Introduction
Clinical guidelines recommend antenatal corticosteroids for pregnant people at risk of preterm birth between 24 and 34 weeks’ gestation to improve newborn health outcomes, 1–3 including reducing risk of respiratory distress syndrome, perinatal and neonatal death and intraventricular haemorrhage. Reference McGoldrick, Stewart, Parker and Dalziel4
Although there is no evidence of clinically important differences between those exposed in utero to antenatal corticosteroids versus placebo from follow-up of randomised controlled trials to 50 years of age, Reference McGoldrick, Stewart, Parker and Dalziel4–Reference St Clair, Walters and Crowther12 concern remains about the potential for long-term adverse effects of antenatal corticosteroid exposure, including in the next generation (F 2), largely based on findings of animal studies. Reference Drake, Walker and Seckl13 A rat study demonstrated reduced birthweight in F 2 rats with both parents exposed to dexamethasone compared with placebo. Reference Drake, Walker and Seckl13 Although there were no differences in glucose metabolism between those exposed to dexamethasone in utero compared to placebo (F 1), in offspring of those exposed to dexamethasone, higher plasma glucose concentrations were seen during glucose tolerance testing at 4 months of age. No difference was seen in plasma insulin concentrations during glucose tolerance testing. At 6 months, those with parents exposed to dexamethasone had higher basal insulin concentrations.
A sheep model has demonstrated reduced birthweight, crown rump length, abdominal and thoracic circumference in F 2 newborn lambs with a parent exposed to dexamethasone compared with placebo, but no difference in weight by 6-8 months of age. Reference Long, Shasa, Ford and Nathanielsz14 At 6 months, lambs with a parent exposed to dexamethasone had increased fasting plasma glucose concentrations and a reduced fasting insulin to glucose ratio compared with those exposed to placebo. During glucose tolerance testing, higher plasma glucose concentrations and a lower insulin to glucose ratio were seen those with a parent exposed to dexamethasone compared with placebo. Additionally, in this sheep model, baseline cortisol and baseline adrenocorticotropic hormone (ACTH) was higher in those with a parent exposed to dexamethasone compared with placebo, and cortisol response to an ACTH stimulus was reduced. Reference Long, Ford and Nathanielsz15
A guinea pig model demonstrated no difference in birthweight between those with a parent exposed to corticosteroid compared with placebo and no differences in body measures or weight gain. Reference Dunn, Kapoor, Leen and Matthews16–Reference Moisiadis, Mouratidis, Kostaki and Matthews19 Findings on the cortisol response to stress were mixed, with some studies and subgroups reporting increased response, some decreased response, and others no change. Reference Iqbal, Moisiadis, Kostaki and Matthews17–Reference Moisiadis, Mouratidis, Kostaki and Matthews19 Changes in hippocampal and anterior pituitary mRNA expression and protein production related to hypothalamic-pituitary-adrenal (HPA) axis function have been reported in those with a parent exposed to betamethasone compared with saline, but these differed between studies. In adulthood, there were no differences between groups in plasma ACTH or cortisol concentrations, oestradiol and progesterone concentrations in females Reference Iqbal, Moisiadis, Kostaki and Matthews17 or testosterone concentrations in males. Reference Moisiadis, Mouratidis, Kostaki and Matthews19 F 2 behaviour was also similar between parental betamethasone and saline groups. Reference Moisiadis, Mouratidis, Kostaki and Matthews19
These animal studies raise concerns about the effect of antenatal corticosteroid exposure on glucose metabolism and HPA axis function in the second generation. However, extrapolating the results of animal studies to humans is challenging. The dosing regimen used by Drake et al. Reference Drake, Walker and Seckl13 included daily administration of glucocorticoids for 7 days, mimicking continuous exposure for a much larger proportion of gestation than in clinical use in humans. Additionally, animal models simulate a ‘normal’ scenario, missing the complexity of preterm birth and pathology. Reference Jobe20
One human study has examined effects of antenatal corticosteroid exposure on the second generation. Reference Abraham21 This was a follow-up of the Auckland Steroid Trial (AST), the first randomised controlled trial assessing the effects of antenatal corticosteroids on newborn health outcomes. Reference Liggins and Howie22 Participants were children aged 5–10 years whose grandmothers were the original (F 0) participants in the AST. Those with a parent exposed to betamethasone had increased fasting glucose concentrations, fasting insulin concentrations and acute insulin release compared with participants with a parent exposed to placebo. No differences were found in the insulin sensitivity index, glucose effectiveness, total or android body fat percentage between groups. Reference Abraham21 However, due to the small sample size (n = 61), differences between groups may have been missed and other important outcomes were not assessed. Reference Abraham21 Additionally, these participants were pre-pubertal, necessitating further follow-up into adulthood, as cardiometabolic risk increases with age. Reference Devers, Campbell and Simmons23
Although studies to date suggest that antenatal corticosteroid exposure may lead to increased metabolic risk factors in the second generation, we have very limited data in humans. Cardiovascular disease is the world’s leading cause of mortality and a major cause of disability Reference Roth, Mensah and Johnson24 and body mass index is a major, age-independent risk factor for cardiometabolic disease. Reference Zhou, Wu, Yang, Li, Zhang and Zhao25,Reference Jousilahti, Tuomilehto, Vartiainen, Pekkanen and Puska26 Therefore, we aimed to assess the effects of parental antenatal corticosteroid exposure on body mass index and other measures of health and wellbeing in the second generation.
Methods
Ethics statement
Ethics approval was granted by Northern A Health and Disability Ethics Committee (20/NTA/37/AM02). Written consent was obtained from F 2 participants 16 years and older. F 2 participants under 16 years assented and written consent was obtained from their parents. If these participants turned 16 years old during the follow-up period, they were asked for their own consent for ongoing participation and data use.
