Children and adolescents with attention–deficit hyperactivity disorder (ADHD) have more frequent contact with child welfare services (CWS) than their peers. Reference Chaulagain, Widding-Havneraas, Lyhmann, Halmøy, Bjelland and Mykletun1–Reference Klein, Damiani-Taraba, Koster, Campbell and Scholz3 This may be due to disruptive behaviour, challenges in academic and social settings, and increased family stress. They are also more often raised in environments characterised by familial conflict, marital breakdown or single-parent households, all factors associated with adverse family dynamics. Reference Chaulagain, Lyhmann, Halmøy, Widding-Havneraas, Nyttingnes and Bjelland4,Reference Huang, Xu, Au, Xu and Wu5 Consequently, children and adolescents with ADHD may be more likely to require CWS support, which provides care, and protection to children and families in vulnerable or at-risk situations. Reference Skivenes, Sørsdal, Falch-Eriksen and Backe-Hansen6
In Norway, CWS are responsible for investigating, providing supportive interventions or out-of-home placements and following up on children and adolescents placed in out-of-home care. By law, local CWS are present in all Norwegian municipalities. Reasons for CWS involvement include child abuse and neglect, children’s psychosocial or behavioural problems and parents’ insufficient caregiving capacity. 7 To ensure comprehensive care, support and development, CWS collaborate closely with health, education and other social sectors. 7 CWS in Norway emphasise supportive interventions at home with the goal of family preservation, similar to other Nordic countries, Germany and the UK. This contrasts somewhat with CWS in other countries, such as the US, Poland, Romania and the Czech Republic, where CWS are more risk-oriented, focus on children’s safety and have a high threshold for interventions that restrict parental rights. Reference Helland8
For over 70 years, pharmacological intervention has been the predominant approach to ADHD management. Randomised controlled trials (RCT) show that pharmacological treatment can reduce short-term ADHD symptoms, Reference Chaulagain, Lyhmann, Halmøy, Widding-Havneraas, Nyttingnes and Bjelland4,Reference Faraone, Bellgrove, Brikell, Cortese, Hartman and Hollis9 improve communication between children and parents, reduce parental stress and foster healthier family dynamics. Reference Chou, Wang, Lin, Liang, Chen and Hou10 Collectively, these improvements may reduce the need for contact with CWS among children and adolescents with ADHD.
To our knowledge, only one previous study, from Denmark, has examined the causal effect of ADHD medication on foster care caseloads. It found that pharmacological treatment for ADHD accounted for a substantial share of the decline in foster care caseloads. Reference Fallesen and Wildeman11 However, no study to date has provided causal evidence on the broader effects of ADHD medication on overall CWS involvement, including separate analyses of supportive interventions and out-of-home placements.
Due to limited causal evidence, this study examines the effects of pharmacological treatment of ADHD on contact with CWS using a quasi-experimental design. We exploit plausibly random variation in providers’ prescribing preferences as an instrumental variable. 7 While Norway’s universal healthcare system and national clinical guidelines aim to standardise healthcare, geographical disparities in ADHD diagnostics and treatment practices persist, even after accounting for symptom severity. Reference Mykletun, Widding-Havneraas, Chaulagain, Lyhmann, Bjelland and Halmøy12,Reference Widding-Havneraas, Markussen, Elwert, Lyhmann, Bjelland and Halmøy13 This real-life variation can be used as an instrumental variable to assess the treatment effect for patients on the margin of treatment, i.e. their need for medication is subject to professional ambivalence due to e.g. less severe or atypical symptom presentation. Reference Mykletun, Widding-Havneraas, Chaulagain, Lyhmann, Bjelland and Halmøy12
We hypothesised that pharmacological treatment of ADHD reduces contact with CWS among children and adolescents on the margin of treatment by reducing behavioural and emotional challenges common in ADHD, thus enabling families to cope without the involvement of CWS.
