Introduction
On Earth, there are several reservoirs where carbon circulates with different residence times and turnover rates. In the biosphere, for example, it resides for an average of 4 to 8 years (Gaudinski et al. Reference Gaudinski, Trumbore, Davidson and Zheng2000) while in the oceans, CO2 dissolves in the water and carbon atoms can remain in the deep layers for thousands of years (e.g. Sigman and Boyle Reference Sigman and Boyle2000).
This behavior has an implication for marine organisms that incorporate dissolved inorganic carbon (DIC) from the marine environment into their structures. Mollusks, for example, take up inorganic CO2, bicarbonate, and carbonate from the water to form their calcite or aragonite shells. Since this carbon pool has remained in the ocean for potentially hundreds or thousands of years, radiocarbon (14C) dating of ocean shells will result in an older “apparent age,” with a difference that can be hundreds of years from contemporary atmosphere (Mangerud Reference Mangerud1972). This phenomenon is known as the “marine reservoir effect” (Stuiver and Polach Reference Stuiver and Polach1977) and can be corrected in two steps. First, by calibrating with a global marine curve, which takes into account offsets between the atmosphere and the global average ocean over time (Heaton et al. Reference Heaton, Köhler, Butzin, Bard, Reimer, Austin, Bronk Ramsey, Grootes, Hughen, Kromer, Reimer, Adkins, Bueke, Cook, Olsen and Skinner2020). Second, it must be taken into account that the reservoir effect depends on local factors such as latitude, currents, temperature and marine topography (e.g. Alves et al. Reference Alves, Macario, Ascough and Bronk Ramsey2018). Quantifying this local effect, known as delta R (ΔR) is possible, and there are generally three ways to do it.
One possibility is the use of coeval marine-terrestrial paired samples recovered from archaeological contexts or buried in coastal sediments; for these cases, it is very important to ensure the contemporaneity of the paired samples (Alves et al. Reference Alves, Macario, Ascough and Bronk Ramsey2018). Another option is by dating marine samples with radiocarbon and in parallel with an alternative dating method (e.g., U/Th). However, the most direct method is by dating shells or bones of marine organisms that accomplish certain requirements. Ideally, they should have been collected alive and before 1950, to avoid perturbations in atmospheric 14C levels caused by nuclear detonations; the year of collection should be known precisely as well as the geographic location of their provenance; species that feed on suspended material are preferred to avoid specimens that scrape and take carbonates of geological origin, or carbon deposited for a long time in sediments (O’Connor et al. Reference O’Connor, Ulm, Fallon, Barha and Loch2010). Samples that accomplish all these conditions can be found in museum collections, that have reliable and complete records of the specimens.
In recent years, publications of ΔR values for localities around the world have increased, as can be seen in the Marine Reservoir Correction database (http://calib.org/marine) (Reimer and Reimer Reference Reimer and Reimer2001). These data are not homogeneously distributed, since there are large coastal extensions for which there is still no information. In the case of Mexico, most of the reported data correspond to the area of the Baja California Peninsula, Gulf of California and Pacific Ocean (Berger et al. Reference Berger, Taylor and Libby1966; Frantz et al. Reference Frantz, Kashgarian, Coale and Foster2000; Goodfriend et al. Reference Goodfriend and Flessa1997; Ingram and Southon Reference Ingram and Southon1996; Treinen et al. Reference Treinen-Crespo, Barbara, Villaescusa, Schmidt, Pearson and Carriquiry2021). In Gulf of Mexico (Wagner et al. Reference Wagner, Guilderson, Slowey and Cole2009) and Caribbean Sea regions (Druffel et al. Reference Druffel1980; Greer et al. Reference Greer, Clark, Waggoner, Busch, Guilderson, Wirth, Zhao and Curran2020), there are also some reported data.
