Introduction
High-quality seeds are central to the sustainability and resilience of agri-food systems. Their germination and transition to seedling growth is one of the most vulnerable plant life cycle stages, with plant establishment at risk by abiotic (e.g. drought and salinity) and biotic (e.g. pathogenic microbes and weeds) environmental stresses and extreme weather events (Finch-Savage and Leubner-Metzger, Reference Finch-Savage and Leubner-Metzger2006; Walck et al., Reference Walck, Hidayati, Dixon, Thompson and Poschlod2011; Weitbrecht et al., Reference Weitbrecht, Müller and Leubner-Metzger2011; Finch-Savage and Bassel, Reference Finch-Savage and Bassel2016; Fernandez-Pascual et al., Reference Fernandez-Pascual, Mattana and Pritchard2019). Despite the benefits realized by fungicide seed treatment to improve crop seedling emergence and protection against damping-off diseases, recent studies raised concerns regarding this practice (Ayesha et al., Reference Ayesha, Suryanarayanan, Nataraja, Prasad and Shaanker2021). Of particular concern are the off-target effects on the seed-borne microbiome and the assembly of the plant microbiome. The transition from seeds to seedlings represents a major bottleneck for the assembly of the plant microbiome which includes beneficial microorganisms derived from the seed and the soil (Card et al., Reference Card, Hume, Roodi, McGill, Millner and Johnson2015; Rybakova et al., Reference Rybakova, Mancinelli, Wikström, Birch-Jensen, Postma, Ehlers, Goertz and Berg2017; Nelson, Reference Nelson2018; Simonin et al., Reference Simonin, Briand, Chesneau, Rochefort, Marais, Sarniguet and Barret2022; Abdelfattah et al., Reference Abdelfattah, Tack, Lobato, Wassermann and Berg2023). Biopriming and other microbial inoculation technologies to deliver beneficial microorganisms via the seed for improved crop performance and yield gains despite environmental stresses is an increasing research interest (O'Callaghan, Reference O'Callaghan2016; Rocha et al., Reference Rocha, Ma, Souza-Alonso, Vosatka, Freitas and Oliveira2019; Li et al., Reference Li, Van Gerrewey and Geelen2022; Paravar et al., Reference Paravar, Piri, Balouchi and Ma2023). While progress has been made in identifying suitable beneficial microorganisms and improving seed inoculation technologies, there are significant technical challenges in maintaining viable microbial seed inocula throughout commercial seed processing and warehouse storage (O'Callaghan, Reference O'Callaghan2016). Further research is therefore needed before the benefits of microbial seed inoculants can be captured for use in sustainable agriculture, ecosystem restoration and beyond.
Environmental-friendly innovative seed technologies include various priming, coating, pelleting and other processing methods to aid sowing and/or enhance crop seed performance and stress resilience in the field (Ignatz et al., Reference Ignatz, Hourston, Tureckova, Strnad, Meinhard, Fischer, Steinbrecher and Leubner-Metzger2019; Bruggink, Reference Bruggink, Buitink and Leprince2022; Hampton, Reference Hampton, Buitink and Leprince2022). The different meanings associated with ‘priming’ as a very broad term fall into two major types which are described in detail by Hampton (Reference Hampton, Buitink and Leprince2022). First, seed priming is a pre-sowing technique to physiologically enhance seed performance and seedling vigour. This involves controlled hydration (phases I and II of the water uptake curve) to allow metabolic activation, but without permitting the seed to proceed to water uptake phase III or visible radicle protrusion (Bruggink, Reference Bruggink, Buitink and Leprince2022; Corbineau et al., Reference Corbineau, Taskiran-Özbingöl and El-Maarouf Bouteau2023; Fatelnig et al., Reference Fatelnig, Chanyalew, Tadesse, Kebede, Hussein, Iza, Tadele, Leubner-Metzger and Steinbrecher2024). The controlled hydration method (e.g. hydropriming, osmopriming) is followed by drying seeds back to a low moisture content similar to the original air-dry state. The effect of classical seed priming (hydration and drying) is accelerated and synchronized germination leading to vigorous seedlings even upon stress (Ibrahim, Reference Ibrahim2016; Corbineau et al., Reference Corbineau, Taskiran-Özbingöl and El-Maarouf Bouteau2023; Fatelnig et al., Reference Fatelnig, Chanyalew, Tadesse, Kebede, Hussein, Iza, Tadele, Leubner-Metzger and Steinbrecher2024). During seed hydropriming, water uptake is restricted by adding a defined limited amount of water or by a defined short time period, while during seed osmopriming, reduced water potential is generated by adding osmolytes (e.g. polyethylene glycol or salts) to restrict the transition from water uptake phases II to III. These priming treatments enhanced germination in that the time of the seed population for 1 and 50%, T1% and T50%, respectively was reached earlier, and that the seed populations uniformity (U90%–10%, the time difference between T10% and T90%) was decreased (Lechowska et al., Reference Lechowska, Kubala, Wojtyla, Nowaczyk, Quinet, Lutts and Garnczarska2019; Sano and Seo, Reference Sano and Seo2019; Batista et al., Reference Batista, Fernandez, da Silva, Maia and da Silva2020). In general, primed seeds of many species have increased ageing sensitivity and therefore a reduced storability (‘shelf-life’) due to deterioration processes (Fabrissin et al., Reference Fabrissin, Sano, Seo and North2021). A second major type of seed or seedling priming includes seed biopriming, it does not have improving germination performance as its main aim but is mainly conducted to protect seedlings and adult plants by enhanced responsiveness to abiotic and biotic stressors (Mauch-Mani et al., Reference Mauch-Mani, Baccelli, Luna and Flors2017; Hampton, Reference Hampton, Buitink and Leprince2022). Biopriming of Brassica seeds with beneficial bacteria resulted for example in enhanced resistance of seedlings to blackleg disease (Abuamsha et al., Reference Abuamsha, Salman and Ehlers2011a) and Verticillium wilt (Müller and Berg, Reference Müller and Berg2008), but not effectively against Fusarium wilt and Phytium damping-off disease (Chin et al., Reference Chin, Lim and Ting2022). However, sugarbeet biopriming protected seedlings against Phytium damping-off disease (Shah-Smith and Burns, Reference Shah-Smith and Burns1997; Moënne-Loccoz et al., Reference Moënne-Loccoz, Naughton, Higgins, Powell, O'Connor and O'Gara1999; O'Callaghan et al., Reference O'Callaghan, Swaminathan, Lottmann, Wright and Jackson2006).
Seed biopriming is a technique involving immersion of seeds in a microbe suspension for a defined period followed by seed drying (Mahmood et al., Reference Mahmood, Turgay, Farooq and Hayat2016; O'Callaghan, Reference O'Callaghan2016; Hampton, Reference Hampton, Buitink and Leprince2022). The seed soaking is usually in a saline suspension of a beneficial microbial inoculant and the method aims to improve crop performance, pathogen defence and stress resilience. Since biopriming involves seed hydration, i.e. similar to osmopriming if a saline suspension is used, it was proposed from works with beneficial bacteria that they are also taken up into the seed (Müller and Berg, Reference Müller and Berg2008; Mahmood et al., Reference Mahmood, Turgay, Farooq and Hayat2016; O'Callaghan, Reference O'Callaghan2016). There are numerous examples which demonstrate the effectiveness of seed biopriming in alleviating stress during germination and seedling growth and in protecting against disease (e.g. Shah-Smith and Burns, Reference Shah-Smith and Burns1997; O'Callaghan et al., Reference O'Callaghan, Swaminathan, Lottmann, Wright and Jackson2006; Müller and Berg, Reference Müller and Berg2008; Moeinzadeh et al., Reference Moeinzadeh, Sharif-Zadeh, Ahmadzadeh and Tajabadi2010; Abuamsha et al., Reference Abuamsha, Salman and Ehlers2011a, Reference Abuamsha, Salman and Ehlers2011b; Deaker et al., Reference Deaker, Hartley and Gemell2012; Mahmood et al., Reference Mahmood, Turgay, Farooq and Hayat2016; O'Callaghan, Reference O'Callaghan2016; Araujo et al., Reference Araujo, da Cruz, Souchie, Martin, Nakatani, Nogueira and Hungria2017; Lally et al., Reference Lally, Galbally, Moreira, Spink, Ryan, Germaine and Dowling2017; Rybakova et al., Reference Rybakova, Mancinelli, Wikström, Birch-Jensen, Postma, Ehlers, Goertz and Berg2017; Costales et al., Reference Costales, Nápoles, Falcón-Rodríguez, González-Anta, Petit and Solá2019; Sandini et al., Reference Sandini, Pacentchuk, Hungria, Nogueira, da Cruz, Nakatani and Araujo2019; Fiodor et al., Reference Fiodor, Ajijah, Dziewit and Pranaw2023). In several of these cases, recovery of the viable beneficial microbes from bioprimed seeds (by either surface wash or surface disinfection and grinding seeds) or from derived seedlings was demonstrated, but direct evidence for within-seed location of the beneficial microbial inoculant is a matter of circumstantial debate. What is also lacking are analyses of long-term storage effects of bioprimed seeds (shelf-life) on germination and seedling growth, and only a few studies investigated microbe viability (shelf-life) beyond 1-month storage of dry bioprimed seeds (Shah-Smith and Burns, Reference Shah-Smith and Burns1997; O'Callaghan et al., Reference O'Callaghan, Swaminathan, Lottmann, Wright and Jackson2006; Abuamsha et al., Reference Abuamsha, Salman and Ehlers2011b; Araujo et al., Reference Araujo, da Cruz, Souchie, Martin, Nakatani, Nogueira and Hungria2017).
Brassica napus (oilseed rape) is an important crop seed model for studying the effects of abiotic (Schopfer and Plachy, Reference Schopfer and Plachy1984; Weitbrecht et al., Reference Weitbrecht, Müller and Leubner-Metzger2011; Derakhshan et al., Reference Derakhshan, Bakhshandeh, Siadat, Moradi-Telavat and Andarzian2018) and biotic (Müller and Berg, Reference Müller and Berg2008; Abuamsha et al., Reference Abuamsha, Salman and Ehlers2011a; O'Callaghan, Reference O'Callaghan2016) environmental stresses on germination and seedling growth, as well as for investigating the roles and mechanisms of the seed microbiome (Card et al., Reference Card, Hume, Roodi, McGill, Millner and Johnson2015; Rybakova et al., Reference Rybakova, Mancinelli, Wikström, Birch-Jensen, Postma, Ehlers, Goertz and Berg2017; Nelson, Reference Nelson2018). The B. napus system was successfully used for seed biopriming, germination and seedling growth experiments with beneficial plant growth-promoting rhizobacteria (PGPR) such as Pseudomonas spp. (Sheng et al., Reference Sheng, Xia, Jiang, He and Qian2008; Abuamsha et al., Reference Abuamsha, Salman and Ehlers2011a, Reference Abuamsha, Salman and Ehlers2011b; Card et al., Reference Card, Hume, Roodi, McGill, Millner and Johnson2015; Lally et al., Reference Lally, Galbally, Moreira, Spink, Ryan, Germaine and Dowling2017; Rybakova et al., Reference Rybakova, Mancinelli, Wikström, Birch-Jensen, Postma, Ehlers, Goertz and Berg2017; Szymańska et al., Reference Szymańska, Dąbrowska, Tyburski, Niedojadło, Piernik and Hrynkiewicz2019). Pseudomonas fluorescens encompasses a group of common, non-pathogenic saprophytes that colonize soil, water and plants as environments. It is a common gram-negative, biofilm forming and well-studied PGPR (Paulsen et al., Reference Paulsen, Press, Ravel, Kobayashi, Myers, Mavrodi, DeBoy, Seshadri, Ren, Madupu, Dodson, Durkin, Brinkac, Daugherty, Sullivan, Rosovitz, Gwinn, Zhou, Schneider, Cartinhour, Nelson, Weidman, Watkins, Tran, Khouri, Pierson, Pierson, Thomashow and Loper2005; Hol et al., Reference Hol, Bezemer and Biere2013; Raio, Reference Raio2024). It inhibits the rhizosphere of many plants and suppresses plant diseases caused by soil-borne pathogens (O'Callaghan et al., Reference O'Callaghan, Swaminathan, Lottmann, Wright and Jackson2006; Chin et al., Reference Chin, Lim and Ting2022). Certain P. fluorescens strains can promote germination and seedling growth (Sheng et al., Reference Sheng, Xia, Jiang, He and Qian2008; Moeinzadeh et al., Reference Moeinzadeh, Sharif-Zadeh, Ahmadzadeh and Tajabadi2010; Lally et al., Reference Lally, Galbally, Moreira, Spink, Ryan, Germaine and Dowling2017; Sandini et al., Reference Sandini, Pacentchuk, Hungria, Nogueira, da Cruz, Nakatani and Araujo2019; Chin et al., Reference Chin, Lim and Ting2022; Hanifah et al., Reference Hanifah, Ghadamgahi, Ghosh, Ortiz, Whisson, Vetukuri and Kalyandurg2023). Shah-Smith and Burns (Reference Shah-Smith and Burns1997) demonstrated that a 10,000-fold decrease in viable Pseudomonas putida during long-term storage to below 103 bacteria per seed protected against seedling damping off, but a general minimal value of required viable bacteria per seed is not known (O'Callaghan et al., Reference O'Callaghan, Swaminathan, Lottmann, Wright and Jackson2006; Bashan et al., Reference Bashan, de-Bashan, Prabhu and Hernandez2014). In commercial applications, a microbial inoculant shelf-life of 1–2 years under warehouse storage conditions is desirable, but this is difficult to achieve for non-spore-forming, gram-negative bacteria.
