Retrograde clay mineral formation on the ocean floor, also called reverse weathering, has strongly influenced the mineralogical and biogeochemical composition of Earth’s marine sediments, the chemistry of its seawater and the composition of its atmosphere across geological time (Mackenzie & Garrels, Reference Mackenzie and Garrels1966a, Reference Mackenzie and Garrels1966b; Mackin & Aller, Reference Mackin and Aller1984; Siever, Reference Siever1992; Mackenzie & Kump, Reference Mackenzie and Kump1995; Michalopoulos & Aller, Reference Michalopoulos and Aller1995; Misra & Froehlich, Reference Misra and Froehlich2012; Isson & Planavsky, Reference Isson and Planavsky2018; Li et al., Reference Li, Penman, Planavsky, Knudsen, Zhao and Wang2021a; Baldermann et al., Reference Baldermann, Banerjee, Czuppon, Dietzel, Farkaš and Löhr2022; Rauzi et al., Reference Rauzi, Foster, Takahashi, Hon, Beaty, Tarhan and Isson2024). In this process, porewater- or seawater-derived Si(OH)4 reacts with HCO3– ions, dissolved cations and metal aquo-complexes, such as Fe2+/3+, Mg2+, K+, Na+, Ca2+ and Al(OH)30, to form a suite of authigenic clay minerals, as well as variable amounts of acidity (H+) during early marine diagenesis (e.g. Siever, Reference Siever1992; Michalopoulos et al., Reference Michalopoulos, Aller and Reeder2000; Hazen et al., Reference Hazen, Sverjensky, Azzolini, Bish, Elmore, Hinnov and Milliken2013; Tosca et al., Reference Tosca, Guggenheim and Pufahl2016; Cuadros et al., Reference Cuadros, Andrade, Ferreira, de Moya Partiti, Cohen and Vidal-Torrado2017; Frings, Reference Frings2017; Cao et al., Reference Cao, Bataille, Song, Saltzman, Cramer and Wu2022; Ma et al., Reference Ma, Shi, Lechte, Zhou, Wang and Huang2022; Steiner et al., Reference Steiner, Rae, Berelson, Adkins, Hou and Dong2022; Wallmann et al., Reference Wallmann, Geilert and Scholz2023). As a result, near-surface clay mineral authigenesis impacts the evolution of seawater pH and the marine element cycles, and potentially serves as a long-term regulator of Earth’s climate by influencing atmospheric CO2 concentrations (e.g. Kump et al., Reference Kump, Brantley and Arthur2000; Isson & Planavsky, Reference Isson and Planavsky2018; Andrews et al., Reference Andrews, Pogge von Strandmann and Fantle2020; Isson et al., Reference Isson, Planavsky, Coogan, Stewart, Ague and Bolton2020; Krissansen-Totton & Catling, Reference Krissansen-Totton and Catling2020; Li et al., Reference Li, Penman, Planavsky, Knudsen, Zhao and Wang2021a; Warr, Reference Warr2022; Yin et al., Reference Yin, Wei, Pogge von Strandmann, Wei, Pogge von Strandmann Philip and Lechte2023). Reverse weathering reactions have been documented in a diverse range of marine settings, including ‘low’-temperature shallow shelf and deep-marine environments, as well as ‘high’-temperature (up to a few hundred °C) settings associated with mid-oceanic ridge sites, or transform faults. Common examples of clay mineral precipitation reactions are given in Equation 1a–e (modified after Isson & Planavsky, Reference Isson and Planavsky2018):
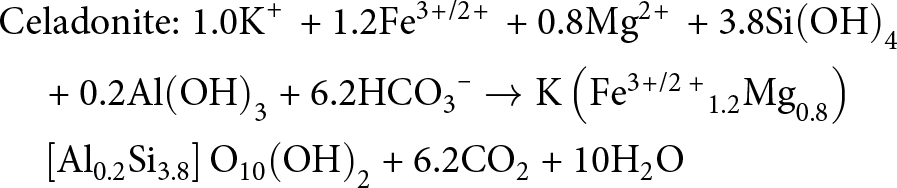 \begin{align}
&{\text{Celadonite:}}\;{\text{1}}{\text{.0}}{{\text{K}}^{\text{\! + }}}{\text{ + 1}}{\text{.2F}}{{\text{e}}^{{\text{3\! +\! /2\! + }}}}{\text{ + 0}}{\text{.8M}}{{\text{g}}^{{\text{2\! + }}}}{\text{ + 3}}{\text{.8Si}}{\left( {{\text{OH}}} \right)_{\text{4}}} \nonumber\\
&\quad {\text{ + 0}}{\text{.2Al}}{\left( {{\text{OH}}} \right)_{\text{3}}}{\text{ + 6}}{\text{.2HC}}{{\text{O}}_{\text{3}}}^{\text{--}} \to {\text{K}}\left( {{\text{F}}{{\text{e}}^{{\text{3\! +\! /2 + }}}}_{{\text\!{1}}{\text{.2}}}{\text{M}}{{\text{g}}_{{\text{0}}{\text{.8}}}}} \right) \nonumber\\
&\quad \left[ {{\text{A}}{{\text{l}}_{{\text{0}}{\text{.2}}}}{\text{S}}{{\text{i}}_{{\text{3}}{\text{.8}}}}} \right]{{\text{O}}_{{\text{10}}}}{\left( {{\text{OH}}} \right)_{\text{2}}}{\text{ + 6}}{\text{.2C}}{{\text{O}}_{\text{2}}}{\text{ + 10}}{{\text{H}}_{\text{2}}}{\text{O}}
\end{align}
\begin{align}
&{\text{Celadonite:}}\;{\text{1}}{\text{.0}}{{\text{K}}^{\text{\! + }}}{\text{ + 1}}{\text{.2F}}{{\text{e}}^{{\text{3\! +\! /2\! + }}}}{\text{ + 0}}{\text{.8M}}{{\text{g}}^{{\text{2\! + }}}}{\text{ + 3}}{\text{.8Si}}{\left( {{\text{OH}}} \right)_{\text{4}}} \nonumber\\
&\quad {\text{ + 0}}{\text{.2Al}}{\left( {{\text{OH}}} \right)_{\text{3}}}{\text{ + 6}}{\text{.2HC}}{{\text{O}}_{\text{3}}}^{\text{--}} \to {\text{K}}\left( {{\text{F}}{{\text{e}}^{{\text{3\! +\! /2 + }}}}_{{\text\!{1}}{\text{.2}}}{\text{M}}{{\text{g}}_{{\text{0}}{\text{.8}}}}} \right) \nonumber\\
&\quad \left[ {{\text{A}}{{\text{l}}_{{\text{0}}{\text{.2}}}}{\text{S}}{{\text{i}}_{{\text{3}}{\text{.8}}}}} \right]{{\text{O}}_{{\text{10}}}}{\left( {{\text{OH}}} \right)_{\text{2}}}{\text{ + 6}}{\text{.2C}}{{\text{O}}_{\text{2}}}{\text{ + 10}}{{\text{H}}_{\text{2}}}{\text{O}}
\end{align}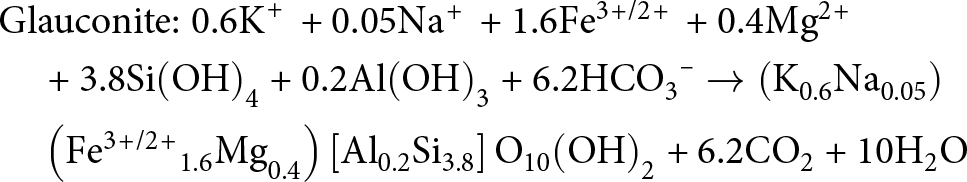 \begin{align}
&{\text{Glauconite:}}\;{\text{0}}{\text{.6}}{{\text{K}}^{\text{\! + }}}{\text{ + 0}}{\text{.05N}}{{\text{a}}^{\text{\! + }}}{\text{ + 1}}{\text{.6F}}{{\text{e}}^{{\text{3\! +\! /2\! + }}}}{\text{ + 0}}{\text{.4M}}{{\text{g}}^{{\text{2\! + }}}} \nonumber\\
&\quad {\text{ + 3}}{\text{.8Si}}{\left( {{\text{OH}}} \right)_{\text{4}}}{\text{ + 0}}{\text{.2Al}}{\left( {{\text{OH}}} \right)_{\text{3}}}{\text{ + 6}}{\text{.2HC}}{{\text{O}}_{\text{3}}}^{\text{--}} \to \left( {{{\text{K}}_{{\text{0}}{\text{.6}}}}{\text{N}}{{\text{a}}_{{\text{0}}{\text{.05}}}}} \right) \nonumber\\
&\quad \left( {{\text{F}}{{\text{e}}^{{\text{3\! +\! /2\! + }}}}_{{\!\text{1}}{\text{.6}}}{\text{M}}{{\text{g}}_{{\text{0}}{\text{.4}}}}} \right)\left[ {{\text{A}}{{\text{l}}_{{\text{0}}{\text{.2}}}}{\text{S}}{{\text{i}}_{{\text{3}}{\text{.8}}}}} \right]{{\text{O}}_{{\text{10}}}}{\left( {{\text{OH}}} \right)_{\text{2}}}{\text{ + 6}}{\text{.2C}}{{\text{O}}_{\text{2}}}{\text{ + 10}}{{\text{H}}_{\text{2}}}{\text{O}}
\end{align}
\begin{align}
&{\text{Glauconite:}}\;{\text{0}}{\text{.6}}{{\text{K}}^{\text{\! + }}}{\text{ + 0}}{\text{.05N}}{{\text{a}}^{\text{\! + }}}{\text{ + 1}}{\text{.6F}}{{\text{e}}^{{\text{3\! +\! /2\! + }}}}{\text{ + 0}}{\text{.4M}}{{\text{g}}^{{\text{2\! + }}}} \nonumber\\
&\quad {\text{ + 3}}{\text{.8Si}}{\left( {{\text{OH}}} \right)_{\text{4}}}{\text{ + 0}}{\text{.2Al}}{\left( {{\text{OH}}} \right)_{\text{3}}}{\text{ + 6}}{\text{.2HC}}{{\text{O}}_{\text{3}}}^{\text{--}} \to \left( {{{\text{K}}_{{\text{0}}{\text{.6}}}}{\text{N}}{{\text{a}}_{{\text{0}}{\text{.05}}}}} \right) \nonumber\\
&\quad \left( {{\text{F}}{{\text{e}}^{{\text{3\! +\! /2\! + }}}}_{{\!\text{1}}{\text{.6}}}{\text{M}}{{\text{g}}_{{\text{0}}{\text{.4}}}}} \right)\left[ {{\text{A}}{{\text{l}}_{{\text{0}}{\text{.2}}}}{\text{S}}{{\text{i}}_{{\text{3}}{\text{.8}}}}} \right]{{\text{O}}_{{\text{10}}}}{\left( {{\text{OH}}} \right)_{\text{2}}}{\text{ + 6}}{\text{.2C}}{{\text{O}}_{\text{2}}}{\text{ + 10}}{{\text{H}}_{\text{2}}}{\text{O}}
\end{align}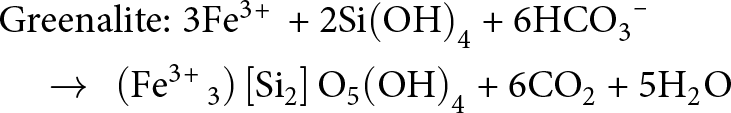 \begin{align}&{\text{Greenalite:}}\;{\text{3F}}{{\text{e}}^{{\text{3\! + }}}}{\text{ + 2Si}}{\left( {{\text{OH}}} \right)_{\text{4}}}{\text{ + 6HC}}{{\text{O}}_{\text{3}}}^{\text{--}} \nonumber\\
&\quad \to {\text{ }}\left( {{\text{F}}{{\text{e}}^{{\text{3\! + }}}}_{\text{3}}} \right)\left[ {{\text{S}}{{\text{i}}_{\text{2}}}} \right]{{\text{O}}_{\text{5}}}{\left( {{\text{OH}}} \right)_{\text{4}}}{\text{ + 6C}}{{\text{O}}_{\text{2}}}{\text{ + 5}}{{\text{H}}_{\text{2}}}{\text{O}}\end{align}
\begin{align}&{\text{Greenalite:}}\;{\text{3F}}{{\text{e}}^{{\text{3\! + }}}}{\text{ + 2Si}}{\left( {{\text{OH}}} \right)_{\text{4}}}{\text{ + 6HC}}{{\text{O}}_{\text{3}}}^{\text{--}} \nonumber\\
&\quad \to {\text{ }}\left( {{\text{F}}{{\text{e}}^{{\text{3\! + }}}}_{\text{3}}} \right)\left[ {{\text{S}}{{\text{i}}_{\text{2}}}} \right]{{\text{O}}_{\text{5}}}{\left( {{\text{OH}}} \right)_{\text{4}}}{\text{ + 6C}}{{\text{O}}_{\text{2}}}{\text{ + 5}}{{\text{H}}_{\text{2}}}{\text{O}}\end{align}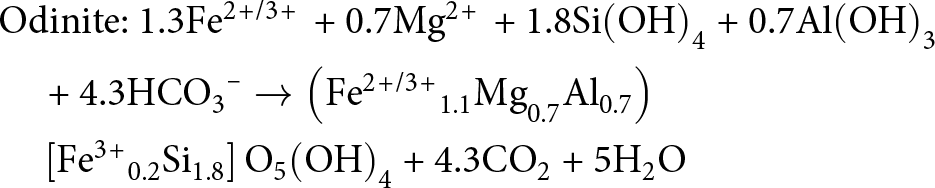 \begin{align}
&{\text{Odinite: 1}}{\text{.3F}}{{\text{e}}^{{\text{2\! +\! /3\! + }}}}{\text{ + 0}}{\text{.7M}}{{\text{g}}^{{\text{2\! + }}}}{\text{ + 1}}{\text{.8Si}}{\left( {{\text{OH}}} \right)_{\text{4}}}{\text{ + 0}}{\text{.7Al}}{\left( {{\text{OH}}} \right)_{\text{3}}} \nonumber\\
&\quad {\text{ + 4}}{\text{.3HC}}{{\text{O}}_{\text{3}}}^{\text{--}} \to \left( {{\text{F}}{{\text{e}}^{{\text{2\! +\! /3\! + }}}}_{{\!\text{1}}{\text{.1}}}{\text{M}}{{\text{g}}_{{\text{0}}{\text{.7}}}}{\text{A}}{{\text{l}}_{{\text{0}}{\text{.7}}}}} \right) \nonumber\\
&\quad \left[ {{\text{F}}{{\text{e}}^{{\text{3\! + }}}}_{{\!\text{0}}{\text{.2}}}{\text{S}}{{\text{i}}_{{\text{1}}{\text{.8}}}}} \right]{{\text{O}}_{\text{5}}}{\left( {{\text{OH}}} \right)_{\text{4}}}{\text{ + 4}}{\text{.3C}}{{\text{O}}_{\text{2}}}{\text{ + 5}}{{\text{H}}_{\text{2}}}{\text{O}}
\end{align}
\begin{align}
&{\text{Odinite: 1}}{\text{.3F}}{{\text{e}}^{{\text{2\! +\! /3\! + }}}}{\text{ + 0}}{\text{.7M}}{{\text{g}}^{{\text{2\! + }}}}{\text{ + 1}}{\text{.8Si}}{\left( {{\text{OH}}} \right)_{\text{4}}}{\text{ + 0}}{\text{.7Al}}{\left( {{\text{OH}}} \right)_{\text{3}}} \nonumber\\
&\quad {\text{ + 4}}{\text{.3HC}}{{\text{O}}_{\text{3}}}^{\text{--}} \to \left( {{\text{F}}{{\text{e}}^{{\text{2\! +\! /3\! + }}}}_{{\!\text{1}}{\text{.1}}}{\text{M}}{{\text{g}}_{{\text{0}}{\text{.7}}}}{\text{A}}{{\text{l}}_{{\text{0}}{\text{.7}}}}} \right) \nonumber\\
&\quad \left[ {{\text{F}}{{\text{e}}^{{\text{3\! + }}}}_{{\!\text{0}}{\text{.2}}}{\text{S}}{{\text{i}}_{{\text{1}}{\text{.8}}}}} \right]{{\text{O}}_{\text{5}}}{\left( {{\text{OH}}} \right)_{\text{4}}}{\text{ + 4}}{\text{.3C}}{{\text{O}}_{\text{2}}}{\text{ + 5}}{{\text{H}}_{\text{2}}}{\text{O}}
\end{align}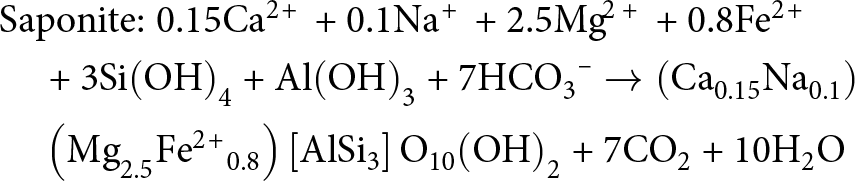 \begin{align}
&{\text{Saponite:}}\;{\text{0}}{\text{.15C}}{{\text{a}}^{{\text{2\! + }}}}{\text{ + 0}}{\text{.1N}}{{\text{a}}^{\text{\! + }}}{\text{ + 2}}{\text{.5M}}{{\text{g}}^{{\!\text{2 + }}}}{\text{ + 0}}{\text{.8F}}{{\text{e}}^{{\text{2\! + }}}} \nonumber\\
&\quad {\text{ + 3Si}}{\left( {{\text{OH}}} \right)_{\text{4}}}{\text{ + Al}}{\left( {{\text{OH}}} \right)_{\text{3}}}{\text{ + 7HC}}{{\text{O}}_{\text{3}}}^{\text{--}}
\to \left( {{\text{C}}{{\text{a}}_{{\text{0}}{\text{.15}}}}{\text{N}}{{\text{a}}_{{\text{0}}{\text{.1}}}}} \right) \nonumber\\
&\quad \left( {{\text{M}}{{\text{g}}_{{\text{2}}{\text{.5}}}}{\text{F}}{{\text{e}}^{{\text{2\! + }}}}_{{\!\text{0}}{\text{.8}}}} \right)\left[ {{\text{AlS}}{{\text{i}}_{\text{3}}}} \right]{{\text{O}}_{{\text{10}}}}{\left( {{\text{OH}}} \right)_{\text{2}}}{\text{ + 7C}}{{\text{O}}_{\text{2}}}{\text{ + 10}}{{\text{H}}_{\text{2}}}{\text{O}}
\end{align}
\begin{align}
&{\text{Saponite:}}\;{\text{0}}{\text{.15C}}{{\text{a}}^{{\text{2\! + }}}}{\text{ + 0}}{\text{.1N}}{{\text{a}}^{\text{\! + }}}{\text{ + 2}}{\text{.5M}}{{\text{g}}^{{\!\text{2 + }}}}{\text{ + 0}}{\text{.8F}}{{\text{e}}^{{\text{2\! + }}}} \nonumber\\
&\quad {\text{ + 3Si}}{\left( {{\text{OH}}} \right)_{\text{4}}}{\text{ + Al}}{\left( {{\text{OH}}} \right)_{\text{3}}}{\text{ + 7HC}}{{\text{O}}_{\text{3}}}^{\text{--}}
\to \left( {{\text{C}}{{\text{a}}_{{\text{0}}{\text{.15}}}}{\text{N}}{{\text{a}}_{{\text{0}}{\text{.1}}}}} \right) \nonumber\\
&\quad \left( {{\text{M}}{{\text{g}}_{{\text{2}}{\text{.5}}}}{\text{F}}{{\text{e}}^{{\text{2\! + }}}}_{{\!\text{0}}{\text{.8}}}} \right)\left[ {{\text{AlS}}{{\text{i}}_{\text{3}}}} \right]{{\text{O}}_{{\text{10}}}}{\left( {{\text{OH}}} \right)_{\text{2}}}{\text{ + 7C}}{{\text{O}}_{\text{2}}}{\text{ + 10}}{{\text{H}}_{\text{2}}}{\text{O}}
\end{align}Many of these authigenic marine clay minerals are Fe-rich, have a greenish colour and show a high but temporally restricted abundance in the rock record. Some of them even form distinct facies, such as the well-known glaucony and verdine facies (e.g. Rubio & López-Pérez, Reference Rubio and López-Pérez2024). These unique Fe-rich clay minerals preferentially precipitate in semi-confined geochemical micro-environments, such as in faecal pellets or foraminifera chambers, within marine sedimentary sequences that cover large areas of modern shelf and deep-sea areas and characterized equivalent ancient environments (e.g. Baldermann et al., Reference Baldermann, Grathoff and Nickel2012, Reference Baldermann, Banerjee, Czuppon, Dietzel, Farkaš and Löhr2022; Banerjee et al., Reference Banerjee, Mondal, Chakraborty and Meena2015, Reference Banerjee, Bansal and Thorat2016).