Study methods
The AST was a single centre, double-blind, randomised controlled trial comparing antenatal corticosteroid treatment to placebo for reducing rates of respiratory distress syndrome. Reference Liggins and Howie22 Women expected to give birth preterm at National Women’s Hospital in Auckland were recruited from 1969 to 1974. Rates of respiratory distress syndrome were lower in the 601 babies exposed to betamethasone compared with the 617 babies exposed to placebo. Reference Walters, Lin, Crowther, Gamble, Dalziel and Harding27
AST F1 participants were followed up at 50 years of age, with findings reported elsewhere. Reference Walters, Gamble and Crowther10–Reference St Clair, Walters and Crowther12 F 2 participants were also followed up using the same methods of questionnaires and data linkage. Grandchildren of F 0 AST participants with written informed consent were eligible to participate.
Because this study focuses on the F 2 generation, F 2 participants will be referred to as study participants, their parent (F 1) referred to as the study parent and their grandmothers (F 0) as the participants’ grandmothers.
F1 participants provided information about their children in the F 1 study questionnaire. Parents were asked for permission for the study team to contact children 16 years and older and for consent to include children under 16 years in the study.
The study questionnaires were adapted from the New Zealand Health Survey 2019/2020 28 and the International Primary care Airways Guidelines questionnaire. Reference Price, Tinkelman, Nordyke, Isonaka and Halbert29 Participants 16 years and older completed the questionnaire themselves. Parents of participants under 16 years completed the questionnaire on their child’s behalf.
Participants 16 years and over, and parents of participants under 16 years, were also asked for consent for the study team to access their data from any of seven New Zealand government agencies, Ministry of Health, Whaikaha (Ministry for Disabled People), Accident Compensation Corporation, Testsafe, Ministry of Education, New Zealand Qualifications Authority and the Ministry of Justice.
Outcomes included questionnaire and linked data. The primary outcome was BMI z-score, calculated from weight and height reported in the questionnaire. Secondary outcomes included a composite of overweight or obesity, any diabetes or prediabetes (including gestational diabetes mellitus for women ≥16 years of age), respiratory disease (any of lower respiratory tract infection, chronic lower respiratory diseases, other respiratory disease), cardiometabolic risk factors (≥16 years of age only, any of diabetes, hypertension, dyslipidaemia), neurodevelopmental difficulties (any of visual impairment, hearing impairment, intellectual impairment, cerebral palsy, epilepsy, autism spectrum disorder, attention deficit disorder (ADD) or attention deficit hyperactivity disorder (ADHD)), mental health difficulties (any mental health condition including depression, bipolar affective disorder, schizophrenia, anxiety disorders, suicide/self-harm), fair or poor self- or parent-reported general health, functional difficulties defined by the Washington Group Short Set (proportion with no moderate or no severe disability, moderate disability, severe disability), 30,Reference Hanass-Hancock, Kamalakannan and Murthy31 ≥1 significant parental developmental concern (Parental Evaluation of Developmental Status Reference Glascoe32 ), emotional or behavioural difficulties (Strengths and Difficulties Questionnaire Reference Goodman33 abnormal or borderline), ambulatory hospital presentations or admissions (any (excluding birth admission if applicable), number per ten years alive, time in hospital per ten years alive), allergic conditions other than asthma, birthweight, gestational age at birth, and number of fractures per ten years alive.
Tertiary outcomes included fair or poor oral health (self- or parent-reported, any tooth extractions or signs of more advanced or advanced tooth decay), any convictions (≥ 16 years of age only), unemployment (≥ 16 years of age only), any secondary school qualification (≥ 18 years of age only), and any educational disciplinary action. Components of the secondary composite outcomes were also included as tertiary outcomes.
If a participant was identified to have a certain condition in at least one dataset, they were considered to have the outcome. Participants were only classified as missing the outcome if they did not have information in any of the datasets that contributed to an outcome.
Analysis
The Statistical Analysis Plan was finalised before any analyses took place. Analyses were conducted using SAS® 9.4 (SAS Institute Inc., Cary, NC, USA). An intention-to-treat approach was used, analysing participants according to the initial treatment group to which their grandmother (F 0) was allocated. For participants who withdrew, data up to the point of withdrawal was used unless consent for this was also removed. No imputation was conducted for missing data.
The median and distribution of ages of participants was compared between the betamethasone and control group (Wilcoxon and Kolmogorov-Smirnov tests respectively) to establish whether adjustments for age were required.
As pre-specified in the Statistical Analysis Plan, the steering group reviewed the baseline demographics prior to the outcomes analysis. Socioeconomic status was classified using the New Zealand Deprivation Index (NZDep), an area-based measure of deprivation based on census data, in this case the 2018 census. Reference Atkinson, Salmond and Crampton34 The steering group noted that participants whose study parent was exposed to betamethasone were more likely to have this parent living in a more socioeconomically deprived area (NZDep 8–10) compared with those whose study parent was exposed to placebo. A decision was made to conduct a post-hoc subgroup analysis rather than adjust for deprivation in the main analysis since it may be on the causal pathway between antenatal corticosteroid exposure and the outcomes measured.
This study involved the follow-up of offspring of participants in a clinical trial and the sample size was limited by the number of participants able to be located for whom consent was obtained. For the primary outcome of BMI z-score, the sample size of 144 allows for detection of mean difference equal to half a standard deviation with 90% power (β = 0.10) at a significance level of 5% (α = 0.05). Reference Champely35
Prespecified subgroup analyses examined the effect of F 2 sex assigned at birth, F 1 sex assigned at birth for the parent who was exposed to the AST study drug, and F 1 born preterm or term for the parent who was exposed to the AST study drug. A post hoc subgroup analysis explored the effect of F 1 socioeconomic status (NZDep deciles 8–10 versus 1–7). We performed these subgroup analyses for each of the primary and secondary outcomes as main and interaction effects with treatment allocation.