In the present study, we utilize the B. napus seed model with P. fluorescens to investigate the effects of duration, temperature, seed input and additives (kimchi paste) on the biopriming process. We conclude that the high salt concentration in the biopriming solution is the cause of the reduced germination uniformity. We investigate how long-term storage of dry bioprimed seeds affects bacterial viability (microbial inoculant shelf-life), and conclude that long-term air-dry storage of bioprimed seeds does not negatively affect germination and seedling growth.
Materials and methods
Plant and inoculant material
Seeds of B. napus L. cv. Hertz and cv. Campus (trial oilseed rape seed, KWS SAAT SE & Co. KGaA, Einbeck, Germany and Thriplow, UK) were stored in hermetically sealed containers containing silica gel at room temperature. Their thousand seed weights (TSWs) were 5.51 and 4.68 g, respectively. Stocks of P. fluorescens Migula 1895, collection number DSM 50090 (taxonomic type strain of the species, Leibniz Institute, DSMZ-German Collection of Microorganisms and Cell Cultures GmbH, Braunschweig, Germany) in 20% v/v glycerol were stored at −20°C. Two separately purchased glycerol stocks were used in the seed biopriming experiments, #1 in the experiments with the Hertz and #2 in the experiments with the Campus cultivar. P. fluorescens DSM 50090 from stocks were grown on King's B (KB, Sigma-Aldrich, Gillingham, UK) nutrient agar (King, Reference King1954) in 9-cm Petri dishes incubated at 28°C in darkness.
Seed biopriming and hydropriming
A liquid P. fluorescens DSM 50090 pre-culture was prepared with single-colony inoculant from the KB plates using 20 ml autoclaved pre-warmed Tryptic Soy Broth (TSB, Sigma-Aldrich, Gillingham, UK), which was incubated at 28°C at 125 rpm in a thermoregulated orbital shaker for 7 h. Of this, 1 ml aliquots were used to inoculate flasks with 100 ml autoclaved pre-warmed TSB, which were incubated under the conditions described above for 17 h. These flasks with grown 100-ml cultures were used for seed biopriming following the protocol of Abuamsha et al. (Reference Abuamsha, Salman and Ehlers2011b). In short, 10, 25 or 50 g B. napus seeds were immersed in the 100 ml bacterial suspensions in TSB (priming solution, PS) for 2, 4, 6 or 24 h incubated at 28°C on the orbital shaker at 125 rpm providing oxygenation and even spread of the contents. Bioprimed seeds were subsequently dried overnight (>12 h, Apaptis-A1000 chamber, Conviron, Isleham, Cambridgeshire, UK) at 28 or 24°C, as indicated, in netted bags in a food dryer (Dörrex Dehydrator, Stöckli AG, Netstal, Switzerland) for the experiments with the Hertz cultivar, or in an Adaptis A1000 chamber with airflow (Conviron Europe LTD., Isleham, UK) for the Campus cultivar. The seed moisture contents prior to and after priming were analysed for each of the different treatments and seed drying temperatures (Supplementary Table S1). For TSB-priming and hydropriming, 100 ml TSB (without bacteria) and deionized water (dH2O) were used, respectively, otherwise, the seed priming was conducted using the same procedure described above. To investigate the effects of the different TSB components, mock seed priming without bacteria was conducted: ‘TSB-primed without glucose’, ‘Salt-primed’ with TSB salts only (85.6 mM NaCl, 14.4 mM K2HPO4) and ‘Glu-primed’ (hydroprimed with 13.9 mM glucose).
For the additive biopriming with kimchi paste (Cooks’ Ingredients Spicy-Sour Kimchi Paste, Waitrose, UK), a hot and spicy paste with garlic and dried chilli for preparing the traditional Korean side dish (Seo et al., Reference Seo, Park, Kim, Cho, Kwon and Son2020; Lee and Ko, Reference Lee and Ko2021; Cha et al., Reference Cha, Kim, Park, Lee, Roh, Son and Whon2023), 1, 5 or 10 g kimchi paste was mixed into the 100 ml bacterial suspensions in TSB prior to the seed immersion. For the initial compatibility assays, 1-cm filter paper discs were soaked in a mixture of kimchi (0, 1, 5 or 10 g) in 100 ml autoclaved dH2O. The discs were placed equidistant to each other onto KB plates evenly spread with P. fluorescens. These plates were subsequently incubated for 24 h at 28°C to evaluate the possible effects of kimchi paste on bacterial growth.
Storage of dry bioprimed seeds and seed moisture content analysis
Seed moisture content (SMC) (in g H2O per g dry weight) was determined using the moisture analyser (HB43-S Mettler Toledo) as described (Fatelnig et al., Reference Fatelnig, Chanyalew, Tadesse, Kebede, Hussein, Iza, Tadele, Leubner-Metzger and Steinbrecher2024). Dry bioprimed seeds were stored for 0 (i.e. no storage), 4, 8, 12 and 16 weeks, as indicated, before the storage effects on bacterial viability, germination and seedling growth were analysed. Two storage conditions were used, as indicated, either at 15°C in vacuum-sealed 95 mm × 150 mm aluminium bags (Lamizip Stazakken, DaklaPack Europe B.V., Lelystad, The Netherlands) for the experiments with the Hertz cultivar, or at 21°C in paper bags inside a water-tight polypropylene sealed container (W-D-H 24-24-10 cm, Klip lock, Sainsbury's, UK) with ~600 ml silica gel which generates <10% relative humidity (RH) verified with a humidity data logger (Testo Ltd, Alton, Hampshire, UK) for the experiments with the Campus cultivar. The seed moisture contents prior to and after storage were analysed for each of the different treatments and seed drying temperatures (Supplementary Table S1).
Quantification of bacterial viability
Directly after biopriming and seed drying (0 weeks) or after the indicated storage period (4–16 weeks), 2.5 g of bioprimed seed samples (~454 and ~535 seeds for the Hertz and Campus cultivars, respectively) were washed to elute the P. fluorescens bacteria from the surface of the dry bioprimed seeds. The washing was conducted with 25 ml of 154 mM NaCl, 0.1% (v/v) Tween 20 for 3 h in flasks on an orbital shaker at 125 rpm at 21°C. Appropriate serial dilutions of the resulting wash solutions were plated on KB agar in triplicates and incubated at 28°C. After 24 h, the colony-forming units (CFUs) were counted and used to calculate the number of viable P. fluorescens bacteria in the 2.5 g seed samples. From these numbers, the number of viable bacteria per seed and the relative bacterial viability in per cent of the PS were calculated. Two (Figs. 3A and 7A) or three (Fig. 4E) biological replicates (independent biopriming samples) and for each of these three technical replicates (CFU plates) were used.
Scanning electron microscopy
For the scanning electron microscopy, dry samples of ungerminated and germinated B. napus cv. Campus seeds were mounted on 12.5 mm aluminium specimen stubs and sputter coated with conductive carbon material. Images were captured using a Hitachi S-3000N SEM device at an acceleration voltage of 15 kV.
Germination and seedling assays
Seed germination assays were performed using 3 replicates of 30 seeds in 60-mm Petri dishes as described (Loades et al., Reference Loades, Perez, Tureckova, Tarkowska, Strnad, Seville, Nakabayashi and Leubner-Metzger2023). Seeds were imbibed with 1.5 ml of autoclaved deionized water on a layer of filter paper (MN713; Macherey-Nagel, Dueren, Germany) and sealed using parafilm. The assays were performed in MLR-352 Versatile Environmental Test Chambers (Panasonic, Bracknell, UK) under constant white light (100 μmol m−2 s−1) at 16 or 24°C, as indicated. Temperature response assays were performed on a GRD1-LH temperature gradient plate device (Grant Instruments Ltd., Cambridge, UK). Germination was recorded daily using a stereomicroscope and scored as completed upon visible radicle emergence (>1 mm) through the ruptured testa (Schopfer and Plachy, Reference Schopfer and Plachy1984). Germination uniformity (U90%–10%) was quantified as the time difference between T10% and T90% (Lechowska et al., Reference Lechowska, Kubala, Wojtyla, Nowaczyk, Quinet, Lutts and Garnczarska2019; Sano and Seo, Reference Sano and Seo2019). Seedling growth was analysed by measuring the length and dry weight of 10-d seedlings. Seedling biomass was determined using a Mettler Toledo HB43-S moisture analyser.
Thermal-time modelling and statistical analyses
The cardinal temperatures are permissible for germination including base temperature (T b), optimal temperature (T o) and ceiling temperature (T c), and the thermal time constant Θcold(50%) were estimated by population-based threshold modelling (Bradford and Bello, Reference Bradford, Bello, Buitink and Leprince2022) for thermal time (‘heat sums’) as described (Loades et al., Reference Loades, Perez, Tureckova, Tarkowska, Strnad, Seville, Nakabayashi and Leubner-Metzger2023). Statistical analyses of obtained germination curves were by using a non-linear fit on GraphPad Prism v10 (GraphPad Software, San Diego, CA, USA). This programme was also used to calculate mean ± SD and SEM values and to conduct ANOVA and t-test statistical analyses which were used as P-values * < 0.05, ** < 0.01, *** < 0.001, **** < 0.0001.
