Lithium is the first-line maintenance treatment for bipolar disorder, Reference Shorter1 an illness that frequently affects women of childbearing age, creating a significant clinical dilemma during the perinatal period. Reference Merikangas, Jin, He, Kessler, Lee and Sampson2,Reference Viguera, Nonacs, Cohen, Tondo, Murray and Baldessarini3 Although untreated bipolar disorder carries risks for both mother and infant, lithium use during pregnancy is associated with a dose-dependent risk of congenital malformations and adverse neonatal outcomes, Reference Mohamed, Elhelbawy, Khalid, Abdallatif and Lialy4–Reference Patorno, Huybrechts, Bateman, Cohen, Desai and Mogun6 leading to a decline in its prescription. This caution is reflected in UK National Institute for Health and Care Excellence (NICE) guidelines, 7,8 which advise considering discontinuation of lithium in pregnant women. Although the core safety information for lithium remained consistent across the 2006 and 2014 guideline updates, the 2014 guidance significantly strengthened the required standard of care, formalising preconception advice and multidisciplinary planning to manage the resulting high risk of postpartum maternal relapse. 8 The guidance maintained lithium as the preferred mood stabiliser compared with high-risk agents like valproate, focusing on rigorous therapeutic drug monitoring throughout gestation. 8,9 Although large-scale studies confirm that lithium use in pregnancy is low, Reference Hastie, Tong, Hiscock, Lindquist, Lindstrom and Wikstrom5 particularly in the UK, Reference Wittstrom, Cesta, Bateman, Bendix, Bliddal and Chan10 they lack the granularity to detail specific prescribing patterns, such as discontinuation rates and patient characteristics, following the 2014 NICE guideline update. Therefore, this study aims to fill this evidence gap by characterising the treatment patterns of lithium, including continuation, discontinuation and dosage trends, among pregnancies in the UK before, during and after pregnancy, from 1995 to 2018.
Method
Data source
We analysed the primary healthcare records of women in the Clinical Practice Research Datalink (CPRD) GOLD Pregnancy Register. CPRD GOLD maintains longitudinally collected data from primary care that uses Vision GP software in the UK, with a large sample population. Reference Herrett, Gallagher, Bhaskaran, Forbes, Mathur and Van Staa11 In the UK, over 98% of the population is registered with a general practitioner (GP) and access to specialist care is typically available only through a GP referral. Reference Herrett, Gallagher, Bhaskaran, Forbes, Mathur and Van Staa11 CPRD efficiently captures prescribing data, because primary care holds the prescribing budget for the National Health Service (NHS). 12 Although lithium may be initiated and dose adjustments made in secondary care, all ongoing and long-term prescribing is managed by primary care. CPRD uses READ codes for recording diagnoses and British National Formulary (BNF) codes for prescriptions. Reference Chisholm13 The CPRD GOLD Pregnancy Register was developed to identify pregnancy episodes among women aged 11–49 years, using an algorithm. Reference Minassian, Williams, Meeraus, Smeeth, Campbell and Thomas14 CPRD GOLD pseudonymises data and has no direct patient involvement, and thus the need for informed consent was waived for the current study. The study protocol was approved by the Clinical Practice Research Datalink Research Data Governance Committee (reference no. 20_000228).
