Introduction
Major depressive disorder (MDD) is a leading cause of disability worldwide and a major public health concern (Kennedy et al., Reference Kennedy, Lam, McIntyre, Tourjman, Bhat, Blier, Hasnain, Jollant, Levitt, MacQueen, McInerney, McIntosh, Milev, Müller, Parikh, Pearson, Ravindran and Uher2016). Antidepressant medications targeting monoaminergic systems, as well as evidence-based psychotherapies such as cognitive-behavioral therapy, behavioral activation, and interpersonal therapies, have demonstrated efficacy (Kennedy et al., Reference Kennedy, Lam, McIntyre, Tourjman, Bhat, Blier, Hasnain, Jollant, Levitt, MacQueen, McInerney, McIntosh, Milev, Müller, Parikh, Pearson, Ravindran and Uher2016). However, 30%–40% of patients fail to achieve full remission even after several treatment phases, rendering their depressive episode treatment-resistant (Demyttenaere & Van Duppen, Reference Demyttenaere and Van Duppen2019; Nemeroff, Reference Nemeroff2007). Following two consecutive antidepressant failures, the probability of a patient’s response to a third treatment attempt falls below 10% (Rush et al., Reference Rush, Trivedi, Wisniewski, Nierenberg, Stewart, Warden, Niederehe, Thase, Lavori, Lebowitz, McGrath, Rosenbaum, Sackeim, Kupfer, Luther and Fava2006; Souery et al., Reference Souery, Amsterdam, de Montigny, Lecrubier, Montgomery, Lipp, Racagni, Zohar and Mendlewicz1999). Treatment-resistant depression (TRD) is associated with marked impairments in global functioning, elevated suicide risk, and persistent cognitive deficits, particularly in memory, attention, processing speed, and executive functions (Rock et al., Reference Rock, Roiser, Riedel and Blackwell2014; Snyder, Reference Snyder2013). These cognitive disturbances often persist despite mood remission and contribute to functional disability and vulnerability to depressive relapse.
A promising non-monoaminergic target is the glutamatergic system. Ketamine, a non-competitive N-methyl-D-aspartate (NMDA) receptor antagonist, first introduced as an anesthetic in the 1970s, has shown rapid antidepressant effects at subanesthetic doses. In Reference Berman, Cappiello, Anand, Oren, Heninger, Charney and Krystal2000, Berman and colleagues reported a robust, rapid (within 24 hours) but transient antidepressant response following a single intravenous (IV) ketamine infusion (0.5 mg/kg over 40 minutes) in seven patients with TRD. This landmark study, since replicated in numerous randomized controlled trials, marked a paradigm shift in the treatment of depression (Marcantoni et al., Reference Marcantoni, Akoumba, Wassef, Mayrand, Lai, Richard-Devantoy and Beauchamp2020). Though evidence remains limited, the Berman dosing protocol remains the most widely used and has shown comparable efficacy to electroconvulsive therapy (ECT) in non-psychotic TRD (Jha et al., Reference Jha, Wilkinson, Krishnan, Collins, Sanacora, Murrough, Goes, Altinay, Aloysi, Asghar-Ali, Barnett, Chang, Costi, Malone, Nikayin, Nissen, Ostroff, Reti, Wolski and Anand2024).
While the antidepressant effects of IV ketamine are well established, its impact on cognitive functioning remains less clearly defined (Gill et al., Reference Gill, Gill, Rodrigues, Lipsitz, Rosenblat, El-Halabi, Nasri, Mansur, Lee and McIntyre2021). Preliminary evidence suggests potential enhancements in processing speed and working memory through glutamatergic and neuroplasticity-related mechanisms (Murrough et al., Reference Murrough, Iosifescu, Chang, Al Jurdi, Green, Perez, Iqbal, Pillemer, Foulkes, Shah, Charney and Mathew2013). The systematic review by Gill et al. (Reference Gill, Gill, Rodrigues, Lipsitz, Rosenblat, El-Halabi, Nasri, Mansur, Lee and McIntyre2021) identified improvements in processing speed and verbal memory but did not include studies published in the past 5 years and did not address cognitive processes associated with suicidal behaviors. Since then, several recent trials (Kumpf et al., Reference Kumpf, Wilkinson, Hu, Chen, Kamini, Chakrabarti, Rhee, Grezmak, Mathew, Sanacora, Murrough, Goes, Collins, Barnett and Anand2025; Oughli et al., Reference Oughli, Gebara, Ciarleglio, Lavretsky, Brown, Flint, Farber, Karp, Mulsant, Reynolds, Roose, Yang, Butters and Lenze2023; Permoda-Osip et al., Reference Permoda-Osip, Kisielewski, Bartkowska-Sniatkowska and Rybakowski2015; Phillips et al., Reference Phillips, Van Geel, Burhunduli, Vasudev, Batten, Norris, Talbot, Ortiz, Owoeye and Blier2022; Richard-Devantoy et al., Reference Richard-Devantoy, Berlim and Jollant2014; Zavaliangos-Petropulu et al., Reference Zavaliangos-Petropulu, McClintock, Khalil, Joshi, Taraku, Al-Sharif, Espinoza and Narr2023; Zhou et al., Reference Zhou, Wang, Lan, Li, Chao, Wu, McIntyre and Ning2022) have expanded the evidence base by exploring cognition beyond mood improvement, partially including domains relevant to suicidal cognition, such as executive control and verbal fluency (Richard-Devantoy et al., Reference Richard-Devantoy, Berlim and Jollant2014, Reference Richard-Devantoy, Berlim, Garel, Inja and Turecki2024).
The aim of the present study was therefore to conduct a systematic and critical review of the literature on the cognitive effects of IV ketamine in TRD, with particular attention to executive functions and cognitive processes related to suicidal behaviors.
Materials and methods
Search strategy and selection criteria
Cochrane, MEDLINE, Embase, and PsycINFO databases were searched up to May 15, 2025, using the terms ‘major depressive disorder’, ‘treatment-resistant depression’, ‘intravenous ketamine’, ‘cognition’, and their synonyms. Search strategies combined controlled vocabulary (e.g. MeSH, Emtree) and free-text terms. Searches were limited to human studies in adults. To reduce heterogeneity, studies involving esketamine or intranasal ketamine were excluded. Inclusion criteria were as follows: (1) adult participants with treatment-resistant major depressive disorder (TRD); (2) the use of single or multiple IV ketamine infusions; intranasal/esketamine and non-IV routes were excluded; (3) cognition assessed as an outcome; (4) publication in a peer-reviewed journal; and (5) articles published in English or French. This review was conducted in accordance with the PRISMA guidelines, and the protocol was registered in PROSPERO (ID: 1160487).
