Introduction
In Europe, the burden of chronic conditions on the healthcare systems is enormous and will continue to grow. For instance, in 2022, the percentage of ≥65-year-old people with two chronic conditions was higher than 30% (OECD and European Union, 2022; Hacker, Reference Hacker2024). The healthcare systems are inadequately and insufficiently prepared to manage such burden (Holman Reference Holman2020; Kabir et al., Reference Kabir, Karim, Islam, Romero and Billah2022). To improve their readiness, care delivery must be reorganized (OECD and European Union, 2022). One of the most important dimensions to consider is the development of a greater place for self-management interventions (SMI) (Straub and Thekkekandam, Reference Straub, Thekkekandam, Daaleman and Helton2023). World Health Organization (WHO) Europe defines SMIs as supportive interventions systematically provided by healthcare staff, peers, or lay persons to increase the patients’ skills and confidence in their ability to manage long-term conditions (WHO European Region, 2024). Evidence from the literature shows that SMI programmes can improve clinical outcomes, patient-reported measures, and the quality of life of people living with chronic conditions (Tsokani et al., Reference Tsokani, Seitidis, Christogiannis, Kontouli, Nikolakopoulos, Zevgiti, Orrego, Ballester, Suñol, Heijmans, Poortvliet, van der Gaag, Alonso-Coello, Canelo-Aybar, Beltran, González-González, de Graaf, Veroniki and Mavridis2023). To improve access to these programmes, primary care has a unique role to play because it can reduce disparities across the population by ensuring equal access to services (Dineen-Griffin et al., Reference Dineen-Griffin, Garcia-Cardenas, Williams and Benrimoj2019; WHO European Region, 2024). Therefore, SMI development in primary care is encouraged, especially in France (Haut conseil pour l’avenir de l’assurance maladie, 2022).
In Europe, SMIs for chronic diseases were introduced in the 2000s. Many healthcare systems offer different SMI programmes, mainly by health care providers (HCPs) including general practitioners (GPs) and more frequently trained nurses (Nolte et al., Reference Nolte, Knai and Saltman2014). Since 2007, in France, the National Health Authority (Haute Autorité de Santé; HAS) considers SMI accessibility a priority. These interventions need to be structured according to the HAS guidelines and to be authorized by the Regional Health Agency (Agence Régionale de Santé, ARS) (Haute Autorité de Santé, 2007). SMIs can be performed in different settings, including by telephone and online (e.g. the ‘Sophia’ programme developed by the French health insurance fund), at hospitals or in education centres, which are located in the main city of each French region, and in primary care practices (PCP). For all these programmes, which are free of charge, each SMI team must complete a structured administrative form to provide information on the hosting structure, type of self-management, content, team, coordination, and local partners. This form is analysed using some quality criteria by the ARS and validated for four years. Since 2021, a less restrictive declaration procedure has been introduced to improve SMI development.
In 2013, the qualitative review by Elissen et al., in 13 European countries, showed that SMI remains relatively underdeveloped (Elissen et al., Reference Elissen, Nolte, Knai, Brunn, Chevreul, Conklin, Durand-Zaleski, Erler, Flamm, Frølich, Fullerton, Jacobsen, Saz-Parkinson, Sarria-Santamera, Sönnichsen and Vrijhoef2013). In 2016, Schäfer et al. highlighted the decreased involvement of European GPs in preventive activities between 1993 and 2012 (Schäfer et al., Reference Schäfer, Boerma, Spreeuwenberg, Schellevis and Groenewegen2016). In France, using data from three national agency reports, Clet et al. showed that SMI development in primary care is hindered particularly by the difficulties to integrate a collective, long-term dimension into preventive clinical practices (Clet et al., Reference Clet, Essilini, Cohidon and Alla2024). However, in the last few years, more and more PCPs have been developed in France (Bahiaoui, Reference Bahiaoui2024). Thanks to their organization with more collaboration among the implicated professionals, they can address the lack of SMIs and improve the quality of care for people with chronic diseases (Cret et al., Reference Cret, Guilhot, Capgras and Pascal2022).