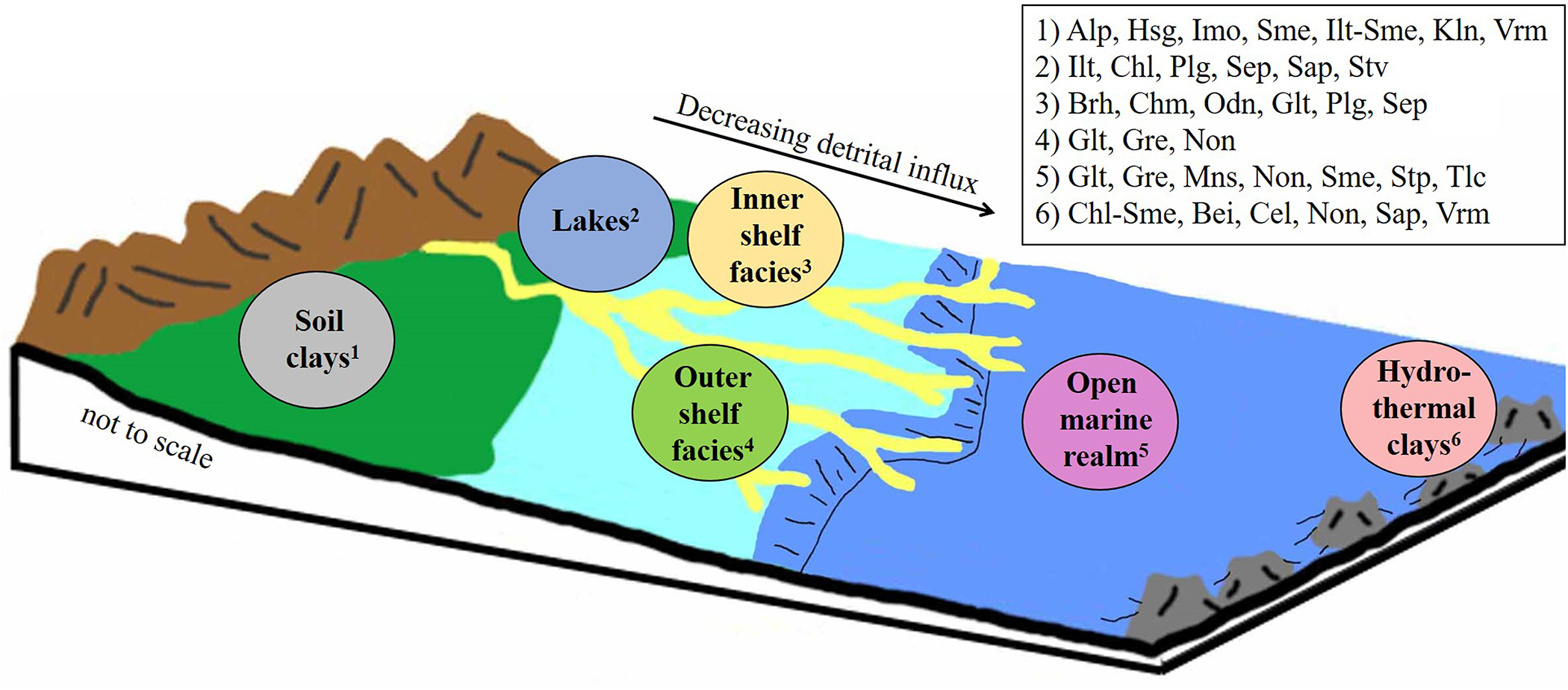
In the geological record, the occurrence of large deposits of authigenic marine clay minerals, such as ironstones and greensands, is often found to be associated with high rates of bio-opal remineralization (Michalopoulos & Aller, Reference Michalopoulos and Aller2004). This relationship suggests a close linkage between the global silica and other element cycles – importantly, C, K, Mg and Fe (e.g. Pogge von Strandmann et al., Reference Pogge von Strandmann, Jenkyns and Woodfine2013; Dunlea et al., Reference Dunlea, Murray, Santiago Ramos and Higgins2017; Baldermann et al., Reference Baldermann, Banerjee, Czuppon, Dietzel, Farkaš and Löhr2022; Isson & Rauzi, Reference Isson and Rauzi2024). However, quantifying the role of reverse weathering in global biogeochemical cycles and the climate system remains difficult because the mechanisms and rates of individual reactions are often not known (e.g. Ehlert et al., Reference Ehlert, Doering, Wallmann, Scholz, Sommer and Grasse2016; Rahman et al., Reference Rahman, Aller and Cochran2016; Krissansen-Totton & Catling, Reference Krissansen-Totton and Catling2020). Nevertheless, there is ample evidence that marine clay mineral authigenesis has had a strong effect on Earth’s climatic evolution and the marine sedimentary element cycles – relationships that are discussed in this article. Moreover, this article reviews the temporal distribution and abundance of authigenic clay minerals in marine sedimentary archives from the Precambrian to recent epochs by focusing on the formation of beidellite, berthierine, chamosite, celadonite, glauconite, greenalite, kaolinite, minnesotaite, odinite, palygorskite, saponite, sepiolite, stevensite, stilpnomelane, talc and vermiculite (Fig. 1). Recent advances in trace element and isotope geochemistry of authigenic clay minerals are also presented, and examples are provided highlighting the importance of clay mineral formation versus weathering in marine geoengineering and in slow-release fertilizer technology.
Illustration showing selected sedimentary environments characterized by authigenic clay mineral formation. Common clay mineral assemblages forming in each depocentre are indicated at the top right. The yellow meandering strands indicate deltas and subaquatic channels. International Mineralogical Association (IMA)-approved clay minerals abbreviations after Warr (Reference Warr2020): Alp = allophane; Bei = beidellite; Brh = berthierine; Cel = celadonite; Chl = chlorite; Chl-Sme = chlorite-smectite; Chm = chamosite; Glt = glauconite; Gre = greenalite; Hsg = hisingerite; Ilt = illite; Ilt-Sme = illite-smectite; Imo = imogolite; Kln = kaolinite; Mns = minnesotaite; Non = nontronite; Odn = odinite; Plg = palygorskite; Sap = saponite; Sep = sepiolite; Sme = smectite; Stp = stilpnomelane; Stv = stevensite; Tlc = talc; Vrm = vermiculite.
Reverse weathering scenarios and climate evolution across geological time
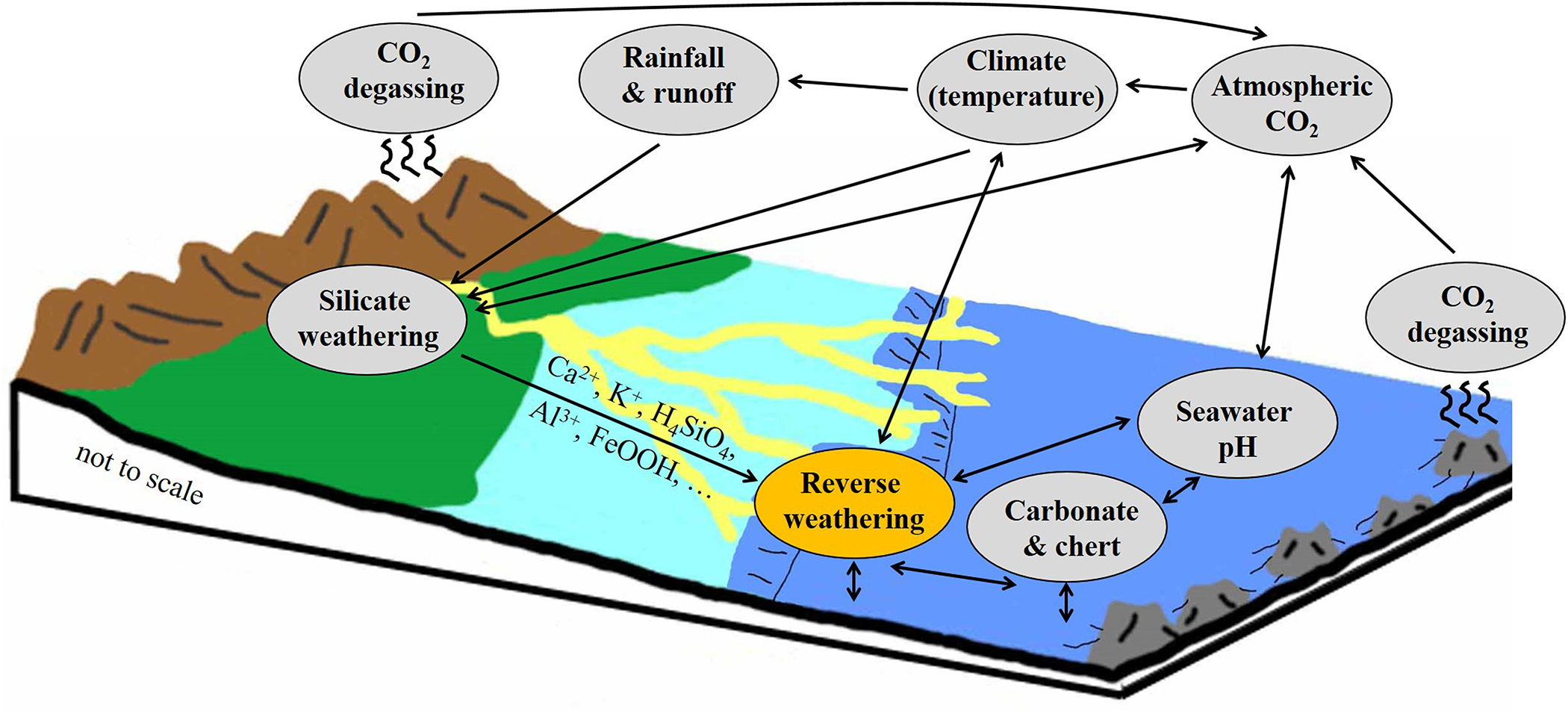
Weathering, erosion and biodegradation are the main mechanisms that cause the breakdown of rocks and minerals on Earth’s surface. These processes trigger congruent and incongruent dissolution of primary siliceous, calcareous and other minerals, providing dissolved elements, colloids, degraded minerals and solid matter to rivers, groundwaters and surface waters, which ultimately enter the oceans (Fig. 2). Thus, by regulating supply, weathering reactions on the continents play a central role in controlling ocean (bio)geochemical reactions at the sediment–seawater interface and in the marine element cycle(s) through time (e.g. Singer, Reference Singer1980, Reference Singer1984; Michalopoulos & Aller, Reference Michalopoulos and Aller1995; Isson & Planavsky, Reference Isson and Planavsky2018; Isson et al., Reference Isson, Planavsky, Coogan, Stewart, Ague and Bolton2020; Kalderon-Asal et al., Reference Kalderon-Asal, Katchinoff, Planavsky, Hood, Dellinger and Bellefroid2021).
Illustration showing the complex interplay between terrestrial (silicate) weathering, marine bioproductivity, reverse weathering reactions, ocean biogeochemistry and atmospheric circulation, which all contribute to Earth’s climatic evolution (after Isson & Planavsky, Reference Isson and Planavsky2018). The yellow meandering strands indicate deltas and subaquatic channels.
Silicate minerals compose over 90% of the rocks found in Earth’s near-surface environments. As most of them are unstable under ambient pressure and temperature conditions, they are susceptible to various forms of physical alteration and chemical degradation (e.g. Wilson, Reference Wilson1999, Reference Wilson2004). The incongruent dissolution of primary igneous silicate minerals, such as olivine, pyroxene, amphibole, feldspar and mica, and the resulting supply of cations are therefore often viewed as prerequisites for the formation of secondary clay minerals in the critical zone (i.e. soils) and in the widespread marine realm (e.g. Banfield et al., Reference Banfield, Jones and Veblen1991; Wilson, Reference Wilson1999, Reference Wilson2004; Gillis & Coogan, Reference Gillis and Coogan2011; Rafiei et al., Reference Rafiei, Löhr, Baldermann, Webster and Kong2020). These reactions are ultimately linked to regulating Earth’s climate and its carbon cycle over the long term. There are various numerical models available (reviewed by Penman et al., Reference Penman, Rugenstein, Ibarra and Winnick2020) that aim at understanding and quantifying (at least parts of) the complex feedback mechanisms and rates between silicate weathering, reverse weathering and climate change. These consider changes in parameters through geological time, such as the partial pressure of CO2 in the atmosphere (pCO2), the rates of continental and seafloor weathering and erosion (e.g. Coogan & Gillis, Reference Coogan and Gillis2013, Reference Coogan and Gillis2018, Reference Coogan and Gillis2020), vegetational cover, topography and temperature, among other factors (e.g. Penman et al., Reference Penman, Rugenstein, Ibarra and Winnick2020). The key parameters controlling climate change and the palaeoclimatic evolution of Earth remain, however, focuses of continued debate. This reflects the complexity of quantifying silicate dissolution (CO2 sink) versus clay mineral formation processes (CO2 source) occurring in Earth’s broad spectrum of near-surface environments and the resulting limitations in modelling and predicting global elemental fluxes and their sensitivity to climate (e.g. Keller & Wood, Reference Keller and Wood1993; Isson et al., Reference Isson, Planavsky, Coogan, Stewart, Ague and Bolton2020; Krissansen-Totton & Catling, Reference Krissansen-Totton and Catling2020). On geological timescales, silicate weathering and clay mineral cycles are also linked to plate tectonic processes, with faster subduction cycles occurring along active plate margins and slower cycles taking place on passive continental margins (reviewed by Warr, Reference Warr2022). This means that there is an intimate relationship between silicate weathering, clay mineral precipitation, climate and the tectono-thermal behaviour of the lithosphere.
It is well-established that Earth’s early climate was characterized by warmth and stability, with ice-free conditions prevailing during most of the Proterozoic eon (2,500–541 Myr ago), except for the extreme glaciation events in the Cryogenian (e.g. Lipp et al., Reference Lipp, Shorttle, Sperling, Brocks, Cole and Crockford2021; Isson & Rauzi, Reference Isson and Rauzi2024). This occurred despite the solar luminosity being ∼5–20% lower than in the present day (e.g. Gough, Reference Gough1981). The long-lasting climate stability changed dramatically in the Phanerozoic eon (541 Myr–present), with the appearance of frequent and rapid icehouse–greenhouse cycles (e.g. Royer et al., Reference Royer, Berner, Montañez, Tabor and Beerling2004). The reasons for this climate transition have been controversially discussed in the literature. Proposed explanations include temporal changes in Earth’s crustal composition (e.g. Gaillardet et al., Reference Gaillardet, Dupré, Louvat and Allegre1999; Coogan & Dosso, Reference Coogan and Dosso2015), increased uplift and erosional rates, as well as changes in ocean currents (e.g. Foster & Rohling, Reference Foster and Rohling2013), which had maintained elevated pCO2 during the Precambrian (e.g. Kump et al., Reference Kump, Brantley and Arthur2000; Isson & Planavsky, Reference Isson and Planavsky2018; Sheldon et al., Reference Sheldon, Mitchell and Dzombak2021). Others have suggested that large variations in seawater pH and certain processes, such as carbonate mineral precipitation and reverse weathering reactions (Equation 1a–e), helped to regulate the global carbon–silica cycle in the Precambrian and thus the input and output fluxes of C and Si between marine sediment sinks, the ocean and the atmosphere (Fig. 2; e.g. Sun & Turchyn, Reference Sun and Turchyn2014; Isson & Planavsky, Reference Isson and Planavsky2018; Kasting, Reference Kasting2019; Isson et al., Reference Isson, Planavsky, Coogan, Stewart, Ague and Bolton2020; Krissansen-Totton & Catling, Reference Krissansen-Totton and Catling2020; Torres et al., Reference Torres, Milliken, Hüpers, Kim and Lee2022). All of the mechanisms discussed above are expected to impact atmospheric CO2 concentrations and climate stability (e.g. Royer et al., Reference Royer, Berner, Montañez, Tabor and Beerling2004).