Method
Study population
We defined the ADHD cohort as individuals aged between 5 and 14 years in 2009–2011 who received an ADHD diagnosis (ICD 10th edition: F90.0, F90.1, F90.8 and F90.9) in Child and Adolescent Mental Health Service (CAMHS) during this period (n = 5930) as registered in the Norwegian Patient Registry. 14 Children under 5 years of age were excluded as it is not common in Norway to diagnose ADHD and provide pharmacological treatment to younger children. Children and adolescents can receive CWS until 18 years of age. Since we follow children up to 4 years from diagnosis, we restricted our sample to age 5 to 14 when diagnosed with ADHD. For descriptive comparisons, we also included a random sample of children and adolescents aged 5 to 14 from the general population with no CAMHS contact between 2009 and 2011 (n = 53 196). While the comparison sample does not inform our main aim, we included this sample to compare the rate of contact with CWS among children and adolescents diagnosed with ADHD and the general population.
Outcome
CWS contacts were obtained from comprehensive population-wide registry data maintained by Statistics Norway including individual-level information from the municipal CWS. 15 We constructed four binary outcome indicators at 1, 2, 3 and 4 years post-diagnosis, taking the value one if any CWS contact was registered within that period and zero otherwise. The indicators were constructed separately for the following three measures: Supportive interventions included measures to enhance parenting skills, measures to enhance child development, supervision and control, networking and cooperation with other services, assessment and treatment from other institutions or housing (excluding out-of-home placement). Out-of-home placement indicated that the children were placed in a foster home or child welfare institution. Any contact with CWS indicated that the children received supportive intervention or out-of-home placement or other CWS measures related to either category.
Pharmacological treatment of ADHD
Pharmacological treatment included information about filled prescriptions from the Norwegian Prescription Database for ADHD medication. 16 Medication includes methylphenidate (87.38% of total prescriptions), atomoxetine (9.88%), lisdexamphetamine (2.17%), dexamphetamine (0.35%) and other amphetamines (0.07%). Pharmacological treatment was measured as the total number of daily defined doses (DDD) filled for any ADHD medication following diagnosis. We constructed four treatment variables for DDD filled across 1, 2, 3 and 4 years post-diagnosis. We scaled each treatment variable so that a one-unit increase corresponded to a change from no treatment to full-time pharmacological treatment throughout the respective follow-up period (e.g. 365 DDD for the 1-year variable and 730 DDD for the 2-year variable). Thus, to estimate the effects of medication on CWS contacts by 1 year post-diagnosis, we used medication filled during that first year. To estimate the effects of medication on CWS contacts by 2 years post-diagnosis, we used medication filled during the first 2 years post-diagnosis, and likewise for the 3- and 4-year analyses.
Covariates
Patient covariates included age, gender, comorbidities at the time of receiving an ADHD diagnosis, country of birth (Norway, Europe or outside of Europe), year of clinic contact and prior contact with CWS, all measured at baseline. Parental covariates included income, educational level (primary school, high school, short- or long-term university), and marital status (married, unmarried, widowed, divorced or separated) when the child was 6 years old. Catchment area characteristics included population size, school drop-out rates and aggregated data on youth immigrant proportions, parental income, educational level and maternal marriage rates, measured for 2009–2011. Data sources for covariates are presented in Supplementary Table S1 available at https://doi.org/10.1192/bjp.2026.10554.
Statistical analyses
We used linear probability models (LPM) to estimate associations between pharmacological treatment of ADHD and contact with CWS. All analyses were adjusted for the covariates mentioned above. To account for potential unobserved confounding that could bias causal inferences in conventional regression models, we used instrumental variable models estimated via two-stage least squares (2SLS), including the same covariates. This instrumental variable approach leveraged variation in provider prescribing preference Reference Widding-Havneraas, Chaulagain, Lyhmann, Zachrisson, Elwert and Markussen17 across clinics, an approach used in previous studies examining the effect of medication on criminal charges, Reference Widding-Havneraas, Zachrisson, Markussen, Elwert, Lyhmann and Chaulagain18 injuries Reference Widding-Havneraas, Elwert, Markussen, Zachrisson, Lyhmann and Chaulagain19 and later comorbidities. Reference Lyhmann, Widding-Havneraas, Bjelland, Markussen, Elwert and Chaulagain20 Provider preference was defined as the clinic-specific average number of DDDs of ADHD medication filled by patients with ADHD and measured over the follow-up period.