Most SMI approaches in Europe involve some form of delivery system design, but the nature of the strategies varies, and more data need to be collected (Nolte et al., Reference Nolte, Knai and Saltman2014). Few data are available on SMI practice at the national level, particularly in primary care (Boerma et al., Reference Boerma, Bourgueil, Cartier, Hasvold, Hutchinson, Kringos and Kroneman2015). Yet, to improve their development in healthcare systems, particularly in primary care, it is important (i) to describe the model of SMI practices in primary care; (ii) to identify strengths and areas of improvement; and (iii) to inform decision makers and HCPs for policy and practice improvement.
Therefore, the main objective of our study was to describe the SMI programmes at PCPs nationwide in France from 2010 to 2022.
Methods
This descriptive retrospective study focused on the SMI programmes authorized/declared by/to the 18 ARS of France between 2010 and 2022. This period was chosen because in 2010 a national decree was published in France that lists the specifications that must be met by each SMI programme. The limit date of 2022 was selected because data collection/analysis started at the beginning of 2023.
In partnership with the French Ministry of Health Chronic Disease department, in January 2023, the self-management mission managers of each ARS were contacted by email and asked to participate in this study. Among them, 15 managers (3 refused to participate) attended an online meeting in March 2023 where the study protocol was explained. After one additional refusal to participate, individual interviews were organized with each of the 14 self-management mission managers to explain in details the study and to list the data and documents that needed to be collected. As one ARS did not have any data for the study, the analysis concerned data from 13 ARS that covered 60,059,521 million French inhabitants (87.8% of the French population).
First, from the administrative SMI files stored at the ARS, the following data were extracted: SMI location (hospital or in primary care), type (group, personalized intervention), and administrative structure responsible of the programme. Specifically, for programmes in PCPs without a specific coordinator for the administrative/financial aspects, local administrative structures can help to perform these duties, for instance, through the Coordination Support Mechanism (dispositif d’appui à la coordination).
After identification of all SMI programmes carried out at PCPs, the set of specifications of the programmes authorized/validated by their regional ARS were analysed. These data were obtained from the SI-ETP website (https://si-etp.ars.sante.fr/ ), an ARS reference site that lists all SMI programmes in France. An access to the website was created specifically for the study. When several files were available for one programme, the most recent file in the system was selected. According to Appendix 2 of the 2010 French decree, these specifications are required for the SMI programme authorization application (Supplementary file 1). The absence of a standardized authorization/declaration justified the programme exclusion from the study.
Statistical analysis
An Excel dataset was created, and the categories of the extracted data were determined on the basis of a literature review (Allory et al., Reference Allory, Scheer, De Andrade, Garlantézec and Gagnayre2024) and exchanges among all authors. They included: PCP type, territory type according to the French National Institute of Statistics and Economics Studies (Institut national de la statistique et des études économiques, Insee) as well as SMI programme duration, coordination, content, funding bodies, description of the team and its local partners (defined as healthcare actors working with the SMI team).
The descriptive statistical analysis of the collected data was carried out using the Stata® software. Qualitative variables were described by numbers and percentages, and quantitative variables by means and standard deviation (SD).
Ethical issues
The Rennes University Hospital Ethics Committee approved the study (approval number 23.51 of 28 April 2023; Supplementary file 2). The project complied with the reference methodology MR-004 defined by the French committee on personal data protection (Commission Nationale Informatique et Libertés, CNIL) and with the European General Data Protection Regulation. The study protocol was registered in Clinical trials® under the number NCT05950451.