CO2 fluxes related to silicate weathering
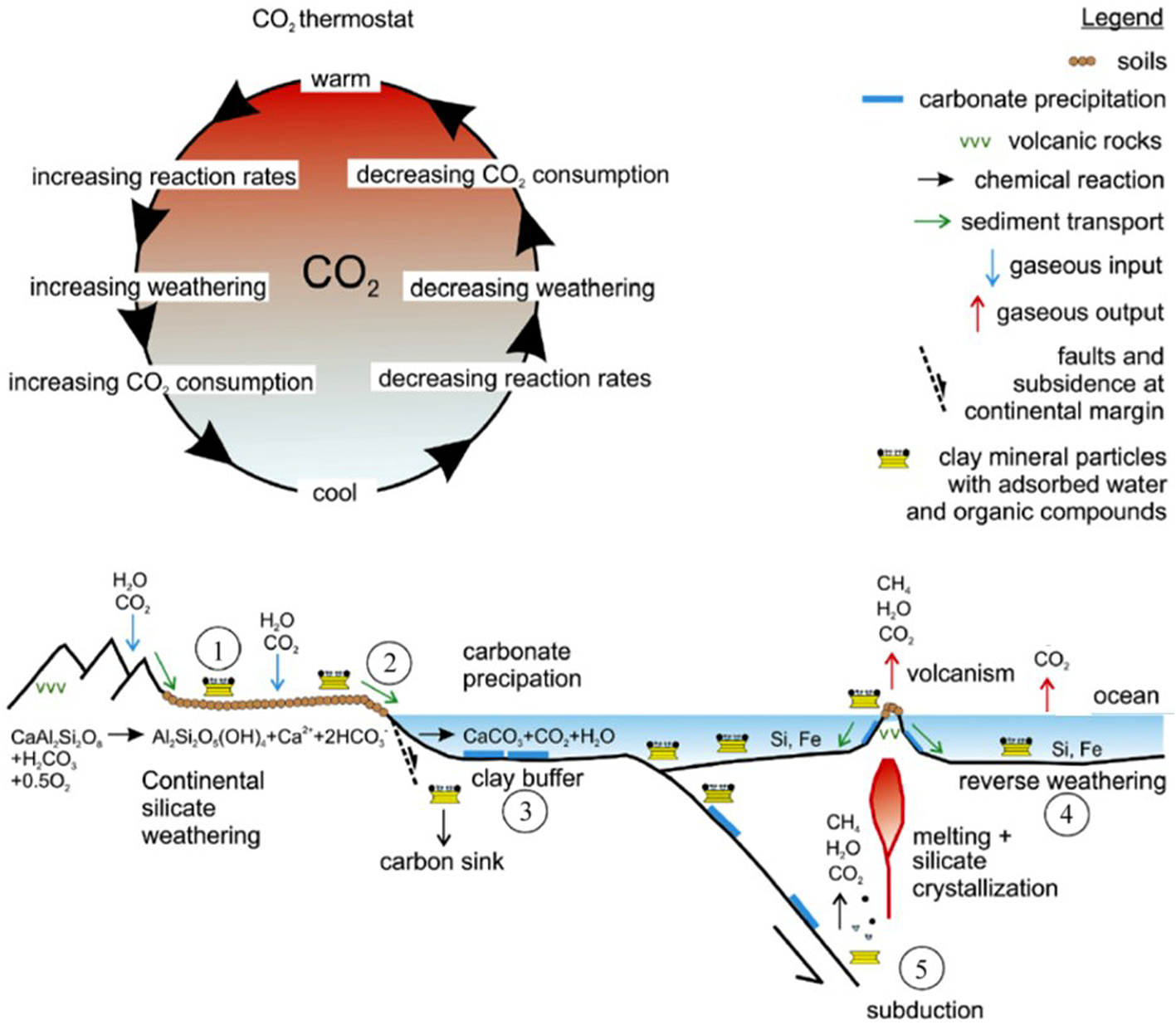
Five main mechanisms control clay mineral-related fluxes of CO2 to the atmosphere, which are described in detail by Warr (Reference Warr2022). These are: (1) silicate weathering on the continents, which creates clay mineral covers and crusts that can slow down the rate of rock alteration; (2) organic–clay mineral complexation, which helps to pass dissolved carbon species into the oceans; (3) clay mineral-based pH buffering, which aids carbonate precipitation in the oceanic environment; (4) reverse weathering reactions, causing clay mineral precipitation onto the seafloor; and (5) crustal diagenesis, metamorphism, melting and volcanism in the upper lithosphere, where (4) and (5) release water and other volatiles (e.g. CO2) from transforming clay minerals (Fig. 3).
Illustration of the interaction of clay minerals, silicate weathering and atmospheric CO2 levels. (1) Formation of continental surface clay mineral deposits and weathered crusts consumes CO2 and slows down rock alteration; (2) organic–clay minerals complexation fixes carbon and passes it into the oceans; (3) clay mineral buffers aid carbonate mineral precipitation in the marine environment; (4) reverse weathering and clay mineral precipitation occur on the seafloor, releasing CO2; and (5) the release of water and volatiles from clay minerals during subduction enhances crustal melting and volcanism (Warr, Reference Warr2022, with minor modifications).
Within this context, Earth’s climate today is controlled mainly by the (im)balance between CO2 emissions to the atmosphere and carbon burial by silicate weathering and carbonate formation (e.g. Friedlingstein et al., Reference Friedlingstein, Houghton, Marland, Hackler, Boden and Conway2010; Colbourn et al., Reference Colbourn, Ridgwell and Lenton2015; Coogan & Gillis, Reference Coogan and Gillis2020). Carbon removal by marine biogenic and diagenetic–abiogenic carbonate mineral formation consumes the majority of the atmospheric CO2 inventory (e.g. Torres et al., Reference Torres, Milliken, Hüpers, Kim and Lee2022), as well as some of the ocean-bound carbonate alkalinity sourced from silicate and carbonate weathering on the continents and the deep ocean floor (e.g. Walker et al., Reference Walker, Hays and Kasting1981; Colbourn et al., Reference Colbourn, Ridgwell and Lenton2015). It is believed that the long-term carbon cycle and thus Earth’s climate are stabilized by a negative-feedback mechanism known as the silicate weathering thermostat (e.g. Berner et al., Reference Berner, Lasaga and Garrels1983; Brantley et al., Reference Brantley, Shaughnessy, Lebedeva and Balashov2023), which involves temperature- or climate-sensitive atmospheric CO2 consumption via the chemical weathering of susceptible silicate minerals (Figs. 2 & 3; e.g. Walker et al., Reference Walker, Hays and Kasting1981; Kasting, Reference Kasting2019; Jin et al., Reference Jin, Wan, Liu, Zhao, Pei and Yu2023).
In this widely accepted negative-feedback climate model, silicate weathering is enhanced when pCO2 and temperature increase, leading to increased consumption of atmospheric CO2 and hence a cooling effect. As pCO2 falls, the silicate weathering rate decreases, permitting the build-up of atmospheric CO2 and consequent warming (e.g. Isson & Planavsky, Reference Isson and Planavsky2018; Kasting, Reference Kasting2019; Penman et al., Reference Penman, Rugenstein, Ibarra and Winnick2020). The timescale of the climate response related to changes in silicate weathering is considered to lie in the range of 170–380 kyr, with an average response time of ∼240 kyr (Colbourn et al., Reference Colbourn, Ridgwell and Lenton2015). As a direct consequence of the described negative-feedback loop, continental and seafloor weathering of primary igneous silicate minerals, such as pyroxene, olivine, amphibole, feldspar and phyllosilicates, is currently considered as a key reaction step for regulating Earth’s climate and seawater composition (e.g. Coogan & Gillis, Reference Coogan and Gillis2013, Reference Coogan and Gillis2018). As an example, this can be represented by the reactions show in Equation 2a–c using wollastonite (CaSiO3; e.g. Walker et al., Reference Walker, Hays and Kasting1981; Berner et al., Reference Berner, Lasaga and Garrels1983; Schott et al., Reference Schott, Pokrovsky, Spalla, Devreux, Gloter and Mielczarski2012; Farkaš et al., Reference Farkaš, Wallmann, Mosley, Staudigel, Zheng, Leyden, Anbar and Weis2025):
These reactions illustrate how released bivalent cations (mainly Ca2+, but also Ba2+, Mg2+, Mn2+, Fe2+, Sr2+ and Zn2+) and carbonate alkalinity are finally deposited as CaCO3 minerals in marine sediments, causing a net reduction of 1 mol of atmospheric CO2 per 1 mol of CaCO3 formed. In addition, dissolved cations (including also Al3+, Na+, K+ and Li+, among others) and Si(OH)4, if transported to the ocean, can trigger the formation of biogenic opal and authigenic clay minerals in various shallow and deep marine settings (Fig. 1; e.g. Michalopoulos & Aller, Reference Michalopoulos and Aller1995; Isson & Planavsky, Reference Isson and Planavsky2018; Baldermann et al., Reference Baldermann, Banerjee, Czuppon, Dietzel, Farkaš and Löhr2022). However, we note here that clay mineral formation also occurs in various continental settings, such as in soils and lakes (e.g. Huggett & Cuadros, Reference Huggett and Cuadros2010; Bristow et al., Reference Bristow, Kennedy, Morrison and Mrofka2012; Pozo & Calvo, Reference Pozo and Calvo2018), although none of these depositional environments belong (by definition) to clay minerals formed through reverse weathering reactions. In any case, the rates of silicate weathering, CO2 uptake and carbonate mineral precipitation are temperature dependent and therefore can be viewed as representing a planetary thermostat or climate regulator. This is indicated, for example, by the climate-sensitive weathering rates of north-eastern Iceland river catchments (e.g. Gislason et al., Reference Gislason, Oelkers, Eiriksdottir, Kardjilov, Gisladottir and Sigfusson2009) or the terrestrial climate archive from the Valley of Lakes (Mongolia, Central Asia; see Fig. 4; e.g. Baldermann et al., Reference Baldermann, Wasser, Abdullayev, Bernasconi, Löhr and Wemmer2021). There, variations in temperature and climatic conditions during the Cenozoic were directly related to pulses of intensified physical versus chemical weathering, as is evident by the correlation between the Chemical Index of Alteration (CIA) and excursions of oxygen isotopes (δ18O) recorded in pedogenic carbonates (e.g. Richoz et al., Reference Richoz, Baldermann, Frauwallner, Harzhauser, Daxner-Höck, Klammer and Piller2017). The Valley of Lakes continental archive thus provides a sensitive record of changing hydroclimatic conditions, as calcrete (i.e. authigenic calcite) deposited in palaeosols resulted in systematically lighter δ18O values during wetter conditions and relatively heavier δ18O values during incursions of drier conditions. Analogously, increased chemical weathering indices (here: the CIA) correspond to periods of wetter conditions, and vice versa (Baldermann et al., Reference Baldermann, Wasser, Abdullayev, Bernasconi, Löhr and Wemmer2021).
Regional response of pedogenic carbonate oxygen isotope (δ18Ocarbonate) geochemistry and silicate-based CIA associated with Cenozoic climate change in the Valley of Lakes, Mongolia (source: Baldermann et al., Reference Baldermann, Wasser, Abdullayev, Bernasconi, Löhr and Wemmer2021). Note the high sensitivity of the study site to regional and global climatic variability during this time, which is expressed as a higher abundance of detrital illite (Ilt) versus authigenic illite-smectite (Ilt-Sme) in periods of increased physical versus chemical weathering (see scanning electron microscopy images on the left). Oi-1a/b = Oi-1a/b glaciation (∼34–33 Myr); EOA = early Oligocene aridification (∼31 Myr); OGM = Oligocene glacial maximum (∼28 Myr); LOW = late Oligocene warming (∼25 Myr); Mi-1 = Mi-1 glaciation (∼23 Myr). Aqu = Aquitanian; Bar = Bartonian; Bur = Burdigalian; Cha = Chattian; Lan = Langhian; Lut = Lutetian; Pri = Priabonian; Rup = Rupelian; Ser = Serravallian; V-PDB = Vienna Peedee Belemnite; Ypr = Ypresian.
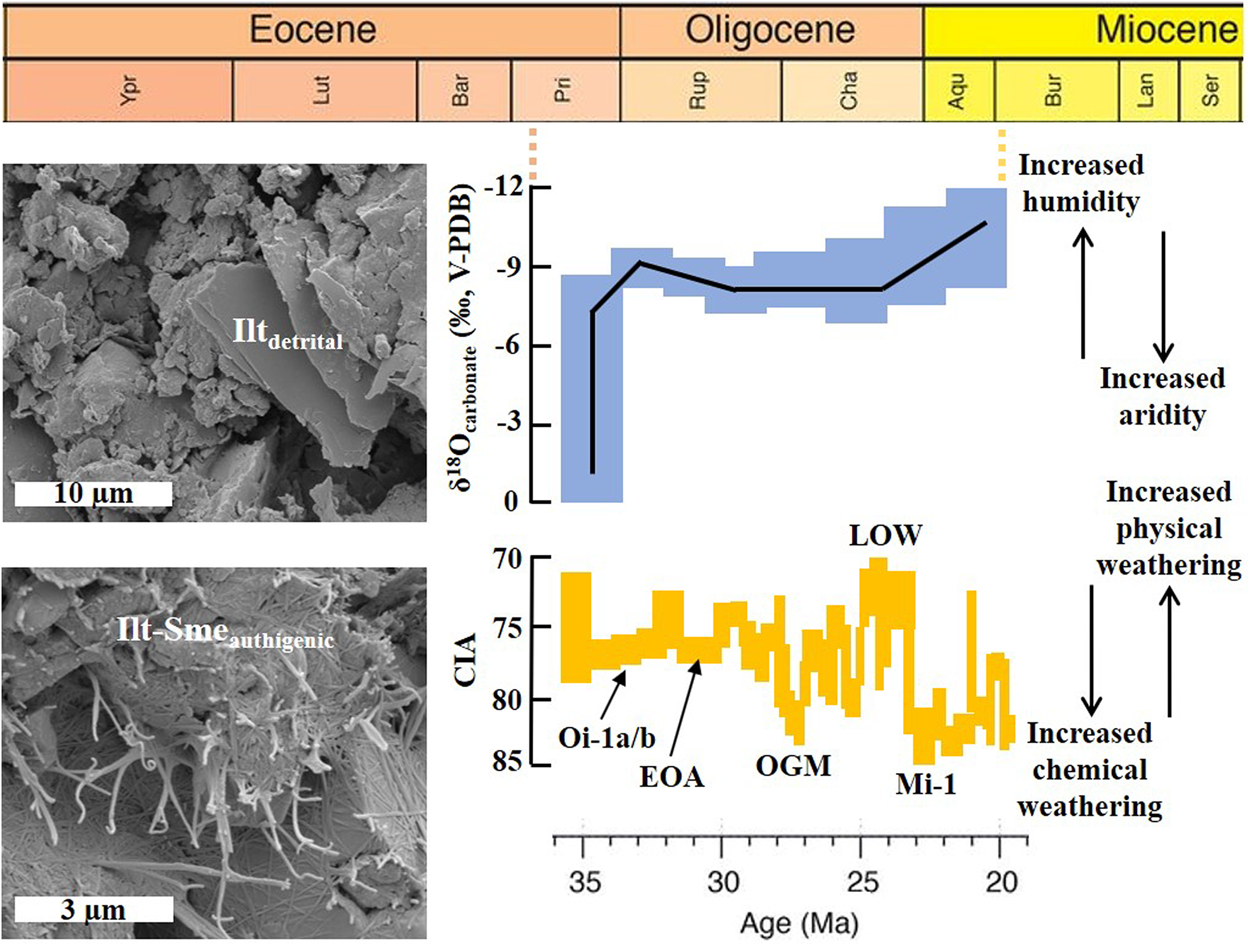
The reactivity of Earth’s crust (e.g. Gaillardet et al., Reference Gaillardet, Dupré, Louvat and Allegre1999; Coogan & Dosso, Reference Coogan and Dosso2015) is also modulated by biological factors, such as the activities of terrestrial plants or soil microbes. As these factors may accelerate or slow down the rates of physical and chemical weathering (e.g. Cuadros, Reference Cuadros2017), they are ultimately linked to the regulation of the carbon cycle. These became more important since the advent of biologically active soils and biogenic sediments approximately in the late Neoproterozoic (e.g. Kennedy et al., Reference Kennedy, Droser, Mayer, Pevear and Mrofka2006; Rafiei et al., Reference Rafiei, Löhr, Baldermann, Webster and Kong2020), as well as the rise of land plants in the Palaeozoic (e.g. Lenton et al., Reference Lenton, Crouch, Johnson, Pires and Dolan2012; Porada et al., Reference Porada, Lenton, Pohl, Weber, Mander and Donnadieu2016; McMahon & Davies, Reference McMahon and Davies2018; Davies et al., Reference Davies, Shillito, Slater, Liu and McMahon2020; Isson & Rauzi, Reference Isson and Rauzi2024). Furthermore, tectonic uplift and topographic elevation (e.g. the Himalayas and the Tibetan Plateau), combined with high precipitation rates, further increased the rate of silicate chemical weathering (and CO2 consumption) by up to 40–60% (e.g. Raymo & Ruddiman, Reference Raymo and Ruddiman1992; Kump & Arthur, Reference Kump, Arthur and Ruddiman1997; Xu et al., Reference Xu, Bufe, Li, Erlanger, Ran and Zhang2024). In addition, the age of an orogen or a mountain belt impacts the rate of silicate weathering. Young orogens weather faster than older ones due to the rapid exhumation of fresh lithologies that are less resistant to chemical weathering processes (e.g. Maher & Chamberlain, Reference Maher and Chamberlain2014).