Study populations
The study included pregnancies that started between 1 January 1995 and 31 December 2018 and that resulted in either a live or stillbirth. Early pregnancy losses and non-specific delivery outcomes were excluded. In accordance with the Pregnancy Register algorithm, Reference Minassian, Williams, Meeraus, Smeeth, Campbell and Thomas14 early pregnancy losses were defined as events occurring up to 12 completed weeks of gestation, encompassing miscarriages, terminations of pregnancy (TOP), ectopic pregnancies, molar pregnancies, blighted ova and unspecified losses. Although the register captures all documented outcomes, events specifically coded as TOP or ‘probable TOP’ are categorised within the early-loss hierarchy, with an assumed maximum duration of 24 weeks. Reference Minassian, Williams, Meeraus, Smeeth, Campbell and Thomas14 Clinical events occurring after 24 weeks are classified as delivery outcomes (live births or stillbirths), unless specifically coded otherwise in the source data. Reference Minassian, Williams, Meeraus, Smeeth, Campbell and Thomas14 Eligible women were registered with an ‘up to standard’ primary care practice for at least 12 months before their pregnancy began. Reference Minassian, Williams, Meeraus, Smeeth, Campbell and Thomas14 Pregnancy periods were defined as ‘first trimester’ from last menstrual period to 13 weeks of pregnancy, ‘second trimester’ from 14 to 26 weeks of pregnancy and ‘third trimester’ for those from 27 weeks to the end of pregnancy. Reference Minassian, Williams, Meeraus, Smeeth, Campbell and Thomas14 Multiple pregnancies, including cases of twins and several pregnancies from the same mother, were included in the analysis. Because this study describes prescribing patterns and prevalence, the primary unit of analysis is the pregnancy rather than the individual woman. Within the overall pregnancy register, 49.97% of women contributed two or more pregnancies. Reference Minassian, Williams, Meeraus, Smeeth, Campbell and Thomas14 Detailed information about the study populations and data sources can be found elsewhere. Reference Madley-Dowd, Rast, Ahlqvist, Zhong, Martin and Davies15
Identification of lithium prescriptions and exposure periods
We extracted information on lithium prescriptions from CPRD GOLD, which contains primary care records. It is important to note that this data-set does not capture prescriptions issued by specialists or those in secondary care settings. The BNF codes used to identify prescriptions are detailed in Supplementary Method 1 available at https://doi.org/10.1192/bjp.2026.10632. We assessed lithium exposure across several distinct periods, including 3-month intervals before and after pregnancy and for each of the three trimesters. A woman was considered as having been exposed to lithium during any given period if a lithium prescription started, ended or spanned that time frame.
Definition of perinatal prescribing patterns
To distinguish between prescribing patterns, we first established a baseline of pre-pregnancy prescription, defined as receiving at least two lithium prescriptions in the year before pregnancy (one in the 12 to 6 months prior, and another in the 6 months immediately preceding pregnancy) to ensure that we captured consistent users. Based on this, we defined the following mutually exclusive patterns:
Continuous prescription: met the criteria for pre-pregnancy prescription and had prescriptions in all three trimesters.
Pre-pregnancy discontinuation: met the criteria for pre-pregnancy prescription but had no prescriptions during any trimester.
Late discontinuation: met the criteria for pre-pregnancy prescription and had a first-trimester prescription, but no prescription in the third trimester (there may or may not have been a prescription during the second trimester).
Initiate during pregnancy: had a lithium prescription in at least one trimester but did not meet the criteria for pre-pregnancy use.
Initiate after delivery: had no lithium prescriptions in the 12 months before the pregnancy or during pregnancy but received a prescription after delivery.
Other patterns: included any prescribing sequence that did not fit the above definitions (see Supplementary Method 2 for further details).
Calculation and categorisation of lithium dosage
We calculated the daily dose (in milligrams) by multiplying tablet strength by the number of tablets prescribed per day. Reference Madley-Dowd, Rast, Ahlqvist, Zhong, Martin and Davies15 These doses primarily reflect lithium carbonate, which accounts for approximately 98% of lithium prescriptions in UK primary care. 16,17 We then categorised lithium doses as either low, medium or high based on the distribution of all prescriptions issued in the pre-pregnancy period. We calculated quartiles of the distribution of daily doses and then consolidated this information into a single dose-level variable, resulting in the following categories:
(a) low-dose: <600 mg/day (first quartile);
(b) moderate-dose: 600–800 mg/day (second and third quartiles);
(c) high-dose: >800 mg/day (fourth quartile).
The dose thresholds were determined to reflect the typical clinical practice, informed by Electronic Medicines Compendium (EMC) guidance, which emphasises individualised dosing based on serum lithium levels. Although the EMC does not specify formal dose thresholds, it notes that initial doses often begin at 400–600 mg, with maintenance dosing adjusted to therapeutical response. 18 If a woman had received prescriptions for different dosages within a single trimester, the highest daily dose was used for the analysis of that period.