Results
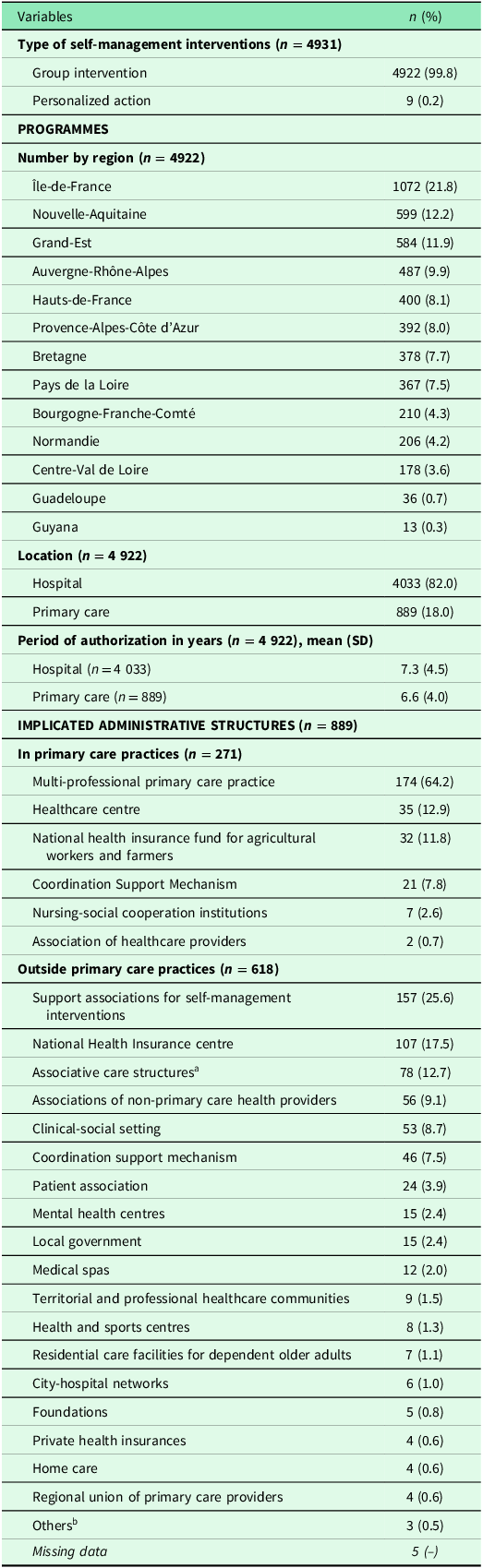
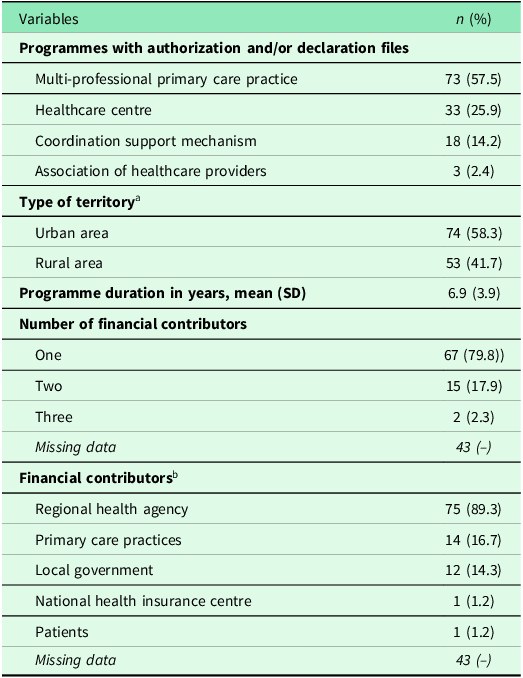
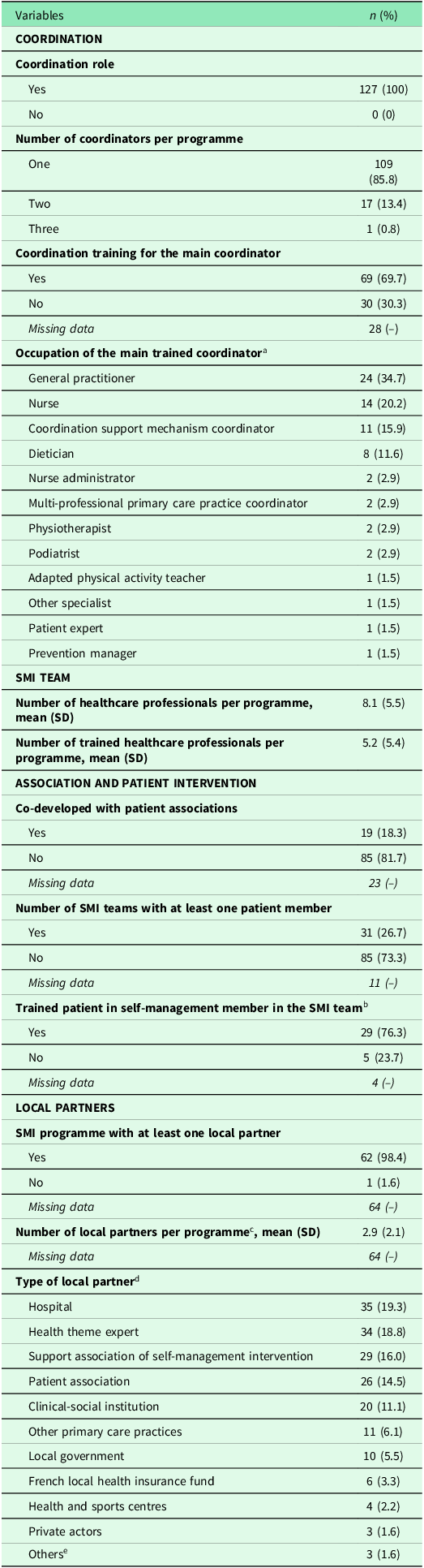
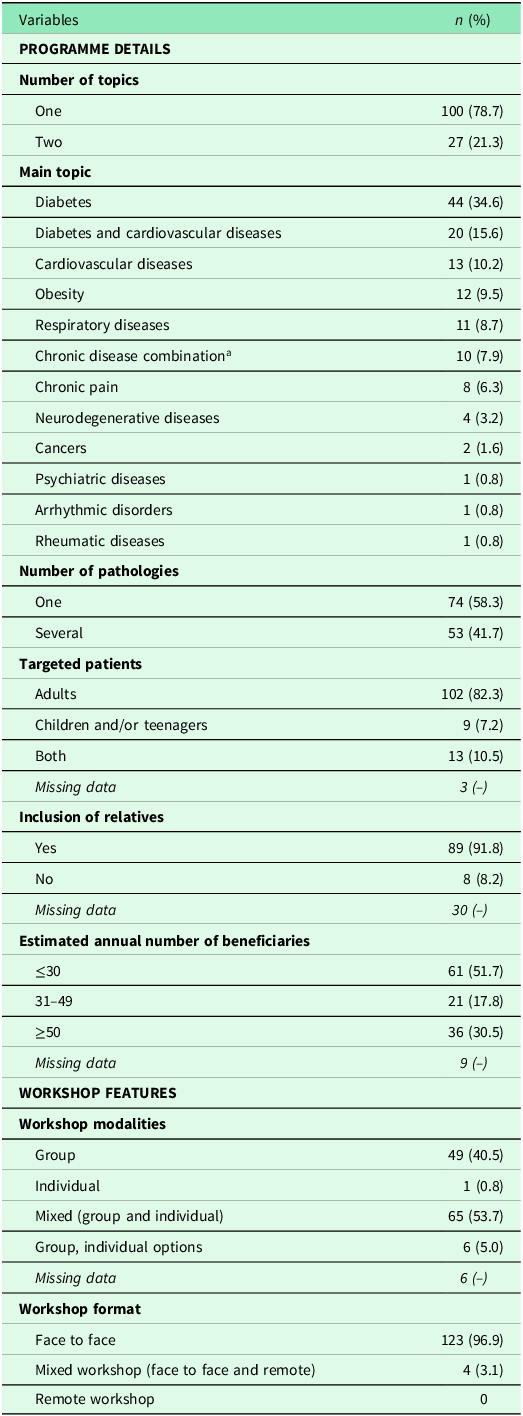
At the 13 ARS that participated in the study, 4922 SMI programmes were identified (Table 1) among which 82% were at hospitals. The mean period of authorization was 6.6 years for SMIs in primary care settings. Among the 889 programmes in primary care settings, 271 were performed at PCPs, and 71.1% of these interventions were administratively supported by the PCP. Then, 144 SMI programmes were excluded for different reasons: the ARS did not give the files due to the work required (n = 115 programmes from three ARS); files not found by the ARS (n = 21); different programmes linked to only one file (n = 5); and absence of standardized authorization/declaration file (n = 3). Programmes excluded for these three last reasons were distributed equally among ARS. Therefore, the analysis focused on the 127 SMI programmes carried out in PCPs for which the authorization and/or declaration files could be accessible (Table 2). More than 80% of these programmes were developed in a multi-professional primary care practice (MPCP) or a healthcare centre, mostly in urban areas (58.3%). Some programmes had more than one funder (20.2%). The main financial contributor was the ARS (89.3%). All SMI programmes had a coordination structure, mainly with one coordinator (85.8%) who was trained in 69.7% of cases (Table 3). The coordinator was mainly a GP (34.7%), followed by a nurse (20.2%). The mean HCP number per programme was 8.1, among whom 5.2 were trained in self-management. The programme was rarely co-developed with patient associations (18.3%). When patients were part of the SMI team (26.7%), they were mainly trained in self-management (76.3%). Almost all SMI programmes worked with at least one local partner (98.4%; mean of 2.9 partners per programme). The main local partner was the hospital (19.3%). Lastly, 79% of SMI programmes covered only one topic, mainly diabetes (34.6%) (Table 4). Almost 42% of SMI programmes were open to patients with more than one disease, mainly adults (82.3%), including their relatives (91.8%). For 51.7% of SMI programmes, the estimated annual number of beneficiaries was lower than 30. The workshop modalities were mostly mixed, with group and individual activities, mainly in the face-to-face format (96.9%).