To summarize, it is widely accepted that changes to global continental weathering have led to fluctuations in atmospheric CO2 over geological timescales (e.g. Berner, Reference Berner1990). However, it remains unclear how the combination of tectonic processes, climate change and rock lithologies, among other factors, has influenced the rate of silicate weathering on a global scale (e.g. Hilley & Porder, Reference Hilley and Porder2008). Hilley & Porder (Reference Hilley and Porder2008) calculated a global annual Si weathering flux of 19–46 Tmol, obtained by considering local erosion rates, dust fluxes, temperature and the water balance of various regions worldwide. These authors concluded that the uplift of the Himalayas since the late Cenozoic has had a significant impact on Earth’s climate. Similarly, (Caves Rugenstein et al., Reference Caves Rugenstein and Chamberlain2018) have argued that the Cenozoic global cooling (∼52 Myr to the present day) was mainly triggered by the increasing exposure of highly reactive lithologies exposed in the Himalayas and the Tibetan Plateau. This underlines the importance of the global weathering feedback to Earth’s climate evolution, and vice versa (Fig. 4; e.g. Caves et al., Reference Caves, Jost, Lau and Maher2016; Baldermann et al., Reference Baldermann, Wasser, Abdullayev, Bernasconi, Löhr and Wemmer2021; Jin et al., Reference Jin, Wan, Liu, Zhao, Pei and Yu2023). Recently, Lipp et al. (Reference Lipp, Shorttle, Sperling, Brocks, Cole and Crockford2021) have estimated global weathering intensities based on the evolution of the continents’ bulk composition over geological time and proposed that silicate weathering was intensified in the Archaean (∼25% more CO2 sequestration between ∼4000 and 2500 Myr). This was followed by reduced but relatively constant rates of silicate weathering during the Proterozoic (∼2500–541 Myr) and the Phanerozoic (<541 Myr to present). However, these authors also identified peaks in the weathering intensities on shorter timescales within the Phanerozoic, such as during the Carboniferous, Triassic and Cretaceous periods (see Fig. 3 in Lipp et al., Reference Lipp, Shorttle, Sperling, Brocks, Cole and Crockford2021), illustrating how silicate weathering acts in response to changes in atmospheric CO2. Similar conclusions were recently drawn by Isson & Rauzi (Reference Isson and Rauzi2024), who have argued that the evolution of Earth’s global climate, seawater pH and changes in silicate weathering triggered distinct pulses or declines in early clay mineral diagenesis (i.e. continental and reverse weathering), which are detailed further in later sections of this article.
Early clay mineral diagenesis and climate feedback
Reverse weathering involves the formation of authigenic clay minerals in near-surface marine sediments, a process that consumes carbonate alkalinity and produces acidity and leads to a net release of CO2 (cf. Equation 1a–e & Fig. 2; e.g. Farkaš et al., Reference Farkaš, Wallmann, Mosley, Staudigel, Zheng, Leyden, Anbar and Weis2025). It can proceed via entirely de novo precipitation of clay minerals, but it can also proceed via the partial dissolution of detrital precursor phases, followed by cation enrichment, substitution and restructuring as a ‘new phase’ (e.g. Garrels, Reference Garrels1965; Mackenzie & Garrels, Reference Mackenzie and Garrels1966b; Mackin & Aller, Reference Mackin and Aller1984). The latter pathway is illustrated by the transformation of kaolinite to berthierine with no additional Si(OH)4 being consumed in this reaction (see Equation 3; Bhattacharyya, Reference Bhattacharyya1983):
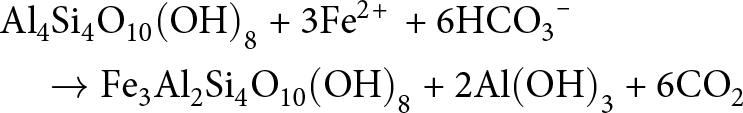 \begin{align}
&{\text{A}}{{\text{l}}_{\text{4}}}{\text{S}}{{\text{i}}_{\text{4}}}{{\text{O}}_{{\text{10}}}}{\left( {{\text{OH}}} \right)_{\text{8}}} {\text{ + 3F}}{{\text{e}}^{{\text{2\! + }}}}{\text{ + 6HC}}{{\text{O}}_{\text{3}}}^{\text{--}} \nonumber\\
&\quad \to {\text{F}}{{\text{e}}_{\text{3}}}{\text{A}}{{\text{l}}_{\text{2}}}{\text{S}}{{\text{i}}_{\text{4}}}{{\text{O}}_{{\text{10}}}}{\left( {{\text{OH}}} \right)_{\text{8}}}{\text{ + 2Al}}{\left( {{\text{OH}}} \right)_{\text{3}}}{\text{ + 6C}}{{\text{O}}_{\text{2}}}\end{align}
\begin{align}
&{\text{A}}{{\text{l}}_{\text{4}}}{\text{S}}{{\text{i}}_{\text{4}}}{{\text{O}}_{{\text{10}}}}{\left( {{\text{OH}}} \right)_{\text{8}}} {\text{ + 3F}}{{\text{e}}^{{\text{2\! + }}}}{\text{ + 6HC}}{{\text{O}}_{\text{3}}}^{\text{--}} \nonumber\\
&\quad \to {\text{F}}{{\text{e}}_{\text{3}}}{\text{A}}{{\text{l}}_{\text{2}}}{\text{S}}{{\text{i}}_{\text{4}}}{{\text{O}}_{{\text{10}}}}{\left( {{\text{OH}}} \right)_{\text{8}}}{\text{ + 2Al}}{\left( {{\text{OH}}} \right)_{\text{3}}}{\text{ + 6C}}{{\text{O}}_{\text{2}}}\end{align}Reverse weathering is thought to have exerted a significant influence on global climate and marine elemental cycling in the past and in the present (e.g. Berner et al., Reference Berner, Lasaga and Garrels1983; Mackenzie & Kump, Reference Mackenzie and Kump1995; Baldermann et al., Reference Baldermann, Banerjee, Czuppon, Dietzel, Farkaš and Löhr2022; Isson & Rauzi, Reference Isson and Rauzi2024), given that the formation of authigenic clay minerals releases CO2 and impacts on Earth’s climate and seawater pH (e.g. Mackenzie & Garrels, Reference Mackenzie and Garrels1966b; Kump et al., Reference Kump, Brantley and Arthur2000; Isson & Planavsky, Reference Isson and Planavsky2018). In simple terms, reverse weathering reactions remobilize carbon species in the ocean–atmosphere system (e.g. Isson & Planavsky, Reference Isson and Planavsky2018; Farkaš et al., Reference Farkaš, Wallmann, Mosley, Staudigel, Zheng, Leyden, Anbar and Weis2025) and act to counterbalance (reverse) the sequestration of carbon linked to silicate weathering (e.g. West et al., Reference West, Galy and Bickle2005). This process can be illustrated via the example of kaolinite formation by incongruous weathering of silicates followed by kaolinite alteration to authigenic illite. A standard example of silicate weathering occurring on the continents is the reaction of anorthite (CaAl2Si2O8) with dissolved CO2 (i.e. carbonic acid) at pH 4–6 and at high leaching rates to form kaolinite and alkalinity in (sub)tropical soils (e.g. West et al., Reference West, Galy and Bickle2005; Galán, Reference Galán2006), according to the reaction in Equation 4:
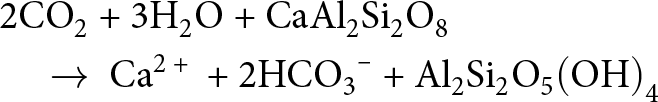 \begin{align}&{\text{2C}}{{\text{O}}_{\text{2}}}{\text{ + 3}}{{\text{H}}_{\text{2}}}{\text{O + CaA}}{{\text{l}}_{\text{2}}}{\text{S}}{{\text{i}}_{\text{2}}}{{\text{O}}_{\text{8}}} \nonumber\\
&\quad \to {\text{ C}}{{\text{a}}^{{\text{2 + }}}}{\text{ + 2HC}}{{\text{O}}_{\text{3}}}^{\text{--}}{\text{ + A}}{{\text{l}}_{\text{2}}}{\text{S}}{{\text{i}}_{\text{2}}}{{\text{O}}_{\text{5}}}{\left( {{\text{OH}}} \right)_{\text{4}}}\end{align}
\begin{align}&{\text{2C}}{{\text{O}}_{\text{2}}}{\text{ + 3}}{{\text{H}}_{\text{2}}}{\text{O + CaA}}{{\text{l}}_{\text{2}}}{\text{S}}{{\text{i}}_{\text{2}}}{{\text{O}}_{\text{8}}} \nonumber\\
&\quad \to {\text{ C}}{{\text{a}}^{{\text{2 + }}}}{\text{ + 2HC}}{{\text{O}}_{\text{3}}}^{\text{--}}{\text{ + A}}{{\text{l}}_{\text{2}}}{\text{S}}{{\text{i}}_{\text{2}}}{{\text{O}}_{\text{5}}}{\left( {{\text{OH}}} \right)_{\text{4}}}\end{align}When delivered to marginal marine settings, such as mangrove forests, either 1 or 2 moles of soil-derived, detrital kaolinite (depending on the Si flux) can react further with dissolved cations, provided from saprolite weathering and marine porewater ingress, to form 1 mole of Fe(III)-illite and other components through kaolinite-illitization according to Equation 5a–b (Cuadros et al., Reference Cuadros, Andrade, Ferreira, de Moya Partiti, Cohen and Vidal-Torrado2017):
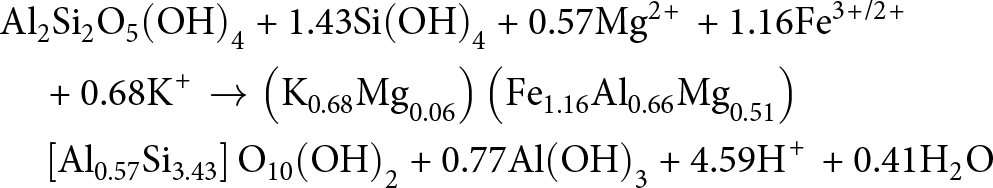 \begin{align}
&{\text{A}}{{\text{l}}_{\text{2}}}{\text{S}}{{\text{i}}_{\text{2}}}{{\text{O}}_{\text{5}}}{\left( {{\text{OH}}} \right)_{\text{4}}}{\text{ + 1}}{\text{.43Si}}{\left( {{\text{OH}}} \right)_{\text{4}}}{\text{ + 0}}{\text{.57M}}{{\text{g}}^{{\text{2\! + }}}}{\text{ + 1}}{\text{.16F}}{{\text{e}}^{{\text{3\! +\! /2\! + }}}} \nonumber\\
&\quad {\text{ + 0}}{\text{.68}}{{\text{K}}^{\text{\! + }}} \to \left( {{{\text{K}}_{{\text{0}}{\text{.68}}}}{\text{M}}{{\text{g}}_{{\text{0}}{\text{.06}}}}} \right)\left( {{\text{F}}{{\text{e}}_{{\text{1}}{\text{.16}}}}{\text{A}}{{\text{l}}_{{\text{0}}{\text{.66}}}}{\text{M}}{{\text{g}}_{{\text{0}}{\text{.51}}}}} \right) \nonumber\\
&\quad \left[ {{\text{A}}{{\text{l}}_{{\text{0}}{\text{.57}}}}{\text{S}}{{\text{i}}_{{\text{3}}{\text{.43}}}}} \right]{{\text{O}}_{{\text{10}}}}{\left( {{\text{OH}}} \right)_{\text{2}}}{\text{ + 0}}{\text{.77Al}}{\left( {{\text{OH}}} \right)_{\text{3}}}{\text{ + 4}}{\text{.59}}{{\text{H}}^{\text{\! + }}}{\text{ + 0}}{\text{.41}}{{\text{H}}_{\text{2}}}{\text{O}}
\end{align}
\begin{align}
&{\text{A}}{{\text{l}}_{\text{2}}}{\text{S}}{{\text{i}}_{\text{2}}}{{\text{O}}_{\text{5}}}{\left( {{\text{OH}}} \right)_{\text{4}}}{\text{ + 1}}{\text{.43Si}}{\left( {{\text{OH}}} \right)_{\text{4}}}{\text{ + 0}}{\text{.57M}}{{\text{g}}^{{\text{2\! + }}}}{\text{ + 1}}{\text{.16F}}{{\text{e}}^{{\text{3\! +\! /2\! + }}}} \nonumber\\
&\quad {\text{ + 0}}{\text{.68}}{{\text{K}}^{\text{\! + }}} \to \left( {{{\text{K}}_{{\text{0}}{\text{.68}}}}{\text{M}}{{\text{g}}_{{\text{0}}{\text{.06}}}}} \right)\left( {{\text{F}}{{\text{e}}_{{\text{1}}{\text{.16}}}}{\text{A}}{{\text{l}}_{{\text{0}}{\text{.66}}}}{\text{M}}{{\text{g}}_{{\text{0}}{\text{.51}}}}} \right) \nonumber\\
&\quad \left[ {{\text{A}}{{\text{l}}_{{\text{0}}{\text{.57}}}}{\text{S}}{{\text{i}}_{{\text{3}}{\text{.43}}}}} \right]{{\text{O}}_{{\text{10}}}}{\left( {{\text{OH}}} \right)_{\text{2}}}{\text{ + 0}}{\text{.77Al}}{\left( {{\text{OH}}} \right)_{\text{3}}}{\text{ + 4}}{\text{.59}}{{\text{H}}^{\text{\! + }}}{\text{ + 0}}{\text{.41}}{{\text{H}}_{\text{2}}}{\text{O}}
\end{align}or
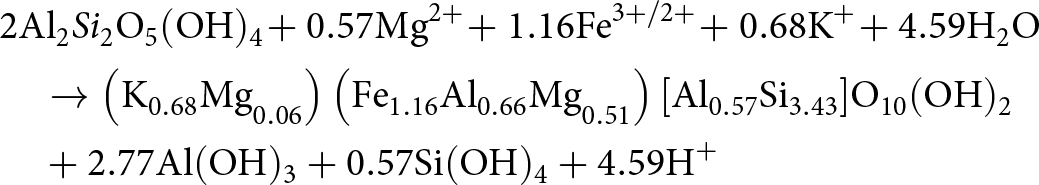 \begin{align}
&2\text{Al}_2Si_2\text{O}_5(\text{OH})_4\! + 0.57\text{Mg}^{2+}\! + 1.16\text{Fe}^{3+/2+}\! + 0.68\text{K}^{+}\! + 4.59\text{H}_2 \text{O} \nonumber\\
&\quad\to \left(\text{K}_{0.68}\text{Mg}_{0.06}\right)\left(\text{Fe}_{1.16}\text{Al}_{0.66}\text{Mg}_{0.51}\right) [\text{Al}_{0.57}\text{Si}_{3.43}]\text{O}_{10}(\text{OH})_2 \nonumber\\
&\quad+ 2.77\text{Al}(\text{OH})_3 + 0.57\text{Si}(\text{OH})_4 + 4.59\text{H}^{+}
\end{align}
\begin{align}
&2\text{Al}_2Si_2\text{O}_5(\text{OH})_4\! + 0.57\text{Mg}^{2+}\! + 1.16\text{Fe}^{3+/2+}\! + 0.68\text{K}^{+}\! + 4.59\text{H}_2 \text{O} \nonumber\\
&\quad\to \left(\text{K}_{0.68}\text{Mg}_{0.06}\right)\left(\text{Fe}_{1.16}\text{Al}_{0.66}\text{Mg}_{0.51}\right) [\text{Al}_{0.57}\text{Si}_{3.43}]\text{O}_{10}(\text{OH})_2 \nonumber\\
&\quad+ 2.77\text{Al}(\text{OH})_3 + 0.57\text{Si}(\text{OH})_4 + 4.59\text{H}^{+}
\end{align}It has been proposed that reverse weathering was more efficient under the pervasively silica-rich conditions found in Earth’s early oceans, helping to maintain warm, ice-free conditions throughout the Precambrian period (e.g. Isson & Planavsky, Reference Isson and Planavsky2018). This probably occurred via the establishment of a strong relationship between global climate, continental silicate weathering rates, seawater pH and authigenic clay mineral formation, according to the empirical reactions show in Equations 6 & 7:
 \begin{align}&{\text{Silicate weathering: Mafic minerals and glass + C}}{{\text{O}}_{\text{2}}}{\text{ + }}{{\text{H}}_{\text{2}}}{\text{O }} \nonumber\\
&\quad \to {\text{ HC}}{{\text{O}}_{\text{3}}}^{\text{--}}{\text{ + Si}}{\left( {{\text{OH}}} \right)_{\text{4}}}{\text{ + cations}}\end{align}
\begin{align}&{\text{Silicate weathering: Mafic minerals and glass + C}}{{\text{O}}_{\text{2}}}{\text{ + }}{{\text{H}}_{\text{2}}}{\text{O }} \nonumber\\
&\quad \to {\text{ HC}}{{\text{O}}_{\text{3}}}^{\text{--}}{\text{ + Si}}{\left( {{\text{OH}}} \right)_{\text{4}}}{\text{ + cations}}\end{align}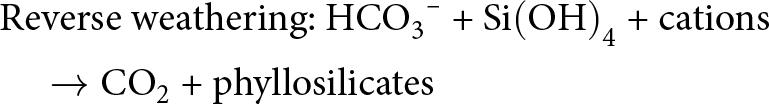 \begin{align}
&{\text{Reverse weathering: HC}}{{\text{O}}_{\text{3}}}^{\text{--}}{\text{ + Si}}{\left( {{\text{OH}}} \right)_{\text{4}}}{\text{ + cations}} \nonumber\\
&\quad \to {\text{C}}{{\text{O}}_{\text{2}}}{\text{ + phyllosilicates}}\end{align}
\begin{align}
&{\text{Reverse weathering: HC}}{{\text{O}}_{\text{3}}}^{\text{--}}{\text{ + Si}}{\left( {{\text{OH}}} \right)_{\text{4}}}{\text{ + cations}} \nonumber\\
&\quad \to {\text{C}}{{\text{O}}_{\text{2}}}{\text{ + phyllosilicates}}\end{align}Banerjee et al. (Reference Banerjee, Bansal and Thorat2016) and Baldermann et al. (Reference Baldermann, Banerjee, Czuppon, Dietzel, Farkaš and Löhr2022) have also argued that glauconite – the most abundant authigenic clay mineral in the Phanerozoic – preferentially formed during warm climate periods with sea-level high stands, such as in the late Palaeozoic, early Mesozoic and Cenozoic. However, the influence of marine authigenic clay mineral formation on the carbon cycle and seawater pH remains poorly quantified, as the reaction rates are unknown and as the amount of CO2 released during reverse weathering is probably much lower than the cooling produced by increased silicate weathering reactions that consume CO2. Further research should assess the kinetics of clay mineral reactions and their influence on the pH sensitivity of aquatic systems, which is required to model the impact of reverse weathering on Earth’s CO2 inventory and climate change (e.g. Isson & Rauzi, Reference Isson and Rauzi2024).
