Cognitive models of depressionReference Beck1 describe low mood and behavioural withdrawal as consequences of distorted emotion processing and maladaptive thinking. Empirical evidence highlights selective attention to, recall of and difficulty disengaging from negative information as key features of these distortions.Reference LeMoult and Gotlib2–Reference Duyser, van Eijndhoven, Bergman, Collard, Schene and Tendolkar6 Cognitive–behavioural therapy (CBT) addresses these biases by, for example, promoting changes in biased attention.Reference Vazquez, Duque, Blanco, Pascual, Poyato and Lopez-Gomez7 Although CBT is a first-line treatment for depressive disorders,8 a notable proportion of patients do not respond sufficientlyReference Cuijpers, Karyotaki, Ciharova, Miguel, Noma and Furukawa9 and research on its underlying mechanisms remains limited. Investigating the neurobiological effects of CBT may offer insights into mechanisms and predictors to improve this therapy.
Functional neuroimaging (fMRI) studies in depressive disorders suggest an imbalance in frontolimbic activity during negative emotion processing, with hyperactivity in the limbic system and hypoactivity in prefrontal regions such as the dorsal anterior cingulate cortex (dACC).Reference Stuhrmann, Suslow and Dannlowski10,Reference Li and Wang11 To date, few longitudinal CBT fMRI studies have been conducted,Reference Fu, Williams, Cleare, Scott, Mitterschiffthaler and Walsh12–Reference Rubin-Falcone, Weber, Kishon, Ochsner, Delaparte and Doré15 yielding a rather inconsistent pattern concerning the reversibility of these activity changes during negative emotion processing. While some studies show activity decreases in the hippocampus–amygdala complexReference Fu, Williams, Cleare, Scott, Mitterschiffthaler and Walsh12 and increases in the ACCReference Fu, Williams, Cleare, Scott, Mitterschiffthaler and Walsh12,Reference Yang, Oathes, Linn, Bruce, Satterthwaite and Cook14 and prefrontal areasReference Ritchey, Dolcos, Eddington, Strauman and Cabeza13,Reference Yang, Oathes, Linn, Bruce, Satterthwaite and Cook14 following CBT, other evidence suggests increased hippocampal activity in response to negative stimuli following CBT.Reference Ritchey, Dolcos, Eddington, Strauman and Cabeza13 Additionally, one studyReference Rubin-Falcone, Weber, Kishon, Ochsner, Delaparte and Doré15 reported no significant changes in brain activity related to negative emotion processing following CBT. Furthermore, the extent to which these neural changes correlate with clinical symptom improvement remains unclear.Reference Yang, Oathes, Linn, Bruce, Satterthwaite and Cook14,Reference Rubin-Falcone, Weber, Kishon, Ochsner, Delaparte and Doré15 Previous studies were probably limited by small sample size (n ≤ 23) and a lack of statistical comparisons with control groups (except for ref. Reference Fu, Williams, Cleare, Scott, Mitterschiffthaler and Walsh12), making it difficult to determine whether the observed effects were due to the treatment itself or general fluctuations. Additionally, predicting treatment response appears crucial due to the heterogeneity in outcome. Previous CBT fMRI studies suggest lower pre-treatment dACC activity during negative emotion processing as a potential predictor of CBT response,Reference Fu, Williams, Cleare, Scott, Mitterschiffthaler and Walsh12,Reference Ritchey, Dolcos, Eddington, Strauman and Cabeza13 but results are based on symptom questionnaires prone to weekly fluctuations. Only one study has applied machine learning,Reference Costafreda, Khanna, Mourao-Miranda and Fu16 identifying pre-treatment dACC and frontal activity as potential classifiers for CBT response. However, because the sample size in this study was rather small (N = 16), further predictive models at the individual patient level are needed for clinical translation. Thus, the present study aimed to examine the effect of CBT on brain function and its link to treatment response using a longitudinal naturalistic design, based on a supraliminal emotion-processing task in patients with depressive disorder before and after approximately 20 naturalistic CBT sessions. We focused on the hippocampus, amygdala and dACC, given their relevance for emotion processing. First, we hypothesised a decrease in amygdala and hippocampus activity and an increase in dACC activity to negative stimuli following CBT in patients (objective a). Furthermore, we investigated whether these activity changes were related to response (objective b). We further expected pre-treatment activity differences between responders and non-responders (objective c). Additionally, we exploratively applied machine learning analyses to test predictive models, aiming to identify reliable neural predictors of CBT response at the individual level and to support personalised treatment decisions.