General description of self-management interventions (group and personalized interventions), in France (13 regions), over the period 2010–2022

a Dialysis centres, non-multidisciplinary medical centres, nurse offices, nursing home care services.
b ‘Centre de Ressource, d’Expertise et de Performance Sportive’ (CREPS), ‘Centre de Coopération Internationale en Recherche Agronomique pour le Développement’ (CIRAD), and one programme in Bretagne identified with the term ‘association’.
Description of the SMI programmes carried out in primary care practices in France (13 regions) over the period 2010–2022 (n = 127)

a According to the Insee classification.
b The total is >100% because each programme could be supported by more than one funder.
Description of the SMI teams in primary care practices, in France (13 regions), over the period 2010–2022 (n = 127)

SD: Standard deviation, SMI: Self-management interventions.
a Among the main coordinators trained in coordination n = 69.
b Among patients participating in SMI programmes n = 38.
c Among the SMI programmes with at least one local partner n = 62.
d Among all local partners n = 181.
e Student and other associations.
Description of the SMI programmes in primary care practices, in France (13 regions), over the period 2010–2022 (n = 127)

a Combination of different chronic diseases for each programme (1 to 10): 1 (obesity and cardiovascular diseases); 2 (diabetes, heart failure, Parkinson’s disease, cancer, depressive disorders and asthma, and/or chronic obstructive pulmonary disease, COPD); 3 (multiple pathologies in the context of ageing without cognitive disorders); 4 (cardiovascular diseases, respiratory diseases, and type II diabetes); 5 (severe chronic kidney disease and primary nephrotic syndrome); 6 (ulcerative colitis and progressive Crohn’s disease); 7 (cardiovascular diseases, obesity and stress management); 8 (asthma, cardiovascular diseases, kidney failure, and type II diabetes); 9 (stroke, diabetes, kidney failure, neurological diseases, cancers, cardiovascular diseases, rheumatic diseases, digestive system diseases, respiratory diseases, and HIV/AIDS); and 10 (type II diabetes, cardiovascular diseases, and COPD.
Discussion
The results of this study showed that in France, less than 20% of SMI programmes are carried out in primary care settings and that 5.5% (n = 271) of them are developed in PCPs. Moreover, they focus mostly on adults with one chronic disease, and they are followed by too few patients. They are mainly supported financially by the ARS; they have a large team, with a quasi-systematic coordination role, carried out by GPs and nurses. Although they are well integrated with the territorial partners (first the hospital), most programmes do not include a patient association partnership.