A 4-year time frame was chosen as the instrumental variable was sufficiently strong only during this period. We used a leave-one-out mean to ensure that an individual patient’s own treatment did not influence their clinic’s average prescribing rate. The targeted parameter was the local average treatment effect (LATE), representing the causal effect of medication for patients whose treatment decisions were affected by the instrument – i.e. patients ‘on the margin’ of receiving medication.
The validity of the instrumental variable approach relies on meeting several key assumptions:
Relevance: The instrumental variable, i.e. provider preference, must strongly predict patient’s treatment status. Exclusion restriction: Provider preference must affect the outcome (i.e. contact with CWS) only through its influence on the patient’s receipt of ADHD medication (i.e. no direct effects of the IV on the outcome). Independence: Provider preference should be as good as randomly assigned, meaning it is not correlated with unobserved factors affecting the outcome after controlling for observed factors. Monotonicity: Provider preference must influence treatment in the same direction for all patients, meaning a higher provider preference should increase the likelihood of receiving ADHD medication for all patients.
We adjusted s.e. for clustering at the clinic level in all models. We report LPM and instrumental variable point estimates with 95% confidence intervals. As robustness analyses, we conducted probit models. All analyses were performed in Stata 17 for Windows 10 (StataCorp LLC, https://www.stata.com).
Ethical approval
The study is approved by the Regional Committee for Medical and Health Research Ethics (REC), South East, Committee D, Norway (REC number 2017/2150/REC south-east D). The study has been evaluated by REC to be ‘of considerable importance to society’ and to ‘protect the integrity and welfare’ of the participants. Based on the fulfilment of these criteria, the project has been exempted from the requirement of collecting informed consent from participants. The research was conducted according to the guidelines of the Norwegian National Research Ethics Committee for Medical and Health Research. The guidelines are based on international conventions such as the Declaration of Helsinki.
Results
Descriptive statistics
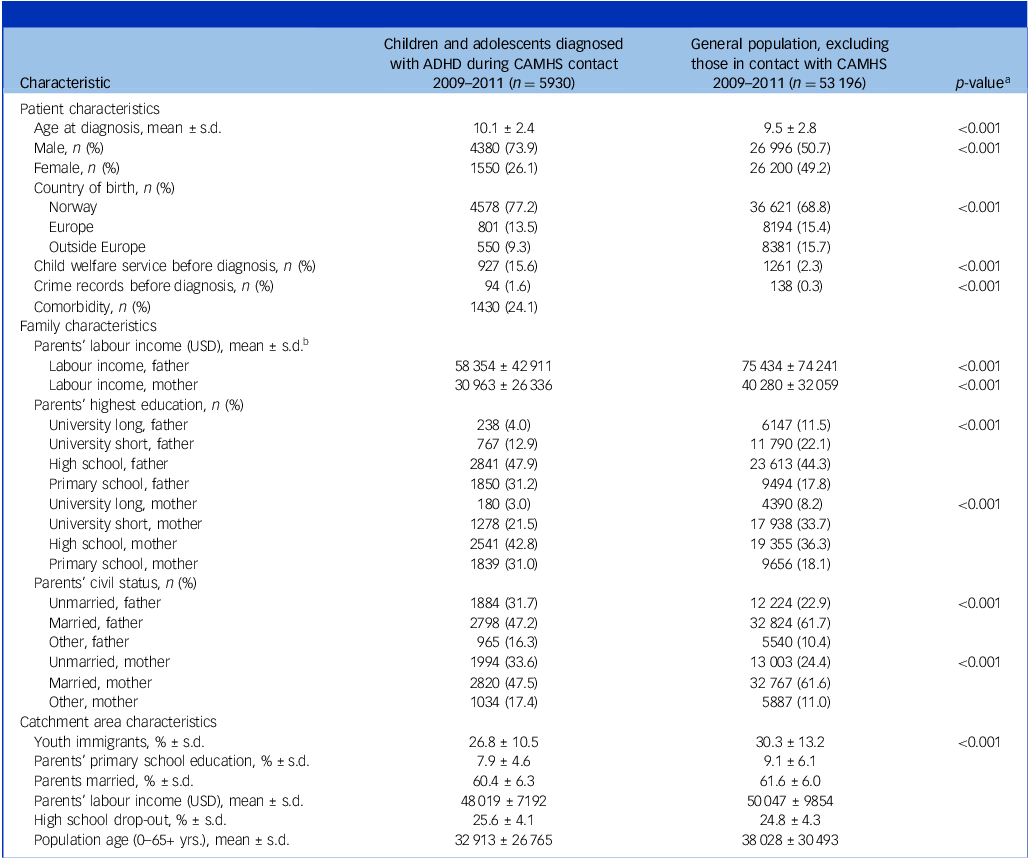