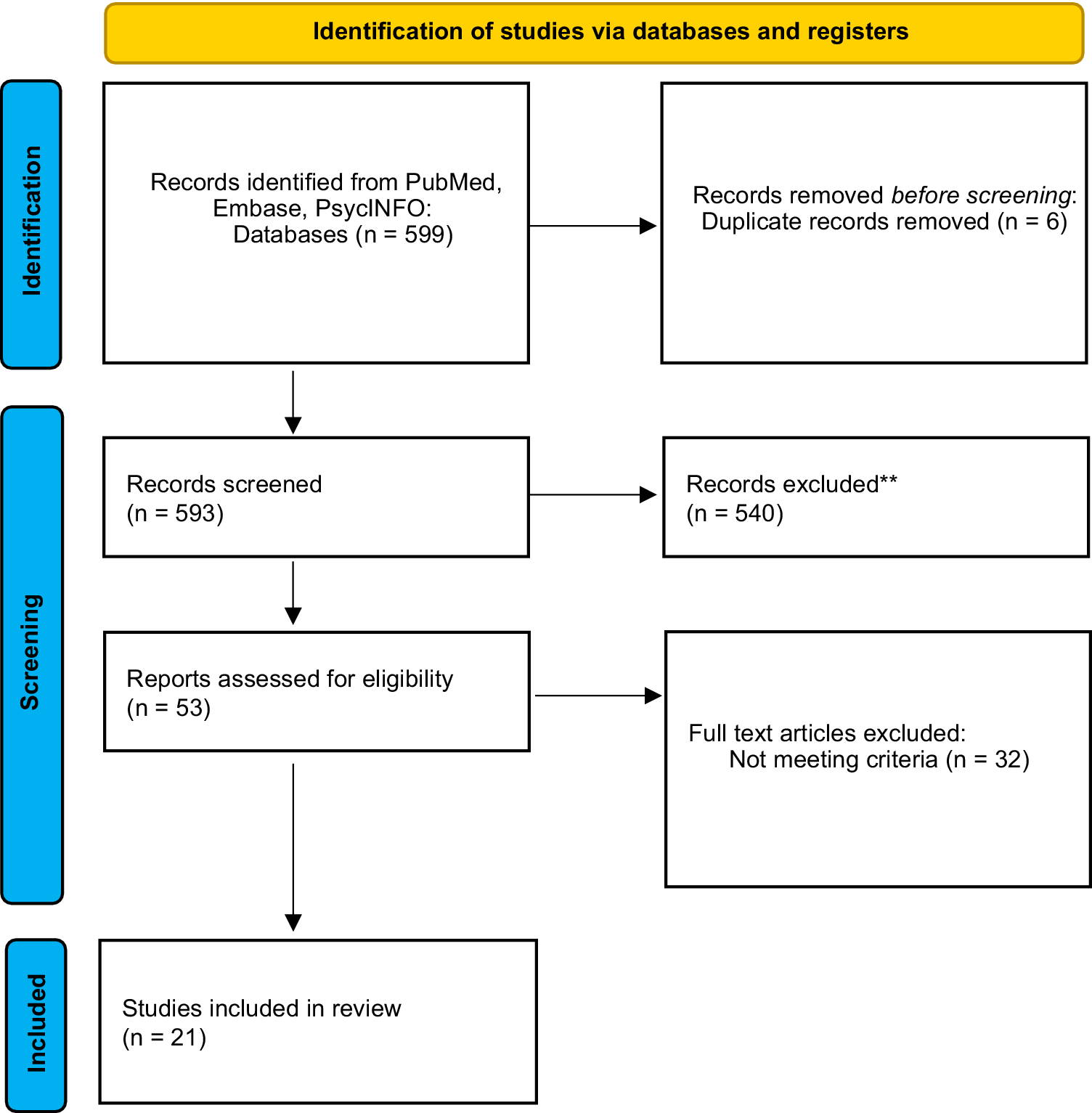
The database search initially identified 599 records. After removal of 6 duplicates, 593 records were screened at the title and abstract level, resulting in 540 exclusions. Fifty-three full-text articles were assessed for eligibility; 32 were excluded primarily for not meeting TRD or IV-ketamine criteria. In total, 21 studies met the inclusion criteria (Figure 1), including several recent trials (Chen et al., Reference Chen, Li, Lin, Hong, Tu, Bai, Cheng and Su2018, Reference Chen, Lin, Li, Tsai, Wu, Bai, Hong, Tu and Su2022; Jha et al., Reference Jha, Wilkinson, Krishnan, Collins, Sanacora, Murrough, Goes, Altinay, Aloysi, Asghar-Ali, Barnett, Chang, Costi, Malone, Nikayin, Nissen, Ostroff, Reti, Wolski and Anand2024; Keilp et al., Reference Keilp, Madden, Marver, Frawley A, Burke, Mohammad, Gluck, Mann and Grunebaum2021; Kumpf et al., Reference Kumpf, Wilkinson, Hu, Chen, Kamini, Chakrabarti, Rhee, Grezmak, Mathew, Sanacora, Murrough, Goes, Collins, Barnett and Anand2025; Liu et al., Reference Liu, Zhou, Zheng, Wang, Zhan, Lan, Zhang, Li, Chen and Ning2019; McIntyre et al., Reference McIntyre, Rosenblat, Nemeroff, Sanacora, Murrough, Berk, Brietzke, Dodd, Gorwood, Ho, Iosifescu, Lopez Jaramillo, Kasper, Kratiuk, Lee, Lee, Lui, Mansur, Papakostas and Stahl2021; Murrough et al., Reference Murrough, Burdick, Levitch, Perez, Brallier, Chang, Foulkes, Charney, Mathew and Iosifescu2015; Oughli et al., Reference Oughli, Gebara, Ciarleglio, Lavretsky, Brown, Flint, Farber, Karp, Mulsant, Reynolds, Roose, Yang, Butters and Lenze2023; Permoda-Osip et al., Reference Permoda-Osip, Kisielewski, Bartkowska-Sniatkowska and Rybakowski2015; Phillips et al., Reference Phillips, Van Geel, Burhunduli, Vasudev, Batten, Norris, Talbot, Ortiz, Owoeye and Blier2022; Price et al., Reference Price, Iosifescu, Murrough, Chang, Al Jurdi, Iqbal, Soleimani, Charney, Foulkes and Mathew2014; Shiroma et al., Reference Shiroma, Albott, Johns, Thuras, Wels and Lim2014, Reference Shiroma, Thuras, Wels, Albott, Erbes, Tye and Lim2020; Singh et al., Reference Singh, Parikh, Vande Voort, Pazdernik, Achtyes, Goes, Yocum, Nykamp, Becerra, Smart, Greden, Bobo, Frye, Burdick and Ryan2024; Zavaliangos-Petropulu et al., Reference Zavaliangos-Petropulu, McClintock, Khalil, Joshi, Taraku, Al-Sharif, Espinoza and Narr2023; Zheng et al., Reference Zheng, Zhou, Liu, Wang, Zhan, Li, Chen, Li and Ning2019; Zhou et al., Reference Zhou, Wang, Lan, Li, Chao, Wu, McIntyre and Ning2022, Reference Zhou, Wang, Lan, Zheng, Li, Chao, Wu, McIntyre and Ning2021, Reference Zhou, Zheng, Liu, Wang, Zhan, Li, Chen, Li and Ning2018). Given the overlap between some research groups, there is a potential for sample overlap across certain studies (Chen et al., Reference Chen, Li, Lin, Hong, Tu, Bai, Cheng and Su2018, Reference Chen, Lin, Li, Tsai, Wu, Bai, Hong, Tu and Su2022; Zhou et al., Reference Zhou, Wang, Lan, Li, Chao, Wu, McIntyre and Ning2022, Reference Zhou, Wang, Lan, Zheng, Li, Chao, Wu, McIntyre and Ning2021, Reference Zhou, Zheng, Liu, Wang, Zhan, Li, Chen, Li and Ning2018). Each study was retained when it reported distinct cognitive outcomes, analyses, or time points; however, this may lead to duplications in participant counts.
Prisma flow chart for studies identified through databases and registers.