Results
Morphological and physiological characterization of B. napus cv. Campus seeds
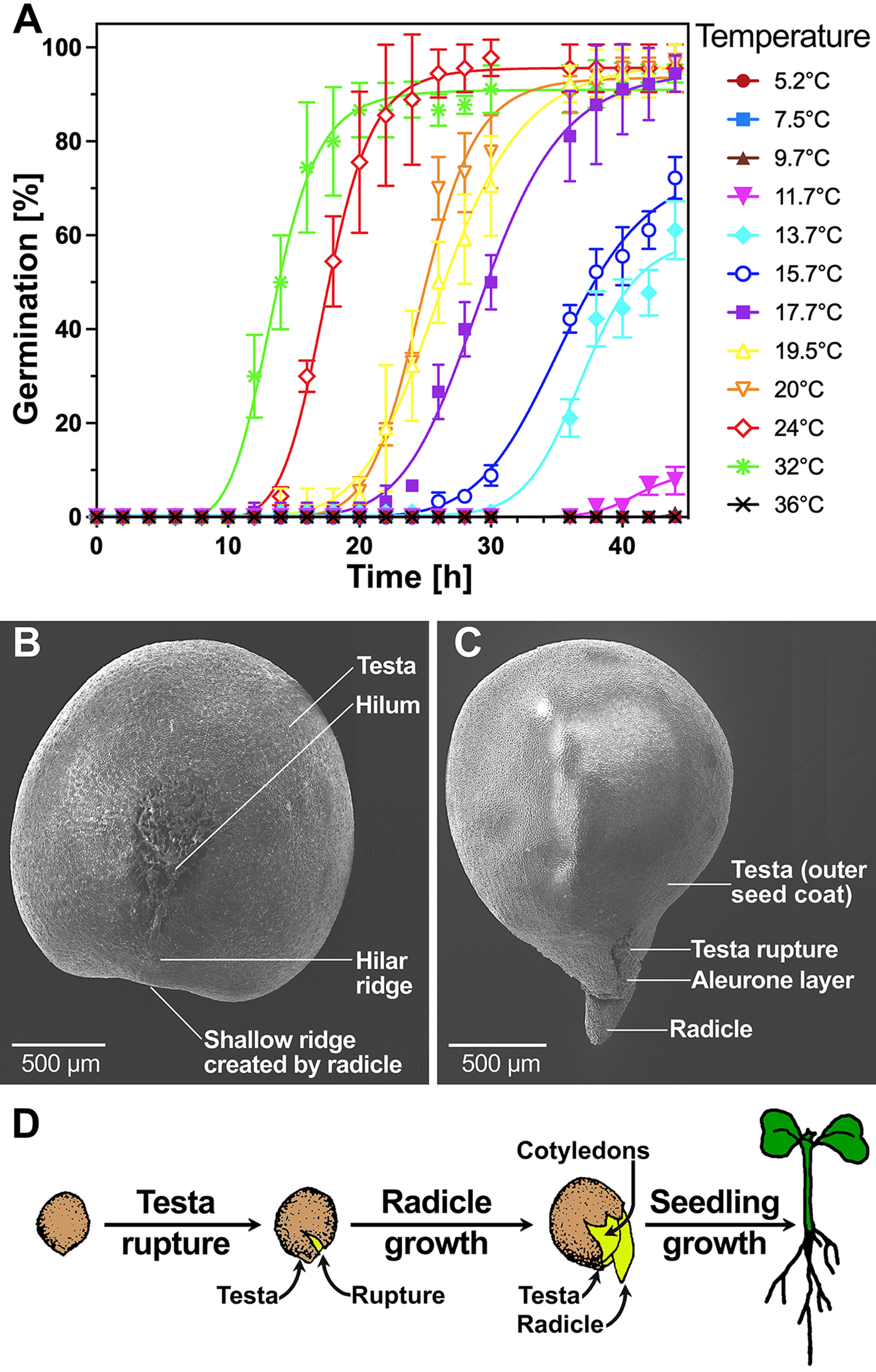
B. napus seeds are ideal in their size, shape and germination physiology (Fig. 1) to study the effects of biopriming with beneficial PGPR such as P. fluorescens (Fig. 2). The maximum germination percentage (G max) of the B. napus cv. Campus seedlot used was ca 95% across the 18–32°C temperature range (Fig. 1A). Population-based thermal-time threshold modelling (Bradford and Bello, Reference Bradford, Bello, Buitink and Leprince2022) was used to estimate the base (T b = 5.7°C), optimal (T o = 32°C, with 24°C similar in germination speed) and ceiling (T c = 34–36°C) temperatures (Supplementary Fig. S1); the observed values were very similar to other B. napus cultivars (Derakhshan et al., Reference Derakhshan, Bakhshandeh, Siadat, Moradi-Telavat and Andarzian2018). Compared with the germination rate GR50% (inverse of T50%, the time required for the seed population to complete 50% germination) at 24°C (optimal), the 16°C temperature (cold stress) reduced germination to about half the speed (Supplementary Fig. S1). The testa (outer seed coat) of dry B. napus seeds (Fig. 1B) is characterized by micromorphological sculpturing (Supplementary Fig. 2A–C) which is known to differ between Brassica species and cultivars (Zeng et al., Reference Zeng, Wang, Liu and Wu2004, Reference Zeng, Wu and Wang2006). The thousand seed weight of B. napus cv. Campus was 4.68 ± 0.16 g. Visible radicle growth (>1 mm) was used as the completion of germination (Fig. 1C) which is followed by seedling growth (Fig. 1D).
Seed morphology and germination physiology of B. napus cv. Campus. (A) The kinetics of germination (scored as visible radicle protrusion over time) at different temperatures in continuous white light. Mean ± SEM values are presented on triplicate plates with 30 seeds each. For thermal-time analysis of these data, see Supplementary Fig. S1. (B, C) SEM of ungerminated and germinated seeds; scale bar indicates size. Specific regions and tissues are indicated, including the testa (outer seed coat) and the hilum region; enlargements of the testa surface and the hilum regions are presented in Supplementary Fig. S2. Note that the aleurone layer constitutes either inner seed coat (Zeng et al., Reference Zeng, Wang, Liu and Wu2004, Reference Zeng, Wu and Wang2006) or endosperm (Munz et al., Reference Munz, Rolletschek, Oeltze-Jafra, Fuchs, Guendel, Neuberger, Ortleb, Jakob and Borisjuk2017) tissue. (D) Visible events during the germination of Brassica seeds and subsequent seedling growth. Source: Image modified from Finch-Savage and Leubner-Metzger (Reference Finch-Savage and Leubner-Metzger2006).
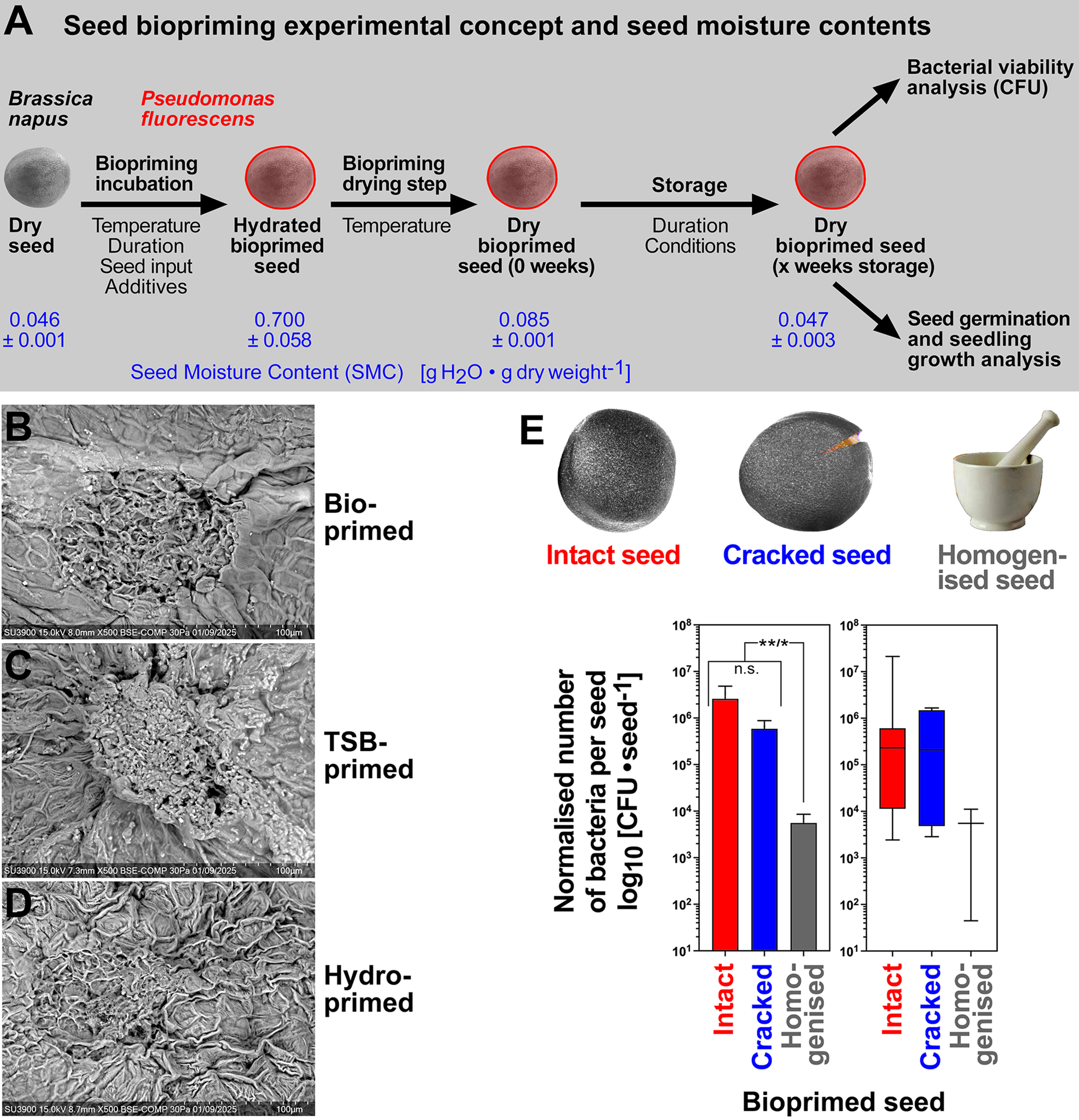
Biopriming concept of B. napus seeds with P. fluorescens DSM 50090. (A) Schematic presentation of the seed biopriming experimental concept used and parameter tested. Note that seed biopriming always includes seed drying after the biopriming incubation. Washing of intact dry bioprimed seeds was used to remove the adhering bacteria to the seed surface, and CFU assays with appropriate dilution plates were used to quantify the number of viable bacteria from the samples. Seed moisture contents are presented exemplarily for 6-h biopriming, 28°C drying and subsequent storage for 3 weeks (21°C, <10% RH). (B–D) SEM of the hilum regions of bioprimed, TSB-primed and hydroprimed seeds. For seed surface areas beyond the hilum regions, see Supplementary Fig. S2. (E) CFUs per seed washed from bioprimed seeds (6-h incubation, 28°C drying). Washings of intact seeds and cracked seeds (artificial testa cracking with defined force) were compared with homogenized seeds. Mean values ± SEM and box plots with minimum to maximum whiskers and medians are presented using a log10 scale of 9, 6 and 3 biological replicates, respectively. One-way ANOVA analyses with the Kruskal–Wallis test demonstrated significant differences between intact and homogenized seeds (P = 0.0094) and between cracked and homogenized seeds (P = 0.0129), but not between CFUs from intact and cracked seeds.
P. fluorescens viability during biopriming and storage of B. napus seeds
Seed biopriming of B. napus was conducted with P. fluorescens DSM 50090 followed by seed drying overnight and subsequent storage of dry bioprimed seeds (Fig. 2A). During the biopriming incubation, the SMC of B. napus cv. Campus increased from 0.046 ± 0.001 to 0.700 ± 0.058 g H2O⋅g dry weight−1. During the subsequent biopriming drying step which completes the process, the SMC was reduced to 0.085 ± 0.001 (drying at 28°C, i.e. just after completion of the ~12 h drying step at the time point corresponding to 0 weeks storage) and decreased further within a week of subsequent storage (21°C, <10%) of bioprimed seeds to 0.045 ± 0.006 g H2O⋅g dry weight−1 (Fig. 2A). Very similar SMC values were obtained for drying at 24°C instead of 28°C, and for mock priming experiments without bacteria (Supplementary Table S1). The biopriming altered the testa surface, especially the ridges of the testa cells, in a different manner than the hydropriming, but this was due to the TSB and not the bacteria (Fig. 2B–D; Supplementary Fig. S2). To quantify the bacteria established on the surface of dry bioprimed seeds, 3 h washings were conducted, followed by quantification of the recovered CFUs. These CFUs represent viable P. fluorescens bacteria which survived the biopriming process including the seed drying and storage (Fig. 2A). Using 6 h biopriming incubation with subsequent seed drying at 28°C, we did not find any evidence for P. fluorescens bacteria inside seeds, the majority if not all were located on the seed surface (Fig. 2E). Comparison of biological replicates demonstrated that there was a high variability in seed surface adherence, and that the CFU counts from homogenized seeds were ~400-fold lower compared to intact seeds. In initial experiments, seed biopriming of B. napus cv. Hertz was conducted with 2–6 h immersion of 10 or 25 g of seeds in 100 ml PS containing P. fluorescens DSM 50090 (Supplementary Fig. S3). Using 25 g as seed input was better than 10 g and resulted in a 60–80% recovery rate (compared to PS) of viable bacteria from the surface of dry seeds directly after biopriming. Subsequent seed storage (15°C in sealed aluminium bags, low oxygen environment) for 4 weeks of dry bioprimed seed reduced the recovery rates to ca 30% (compared to PM) or 40–50% (compared to week 0, i.e. no storage).