Table 1 shows baseline characteristics of children and adolescents with ADHD and the general population. Children and adolescents with ADHD differed significantly from the general population across a range of individual, family and clinic catchment area characteristics; the ADHD patient sample consisted of a higher proportion of males, were more often born in Norway and had more contacts with CWS before diagnosis. Parents of ADHD patients had lower levels of education and income and were less likely to be married. The share of patients who filled ≥1 prescription for ADHD medication was 77% during the first year post-diagnosis, 84% over the first 2 years post-diagnosis, 86% over 3 years post-diagnosis and 87% over 4 years post-diagnosis. The average percentage of patients who had filled ≥1 ADHD prescription varied from 43 to 100% between clinics by the 4-year follow-up.
Baseline characteristics for patients with attention–deficit hyperactivity disorder (ADHD), aged 5 to 14 in 2009–2011 (n = 5930)

CAMHS, Child and Adolescent Mental Health Services.
a P-value from Pearson’s chi-squared test or independent sample t-test.
b USD/NOK exchange rate average for 2010 (USD 1/NOK 6.0453).
Figure 1 shows the rate of contact with CWS among children and adolescents with ADHD compared with the general population. Over a 4-year follow-up period, children and adolescents with ADHD had significantly higher rates of contact with any CWS (22.7%) compared with the general population (4.1%). This pattern was consistent across both males and females, as well as across CWS categories, including supportive interventions and out-of-home placement.
Contact with child welfare service within 4 years of baseline, attention–deficit hyperactivity disorder (ADHD) versus general population.

Association between pharmacological treatment of ADHD and contact with CWS
Figure 2 presents estimated associations between pharmacological treatment and the probability of any contact with CWS, receiving any supportive interventions and out-of-home placement using LPMs for 1–4 years follow-up after ADHD diagnosis for all patients. All point estimates were substantively small (less than 2% difference in outcomes between patients who did versus did not receive the pharmacological treatment), and all but one coefficient failed to reach statistical significance. We conclude that there was no strong evidence of associations between pharmacological treatment and any contact with CWS or out-of-home placement and only weak evidence for receiving a supportive intervention, limited to year 2 in these LPMs, net of observed covariates. Stratified analysis by gender showed similar results (Supplementary Figure S4).
Associations between attention–deficit hyperactivity disorder (ADHD) medication and contact with child welfare services (CWS) from linear probability models net of covariates. Patients with ADHD diagnosis in Norway 2009–2011 aged 5 to 14 at time of diagnosis. Coefficient plots for regressions with 95% confidence intervals from linear probability models adjusted for patient mix.