Maternal characteristics
Lithium users and non-users
We described maternal and clinical characteristics between pregnancies with and without lithium prescriptions during the gestational period. These characteristics included age; ethnicity; the Index of Multiple Deprivation; smoking habits; body mass index (BMI); parity; frequency of primary care consultations in the year before the commencement of pregnancy; and co-prescription of antidepressants and antipsychotics in the same period. We identified the earliest recorded date of each maternal health diagnosis using information from the 3 years preceding the start of pregnancy. The full definitions of maternal health diagnoses and other covariates are provided in Supplementary Method 3 and 4.
Lithium continuation and discontinuation
Similar to the above, we compared pregnancies where lithium was continued throughout the gestation with those where it was discontinued.
Statistical analysis
Statistical analysis was conducted using Stata version 18 for Windows (StatCorp LLC, College Station, Texas, USA; https://www.stata.com). Baseline maternal and clinical characteristics of pregnancies exposed to lithium were compared descriptively with those unexposed during pregnancy, as well as for those who continued versus discontinued lithium use. Differences were analysed using chi-squared tests.
Prevalence of lithium prescription before, during and after pregnancy
We calculated the yearly prevalence of lithium prescriptions at any point before, during and after pregnancy for each calendar year, from 1 January 1995 to 31 December 2018. The annual prevalence of lithium prescription for each year was calculated by dividing the number of pregnancies prescribed lithium in that year by the total number of pregnancies under observation during the same year.
Pattern of lithium prescription before, during and after pregnancy
The NICE guidelines provide a framework for managing bipolar disorder using lithium, particularly with considerations for pregnancy. We evaluated the implementation of NICE guidelines over time frames 1995–2006, 2007–2014 and 2015–2018. These intervals were selected based on the publication years of these guidelines in 2006 and 2014. Implementation of NICE guidelines could inform lithium prescribing rates and patterns before, during and after pregnancy, which were explored across the stratified time periods.
We calculated the proportions of pregnancies where lithium was continued, discontinued pre-pregnancy and later into pregnancy, initiated during or after pregnancy and other patterns. These analyses were conducted overall and stratified by bipolar disorder. Additionally, we assessed variation in lithium doses (low, medium and high) during the perinatal period.
Results
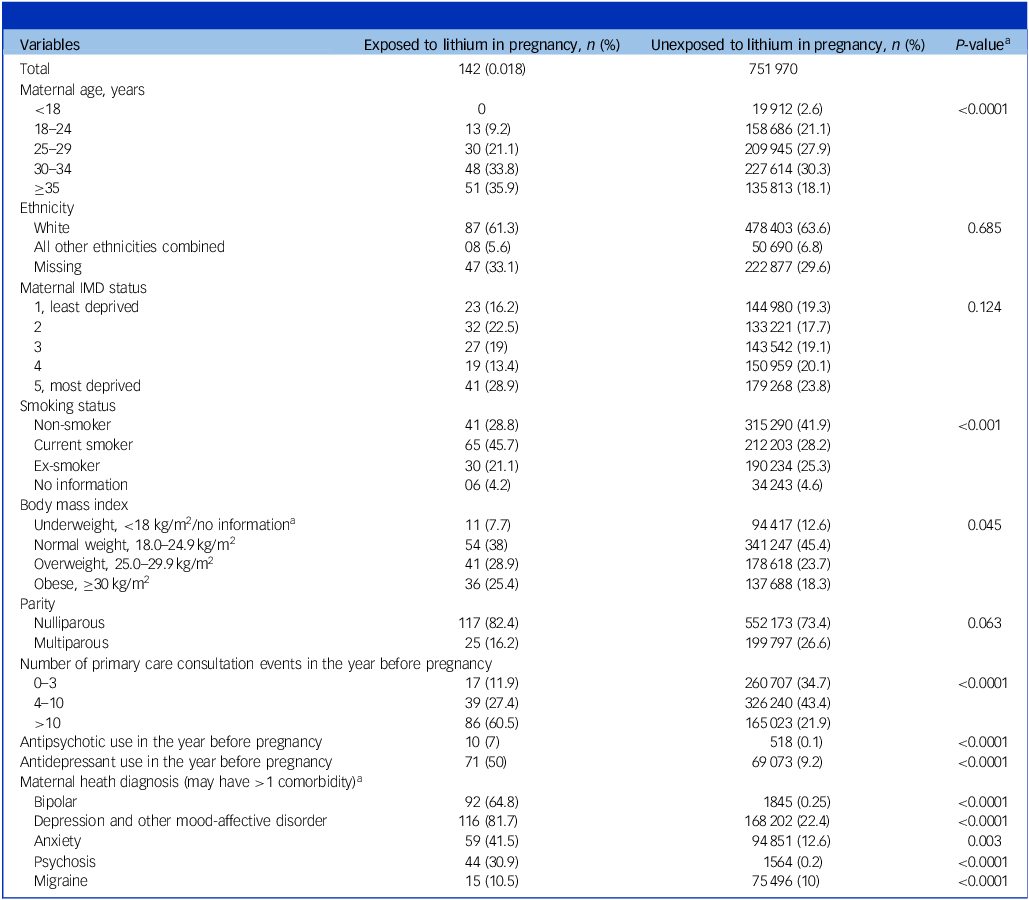