Data collection and extraction
Abstracts retrieved through the search were independently screened by three reviewers (LAC, SRD, and VDJ) to determine eligibility. Full texts were then obtained for studies that met the inclusion criteria. For each eligible study, the following data were independently extracted: (1) study characteristics (first author, year, country, eligibility criteria, and sample size per group); (2) participant characteristics (age, sex, and psychiatric diagnoses); (3) ketamine treatment details (dose, frequency, route of administration, and duration); (4) depression measures; (5) cognitive measures (neuropsychological tests, cognitive domains assessed, and timing of assessment); and (6) a summary of the study’s main findings. When multiple publications originated from the same registered trial or clearly shared cohort, identified based on trial registration number, recruitment site, recruitment period, and study design, we designated a primary cohort report for participant counting and overall synthesis. Additional publications from the same cohort were retained when they contributed distinct cognitive domains, mediation analyses, or non-overlapping follow-up intervals not reported in the primary report. This strategy was implemented to enhance methodological transparency and to prevent inflation of the total sample size due to cohort reuse.
Outcomes
The primary outcome was a change in cognitive performance across domains, assessed using standardized neuropsychological tests. Owing to substantial heterogeneity in study design, ketamine protocols, timing of assessment, and cognitive measures, quantitative meta-analysis was not feasible. Therefore, a structured narrative synthesis using descriptive vote-counting was conducted to summarize the number of studies reporting improvement, no change, or worsening within each cognitive domain.
Although this approach does not yield pooled effect size estimates or formally weight studies by sample size, larger randomized trials were given particular interpretative emphasis in the narrative discussion. This strategy allowed identification of consistent patterns across heterogeneous studies while maintaining transparency regarding methodological variability.
Assessment of methodological quality
The methodological quality of the included studies was assessed independently by three reviewers using validated tools recommended by the Cochrane Collaboration. Inter-rater agreement between the three reviewers was substantial (Fleiss’ κ = 0.72), indicating a reliable assessment of study eligibility and risk of bias. Randomized controlled trials (RCTs) were evaluated with the Cochrane Risk of Bias 2 (RoB 2) tool, which considers five domains: (1) randomization process, (2) deviations from intended interventions, (3) missing outcome data, (4) measurement of the outcomes, and (5) selection of the reported results. Non-randomized or open-label studies were evaluated with the Risk of Bias in Non-randomized Studies of Interventions (ROBINS-I) tool, which covers seven domains: confounding variables, selection of participants, classification of interventions, deviations from intended interventions, missing data, measurement of outcomes, and selection of the reported results. Discrepancies were resolved through consensus. Given the heterogeneity across study designs, cognitive assessment tools, and timing of post-treatment evaluations, a quantitative meta-analysis was not feasible. Therefore, findings were synthesized narratively, emphasizing patterns of improvement across cognitive domains. Risk-of-bias outcomes for all included studies are summarized in Table 1.
Risk of bias assessment of included studies (RoB 2 and ROBINS-I)

Results
Study characteristics
A total of 21 studies were included in the systematic review, encompassing approximately 1,180 reported participants overall (Figure 1). However, accounting for confirmed cohort overlap – most notably across the Zhou et al. (Reference Zhou, Zheng, Liu, Wang, Zhan, Li, Chen, Li and Ning2018, Reference Zhou, Wang, Lan, Zheng, Li, Chao, Wu, McIntyre and Ning2021, Reference Zhou, Wang, Lan, Li, Chao, Wu, McIntyre and Ning2022) publications originating from a single registered trial (ChiCTR-OOC-17012239), related reports from the same Guangzhou research program, and secondary analyses of the ELEKT-D trial – as well as possible overlap within additional single-center programs, the number of unique participants likely ranges between approximately 900 and 1,180. The aggregated sample comprised approximately 900 patients treated with IV ketamine, about 230–250 comparator participants across midazolam, placebo, or ECT conditions, and a small group of healthy controls. Exact totals vary due to differences in study design, sample structure, and partial cohort reuse. A detailed cohort map grouping publications into probable cohort families is provided in Supplementary Table S1.
Across studies, the mean age was approximately 40–41 years, and 55–58% of participants were male. Ketamine was administered intravenously in all studies, most commonly at a subanesthetic dose of 0.5 mg/kg, with a median of six infusions delivered over approximately 12 days. Baseline cognitive evaluation was conducted prior to the first infusion in nearly all trials. Table 2 summarizes the main study characteristics and cognitive outcomes.
Summary of articles from the systematic literature review studying the association between cognition and Ketamine in treatment resistant depression

AMI-SF, Columbia autobiographical memory interview; BD, bipolar disorder; CDRS-R, children’s depression rating; CogState, Cogstate battery; COWAT, controlled oral word association test; CPT, continuous performance test; CRT, choice reaction test; C-SSRS: Columbia-suicide severity rating; CVLT-II, California verbal learning test-II; DSST, digit symbol substitution test; HAMD, Hamilton depression rating scale; HC, healthy controls; HVLT-R, Hopkins verbal learning test revised; IAT, implicit association test; KET, ketamine; PC, patient controls; MADRS, Montgomery-Asberg depression rating scale; MATRICS, measurement and treatment research to improve cognition in schizophrenia; MCCB, MATRICS consensus cognitive battery; M, men; MID, midazolam; MoCA, Montreal cognitive assessment; MMSE, mini-mental state examination; NART-35, North American adult reading test-35; N-Back Task, N-back task; NIHToolbox, NIH toolbox for the assessment of neurological and behavioral function; PDQ-D5, Perceived Deficits Questionnaire - Depression; POMS, profile of mood states; QIDS-SR-16, quick inventory of depressive symptoms – short version; RT, reaction time; RBANS, repeatable battery for the assessment of neuropsychological status; SCWT, stroop color and word test; SRT, simple reaction time; SSI, scale for suicide ideation; TDM, resistant major depression; TMT A et B, trail making test A et B; WAI-II, working alliance inventory – second version; WAIS-II, Wechsler adult intelligence scale.
Most included studies explicitly excluded participants with current alcohol or substance use disorders, frequently requiring negative urine toxicology screening and excluding substance abuse or dependence within the preceding 3–6 months. These exclusions were reported in the majority of randomized controlled trials. One large randomized trial (ELEKT-D secondary analysis) reported inclusion of a small proportion of participants with substance use comorbidity (Kumpf et al., Reference Kumpf, Wilkinson, Hu, Chen, Kamini, Chakrabarti, Rhee, Grezmak, Mathew, Sanacora, Murrough, Goes, Collins, Barnett and Anand2025), whereas a small number of studies did not clearly specify substance-related exclusion criteria (Kumar et al., 2024; Permoda-Osip et al., Reference Permoda-Osip, Kisielewski, Bartkowska-Sniatkowska and Rybakowski2015; Price et al., Reference Price, Iosifescu, Murrough, Chang, Al Jurdi, Iqbal, Soleimani, Charney, Foulkes and Mathew2014). Overall, the available evidence primarily reflects TRD populations without active substance misuse.
Timing of baseline cognitive assessment
The timing of pre-treatment cognitive assessment varied slightly across studies. In most trials, cognition was evaluated within 1 week before the first infusion (Chen et al., Reference Chen, Lin, Li, Tsai, Wu, Bai, Hong, Tu and Su2022; Liu et al., Reference Liu, Zhou, Zheng, Wang, Zhan, Lan, Zhang, Li, Chen and Ning2019; Murrough et al., Reference Murrough, Burdick, Levitch, Perez, Brallier, Chang, Foulkes, Charney, Mathew and Iosifescu2015; Oughli et al., Reference Oughli, Gebara, Ciarleglio, Lavretsky, Brown, Flint, Farber, Karp, Mulsant, Reynolds, Roose, Yang, Butters and Lenze2023; Shiroma et al., Reference Shiroma, Albott, Johns, Thuras, Wels and Lim2014; Zhou et al., Reference Zhou, Wang, Lan, Zheng, Li, Chao, Wu, McIntyre and Ning2021). Chen et al. (Reference Chen, Li, Lin, Hong, Tu, Bai, Cheng and Su2018) assessed cognition 2 days prior to infusion, whereas Zheng et al. (Reference Zheng, Zhou, Liu, Wang, Zhan, Li, Chen, Li and Ning2019) and Price et al. (Reference Price, Iosifescu, Murrough, Chang, Al Jurdi, Iqbal, Soleimani, Charney, Foulkes and Mathew2014) did not specify the exact timing of baseline evaluation.