Carbon cycle models (e.g. Isson & Planavsky, Reference Isson and Planavsky2018) can incorporate silica (re)cycling and alkalinity consumption through reverse weathering reactions (Equation 1a–e) to account for the formation of berthierine, chamosite, clinochlore, corrensite, glauconite, greenalite, minnesotaite, odinite, saponite, sepiolite and sudoite. These models attempt to provide quantitative estimates on long-term fluctuations in Earth’s climate versus seawater pH and pCO2 (e.g. Isson & Planavsky, Reference Isson and Planavsky2018). It should be noted that the rates of reverse weathering and clay mineral authigenesis in marine sediments are very often considered to be slow (e.g. Ehlert et al., Reference Ehlert, Doering, Wallmann, Scholz, Sommer and Grasse2016; Baldermann et al., Reference Baldermann, Mavromatis, Frick and Dietzel2018). In contrast, faster reverse weathering rates may occur in proximal and distal marine settings, such as in deltas, estuaries and in some deep-sea environments (e.g. Michalopoulos & Aller, Reference Michalopoulos and Aller1995, Reference Michalopoulos and Aller2004; Wallmann et al., Reference Wallmann, Aloisi, Haeckel, Tishchenko, Pavlova and Greinert2008, Reference Wallmann, Geilert and Scholz2023; Rahman et al., Reference Rahman, Aller and Cochran2017; Steiner et al., Reference Steiner, Rae, Berelson, Adkins, Hou and Dong2022; Geilert et al., Reference Geilert, Frick, Garbe-Schönberg, Scholz, Sommer and Grasse2023). In proximal marine settings, it has been estimated that reverse weathering may sequester 10–50% of the total dissolved silica exported to the modern oceans (e.g. Tréguer & De La Rocha, Reference Tréguer and La Rocha C.L2013; Rahman et al., Reference Rahman, Aller and Cochran2016; Tréguer et al., Reference Tréguer, Sutton, Brzezinski, Charette, Devries and Dutkiewicz2021).
The marine silica cycle: biogenic silica versus authigenic clay mineral precipitation
Reverse weathering strongly influences the global marine Si cycle via the transformation of biogenic and amorphous silica (SiO2), reactive silicates (e.g. feldspar) or dissolved Si(OH)4 into authigenic clay minerals (e.g. Rahman et al., Reference Rahman, Aller and Cochran2016; Geilert et al., Reference Geilert, Frick, Garbe-Schönberg, Scholz, Sommer and Grasse2023; Wallmann et al., Reference Wallmann, Geilert and Scholz2023). The individual transformation rates are believed to be relatively slow in open marine settings but may be significantly faster in estuaries, deltas and hydrothermal (deep-sea) environments. Importantly, it is the chemical composition (and temperature) of local marine porewaters that controls the rate of clay mineral authigenesis, with continental margin and shelf settings being particularly favourable. This is probably because the faster and quantitatively more important precipitation of biogenic opal by marine silicifying organisms in the upper water column of the oceans consumes dissolved Si(OH)4, resulting in low Si(OH)4 concentrations in open, deep marine porewaters, according to the reaction shown in Equation 8:
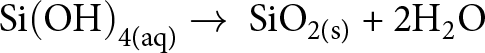 \begin{equation}{\text{Si}}{\left( {{\text{OH}}} \right)_{{\text{4(aq)}}}} \to {\text{ Si}}{{\text{O}}_{{\text{2(s)}}}}{\text{ + 2}}{{\text{H}}_{\text{2}}}{\text{O}}\end{equation}
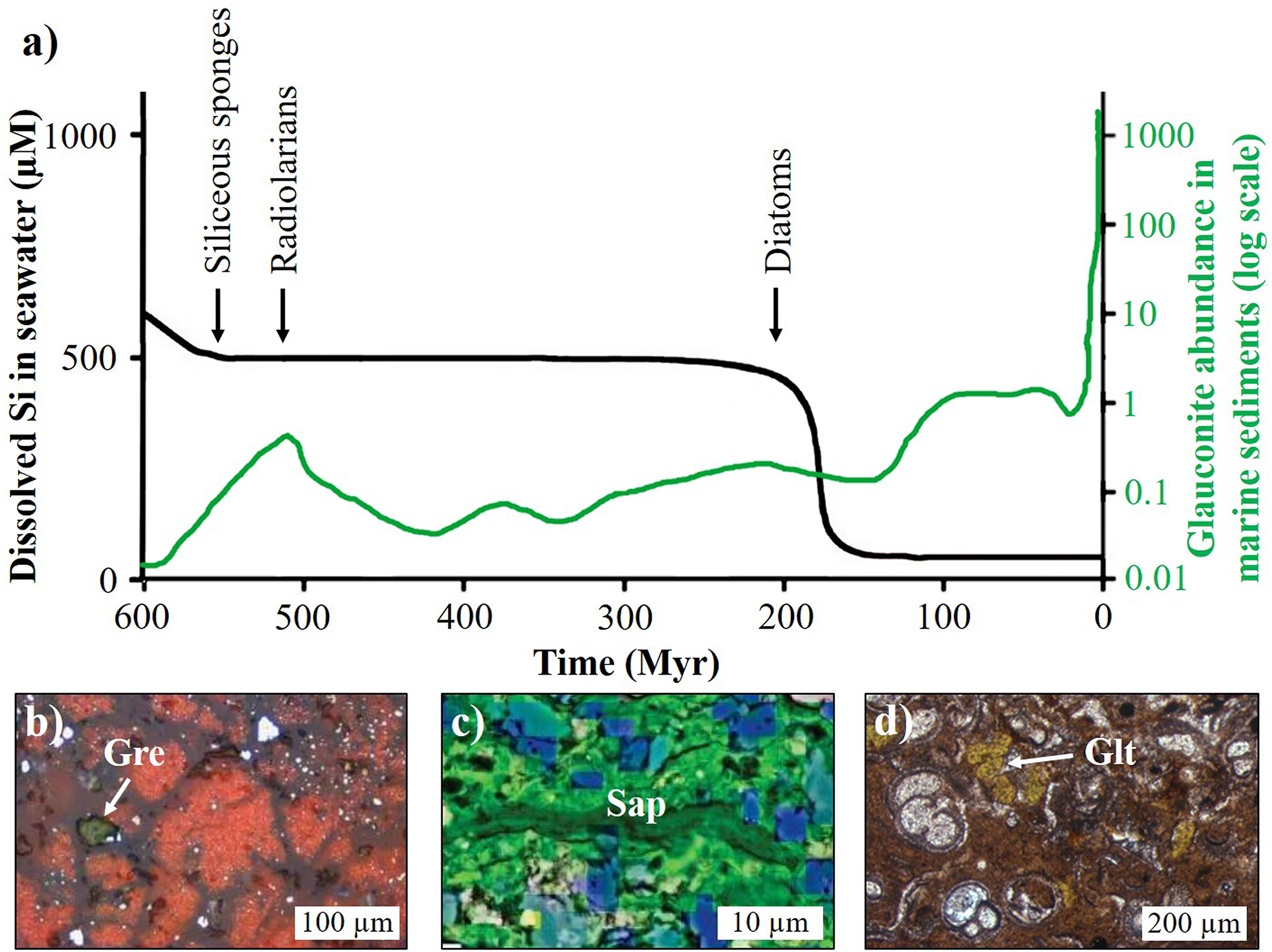
\begin{equation}{\text{Si}}{\left( {{\text{OH}}} \right)_{{\text{4(aq)}}}} \to {\text{ Si}}{{\text{O}}_{{\text{2(s)}}}}{\text{ + 2}}{{\text{H}}_{\text{2}}}{\text{O}}\end{equation}Throughout the Phanerozoic, the main silicifying organisms have changed, with siliceous sponges expanding in the Cambrian, followed by radiolarians in the Ordovician and diatoms in the Jurassic–Cretaceous (Fig. 5a). Today, such marine biomineralizers account for ∼70–90% of the global silica sink (e.g. Tréguer & De La Rocha, Reference Tréguer and La Rocha C.L2013; Tréguer et al., Reference Tréguer, Sutton, Brzezinski, Charette, Devries and Dutkiewicz2021). Consequently, modern seawater has a Si(OH)4 concentration (<0.1 mM) that is at least an order of magnitude lower than that inferred for the Precambrian oceans (∼1.00–2.21 mM; cf. Fig. 5a; e.g. Siever, Reference Siever1992; Conley et al., Reference Conley, Frings, Fontorbe, Clymans, Stadmark and Hendry2017). The modern ‘Si-depleted’ seawater is therefore unsaturated with respect to the conditions required to precipitate most marine clay minerals (e.g. Baldermann et al., Reference Baldermann, Mavromatis, Frick and Dietzel2018; Isson & Planavsky, Reference Isson and Planavsky2018), contributing to the reduced number of authigenic clay mineral species forming in the modern ocean compared to the wider array documented in Precambrian marine sedimentary rocks. For example, Han et al. (Reference Han, Löhr, Abbott, Baldermann, Shields and Cui2024) have proposed that widespread evaporitic conditions on the Yangtze Gorges shelf (south China) provided favourable conditions during the Ediacaran for authigenic saponite formation and the development of the Doushantuo Biota.
Isson & Planavsky (Reference Isson and Planavsky2018) have also argued that the rise of silica-biomineralizing eukaryotes may have caused a substantial reduction of the reverse weathering rate in the ancient oceans, resulting in a drop in pCO2 and a change to a less stable climate system by reducing the pH buffer capacity of the global oceans. Such a scenario is consistent with the high diversity of authigenic clay minerals, such as greenalite (Fig. 5b), saponite (Fig. 5c), glauconite and minnesotaite, in the Precambrian geological record, but it is incompatible with the significant increase of glauconite in the marine sediments since the Phanerozoic (Fig. 5a, d). The negative correlation between the Si(OH)4 concentration in Phanerozoic seawater and the abundance of glauconite in the rock record further suggests that seawater Si was not a limiting factor for this type of marine clay mineral authigenesis. Consequently, the advent of biomineralization and the resulting drop in the seawater Si concentration did not result in the cessation of authigenic clay mineral formation during the Phanerozoic (cf. Equation 8, reverse reaction, & Fig. 5a). Future research should explore the abundance and type of authigenic clay minerals in Precambrian versus Phanerozoic marine sedimentary rocks.
Since the emergence and the proliferation of diatoms in the Jurassic–Cretaceous, a drop in the seawater Si(OH)4 concentration occurred (cf. Fig. 5, ∼200 Myr; e.g. Conley et al., Reference Conley, Frings, Fontorbe, Clymans, Stadmark and Hendry2017), which came along with a (time-displaced) increase in the abundance of glauconite in the marine rock record (Fig. 5a). Such a scenario appears to be consistent with the comparatively high abundance of glauconite and celadonite observed in modern coastal and oceanic sediments (e.g. Banerjee et al., Reference Banerjee, Bansal and Thorat2016; Cuadros et al., Reference Cuadros, Andrade, Ferreira, de Moya Partiti, Cohen and Vidal-Torrado2017; López-Quirós et al., Reference López-Quirós, Escutia, Sánchez-Navas, Nieto, Garcia-Casco and Martín-Algarra2019, Reference López-Quirós, Lobo, Mendes and Nieto2023; Baldermann et al., Reference Baldermann, Banerjee, Czuppon, Dietzel, Farkaš and Löhr2022; Geilert et al., Reference Geilert, Frick, Garbe-Schönberg, Scholz, Sommer and Grasse2023). There, re-mineralization of reactive biogenic opal in the sedimentary pile provides an early diagenetic (secondary) pool of dissolved Si(OH)4, which is required for clay mineral formation (e.g. Vorhies & Gaines, Reference Vorhies and Gaines2009; Isson & Planavsky, Reference Isson and Planavsky2018; Woltz et al., Reference Woltz, Anderson, Tosca and Porter2023). This process illustrates the linkage between biogenic opal formation and early diagenetic re-mineralization and clay mineral authigenesis, which is expressed by Equation 9:
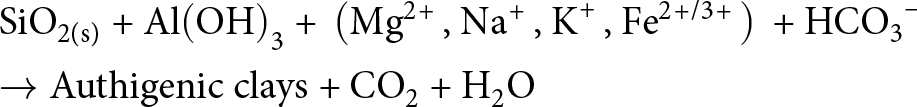 \begin{equation}\begin{gathered}
{\text{Si}}{{\text{O}}_{{\text{2(s)}}}}{\text{ + Al}}{\left( {{\text{OH}}} \right)_{\text{3}}}{\text{ + }}\left( {{\text{M}}{{\text{g}}^{{\text{2\! + }}}}{\text{, N}}{{\text{a}}^{\text{\! + }}}{\text{, }}{{\text{K}}^{\text{\! + }}}{\text{, F}}{{\text{e}}^{{\text{2\! +\! /3\! + }}}}} \right){\text{ + HC}}{{\text{O}}_{\text{3}}}^{\text{--}} \hfill \\
\to {\text{Authigenic clays + C}}{{\text{O}}_{\text{2}}}{\text{ + }}{{\text{H}}_{\text{2}}}{\text{O}} \hfill \\
\end{gathered} \end{equation}
\begin{equation}\begin{gathered}
{\text{Si}}{{\text{O}}_{{\text{2(s)}}}}{\text{ + Al}}{\left( {{\text{OH}}} \right)_{\text{3}}}{\text{ + }}\left( {{\text{M}}{{\text{g}}^{{\text{2\! + }}}}{\text{, N}}{{\text{a}}^{\text{\! + }}}{\text{, }}{{\text{K}}^{\text{\! + }}}{\text{, F}}{{\text{e}}^{{\text{2\! +\! /3\! + }}}}} \right){\text{ + HC}}{{\text{O}}_{\text{3}}}^{\text{--}} \hfill \\
\to {\text{Authigenic clays + C}}{{\text{O}}_{\text{2}}}{\text{ + }}{{\text{H}}_{\text{2}}}{\text{O}} \hfill \\
\end{gathered} \end{equation}Despite the above relationships, porewater data collected from shelf, slope and deep-sea sites across modern oceans indicate that the dissolved Si(OH)4 concentration is generally too low and not favourable for more extensive marine clay mineral authigenesis (e.g. Isson & Planavsky, Reference Isson and Planavsky2018). This is mainly due to the high degree of bio-opal remineralization occurring in the upper water column of the oceans (∼70%), the regionally uneven distribution of bio-opal production and the rapid vertical diffusion of porewater-related Si(OH)4 to the Si-depleted bottom seawater. All of these factors prevent the establishment of the high concentrations of Si(OH)4 at the sediment–seawater interface that are required for authigenic clay mineral formation (e.g. Tréguer & De La Rocha, Reference Tréguer and La Rocha C.L2013). The notable exceptions are glauconite, nontronite, saponite and sepiolite-palygorskite group clay minerals with the right compositions to form in marine environments. According to hydrochemical modelling of modern seawater and porewater compositions, these require comparatively low amounts of dissolved Si(OH)4 to precipitate (e.g. Baldermann et al., Reference Baldermann, Mavromatis, Frick and Dietzel2018; Isson & Planavsky, Reference Isson and Planavsky2018). However, it has been frequently documented that authigenic clay minerals can precipitate directly on dissolving biogenic silica particles, which could imply that at least the reactive parts of siliceous skeletons react in situ to form phyllosilicates (e.g. Michalopoulos et al., Reference Michalopoulos, Aller and Reeder2000; Presti & Michalopoulos, Reference Presti and Michalopoulos2008; Loucaides et al., Reference Loucaides, Michalopoulos, Presti, Koning, Behrends and Van Cappellen2010; Baldermann et al., Reference Baldermann, Warr, Grathoff and Dietzel2013; López-Quirós et al., Reference López-Quirós, Escutia, Sánchez-Navas, Nieto, Garcia-Casco and Martín-Algarra2019; Geilert et al., Reference Geilert, Frick, Garbe-Schönberg, Scholz, Sommer and Grasse2023). This is presumably because the local pore fluid Si(OH)4 concentration at this reactive interface is much higher than that measured in the bulk fluid, generating geochemical microenvironments that favour clay mineral authigenesis, as confirmed by the hydrochemical modelling of modern porewater compositions (e.g. Wallmann et al., Reference Wallmann, Geilert and Scholz2023).
Consequently, it is the variation of the local dissolved Si(OH)4 concentration in porewaters across geological time that controlled the rate and magnitude of authigenic clay mineral formation. Whereas biogenic opal mineralization may have decreased the average seawater Si concentration, the massive sedimentation of siliceous components may have favoured large local porewater or early diagenetic micromilieu Si(OH)4 concentrations that helped to boost the authigenesis of diverse clay mineral species. In the Precambrian, the global ocean was likely to have been anoxic (Fe-rich) and rich in Si(OH)4 (e.g. Siever, Reference Siever1992; Poulton & Canfield, Reference Poulton and Canfield2011; Conley et al., Reference Conley, Frings, Fontorbe, Clymans, Stadmark and Hendry2017; Tosca et al., Reference Tosca, Jiang, Rasmussen and Muhling2019), favouring the deposition of, for example, extensive silica-rich banded iron formations (BIFs), as well as greenalite and other marine clay minerals (e.g. Rasmussen et al., Reference Rasmussen, Bay, Co and Road1998, Reference Rasmussen, Meier, Krapež and Muhling2013, Reference Rasmussen, Muhling, Suvorova and Krapež2017, Reference Rasmussen, Muhling and Krapež2021; Klein, Reference Klein2005; Johnson et al., Reference Johnson, Muhling, Cosmidis, Rasmussen and Templeton2018; Tosca et al., Reference Tosca, Jiang, Rasmussen and Muhling2019; Muhling & Rasmussen, Reference Muhling and Rasmussen2020). The latter Fe-phyllosilicates not only occur in distinct and separate sedimentary units; they also appear as encapsulated pockets and inclusions in early diagenetic chert (Fig. 5b). This implies that authigenic clay minerals, despite their tendency to dissolve, dehydrate and/or transform during diagenesis and metamorphism, could have been more abundant in the Precambrian record than recently thought or preserved in the rock record (e.g. Morris, Reference Morris1993; Klein, Reference Klein2005; Rasmussen et al., Reference Rasmussen, Muhling, Suvorova and Krapež2017; Isson & Planavsky, Reference Isson and Planavsky2018; Huang et al., Reference Huang, Shen, Wang, Ma, Li and Zhou2024; Isson & Rauzi, Reference Isson and Rauzi2024). This abundance of clay minerals formed by reverse weathering would have potentially impacted the global climate at that time.
In contrast, Dunlea et al. (Reference Dunlea, Murray, Santiago Ramos and Higgins2017) suggested that during the Cenozoic (∼66 Myr until today) stagnating rates of marine authigenic clay mineral formation (Fig. 5a) caused seawater Mg/Ca to rise and atmospheric CO2 to decline over the past ∼50 Myr. This was linked to the remarkable Cenozoic global cooling that continued until the beginning of the temperature rise in the late Pleistocene. This latter relationship highlights the complex links between the global marine element cycles and Earth’s climate response – features that are evident in the diversity of authigenic clay minerals found in the geological record.