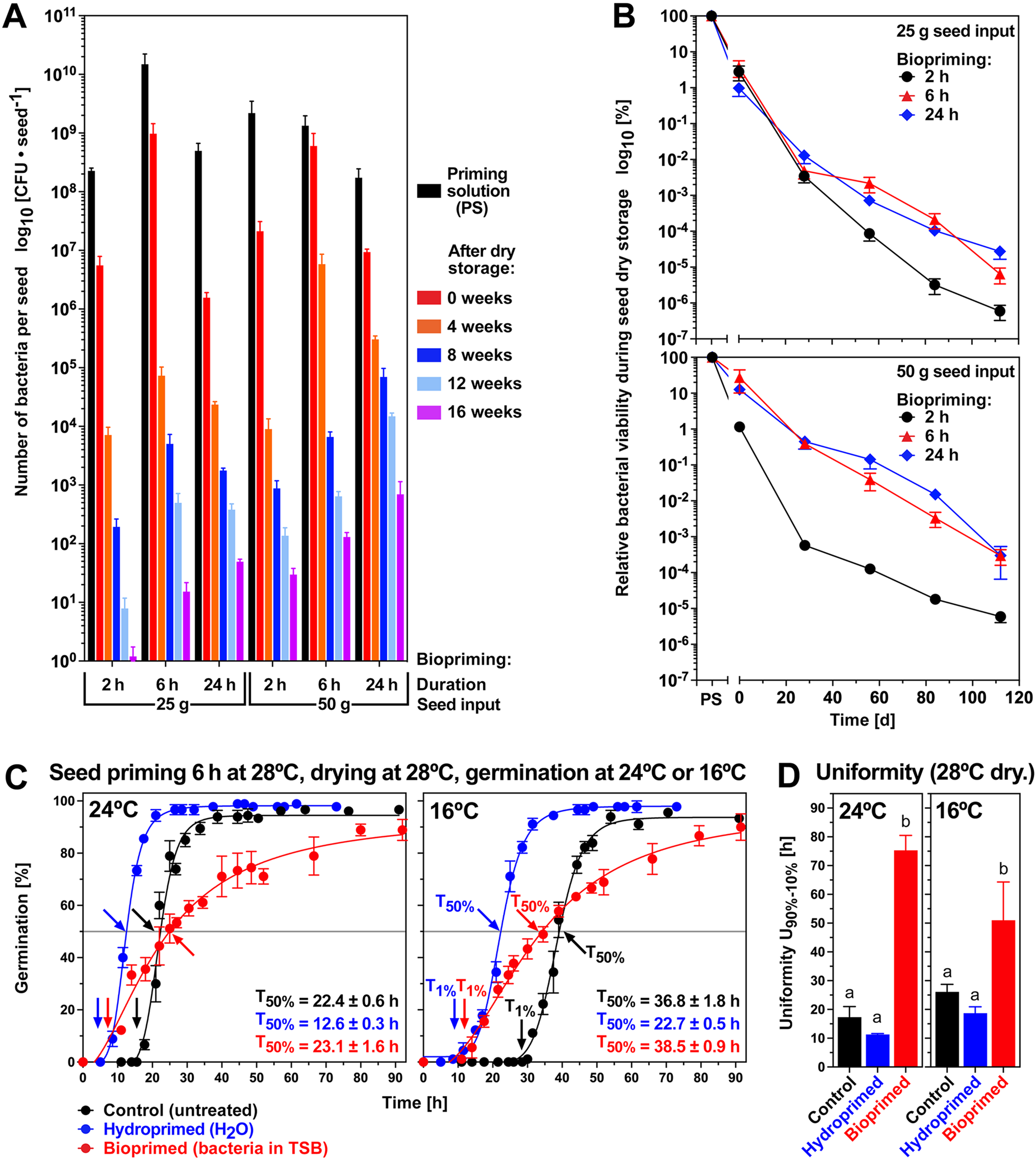
Seed biopriming with B. napus cv. Campus followed the same experimental concept (Fig. 2A). P. fluorescens DSM 50090 was used and further variations of the biopriming and storage conditions were tested. Figure 3A shows that the number of viable bacteria attached per seed was highest for the 6 h biopriming (6.0–9.8 × 108 at 0 weeks). During subsequent storage of dry bioprimed seeds, these numbers of viable bacteria per seed declined rapidly for the 25 g seed input series to 7.3 × 104 (4 weeks) and 5.0 × 102 (12 weeks), and for the 50 g seed input series to 5.8 × 106 (4 weeks) and 6.4 × 102 (12 weeks); the recoveries at 16 weeks were very low in these series. Interestingly, for the 24 h biopriming duration with 50 g seed input which had a lower number of viable bacteria attached per seed (9.4 × 106 at 0 weeks), the best long-term storage recovery was observed with 1.5 × 104 (12 weeks) and 4.5 × 102 (16 weeks). The average recovery rates (compared to PS) of viable bacteria from the surface of dry bioprimed seeds were ca 3 and 14% for the 25 and 50 g seed inputs, respectively. Subsequent seed storage was conducted at dry, aerobic conditions (21°C over silica gel, <10% RH, 21% oxygen) and for longer periods to test the effects on both bacteria (Fig. 3A) and seed quality (see later). Seed storage for 4 weeks led to recovery rates of viable bacteria from the surfaces of dry bioprimed seeds of <2% (compared to week 0). For long-term seed storage, longer biopriming durations (6 and 24 h) and higher seed input (50 g) were better than the 2 h duration and 25 g seed input (Fig. 3B). After a 12-week seed storage period, the recovery rates were down to ca 0.01 and 0.10% for the 25 and 50 g seed input, respectively (Fig. 3A and C).
The effects of seed biopriming of B. napus cv. Campus (unaged seedlot) with P. fluorescens DSM 50090 on bacterial survival during storage and seed germination. (A) Effects of biopriming at (28°C incubation and 28°C drying) duration and seed input, and long-term storage of dry bioprimed seeds at 21°C over silica gel at <10% RH and 21% oxygen. Mean values ± SEM of the number of viable bacteria per seed from seed surface washes are presented on a log10 scale. Corresponding one-way ANOVA analyses of the results are presented in Supplementary Table S2. (B) Time course of relative (compared to PS) bacterial viability during the storage of dry bioprimed seeds (21°C, <10% RH) over a longer period. Mean values ± SEM are presented using a log10 scale. (C) Effect of seed biopriming with P. fluorescens DSM 50090 and hydropriming (6 h with 25 g seed input into the 100 ml PS) on the seed germination of the unaged B napus cv. Campus seedlot. Seed hydropriming was conducted in the same way but without bacteria and with using deionized water (dH2O). Germination was using dH2O and untreated control (non-primed) seed germination is presented for comparison. The kinetics of germination (scored as visible radicle protrusion over time) at 24 (optimal temperature) and 16°C (cold-stress temperature) in continuous white light. Mean ± SEM values are presented of triplicate plates with 30 seeds each. Arrows indicate T1% (onset of the completion of germination) and T50% (50% germination; mean ± SEM T50% values are presented) times required for the unaged seed populations. (D) Germination uniformity U90%–10% calculated as the time difference between T10% and T90% from the germination curves presented in panel C. Significance in one-way ANOVA analysis (P < 0.0001) is indicated by letter code. Note that a high value indicates a reduced germination uniformity.
Effects of biopriming and seed storage of B. napus germination and seedling growth
Figure 3C shows that compared with the untreated controls, seed biopriming did not appreciably affect the maximum germination percentage G max of the B. napus seedlot. It also did not appreciably affect the germination rates GR50% (1/T50%; arrows indicate T50%) at 24 (optimal) and 16°C (cold stress), but it reduced germination uniformity ~4-fold (Fig. 3D) with the seed population exhibiting an earlier onset (T1%; arrows indicate T1%) and later completion (G max) of the germination process (Fig. 3C). In contrast to this, classical hydropriming accelerated the seed population's germination by shifting the entire germination curve (including T1%, T50% and reaching G max) towards earlier times (Fig. 3C). Classical seed hydropriming is a technique involving seed hydration (phases I and II) without permitting the seeds to proceed to water uptake phase III, which is followed by drying them back to a low moisture content similar to the original air-dry state. The effects of hydropriming in accelerating germination were more pronounced upon cold stress at 16°C compared with the optimal temperature at 24°C (Fig. 3C), and hydropriming further increased germination uniformity (Fig. 3D). Short washing pre-treatments preceding the seed biopriming or hydropriming had no additional effects on the germination curves (Supplementary Fig. S4A and B). The seed biopriming experiments presented in Fig. 3 were conducted with the B. napus cv. Campus seedlot acquired in 2021, and by using 28°C for the biopriming incubation (6 h) and the biopriming drying step, which raises the question if different temperatures affect the process (Fig. 2A).