Method
Participants and study design
In this longitudinal naturalistic study, participants were assessed by fMRI, the structured clinical interview for DSM-IV (SCID-I),Reference Wittchen, Wunderlich, Gruschwitz and Zaudig17 as well as by the Hamilton Depression Rating Scale (HDRS)Reference Hamilton18 and Beck Depression Inventory (BDI).Reference Beck and Steer19 Additionally, the course of illness was obtained. For patients with depressive disorders, assessments took place before the start (baseline, t 0) and following approximately 20 sessions of naturalistic CBT (follow-up, t 1; Supplement 1). The healthy control (HC) group was assessed at equivalent time points (time [T], T0–T1, in months: M patients = 7.81, s.d.patients = 2.42; M HC = 7.70, s.d.HC = 1.83; P = 0.773). Patients were recruited from the psychotherapeutic outpatient unit of the University of Muenster (Psychotherapie-Ambulanz), while healthy controls were recruited via public notices and newspaper announcements. Both groups were part of the ongoing Prevention and Intervention Neuroimaging Cohort study, with data collection for these groups conducted between August 2017 and September 2022. The authors assert that all procedures contributing to this work comply with the ethical standards of the relevant national and institutional committees on human experimentation, and with the Helsinki Declaration of 197 as revised in 2013. All procedures involving human participants were approved by the Ethics Committee of the University of Münster (amendment nos 2016-173-f-S and 2020-205-f-S). Written informed consent was obtained from all participants.
The final study sample consisted of n = 59 patients with a depressive disorder and n = 60 healthy controls (for exclusion criteria and sample selection, see Supplement 2 and Supplementary Fig. 1). Sample characteristics can be found in Table 1. The patient group was further divided into two subgroups (Table 2) based on the change in diagnosis from t 0 to t 1 according to DSM-IV.Reference Wittchen, Wunderlich, Gruschwitz and Zaudig17 Patients who transitioned from acute to partial or full remission, or from partial to full remission, between t 0 and t 1, were considered as responders (n = 40). Patients who had suffered from an ongoing acute or partially remitted depressive disorder since t 0 were considered as non-responders (n = 19). Detailed information on prior treatments in patients is presented in Supplementary Table 1.
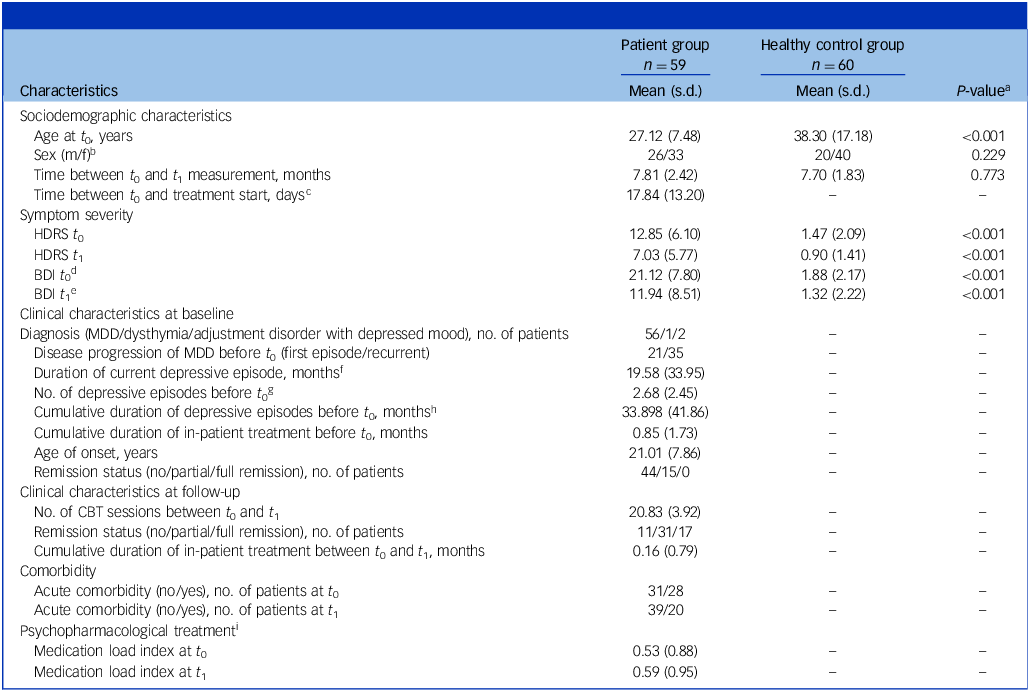
Sociodemographic and clinical characteristics of the sample

HDRS, Hamilton Depression Rating Scale; BDI, Beck Depression Inventory; MDD, major depressive disorder; CBT, cognitive–behavioural therapy.
a . P-values were obtained using the unpaired two-tailed t-test except where noted.
b . P-values were obtained using the χ 2-test.
c . Information was missing for n = 21 in the patient group.
d . Information was missing for n = 1 in the healthy controls group.
e . Information was missing for n = 2 in the patient group and n = 1 in the healthy controls group.
f . Information was missing for n = 6 in the patient group.
g . Information was missing for n = 2 in the patient group.
h . Information was missing for n = 3 in the patient group.
i . For information on medication load index, see Supplement 10.