Table 1 summarises the characteristics of 752 112 eligible pregnancies identified between 1995 and 2018. In total, 337 (4.48 per 10 000) pregnancies were exposed to lithium at any time before, during or after pregnancy. Of these, 227 (3.02 per 10 000, 95% CI: 2.64, 3.44) were exposed to lithium before pregnancy, 142 (1.89 per 10 000, 95% CI: 1.59, 2.23) during pregnancy and 211 (2.81 per 10 000, 95% CI: 2.44, 3.21) after delivery. Compared with unexposed pregnancies, pregnancies exposed to lithium were more likely to be among women aged 35 and older; current smokers; women with a BMI ≥30; those with >10 primary care consultation events in the year before pregnancy; women who used antipsychotics and antidepressants in the year before pregnancy; and those with bipolar disorder, depression or other mood affective disorder, anxiety and psychosis as their comorbidities (P < 0.05; Table 1).
Characteristics of eligible pregnancies included in analyses, with and without lithium prescription during pregnancy

IMD, Index of Multiple Deprivation.
a Calculated using chi-squared test for categorical variables.
Yearly prevalence varied from 6.23 (95% CI: 3.12, 12.46) per 10 000 in 1996 to 0.49 (95% CI: 0.12, 1.96) per 10 000 in 2012, with the most recent prevalence (in 2018) of 1.03 (95% CI: 0.26, 4.11) per 10 000 (Fig. 1). Following an initial fluctuation in 1996, the annual prevalence of lithium use during pregnancy remained steady throughout the study period. Yearly prevalence is reported in Supplementary Table 1. Figure 2 illustrates the prevalence of lithium prescription among exposed pregnancies before, during and after pregnancy, stratified by the time periods (1995–2006, 2007–2014, 2015–2018) in relation to NICE guidelines published in 2006 and updated in 2014 (see Supplementary Table 2). During all three time periods the prevalence was more common before pregnancy, declined starting in the first trimester and remained low during the second and third trimesters. Notably, the introduction of the 2006 and 2014 NICE guidelines did not correspond with significant shifts in prescribing patterns, with lithium prevalence remaining stable across all 3 time periods.
Annual prevalence of lithium prescriptions during pregnancy, per 10 000 pregnancies in the UK, 1995–2018. The solid line represents the estimated annual prevalence and the shaded region denotes the 95% confidence interval. NICE, National Institute for Health and Care Excellence.

Prevalence of lithium prescriptions per 10 000 pregnancies, from 12 months before pregnancy to 12 months after delivery. Comparisons are shown across periods defined by the 2006 and 2014 National Institute for Health and Care Excellence guidelines. Vertical error bars indicate 95% confidence intervals for each estimate.