Obtaining new ΔR values from Mexican littorals is of interest for doing more precise dating of malacological samples, especially in archaeological contexts where it is hard to find other types of organic materials, due to the difficulty of their preservation in coastal environments. In Mexico, mollusks were highly appreciated by pre-Columbian cultures, and were used for different purposes: food, artistic, commercial, ritual, tool making, construction, and ornamental objects. It is common to find them in burial offerings, in decorative objects, or as food remains throughout all the territory (Martínez Becerril Reference Martínez Becerril2013).
However, it should be noted that one of the drawbacks of using known-collection-year shells for ΔR determination, is that they have very recent collection dates. Therefore, it may not be always appropriate to assume that ΔR values from the early 20th century, are compatible with those from an earlier past (Hadden et al. Reference Hadden, Hutchinson and Martindale2023)
Methods
The set of selected samples for this study was obtained from a collection of shells belonging to the Natural History Museum Felipe Poey, from the Universidad de La Habana, Cuba. Samples were derived from five sites: four of them in the Pacific Coast (San Pedro, in the California USA coast; Todos Los Santos and Cedros Island in Baja California coasts; and Mazatlan), and the Isla Mujeres site in the Caribbean Sea (Figure 1). According to the museum records, all specimens were collected alive, with known collection date, and prior to the beginning of atmospheric nuclear weapons tests.
Map showing the locations of studied sites.

Among the corresponding species we have seven bivalves and six gastropods (Table 1). Bivalves Tellina radiate (Linne 1758), Eurytellina lineata (Turton1819), Eurytellina angulosa (Gmelin1791) and Solecurtus sanctaemarthae (d’Orbigny 1842) are usually found buried in sandy or muddy substrates in shallow waters, often in coastal areas, estuaries, lagoons and intertidal zones. Like other bivalves, these species are suspension feeders, filtering plankton and organic particles from the water. Macron lividus (Adams 1855) and Amiantis callosa (Conrad 1837) inhabits sandy or muddy substrates in shallow coastal waters, including estuaries, lagoons, and tidal flats. They are filter feeders commonly found buried just below the surface of the sediment. Leptopecten latiauratus (Conrad1837) are often found in kelp forests, where they attach themselves to algae or other substrates with byssal threads. They are usually found in shallow waters, but can also inhabit deeper regions, up to 250 meters. This species feeds by filtering water for plankton, which is its primary food source (Abbott and Percy Reference Abbott and Morris2001; Rosenberg Reference Rosenberg2009; Ruppert and Richard Reference Ruppert and Fox1988).
Analyzed samples grouped by location and species

In the gastropods group, Sinum perspectivumy (Say 1831) and Ficus papyraceus (Say 1822) are predators. They are typically found in sandy or muddy substrates in shallow coastal environments, often burrowing in the sediment just beneath the surface. Mitrella ocellata (Gmelin 1791) and Mazatlania fulgurita (Philippi 1846) are typically found in shallow marine environments, such as sandy or muddy substrates, from the intertidal zone down to deeper waters in the case of Mazatlania. They are both carnivorous predators, feeding on small invertebrates and other mollusks. Ceratostoma nuttalli (Conrad 1837) is found in shallow marine environments, including rocky reefs and coral substrates. As a predator it feeds on other mollusks and small invertebrates. Columbella major (Sowerby 1832) is typically found in shallow coastal waters, often on sandy or rocky substrates, and among seagrasses or coral rubble. It is also found in intertidal zones where it can tolerate some exposure to air. This species is a carnivorous scavenger, feeding on small invertebrates and organic detritus (Abbott and Percy Reference Abbott and Morris2001; Rosenberg Reference Rosenberg2009; Ruppert and Richard Reference Ruppert and Fox1988)
For some species, we measured two different specimens from the same museum lot. Moreover, we measured some of the shell specimens twice with the aim of finding some variations in the same body shell or possible error in the measuring process. All dated shells correspond to short-term life species with lifespan of 1 to 5 years; nevertheless, we selected for dating the part in the shell associated to the last stage of growing.