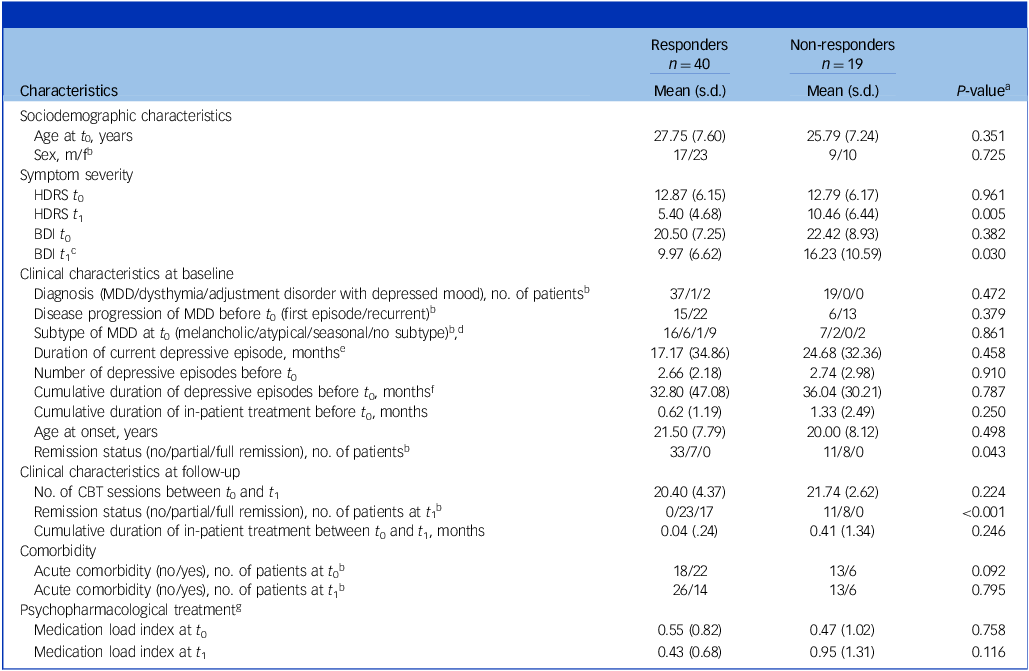
Sociodemographic and clinical characteristics of responders versus non-responders

HDRS, Hamilton Depression Rating Scale; BDI, Beck Depression Inventory; MDD, major depressive disorder; CBT, cognitive–behavioural therapy.
a . P-values were obtained using the unpaired two-tailed t-test except where noted.
b . P-values were obtained using the χ 2-test.
c . Information was missing for n = 1 in the responder group.
d . Subtypes that did not occur in our sample (psychotic, catatonic, postpartal) were not listed; n = 8 responders and n = 8 non-responders were partially remitted at t 0 and therefore did not exhibit any subtype.
e . Information was missing for n = 4 in the responder group and n = 2 in the non-responder group.
f . Information was missing for n = 3 in the responder group.
g . For information on medication load index, see Supplement 10.
fMRI paradigm, data acquisition and preprocessing
For details on the paradigm, data acquisition and preprocessing, as well as on first-level analyses of fMRI data, see Supplements 3–5 and Supplementary Fig. 2. Briefly, for the fMRI paradigm, a negative emotional face-processing task was implemented. This consisted of four blocks of a face-processing task using photographs of faces expressing fear or anger from the Ekman and FriesenReference Ekman and Friesen20 stimulus set, and five blocks of a sensorimotor control task featuring geometric figures in the shape of circles or ellipses. T2* functional data were obtained using a 3 Tesla scanner (Prisma, Siemens, Erlangen, Germany) at t 0 and t 1, and preprocessed using statistical parametric mapping software (SPM8, Wellcome Department of Cognitive Neurology, London, UK; http://www.fil.ion.ucl.ac.uk/spm). A contrast image was generated for each participant in their individual first-level analysis (faces versus shapes), comparing activation while viewing negative faces versus shapes.
Statistical analyses
Clinical and demographic data were analysed using SPSS Statistics version 29.0, IBM Corporation. To assess the impact of CBT on depressive symptom severity, paired t-tests were conducted to compare HDRS and BDI scores at t 0 and t 1 within patients. Additionally, 2 × 2 repeated-measures analyses of variance (ANOVAs) were performed on HDRS and BDI scores, with response (responders versus non-responders) as the between-subjects factor and time (t 0 v. t 1) as the within-subjects factor. Parametric tests were used in these analyses to enhance comparability with prior treatment fMRI studies. As a robustness check, statistical analyses were additionally performed using non-parametric tests.
Functional activity analysis
The fMRI data were analysed using statistical parametric mapping software (SPM12, v6685, Wellcome Department of Cognitive Neurology, London, UK; http://www.fil.ion.ucl.ac.uk/sp), with age and sex as covariates of no interest. For each of the following analyses, region of interest (ROI) analyses of the bilateral hippocampus, bilateral amygdala and bilateral dACC were performed separately. ROIs were created using the Wake Forest University PickAtlas,Reference Maldjian, Laurienti, Kraft and Burdette21 with hippocampus and amygdala ROI defined according to the AAL-atlasReference Tzourio-Mazoyer, Landeau, Papathanassiou, Crivello, Etard and Delcroix22 and dACC ROI defined as BA 32Reference Rive, Van Rooijen, Veltman, Phillips, Schene and Ruhé23 (dilated by 1 mm). Whole-brain analyses were also performed, reporting results with a cluster size threshold of k ≥ 100. Significance thresholds for multiple testing were obtained at the cluster level using threshold-free cluster enhancement (TFCE) implemented in the TFCE toolbox, version 174 (http://dbm.neuro.uni-jena.de/tfce). For all following analyses, a conservative family-wise error (FWE)-corrected threshold of P < 0.05 was established by performing 10 000 permutations per test.