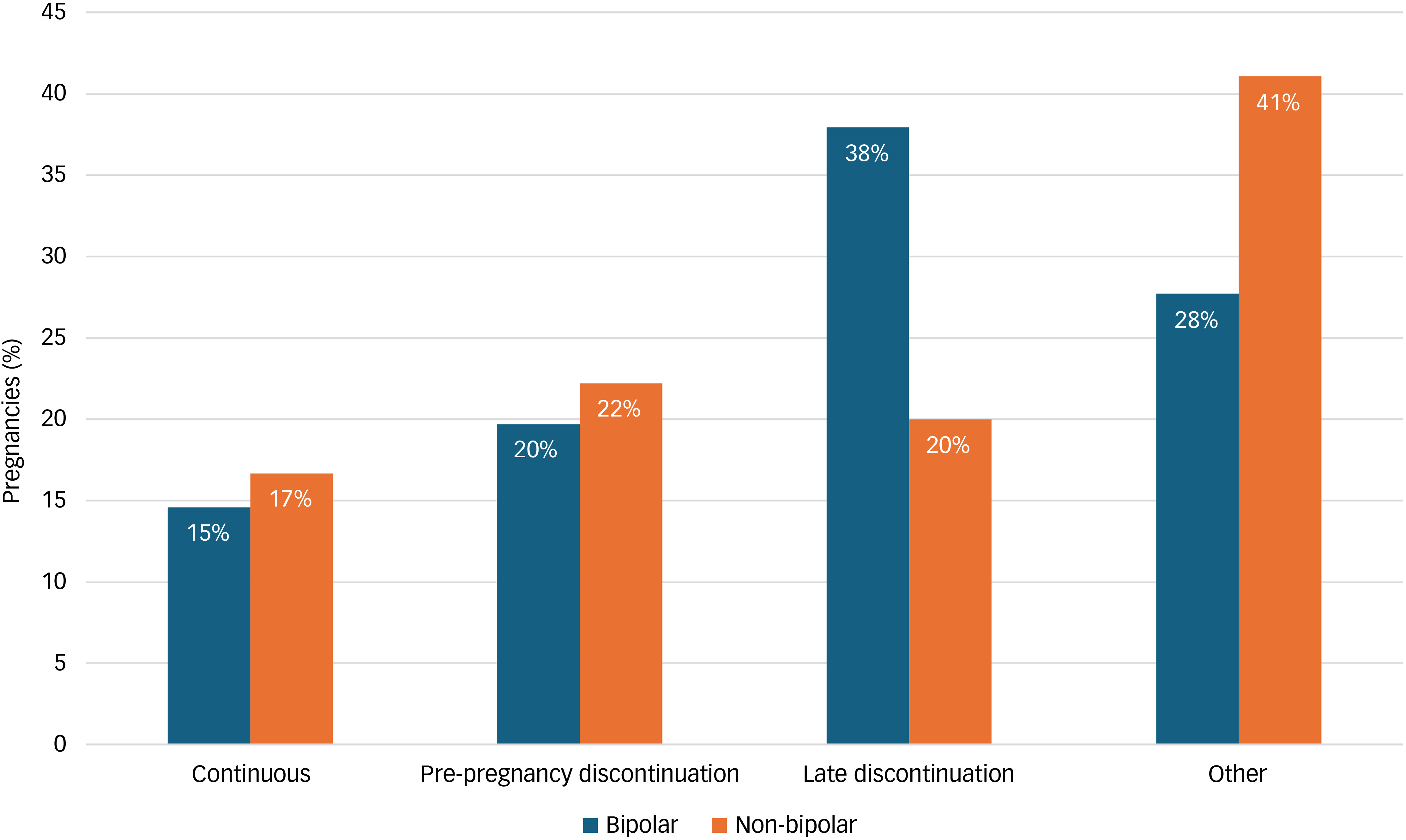
Of the 337 pregnancy episodes with any lithium prescription during the perinatal period, 163 (48.4%) were episodes where the women had a recorded diagnosis of bipolar disorder. This cohort included 227 pregnancies with established pre-pregnancy lithium use and 110 that initiated treatment during or after pregnancy. Among the 227 pregnancies during which lithium was used prior to conception, treatment discontinuation was the most common outcome. Only 15.4% (n = 35) continued treatment throughout all three trimesters. By contrast, 20.7% (n = 47) discontinued before pregnancy began and 30.8% (n = 70) discontinued later in pregnancy (during the second or third trimester). A third (33.0%, n = 75) followed other, more varied, prescribing patterns. These patterns differed based on maternal diagnosis (Fig. 3). Among the 137 pre-pregnancy users with bipolar disorder, 14.6% (n = 20) continued treatment whereas a majority either discontinued late (37.9%, n = 52) or before pregnancy (19.7%, n = 27). Among the 90 pre-pregnancy users without bipolar disorder, continuation (16.7%, n = 15) and discontinuation rates (42.2%, n = 38) were different, and a notably larger proportion (41.1%, n = 37) followed other prescribing patterns.