For participants whose age at assessment was 4–6 years and for whom weight and/or height was missing in the questionnaire, a sensitivity analysis was performed including BMI z-score from the B4 School Check (B4SC) in the analysis. The B4SC is a free assessment of health and development offered to all New Zealand children at four years of age and includes measurement of weight and height. 36
Baseline variables are reported using frequency and percentages for categorical variables and mean and standard deviation or median and interquartile range, as appropriate, for continuous variables. They were compared using chi square tests for categorical outcomes, or Fisher’s exact test where numbers in more than 20% of cells were <5. Student’s T-tests were used for normally distributed continuous outcomes and Wilcoxon tests for non-parametric continuous outcomes.
Statistical analyses comparing the parental betamethasone and parental placebo exposure groups were conducted for all primary, secondary and tertiary outcomes, with a two-sided test at the 5% significance level. No adjustment to the significance level was made for multiple comparisons as is usual for safety analyses. However, all comparisons are clearly reported and potential for type 1 (and 2) errors is acknowledged as a limitation.
Fully saturated models comprising treatment allocation, the age category of participants (≥16 years versus <16 years) and their interaction were created for outcomes that pool data from child and adult questionnaires. Where the main effect of age group or the interaction effect failed to reach conventional statistical significance, a reduced model with only the fixed effect of age category formed the primary analysis method. To account for the nonindependence of twins, siblings and cousins in this cohort, F 2 participants descending from the same F 0 study pregnancy were identified by a unique F 0 identifier that was included as a random effect in all models, except where noted in the subgroup analyses, where it was removed so the model would converge.
Binary outcomes were analysed using generalised linear mixed modelling (log link function, binomial distribution). Results were reported as relative risk (RR) with 95% confidence intervals.
Ordinal outcomes were analysed using ordinal logistic regression (cumulative logit link function, multinomial distribution), reported as an odds ratio (OR) with 95% confidence intervals.
Continuous outcomes were analysed using generalised linear mixed modelling (identity link function, normal distribution). Results were reported as mean difference (MD) with 95% confidence intervals.
Rates were analysed using a Poisson distribution (log link function, offset by the natural logarithim of age) and results were reported as rate ratios with 95% confidence intervals. OpenEpi was used to calculate rates in each treatment group to report in outcome tables. Reference Dean, Sullivan and Soe37
BMI z-scores were calculated using an R package Reference Myatt and Guevarra38 based on World Health Organisation growth charts Reference de Onis, Onyango, Borghi, Siyam, Nishida and Siekmann39,40 using age in days. These growth charts extend to 19 years of age, so z-scores for 19 years were used for participants ≥19 years old. For participants aged 5–19 years, those with a z-score of >1 met the criteria for overweight and those with a z-score of >2 the criteria for obesity. 41 For participants aged 0–5 years, the criteria were a z-score of >2 for overweight and >3 for obesity. 42
Results
Of the 415 F 1 individuals who completed the questionnaire in the 50-year follow up, 255 were known to have children (Fig 1). Of these, 129 had at least one child included in this study. A total of 213 F 2 participants were followed up.
Flow diagram for the Auckland Steroid Trial 50-year F2 follow-up.

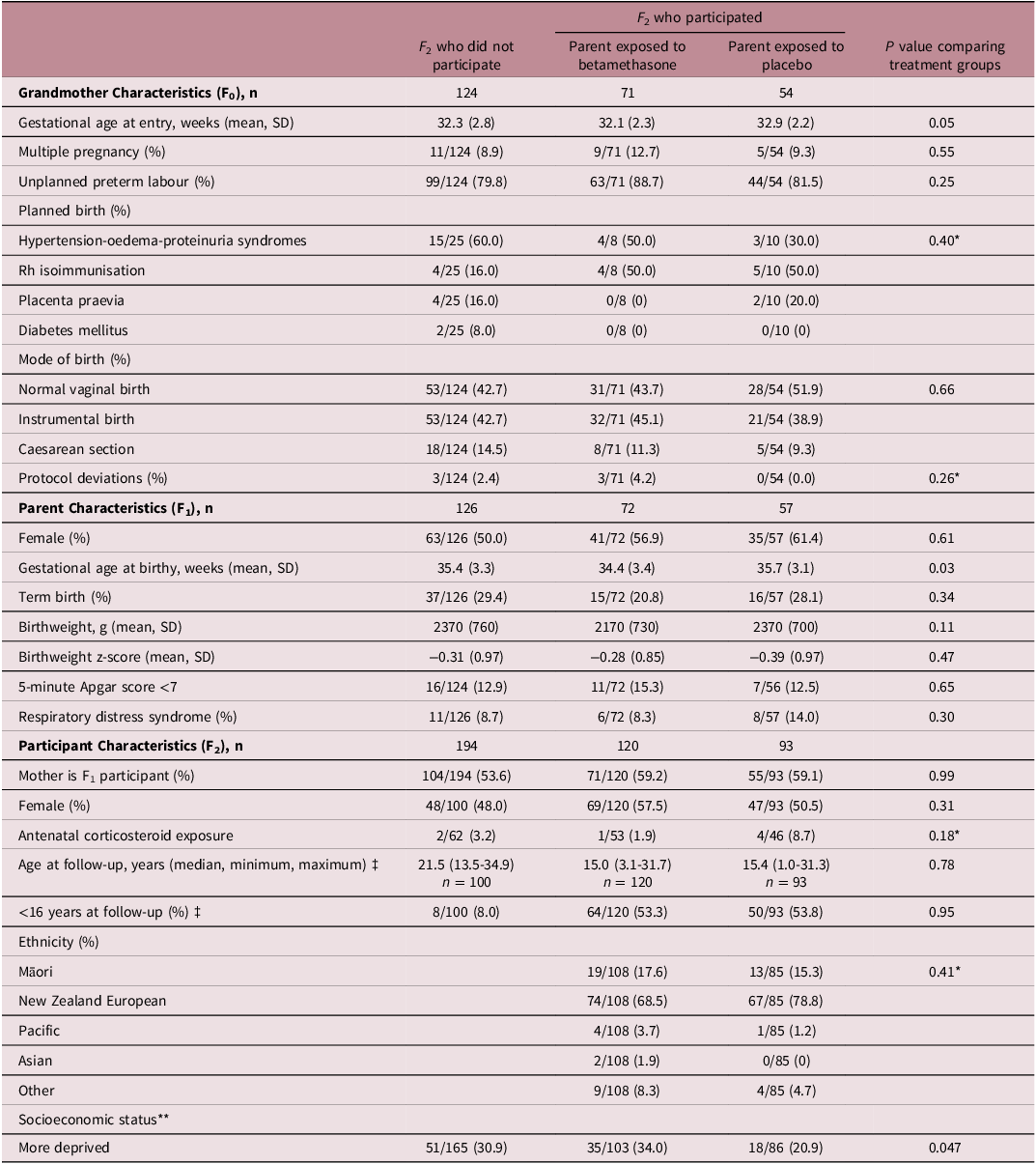
There were 125 grandmothers who had at least one participating grandchild and 124 grandmothers with known grandchildren who did not participate (Table 1). Clinical characteristics were similar at the time of recruitment to the AST in these two groups of grandmothers.