To test the hypothesis regarding CBT-related activity changes (objective a), a 2 × 2 ANOVA, with group (patients versus healthy controls) as a between-subjects factor and time (t 0 v. t 1) as a within-subjects factor, was conducted. To investigate whether pre-treatment activity and activity changes are associated with response (objectives b and c), a subsequent 2 × 2 ANOVA was performed within the patient group, with response (responders versus non-responders) as a between-subjects factor. Post hoc paired t-tests in each group followed significant main effects of time and interaction effects. Group differences in activity between patients and healthy controls at t 0 and t 1 were tested via two-sample t-tests. Additionally, a multivariate machine learning approach, based on Winter et al.,Reference Winter, Blanke, Leenings, Ernsting, Fisch and Sarink24 was applied to distinguish responders from non-responders using pre-treatment ROI and whole-brain activity. For this approach, classification algorithms including support vector machines, random forests, logistic regression, k-nearest neighbours, Gaussian naive Bayes and boosting classifiers were trained; detailed information is provided in Supplement 6. To explore whether CBT-related activity changes are linked to symptom changes within patients (objective b), Spearman’s correlations were computed between significant activity changes and ΔHDRS/ΔBDI scores. Functional activity data from significant clusters identified in 2 × 2 ANOVA (patients versus healthy controls) at t 0 and t 1 were extracted. Percentage change scores were calculated for brain activity, HDRS and BDI (Δvariable = (variable t1 – variable t0)/variable t0 × 100) using SPSS Statistics. Higher scores reflect increased activity or symptoms.
Results
Clinical effects of CBT
HDRS and BDI scores significantly decreased in patients from t 0 to t 1 (HDRS: M Diff = 5.816, 95% confidence interval [CI] [4.099, 7.533], P < 0.001, partial η 2 [η p 2] = 0.442, 95% CI [0.2476, 0.5784]; BDI: M Diff = 9.266, 95% CI [6.952, 11.580], P < 0.001, η p 2 = 0.535, 95% CI [0.3461, 0.6534]). Based on our response criteria, 67.8% (n = 40) of patients were classified as responders and 32.2% (n = 19) as non-responders. There were no significant differences between responders and non-responders with regard to sociodemographic and clinical variables, with the exception of BDI and HDRS scores at t 1 and remission status at both time points (Table 2). A 2 × 2 ANOVA (responders versus non-responders, t 0 v. t 1) on HDRS and BDI scores showed that the significant HDRS decrease was driven by responders (M DIFF = 7.473, P < 0.001, η p 2 = 0.566, 95% CI [0.3396, 0.6932]), while both groups exhibited a reduction in BDI scores (responders: M DIFF = 10.621, P < 0.001, η p 2 = 0.569, 95% CI [0.3396, 0.6964]; non-responders: M DIFF = 6.331, P < 0.001, η p 2 = 0.506, 95% CI [0.1362, 0.6924]; Supplement 7). The results remained valid following robustness checks with non-parametric tests (Supplement 8).
Effects of CBT on negative emotion processing (objective a)
There was a significant group (patients versus healthy controls) × time (t 0 v. t 1) interaction within the right hippocampus (P FWE = 0.022, η p 2 = 0.101, 95% CI [0.0210, 0.2100]; Fig. 1). The observed interaction resulted from significant activity decreases within patients (left: P FWE = 0.024, η p 2 = 0.204, 95% CI [0.0489, 0.3686]; right: P FWE = 0.022, η p 2 = 0.185, 95% CI [0.0383, 0.3497]), whereas there was no significant change in hippocampal activity within healthy controls between t 0 and t 1 (P FWE = 0.420). Furthermore, a main effect of time was identified in the right hippocampus (P FWE =0.028, η p 2 = 0.124, 95% CI [0.0334, 0.2372]). Moreover, a main effect of time was observed in the right dACC (P FWE = 0.043, η p 2 = 0.098, 95% CI [0.0195, 0.2063]), marked by a significant decrease in dACC activity within patients (P FWE = 0.022, η p 2 = 0.233, 95% CI [0.0669, 0.3965]; Fig. 1), while no significant activity changes were evident in healthy controls (P FWE = 0.120). However, no significant interaction effect was detected within the dACC (P FWE = 0.173), and there was no significant interaction effect or main effect of time in the amygdala (both P FWE ≥ 0.198). Supplementary Table 2 provides detailed information on the obtained results. Exploratory whole-brain analyses revealed a significant main effect of time (all P FWE ≤ 0.040) resulting from significant activity decreases from t 0 to t 1 in clusters encompassing only the precuneus, middle temporal gyrus and hippocampus in patients (all P FWE ≤ 0.033; Supplementary Table 3). There was no significant change within healthy controls (P = 0.159). Notably, no significant interaction effect was identified (P FWE ≥ 0.120).