Lithium use patterns among pregnancies exposed to lithium before pregnancy, categorised by maternal bipolar disorder diagnosis. ‘Other’ category comprises remaining diagnoses that do not fit within the listed categories, and those of limited size.

We identified several differences between pregnancies where lithium was continued throughout pregnancy and those where it was discontinued (see Supplementary Table 3 for full details). Compared with pregnancies with continued use, those involving discontinued treatment were more likely to be in younger women (under 35 years, 66 v. 51%), in those having a diagnosis of bipolar disorder (68 v. 57%), in those residing in more deprived areas (62 v. 51%) and in those with a history of smoking (68 v. 54%). Conversely, women who continued treatment were more likely to have had fewer primary care consultations (≤10) in the year before pregnancy.
Among the 227 pregnancies with lithium exposure pre-conception, the most common dose prescribed before pregnancy was medium (600–800 mg/day; 51.1%), followed by low (<600 mg/day; 34.4%) and high (>800 mg/day; 14.5%). Data on dosage during pregnancy were limited, with dosage information available for only 129 pregnancies (56.8%) in the first trimester, declining to just 57 pregnancies (25.1%) by the third trimester. For pregnancies with available dosing data, the proportion requiring high doses (>800 mg/day) rose from 13.9% in the first trimester to 17.5% by the third trimester. The distribution of other prescribed doses for this smaller group across the trimesters is detailed in Supplementary Table 4.
Discussion
Interpretation of findings
This population-based study confirms that lithium use during pregnancy in UK primary care is rare and has remained so over two decades. Our calculated prevalence of 1.9 per 10 000 pregnancies aligns with previous UK and European reports, reinforcing the picture of widespread clinical caution. Whereas large, multinational studies have established this low prevalence, Reference Wittstrom, Cesta, Bateman, Bendix, Bliddal and Chan10 our analysis of patient-level data reveals that pregnancy for women taking lithium is most often characterised by treatment discontinuation. Our most significant finding is that fewer than one in six women on lithium prior to pregnancy continued the medication throughout all three trimesters. This high rate of discontinuation, particularly before or during the first trimester, may reflect clinician and patient concerns about lithium’s teratogenic risk. Reference Hidalgo-Mazzei, Mantingh, Perez de Mendiola, Samalin, Undurraga and Strejilevich19 However, this risk-mitigation strategy creates a clinical paradox. Discontinuing lithium, especially abruptly, is known to carry a high risk of relapse, particularly in the perinatal period when hormonal and psychosocial stressors are heightened. Reference Viguera, Nonacs, Cohen, Tondo, Murray and Baldessarini3 Therefore, although avoidance of fetal harm is a primary driver of discontinuation, it concurrently exposes the mother to the significant morbidity associated with untreated bipolar disorder.
A novel finding from our study is the difference in discontinuation timing based on diagnosis. Women with bipolar disorder were more likely to discontinue later in pregnancy compared with those without a bipolar diagnosis. This finding is suggestive of a more complex and prolonged risk–benefit calculation for this group, where the immediate risk of maternal relapse may delay the decision to stop treatment. This is a purely descriptive finding, and its interpretation is constrained: first, by the substantial contribution of the ‘other patterns’ category in Fig. 3, which indicates significant heterogeneity in discontinuation across diagnoses; and second, by the potential for unmeasured or unanalysed confounding by other relevant factors, such as deprivation and engagement with healthcare services, which may also influence the observed timing of discontinuation. Our analysis also reveals that the non-bipolar group is primarily composed of women with severe depression (91.7%) and anxiety (54.2%), suggesting that lithium is frequently utilised as an augmentation therapy for treatment-resistant unipolar illness (Supplementary Table 5). Reference Bauer, Adli, Ricken, Severus and Pilhatsch20
Furthermore, our data on dosage reflect the clinical complexity of managing bipolar disorder during pregnancy. Dose reduction across pregnancy is probably an attempt to minimise fetal exposure. However, the physiological changes of pregnancy, including increased glomerular filtration rate and increased lithium clearance, may necessitate a higher dose to maintain a therapeutic serum level. Reference Wesseloo, Wierdsma, van Kamp, Munk-Olsen, Hoogendijk and Kushner21 Although our findings show that the proportion of high-dose prescriptions (>800 mg) rose from 13.9% in the first trimester to 17.5% by the third, the majority of pregnancies remained on stable or lower doses throughout the gestational period. These patterns highlight the need for individualised monitoring to balance the risks of maternal relapse against those of fetal exposure, particularly because physiological demands peak in the third trimester.