Baseline characteristics of each generation in the Auckland Steroid Trial included in second generation follow-up

SD – standard deviation.
* Fisher’s exact test used rather than chi square test due to small numbers in most cells
** More deprived defined as NZDep 8–10, less deprived defined as NZDep 1–7, based on F1 parent address
‡ Difference between F 2 not participated and participated groups statistically significant (p < 0.001).
For those who were not assessed, age at follow-up was calculated as age at the end of the study period (May 2022).
Of the 125 grandmothers who had at least one grandchild participate, 71 were allocated to betamethasone and 54 allocated to placebo groups (Table 1). Again, clinical characteristics were similar at the time of recruitment to the AST between grandmothers in each treatment group.
There were 129 parents who had at least one child who participated in the study and 126 parents who told us they had eligible children who did not participate (Table 1). There were no differences in demographic or clinical characteristics between these groups of parents.
Of the 129 parents who had at least one child participating, 72 were exposed to betamethasone and 57 were exposed to placebo (Table 1). Those exposed to betamethasone in utero had a lower gestational age at birth compared with those exposed to placebo. As expected, the parents exposed to betamethasone in utero were less likely to experience respiratory distress syndrome compared with those exposed to placebo (8% vs 14%). There were no differences in the other reported demographic or clinical characteristics at birth between these groups of parents.
There were 213 participants and 194 known eligible individuals who did not participate in this study (Table 1). Study participants were younger than those who did not participate (median (minimum, maximum) 15.4 years (1.0–31.7) versus 21.5 years (13.5–4.9), p < 0.001, and 54% were younger than 16 years of age, compared with only 8% of those who did not participate. There were no other differences in demographic or clinical characteristics between participants and those who did not participate.
Of the 213 participants, 120 had a parent exposed to betamethasone and 93 had a parent exposed to placebo (Table 1). For participants whose study parent was exposed to betamethasone, this parent was more likely to live in a more deprived area (NZDep deciles 8–10) compared with those whose parent was exposed to placebo (35/103, 34% versus 18/86, 21%, p = 0.047). There were no other demographic differences between those with a parent exposed to betamethasone compared with placebo and no difference in the proportion of F 2 participants exposed to corticosteroids themselves. No further adjustment was made for age since the median age and age distribution was similar between betamethasone and placebo groups (p > 0.70 for Wilcoxon and Kolmogorov-Smirnov tests).
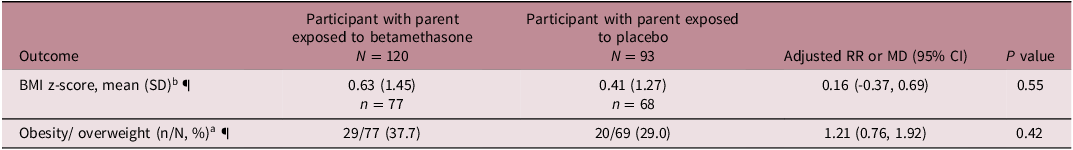
Data for the primary outcome of BMI z-score were available for 144 of 213 participants (68%). There was no evidence of a difference in BMI z-score between those with a parent exposed to betamethasone and those with a parent exposed to placebo (mean (SD) 0.63 (1.45), n = 77 vs 0.41 (1.28), n = 67, adjusted mean difference (aMD) 0.16 (95% confidence interval (CI) (−0.37, 0.69), p = 0.55, adjusted for age category and clustering). The 69 participants who did not have BMI z-score data available were missing weight and/or height from the questionnaire. The proportion of participants missing weight and/or height data did not significantly differ between those with a parent exposed to betamethasone compared with placebo (43/120, 36% vs 26/93, 28%, p = 0.22).