Causal effect of pharmacological treatment of ADHD on contact with CWS
Figure 3 presents the estimate of LATEs from 2SLS instrumental variables models for all patients. For our assessment of the instrumental variable assumptions, see Supplementary Material (S1–S3). The instrumental variable results demonstrate that pharmacological treatment reduced the probability of receiving a supportive intervention at 1- and 2-year follow-up, and of out-of-home-placement at 2-years follow-up. For patients on the margin of treatment, pharmacological treatment reduced contact with CWS by 6.75% (95% CI: −13.04, −0.47) at 1-year follow-up. This corresponds to a number-needed-to-treat (NNT) estimate of 15 indicating that treatment intensity would have to be increased from 0 to 1 years DDDs for 15 patients to avoid one patient having any contact with CWS. At a 2-year follow-up, pharmacological treatment reduced the probability of CWS contact by 12.3% (95% CI: −13.04, −0.47), with an NNT of 8. Similarly, pharmacological treatment reduced the contact with supportive intervention by 11.9% (95% CI: −20.12, −3.80) at 2-year follow-up and out-home-placement by 3.30% (95% CI: −6.44,−0.15). This corresponds to NNT estimates of 8 and 30, respectively. We found no statistically significant evidence that pharmacological treatment of ADHD reduced contact with CWS over 3 or 4 years post-diagnosis but point estimates for these durations were less precise. Estimated LATEs from instrumental variable probit models showed similar effect estimates (Supplementary Figure S5). The stratification analysis by gender showed similar effect estimates for males but imprecise estimates for females (Supplementary Figure S6).
Effect estimates of attention–deficit hyperactivity disorder (ADHD) medication on contact with child welfare services (CWS) from instrumental variable analyses. Patients with ADHD diagnosis in Norway 2009–2011 aged 5 to 14 at time of diagnosis. Coefficient plots for regressions with 95% confidence intervals. Two stage least squares estimates adjusted for patient mix.

Discussion
This study aimed to estimate the causal effect of ADHD medication on contact with CWS among children and adolescents diagnosed with ADHD in Norway, utilising provider preference as an instrumental variable to address unmeasured confounding. We found evidence for a causal effect of pharmacological treatment of ADHD in reducing CWS contact among children and adolescents at the margin of treatment. Specifically, we found a reducing effect on receiving any CWS and supportive intervention in the first and second years and out-of-home placement across 2 years of follow-up.
The observed effect of ADHD medication reducing CWS involvement may be explained by improvements in core ADHD symptoms, such as impulsivity, inattention and hyperactivity, known to contribute to challenging family dynamics. Reductions in these symptoms can alleviate family stress, mitigate conflict and improve children’s relationships with peers and adults. As a result, children on medication may require fewer supportive interventions and have lower risk of major disruptive behavioural patterns that could lead to out-of-home placements. The effect may also operate through reduced delinquent behaviour, which may also be a pathway to CWS involvement. We have previously shown that medication reduces crime rates, Reference Widding-Havneraas, Zachrisson, Markussen, Elwert, Lyhmann and Chaulagain18 supporting this potential mechanism for medication’s impact on reducing CWS contact.
The reduction of CWS involvement observed during the first 2 years likely reflects an initial period of symptom stabilisation, high adherence and active engagement with treatment when symptom management is most critical. Reference Kamimura-Nishimura, Brinkman and Froehlich21,Reference Ahmed and Aslani22 The decline in the estimated effect of medication in the third and fourth years, might be due to factors such as reduced adherence, Reference Lillemoen, Kjosavik, Hunskår and Ruths23,Reference Brikell, Yao, Li, Astrup, Gao and Gillies24 developmental changes and shifts in family or treatment focus, which could have an impact on the observed benefits of medication over time. Reference Kamimura-Nishimura, Brinkman and Froehlich21 Our conventional LPM analysis found no statistically significant association between ADHD medication and subsequent involvement with CWS after adjusting for covariates. By contrast, the instrumental variable approach used in this study showed some positive effects of pharmacological treatment. This difference suggests that unobserved confounding, for example, related to family or provider-related characteristics, measures of symptom load and nonpharmacological treatment, may have biased the LPM results upwardly toward zero. Reference Fernainy, Cohen, Murray, Losina, Lamontagne and Sourial25 Children with more severe ADHD symptoms are more likely to both receive medication and be in contact with CWS. This leads to a positive confounding, which can mask the true contact-reducing effect of ADHD medication. The instrumental variable approach used the natural variation in providers’ prescription preference to reduce bias and estimate causal effects for children and adolescents on the margin of treatment. This finding highlights the potential of ADHD medication to reduce the need for CWS contact for this specific subgroup. Importantly, the instrumental variable approach estimates the LATE, which pertains to patients whose treatment status is influenced by the instrument and who may differ substantially from the overall treated population. The most plausible reason for variation in medication use is that these children have less severe or less clear-cut ADHD symptoms e.g. symptoms being over-shadowed by comorbidities or affected by an adverse psychosocial environment. As a result, the estimated effect applies specifically to this subgroup rather than to all individuals receiving ADHD medication.