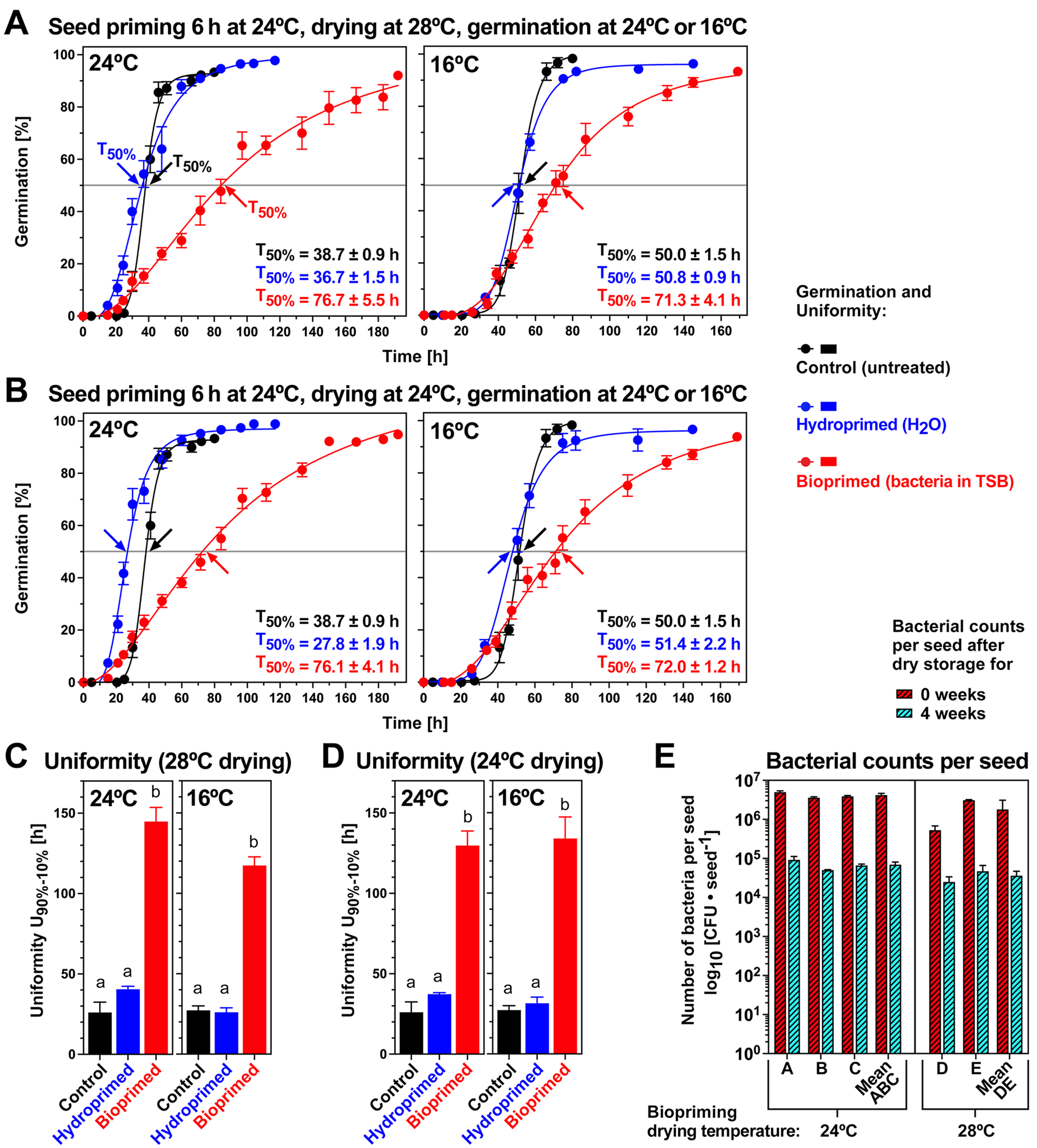
To test if biopriming is also possible at 24°C as biopriming incubation (6 h) temperature and how different drying temperatures may affect the process (Fig. 2A), we conducted seed biopriming with the same B. napus cv. Campus seedlot in 2024 (Fig. 4). The seedlot had, therefore, been stored for 3 years (21°C over silica gel, <10% RH, 21% oxygen) and the germination results demonstrate that it had aged by losing seed vigour, but seed viability was not reduced. The T50% times of the control were increased 1.7-fold (24°C) and 1.4-fold (16°C) without affecting G max (Fig. 4A and B), and the germination uniformity of the control was also not appreciably affected (Fig. 4C and D). Seed biopriming at 24°C biopriming incubation and either 24 or 28°C biopriming drying resulted in similarly delayed germination with 3.3-fold (24°C germination temperature) and 1.9-fold (16°C germination temperature) increased T50% values in the aged compared with the unaged seedlot (Figs. 3C and 4A and B). Germination uniformity of the bioprimed seeds was ~2-fold reduced in the aged compared with the unaged seedlot (Figs. 3D and 4C and D). Further to this, the aged seedlot became less sensitive to hydropriming which only had a small effect when compared to the unaged seedlot. Despite these differences, the general effects on the patterns of the germination curves were the same for aged and unaged seeds, especially with the delayed germination and reduced uniformity caused by the biopriming treatment (Figs. 3 and 4). To test if the temperature of the biopriming drying step affects the number of bacteria adhering to the seed surface, we compared 28 and 24°C as drying temperatures (Fig. 4E). The two drying temperatures did not appreciably differ in the CFU values obtained just after the biopriming drying (0 weeks) and also not after 4 weeks storage of bioprimed seeds (21°C over silica gel, <10% RH, 21% oxygen). We, therefore, conclude that the two drying temperatures did not affect bacterial adherence to the seed surface and also not bacterial survival during storage of bioprimed seeds for which a >100-fold decrease was observed within 4 weeks for the unaged (Fig. 3) and aged (Fig. 4) seeds.
The effects of different temperatures on seed biopriming of B. napus cv. Campus (aged seedlot) with P. fluorescens DSM 50090. (A) Effect of seed biopriming and hydropriming (24°C incubation and 28°C drying; 6 h with 25 g seed input into the 100 ml PS) on the seed germination of the naturally aged B. napus cv. Campus seedlot. Germination was using dH2O and untreated control (non-primed) seed germination is presented for comparison. Mean ± SEM values are presented of triplicate plates with 30 seeds each. Arrows indicate T50% (50% germination; mean ± SEM T50% values are presented) times required for the aged seed populations. (B) Effect of seed biopriming and hydropriming (24°C incubation and 24°C drying) on the seed germination of the naturally aged B. napus cv. Campus seedlot. (C) Germination uniformity U90%–10% calculated as the time difference between T10% and T90% from the germination curves presented in panel A (28°C drying). (D) Germination uniformity U90%–10% of germination curves presented in panel B (24°C drying). Significance in one-way ANOVA analysis (P < 0.0001) is indicated by letter code. (E) Effect of drying temperatures 24°C (left) and 28°C (right) on bacterial survival during biopriming (24°C incubation temperature; 6 h with 25 g seed input into the 100 ml PS) and storage (21°C, <10% RH and 21% oxygen). Mean values ± SEM of the number of viable bacteria per seed from seed surface washes are presented on a log10 scale for three (A–C) and two (D, E) biological replicates using the aged B. napus cv. Campus seedlot.
To test if the bacteria or TSB caused the germination delay of bioprimed seeds, we conducted mock priming experiments of the unaged (Supplementary Fig. S4C and D) and aged (Fig. 5) seedlot without bacteria (TSB-priming: TSB only) and compared it with the bioprimed seeds (TSB + bacteria) by analysing the resultant germination in water at 24 and 16°C. In both experiments, the TSB-primed seeds displayed similarly delayed germination curves compared with the corresponding bioprimed seeds, demonstrating that it is the TSB and not the bacteria that cause this effect. Seed hydration also occurs during biopriming, but in contrast to hydropriming which uses pure water (dH2O), the hydration during biopriming occurs in TSB which contains 85.6 mM NaCl, 14.4 mM K2HPO4 as TSB salts, 13.9 mM glucose and peptones (17 g pancreatic digest of casein and 3 g peptic digest of soybean per litre) which could affect germination by conferring higher osmolarity and other inhibitory effects. To investigate which of the TSB components causes the negative effects on germination, we compared mock priming with TSB (TSB-priming), priming with TSB without glucose, priming with TSB salts (salt-priming with 85.6 mM NaCl and 14.4 mM K2HPO4), hydropriming (H2O) and hydopriming with 13.9 mM glucose (Fig. 5). The comparison of these mock priming experiments (drying at 28 or 24°C) with germination analysis on water at 24 and 16°C demonstrated that the priming with TSB salts alone delivered the same reduction on germination speed and uniformity (Fig. 5). Priming with glucose alone essentially delivered the same effects as hydropriming (Figs. 4 and 5). We, therefore, conclude that the observed negative effects of the biopriming on seed germination speed and uniformity are in fact a salinity effect.
The effects of temperatures and TSB components on the seed priming of B. napus cv. Campus (aged seedlot). (A) Effect of mock priming and hydropriming (24°C incubation and 28°C drying; 6 h with 25 g seed input into the 100 ml PS) on the seed germination of the naturally aged B. napus cv. Campus seedlot. As mock priming solutions TSB, TSB without glucose, TSB salts and glucose were compared in the concentrations indicated. Germination was using dH2O and untreated control (non-primed) seed germination is presented for comparison. Mean ± SEM values are presented on triplicate plates with 30 seeds each. T50% mean ± SEM values are presented. (B) Effect of mock priming and hydropriming (24°C incubation and 24°C drying) on the seed germination of the naturally aged B. napus cv. Campus seedlot. (C) Germination uniformity U90%–10% calculated as the time difference between T10% and T90% from the germination curves presented in panel A (28°C drying). (D) Germination uniformity U90%–10% of germination curves presented in panel B (24°C drying). Significance in one-way ANOVA analysis (P < 0.0001) is indicated by letter code.
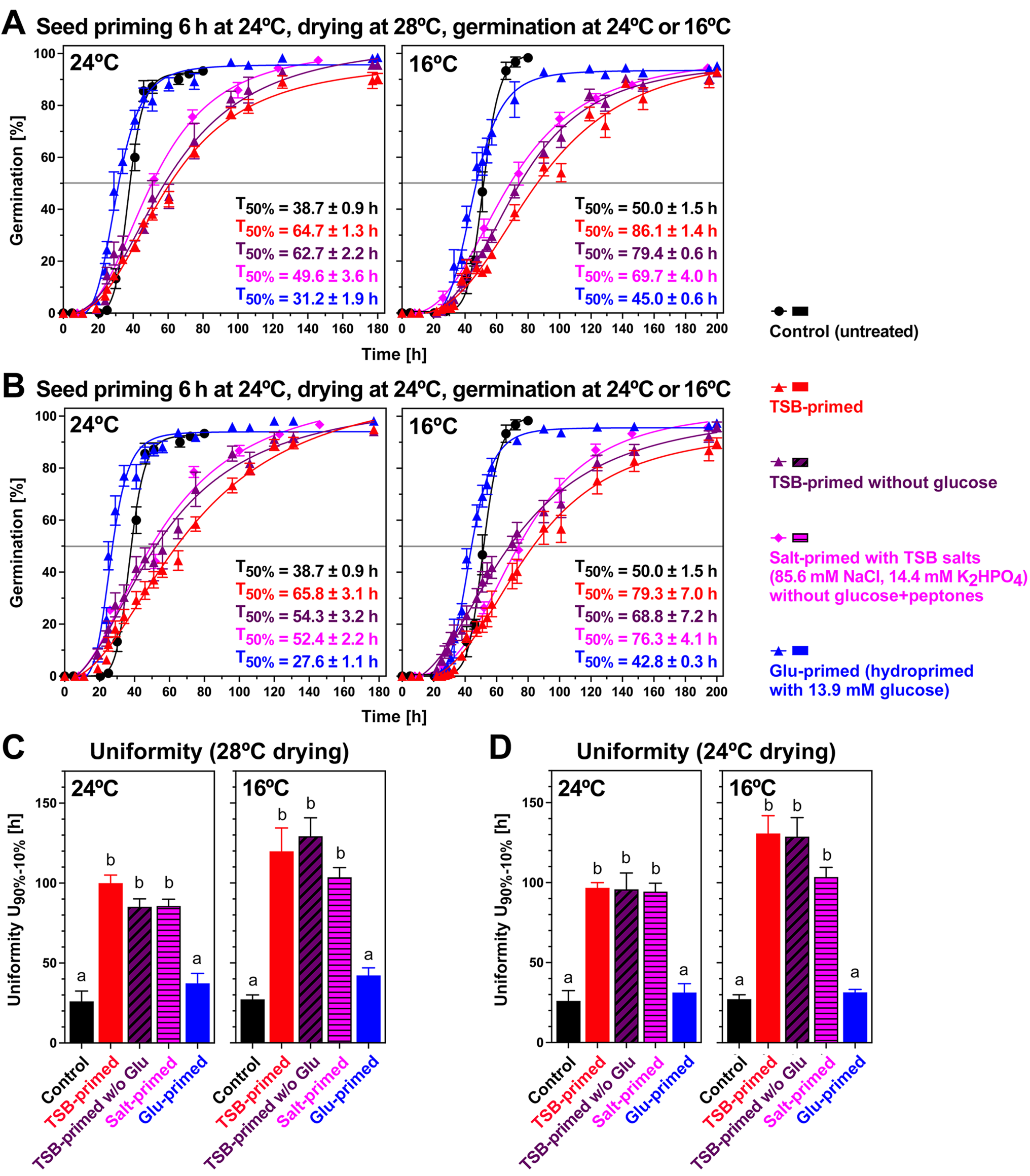
Figure 6 shows that neither TSB-priming nor biopriming with different priming incubation times (2, 6 or 24 h) and seed input (25 or 50 g) appreciably affected germination and seedling growth. The reducing effects of TSB-priming and biopriming on germination speed, as compared to hydropriming, were the same directly after the priming treatment (Fig. 6A, top panel) and after 4 months of seed storage (Fig. 6B, top panel). Neither of the priming treatments changed G max, which was also not affected after 4 months of seed storage (Fig. 6A and B, middle panels). In contrast to hydropriming, which did not affect the biomass of 10-d seedlings, biopriming slightly reduced the seedling biomass (Fig. 6A and B, bottom panel). In summary, seed biopriming and the subsequent 4-month storage did not have any negative effects on B. napus germination and seedling viability.