More recent studies generally performed baseline assessments either on the day of the first infusion (Permoda-Osip et al., Reference Permoda-Osip, Kisielewski, Bartkowska-Sniatkowska and Rybakowski2015; Phillips et al., Reference Phillips, Van Geel, Burhunduli, Vasudev, Batten, Norris, Talbot, Ortiz, Owoeye and Blier2022; Singh et al., Reference Singh, Parikh, Vande Voort, Pazdernik, Achtyes, Goes, Yocum, Nykamp, Becerra, Smart, Greden, Bobo, Frye, Burdick and Ryan2024), within the week before treatment initiation (Kumpf et al., Reference Kumpf, Wilkinson, Hu, Chen, Kamini, Chakrabarti, Rhee, Grezmak, Mathew, Sanacora, Murrough, Goes, Collins, Barnett and Anand2025; Zavaliangos-Petropulu et al., Reference Zavaliangos-Petropulu, McClintock, Khalil, Joshi, Taraku, Al-Sharif, Espinoza and Narr2023), or prior to randomization (Zhou et al., Reference Zhou, Wang, Lan, Li, Chao, Wu, McIntyre and Ning2022).
Timing of post-treatment cognitive assessments
The timing of post-treatment cognitive assessments also varied considerably across studies.
Shiroma et al. (Reference Shiroma, Albott, Johns, Thuras, Wels and Lim2014) conducted repeated assessments after each infusion (days 3, 5, 8, 10, and 12), while Murrough et al. (Reference Murrough, Burdick, Levitch, Perez, Brallier, Chang, Foulkes, Charney, Mathew and Iosifescu2015) evaluated cognition 7 days post-infusion. Chen et al. (Reference Chen, Li, Lin, Hong, Tu, Bai, Cheng and Su2018) included both short-term (day 3) and medium-term (day 14) assessments.
Oughli et al. (Reference Oughli, Gebara, Ciarleglio, Lavretsky, Brown, Flint, Farber, Karp, Mulsant, Reynolds, Roose, Yang, Butters and Lenze2023) used a longitudinal design with evaluations after six infusions across 3 weeks, and again 1 month later, focusing on episodic memory, processing speed, and executive functions. Phillips et al. (Reference Phillips, Norris, Talbot, Birmingham, Hatchard, Ortiz, Owoeye, Batten and Blier2019) performed repeated testing after a single infusion (2 hours, 1 day, and 7 days) to track temporal evolution.
More recent studies extended follow-up windows: Phillips et al. (Reference Phillips, Van Geel, Burhunduli, Vasudev, Batten, Norris, Talbot, Ortiz, Owoeye and Blier2022) assessed cognition at day 1, week 1, and week 4, and Zhou et al. (Reference Zhou, Wang, Lan, Li, Chao, Wu, McIntyre and Ning2022) evaluated participants after six infusions and again at 2- and 4-week follow-up. Zavaliangos-Petropulu et al. (Reference Zavaliangos-Petropulu, McClintock, Khalil, Joshi, Taraku, Al-Sharif, Espinoza and Narr2023) measured outcomes at day 3, day 7, and week 4; Permoda-Osip et al. (Reference Permoda-Osip, Kisielewski, Bartkowska-Sniatkowska and Rybakowski2015) reassessed participants 3 days after the final infusion. Lastly, Singh et al. (Reference Singh, Parikh, Vande Voort, Pazdernik, Achtyes, Goes, Yocum, Nykamp, Becerra, Smart, Greden, Bobo, Frye, Burdick and Ryan2024) included additional evaluations at the end of the acute phase and during the continuation phase, and Kumpf et al. (Reference Kumpf, Wilkinson, Hu, Chen, Kamini, Chakrabarti, Rhee, Grezmak, Mathew, Sanacora, Murrough, Goes, Collins, Barnett and Anand2025) compared cognitive performance at weeks 3 and 6 between IV ketamine and ECT treatments.
Cognitive domains assessed
Across the 21 studies, cognitive domains evaluated included working memory (16 studies, 76.2%), processing speed (15 studies, 71.4%), attention (11 studies, 52.4%), verbal memory (13 studies, 61.9%), visual memory (9 studies, 42.9%), episodic memory (4 studies, 19%), cognitive flexibility (8 studies, 38.1%), executive control (8 studies, 38.1%), and implicit cognition (1 study, 4.8%).
A synthesis of outcomes and the tests performed is presented in Table 2. The most frequent procognitive effects were observed in processing speed and working memory, whereas results for verbal and visual memory were more heterogeneous. In most cases, improvements in working memory and attention correlated with antidepressant response, while gains in processing speed and executive control were often independent of mood change. A detailed synthesis of improvement versus non-improvement across domains is presented in Table 3. For domain-level counts, publications arising from the same cohort were considered as separate reports when they contributed distinct cognitive domains or follow-up intervals. Cohort overlap is detailed in Supplementary Table S1 and should be considered when interpreting frequency counts.
Cognitive domains improved or not with IV Ketamine in treatment resistant depression

Processing speed
Across the included studies, processing speed was one of the most consistently improved domains following IV ketamine treatment. Thirteen of 15 studies reported significant post-treatment gains, often emerging after multiple infusions and persisting up to four weeks after the last session (Oughli et al., Reference Oughli, Gebara, Ciarleglio, Lavretsky, Brown, Flint, Farber, Karp, Mulsant, Reynolds, Roose, Yang, Butters and Lenze2023; Permoda-Osip et al., Reference Permoda-Osip, Kisielewski, Bartkowska-Sniatkowska and Rybakowski2015; Zavaliangos-Petropulu et al., Reference Zavaliangos-Petropulu, McClintock, Khalil, Joshi, Taraku, Al-Sharif, Espinoza and Narr2023). Recent trials have further strengthened this observation. Zhou et al. (Reference Zhou, Wang, Lan, Li, Chao, Wu, McIntyre and Ning2022) reported significant improvement on MATRICS processing speed tasks, partially independent of mood response. Permoda-Osip et al. (Reference Permoda-Osip, Kisielewski, Bartkowska-Sniatkowska and Rybakowski2015) found enhanced performance on the Trail Making Test (TMT) and Stroop speed measures, also unrelated to depressive symptom change. In Singh et al. (Reference Singh, Parikh, Vande Voort, Pazdernik, Achtyes, Goes, Yocum, Nykamp, Becerra, Smart, Greden, Bobo, Frye, Burdick and Ryan2024), immediate memory improvement correlated with depression reduction, whereas gains in attention and language were independent of mood change. Similarly, Chen et al. (Reference Chen, Lin, Li, Tsai, Wu, Bai, Hong, Tu and Su2022) reported faster completion times on psychomotor speed tasks.
