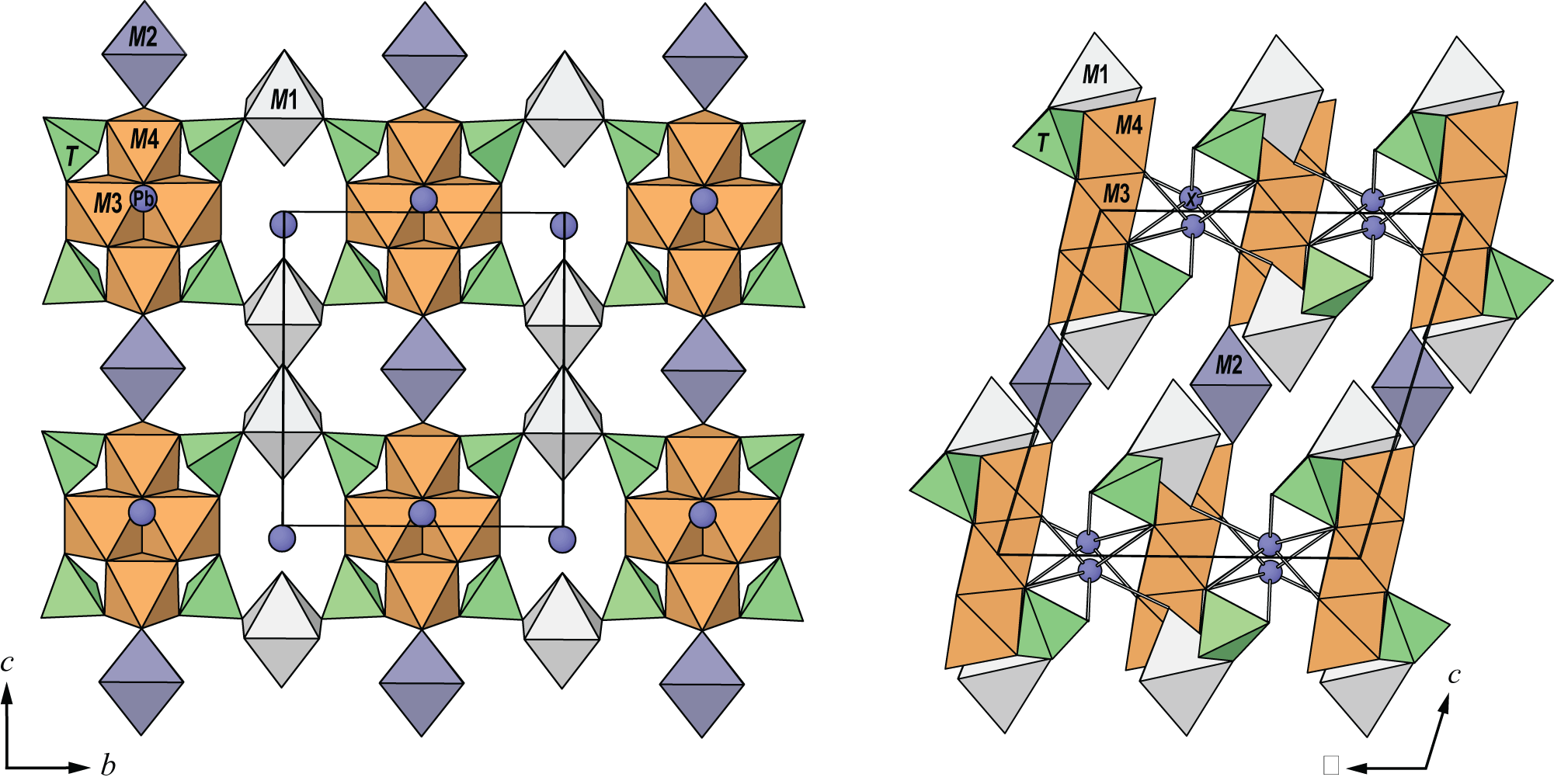
Two M3 and two M4 octahedra share edges to form a M 4O16 tetrameric cluster. Four TO4 tetrahedra share O3 and O4 anions with each M 4O16 cluster to form a M 4T 4O24 cluster. M1 octahedra link to TO4 tetrahedra via corner sharing to link clusters in the [010] direction and M2 octahedra link to M4 octahedra via corner sharing to link clusters in the [001] direction to form sheets parallel to {100}. Sheets link in the [100] direction by Pb–O bonds. The structure is shown in Fig. 6.
The crystal structure of puttapaite viewed (left) along [100] and (right) along [010]. The unit cell is outlined.
The structure contains four H2O molecules and two OH groups. Although the H atoms could not be located during the refinement, a possible hydrogen bonding scheme based on O···O bond distances is summarised in Table 5. Under this scheme, the underbonded O1, O2, O3 and O4 anions are receptors of hydrogen bonds as are each of the H2O molecules. The observed O⋯O distances range from ∼2.7 to ∼3.3 Å indicating strong to very weak hydrogen bonds.
Relationship to other minerals
Puttapaite represents a unique chemistry and structure type for minerals and inorganic compounds. There are many arsenate minerals that contain Pb and Zn and five arsenate minerals that contain Pb and Mn. Besides puttapaite, there is only one other arsenate mineral that contains Pb and Cr, fornacite. In fornacite, Cr is hexavalent whereas in puttapaite Cr is trivalent. The crystal structures of anthoinite, AlWO3(OH)3 (Grey et al., Reference Grey, Madsen, Mills, Hatert, Paterson and Bastow2010), and bamfordite, Fe3+Mo2O6(OH)3⋅H2O, (Birch et al., Reference Birch, Pring, McBriar, Gatehouse and McCammon1998) contain M 4O16 tetrameric clusters of edge-sharing octahedra and they have a similar layer structure. In the anthoinite structure there are two types of tetramer that share octahedral vertices with four adjacent tetramers to form stepped layers parallel to (001). Connectivity between layers is by hydrogen bonding. In the bamfordite structure, tetramers interconnect via octahedral dimers, Fe3+2(O,OH,H2O)10, to form stepped layers parallel to (100). The layers are linked by hydrogen bonding.
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1180/mgm.2024.60.
Acknowledgements
The authors thank Ben Wade of Adelaide Microscopy, The University of Adelaide for assistance with the microprobe analysis. The infrared spectrum was acquired with the assistance of the Forensic Science Centre, Adelaide. This research was undertaken in part using the MX2 beamline at the Australian Synchrotron, part of ANSTO, and made use of the Australian Cancer Research Foundation (ACRF) detector. The authors thank two anonymous reviewers and Structures Editor Peter Leverett for their comments and corrections.
Competing interests
The authors declare none.