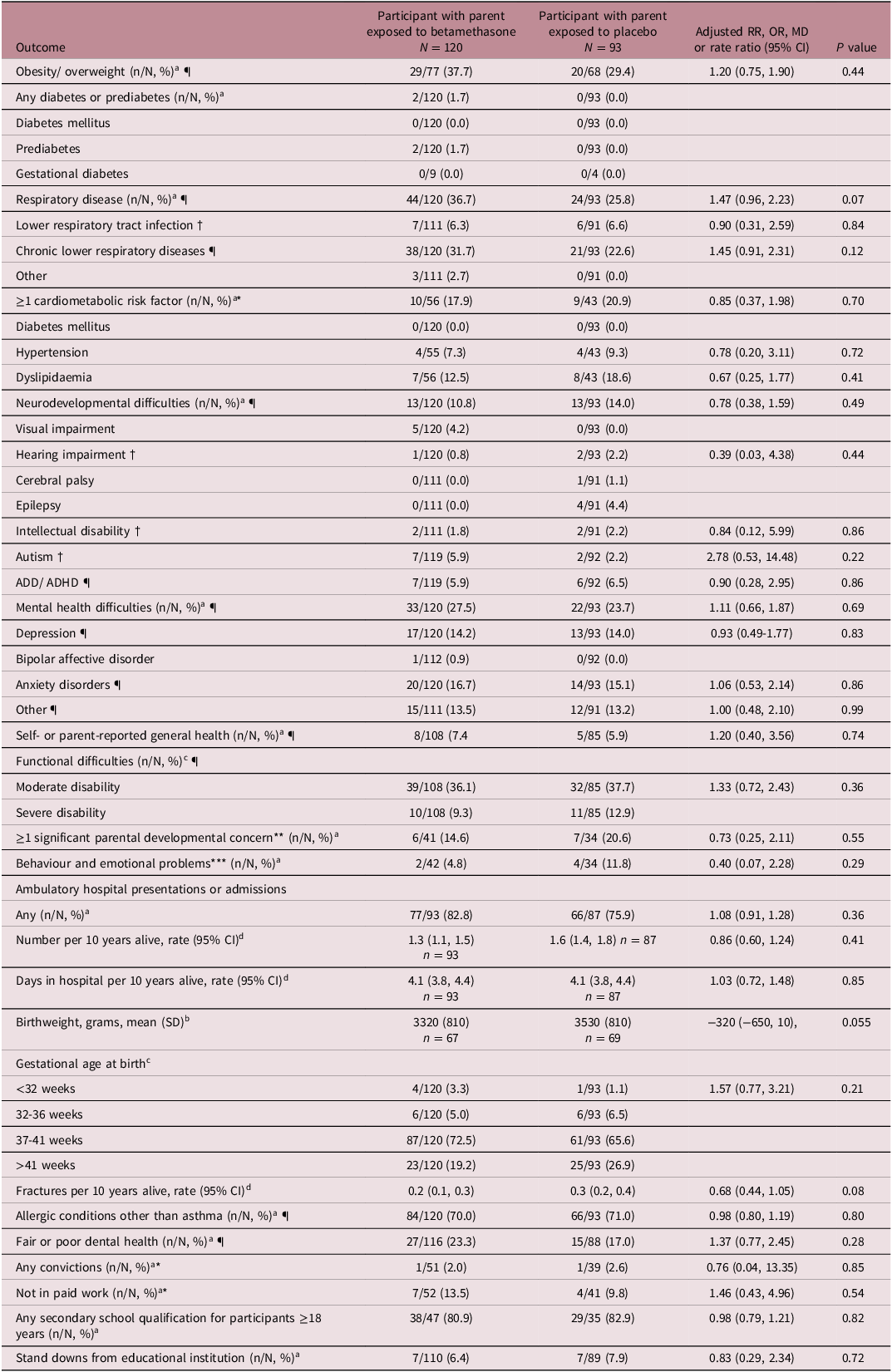
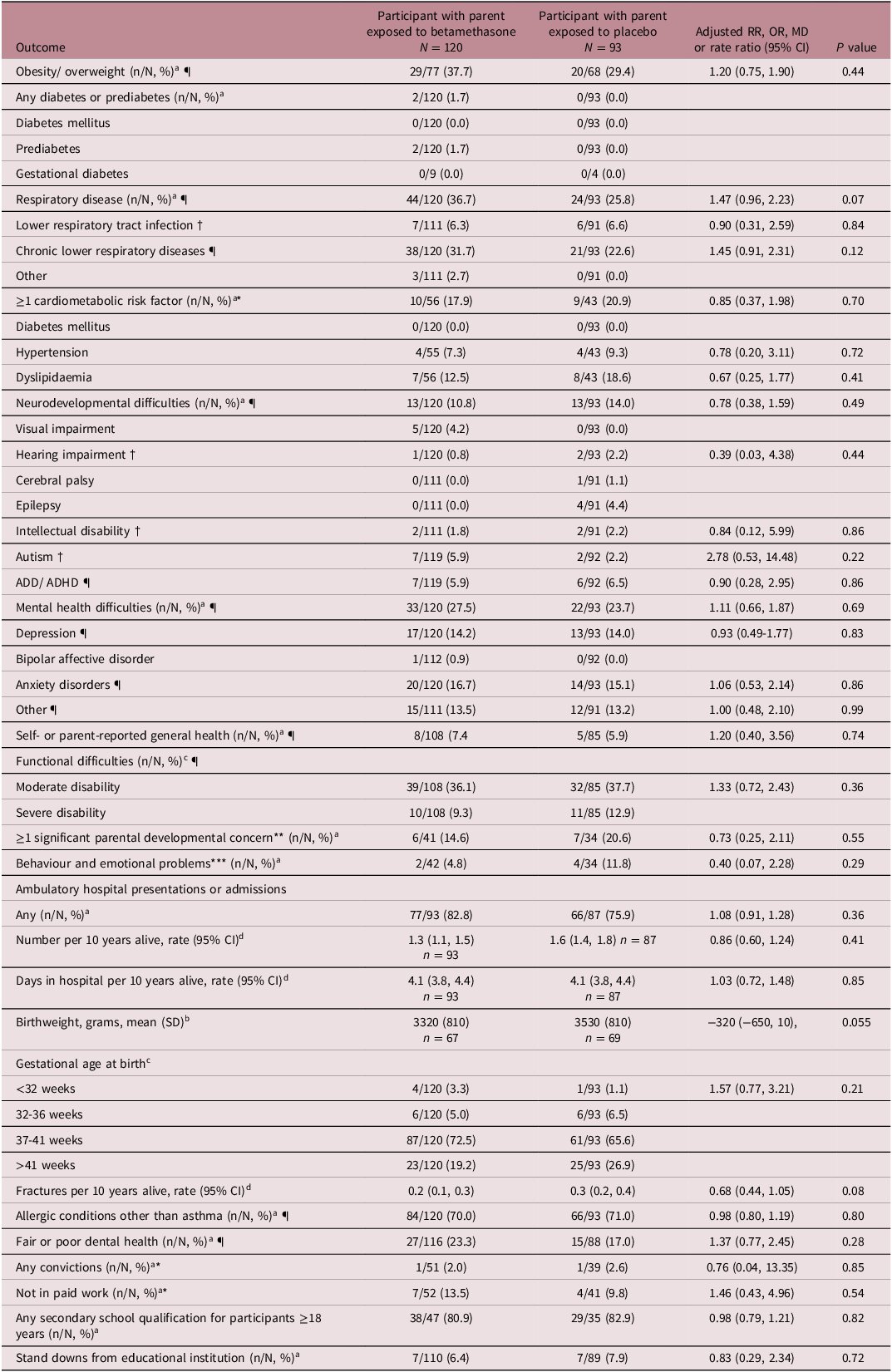
Participants with a parent exposed to betamethasone were more likely than those with a parent exposed to placebo to be overweight or obese, and to have respiratory disease, but these differences were not statistically significant (overweight/obesity adjusted relative risk (aRR) 1.20 (0.75, 1.90), p = 0.44, respiratory disease aRR 1.47 (0.96, 2.23), p = 0.07) (Table 2). Only two participants had prediabetes, and none had diabetes mellitus or gestational diabetes. Of the participants 16 years and older, 19% had at least one cardiometabolic risk factor and of the entire cohort, 12% had a neurodevelopmental difficulty and 26% a mental health difficulty. For these outcomes, there was no evidence of a difference between groups. There was no evidence of a difference between groups for self- or parent-reported general health or functional difficulties. There was also no evidence of a difference between groups for at least one significant parental developmental concern or behavioural or emotional problems at 4 years of age in data from the B4SC, but data were only available for 35% of participants as the B4SC programme was introduced nationally in 2008. 43
Secondary and tertiary outcomes in participants whose parent was exposed to betamethasone or placebo
aRelative risk (RR), bmean difference (MD), codds ratio (OR), adjusted for clustering and age category of participants (<16 years or≥16 years), drate ratio
CI – confidence interval, ADD - attention deficit disorder, ADHD - attention deficit hyperactivity disorder, SDQ – Strengths and Difficulties Questionnaire, LQ – lower quartile, UQ - upper quartile.
*F2 ≥16 years of age only.
The rate of any ambulatory hospital presentations or admissions, number of hospital admissions per ten years alive and time in hospital per ten years alive was similar between groups (Table 2). Those with a parent exposed to betamethasone had a lower birthweight compared with those with a parent exposed to placebo but the confidence interval included no effect (aMD -320 g (-650, 10)). There was a higher rate of term birth and lower rate of post-term birth in participants with a parent who was exposed to betamethasone compared with those with a parent who was exposed to placebo, but these differences were not statistically significant (term births = 87/120 (73%) versus 61/93 (66%), post-term births = 23/120 (19%), versus 25/93 (27%), adjusted odds ratio = 1.57 (0.77, 3.21)). There were similar rates of fractures per ten years