Samples were prepared at the AMS Laboratory LEMA (Laboratorio de Espectrometría de Masas con Aceleradores) of the Physics Institute, Universidad Nacional Autónoma de México. Between 20 and 30 mg of shells samples were selected. They were first washed with deionized water. After drying, they were treated with a volume of HCl 0.1M (Hydrochloric acid 37%, Meyer) specifically calculated to remove 50% of the mass. The purpose was to eliminate the most external part, usually exposed to contamination and probable chemical alterations. Samples remained in acid for 24 hours at 75°C. Finally, they were washed with ultrapure water until reaching neutral pH and left to dry at 50°C in the oven.
After injecting H3PO4 (orthophosphoric acid 85%, Merck) at 70°C to obtain CO2, samples were graphitized in a Carbonate Handling System (Ion Plus), using iron as catalyst of the reaction (Wacker et al. Reference Wacker, Fülöp, Hajdas, Molnár and Rethemeyer2013). Accelerator mass spectrometry (AMS) analysis of pressed graphite was performed in the High Voltage Engineering Europa (HVEE) 1MV AMS-LEMA system (Solís et al. Reference Solís, Chávez-Lomelí, Ortiz, Huerta, Andrade and Barrios2014).
Results and discussions
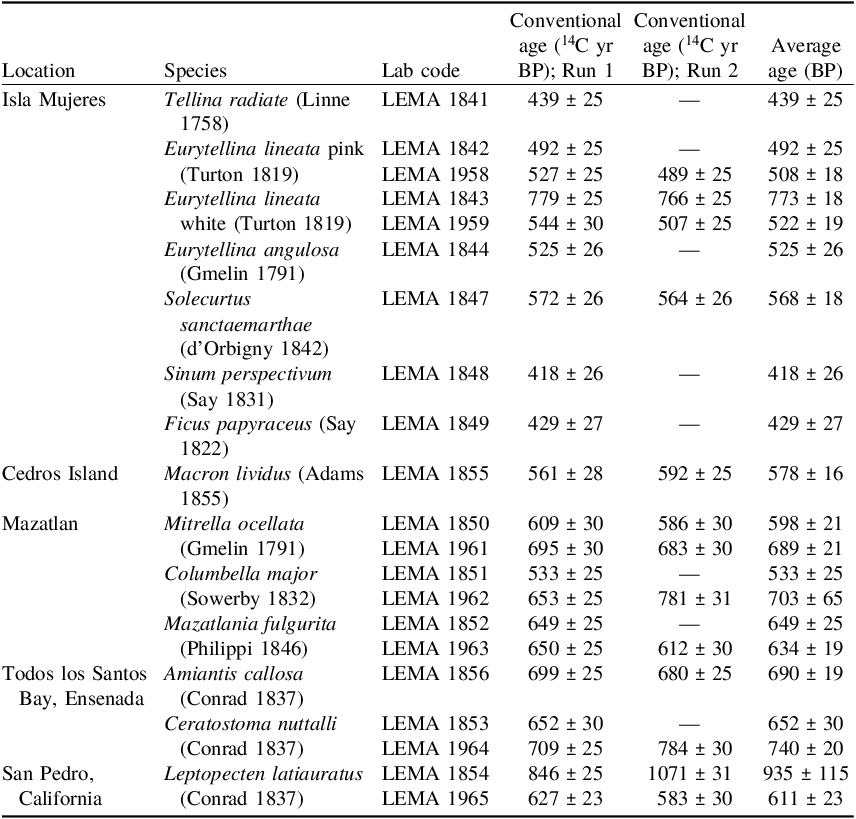
Table 1 presents the results obtained for the samples grouped by location and species. The last column is the weighted-average of the two runs for the shells measured twice and the corresponding error is the statistic error if both values fit the χ2-test, or the standard deviation if they do not (Ward and Wilson Reference Ward and Wilson1978). All samples present a good agreement between both runs, with the exception of LEMA 1854 and 1962.