Effects of cognitive–behavioural therapy on functional activity in emotion-processing areas. (a) Visualisation of significant cluster (right: x = 24, y = −40, z = 6, TFCE(232) = 120.33, T (232) = 4.10, k = 26, P FWE = 0.022, η p 2 = 0.101) of the hippocampus region of interest (ROI) analysis for the group × time interaction effect (one-tailed) on a Montreal Neurological Institute (MNI) template, and (b) driven by activity decreases in the patient group from baseline to follow-up (left: x = −16, y = −34, z = 10, TFCE(232) = 127.32, T (232) = 4.48, k = 9, P FWE = 0.024, η p 2 = 0.204; right: x = 26, y = −34, z = 8, TFCE(232) = 130.48, T (232) = 3.93, k = 34, P FWE = 0.022, η p 2 = 0.185. (a,b), Clusters significant at P TFCE-FWE < 0.05. Scale bars indicate TFCE values. (c) Plot depicting hippocampal activity at baseline and follow-up for the healthy control and patient groups during the study interval. Functional magnetic resonance imaging (fMRI) contrast values were calculated using the eigenvariate function of the significant right cluster from the hippocampus ROI analysis of the group × time interaction (one-tailed). This function extracts the first eigenvariate from the significant cluster through singular value decomposition of the time series across all voxels within the cluster. Because all second-level analyses were based on the contrast between each individual’s face and shape (defined in the first-level analysis), the plot represents typical responses for faces versus shapes in this cluster by yielding a vector of fMRI contrast values for each subject and time point. (d) Visualisation of significant cluster (right: x = 20, y = 36, z = 22, TFCE(232) = 134.22, T (232) = 3.94, k = 5, P FWE = 0.043, η p 2 = 0.098) of the dorsal anterior cingulate cortex (dACC) ROI analysis for the main effect of time (one-tailed) on a MNI template, and (e) driven by activity decreases in the patient group from baseline to follow-up (right: x = 20, y = 36, z = 22, TFCE(232) = 174.60, T (232) = 4.94, k = 11, P FWE = 0.022, η p 2 = 0.245). (d,e) Clusters significant at P TFCE-FWE < 0.05. Scale bars indicate TFCE values. (f) Plot depicting dACC activity at baseline and follow-up for the healthy control and patient groups during the study interval. fMRI contrast values were computed by extracting the first eigenvariate of the significant right cluster resulting from the dACC ROI analysis of the main effect of time (one-tailed; for details, see (c)). P FWE, P family-wise error; T, test statistic (t-value); TFCE, threshold-free cluster enhancement.

Association of activity changes with response (objective b)
Even if the subsequent analysis of responders versus non-responders showed only a nominally significant response × time interaction within the hippocampus (P FWE = 0.068), post hoc t-tests revealed that responders showed a decrease in bilateral hippocampal activity during negative emotion processing between t 0 and t 1 (left: P FWE = 0.020, η p 2 = 0.204, 95% CI [0.0269, 0.3999]; right: P FWE = 0.005, η p 2 = 0.260, 95% CI [0.0556, 0.4509]; Supplementary Table 2). There was no significant change in hippocampal activity in non-responders following CBT (P FWE = 0.220). The main effect of time was not statistically significant (P FWE = 0.087). Within the right dACC, analysis of responders versus non-responders showed a significant main effect of time (P FWE = 0.018, η p 2 = 0.219, 95% CI [0.0584, 0.3839]; Supplementary Table 2). Post hoc tests, however, demonstrated no significant dACC activity changes in either responders (P FWE = 0.179) or non-responders (P FWE = 0.092), and no significant interaction effect was observed within the dACC (P FWE = 0.276). Additionally, there was no significant interaction effect or main effect of time in the amygdala (both P FWE ≥ 0.416), or at whole-brain level in clusters k ≥ 100 (P FWE ≥ 0.256).
Viewed dimensionally, reductions in left hippocampal activity within patients from t 0 to t 1 were associated with ΔHDRS (left: r = 0.293, P = 0.024; right: P = 0.935). There was no significant correlation between reductions in bilateral hippocampal activity and BDI score changes (both P ≥ 0.114). Reductions in right dACC activity within patients from t 0 to t 1 were not significantly associated with overall depressive symptom improvement (HDRS: P = 0.759; BDI: P = 0.648).
Cross-sectional group differences and response prediction (objective c)
Cross-sectional analyses revealed no significant differences in brain activity during negative emotion processing between patients and healthy controls at either t 0 or t 1 across the hippocampus (both P FWE ≥ 0.232), amygdala (both P FWE ≥ 0.125), dACC (both P FWE ≥ 0.424) or at whole-brain level (both P FWE ≥ 0.221).