alive and allergic conditions other than asthma between groups. There was no evidence of a difference between groups for any of the social outcomes; fair or poor dental health, any convictions, unemployment, any secondary school qualification or stand downs from educational institutions. There was also no evidence of a difference between groups for any of the components of composite outcomes.
In the prespecified subgroup analysis of participant sex, there was no evidence of a difference in outcomes between those with a parent exposed to betamethasone or placebo in female and male participants, and no significant interactions between parental betamethasone exposure and participant sex (Table 3).
Subgroup analysis - primary and secondary outcomes in participants assigned female or male sex at birth whose parent was exposed to betamethasone or placebo

aRelative risk (RR), bmean difference (MD), codds ratio (OR) adjusted for clustering and age category of participants (<16 years or≥16 years), drate ratio.
CI – confidence interval, BMI – body mass index, SD – standard deviation, LQ – lower quartile, UQ – upper quartile. ¶ Fully saturated model not reported as interaction term not statistically significant (p > 0.05).
§ Clustering term removed to get model to converge.
However, in the subgroup analysis investigating the effect of sex of the exposed parent, participants whose mothers were exposed to betamethasone were more likely to have any ambulatory hospital presentations or admissions than those whose mother was exposed to placebo, but this difference was not seen when the study parent was the father (p = 0.007 for interaction, Table 4). In contrast, participants whose father was exposed to betamethasone had a lower birthweight compared with those whose father was exposed to placebo, but this difference was not seen when the study parent was the mother (p = 0.051 for interaction, Table 4).
Subgroup analysis - primary and secondary outcomes in participants whose parent was assigned female or male sex at birth and exposed to betamethasone or placebo

aRelative risk (RR), bmean difference (MD), codds ratio (OR), adjusted for clustering and age category of participants (<16 years or≥16 years), drate ratio.
CI – confidence interval, BMI – body mass index, SD – standard deviation, LQ – lower quartile, UQ – upper quartile.
¶ Fully saturated model not reported as interaction term not statistically significant (p > 0.05).
§ Clustering term removed to get model to converge. † Fully saturated model not reported due to non-convergence.