The effects of seed biopriming, TSB-priming and hydropriming and seed storage on germination and seedling growth of B. napus cv. Campus (unaged seedlot). (A) The effects of seed biopriming, TSB-priming and hydropriming without seed storage; analysis was directly after seed drying. Seed biopriming was conducted for 2, 6 or 24 h with 25 or 50 g seed input, as indicated, into the 100 ml priming medium with P. fluorescens DSM 50090 #2. The corresponding TSB-priming and hydropriming was conducted in the same way, but without bacteria and with using TSB and deionized water (dH2O), respectively. Seed germination speed (germination percentage at 48 h), maximum germination percentage G max (at 10 d), and seedling biomass (at 10 d) were analysed for germination and seedling growth at 16°C in continuous white light. (B) The effects of 4-month storage (21°C, <10% RH) of bioprimed, TSB-primed and hydroprimed dry seeds on germination speed, G max, and seedling biomass analysed for germination and seedling growth at 16°C in continuous white light. These results correspond to the bacterial viability at 16 weeks presented in Fig. 3A. (A, B) Mean ± SEM values are presented of triplicate plates with 30 seeds each. One-way ANOVA demonstrated that germination speed, but not G max or seedling biomass differed significantly with P-values < 0.0001. Unpaired t-tests were therefore conducted in addition to compare hydroprimed versus bioprimed, hydroprimed versus TSB-primed and TSB-primed versus bioprimed. These results are presented in the top panels with P-values indicated (**** < 0.0001, **0.0022); TSB-primed and bioprimed seeds were not significantly (n.s.) different.

Additive seed biopriming using kimchi as an example
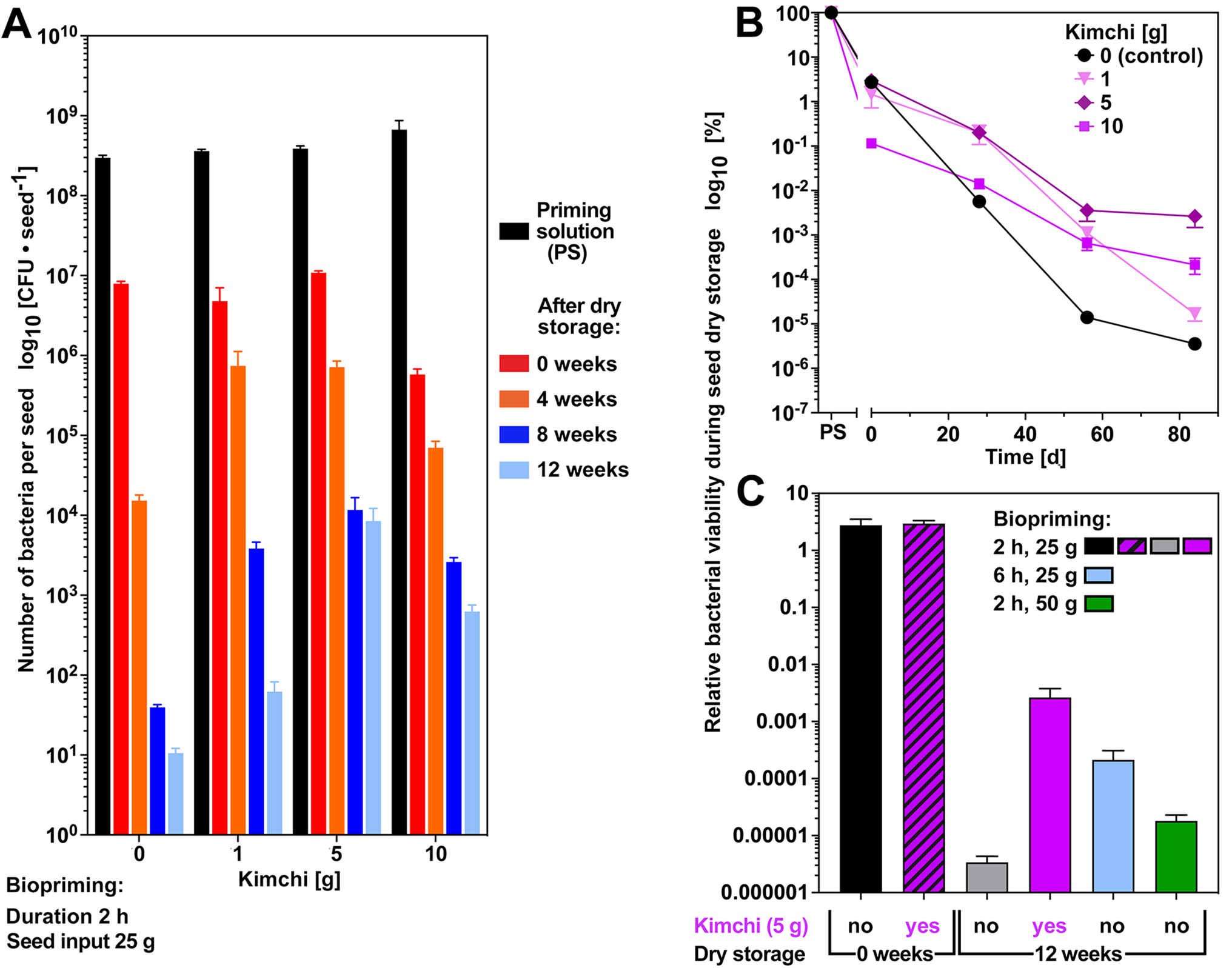
To investigate if additions to the biopriming solution can improve the storability bacteria on dry bioprimed seeds, we decided to use kimchi paste as an example of the case study. Kimchi is a traditional fermented Korean food product prepared by salt-blended vegetables (Lee and Ko, Reference Lee and Ko2021) and the details of the kimchi paste composition used are provided in the discussion. Initial compatibility assays demonstrated that P. fluorescens growth was not inhibited by kimchi paste (Supplementary Fig. S5). Figure 7 shows that the addition of 1–5 g kimchi paste (‘kimchi’ hereafter) to the biopriming solution (2 h biopriming, 25 g seed input) did not negatively affect the number and viability of bacteria per seed recovered from dry seeds directly after the biopriming (0 weeks). It was 7.9 × 106 CFU per seed without kimchi and with kimchi 4.8 × 106 (1 g) and 1.1 × 107 (5 g) (Fig. 7A). Analysis of subsequent storage (21°C, <10% RH, 21% oxygen) of dry bioprimed seeds for 4–12 weeks revealed that 1–10 g kimchi significantly improved the recovery rates of viable bacteria from the surfaces of dry bioprimed seeds (Fig. 7A). All concentrations of kimchi were equally effective after 8 weeks of storage, but the best stabilizing effect long-term was obtained with 5 g kimchi (Fig. 7B). The recovery rate (compared with week 0) of viable bacteria from the surfaces of dry bioprimed seeds after 12 weeks storage was 0.1% for 5 g kimchi. Compared with only 0.0001% without kimchi, this is an almost 800-fold stabilization of bacterial viability in long-term seed storage. Without kimchi, a decline in bacteria per seed from 7.9 × 106 (0 weeks) to 1.5 × 104 (4 weeks) and 1.1 × 101 (12 weeks) was obtained. For the 5 g kimchi, the stabilization of viability was obvious in the higher number of bacteria per seed values: 1.1 × 107 (0 weeks) to 7.1 × 105 (4 weeks) and 8.5 × 103 (12 weeks). When compared with other parameter such as increased biopriming duration and increased seed input, the 5 g kimchi provided the best stabilization result in the long-term storage (Fig. 7C). Additive biopriming with kimchi did not appreciably affect the initial attachment of bacteria to seeds (0 weeks), but improved bacterial viability in long-term seed storage.
The effects of additive biopriming with kimchi paste on bacterial viability during seed storage. (A) Seed biopriming of B. napus cv. Campus was conducted with P. fluorescens DSM 50090 and different concentrations of kimchi paste as an additive in the PS. Biopriming was for 2 h with 25 g seed input, and long-term storage of dry bioprimed seeds was at 21°C over silica gel at <10% RH. Mean values ± SEM of number and percentages (relative to PS) of viable bacteria from seed surface washes are presented on a log10 scale. See Supplementary Table S3 for the corresponding one-way ANOVA analysis. (B) Time course of relative (compared to PS) bacterial viability during the storage of dry bioprimed seeds (21°C, <10% RH) over a longer period. Mean values ± SEM are presented using a log10 scale. (C) Relative bacterial viability during 12-weeks storage of dry bioprimed seeds (21°C, <10% RH) with the stabilizing effect of 5 g kimchi compared to other biopriming parameter. Note that an initial compatibility experiment verified that the kimchi paste itself did not inhibit bacterial growth (Supplementary Fig. S5).
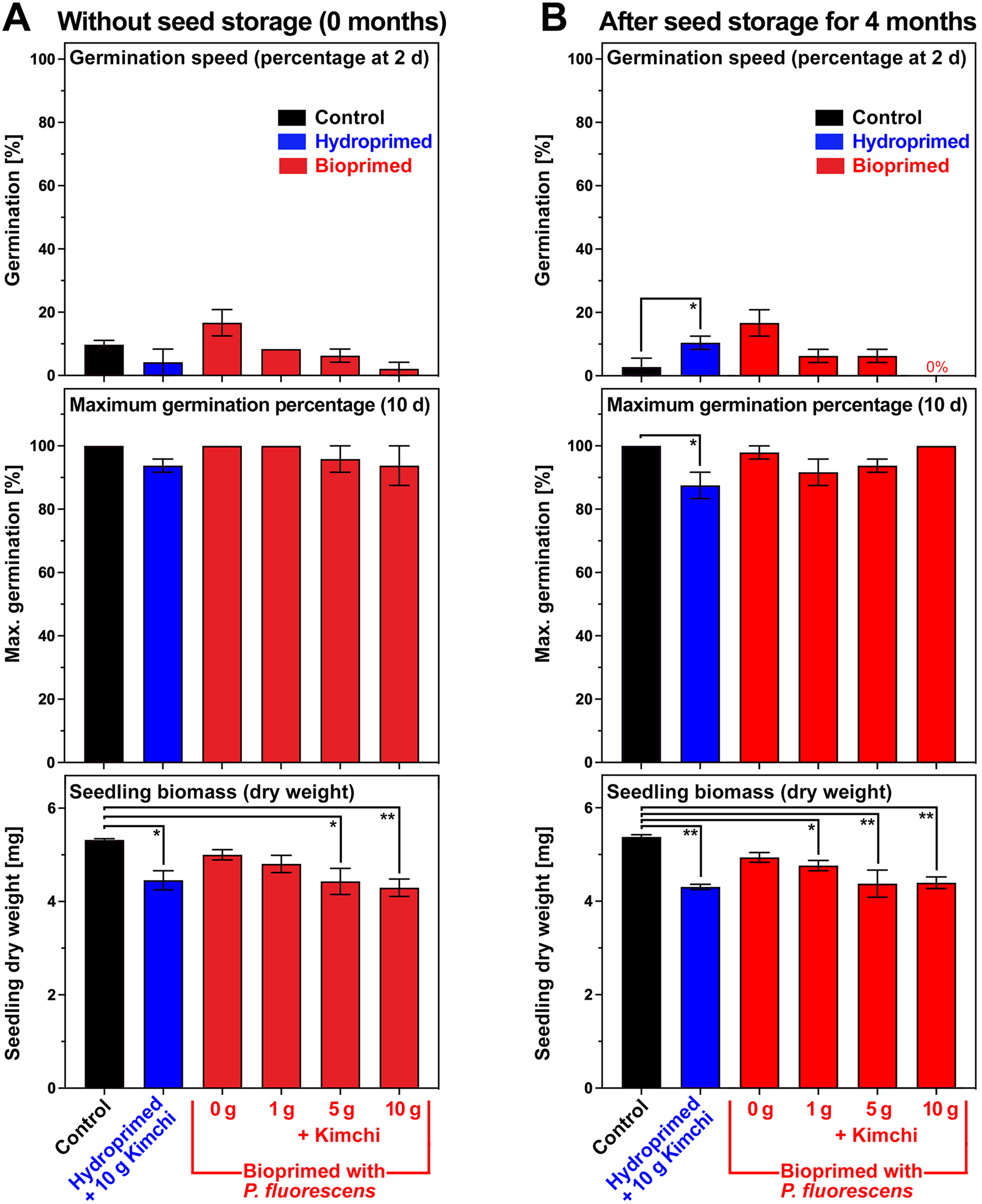
To test if kimchi affected germination and seedling growth during seed biopriming and storage, we analysed germination speed, G max and seedling biomass without and with 4 months of seed storage (Fig. 8). Kimchi did not appreciably affect germination speed and G max of dry bioprimed seeds (Fig. 8A), and also not after 4 months of seed storage (Fig. 8B). Hydropriming of seeds with 10 g kimchi did not appreciably affect their germination, but G max after 4 months of seed storage was slightly reduced (Fig. 8). Seedling biomass was slightly reduced after hydropriming or biopriming with 5–10 g kimchi, as well as after 4 months of seed storage. Therefore, taken together, no obvious major negative effects of additive seed biopriming with kimchi on germination and production of viable seedlings were observed, but kimchi had a major stabilization effect on P. fluorescens viability during the long-term storage of dry bioprimed seeds.