Concerning pre-treatment differences between the responder and non-responder groups, the subsequent analysis of responders versus non-responders revealed that the former had higher pre-treatment activity within the right hippocampus during negative emotion processing compared with non-responders (P FWE = 0.017, η p 2 = 0.189, 95% CI [0.0397, 0.3554]; Supplementary Table 2). However, no significant pre-treatment differences were observed between responders and non-responders in either the dACC (P FWE = 0.371) or amygdala (P FWE = 0.267; Supplementary Table 2). Similarly, no significant differences were observed between both response groups and healthy controls (all P FWE ≥ 0.347; Supplement 9). Additionally, no pre-treatment differences in whole-brain activity were identified between responders and non-responders (P FWE ≥ 0.264). In our machine learning analyses, the best hyperparameter configurations were achieved with random forest classification (Supplementary Table 4). Across tested brain regions, the highest mean balanced accuracy was found for pre-treatment dACC activity (60.4%). For detailed results, see Supplementary Table 5 and Supplementary Fig. 3.
Robustness checks
Robustness checks (Supplement 10 and Supplementary Tables 6 and 7) were performed to validate the main findings, including adjustments for non-linear age effects and clinical characteristics in patients (baseline depressive symptom severity, number of depressive episodes before t 0, remission status at t 0, acute comorbidity and medication intake). Overall, robustness checks confirmed a consistent pattern of results.
Discussion
The present study investigated changes in functional activity during negative emotion processing following naturalistic CBT in patients with a depressive disorder. Our results revealed that activity in emotion-processing areas (hippocampus and dACC) was decreased following CBT in patients and that hippocampal activity changes were related to CBT response. Furthermore, responders showed higher pre-treatment hippocampal activity to negative stimuli in our standard univariate analyses. Notably, these findings largely persisted after controlling for non-linear age effects and clinical characteristics.
Effects of CBT on negative emotion processing and association with response
Consistent with objective a and one previous study,Reference Fu, Williams, Cleare, Scott, Mitterschiffthaler and Walsh12 our results showed a significant interaction in the right hippocampus, driven by decreased hippocampal activity during negative emotion processing in patients following naturalistic CBT, with no changes in healthy controls. The hippocampus often shows hyperactivity in depressive disorders during emotion processing.Reference Li and Wang11 In our study, the significant cluster was primarily located in the posterior hippocampus, an area linked to cognitive and memory processes.Reference Grady25 This suggests that the observed activity decrease may indirectly support improved emotion processing, because previous research indicates that reduced posterior hippocampal activity helps individuals with depression manage negative affect by limiting the reinstatement of mood-congruent negative memories.Reference Doré, Rodrik, Boccagno, Hubbard, Weber and Stanley26 Importantly, hippocampal activity reductions were observed only in CBT responders, potentially suggesting that such changes may occur primarily following successful treatment. Moreover, these decreases correlated positively with greater symptom reductions as measured by HDRS but not by BDI (objective b). One explanation is the methodological variance between a clinician-rated interview and a self-report scale. HDRS may be better aligned with DSM-IV criteria, capturing psychopathological elements more apparent to external raters and thereby enhancing its capacity to detect clinically relevant changes, whereas BDI may also be influenced by maladaptive personality traits.Reference Schneibel, Brakemeier, Wilbertz, Dykierek, Zobel and Schramm27,Reference Svanborg and Åsberg28 Additionally, HDRS encompasses a broader range of symptom dimensions, including somatic aspects, while BDI primarily focuses on cognitive-affective symptoms.Reference Steer, Beck, Riskind and Brown29,Reference Grunebaum, Keilp, Li, Ellis, Burke and Oquendo30 Consequently, hippocampal activity may be more closely linked to symptom domains as assessed by HDRS, suggesting that its reduction may not solely reflect improved emotion processing. Future research should incorporate targeted behavioural experiments to objectively evaluate emotion-processing deficits and treatment effects. Building on our findings, it remains uncertain whether hippocampal activity decreases are central to the antidepressant mechanisms of naturalistic CBT. Although healthy controls showed no significant activity changes over time, suggesting that the observed decreases in patients may not merely reflect the passage of time, these reductions could still represent a general correlate or consequence of symptom improvement rather than a CBT-specific effect. Alternatively, they may arise from common therapeutic factors such as the therapeutic alliance. To clarify these influences, future research would benefit from the use of standardised CBT protocols and the inclusion of waitlist and active treatment control groups. Notably, hippocampal activity decreases are not unique to CBT. Similar reductions have been observed with antidepressants, although the majority of studies found no association with treatment response.Reference Delaveau, Jabourian, Lemogne, Guionnet, Bergouignan and Fossati31,Reference Fitzgerald, Laird, Maller and Daskalakis32 In line with this, the lack of a significant response × time interaction in our study warrants caution in interpreting hippocampal activity decreases as strictly response-dependent CBT effects.