(a) Relationship between the evolution of marine siliceous organisms, the dissolved Si concentration in the ocean (data source: Conley et al., Reference Conley, Frings, Fontorbe, Clymans, Stadmark and Hendry2017) and the abundance of glauconite in the geological record (data source: Banerjee et al., Reference Banerjee, Bansal and Thorat2016) from the early Precambrian to the present. The glauconite abundance (in %) is expressed by the logarithm of the actual number of occurrences in different intervals divided by the time duration. The sedimentation and early diagenetic re-mineralization of biogenic opal probably provided porewater Si(OH)4 favouring clay mineral authigenesis. (b) Photomicrograph showing authigenic greenalite (Gre; marked with white arrow) embedded in a greyish chert and reddish hematite matrix from the Late Archean Kushtagi-Hungund Schist Belt (India). (c) Artificially coloured scanning electron microscopy with energy-dispersive X-ray spectroscopy mineral map displaying authigenic saponite (Sap) hosted in a blueish dolomite matrix from the Ediacaran Doushantuo Formation (China). (d) Photomicrograph showing glauconite (Glt; marked with white arrow) growing in foraminiferous ooze in Pleistocene–Holocene-aged Ivory Coast–Ghana Marginal Ridge sediments. Images (b)–(d) are provided by AB.
Stratigraphic distribution of authigenic clay minerals in the marine rock record
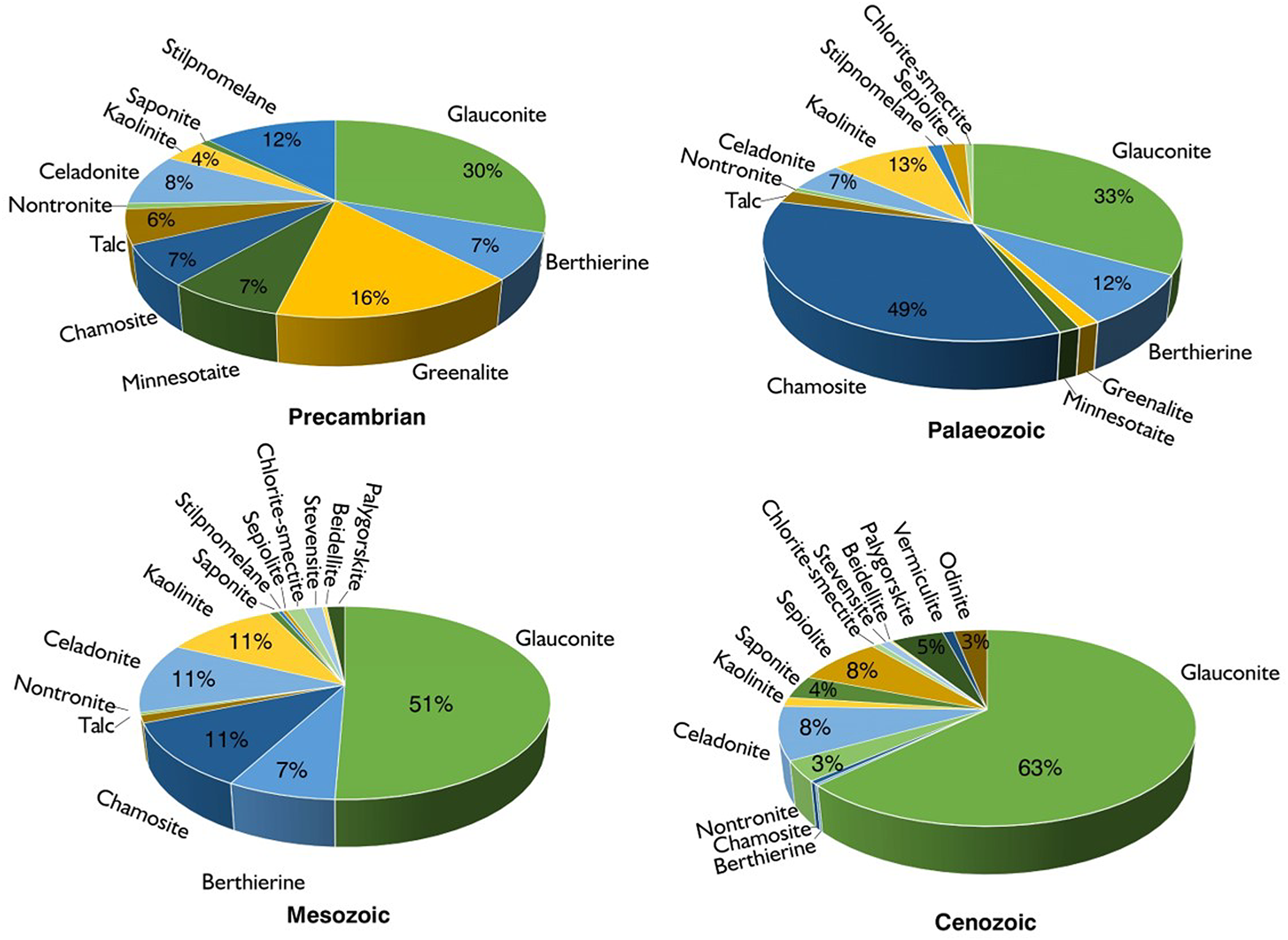
In this section, an overview is given of the stratigraphic and temporal distributions of authigenic clay minerals that typically form in the marine realm during early diagenesis (cf. Fig. 1). Secondary clay mineral assemblages that may precipitate in the continental critical zone and during late diagenesis are not considered, as these minerals do not represent reverse weathering products. However, we emphasize that recognizing and quantifying the frequently fine-grained products of reverse weathering reactions in the marine rock record are challenging (e.g. Hazen et al., Reference Hazen, Sverjensky, Azzolini, Bish, Elmore, Hinnov and Milliken2013; Han et al., Reference Han, Löhr, Abbott, Baldermann, Farkaš and McMahon2022a), given that authigenic clay minerals are commonly intermingled with clay minerals of detrital origin or have been partly overprinted by deep burial diagenetic reactions. Therefore, marine clay minerals are typically of diverse origin. However, most of the truly authigenic clay minerals found in marine sediment archives are Fe-rich (here: 12 out of the 18 clay minerals considered; Hazen et al., Reference Hazen, Sverjensky, Azzolini, Bish, Elmore, Hinnov and Milliken2013). The reasons for the dominance of authigenic clay minerals enriched in Fe(III) and/or Fe(II) over Fe-poor or Fe-depleted clay minerals in marine sediments remain disputed, but controlling factors may include the early diagenetic Fe(III) reduction coupled to complex inorganic and biological processes and the progressive decay of organic matter. These reactions provide (partly organo-complexed) reactive Fe2+ and also Fe3+ ions to the marine porewater inventory, from which authigenic Fe-rich clay minerals tend to precipitate (see earlier).
Beidellite and chlorite-smectite
Beidellite is a rather uncommon mineral of the dioctahedral smectite group that has been documented mainly in Cretaceous sequences (Figs. 6 & 7). It is thought to have formed by the alteration of montmorillonite in the presence of dissolved Fe3+/2+, typically under the elevated temperatures of mid-oceanic ridge sites (e.g. Post et al., Reference Post, Cupp and Madsen1997; Bouna et al., Reference Bouna, Rhouta, Daoudi, Maury, Amjoud and Senocq2012). Orange-coloured beidellite has been found in macropores and intergranular pores of altered mafic host rocks, together with intergrowths of zeolites, goethite and greenish-brown chlorite-smectite mixed-layered clay minerals (e.g. Hu et al., Reference Hu, Yang, Liu, Li, Dang and Zhu2023). Corrensite, an ordered chlorite-smectite group mineral, is locally found in hydrothermally altered marine clastic sediments and ophiolitic rocks. For example, Buatier et al. (Reference Buatier, Früh-Green and Karpoff1995) reported that authigenic corrensite formed ∼32 m below the sediment–seawater interface at 300°C ± 30°C during active hydrothermal venting at the axial rift valley of the northern Juan de Fuca Ridge.
Temporal distribution of authigenic clay minerals found in different sedimentary environments across geological time. The occurrence of authigenic clay minerals is provided in Table S1; however, the nature of kaolinite is disputed – it can be of detrital or authigenic origin. Note that glauconite is the most abundant authigenic clay mineral across time, whereas chamosite is particularly abundant in the Palaeozoic.
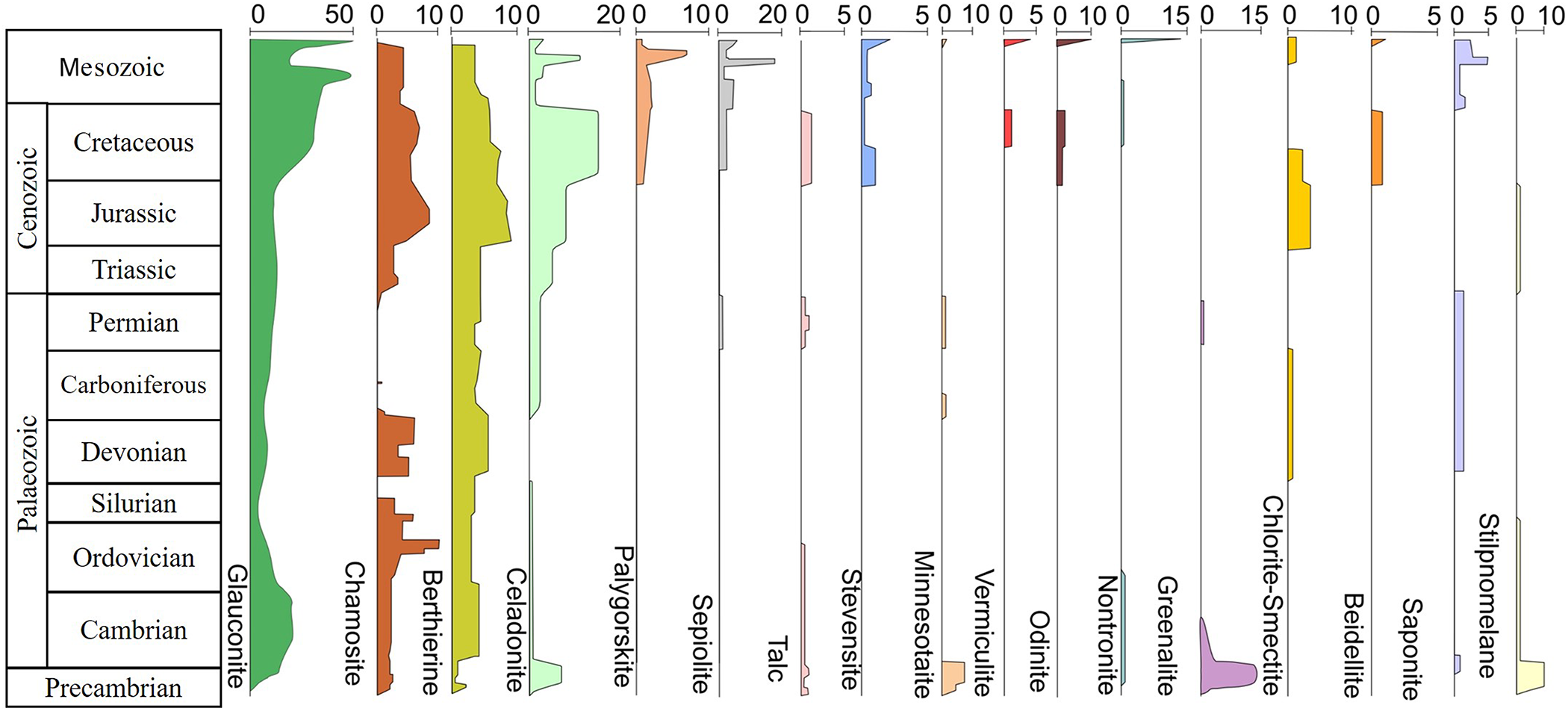
Stratigraphic distribution of common clay minerals found in various sedimentary environments. Kaolinite is not shown because the great majority of kaolinite occurrences are considered to be of detrital rather than marine authigenic origin. The numbers at the top represent the numbers of published occurrences of specific authigenic clay minerals provided in Table S1.
Berthierine
Berthierine, the Fe member of the serpentine mineral group, formed preferentially as oolitic grain infills (together with chamosite) during Palaeozoic and Mesozoic times (Figs. 6 & 7). In the Jurassic and Cretaceous, it formed by the alteration of volcanogenic particles in oxygen-depleted seawater (e.g. Maynard, Reference Maynard1986; Rudmin et al., Reference Rudmin, Banerjee, Maximov, Novoselov, Trubin and Smirnov2022a; Kalinina et al., Reference Kalinina, Maximov, Molukpayeva, Sherstyukov, Kerimov and Rudmin2024; Roy Choudhury et al., Reference Roy Choudhury, Srimani, Mondal, Chakrabarty, Banerjee and Sarkar2024). Reactions involved the transformation of siderite, kaolinite, odinite, chamosite and glauconite precursor grains under reducing conditions (e.g. Iijima & Matsumoto, Reference Iijima and Matsumoto1982; Bhattacharyya, Reference Bhattacharyya1983; Hornibrook & Longstaffe, Reference Hornibrook and Longstaffe1996; Tang et al., Reference Tang, Shu, Ma, Jiang, Zhou and Shi2017) or the alteration of transported laterite-derived materials under reducing, marine porewater conditions (e.g. Fritz & Toth, Reference Fritz and Toth1997; Toth & Fritz, Reference Toth and Fritz1997). The availability of dissolved Fe2+ is key to the formation of yellowish-greenish-brownish berthierine in shallow marine, estuarine and fresh to brackish settings under (sub)tropical conditions (Fig. 8a). In some cases, berthierine has been documented in near-coastal coal-forming environments (Iijima & Matsumoto, Reference Iijima and Matsumoto1982) and marine hydrothermal surroundings (Taylor, Reference Taylor1990; Roy Choudhury et al., Reference Roy Choudhury, Srimani, Mondal, Chakrabarty, Banerjee and Sarkar2024).
Transmission electron microscopy and scanning electron microscopy images of common authigenic clay minerals formed during reverse weathering across geological time. (a) Fibrous berthierine (Bth) crystals (∼7 Å) infilling ooids (inset image) from the Oligocene channel ironstones of the Lisakovsk deposit in Kazakhstan (Rudmin et al., Reference Rudmin, Kalinina, Banerjee, Reva, Kondrashova and Kanaki2021). (b) Lath-like celadonite (Cel) particles occurring in interstitial and void spaces in green bole beds (inset image) within the Late Cretaceous Deccan Volcanic Province in India. This Cel formed in a non-marine environment, but it otherwise precipitated under submarine conditions (Singh et al., Reference Singh, Banerjee, Pande, Bhattacharya, Sarkar and Le Pera2022). (c) Microcrystalline chamosite (Chm) and siderite (Sd) spar from the Middle Triassic Kremikovtsi Sedimentary Exhalative Siderite Iron Formation in Bulgaria (Damyanov & Vassileva, Reference Damyanov and Vassileva2001). (d) Lath-like glauconite (Glt) infilling foraminifera tests (inset image) at the Ivory Coast basin close to the Ghana continental margin (Baldermann et al., Reference Baldermann, Warr, Grathoff and Dietzel2013). (e) Fibrous greenalite (Gre) crystals in polygon-shaped quartz (Qz) grains from the Proterozoic-aged Brockman Iron Formation in Australia (Rasmussen et al., Reference Rasmussen, Muhling and Krapež2021). (f) Honeycomb-like saponite (Sap) aggregates covering the surface of altered basalt grains (Voigt et al., Reference Voigt, Pearce, Fries, Baldermann and Oelkers2020). Coloured stars in the low-resolution images indicate the positions of regions of interest used for high-resolution imaging.

Celadonite
This mineral of the Fe- and K-rich mica group preferentially forms as vesicle fillings and as massive aggregates in altered basaltic rocks (Fig. 8b). It is common in the Proterozoic, often in association with BIFs (e.g. Savko, Reference Savko2006), but it is most abundant in the well-preserved Mesozoic rock record (Figs. 6 & 7), probably because of the peak mid-oceanic ridge length and fast seafloor spreading rates at that time (e.g. Müller et al., Reference Müller, Dutkiewicz, Seton and Gaina2013, Reference Müller, Mather, Dutkiewicz, Keller, Merdith and Gonzalez2022). The most favourable conditions for the formation of bluish-green celadonite developed in the Cretaceous, when this mineral was primarily formed by the in situ alteration of mafic and intermediate volcanic rocks in the presence of either marine or meteoric fluids rich in K+, Fe3+/2+ and Mg2+ ions (e.g. Baker et al., Reference Baker, Rember, Sprenke and Strawn2012). Even though celadonite and glauconite can precipitate under similar physicochemical conditions, celadonite is rarely found in Palaeozoic successions and modern oceanic sediments and volcanic rocks (e.g. Singh et al., Reference Singh, Banerjee, Roy Choudhury, Bhattacharya and Pande2023).
Chamosite
Even though chamosite, the Fe2+ member of the chlorite group, has formed in marine sediments since the Precambrian, the best conditions for its crystallization prevailed during the Ordovician, Silurian, Devonian and Jurassic to Neogene (Figs. 6, 7 & 8c). Favourable conditions for the precipitation of authigenic chamosite often occurred in close association with the formation of ooidal ironstones, where the supply of dissolved Fe2+ and Mg2+ ions from detrital sources or reducing marine fluids and the leaching of Si(OH)4 and K+ ions from reactive precursor clays, such as berthierine or glauconite, triggered the formation of this mineral (e.g. Mücke, Reference Mücke2006; Tang et al., Reference Tang, Shu, Ma, Jiang, Zhou and Shi2017). However, the burial diagenetic or hydrothermal formation of chamosite from smectite is also possible. The stratigraphic distribution and the abundance of authigenic chamosite, a relatively common marine clay mineral, which often appears with a greyish-greenish-brown colour, commonly overlap to some degree with those of glauconite (e.g. Van Houten & Bhattacharyya, Reference Van Houten and Bhattacharyya1982; Rudmin et al., Reference Rudmin, Mazurov and Banerjee2019, Reference Rudmin, Kalinina, Banerjee, Reva, Kondrashova and Kanaki2021). Chamosite has, however, also been reported in Proterozoic sequences, where Fe2+- and Si(OH)4-rich seawater facilitated its formation by ion exchange between the sediment, porewater and seawater (e.g. Tang et al., Reference Tang, Shu, Ma, Jiang, Zhou and Shi2017).