In comparative studies, Kumpf et al. (Reference Kumpf, Wilkinson, Hu, Chen, Kamini, Chakrabarti, Rhee, Grezmak, Mathew, Sanacora, Murrough, Goes, Collins, Barnett and Anand2025) demonstrated that processing speed (Digit Symbol Substitution Test, TMT-A) and cognitive flexibility (COWAT, Stroop interference) were better preserved in ketamine-treated patients than in those receiving ECT. These cognitive benefits were largely independent of changes in depressive symptoms. Conversely, Phillips et al. (Reference Phillips, Van Geel, Burhunduli, Vasudev, Batten, Norris, Talbot, Ortiz, Owoeye and Blier2022) and a minority of earlier studies did not find significant effects (Murrough et al., Reference Murrough, Burdick, Levitch, Perez, Brallier, Chang, Foulkes, Charney, Mathew and Iosifescu2015).
Working memory
Working memory was the most frequently examined cognitive domain, typically assessed using the Digit Span, n-back, or Trail Making Test–B tasks.
Across studies, nine of 16 trials reported significant improvements following ketamine treatment, often in parallel with mood improvement (Chen et al., Reference Chen, Li, Lin, Hong, Tu, Bai, Cheng and Su2018, Reference Chen, Lin, Li, Tsai, Wu, Bai, Hong, Tu and Su2022; Singh et al., Reference Singh, Parikh, Vande Voort, Pazdernik, Achtyes, Goes, Yocum, Nykamp, Becerra, Smart, Greden, Bobo, Frye, Burdick and Ryan2024; Zavaliangos-Petropulu et al., Reference Zavaliangos-Petropulu, McClintock, Khalil, Joshi, Taraku, Al-Sharif, Espinoza and Narr2023; Zhou et al., Reference Zhou, Wang, Lan, Zheng, Li, Chao, Wu, McIntyre and Ning2021). In the RBANS-based trial by Singh et al. (Reference Singh, Parikh, Vande Voort, Pazdernik, Achtyes, Goes, Yocum, Nykamp, Becerra, Smart, Greden, Bobo, Frye, Burdick and Ryan2024), both immediate and delayed memory scores improved significantly after three infusions and remained stable during the continuation phase, indicating sustained enhancement in working memory and attention. Similarly, Chen et al. (Reference Chen, Lin, Li, Tsai, Wu, Bai, Hong, Tu and Su2022) found improved performance on working memory and attentional control tasks in the higher-dose ketamine group, suggesting dose-dependent facilitation of executive resources. Zhou et al. (Reference Zhou, Wang, Lan, Li, Chao, Wu, McIntyre and Ning2022) also reported significant gains on MATRICS working memory measures correlated with antidepressant response, while Zavaliangos-Petropulu et al. (Reference Zavaliangos-Petropulu, McClintock, Khalil, Joshi, Taraku, Al-Sharif, Espinoza and Narr2023) observed modest but consistent improvements on NIH Toolbox working memory scores. Permoda-Osip et al. (Reference Permoda-Osip, Kisielewski, Bartkowska-Sniatkowska and Rybakowski2015) identified discrete gains on complex executive working memory tasks, such as TMT-B and Stroop interference; however, these were unrelated to mood change. In contrast, Phillips et al. (Reference Phillips, Van Geel, Burhunduli, Vasudev, Batten, Norris, Talbot, Ortiz, Owoeye and Blier2022) and Murrough et al. (Reference Murrough, Burdick, Levitch, Perez, Brallier, Chang, Foulkes, Charney, Mathew and Iosifescu2015) reported no significant effects, whereas Kumpf et al. (Reference Kumpf, Wilkinson, Hu, Chen, Kamini, Chakrabarti, Rhee, Grezmak, Mathew, Sanacora, Murrough, Goes, Collins, Barnett and Anand2025) reported that working memory was preserved but not improved compared to ECT.
Verbal memory
Verbal memory, typically assessed using immediate and delayed recall tasks such as the California Verbal Learning Test (CVLT), Hopkins Verbal Learning Test (HVLT), or story recall measures, was examined in most trials.
Eight studies out of 13 reported significant improvements in verbal memory following repeated ketamine infusions, frequently independent of changes in depressive symptoms (Singh et al., Reference Singh, Parikh, Vande Voort, Pazdernik, Achtyes, Goes, Yocum, Nykamp, Becerra, Smart, Greden, Bobo, Frye, Burdick and Ryan2024; Zavaliangos-Petropulu et al., Reference Zavaliangos-Petropulu, McClintock, Khalil, Joshi, Taraku, Al-Sharif, Espinoza and Narr2023; Zhou et al., Reference Zhou, Wang, Lan, Li, Chao, Wu, McIntyre and Ning2022, Reference Zhou, Zheng, Liu, Wang, Zhan, Li, Chen, Li and Ning2018). Specifically, Singh et al. (Reference Singh, Parikh, Vande Voort, Pazdernik, Achtyes, Goes, Yocum, Nykamp, Becerra, Smart, Greden, Bobo, Frye, Burdick and Ryan2024) demonstrated robust improvements in immediate and delayed memory as well as language performance after three infusions, with effects persisting during the continuation phase. Zhou et al. (Reference Zhou, Wang, Lan, Li, Chao, Wu, McIntyre and Ning2022) similarly observed gains in verbal learning and recall on MATRICS tasks, while Zavaliangos-Petropulu et al. (Reference Zavaliangos-Petropulu, McClintock, Khalil, Joshi, Taraku, Al-Sharif, Espinoza and Narr2023) noted stable improvement across serial assessments. In contrast, Phillips et al. (Reference Phillips, Van Geel, Burhunduli, Vasudev, Batten, Norris, Talbot, Ortiz, Owoeye and Blier2022), Oughli et al. (Reference Oughli, Gebara, Ciarleglio, Lavretsky, Brown, Flint, Farber, Karp, Mulsant, Reynolds, Roose, Yang, Butters and Lenze2023) and Permoda-Osip et al. (Reference Permoda-Osip, Kisielewski, Bartkowska-Sniatkowska and Rybakowski2015) found no significant post-treatment change, and Kumpf et al. (Reference Kumpf, Wilkinson, Hu, Chen, Kamini, Chakrabarti, Rhee, Grezmak, Mathew, Sanacora, Murrough, Goes, Collins, Barnett and Anand2025) reported that although verbal fluency did not increase, it was better preserved in the ketamine group compared with patients receiving ECT.