As previously shown in other European countries, and despite the recommendations to integrate SMI in primary care service routine, SMI programmes in primary care in France remains rare (Boerma et al., Reference Boerma, Bourgueil, Cartier, Hasvold, Hutchinson, Kringos and Kroneman2015; Elissen et al., Reference Elissen, Nolte, Knai, Brunn, Chevreul, Conklin, Durand-Zaleski, Erler, Flamm, Frølich, Fullerton, Jacobsen, Saz-Parkinson, Sarria-Santamera, Sönnichsen and Vrijhoef2013). Moreover, as their content focuses often on only one chronic disease, they are inadequate to meet the population’s needs. This result is in line with an international systematic review of 2024 showing that only 20% of SMI programmes in primary care focused on multimorbidity (Allory et al., Reference Allory, Scheer, De Andrade, Garlantézec and Gagnayre2024). However, a retrospective cohort study on more than 400,000 people in England found that patients with multimorbidity accounted for more than half of GP consultations and 78.7% of their prescriptions (Cassell et al., Reference Cassell, Edwards, Harshfield, Rhodes, Brimicombe, Payne and Griffin2018). Moreover, a 2025 study by the Organization for Economic Co-operation and Development on 107,000 people from 19 countries showed that 54% of individuals older than 45 years followed in primary care had at least ≥2 chronic diseases (OECD, 2025). It seems that the recommendations by Rijken et al. in 2014 to develop a multimorbidity approach for SMI in primary care are still relevant, but have not been followed (Rijken et al., Reference Rijken, Bekkema, Boeckxstaens, Schellevis, De Maeseneer and Groenewegen2014). Some qualitative studies with primary care professionals showed that self-management among people with multimorbidity is perceived ‘as “chipping away”, using regular consultations, continuity and ongoing education to encourage change’ (Kristensen et al., Reference Kristensen, Hølge-Hazelton, Waldorff and Guassora2017). Thus, the current SMI programmes may not be adapted to all patient needs and may hinder the development of self-management for people with multimorbidity. Based on these results, the French national health authority, which is not currently supporting the development of multimorbidity approaches in SMI, could encourage them in different ways (e.g. continuing professional development, financial incentives). Another solution could be to combine individual and collective self-management activities (Traynard and Gagnayre, Reference Traynard and Gagnayre2013). Thus, the individual approach, which can be repetitive, during and after the SMI programme, can favour a more patient-centred approach, and the collective approach can help the development of soft skills. In any case, although SMI effectiveness on multimorbidity remains unclear, it seems a priority to work on new SMI models to meet the needs of patients with multimorbidity (Smith et al., Reference Smith, Wallace, O’Dowd and Fortin2021).
In our study, the SMI programme coordinator, who is required in France, was mainly a GP or a nurse. This is in line with the 2014 study by Nolte et al. on chronic disease management in European health systems showing that in the majority of cases, SMIs are provided by GPs or trained nurses (Nolte et al., Reference Nolte, Knai and Saltman2014). However, in many European countries, it is uncommon to have nurses as SMI programme coordinators. For instance, Kringos et al. found that nurses provide health education in primary care only in 12/31 European countries (Kringos et al., Reference Kringos, Boerma, Bourgueil, Cartier, Dedeu, Hasvold, Hutchinson, Lember, Oleszczyk and Pavlič2015). Our result could be explained by the fact that in France, SMI programmes in primary care are mainly developed at PCPs and that the nurses’ implication in care delivery and coordination is common in systems with multidisciplinary teams (Nolte, Reference Nolte2009). This involvement could be partially due to the new care organization in France that gives a bigger place to PCPs and includes more collaboration between HCPs. This leads to responsibility delegation and new competencies, especially for nurses (Suchier and Michel, Reference Suchier and Michel2021). In the last years, the implementation of more advanced roles for nurses (e.g. advanced practice nurses) is a sign of this evolution. Another reason of the nurses’ implication is the increasing gap between the significant population’s demand for managing chronic diseases and the insufficient supply of GPs in the healthcare system. This change is a great opportunity because nurses seem to be supported in their mission by the population and considered by the SMI team as an important educator, with a better understanding of psychosocial issues (Barreto et al., Reference Barreto, de Almeida Rebouças, de Aguiar, Barbosa, Rocha, Cordeiro, de Melo and de Freitas2019).