From the obtained results, we can do some analyses. We start looking at the interspecies behavior of radiocarbon age. Since the collection year (see Table 2) of all samples goes from AD 1925 to 1945, no significant variation for marine modelled age (see Marine20 curve; Heaton et al. Reference Heaton, Köhler, Butzin, Bard, Reimer, Austin, Bronk Ramsey, Grootes, Hughen, Kromer, Reimer, Adkins, Bueke, Cook, Olsen and Skinner2020) is observed in this period. Therefore, the variation of ∆R is totally due to the variation of radiocarbon ages among samples.
ΔR estimation calculated using Table 1 values

We conducted χ2-tests considering all runs for each species together, to test for homogeneity within species. We observed good agreement for most species. Among bivalves, the specie Eurytellina lineata pink (LEMA 1842 and LEMA 1958) (T = 1.43, χ2 0.05 = 5.99) show the better agreement for χ2-test and then reliability of this specie for ∆R studies and radiocarbon dating as well. The poor agreement for Eurytellina lineata white (LEMA 1843 and LEMA 1959) (T = 90.3, χ2 0.05 = 7.82) is due to sample LEMA 1843 yields too high. However, both runs of LEMA 1843 gave close radiocarbon ages, so this result can best be explained as bad sample classification/selection in the museum (e.g., the sample was not actually live collected). Anyway, there are no reasons to distrust this species mainly because the other shell LEMA 1959 has quite similar values of the other sub-species Eurytellina lineata pink (LEMA 1842 and LEMA 1958). Similar issues come from species Leptopecten latiauratus. Sample LEMA 1854 shows high and dispersed values, however, LEMA 1965 gave less scattered and more representative radiocarbon dates. The results obtained for LEMA 1854 are difficult to explain in terms of a reservoir effect or with the feeding habits of a bivalve species. The variability in local carbon sources or recrystallization due to the fact that the shell of this species is extremely thin compared to most bivalves, could be a more plausible explanation.
On the other hand, the gastropod Mazatlania fulgurita (LEMA 1852 and LEMA 1963) (T = 1.16, χ2 0.05 = 5.99) also show good agreement, while Mitrella ocellata (LEMA 1850 and LEMA 1961) (T = 9.68, χ2 0.05 = 7.82) and Ceratostoma nuttalli (LEMA 1853 and LEMA 1964) (T = 7.76, χ2 0.05 = 5.99) even failing χ2-test both show small dispersion. However, Columbella major (LEMA 1851 and LEMA 1962) (T = 40.7, χ2 0.05 = 5.99) presents high dispersion not only among the species but also in the same shell too. The issues with some gastropods species for radiocarbon dating has been described by several authors (e.g., Douka et al. Reference Douka, Higham and Hedges2010; Hogg et al. Reference Hogg, Higham and Dahm1997; Reimer et al. Reference Reimer, McCormac, Moore, McCormick and Murray2002). The carbon source of shell formation comes mainly from dissolved inorganic carbon (DIC) but may also be obtained metabolically. Columbella major is classified as omnivore mostly herbivorous grazer (Ruiz and Bonilla Reference Ruiz and Reyes Bonilla2021), so the 14C concentration may be more sensitive to particulate sources of carbonate (Hogg et al. Reference Hogg, Higham and Dahm1997) and it may also incorporate old carbonates when scrape microflora from the substrate (Reimer et al. Reference Reimer, McCormac, Moore, McCormick and Murray2002). For the species Mazatlania fulgurita and Mitrella ocellata, which are predators, their carbonate is derived principally from seawater DIC and the results seem to confirm that they are more reliable for radiocarbon analyses.