Visual memory
Visual memory, encompassing visuospatial recall, constructional ability, and complex figure learning, was evaluated in several trials. Four studies out of nine reported significant improvement following repeated ketamine infusions, generally after six to seven sessions, with effects largely independent of mood improvement (Shiroma et al., Reference Shiroma, Albott, Johns, Thuras, Wels and Lim2014; Singh et al., Reference Singh, Parikh, Vande Voort, Pazdernik, Achtyes, Goes, Yocum, Nykamp, Becerra, Smart, Greden, Bobo, Frye, Burdick and Ryan2024; Zavaliangos-Petropulu et al., Reference Zavaliangos-Petropulu, McClintock, Khalil, Joshi, Taraku, Al-Sharif, Espinoza and Narr2023; Zhou et al., Reference Zhou, Wang, Lan, Li, Chao, Wu, McIntyre and Ning2022). For instance, Zhou et al. (Reference Zhou, Wang, Lan, Li, Chao, Wu, McIntyre and Ning2022) documented enhanced performance on MATRICS visual learning tasks, while Zavaliangos-Petropulu et al. (Reference Zavaliangos-Petropulu, McClintock, Khalil, Joshi, Taraku, Al-Sharif, Espinoza and Narr2023) observed gains on visual episodic recall that persisted up to 4 weeks post-treatment. In the RBANS-based study by Singh et al. (Reference Singh, Parikh, Vande Voort, Pazdernik, Achtyes, Goes, Yocum, Nykamp, Becerra, Smart, Greden, Bobo, Frye, Burdick and Ryan2024), improvements were also noted on visuospatial/constructional indices during the continuation phase, suggesting potential enhancement of visual encoding and retrieval processes with sustained treatment. Conversely, Oughli et al. (Reference Oughli, Gebara, Ciarleglio, Lavretsky, Brown, Flint, Farber, Karp, Mulsant, Reynolds, Roose, Yang, Butters and Lenze2023), Phillips et al. (Reference Phillips, Van Geel, Burhunduli, Vasudev, Batten, Norris, Talbot, Ortiz, Owoeye and Blier2022), and Permoda-Osip et al. (Reference Permoda-Osip, Kisielewski, Bartkowska-Sniatkowska and Rybakowski2015) did not observe significant post-infusion changes, and Kumpf et al. (Reference Kumpf, Wilkinson, Hu, Chen, Kamini, Chakrabarti, Rhee, Grezmak, Mathew, Sanacora, Murrough, Goes, Collins, Barnett and Anand2025) found visual memory to be preserved but not improved relative to ECT.
Executive functions
Executive functions were evaluated across studies through measures of executive control, inhibitory control, cognitive flexibility, and implicit cognition. Significant improvements following repeated ketamine infusion were reported, particularly on tasks engaging prefrontal regulation and conflict monitoring.
Inhibitory control. Four studies out of four reported significant improvement following repeated ketamine infusion. Improvements in Stroop performance were observed within 1–3 days after ketamine infusion, consistent with enhanced interference control (Keilp et al., Reference Keilp, Madden, Marver, Frawley A, Burke, Mohammad, Gluck, Mann and Grunebaum2021; Permoda-Osip et al., Reference Permoda-Osip, Kisielewski, Bartkowska-Sniatkowska and Rybakowski2015). Most improvements in inhibitory control occurred independently of changes in depressive symptoms, as reported across several studies (Keilp et al., Reference Keilp, Madden, Marver, Frawley A, Burke, Mohammad, Gluck, Mann and Grunebaum2021; Kumpf et al., Reference Kumpf, Wilkinson, Hu, Chen, Kamini, Chakrabarti, Rhee, Grezmak, Mathew, Sanacora, Murrough, Goes, Collins, Barnett and Anand2025; Oughli et al., Reference Oughli, Gebara, Ciarleglio, Lavretsky, Brown, Flint, Farber, Karp, Mulsant, Reynolds, Roose, Yang, Butters and Lenze2023; Permoda-Osip et al., Reference Permoda-Osip, Kisielewski, Bartkowska-Sniatkowska and Rybakowski2015). Phillips et al. (Reference Phillips, Norris, Talbot, Birmingham, Hatchard, Ortiz, Owoeye, Batten and Blier2019, Reference Phillips, Van Geel, Burhunduli, Vasudev, Batten, Norris, Talbot, Ortiz, Owoeye and Blier2022) reported gains on Stroop and Go/No-Go tasks; however, these effects were attenuated after adjusting for mood improvement. Zavaliangos-Petropulu et al. (Reference Zavaliangos-Petropulu, McClintock, Khalil, Joshi, Taraku, Al-Sharif, Espinoza and Narr2023) found reduced interference scores and improved inhibitory control following six infusions, whereas Oughli et al. (Reference Oughli, Gebara, Ciarleglio, Lavretsky, Brown, Flint, Farber, Karp, Mulsant, Reynolds, Roose, Yang, Butters and Lenze2023) reported stable performance in older adults, indicating cognitive safety even in vulnerable populations. Notably, Keilp et al. (Reference Keilp, Madden, Marver, Frawley A, Burke, Mohammad, Gluck, Mann and Grunebaum2021) demonstrated that improved Stroop performance paralleled the rapid antisuicidal effects of ketamine, supporting a link between inhibitory control and the modulation of cognitions related to suicidal behaviors.
Cognitive flexibility. Oughli et al. (Reference Oughli, Gebara, Ciarleglio, Lavretsky, Brown, Flint, Farber, Karp, Mulsant, Reynolds, Roose, Yang, Butters and Lenze2023) and Zavaliangos-Petropulu et al. (Reference Zavaliangos-Petropulu, McClintock, Khalil, Joshi, Taraku, Al-Sharif, Espinoza and Narr2023) both documented significant improvements in set-shifting and mental flexibility tasks (e.g. TMT-B, switching paradigms). Most improvements in cognitive flexibility occurred independently of changes in depressive symptoms, supporting a direct procognitive effect of ketamine. Similarly, Kumpf et al. (Reference Kumpf, Wilkinson, Hu, Chen, Kamini, Chakrabarti, Rhee, Grezmak, Mathew, Sanacora, Murrough, Goes, Collins, Barnett and Anand2025) reported superior preservation of flexibility and verbal fluency in ketamine-treated patients compared to those receiving ECT, suggesting that ketamine may protect against ECT-related executive slowing.
Implicit cognition
Price et al. (Reference Price, Iosifescu, Murrough, Chang, Al Jurdi, Iqbal, Soleimani, Charney, Foulkes and Mathew2014) uniquely assessed implicit suicidal cognition using the Implicit Association Test (IAT). A significant reduction in maladaptive implicit associations was observed among clinical responders, underscoring ketamine’s potential influence on automatic, non-conscious processes relevant to suicidal risk.
Other cognitive outcomes
Attention was generally stable or modestly improved following ketamine treatment. Five studies out of 11 reported significant improvement following repeated ketamine infusions. Zavaliangos-Petropulu et al. (Reference Zavaliangos-Petropulu, McClintock, Khalil, Joshi, Taraku, Al-Sharif, Espinoza and Narr2023) reported stable attentional performance across six infusions using NIH Toolbox measures, while Phillips et al. (Reference Phillips, Van Geel, Burhunduli, Vasudev, Batten, Norris, Talbot, Ortiz, Owoeye and Blier2022) and Permoda-Osip et al. (Reference Permoda-Osip, Kisielewski, Bartkowska-Sniatkowska and Rybakowski2015) found maintained or slightly improved scores after a single infusion.
Recent data confirmed and extended these findings: Zhou et al. (Reference Zhou, Wang, Lan, Li, Chao, Wu, McIntyre and Ning2022) demonstrated significant gains in sustained attention on MATRICS tasks; Singh et al. (Reference Singh, Parikh, Vande Voort, Pazdernik, Achtyes, Goes, Yocum, Nykamp, Becerra, Smart, Greden, Bobo, Frye, Burdick and Ryan2024) observed parallel improvements in attention and language that persisted during the continuation phase; and Oughli et al. (Reference Oughli, Gebara, Ciarleglio, Lavretsky, Brown, Flint, Farber, Karp, Mulsant, Reynolds, Roose, Yang, Butters and Lenze2023) reported preserved attentional functioning in older adults without evidence of decline. In the ELEKT-D trial, Kumpf et al. (Reference Kumpf, Wilkinson, Hu, Chen, Kamini, Chakrabarti, Rhee, Grezmak, Mathew, Sanacora, Murrough, Goes, Collins, Barnett and Anand2025) further demonstrated that attention remained stable in ketamine-treated patients, in contrast to the transient attentional slowing observed in those receiving ECT.