The effects of additive biopriming with kimchi paste on B. napus seed germination and seedling growth. (A) Seed biopriming of B. napus cv. Campus was conducted with P. fluorescens DSM 50090 and different concentrations of kimchi paste as an additive as described in Fig. 7. Hydropriming (dH2O, without bacteria) with 10 g kimchi paste addition was used for comparison. Seed germination speed (germination percentage at 48 h), maximum germination percentage G max (at 10 d) and seedling biomass (at 10 d) were analysed for germination and seedling growth at 16°C in continuous white light. (B) Long-term storage of dry bioprimed seeds was at 21°C over silica gel at <10% RH. Mean ± SEM values are presented on triplicate plates with 30 seeds each. Results from statistical analysis using one-way ANOVA with Dunnett's multiple comparison test are indicated where they were statistically significant with P-values < 0.01 (**) or <0.05 (*).
Discussion
Distinct effects of biopriming and hydropriming on seed germination
B. napus seeds are known to fully imbibe (phase I of the triphasic water uptake curve) within ca 5 h (Schopfer and Plachy, Reference Schopfer and Plachy1984; Weitbrecht et al., Reference Weitbrecht, Müller and Leubner-Metzger2011; Munz et al., Reference Munz, Rolletschek, Oeltze-Jafra, Fuchs, Guendel, Neuberger, Ortleb, Jakob and Borisjuk2017) and at optimal temperature will stay in water uptake phase II (metabolic activation) until ca 12 h before transitioning to water uptake phase III and the completion of germination with testa rupture and radicle growth (Fig. 1). Seed hydration during hydropriming, in which dH2O was used, occurs with a similar pattern to reach and stay in phase II, and this led to accelerated germination by a shift of the germination curve to earlier times, but without changing the curve's shape, e.g. T1%, T50% and T90% were all earlier at 16 and 24°C in the unaged seedlot when compared to the control, and germination uniformity was roughly similar (Fig. 3). Hydropriming was not very effective with the naturally aged seedlot which had lost seed vigour but retained full seed viability (Fig. 4). The effects of seed biopriming were different: T1% was also earlier, but T50% was roughly unchanged in unaged seeds and much later in aged seeds, and T90% was later at 16 and 24°C, leading to a ~4-fold reduced germination uniformity (Figs. 3 and 4). Using mock priming with TSB and TSB components, we identified that this reduction in germination speed and uniformity was not caused by the bacteria, but solely by the TSB salts (85.6 mM NaCl, 14.4 mM K2HPO4) conferring higher osmolarity, and not any other ingredient of the TSB medium (Fig. 5). B. napus seed germination is known to be inhibited by >100 mM NaCl and water uptake into the seed is severely compromised by 200–300 mM NaCl (Bakhshandeh and Jamali, Reference Bakhshandeh and Jamali2020). Hydropriming (with dH2O) of B. napus compared to osmopriming with 150 mM NaCl resulted in a much lower hydrotime constant and base water potential (Tatari et al., Reference Tatari, Ghaderi-Far, Yamchi, Siahmarguee, Shayanfar and Baskin2020). These authors found that NaCl-osmopriming improved water stress tolerance most likely by the absorption of NaCl during the priming treatment, which results in osmotic adjustment and allows to germinate at lower water potentials. On the other hand, this change in germination speed due to the seed biopriming was only a transitionary TSB effect as neither G max nor seedling growth was appreciably affected by the remnants of the TSB ingredients associated with the bioprimed seeds (Fig. 6; Abuamsha et al., Reference Abuamsha, Salman and Ehlers2011b). The seed water contents did not differ between TSB-primed and bioprimed (TSB + bacteria) B. napus seeds, but were slightly lower compared to hydroprimed seeds (Abuamsha et al., Reference Abuamsha, Salman and Ehlers2011b). Higher K2HPO4 concentrations (400 mM) can considerably reduce the germination of B. napus seeds (Abuamsha et al., Reference Abuamsha, Salman and Ehlers2011b), but not TSB (86 mM NaCl, 14.4 mM K2HPO4) which therefore is a suitable seed biopriming medium despite the salinity effect caused by the NaCl, and is therefore not negatively affecting germination and the growth of viable seedlings.
Our findings demonstrate that knowledge from classical seed priming (including hydropriming and osmopriming) is relevant for improving seed biopriming. This is especially true for the timing and patterns of the water uptake phases (key in hydropriming) to optimize the duration of the biopriming, as well as the threshold values for water uptake in solutions with osmolytes (key in osmopriming). The observed reduced germination speed and uniformity (Figs. 3–5) may not be of relevance in practical applications and for seed germination in soil. However, if further optimization of the biopriming procedure (Fig. 2) is required, reducing the NaCl concentration in the biopriming medium is a good starting point. Bennett and Whipps (Reference Bennett and Whipps2008) demonstrated that carrot and onion seed drum priming can be used for the application of beneficial microorganisms including P. fluorescens. Upscale to commercial seed treatment levels is possible as demonstrated for sugar beet seeds (Shah-Smith and Burns, Reference Shah-Smith and Burns1997). Since biopriming involves seed hydration (i.e. similar to osmopriming if a saline suspension is used), it was proposed that the beneficial bacteria are also taken up into the seed (Müller and Berg, Reference Müller and Berg2008; Mahmood et al., Reference Mahmood, Turgay, Farooq and Hayat2016; O'Callaghan, Reference O'Callaghan2016). Bioprimed dry seeds, therefore, should not only have the beneficial bacteria attached to their surfaces (seed coats) but also be located within the seed. Although this is likely, there is to our knowledge no published work that provides direct evidence for within-seed location of the beneficial microbial inoculant. In our own experiments (Fig. 2E), no additional leaching of bacteria occurred from washing of seeds with cracked testa. Homogenizing B. napus seeds provided ~400-fold lower CFU per seed values compared with washing adhering bacteria off from the surface intact seeds. This could be due to the antimicrobial or lytic activity of seed/plant components which kills bacteria in the seed homogenate. The effectiveness of seed biopriming in alleviating stress during germination and seedling growth and in protecting against the disease was however demonstrated in numerous publications (e.g. Shah-Smith and Burns, Reference Shah-Smith and Burns1997; O'Callaghan et al., Reference O'Callaghan, Swaminathan, Lottmann, Wright and Jackson2006; Müller and Berg, Reference Müller and Berg2008; Moeinzadeh et al., Reference Moeinzadeh, Sharif-Zadeh, Ahmadzadeh and Tajabadi2010; Abuamsha et al., Reference Abuamsha, Salman and Ehlers2011a, Reference Abuamsha, Salman and Ehlers2011b; Deaker et al., Reference Deaker, Hartley and Gemell2012; Mahmood et al., Reference Mahmood, Turgay, Farooq and Hayat2016; O'Callaghan, Reference O'Callaghan2016; Araujo et al., Reference Araujo, da Cruz, Souchie, Martin, Nakatani, Nogueira and Hungria2017; Lally et al., Reference Lally, Galbally, Moreira, Spink, Ryan, Germaine and Dowling2017; Rybakova et al., Reference Rybakova, Mancinelli, Wikström, Birch-Jensen, Postma, Ehlers, Goertz and Berg2017; Costales et al., Reference Costales, Nápoles, Falcón-Rodríguez, González-Anta, Petit and Solá2019; Sandini et al., Reference Sandini, Pacentchuk, Hungria, Nogueira, da Cruz, Nakatani and Araujo2019; Fiodor et al., Reference Fiodor, Ajijah, Dziewit and Pranaw2023). Some bacterial isolates from rhizospheric soil samples, including Serratia marcescens, promoted carrot seed germination (Fiodor et al., Reference Fiodor, Ajijah, Dziewit and Pranaw2023). The P. fluorescens isolate did not affect carrot seed germination which is in agreement with our findings for B. napus seeds. In both studies, the germination and seedling assays were conducted without soil to test whether the seed biopriming affected these processes; the experiments were not designed to investigate the beneficial effects of the bacteria on later growth stages in soil. Seed biopriming with P. fluorescens did also not affect the seed germination of Brassica rapa and Capsicum annuum but increased the seedling root growth of C. annuum (Chin et al., Reference Chin, Lim and Ting2022). Others demonstrated that P. fluorescens can promote growth processes beyond the early seedling state by increasing plant height and harvest yield (Moeinzadeh et al., Reference Moeinzadeh, Sharif-Zadeh, Ahmadzadeh and Tajabadi2010; Mahmood et al., Reference Mahmood, Turgay, Farooq and Hayat2016; Lally et al., Reference Lally, Galbally, Moreira, Spink, Ryan, Germaine and Dowling2017; Sandini et al., Reference Sandini, Pacentchuk, Hungria, Nogueira, da Cruz, Nakatani and Araujo2019; Hanifah et al., Reference Hanifah, Ghadamgahi, Ghosh, Ortiz, Whisson, Vetukuri and Kalyandurg2023).
Adhesion of beneficial bacteria to seed coats during biopriming
Our scanning electron micrographs (SEM) of dry Brassica seeds reveal the microsculpturing of their testa surfaces (Fig. 1; Supplementary Fig. S1; Zeng et al., Reference Zeng, Wang, Liu and Wu2004; Chin et al., Reference Chin, Lim and Ting2022). The surface patterns and properties differ between species and can be altered by coating with biopolymers. This may affect the successful attachment of P. fluorescens to the seed surface as visualized by SEM for B. rapa (Chin et al., Reference Chin, Lim and Ting2022). Biopriming for 24 h resulted in ~104 bacteria attached to the surface of a dry B. rapa seed. Bacteria attachment per seed was, depending on the conditions, 106 to 108 for P. fluorescens on B. napus (this work), ~107 for Pseudomonas chlororaphis and ~106 for Serratia plymuthica on B. napus (Müller and Berg, Reference Müller and Berg2008; Abuamsha et al., Reference Abuamsha, Salman and Ehlers2011b). Compared to the biopriming control (9.4 × 106), our additive priming with kimchi resulted in a similar number (1.1 × 107) of P. fluorescens bacteria attached per B. napus seed. Different forms of formulations including using biopolymers (e.g. methylcellulose, xanthum gum or alginate) or thin film coats were used by others and can increase the number of bacteria attached to bioprimed seeds (Müller and Berg, Reference Müller and Berg2008; Abuamsha et al., Reference Abuamsha, Salman and Ehlers2011b; O'Callaghan, Reference O'Callaghan2016; Rocha et al., Reference Rocha, Ma, Souza-Alonso, Vosatka, Freitas and Oliveira2019; Chin et al., Reference Chin, Lim and Ting2022; Paravar et al., Reference Paravar, Piri, Balouchi and Ma2023). The mechanisms for this can be manifold and have not been fully explored in these examples. Microbe adherence to seeds and seed colonization with a high inoculant abundance make bioprimed seeds a relevant vector to transmit beneficial microbes to seedlings and change the trajectory of plant microbiota assembly (Arnault et al., Reference Arnault, Marais, Preveaux, Briand, Poisson, Sarniguet, Barret and Simonin2024).