Strengths and limitations
The primary strength of this study is its use of CPRD GOLD, a large, nationally representative and longitudinal data-set that provides robust, real-world data on prescribing trends over 24 years. Reference Herrett, Gallagher, Bhaskaran, Forbes, Mathur and Van Staa11 The validated pregnancy register enhances the reliability of our perinatal cohort. Reference Minassian, Williams, Meeraus, Smeeth, Campbell and Thomas14
However, several limitations must be acknowledged. First, our data are restricted to primary care records. Although the majority of lithium prescriptions in the UK are issued by GPs under shared-care arrangements with secondary services, some patients may receive prescriptions via in-patient services. Reference Herrett, Gallagher, Bhaskaran, Forbes, Mathur and Van Staa11 If these were not captured in the primary care record, our findings could have overestimated the rate of discontinuation. However, because long-term lithium management is typically reflected in GP records for reimbursement and monitoring purposes, we believe that the observed trends remain a strong indicator of prescribing patterns in the UK. Second, CPRD does not record treatment indications or illness severity, preventing us from definitively knowing why lithium was prescribed, or correlating patterns with the intensity of the underlying condition. Third, missing data for variables such as BMI and smoking status may impact the interpretation of user characteristics. Fourth, prescription data are not a direct measure of medication adherence. Fifth, our study primarily captures lithium exposure in pregnancies resulting in live births or stillbirths, and it probably underestimates total first-trimester prevalence by excluding early pregnancy losses. Based on established miscarriage rates in lithium-exposed populations, Reference Poels, Kamperman, Vreeker, Gilden, Boks and Kahn22 we estimate that approximately 20% of the initial cohort were excluded as early losses (Supplementary Method 5); however, given that such losses are often confounded by underlying maternal illness, Reference Boden, Lundgren, Brandt, Reutfors, Andersen and Kieler23 their exclusion was a methodological necessity to ensure data fidelity and accurate trimester-specific windows. Finally, as this study is primarily descriptive in nature, we did not perform multivariate statistical modelling to control for confounding factors. Although we have provided univariate comparisons for baseline characteristics, the lack of adjusted analyses means that the associations reported should be interpreted strictly as descriptive differences between the cohorts.
Clinical implications and future directions
Our findings reflect the prevailing clinical landscape in the UK, where lithium use during pregnancy is characterised by caution and frequent treatment interruption. The high rates of discontinuation are often part of a coordinated clinical strategy to minimise fetal exposure while preparing for a prompt postpartum restart. Given that the risk of relapse is particularly acute in the postpartum period, Reference Viguera, Nonacs, Cohen, Tondo, Murray and Baldessarini3,Reference Wesseloo, Kamperman, Munk-Olsen, Pop, Kushner and Bergink24 this approach is a recognised and pragmatic management option. However, to ensure the success of this strategy, proactive perinatal planning remains essential. This reinforces guidance from expert bodies such as the Royal College of Psychiatrists and the UK Teratology Information Service, which emphasise facilitating planned, rapid re-initiation of treatment immediately after delivery for those who opt to discontinue during pregnancy. 25,26 For the majority who discontinue lithium, risk-mitigation strategies include preconception counselling to facilitate a planned, gradual tapering and the establishment of ‘rapid-response’ plans for medication re-initiation should mood destabilisation occur. For the minority who continue lithium, therapeutic drug monitoring is recommended to navigate the significant physiological changes of pregnancy and the immediate postpartum period. Reference Wesseloo, Wierdsma, van Kamp, Munk-Olsen, Hoogendijk and Kushner21 By providing tailored support for both clinical pathways, clinicians can better manage the high risk of postpartum relapse while respecting the patient’s treatment preferences.