Risk of bias assessment
The final selection of studies includes several recent trials, which expand the evidence base beyond early proof-of-concept reports. Risk-of-bias assessment indicated predominantly low to some concerns among randomized trials (RoB 2), while non-randomized studies frequently showed serious risk due to confounding (ROBINS-I) (Kumpf et al., Reference Kumpf, Wilkinson, Hu, Chen, Kamini, Chakrabarti, Rhee, Grezmak, Mathew, Sanacora, Murrough, Goes, Collins, Barnett and Anand2025; Oughli et al., Reference Oughli, Gebara, Ciarleglio, Lavretsky, Brown, Flint, Farber, Karp, Mulsant, Reynolds, Roose, Yang, Butters and Lenze2023; Phillips et al., Reference Phillips, Van Geel, Burhunduli, Vasudev, Batten, Norris, Talbot, Ortiz, Owoeye and Blier2022; Permoda-Osip et al., Reference Permoda-Osip, Kisielewski, Bartkowska-Sniatkowska and Rybakowski2015; Singh et al., Reference Singh, Parikh, Vande Voort, Pazdernik, Achtyes, Goes, Yocum, Nykamp, Becerra, Smart, Greden, Bobo, Frye, Burdick and Ryan2024; Zavaliangos-Petropulu et al., Reference Zavaliangos-Petropulu, McClintock, Khalil, Joshi, Taraku, Al-Sharif, Espinoza and Narr2023; Zhou et al., Reference Zhou, Wang, Lan, Li, Chao, Wu, McIntyre and Ning2022). Table 1 provides the full risk-of-bias ratings for each study according to RoB 2 and ROBINS-I criteria. These risk-of-bias considerations further informed the interpretative emphasis placed on larger randomized trials in the narrative synthesis.
Discussion
This systematic review indicates that IV ketamine administered at subanesthetic doses in patients with TRD is associated with improvements in selected cognitive domains, most consistently in processing speed, working memory, and attention (Table 3). Approximately three quarters of included reports described procognitive effects, (Chen et al., Reference Chen, Li, Lin, Hong, Tu, Bai, Cheng and Su2018, Reference Chen, Lin, Li, Tsai, Wu, Bai, Hong, Tu and Su2022; Keilp et al., Reference Keilp, Madden, Marver, Frawley A, Burke, Mohammad, Gluck, Mann and Grunebaum2021; Kumpf et al., Reference Kumpf, Wilkinson, Hu, Chen, Kamini, Chakrabarti, Rhee, Grezmak, Mathew, Sanacora, Murrough, Goes, Collins, Barnett and Anand2025; Liu et al., Reference Liu, Zhou, Zheng, Wang, Zhan, Lan, Zhang, Li, Chen and Ning2019; Oughli et al., Reference Oughli, Gebara, Ciarleglio, Lavretsky, Brown, Flint, Farber, Karp, Mulsant, Reynolds, Roose, Yang, Butters and Lenze2023; Permoda-Osip et al., Reference Permoda-Osip, Kisielewski, Bartkowska-Sniatkowska and Rybakowski2015; Price et al., Reference Price, Iosifescu, Murrough, Chang, Al Jurdi, Iqbal, Soleimani, Charney, Foulkes and Mathew2014; Shiroma et al., Reference Shiroma, Albott, Johns, Thuras, Wels and Lim2014, Reference Shiroma, Thuras, Wels, Albott, Erbes, Tye and Lim2020; Singh et al., Reference Singh, Parikh, Vande Voort, Pazdernik, Achtyes, Goes, Yocum, Nykamp, Becerra, Smart, Greden, Bobo, Frye, Burdick and Ryan2024; Zavaliangos-Petropulu et al., Reference Zavaliangos-Petropulu, McClintock, Khalil, Joshi, Taraku, Al-Sharif, Espinoza and Narr2023; Zheng et al., Reference Zheng, Zhou, Liu, Wang, Zhan, Li, Chen, Li and Ning2019; Zhou et al., Reference Zhou, Wang, Lan, Li, Chao, Wu, McIntyre and Ning2022, Reference Zhou, Wang, Lan, Zheng, Li, Chao, Wu, McIntyre and Ning2021, Reference Zhou, Zheng, Liu, Wang, Zhan, Li, Chen, Li and Ning2018), whereas the remaining studies reported limited or absent effects (Chen et al., Reference Chen, Lin, Li, Tsai, Wu, Bai, Hong, Tu and Su2022; Murrough et al., Reference Murrough, Burdick, Levitch, Perez, Brallier, Chang, Foulkes, Charney, Mathew and Iosifescu2015; Phillips et al., Reference Phillips, Norris, Talbot, Birmingham, Hatchard, Ortiz, Owoeye, Batten and Blier2019, Reference Phillips, Van Geel, Burhunduli, Vasudev, Batten, Norris, Talbot, Ortiz, Owoeye and Blier2022).
Among the most replicated findings, working memory and processing speed improved after repeated infusions, with some effects persisting for up to 4 weeks. Gains in working memory were often linked to antidepressant response, while improvements in processing speed appeared partially independent of mood change, suggesting distinct underlying cognitive mechanisms. Ketamine may exert direct neurocognitive effects distinct from its antidepressant action. For example, Chen et al. (Reference Chen, Lin, Li, Tsai, Wu, Bai, Hong, Tu and Su2022) reported faster completion times on psychomotor speed tasks, suggesting a potential facilitation of cognitive efficiency even in patients with higher baseline depressive severity. Overall, ketamine appears to enhance processing speed and aspects of executive functioning, with effects that are partly independent of mood change and may reflect direct glutamatergic neuroplasticity. Working memory tends to improve or at least stabilize, particularly after repeated infusions, with effects that often parallel antidepressant response, potentially mediated by glutamatergic modulation of frontoparietal networks.
Effects on verbal and visual memory were more heterogeneous, with some studies reporting mood-independent improvements (Singh et al., Reference Singh, Parikh, Vande Voort, Pazdernik, Achtyes, Goes, Yocum, Nykamp, Becerra, Smart, Greden, Bobo, Frye, Burdick and Ryan2024; Zhou et al., Reference Zhou, Wang, Lan, Li, Chao, Wu, McIntyre and Ning2022). Ketamine may enhance or preserve verbal memory and language-related performance, particularly after repeated infusions. However, the magnitude and persistence of these effects remain heterogeneous, likely influenced by differences in cognitive batteries, follow-up duration, and baseline severity of cognitive impairment. Evidence indicates that ketamine may confer modest benefits on visual and visuospatial memory, particularly following repeated or extended treatment. However, findings remain inconsistent across study designs and cognitive batteries. Finally, ketamine does not impair attentional performance and may confer mild procognitive effects, particularly in selective attention domains. These findings reinforce ketamine’s favorable neurocognitive safety relative to traditional antidepressant interventions.