Attachment of bacteria to environmental surfaces, including seed coats, plant roots and soil particles, may lead to surface-associated multicellular communities known as biofilms (Branda et al., Reference Branda, Vik, Friedman and Kolter2005; Collins et al., Reference Collins, Smith, Sondermann and O'Toole2020). Pseudomonas species, including P. fluorescens, P. putida and P. aeruginosa, are extensively studied model organisms for biofilm formation of gram-negative bacteria (Collins et al., Reference Collins, Smith, Sondermann and O'Toole2020; Costa-Gutierrez et al., Reference Costa-Gutierrez, Adler, Espinosa-Urgel and de Cristobal2022; Espinosa-Urgel and Ramos-Gonzalez, Reference Espinosa-Urgel and Ramos-Gonzalez2023). Mutants of P. fluorescens and P. putida defective in surface attachment and biofilm formation were used to elucidate the underpinning genes and mechanism; many of these mutants were also defective in attachment to seeds (Deflaun et al., Reference Deflaun, Marshall, Kulle and Levy1994; Espinosa-Urgel et al., Reference Espinosa-Urgel, Salido and Ramos2000; Hinsa et al., Reference Hinsa, Espinosa-Urgel, Ramos and O'Toole2003; Espinosa-Urgel and Ramos, Reference Espinosa-Urgel and Ramos2004; Espinosa-Urgel and Ramos-Gonzalez, Reference Espinosa-Urgel and Ramos-Gonzalez2023). During biofilm formation, free-living, planktonic bacteria encounter a surface and undergo numerous physiological changes to attach to that surface. Once attached, bacteria in a developing biofilm produce an extracellular adhesive matrix consisting of proteins, polysaccharides and extracellular DNA as key components securing the bacterial cells to the surface and each other. Bacteria are able to leave the biofilm through a process of dispersal and become free-living, planktonic cells (Collins et al., Reference Collins, Smith, Sondermann and O'Toole2020; Costa-Gutierrez et al., Reference Costa-Gutierrez, Adler, Espinosa-Urgel and de Cristobal2022; Espinosa-Urgel and Ramos-Gonzalez, Reference Espinosa-Urgel and Ramos-Gonzalez2023). The large adhesion protein LapA, a conserved key element of P. fluorescens, P. putida and other plant-beneficial Pseudomonas species, is secreted to the cell surface where it remains tethered to the bacterial outer membrane and acts to adhere to cell to biotic or abiotic surfaces through various adhesive domains. LapA-mediated biofilm formation is triggered by cyclic di-guanosine monophosphate (c-di-GMP) and involves additional Lap proteins, secretion of extracellular polysaccharides. Knowledge of the Lap/c-di-GMP biofilm regulatory circuit and attachment mechanisms to seeds could be instructive for further improving seed biopriming.
P. fluorescens viability during long-term storage of bioprimed seeds
B. napus germination and seedling growth were not negatively affected by seed biopriming (hydration and drying) with P. fluorescens and the subsequent long-term storage of dry bioprimed seeds as discussed above. In contrast to this, bacterial viability declined during the long-term storage of dry bioprimed seeds; the bioprimed seed product therefore has a limited shelf-life. Recovery rates of viable bacteria during the storage (21°C, <10°C) of dry bioprimed seeds depended on the biopriming conditions (Figs. 2 and 5). In the best case, 24 h biopriming with 50 g seed input, viable P. fluorescens bacteria per seed declined from 9.4 × 106 to 3.0 × 105 after 4 weeks, to 1.6 × 104 after 12 weeks and 4.5 × 102 after 16 weeks (Fig. 2). This is a 630-fold decline during 3 months of dry storage at room temperature. Very similar results were obtained by Abuamsha et al. (Reference Abuamsha, Salman and Ehlers2011b) with P. chlororaphis seed biopriming of B. napus. During their storage experiments of dry bioprimed seeds at 20°C, the recovery of viable bacteria declined from ~107 to 4.1 × 104 after 4 weeks to ~102 after 12 weeks. This decline was reduced when dry bioprimed seeds were stored at 4°C which led to recoveries of 3 × 106 after 4 weeks to 8 × 104 after 12 weeks (a ~800-fold improvement). Further to this, seed storage in addition under anaerobic conditions led to a further improvement with 2 × 106 viable bacteria after 12 weeks (Abuamsha et al., Reference Abuamsha, Salman and Ehlers2011b). These authors made similar observations for Serratia plymuthica, suggesting that temperature and oxygen are major factors affecting the shelf-life of viable bacteria during the storage of dry bioprimed seeds.
Similar findings for the storage conditions were made for the biopriming of onion seeds with P. fluorescens and storage up to 10 weeks (O'Callaghan et al., Reference O'Callaghan, Swaminathan, Lottmann, Wright and Jackson2006), and of sugar beet seeds with P. putida and storage up to 52 weeks (Shah-Smith and Burns, Reference Shah-Smith and Burns1997). In both cases, 4°C was ~1000-fold better than 20°C during the long-term storage. Inoculum preparation, formulation and commercial coatings also affected the recovery of viable PGPB positively or negatively during the storage of dry bioprimed seeds (Shah-Smith and Burns, Reference Shah-Smith and Burns1997; Moënne-Loccoz et al., Reference Moënne-Loccoz, Naughton, Higgins, Powell, O'Connor and O'Gara1999; Müller and Berg, Reference Müller and Berg2008). Similar findings for the shelf-life of rhizobia bacteria were also made for the biopriming of legume seeds (Deaker et al., Reference Deaker, Hartley and Gemell2012; Araujo et al., Reference Araujo, da Cruz, Souchie, Martin, Nakatani, Nogueira and Hungria2017; Costales et al., Reference Costales, Nápoles, Falcón-Rodríguez, González-Anta, Petit and Solá2019). Different drying regimes (20 versus 3 h) of bioprimed seeds can also affect the survival of Pseudomonas bacteria during storage (Moënne-Loccoz et al., Reference Moënne-Loccoz, Naughton, Higgins, Powell, O'Connor and O'Gara1999). The cell surface properties of P. fluorescens are affected by drying as well as by bacterial adaptation to abiotic stresses (Kjærvik et al., Reference Kjærvik, Ramstedt, Schwibbert, Dietrich and Unger2021; Abu Quba et al., Reference Abu Quba, Goebel, Karagulyan, Miltner, Kastner, Bachmann, Schaumann and Diehl2023). Studies of the freeze-drying process of P. fluorescens agree with the role of the drying process and the subsequent storage environment for retaining bacterial viability (Kanyinda et al., Reference Kanyinda, Pierart, Destain, Noki and Thonart2014). These authors found that depending on temperature and the addition of protective compounds, cell damage by freeze-drying can be reduced. Much of the damage, including peroxidation of fatty acids and proteins as well as DNA oxidation, was caused by oxygen. Environmental conditions during seed biopriming, drying and storage are therefore of key importance for optimizing the process.
We found that additive seed priming with kimchi paste significantly improved the shelf-life of P. fluorescens during the long-term storage of dry bioprimed seeds of B. napus seeds (Fig. 5). Without kimchi, a decline in the recovery of viable bacteria per seed from 7.9 × 106 (0 weeks) to 1.5 × 104 (4 weeks) and 1.1 × 101 CFU (12 weeks) was observed. In contrast to this, with 5 g kimchi paste as an additive, 7.1 × 105 (4 weeks) and 8.5 × 103 CFU (12 weeks) were obtained, a ~800-fold stabilization of bacterial viability during long-term storage of dry bioprimed seeds. Kimchi is a traditional Korean food product prepared with salt-blended vegetables (Lee and Ko, Reference Lee and Ko2021). It is fermented food of which we used a kimchi paste in our experiments (made from fructose syrup, garlic, dried chili, yellow onion, sugar, rice vinegar, raw soya sauce, fish sauce, garlic and onion powder, citric and malic acid, cayenne pepper) which contains 28% carbohydrates (including 24% sugar), 5.3% fibres, 2.3% protein, 0.8% fat and 3.34% salt (detail information from the manufacturer for the kimchi paste used in our work). Which of these components is causing the stabilizing effect on bacterial viability during the storage of dry bioprimed seeds is not known, but as a product of fermentation kimchi is known as an excellent growth medium for a multitude of fungi and bacteria (Seo et al., Reference Seo, Park, Kim, Cho, Kwon and Son2020; Cha et al., Reference Cha, Kim, Park, Lee, Roh, Son and Whon2023). During the fermentation process, lactic acid bacteria reach 109 to 1010 CFU per gram of food. Based on this, we speculate that kimchi sauce may provide better conditions for bacterial survival similar to encapsulation and coating technologies (Rocha et al., Reference Rocha, Ma, Souza-Alonso, Vosatka, Freitas and Oliveira2019; Szopa et al., Reference Szopa, Mielczarek, Skrzypczak, Izydorczyk, Mikula, Chojnacka and Witek-Krowiak2022; Paravar et al., Reference Paravar, Piri, Balouchi and Ma2023). These alginate-based matrices are not only enhancing microbe survival during seed biopriming and storage but are also environment-friendly biodegradable polymers. Chin et al. (Reference Chin, Lim and Ting2022) reported that all biopolymers tested (alginate, xanthan gum, gum arabic, carboxymethylcellulose) were compatible with P. fluorescens and some of these retained bacterial viability. A recent meta-analysis of biostimulants, which include beneficial microbes as well as compounds of microbial and non-microbial origin, concludes that their add-on yield is on average 18% and reached the highest potential via soil treatment (Li et al., Reference Li, Van Gerrewey and Geelen2022). Kimchi is also rich in minerals such as calcium, iron, magnesium and sodium, which have been shown in some systems to enhance biofilm establishment and maintenance (Song and Leff, Reference Song and Leff2006; Baum et al., Reference Baum, Kainovic, O'Keeffe, Pandita, McDonald, Wu and Webster2009; Cen et al., Reference Cen, Wang, Li, Chen and Wang2024). Future research is therefore needed to further improve the effectiveness of seed biopriming as a promising and innovative technology to deliver beneficial PGPR to sustainable agriculture.
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1017/S0960258525000054.
Data availability
All data is available in the article and supplementary materials.
Acknowledgements
We would like to thank Lena Fatelnig for expert technical assistance.
Author contributions
B.M., J.K., C.P., A.P., M.D.-H., T.S. and G.L.-M. designed research. B.M., J.K., T.S. and C.P. performed research. B.M., J.K., C.P., T.S. and G.L.-M. analysed data. T.S. and G.L.-M. wrote the paper with contributions from all authors.
Funding statement
This research was funded by the Biotechnology and Biological Sciences Research Council to G.L.-M. and T.S. (BBSRC DeTecSeedD grant no. BB/V017462/1) and was co-funded by the BBSRC and Incotec via Croda Europe Limited (UK) and through the Croda CTP training grant to B.M. (grant no. BB/T508913/1). J.K. was funded by Royal Holloway University of London with a Living Sustainability Catalyst PhD Studentship.
Competing interests
The authors declare no conflict of interest.