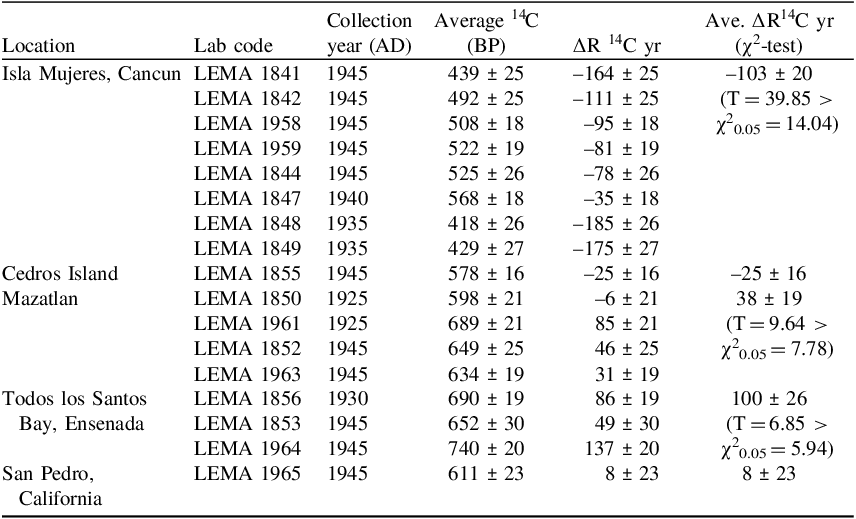
Taking into account the above considerations we excluded from ∆R studies: the outlet samples LEMA 1843, LEMA 1854 and both samples of species Columbella major, LEMA1851 and LEMA 1962. The ∆R values presented in the Table 2 were calculated following a straightforward method for known-age samples ∆R = P – Q, where P is the measured radiocarbon age of the shell and Q is the expected radiocarbon age, for the collection year according to the Marine20 curve (Russell et al. Reference Russell, Cook, Ascough, Scott and Dugmore2011; Stuiver et al. Reference Stuiver, Pearson and Braziunas1986). The error reported for ∆R is the uncertainty of the marine sample 14C age measurement (Reimer and Reimer Reference Reimer and Reimer2016). The ∆R average for locations with several values was calculated as the weighted-average of the set with the standard deviation of the mean as error.
In Isla Mujeres site, the eight samples gave negative ∆R values from –185 14C yr to –35 14C yr, with a weighted average of ∆R = –103 ± 20 14C yr (T = 39.85, χ2 0.05 = 14.04). Even when χ2-test fails (mainly due to sample LEMA 1847) there is not a big dispersion taking into account the number of samples and different species used for ∆R estimation. Isla Mujeres is a little island just in front of Cancun in the Yucatan Channel. The negative value can be explained by the influence of the Yucatan Current (Athie et al. Reference Athié, Candela, Sheinbaum, Badan and Ochoa2011; Sheinbaum et al. Reference Sheinbaum, Candela, Badan and Ochoa2002; Simanca et al. Reference Simanca, Ramírez, Fernandez, Arriaza, Rodas, Esponda, Garcia, Alburquerque and García2012), that is a fast-warm flux of water coming from the Caribbean Sea. Similar negative values are reported by Díaz et al. (Reference Díaz, Macario, Gomes, Álvarez-Lajonchere, Aguilera and Alves2017) on the northwest Cuban coast, just a few kilometers away, crossing the Yucatan strait.