Among the PCPs that deliver SMIs, MPCPs and healthcare centres were the most important. In France, both structures need to have a health project validated by the ARS, but have a different organization. In a MPCP, HCPs are self-employed and work with other self-employed HCPs in the same territory (sometimes the same building), whereas in a healthcare centre, HCPs are employees and work in a building with other HCPs. In 2024, a qualitative study with different HCPs showed that in the context of chronic disease, a collaborative approach among SMI actors is one of the necessary skills for developing SMI programmes in primary care (Timmermans et al., Reference Timmermans, Boeykens, Sirimsi, Van de Velde, De Vriendt, Decat, Foulon, Van Hecke, Vermandere, Schoenmakers and Care Academy2024). In addition, our study found that PCPs are developing many local partnerships, mainly with the hospital, as discussed by Cret (Cret et al., Reference Cret, Guilhot, Capgras and Pascal2022). Nevertheless, our result is in line with the perspectives by Boerma et al. who wrote that the cooperation and coordination between primary and secondary care can benefit from the creation of multidisciplinary teamwork and practices (Boerma et al., Reference Boerma, Bourgueil, Cartier, Hasvold, Hutchinson, Kringos and Kroneman2015).
Lastly, our analysis highlights the limited involvement of patients in SMI development and delivery. The recommendation to better involve the patients in the SMI teams is not followed yet (Reynolds et al.. Reference Reynolds, Dennis, Hasan, Slewa, Chen, Tian, Bobba and Zwar2018). However, some key decisions have been recently taken in France to facilitate the patients’ engagement in SMI. Indeed, the national health authority wrote in 2020 a recommendation on how to support the engagement of the population at all levels of the healthcare system, including SMI delivery in primary care (Haute Autorité de Santé, 2020).
Our study confirms European data on self-management and gives a national perspective using centralized data and a standardized approach based on ARS data. Although not all ARS participated, the collected data were from 13 ARS that cover more than 85% of the French population. However, we cannot exclude a selection bias, although we think that data were missing at random for several programmes.
Another strength of our study is to have worked using a structured administrative form. As we did not find publications reporting similar data from elsewhere in Europe, it should encourage other European countries to systematically describe who offers SMI in the healthcare system.
A limitation of the study is that we lacked data especially on the coordination training for the coordinator and on the engagement of associations or patients in the SMI team. This indicates that when SMI teams start the SMI process, they are often not trained in coordination and in collaboration with patients and/or associations. To explain this lack of patient engagement, Ocloo et al. showed in a systematic review of reviews that the patients’ engagement in the healthcare system needs a whole system approach (Ocloo et al., Reference Ocloo, Garfield, Franklin and Dawson2021). Indeed, both the individual level (especially attitudes and representations among patients and healthcare professionals) and the collective level (communication and working with communities, funding, and general support to engage people especially) need to be taken into account to improve patient engagement. This systematic review did not include any study on patient engagement in self-management in primary care. However, the authors stressed that organizational factors play a key role in patient engagement. Therefore, more studies are needed on patient engagement in self-management programmes in primary health care. Furthermore, the set of specifications was completed by the SMI teams at the SMI beginning and was not re-evaluated afterwards. As the mean programme duration was 6.9 years (3.9), the SMI team probably underwent some changes over time. Therefore, we may have described the facilitating factors to implement SMI programmes in PCPs, but not the facilitating factors to maintain and pursue them.
Based on our results, we propose the following recommendations to French national health authority to improve SMI practice and access in primary care. First, a multimorbidity approach should be encouraged in SMI programmes in primary care. Second, nurses should be seen and supported as the main educator in PCPs. Third, policymakers should support the development of MPCPs and healthcare centres. Fourth, they should give financial incentives to the centres that develop SMI programmes. Fifth, the involvement of patients in SMI development and delivery should be promoted, in accordance with the French health authority recommendations (Haute Autorité de Santé, 2020).
Conclusion
In France, self-management programmes in PCPs are still rare, lack a multimorbidity approach, and are carried out mainly in PCPs with inter-professional collaboration. GPs and nurses are the main actors in SMI programmes, whereas patients are rarely involved in their development and delivery. A qualitative study could be conducted among SMI educational teams in PCPs in France to better identify the SMI strengths, obstacles, and areas of improvement in order to promote a better model of SMI practice in PCPs.
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1017/S1463423626100929.
Acknowledgements
We thank the French Ministry of Health and all the self-management managers at the Regional Health Agencies.
Funding statement
This study was supported by a grant from the French Ministry of Health (ETHOPEE ReSP-IR 2021 2022-19C).
Competing interests
The authors declare none.