In the subgroup analysis investigating the effect of parental gestation at birth, in participants whose study parent was born at term, those with a parent exposed to betamethasone were more likely to have any ambulatory hospital presentations or admissions compared with those with a parent exposed to placebo, but this difference was not seen in participants whose study parent was born preterm (p = 0.03 for interaction, Table 5). Additionally, for those whose study parent was born at term, those with a parent exposed to betamethasone were more likely to be born at term and less likely to be born post-term compared those with a parent exposed to placebo, but this difference was not seen in participants whose study parent was born preterm (p = 0.03 for interaction, Table 5).
Subgroup analysis - primary and secondary outcomes in participants whose parent was born preterm or at term and exposed to betamethasone or placebo

aRelative risk (RR), bmean difference (MD), codds ratio (OR), adjusted for clustering and age category of participants (<16 years or≥16 years), drate ratio.
CI – confidence interval, BMI – body mass index, SD – standard deviation, LQ – lower quartile, UQ – upper quartile.
¶ Fully saturated model not reported as interaction term not statistically significant (p > 0.05).
§ Clustering term removed to get model to converge.
† Fully saturated model not reported due to non-convergence.
There was no evidence of a difference in outcomes between those whose study parent lived in a more deprived versus less deprived area, and no significant interactions between parental betamethasone exposure and socioeconomic deprivation (Table 6).
Post hoc subgroup analysis – primary and secondary outcomes in participants whose parent who was exposed to betamethasone or placebo and lived in more deprived (NZDep deciles 8-10) compared with less deprived (NZDep deciles 1–7) at 50-year follow-up

aRelative risk (RR), bmean difference (MD), codds ratio (OR), adjusted for clustering and age category of participants (<16 years or≥16 years) in the minimally adjusted model, also including an interaction term between the age category of participants and treatment given in the adjusted model, drate ratio
CI – confidence interval, BMI – body mass index, SD – standard deviation, LQ – lower quartile, UQ – upper quartile.
¶ Fully saturated model not reported as interaction term not statistically significant (p > 0.05).
§ Clustering term removed to get model to converge.
The sensitivity analysis that included data from the B4SC for participants aged between 4 and 6 years whose weight was missing in the questionnaire showed no evidence of a difference in outcomes between those whose study parent was exposed to betamethasone compared with placebo (Table 7).
Sensitivity analysis – primary and key secondary outcome in participants whose parent was exposed to betamethasone or placebo, including data from the B4SC for participants whose age at assessment was <= 6 years and for whom data was missing in the questionnaire

aRelative risk (RR), bmean difference (MD), adjusted for clustering and age category of participants (<16 years or≥16 years)
CI – confidence interval, BMI – body mass index, SD – standard deviation.
¶ Fully saturated model not reported as interaction term not statistically significant (p > 0.05).
Discussion
We aimed to assess the effects of parental antenatal corticosteroid exposure on the health and wellbeing of the second generation. We found no evidence of differences between participants with a parent exposed to betamethasone compared with placebo for any primary, secondary or tertiary outcomes. This is reassuring for families and healthcare providers. Alongside the evidence showing no long-term clinically significant adverse effects in F 1 participants, Reference McGoldrick, Stewart, Parker and Dalziel4,Reference Walters, Gamble and Crowther10–Reference St Clair, Walters and Crowther12 our findings suggest intergenerational safety of antenatal corticosteroids in current clinical use.
Many babies are exposed to antenatal corticosteroids. Globally, the incidence of preterm birth is 10% Reference Ohuma, Moller and Bradley44 and in Aotearoa New Zealand it is 7%. 45 Since 2009, rates of preterm birth in New Zealand have remained between 6–8%. 45 Because preterm birth is difficult to predict, some babies exposed to antenatal corticosteroids go on to be born near or at term, at low risk of respiratory distress syndrome. A Finnish birth registry study reported 44% of women who received antenatal corticosteroids had their babies at term Reference Rodriguez, Wang, Ali Khan, Cartwright, Gissler and Järvelin46 and a Canadian population-based study reported that 52% of women administered antenatal corticosteroids gave birth at 35 weeks’ gestation or beyond. Reference Razaz, Skoll, Fahey, Allen and Joseph47 These babies do not experience the benefits of antenatal corticosteroids, but they and their children would be affected by any long term adverse effects. Therefore, it is reassuring that we found no evidence of adverse effects of antenatal corticosteroid exposure in the second generation.
We saw some significant effects in subgroup analyses. However, these should be interpreted with caution, due to small numbers in each subgroup. To draw more robust conclusions regarding these findings, larger studies would be required that are adequately powered to examine differences between groups. These potential differences in outcomes depending on parental sex may be due to differing effects of antenatal corticosteroids on F1 participants of each sex, or differences in transmission of effects to offspring between sexes.
Altered immunity may explain increased rates of any ambulatory hospital presentations or admissions in those with a mother exposed to betamethasone compared with placebo. Antenatal corticosteroids increase glucocorticoid levels in the fetus, known to have immunosuppressive effects. Reference Solano, Holmes, Mittelstadt, Chapman and Tolosa48 No other animal or human studies have reported the effect of antenatal corticosteroids on immune function or hospitalisation rates in the second generation. One randomised controlled trial (n = 84) found that children exposed to antenatal betamethasone compared with placebo had increased risk of parent-reported hospital admissions for infectious disease and lung infections in the first 8–10 years. Reference Smolders-de Haas, Neuvel, Schmand, pE, Koppe and Hoeks49 There were no differences between groups observed in the two years before the study, so investigators suggested the difference was largely due to differential rates in early childhood. However, the very limited evidence that betamethasone exposure increases F 1 infectious disease rates in later life increases the likelihood that our finding is due to type I error. In this study, we made 173 outcome comparisons (2.3% statistically significant, p < 0.05), increasing the likelihood of type 1 error.