The rest of the sites are located in the Pacific coast, throughout which, the upwelling of deep waters is common (Dawson Reference Dawson1951; Delgadillo-Hinojosa et al. Reference Delgadillo-Hinojosa, Camacho-Ibar, Huerta-Díaz, Torres-Delgado, Pérez-Brunius, Lares, Marinone, Segovia and Peña-Manjarrez2015; Lluch-Cota Reference Lluch-Cota2000; Varela et al. Reference Varela, Lima, Seabra, Meneghesso and Gómez-Gesteira2018; Zaytsev et al. Reference Zaytsev, Cervantes-Duarte, Montante and Gallegos-García2003;) so positive ∆R values are expected. In the case of Mazatlán, we obtained three positive similar values and a slightly negative one. The average value ∆R = 38 ± 19 14C yr (T = 9.64, χ2 0.05 = 7.78) is close to the previously reported at Mazatlán (∆R = 59 ± 48 14C yr) and Isabel Island (∆R = 85 ± 50 14C yr), less than 100 Km away (Berger et al. Reference Berger, Taylor and Libby1966). For Todos los Santos Bay, ∆R = 100 ± 26 14C yr (T = 6.85, χ2 0.05 = 5.94) with three samples of two species. There are not previous references in the MRCD, but Delgadillo-Hinojosa et al. (Reference Delgadillo-Hinojosa, Camacho-Ibar, Huerta-Díaz, Torres-Delgado, Pérez-Brunius, Lares, Marinone, Segovia and Peña-Manjarrez2015) reported upwelling influence inside the bay, in a similar way to that of the California Current System. The obtained value ∆R = 8 ± 23 14C yr for San Pedro site is different from that reported in MRCD, ∆R = 111 ± 55 14C yr (Ingram and Southon Reference Ingram and Southon1996). This result can be related to the species Leptopecten latiauratus as pointed above, but this is not conclusive. Because there are five decades of difference for the collection year between the samples in both studies, there could be real variation of ∆R during that period in a location with a strong anthropogenic influence. More data are needed to accurately estimate ∆R for this place.
Unfortunately, the exact collection place of Mazatlán, Todos los Santos Bay and San Pedro is too ambiguous to find a local detailed explanation for ∆R value. Nevertheless, the ∆R values obtained for these three locations confirm a pattern of positive values for the west coast of Mexico (Treinen-Crespo et al. Reference Treinen-Crespo, Barbara, Villaescusa, Schmidt, Pearson and Carriquiry2021, Berger et al. Reference Berger, Taylor and Libby1966).
Finally, the negative value of ∆R = –27 ± 16 14C yr obtained for Cedros Island can look a bit unexpected. However, Berger et al. (Reference Berger, Taylor and Libby1966) obtained a value of ∆R = 11 ± 51 14C yr, also lower compared to other values reported by those authors for Baja California coast. Therefore, a predator gastropod in the present study and a deposit feeder bivalve in the previous study (Berger et al. Reference Berger, Taylor and Libby1966) seems to confirm more “enriched” 14C content in Cedros Island, probably associated with being located several kilometers offshore, less influenced by coast upwelling (Dawson Reference Dawson1951).
Conclusions
The dating of known-age shells from museum collections is a suitable tool not only for ∆R studies but also can provide information about the reliability of the different species of shells for radiocarbon dating. In this research twenty-one samples of fourteen different species were dated, and their values used to estimate the marine reservoir correction in five locations. The values obtained for Todos los Santos (100 ± 26 14C yr) and Isla Mujeres (–103 ± 20 14C yr) represent the first ∆R estimation in these places. The other two sites in Mexican coasts, Mazatlan (38 ± 19 14C yr) and Cedros Island (–27 ± 16 14C yr), show consistent values with previous studies, only the ∆R obtained in San Pedro, California (8 ± 23 14C yr) presents a divergent value compared with a first report. On the other hand, all species of bivalves and gastropods predators dated seems to be reliable for radiocarbon studies. Only Columbella major and Leptopecten latiauratus showed high dispersion of radiocarbon ages, therefore we recommend using these species with caution in absent of more data. New ∆R values obtained from Mexican littorals, are of interest for doing more precise malacological samples dating, especially in archeological contexts where it is difficult to find other types of organic materials, due to the difficulty of their preservation in coastal environments.
Acknowledgments
We are grateful to the editor and the anonymous reviewers whose insights improved this paper. We also thank Arcadio Huerta and Sergio Martínez for technical assistance. This project received partial support from grants DGAPA-PAPIIT-UNAM IN112023, IA101323 and CONAHCYT 2023-LNC-58.