Executive control emerged as a particularly robust cognitive target. Improvements in Stroop performance were observed across several independent cohorts, with some effects detectable within 24 hours of infusion and persisting beyond the acute phase (Keilp et al., Reference Keilp, Madden, Marver, Frawley A, Burke, Mohammad, Gluck, Mann and Grunebaum2021; Permoda-Osip et al., Reference Permoda-Osip, Kisielewski, Bartkowska-Sniatkowska and Rybakowski2015; Phillips et al., Reference Phillips, Norris, Talbot, Birmingham, Hatchard, Ortiz, Owoeye, Batten and Blier2019, Reference Phillips, Van Geel, Burhunduli, Vasudev, Batten, Norris, Talbot, Ortiz, Owoeye and Blier2022; Zavaliangos-Petropulu et al., Reference Zavaliangos-Petropulu, McClintock, Khalil, Joshi, Taraku, Al-Sharif, Espinoza and Narr2023). Most improvements in executive and inhibitory control appeared independent of mood change, supporting a direct procognitive effect of ketamine. Notably, Keilp et al. (Reference Keilp, Madden, Marver, Frawley A, Burke, Mohammad, Gluck, Mann and Grunebaum2021) demonstrated that enhanced Stroop performance paralleled ketamine’s rapid antisuicidal effect, supporting the hypothesis that inhibitory control and attentional regulation may mediate its impact on suicidal cognition. Beyond inhibition, cognitive flexibility improved or was better preserved in ketamine-treated patients as well (Kumpf et al., Reference Kumpf, Wilkinson, Hu, Chen, Kamini, Chakrabarti, Rhee, Grezmak, Mathew, Sanacora, Murrough, Goes, Collins, Barnett and Anand2025; Oughli et al., Reference Oughli, Gebara, Ciarleglio, Lavretsky, Brown, Flint, Farber, Karp, Mulsant, Reynolds, Roose, Yang, Butters and Lenze2023). These findings further contribute to evidence for ketamine’s positive impact on prefrontal executive functioning. Taken together, these findings indicate that executive functions, particularly inhibitory control, represent a key cognitive target of ketamine’s effects. Improvements on Stroop paradigms and related measures may not only index frontocingulate neuroplasticity but also mediate rapid reductions in suicidal ideation, highlighting a mechanistic bridge between cognition and clinical response.
Altogether, subanesthetic IV ketamine appears to preserve, and in some cases enhance, neurocognitive functioning in TRD. These results reflect a clinically meaningful outcome for patients with TRD, given the cognitive burden typically observed in this population. Nevertheless, the considerable interindividual variability and short duration of follow-up in most trials limit firm conclusions regarding the durability and functional significance of these effects.
Furthermore, several moderators may influence cognitive outcomes. Treatment characteristics, such as repeated-dose regimens, generally produced stronger effects than single infusions and play a central role in the cognitive analysis (Grunebaum et al., Reference Grunebaum, Galfalvy, Choo, Keilp, Moitra, Parris, Marver, Burke, Milak, Sublette, Oquendo and Mann2018; Phillips et al., Reference Phillips, Norris, Talbot, Birmingham, Hatchard, Ortiz, Owoeye, Batten and Blier2019). Patient characteristics are also relevant to validity, as younger age and greater baseline cognitive impairment were associated with more pronounced improvement in several studies (X et al., 2023). These findings emphasize the need to incorporate cognitive endpoints into clinical trial design and individualized treatment planning. This variability in timing may contribute to differences in practice effects and test–retest sensitivity across studies.
At a mechanistic level, ketamine’s NMDA receptor antagonism and downstream glutamatergic modulation promote synaptic plasticity, which may underpin its dual antidepressant and procognitive actions (Glue et al., Reference Glue, Loo, Fam, Lane, Young and Surman2024). There is growing interest in sustained-release or combined-modality approaches (e.g. pairing ketamine with neuromodulation or psychotherapy) to prolong these benefits, although interindividual sensitivity and theoretical risks such as excitotoxicity warrant careful monitoring (Luo et al., Reference Luo, Wu, Dai, Yang, Xu, Jing, Rao, Xu, Gao, Fei and Lu2019).
The complexity of TRD is further compounded by psychiatric and medical comorbidities. Because most included trials excluded individuals with active substance use disorders, the generalizability of the present findings to TRD populations with comorbid addiction remains uncertain. Given the known effects of substance use on glutamatergic and executive networks, future research should directly examine whether substance-related neurobiological alterations moderate ketamine’s cognitive effects. Substance use disorders, for instance, may alter glutamatergic and dopaminergic circuits, attenuating ketamine’s cognitive effects, while metabolic or vascular conditions could exacerbate cognitive vulnerability through inflammatory (Zhou et al., Reference Zhou, Jin, Meng, ZHu, Bai, Tian, Mao, Wang, Xie, Zhong, Zhang, Luo, Tao, Wang, Li, Li, Qui, Zhou, Li and Zhang2019) or cerebrovascular pathways (Haroon et al., Reference Haroon, Raison and Miller2012). Such interactions remain insufficiently characterized and should be systematically evaluated in future studies.
This review has several limitations. Substantial heterogeneity in study designs, ketamine protocols, assessment timing, and cognitive measures prevented quantitative meta-analysis, and findings were therefore synthesized using a narrative vote-counting approach, which precludes formal effect-size estimation. The search was limited to English- and French-language publications, which may have excluded relevant studies. In addition, inclusion was restricted to TRD populations. While this decision enhances clinical relevance, as IV ketamine is primarily used in later-line treatment settings, findings may not generalize to individuals with non-resistant MDD. Partial cohort overlap is also possible, as several reports originated from the same research groups (Chen et al., Reference Chen, Li, Lin, Hong, Tu, Bai, Cheng and Su2018, Reference Chen, Lin, Li, Tsai, Wu, Bai, Hong, Tu and Su2022; Singh et al., Reference Singh, Parikh, Vande Voort, Pazdernik, Achtyes, Goes, Yocum, Nykamp, Becerra, Smart, Greden, Bobo, Frye, Burdick and Ryan2024; Zhou et al., Reference Zhou, Wang, Lan, Li, Chao, Wu, McIntyre and Ning2022, Reference Zhou, Wang, Lan, Zheng, Li, Chao, Wu, McIntyre and Ning2021, Reference Zhou, Zheng, Liu, Wang, Zhan, Li, Chen, Li and Ning2018), and the total sample size should therefore be interpreted as an approximation of unique participants. Practice effects may have contributed to apparent cognitive gains, although some trials attempted to mitigate this through alternate test forms or extended follow-up (e.g. Singh et al., Reference Singh, Parikh, Vande Voort, Pazdernik, Achtyes, Goes, Yocum, Nykamp, Becerra, Smart, Greden, Bobo, Frye, Burdick and Ryan2024). Finally, several cognitively and clinically relevant domains, such as decision-making, verbal fluency, and suicidal cognition, remain underexplored, despite their importance for functional recovery and relapse prevention.
Conclusion
This systematic review suggests that IV ketamine administered at subanesthetic doses is associated with procognitive effects in patients with TRD, most consistently in processing speed, working memory, and attention, without evidence of cognitive deterioration. However, cognitions related to suicide behaviors, such as decision-making and verbal fluency, remain underexplored, despite their potential relevance for functional recovery and relapse prevention. Executive functions, particularly inhibitory control as assessed through Stroop paradigms, emerged as promising cognitive targets that may mediate ketamine’s rapid antisuicidal effects. To our knowledge, this is the first systematic review to explicitly highlight suicide-related cognitions, a clinically critical yet neglected dimension within the context of ketamine treatment.
Future studies should incorporate standardized executive-function endpoints, longitudinal designs, and multi-timepoint cognitive assessments to determine the durability, mechanisms, and functional significance of ketamine’s cognitive effects. A better understanding of these cognitive pathways may ultimately guide personalized interventions and improve suicide prevention strategies in TRD.
Author contribution
Conceptualization: S.R.D. and V.D.J.; Formal analysis: L.A.C., V.D.J., and S.R.D.; Writing – original draft: L.A.C., V.D.J., and S.R.D.; Writing – review and editing: V.D.J., S.R.D., N.G., M.S.N., L.O., and G.T.; Supervision: V.D.J. and S.R.D.
Funding statement
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Competing interests
The authors declare no conflict of interest.