Contrary to objective a, our analyses yielded a significant main effect of time, reflecting significant dACC activity decreases during negative emotion processing following naturalistic CBT in patients, while no significant changes were observed in healthy controls. Involvement of the (dorsal) ACC has been implicated in both top-down emotional regulationReference Pizzagalli and Roberts33 and – within the salience network – rumination and self-referential processing.Reference Murray, Jaffe, Tierney, Pidvirny, Balkind and Abbasi34 The observed reduction in dACC activity following CBT may therefore either indicate improved efficiency in emotion regulation – for example, through a reduced need for cognitive control and limbic inhibition,Reference Dichter, Felder and Smoski35 given the concurrent hippocampal activity decrease – or improvements in maladaptive processes such as rumination. There was no significant association between dACC activity decreases and symptom changes, nor any differential changes in dACC activity between responders and non-responders, despite the main effect of time (objective b). This suggests that dACC activity alterations may be unrelated to symptom changes, although our analysis focused on overall symptoms rather than specific domains such as rumination, which could explain the lack of association. The specificity of dACC decreases during negative emotion processing to CBT remains unclear. While psychotherapies generally show reduced dACC activity,Reference Marwood, Wise, Perkins and Cleare36 electroconvulsive therapy (ECT)Reference Enneking, Dzvonyar, Dück, Dohm, Grotegerd and Förster37 and antidepressantsReference Fitzgerald, Laird, Maller and Daskalakis32 often exhibit ACC activity increases. However, evidence is mixed, with some studies reporting opposing effects following CBTReference Fu, Williams, Cleare, Scott, Mitterschiffthaler and Walsh12,Reference Yang, Oathes, Linn, Bruce, Satterthwaite and Cook14 and antidepressants.Reference Delaveau, Jabourian, Lemogne, Guionnet, Bergouignan and Fossati31 Further research should systematically compare different treatments to disentangle the effects of time, remission, treatment modality and common therapeutic factors on dACC activity, while also carefully considering ACC subregion variations.
Surprisingly, no significant amygdala activity changes were observed following naturalistic CBT, either between patients and healthy controls (objective a) or between responders and non-responders (objective b). This contrasts with studies showing reduced amygdala activity following treatments such as CBT,Reference Fu, Williams, Cleare, Scott, Mitterschiffthaler and Walsh12 antidepressantsReference Delaveau, Jabourian, Lemogne, Guionnet, Bergouignan and Fossati31 and ECT.Reference Redlich, Bürger, Dohm, Grotegerd, Opel and Zaremba38 Our sample included patients with milder symptoms, such as adjustment disorder and partially remitted major depressive order, which may have limited the scope for observable amygdala changes. Additionally, detecting significant effects in a small region like the amygdala with a modest sample size remains challenging. A brief discussion of the whole-brain results is provided in Supplement 11.
Cross-sectional group differences and response prediction
Contrary to previous studies,Reference Stuhrmann, Suslow and Dannlowski10,Reference Li and Wang11 we found no significant pre-treatment differences in amygdala, hippocampus or dACC activity between patients and healthy controls. Given the proposed links between depression severity and the extent of brain activity alterations,Reference Lemke, Probst, Warneke, Waltemate, Winter and Thiel39 the inclusion of outpatients with varying symptom levels may have obscured more pronounced group differences. Future studies should investigate activity differences based on symptom severity to clarify their influence on brain alterations. Our still relatively small sample size for cross-sectional analyses also probably limited the statistical power. Nevertheless, patients showed descriptively higher pre-treatment and lower post-treatment hippocampal and dACC activity compared with healthy controls, aligning with a previous study suggesting normalisation of limbic hyperactivity to the level of healthy controls following CBT.Reference Fu, Williams, Cleare, Scott, Mitterschiffthaler and Walsh12 Although descriptive activity differences hint that individuals with depression may require lower-than-average activity in these emotion-processing regions, caution is warranted due to the lack of significant findings. Consequently, our results do not clearly support the reversibility of depression-related activity alterations.