Future research should prioritise linking primary and secondary care data to create a complete picture of perinatal lithium prescribing. Qualitative studies are also needed to understand the decision-making process from both patient and clinician perspectives. Finally, long-term follow-up studies are crucial to understanding the outcomes for both mothers who discontinue lithium and their children, providing the evidence needed to refine future clinical guidelines.
In conclusion, although the low prevalence of lithium use reflects a cautious clinical approach, the high rate of discontinuation highlights the importance of integrated and proactive perinatal mental health frameworks. Such strategies are essential to support patients through this clinical transition, and to ensure that robust plans are in place for the postpartum period, when the risk of relapse is most acute.
Supplementary material
The supplementary material is available online at https://doi.org/10.1192/bjp.2026.10632
Data availability
This study is based on data from CPRD, obtained under licence from the UK Medicines and Healthcare products Regulatory Agency. The data are not publicly available due to patient confidentiality and licensing restrictions. Access to CPRD data is subject to protocol approval by the CPRD Research Data Governance process. Researchers may apply for access to CPRD data via the CPRD website (https://www.cprd.com). The authors do not have permission to share the data directly. The analytical codes that support the findings of this study are available on request from the corresponding author, B.K.L.
Author contributions
B.K.L. and D.R. developed the research idea. D.C. and B.K.L. formulated the original research question and designed the study. D.C. conducted the analysis, interpreted the results and provided the initial drafts of the study findings. D.R. and J.F.H. contributed clinical and subject matter expertise during the study design and interpretation of results. P.M.-D., V.H.A., J.E.R., F.Z.M. and C.M. contributed feedback on the study design. P.M.-D. accessed and verified the data for the study. D.C. drafted the initial manuscript and B.K.L. contributed to the final editing. All authors critically reviewed and revised the manuscript and approved the final version. D.C. and B.K.L. hold final responsibility for the decision to submit the manuscript for publication.
Funding
This study was supported by NIH nos 1R01NS107607 (B.K.L., D.C., J.E.R., V.H.A., D.R., C.M.) and 1R01NS131433 (B.K.L., J.E.R., V.H.A., D.C.). This study was also supported by the NIHR Biomedical Research Centre at the University of Bristol and University Hospitals Bristol and Weston NHS Foundation Trust (no. NIHR203315; P.M.-D., D.R.). P.M.-D. and D.R. work in the Medical Research Council Integrative Epidemiology Unit at the University of Bristol, which is supported by the UK Medical Research Council and the University of Bristol (nos MC_UU_00032/02, MC_UU_00032/06; P.M.-D., D.R.). V.H.A. is funded by the Swedish Society for Medical Research (no. PG-24-0427). No funder had any role in the study design, data collection, analysis or interpretation, writing of the report or in the decision to submit the article for publication. The views expressed are those of the authors and not necessarily those of any of the funders or organisations they represent.
Declaration of interest
J.F.H. is a member of the Editorial Board for the British Journal of Psychiatry and was not involved in the peer review or decision-making process for this manuscript. B.K.L. reports receiving consulting fees from Beasley Allen Law Firm, Patterson Belknap Webb & Tyler LLP, Gerson Lehrman Group and AlphaSights outside of this work. V.H.A. has received speaker fees from Angelini Pharma outside of the submitted work. D.C. reports that this work forms part of her PhD thesis, and that she serves as a contracted service provider for Amgen Inc., unrelated to and outside of the submitted study. P.M.-D., D.R., C.M., J.E.R. and F.Z.M. have nothing to disclose.
Ethical standards
Ethical approval was obtained from the CPRD Research Data Governance Committee (reference no. 20_000228). Informed consent from study participants was not obtained, because CPRD pseudonymises data and there was no direct patient involvement.





eLetters
No eLetters have been published for this article.