Glauconite
This Fe-rich mica group mineral was commonly precipitated as light to dark green rounded concretions (pellets) during the Precambrian, Mesozoic and Cenozoic (Figs. 6 & 7). The Late Cretaceous and Palaeogene marked notable periods of glauconite formation on the continental shelves of that time. Factors that facilitated the formation of glauconite (and of its precursor, Fe(III)-smectite) during greenhouse climates included the availability of dissolved Si(OH)4, Fe2+/3+, K+ and Mg2+ in seawater or sediment porewater, the presence of microenvironments, such as faecal pellets, bioclasts and corroded feldspar, and the development of oxygen-depleted conditions at the sediment–seawater interface (e.g. Meunier & El Albani, Reference Meunier and El Albani2007; Charpentier et al., Reference Charpentier, Buatier, Jacquot, Gaudin and Wheat2011; Baldermann et al., Reference Baldermann, Grathoff and Nickel2012; Banerjee et al., Reference Banerjee, Bansal and Thorat2016; Zhang et al., Reference Zhang, Gaillardet, Barrier and Bouchez2022b). Glauconite formation was also favoured by global sea-level fluctuations and low sedimentation rates (e.g. Roy Choudhury et al., Reference Roy Choudhury, Khanolkar and Banerjee2022). The early diagenetic precipitation of glauconite and other ferric illites contributed significantly to the marine sinks of Fe, Mg, K, Si and Al (e.g. Cuadros et al., Reference Cuadros, Andrade, Ferreira, de Moya Partiti, Cohen and Vidal-Torrado2017; Baldermann et al., Reference Baldermann, Banerjee, Czuppon, Dietzel, Farkaš and Löhr2022). In contrast, during the Holocene the authigenic glauconite grains formed almost exclusively in deep marine sediments (Fig. 8d).
Greenalite
This uncommon Fe-rich mineral of the serpentine group primarily formed in the Precambrian in close association with BIF deposits (Figs. 6, 7 & 8e). Some appearances have also been documented in the Palaeozoic and Mesozoic rock records (e.g. Rasmussen et al., Reference Rasmussen, Bay, Co and Road1998, Reference Rasmussen, Muhling and Krapež2021; Grenne & Slack, Reference Grenne and Slack2019; Ghosh, Reference Ghosh2020). Greenalite formation from amorphous Si- and Fe-bearing precursor phases in the Archaean ocean (e.g. Tosca et al., Reference Tosca, Guggenheim and Pufahl2016, Reference Tosca, Jiang, Rasmussen and Muhling2019; Rasmussen et al., Reference Rasmussen, Muhling, Suvorova and Krapež2017, Reference Rasmussen, Muhling and Krapež2021) was likely favoured by the enrichment of Fe2+, as well as Al3+, Mg2+ and Si(OH)4 in near-surface sediments (e.g. Johnson et al., Reference Johnson, Muhling, Cosmidis, Rasmussen and Templeton2018; Mohanty & Mishra, Reference Mohanty and Mishra2023). Hydrothermal sources have been suggested to have contributed to the source of Fe in greenalites (e.g. Tosca et al., Reference Tosca, Jiang, Rasmussen and Muhling2019; Muhling & Rasmussen, Reference Muhling and Rasmussen2020; Tosca & Tutolo, Reference Tosca and Tutolo2023; Tostevin & Ahmed, Reference Tostevin and Ahmed2023).
Kaolinite
Kaolinite, the dioctahedral member of the kaolinite-serpentine group, occurs as a chemical weathering product in all rock types (e.g. Stoch & Sikora, Reference Stoch and Sikora1976). Its formation and enrichment in soils and shelf sediments were favoured during the Jurassic and Cretaceous (Fig. 7). Although most of the kaolinite found in continental shelf and deep-sea sediments is detrital in origin, some occurrences have been attributed to reverse weathering (e.g. Maliva et al., Reference Maliva, Dickson and Fallick1999; Pe-Piper et al., Reference Pe-Piper, Dolansky and Piper2005). Hydrothermal alteration also induces kaolinization by the removal of Si(OH)4, K+, Mg2+ and Fe2+/3+ from volcanic glasses and mafic minerals in both terrestrial and marine environments (e.g. Altschuler et al., Reference Altschuler, Dwornik and Kramer1963; Erkoyun & Kadіr, Reference Erkoyun and Kadіr2011; Kadіr et al., Reference Kadir, Erman and Erkoyun2011).
Minnesotaite
This Fe-silicate mineral belonging to the pyrophyllite-talc group mainly formed in the Precambrian, but minor occurrences have also been documented in Lower Carboniferous and Permian sequences (Figs. 6 & 7). Minnesotaite deposits are often associated with BIFs, where greenish-grey to olive-green aggregates of fine needles and platelets probably formed via the alteration of greenalite under reducing conditions and in the presence of fluids with a high Mg/Fe ratio (e.g. Blake, Reference Blake1965; Rasmussen et al., Reference Rasmussen, Bay, Co and Road1998; Lempart-Drozd et al., Reference Lempart-Drozd, Blachowski, Gumsley and Ciesielska2022).
Nontronite
This yellowish-green (brownish-orange when weathered) Fe3+ member of the smectite group formed dominantly in the Precambrian, Cambrian, Cretaceous, Palaeogene and Neogene as an alteration product of mafic minerals at mid-oceanic ridge sites or in close association with ironstone deposits, as well as in continental soils (e.g. Köhler et al., Reference Köhler, Singer and Stoffers1994; Dekov et al., Reference Dekov, Kamenov, Stummeyer, Thiry, Savelli and Shanks2007; Afify et al., Reference Afify, Sanz-Montero and Calvo2015, Reference Afify, Sanz-Montero and Calvo2018; Rudmin et al., Reference Rudmin, Banerjee, Sinkina, Ruban, Kalinina and Smirnov2022b). However, nontronite (or Fe(III)-smectite in general) may also precipitate in (semi)confined marine microenvironments at low temperatures and subsequently act as a precursor phase to glauconite formation (e.g. Baldermann et al., Reference Baldermann, Warr, Grathoff and Dietzel2013).
Odinite
Odinite is a rare, Fe(III)-enriched clay mineral of the kaolinite group. It formed in ancient and modern shallow marine settings, lagoons, pro-deltas and estuaries characterized by warm waters enriched in dissolved Fe3+, Mg2+ and Si(OH)4, such as in the Cretaceous (Figs. 6 & 7; e.g. Bailey, Reference Bailey1988). Due to its high reactivity, odinite rapidly alters to berthierine and chlorite (e.g. Hornibrook & Longstaffe, Reference Hornibrook and Longstaffe1996; Šegvić et al., Reference Šegvić, Zanoni and Moscariello2020).
Palygorskite
Palygorskite is a relatively uncommon mineral in the marine rock record. It formed either by the transformation of sepiolite, smectite and volcanic ash or as a precipitate from pore fluids rich in Si(OH)4 and Al3+ and Mg2+ ions (e.g. Yalçin & Bozkaya, Reference Yalçin and Bozkaya2011), preferentially in the Cretaceous and Neogene (Figs. 6 & 7; e.g. Akbulut & Kadir, Reference Akbulut and Kadir2003; Galán & Pozo, Reference Galán and Pozo2011). Microstructural and mineralogical evidence suggests that the purest palygorskite deposits of Cretaceous to Palaeogene age were formed in situ by reverse weathering on the seafloor (e.g. Thiry & Pletsch, Reference Thiry and Pletsch2011). Palygorskite forms a complete solid solution with sepiolite and primarily occurs in lacustrine, fluvial, (peri)marine and soil environments (e.g. Isphording, Reference Isphording1973; Galán & Pozo, Reference Galán and Pozo2011).
Saponite
The trioctahedral (Mg-rich) smectite group mineral saponite mainly formed during the Cenozoic, although rare occurrences of this mineral have been reported in the Precambrian, Devonian and Permian (Figs. 6 & 7). Precambrian saponite preferentially formed in restricted, evaporitic lagoons that were temporally connected to the ocean (e.g. Han et al., Reference Han, Löhr, Abbott, Baldermann, Voigt and Yu2022b). Bluish-green (reddish-brown when weathered) saponite is also known to form via the alteration of mafic rocks or volcanic matter under the influence of hydrothermal fluids in marine settings (Fig. 8f), as well as in evaporitic and alkaline lakes (e.g. Andrews, Reference Andrews1980; Post, Reference Post1984; Akbulut & Kadir, Reference Akbulut and Kadir2003; Rudmin et al., Reference Rudmin, López-Quirós, Banerjee, Ruban, Shaldybin and Bernatonis2023a).
Sepiolite
This hydrous Mg-rich silicate mineral is found in abundance in Cenozoic marine sediments (together with palygorskite), but minor occurrences have also been reported from Permian and Cretaceous strata (Figs. 6 & 7). Fluids rich in Mg2+ and Si(OH)4 facilitated the formation of sepiolite in (peri)marine, transitional and lacustrine environments (e.g. Akbulut & Kadir, Reference Akbulut and Kadir2003; Yalçin & Bozkaya, Reference Yalçin and Bozkaya2011; Sánchez-Roa et al., Reference Sánchez-Roa, Jiménez-Millán, Abad, Faulkner, Nieto and García-Tortosa2016). Sepiolite may also form via the hydrothermal alteration of basalts and volcanic ash (e.g. Akbulut & Kadir, Reference Akbulut and Kadir2003; Galán & Pozo, Reference Galán and Pozo2011; Pozo & Calvo, Reference Pozo and Calvo2018).
Stevensite
Stevensite is a trioctahedral (Mg-rich) smectite group mineral that often formed by precipitation in alkaline and playa lakes (e.g. de Oliveira Nardi Leite et al., Reference de Oliveira Nardi Leite, de Assis Silva and de Ros2020; Netto et al., Reference Netto, Pozo, da Silva, Mexias, Gomes, Borghi and Rios-Netto2022). This mineral mainly formed from the Cretaceous to recent (Figs. 6 & 7) within ooids, peloids and intraclasts in Mg2+- and Si(OH)4-rich lacustrine and coastal marine settings, and more rarely by the alteration of oceanic crust under the influence of hydrothermal fluids (e.g. Banfield et al., Reference Banfield, Jones and Veblen1991; Cuevas et al., Reference Cuevas, de la Villa, Ramirez, Petit, Meunier and Leguey2003; Armelenti et al., Reference Armelenti, Goldberg, Kuchle and de Ros2016; Lima & De Ros, Reference Lima and De Ros2019).
Stilpnomelane
This blackish-greenish-yellowish smectite group mineral formed mainly during the Precambrian and Mesozoic (Figs. 6 & 7) within marine Fe-rich deposits, such as BIFs, that received upwelling of Fe2+- and Si(OH)4-rich deep-seated (hydrothermal) fluids mixed with shallower and slightly oxygenated seawater (e.g. Garzanti et al., Reference Garzanti, Haas and Jadoul1989; Pickard, Reference Pickard2003; Wang et al., Reference Wang, Konhauser and Zhang2015).
Talc
This Mg member of the talc-pyrophyllite group is a relatively rare authigenic mineral in the sedimentary rock record. It mostly formed in Precambrian, Permian and Cretaceous strata (Figs. 6 & 7) under the influence of Al-poor and Si(OH)4- and Mg-rich fluids under marine (or lacustrine) conditions (e.g. Noack et al., Reference Noack, Decarreau, Boudzoumou and Trompette1989; Chen et al., Reference Chen, Lu, Cai and Zhai2002; Tosca et al., Reference Tosca, Macdonald, Strauss, Johnston and Knoll2011; Han et al., Reference Han, Löhr, Abbott, Baldermann, Shields and Cui2024). Rarely, talc also formed by the alteration of sepiolite under the influence of microbial activity and in the presence of Si(OH)4-rich marine pore fluids.
Vermiculite
Dioctahedral vermiculite, an expandable and abundant clay mineral in continental soils, typically formed by the chemical weathering or the alteration of amphibole, biotite and phlogopite precursors (e.g. Wilson, Reference Wilson2004). However, vermiculite is a rather uncommon mineral in the marine rock record, with some rare occurrences being documented in the Cretaceous and Pleistocene to recent times (Figs. 6 & 7). In these rocks, vermiculite appears in its trioctahedral form, typically within interstratified illite-vermiculite and chlorite-vermiculite (e.g. Craw et al., Reference Craw, Smith and Youngson1995; Gutierrez-Mas et al., Reference Gutierrez-Mas, Lopez-Galindo and Lopez-Aguayo1997; Yin et al., Reference Yin, Hong, Churchman, Li and Fang2018).
Reverse weathering and clay mineral authigenesis across geological time
Earth’s greenhouse climatic periods clearly influenced the rates of reverse weathering and the associated distribution and abundance of authigenic clay minerals in marine sediments across geological time. Such periods were characterized by an elevated seawater temperature (on the shelf), a higher continental (dissolved) element export to the oceans, comparatively higher Si(OH)4 concentrations in marine porewaters and enhanced microbial activity that catalysed reverse weathering reactions (e.g. Schieber et al., Reference Schieber, Sur, Banerjee, Schieber, Bose, Eriksson, Banerjee, Sarkar, Altermann and Catuneau2007; Aubineau et al., Reference Aubineau, El, Bekker, Somogyi, Bankole and Macchiarelli2019, Reference Aubineau, El, Bekker, Fru, Somogyi and Medjoubi2020). In this section, some examples are provided of specific periods that favoured clay mineral authigenesis in the oceans and along the submerged continental margins.
Precambrian palaeoclimate
The authigenesis of a suite of Fe(II/III)-phyllosilicates (cf. Fig. 7) in the Precambrian ocean is believed to have released significant amounts of CO2, which helped maintain the warm and ice-free global climate despite the much lower solar luminosity at that time (e.g. Isson & Planavsky, Reference Isson and Planavsky2018; Han et al., Reference Han, Löhr, Abbott, Baldermann, Shields and Cui2024). Glauconite formed dominantly on marine shelves during the Late to Middle Proterozoic, resulting in thick glauconitic sandstones and glauconitic mudrock deposits in many places worldwide (e.g. Banerjee et al., Reference Banerjee, Jeevankumar and Eriksson2008, Reference Banerjee, Bansal and Thorat2016; Mandal et al., Reference Mandal, Banerjee, Sarkar, Mondal and Roy Choudhury2020, Reference Mandal, Roy Choudhury, Das, Sarkar and Banerjee2022). The formation of sedimentary greenalite in the Palaeoproterozoic and of authigenic talc in the Neoproterozoic was facilitated by high Fe2+, Al3+, Si(OH)4 and Mg2+ concentrations in seawater (e.g. Tosca et al., Reference Tosca, Macdonald, Strauss, Johnston and Knoll2011, Reference Tosca, Guggenheim and Pufahl2016; Mohanty & Mishra, Reference Mohanty and Mishra2023; Tostevin & Ahmed, Reference Tostevin and Ahmed2023). Moreover, authigenic saponite formed in restricted evaporitic lagoons over large areas of the Yangtze Gorges shelf in south China from Mg2+- and Si(OH)4-rich Ediacaran seawater (e.g. Han et al., Reference Han, Löhr, Abbott, Baldermann, Voigt and Yu2022b, Reference Han, Löhr, Abbott, Baldermann, Shields and Cui2024). A combination of palaeo-environmental factors, such as the widespread presence of suboxic seawater masses, the absence of marine silicifying organisms, adequate seawater chemistry and the overall low sedimentation rates (e.g. Eriksson et al., Reference Eriksson, Martins-Neto, Nelson, Martins-Neto, Nelson and Aspler2001; Banerjee et al., Reference Banerjee, Jeevankumar and Eriksson2008), favoured the authigenesis of Fe-silicates in the Precambrian (e.g. Banerjee et al., Reference Banerjee, Jeevankumar and Eriksson2008; Mandal et al., Reference Mandal, Banerjee, Sarkar, Mondal and Roy Choudhury2020). Reverse weathering was also facilitated by microbial activity, as is evident from the Precambrian-aged microbial mat deposits enriched in Fe-illite, ferroan illite-smectite, stilpnomelane and minnesotaite (e.g. Konhauser & Urrutia, Reference Konhauser and Urrutia1999; Schieber et al., Reference Schieber, Sur, Banerjee, Schieber, Bose, Eriksson, Banerjee, Sarkar, Altermann and Catuneau2007; Rasmussen et al., Reference Rasmussen, Meier, Krapež and Muhling2013; Konhauser et al., Reference Konhauser, Planavsky, Hardisty, Robbins, Warchola and Haugaard2017; Aubineau et al., Reference Aubineau, El, Bekker, Fru, Somogyi and Medjoubi2020). These microbial biofilms trapped K+ ions from the seawater and released them into the porewater during the degradation of organic matter at early diagenesis, but they also provided the porewater with other important ions, such as Fe2+/3+, during respiration. This resulted in the progressive illitization of smectite precursors, as well as in the neoformation of K- and Fe-phyllosilicates, which subsequently acted as mineral archives that allow us to identify biomineralization processes on ancient Earth (e.g. Vorhies & Gaines, Reference Vorhies and Gaines2009; Aubineau et al., Reference Aubineau, El, Bekker, Somogyi, Bankole and Macchiarelli2019).
Late Palaeozoic to early Mesozoic warming
The Ordovician to Devonian timespan, as well as the Jurassic, witnessed the formation of authigenic chamosite and berthierine in massive ooidal ironstone facies (cf. Fig. 7). Mid-Ordovician-aged chamositic ironstones formed on storm-dominated shelves characterized by Fe2+-rich sea-bottom conditions (e.g. Dunn et al., Reference Dunn, Pufahl and Lokier2021). The authigenesis of these Fe(II)-rich clay minerals that occurred during such periods was linked to a sea-level highstand, the presence of oxygen-depleted but cation-rich seawater, microbial mediation and a warm climate (e.g. Van Houten & Arthur, Reference Van Houten, Arthur, Young and Taylor1989; Young, Reference Young1989; Li et al., Reference Li, Zhang, Ma and Yuan2021b).