Concerning pre-treatment predictors of CBT response, responders showed elevated pre-treatment hippocampal activity compared with non-responders, despite no differences at post-treatment or in amygdala and dACC activity (objective c). This suggests that the potential pathophysiological differences that may underlie the observed hippocampal activity decrease in responders following CBT, and also explains the absence of treatment response observed in non-responders. However, another study has linked pre-treatment hippocampal hypoactivity during emotion processing with CBT response,Reference Rubin-Falcone, Weber, Kishon, Ochsner, Delaparte and Doré15 complicating the role of hippocampal hyperactivity as a predictive marker. The lack of pre-treatment dACC activity differences between responders and non-responders could hint at a response-independent effect of dACC activity decreases. It may also be explained by either the limited statistical power, the phenotypic variability of our sample or the use of an emotion-processing rather than a regulation paradigm.Reference Picó-Pérez, Alemany-Navarro, Dunsmoor, Radua, Albajes-Eizagirre and Vervliet40 Understanding why some patients respond while others do not remains an open and critical question, particularly because responders and non-responders in our sample did not significantly differ in regard to prior illness course, baseline depressive symptom severity, comorbidities, medication use or baseline brain activity relative to healthy controls. These findings imply that non-response cannot be fully explained by these factors alone, and point to the potential role of neurobiological differences. Indeed, the group-level distinction in pre-treatment hippocampal activity underscores depression’s neurobiological heterogeneity. However, our exploratory machine learning analyses indicate that functional imaging data are not yet reliable for predicting individual CBT response, with classification accuracy not exceeding 61%. This is in line with recent researchReference Winter, Blanke, Leenings, Ernsting, Fisch and Sarink24 raising questions about the clinical utility of brain imaging markers for response prediction. Larger samples and careful consideration of clinical heterogeneity may be needed to identify robust predictors of treatment outcome. Ultimately, pinpointing reliable neural markers could enable tailored interventions, such as more intensive CBT, alternative therapies or adjusted treatment durations.Reference Fu, Fan and Davatzikos41 Meanwhile, factors beyond the scope of this study, such as environmental influences (e.g. adverse life events, social support) or genetic predispositions, may also contribute to non-response, warranting further investigation to refine personalised treatment strategies.
Strengths and limitations
Our study comprehensively examined activity changes during negative emotion processing and their association with CBT response in patients with depressive disorders compared with healthy controls, while also exploring pre-treatment neural markers using group statistics and a machine learning approach. The naturalistic study design provides high external validity. Unlike previous studies, we defined CBT response based on SCID-I-validated changes in clinical diagnosis, offering a more reliable measure than symptom rating scales by capturing significant suffering and functional impairment, being less susceptible to weekly fluctuations and reflecting clinical progression throughout CBT. However, some limitations must be acknowledged. First, the naturalistic study design limited control over CBT interventions because sessions were not standardised, although psychotherapists followed established manuals and national care guidelines for unipolar depression.8 Second, the inclusion of patients with different depressive diagnoses adds heterogeneity, possibly explaining the absence of significant cross-sectional differences between patients and healthy controls and limiting the scope of our interpretations. However, this diversity and the application of naturalistic CBT provide higher external validity and a more realistic portrayal of those seeking CBT for depression in our healthcare system. Third, our paradigm raises uncertainty about whether the findings reflect emotion processing of facial stimuli or face processing in general. Future research should include a neutral face or stimulus-absent condition. Despite this, our paradigm is well established for exploring negative emotion processing.Reference Enneking, Dzvonyar, Dück, Dohm, Grotegerd and Förster37,Reference Borgers, Rinck, Enneking, Klug, Winter and Gruber42 Additionally, reliability constraints of paradigm-based fMRI and partial volume effects, particularly in regions close to grey–white matter boundaries, may impact result accuracy (e.g. the clusters in Fig. 1). Additional large-sample longitudinal studies are needed to validate our findings, especially given that pη 2 can overestimate effect size in small samples.
Following naturalistic CBT in patients with depressive disorders, reductions in both hippocampal and dACC activity were observed during negative emotion processing. The identified association between hippocampal activity decreases and response suggests that these changes may occur following only successful CBT, potentially reflecting improvements in biased emotion processing. Univariate analyses showed that responders and non-responders differed in pre-treatment hippocampal activity, although machine learning approaches did not support this finding. In contrast, dACC activity reductions do not seem to be linked to treatment response. Overall, these results provide insights into how naturalistic CBT may influence the neural circuitry underlying negative emotion processing in depressive disorders.
Supplementary material
The supplementary material can be found at https://doi.org/10.1192/bjp.2025.71
Data availability
The data that support the findings of this study are available from the corresponding author, R.R., upon reasonable request.
Acknowledgments
This study is supported by the German Center for Mental Health (DZPG).
Author contributions
T.B., E.Z., M.K., V.E. and R.R. contributed to the conception and design of the study. All authors contributed to organisation of the conduct of the study (including acquisition of data). T.B., E.Z., L.K., L.N. and R.R. analysed and interpreted the data. T.B., E.Z. and R.R. drafted the work, and all other authors revised it critically for important intellectual content. All authors have read and approved the final version of the manuscript.
Funding
The study was supported by grants from the German Research Foundation (DFG grant no. RE4458/1-1 to R.R., and SFB/TRR 393 grant no. 521379614 to U.D., S.M., E.J.L.), and by the Federal Ministry of Education and Research (BMBF) and the Ministry of Saxony-Anhalt within the initial phase of DZPG (BMBF grant no. 01EE2305C, to R.R.).
Declaration of interest
None.
Transparency declaration
We, T.B., E.Z. and R.R., confirm that this manuscript provides an honest, accurate and transparent representation of the reported study; that all significant aspects of the study have been included; and that any deviations from the original study plan have been fully explained.
Analytic code availability
The analytic code is available from the corresponding author, R.R., upon reasonable request.
Research material availability
The research material is available from the corresponding author, R.R., upon reasonable request.




eLetters
No eLetters have been published for this article.