However, several animal studies have demonstrated differential effects on F 2 offspring according to the sex of the exposed parent. Two rat studies have demonstrated birthweight findings with the same direction of effect as our study. In rats, both maternal and paternal antenatal dexamethasone exposure led to significantly decreased birthweight, but the effect was greater with paternal dexamethasone exposure. Reference Drake, Walker and Seckl13,Reference Drake, Liu, Kerrigan, Meehan and Seckl50 This may be due to a greater reduction in hepatic insulin like growth factor 2 mRNA expression with paternal exposure compared with maternal exposure. Reference Drake, Liu, Kerrigan, Meehan and Seckl50 In rats, maternal dexamethasone exposure also led to a greater increase in phosphoenolpyruvate carboxykinase activity than paternal exposure, which may result in hyperinsulinemia and hyperglycaemia. Reference Drake, Walker and Seckl13 In guinea pigs, the HPA response to stress reduced with maternal betamethasone exposure and increased with paternal betamethasone exposure, but findings were not always consistent. Reference Moisiadis, Constantinof, Kostaki, Szyf and Matthews18
Animal studies have suggested that effects of excess glucocorticoid exposure may be transmitted to the second generation through epigenetic changes, including DNA methylation and histone modification or small non-coding RNA. Reference Drake, Liu, Kerrigan, Meehan and Seckl50,Reference Crudo, Petropoulos and Moisiadis51 Epigenetic changes in utero may be transmitted to offspring since they affect germ cells. Drake et al. Reference Drake, Liu, Kerrigan, Meehan and Seckl50 also proposed that excess glucocorticoid exposure in utero may alter maternal physiology, affecting fetal development directly.
Participants with a study parent born at term exposed to betamethasone were more likely to have any ambulatory hospital presentations or admissions compared with those with a term parent exposed to placebo. This may be due to the immunosuppressive effects of antenatal corticosteroids discussed above; however, this does not explain why no difference was seen between exposure groups in those whose study parent was born preterm. Data were not collected on the gestational age at birth of the non-study parent, so it was not possible to further explore possible effects of both parents born preterm.
Those with a study parent born at term exposed to betamethasone were more likely to be born at term rather than post-term compared with those with a term parent exposed to placebo, with no difference seen between exposure groups in those whose study parent was born preterm. Since term babies have generally better health outcomes than post-term babies, including lower rates of neonatal intensive care unit admission, respiratory disease and infectious disease, Reference Linder, Hiersch and Fridman52 this would be unlikely to be of clinical concern. In the wider study of the F 1 generation from which our cohort was derived (n = 415), findings were similar. Reference St Clair, Walters and Crowther12 Those with a parent born at term exposed to betamethasone were born at an earlier gestational age compared with those with a parent born at term exposed to placebo. In contrast, those with a parent born preterm exposed to betamethasone were born at a later gestational age compared with those with a parent born preterm exposed to placebo. Because these differences were small (0.8–1.3 weeks), this is unlikely to be clinically significant.
A previous follow-up of a subset of the AST F 2 cohort at 5–10 years of age is the only other human study assessing effects of antenatal corticosteroids on the second generation. Participants with a parent exposed to betamethasone had increased fasting glucose concentrations, fasting insulin concentrations and acute insulin release compared with participants with a parent exposed to placebo, but no other outcomes were assessed. We were unable to test these parameters in our study, as no in person assessment was conducted. We found no cases of diabetes mellitus or gestational diabetes and only two cases of prediabetes in the exposure group, meaning we were unable to calculate a risk estimate for the diabetes composite outcome. Without longer follow-up and in person assessment, we cannot conclude whether the previous finding of reduced insulin sensitivity has a clinically important effect in later life.
This is the first human follow-up study assessing the effects of parental betamethasone exposure into adolescence and adulthood. It includes social and educational outcomes as well as health outcomes to provide a more comprehensive overview of participant wellbeing. Another strength of this study is that it included both questionnaires and linked data, allowing a wider range of questions to be answered and increasing case detection. Reference Shahbaz, Harding and Milne53
This project had a limited sample size, as to be eligible, participants had to have a grandmother involved in the original AST and parents who shared their details or consented to their involvement. This led to wide confidence intervals for some outcomes and limited power to detect small differences between groups. However, it is unlikely that small differences between groups in the second generation would alter clinical decision-making around use of antenatal corticosteroids.
Because all participants were under 35 years, BMI was used as the primary outcome as a risk factor for cardiometabolic disease. If in-person assessment was conducted, measurements that better predict cardiometabolic risk could have been used, such as waist and hip circumference. Reference Cameron, Magliano and Söderberg54,Reference Yusuf, Hawken and ⓞunpuu55 We also may have obtained data for more participants, as only 68% of participants reported both weight and height. However, using questionnaires and linked data in this study did mean there were no geographical constraints on participation.
Despite these limitations, these data provide the first evidence of intergenerational safety of antenatal corticosteroid exposure in a cohort of adolescents and adults.
Acknowledgements
We thank the families who participated in the original trial and this follow-up study who made this work possible. We acknowledge the seminal contributions of the original trialists, the late Ross Howie and the late Mont Liggins, who generously made available the original data and documentation from the AST. We also thank Trisha Meagher-Lundberg for study coordination and Coila Bevan for participant tracing.
Financial support
The 50-year follow-up study was funded by grant 19/690 from the Health Research Council of New Zealand. AW and RM were funded through the Aotearoa Foundation (9909494). LL was funded by the University of Auckland Postgraduate Honours Scholarship. The work was also supported through the Auckland Medical Research Foundation (1 421 003, AW), Cure Kids New Zealand (SD), and the Health Research Council of New Zealand (19/690, CC, GG, CM, BM, JH).
Competing interests
None.
Ethical standard
The authors assert that all procedures contributing to this work comply with the ethical standards of the relevant national guidelines on human experimentation (National Ethical Standards) and with the Helsinki Declaration of 1975, as revised in 2008, and has been approved by the Health and Disability Ethics Committee.