Our empirical finding that pharmacological treatment of ADHD reduces contact with CWS does not in itself inform the desirability of pharmacological treatment over contact with CWS, or vice versa. Supportive CWS interventions may help children and adolescents with ADHD and their parents, for example by building skills and coping strategies. At the same time, CWS contact among children and adolescents with ADHD and their families may signal underlying difficulties related to behavioural challenges, unmet family needs or disruptions in school and home life. Children with ADHD may exhibit behaviours that strain family relationships, challenge school environments or lead to unsafe situations. Pharmacological treatment of ADHD to manage symptoms might reduce the escalation of problems that would otherwise require formal intervention. Therefore, the estimated reduction in contact with CWS in response to pharmacological treatment may reflect a reduction in severe behavioural symptoms and improved emotional regulation, which in turn may ease family stress and reduce the likelihood of contact with CWS. Our findings about the reducing effect of ADHD medication on contact with CWS align with the previous study from Denmark. Reference Fallesen and Wildeman11 However, differences in methodology and outcome prevent a direct comparison of estimates.
Strengths and limitations
This study has several methodological and contextual strengths. Norway’s universal, publicly funded healthcare system ensures equitable access to ADHD diagnosis and treatment, minimising selection biases based on treatment accessibility. Furthermore, patients are assigned to clinics based on residential location, limiting patient and caregiver choice in selecting providers. Norway’s limited private healthcare sector strengthens the validity of using provider preference as an instrumental variable since provider assignment and variation are largely independent of patient characteristics. This design supports causal inference, particularly for patients on the margin of the treatment, where the estimated LATE offers valuable insights for clinical decision-making for this less clear-cut patient group. Reference Widding-Havneraas, Zachrisson, Markussen, Elwert, Lyhmann and Chaulagain18,Reference Stein, Sibley and Newcorn26,Reference Arnstein, Tarjei, Ashmita, Ingvild, Ingvar and Anne27
The linkage of several Norwegian registries in this study provides comprehensive, population-level information, including ADHD diagnoses, medication prescriptions and CWS interventions. These extensive data minimise selection bias, offer reliable descriptive statistics and support the representativeness of our findings. Additionally, the longitudinal data allowed us to assess the causal effect of ADHD medication effects over time, offering insights into how treatment may influence the need for CWS in a real-world setting. We adjusted for a wide range of confounding variables at individual and community levels, including demographics, comorbidities and socioeconomic indicators, which helped strengthen the validity of our findings. By employing both conventional regression analysis and the IV approach, we addressed potential biases and assessed the robustness of our results. While the LPM did not reveal significant associations, the instrumental variable model, which corrects for unobserved confounding, identified a reducing effect of ADHD medication on contact with CWS. This approach highlights the importance of causal inference methods in observational research and underscores the importance of addressing endogeneity in estimating medication effects. Reference Sajons28
The potential limitations of this study merit consideration. While instrumental variable analysis can help address confounding by indication, it relies on strong assumptions. We have attempted to justify these assumptions based on prior literature and empirical evidence, but we cannot exclude the possibility of violations. Reference Widding-Havneraas and Zachrisson29 For instance, our estimates may be biased by unmeasured common causes of provider preference and contact with CWS. The assumption that provider preferences are effectively random might be affected by residual confounding Reference Widding-Havneraas and Zachrisson29 if providers inclined to prescribe medication are also more likely to recommend non-pharmacological treatments, such as behavioural therapies. Reference Stein, Sibley and Newcorn26 We lack data to measure this overlap, which complicates isolating the effect of medication. Reference Widding-Havneraas, Zachrisson, Markussen, Elwert, Lyhmann and Chaulagain18 However, Norwegian national guidelines recommend that non-pharmacological treatment should be tried before initiating medication in children and adolescents with ADHD. 30 Hence, our finding still reflects the real-world effect of pharmacological treatment within this clinical context.
Although filled prescriptions serve as a common proxy for adherence in registry studies, they do not capture patterns of treatment discontinuation or dosage adjustments, which may have an impact on treatment effectiveness over time, especially for adolescents, whose adherence often fluctuates. Reference Tibble, Sheikh and Tsanas31,Reference Lam and Fresco32
Furthermore, ADHD diagnostic and treatment standards have evolved, potentially affecting how providers interpret and apply these criteria in practice, especially for patients at the margin of treatment. This evolution introduces variability in medication assignment across studies conducted in different periods, challenging comparisons across different countries and affecting generalisability as diagnostic standards continue to adapt. Similarly, variation in interventions across countries and time also hampers the generalisability.
Additionally, as is common with instrumental variable analyses, our approach results in wider confidence intervals, reducing statistical precision. Reference Widding-Havneraas and Zachrisson29 Our findings that pharmacological treatment reduced the probability of out-of-home placement at 2 years warrant some caution in interpretation as no statistically significant evidence was observed in the other follow-up years. The instrumental variable model estimates the LATE for patients at the margin of treatment. This underscores the importance of carefully interpreting instrumental variable results, as they provide insights into the causal relationship for specific subgroups of patients, and the estimate cannot be generalised to all children and adolescents with higher symptom loads of ADHD.
Implications
These findings may be interpreted differently depending on one’s a priori position in the debate over ADHD treatment practices. Clinicians with a liberal position Reference Arnstein, Tarjei, Ashmita, Ingvild, Ingvar and Anne27,Reference Lyhmann, Widding-Havneraas, Zachrisson, Bjelland, Chaulagain and Mykletun33 may interpret these findings as supporting the use of pharmacological treatment of ADHD, to the extent that the reduction in contact with CWS is an indicator of reduction in behavioural symptoms in children and adolescents that reduces the need for CWS involvement. By contrast, clinicians following a more restrictive approach Reference Arnstein, Tarjei, Ashmita, Ingvild, Ingvar and Anne27,Reference Lyhmann, Widding-Havneraas, Zachrisson, Bjelland, Chaulagain and Mykletun33 may argue that the effect is moderate and contact with CWS is not necessarily a negative outcome to the extent that children and families are receiving necessary support as an alternative to pharmacological treatment. Most evidence on treatment effects comes from RCTs conducted under highly controlled conditions. While such studies provide strong evidence on efficacy, their findings may have limited generalisability to routine clinical practice. In contrast, our study estimates the effectiveness of ADHD medication in real-world settings. Our findings therefore complement existing efficacy evidence and are particularly relevant for clinical guidelines and policy-makers.
Children and adolescents with ADHD have higher rates of contact with CWS compared with the general population. Our instrumental variable analysis suggests that pharmacological treatment of ADHD reduces the need for both supportive and out-of-home placements among children and adolescents whose treatment is influenced by clinicians’ prescribing variations in Norway.
Supplementary material
The supplementary material is available online at https://doi.org/10.1192/bjp.2026.10554
Acknowledgement
The authors thank Henrik Daae Zachrisson for his contributions in developing the instrumental variable design used in this study.
Author contributions
Conceptualisation: A.C., T.W.-H., A.H., I.B. and A.M.; methodology: A.C., T.W.-H., S.M., F.E. and A.M.; analysis: A.C. and T.W.-H.; writing – original draft preparation: A.C.; writing – review and editing: I.L., T.W.-H., F.E., S.M., I.B., A.H. and A.M.; supervision: A.M., A.H. and I.B. Principal Investigator and funding acquisition: A.M.
Funding
A.C., T.W.-H., I.L., F.E., S.M. and A.M. are funded by Research Council Norway (288585/IAR). T.W.-H. was also funded by the Western Norway Regional Health Authority (912197). The funders have not been involved in the creation or carrying out of the study.
Declaration of interest
None.






eLetters
No eLetters have been published for this article.