Mesozoic greenhouse
Glauconite authigenesis was significant during transgressive events in the Cretaceous (cf. Fig. 7), especially from the Aptian to the Maastrichtian (e.g. Bansal et al., Reference Bansal, Banerjee, Pande and Ruidas2020, Reference Bansal, Banerjee, Chauhan, Rudmin, Borgohain, Upadhyay, Banerjee and Sarkar2021). The higher temperature and suboxic seawater enriched in K+, Mg2+, Si(OH)4 and Fe2+ ions, coupled with a sea-level highstand during the Cenomanian greenhouse conditions, facilitated the high abundance of glauconite on the marine shelves (e.g. Haq, Reference Haq2014; Banerjee et al., Reference Banerjee, Bansal and Thorat2016; Baioumy et al., Reference Baioumy, Farouk and Al-Kahtany2020; Wilmsen & Bansal, Reference Wilmsen and Bansal2021; Scheiblhofer et al., Reference Scheiblhofer, Moser, Löhr, Wilmsen, Farkaš and Gallhofer2022). The formation of glauconite probably sequestered significant amounts of K, Fe, Mg, Si and Al from Cretaceous seawater (e.g. Baldermann et al., Reference Baldermann, Banerjee, Czuppon, Dietzel, Farkaš and Löhr2022). Upper Cretaceous glauconite deposits mainly formed along the margins of the Tethys Ocean (e.g. Bansal et al., Reference Bansal, Banerjee, Pande and Ruidas2020). In addition, a high abundance of other authigenic clay minerals has been documented for the Cretaceous, such as berthierine, celadonite, palygorskite, talc, stevensite and beidellite (cf. Fig. 7), which was possibly related to a fast sea-level rise, warm seawater and high seafloor spreading rates at mid-oceanic ridges at that time (e.g. Jeans, Reference Jeans2006; Müller et al., Reference Müller, Dutkiewicz, Seton and Gaina2013, Reference Müller, Mather, Dutkiewicz, Keller, Merdith and Gonzalez2022; Singh et al., Reference Singh, Banerjee, Roy Choudhury, Bhattacharya and Pande2023). In addition, chamositic-siderite ironstone deposits formed during Oceanic Anoxic Event 3, and abundant Jurassic chamositic-ooidal ironstones precipitated at the Neo-Tethyan margin due to upwelling and fluctuating redox conditions against the backdrop of Mesozoic greenhouse conditions (e.g. Van Houten & Arthur, Reference Van Houten, Arthur, Young and Taylor1989; Han et al., Reference Han, Han, Garzanti, Zhu, Yao and Guo2023).
Cenozoic greenhouse climate
The Cenozoic era was marked by several hyperthermal events, which were associated with abundant authigenic Fe(II/III)-phyllosilicates preserved in the marine sedimentary record (cf. Fig. 7). Glauconitization was frequent due to a high sea level and warm greenhouse conditions since at least the Cretaceous (e.g. Baldermann et al., Reference Baldermann, Banerjee, Czuppon, Dietzel, Farkaš and Löhr2022), with maximum occurrences documented during hyperthermal events in the Palaeogene, Eocene and Oligocene (e.g. Banerjee et al., Reference Banerjee, Choudhury, Saraswati and Khanolkar2020). A multi-proxy study by Roy Choudhury et al. (Reference Roy Choudhury, Khanolkar and Banerjee2022), which applied biostratigraphy, carbon isotopes, Sr concentrations and 40Ar/39Ar chronostratigraphic data, demarcated the Palaeogene hyperthermal events as being remarkably rich in glauconite deposits, such as those reported for the Palaeocene–Eocene Thermal Maximum. The combination of oceanic hypoxia, warm climate conditions and rapid sea-level changes during the Palaeogene temperature maxima favoured the formation of glauconite deposits (e.g. Banerjee et al., Reference Banerjee, Choudhury, Saraswati and Khanolkar2020; Roy Choudhury et al., Reference Roy Choudhury, Khanolkar and Banerjee2022). Before that time, around the Cretaceous–Paleogene boundary, ooidal ironstones were deposited, which contain abundant chamosite, berthierine and glauconite (e.g. Rudmin et al., Reference Rudmin, Banerjee and Mazurov2017).
Modern-day geochemical fluxes associated with reverse weathering
Clay mineral reactions, such as smectite-illitization, kaolinization and chloritization, which occur during late burial diagenesis, are often considered to be slow (e.g. Cuadros et al., Reference Cuadros, Andrade, Ferreira, de Moya Partiti, Cohen and Vidal-Torrado2017). In contrast, the formation of authigenic clay minerals in modern marine sediments by reverse weathering reactions is increasingly recognized as a faster process and can therefore influence modern and past ocean chemistry and related element (re)cycling at the sediment–seawater interface (e.g. Baldermann et al., Reference Baldermann, Banerjee, Czuppon, Dietzel, Farkaš and Löhr2022).
The first indications for the substantial removal of Na+, Mg2+ and K+ ions and Si(OH)4 from present-day seawater by clay mineral authigenesis were provided by Mackenzie & Garrels (Reference Mackenzie and Garrels1966a). They concluded that element sequestration by reverse weathering balances the riverine influx of the dissolved components from the continents. Since then, extensive field, modelling and laboratory studies have been published that strengthen the evidence for reverse weathering as a key process impacting modern marine element cycles (e.g. Michalopoulos & Aller, Reference Michalopoulos and Aller1995; Michalopoulos et al., Reference Michalopoulos, Aller and Reeder2000; Wallmann et al., Reference Wallmann, Aloisi, Haeckel, Tishchenko, Pavlova and Greinert2008, Reference Wallmann, Geilert and Scholz2023; Baldermann et al., Reference Baldermann, Warr, Letofsky-Papst and Mavromatis2015; Reference Baldermann, Banerjee, Czuppon, Dietzel, Farkaš and Löhr2022; Ehlert et al., Reference Ehlert, Doering, Wallmann, Scholz, Sommer and Grasse2016; Cuadros et al., Reference Cuadros, Andrade, Ferreira, de Moya Partiti, Cohen and Vidal-Torrado2017; Geilert et al., Reference Geilert, Frick, Garbe-Schönberg, Scholz, Sommer and Grasse2023). Although the reverse weathering fluxes of individual elements are relatively small (<10%) compared to the global marine carbon and silicon budgets, clay mineral authigenesis can sequester various chemical elements, such as Na, K, Li, Mg, Fe, Al and Si, implying that the varying rates of clay mineral formation during marine diagenesis can impact global element budgets.
Isson & Planavsky (Reference Isson and Planavsky2018) provided quantitative estimates of the significance of reverse weathering on the modern global silica cycle. They calculated that the oceanic Si output fluxes related to chert formation and reverse weathering reactions account for 10.4 and 0.5 Tmol year–1, respectively. The latter flux is equivalent to 4.6% of the total continental Si input to the oceans by combined riverine, hydrothermal, aeolian and groundwater sources (10.9 Tmol year–1). However, Tréguer et al. (Reference Tréguer, Sutton, Brzezinski, Charette, Devries and Dutkiewicz2021) proposed that the Si output flux due to reverse weathering is much higher than previously calculated, at ∼5 Tmol year–1. This 10-fold difference in the calculated Si output flux reflects the problems associated with model parameterization of reverse weathering reactions occurring in the modern oceans.
More recently, Wallmann et al. (Reference Wallmann, Geilert and Scholz2023) reported on the modern oceanic element fluxes associated with marine silicate weathering and authigenic clay mineral formation. They calculated net fluxes across the sediment–seawater interface in the order of +1.5 Tmol Na year–1, –2.5 Tmol K year–1, –2.0 Tmol Mg year–1, +2.5 Tmol Ca year–1 and +1.9 Tmol Si year–1, where the positive numbers indicate cation release into seawater mainly due to feldspar (e.g. plagioclase) dissolution and the negative numbers denote cation uptake by the precipitation of unspecified authigenic clay minerals and carbonates. However, these estimates did not consider Si burial through biogenic opal precipitation.
More specifically, it has been reported that element sequestration associated with glauconite formation in modern shallow marine settings (0–200 m water depth) sequesters ∼0.06 Tmol year–1 of K, Mg and Al, ∼0.1 Tmol year–1 of Fe and ∼0.3 Tmol year–1 of Si, respectively (see Fig. 4 in Baldermann et al., Reference Baldermann, Banerjee, Czuppon, Dietzel, Farkaš and Löhr2022). This is equivalent to ∼3–6% of the total oceanic K inventory that is delivered from riverine and hydrothermal sources and thus of the same order of magnitude as the K burial flux attributed to authigenic Fe-illite formation in mangrove forests (up to 0.1 Tmol year–1; Cuadros et al., Reference Cuadros, Andrade, Ferreira, de Moya Partiti, Cohen and Vidal-Torrado2017). Although these estimates have a high degree of uncertainty, Li et al. (Reference Li, Liu, Wang, McManus, Haley and Takahashi2022a) proposed that glauconite is an important sink of seawater K (∼0.2 Tmol year–1) based on K isotope measurements of modern marine ‘bulk’ sediments. Regarding the marine Mg cycle, glauconite formation at the marine shelf has been estimated to constitute ∼10–38% of the Mg consumption by authigenic clays forming in the Amazon deltaic sediments (∼0.21 Tmol year–1) and ∼1–3% of the Mg sink that is associated with fresh oceanic crust alteration (Sun et al., Reference Sun, Higgins and Turchyn2016; Huang et al., Reference Huang, Teng, Plank, Staudigel, Hu and Bao2018). Shallow-water glauconite formation may be even more important for the marine Al and Si cycles. It involves ∼27–82% removal of the dissolved riverine Al influx entering the modern oceans (∼0.1 Tmol year–1; Maring & Duce, Reference Maring and Duce1987) and ∼10% removal of the total Si uptake by authigenic clay minerals on a global scale (∼4.7 ± 2.3 Tmol year–1; Rahman et al., Reference Rahman, Aller and Cochran2016; Tréguer et al., Reference Tréguer, Sutton, Brzezinski, Charette, Devries and Dutkiewicz2021). In addition, glauconite formation acts as a significant sink for Fe, sequestering ∼8–33% of the dissolved and particulate riverine fluxes of Fe that enter the ocean (∼0.48–0.86 and ∼0.63 Tmol year–1, respectively; Raiswell, Reference Raiswell2006).
In contrast, the elemental uptake rates by glauconite formation in modern deep-marine sediments are currently poorly constrained, but Fe sequestration at Ocean Drilling Project (ODP) Site 959 (Ivory Coast–Ghana Marginal Ridge) has been estimated to be ∼80 µmol cm–2 kyr–1. Although this oceanic sink of Fe is two orders of magnitude lower than that of modern shelf areas, it is (on average) six times higher than that related to the formation of pyrite in suboxic subsurface sediments 5 m below the seafloor or shallower (Baldermann et al., Reference Baldermann, Warr, Letofsky-Papst and Mavromatis2015). This indicates that glauconite formation in the deep sea may also act as an important Fe sink, in addition to marine Fe-sulfide minerals, which develop under euxinic to anoxic conditions deeper within the sedimentary pile (e.g. Raiswell & Canfield, Reference Raiswell and Canfield2012). As for the modern deep-sea Al, Si, K and Mg cycles, however, glauconite precipitation seems to be less important, which has been attributed to the slower reaction rates at the lower temperatures (<5°C) of deep-sea settings (e.g. Baldermann et al., Reference Baldermann, Warr, Grathoff and Dietzel2013, Reference Baldermann, Warr, Letofsky-Papst and Mavromatis2015, Reference Baldermann, Banerjee, Czuppon, Dietzel, Farkaš and Löhr2022). However, Logvinenko (Reference Logvinenko1982) has reported that modern deep-sea sediments contain ∼1–2 wt.% glauconite or glauconite-smectite on average. In modern calcareous and fossiliferous deep-sea sediments, the glauconite content is even higher, reaching 2–3 wt.% on average, according to Giresse et al. (Reference Giresse, Wiewióra and Grabska2004) and Wigley & Compton (Reference Wigley and Compton2007). These relationships may suggest that the elemental uptake rates of Al, Si, K and Mg by glauconite formation in the deep ocean are currently underestimated.
In hydrothermally influenced sediments, such as at the Galapagos spreading centre, the Mid-Atlantic Ridge, the Crozet Island Archipelago, the Costa Rica Margin, the Pacific–Antarctic Ridge and the Chile Ridge, precipitation of Fe-bearing smectites occurs in the form of nontronite, Fe(III)-montmorillonite and saponite. The formation of these minerals can locally take up ∼1000 µmol Fe cm–2 kyr–1, suggesting that smectite authigenesis at thermally active sites of the deep sea can also constitute an important (but locally restricted) Fe sink in the modern oceans (e.g. Chester, Reference Chester2000). Additional reverse weathering reactions occur via the low- and high-temperature interaction of fresh oceanic crust and seawater in the vicinity of mid-ocean ridge sites (e.g. Elderfield & Schultz, Reference Elderfield and Schultz1996; Higgins & Schrag, Reference Higgins and Schrag2015; Voigt et al., Reference Voigt, Pearce, Fries, Baldermann and Oelkers2020). These have been proposed to contribute strongly to the elemental and isotopic composition of seawater, as indicated by in situ Rb-Sr dating of celadonite forming within veins and amygdales ∼20 Myr after ocean crust accretion (Laureijs et al., Reference Laureijs, Coogan and Spence2021a). That study also concluded that widespread celadonite formation occurring in ocean floor sediments and ophiolite lavas could act as an important sink for seawater K (but also Al, Si, Li, B, Mg and Fe), but without providing robust mass balance constraints to support their hypothesis. This could not be achieved due to the incomplete sample recovery associated with ocean-floor drilling and related challenges in quantifying the heterogeneous distribution of K-rich phases in the upper oceanic crust (Laureijs et al., Reference Laureijs, Coogan and Spence2021b).
While the influence of reverse weathering on major element cycles is becoming increasingly recognized, especially with regard to often easily extractable (millimetre-sized) and quantifiable glauconite and celadonite grains, there are few constraints on the impacts of reverse weathering on the cycling of minor and trace elements in the ocean (e.g. Tostevin & Ahmed, Reference Tostevin and Ahmed2023). This is largely due to the difficulty of separating and purifying typically micrometre-scale authigenic clay minerals, which are usually closely intergrown with detrital clay minerals of similar size. It has been proposed, based on porewater analyses, that the modern marine rare earth element (REE) cycle is largely controlled by the balance of REE release from detrital clay mineral dissolution versus uptake of REEs by authigenic clay minerals, Fe/Mn-(hydr)oxides and phosphates (e.g. Abbott et al., Reference Abbott, Löhr and Trethewy2019). Recent analyses of authigenic glauconite separates from the West African continental margin and comparison to the detrital clay mineral-rich muds in which they form support this model (Bayon et al., Reference Bayon, Giresse, Chen, Rouget, Gueguen and Moizinho2023), whereby detrital clay mineral dissolution coupled with REE uptake by marine authigenic clay minerals is responsible for the large sedimentary flux of REEs in the modern oceans (e.g. Abbott et al., Reference Abbott, Haley, McManus and Reimers2015, Reference Abbott, Löhr and Trethewy2019; Haley et al., Reference Haley, Du, Abbott and McManus2017). In addition, the work of Bayon et al. (Reference Bayon, Giresse, Chen, Rouget, Gueguen and Moizinho2023) suggests that glauconite formation from a detrital clay mineral precursor assemblage is associated with net sinks of Mo, Cr, V and Rb but represents a source of U, Li, Co, Ni, Hf, Zr, Th and Ba. Similarly, authigenic smectites in a Cenozoic sequence from the Ross Sea (Antarctica) have been reported to be enriched in V, Cr, Co, Ni and Sc compared to co-occurring detrital clay minerals (Setti et al., Reference Setti, Marinoni and López-Galindo2004). Other recent work suggests that authigenic clay mineral formation may act as a Be sink (Bernhardt et al., Reference Bernhardt, Oelze, Bouchez, von Blanckenburg, Mohtadi, Christl and Wittmann2020). In addition, clay mineral synthesis experiments suggest that greenalite may have been an important sink of Cu, V and Zn in the Precambrian oceans (Tostevin & Ahmed, Reference Tostevin and Ahmed2023).
Isotope proxies used to trace clay mineral reactions
The development of multi-collector inductively coupled plasma mass spectrometry (MC-ICP-MS) and thermal ionization mass spectrometry (TIMS) marked a significant step towards the improved measurement of the isotopic composition of minerals. These instruments are used to accurately measure elements and their isotopes down to ppm and ppb concentrations with light to large atomic masses (Li, B versus Ca, Fe, Mg, Sr, Zn, etc.) in a suite of components, such as carbonates, phosphates and silicates. These analytical techniques allowed the establishment of a range of new tools and proxy signals to identify and quantify (bio)geochemical processes occurring in Earth’s (surface) settings, such as chemical weathering reactions, element cycles and fluid–mineral interactions (e.g. Pogge von Strandmann et al., Reference Pogge von Strandmann, Burton, James, van Calsteren, Gislason and Sigfusson2008; Oelkers et al., Reference Oelkers, Benning, Lutz, Mavromatis, Pearce and Plümper2015; Schott et al., Reference Schott, Mavromatis, Fujii, Pearce and Oelkers2016; Andrews et al., Reference Andrews, Pogge von Strandmann and Fantle2020; Chanda et al., Reference Chanda, Kohli, Teng and Fantle2023; Farkaš et al., Reference Farkaš, Wallmann, Mosley, Staudigel, Zheng, Leyden, Anbar and Weis2025). While the elemental and isotopic compositions of carbonates have long been used to reconstruct conditions prevailing at the time of their formation (i.e. palaeo-pH, temperature, redox conditions and growth rate), and thus seawater chemistry over time (e.g. Eiler, Reference Eiler2007), applying such methods to determine palaeo-environmental information from authigenic and detrital clay minerals is less well established. This is, among other factors, due to: (1) the lack of well-constrained fractionation factors for clay mineral–fluid systems and substantial isotopic differences between adsorbed/exchangeable and structurally incorporated constituents (e.g. Köster et al., Reference Köster, Williams, Kudejova and Gilg2019; Li et al., Reference Li, Liu, Wang, McManus, Haley and Takahashi2022a); (2) the difficulties in synthesizing well-crystallized and close to pure clay minerals at ambient temperature (e.g. Kloprogge, Reference Kloprogge1998; Baldermann et al., Reference Baldermann, Stamm, Farkaš, Löhr, Ratz, Letofsky-Papst and Dietzel2024); and (3) the challenges associated with isolating authigenic clay minerals from phyllosilicates of detrital origin (e.g. Zhang et al., Reference Zhang, Zhou, Lin, Tong and Yu2010; Tosca & Masterson, Reference Tosca and Masterson2014; Han et al., Reference Han, Löhr, Abbott, Baldermann, Farkaš and McMahon2022a, Reference Han, Löhr, Abbott, Baldermann, Shields and Cui2024; Bayon et al., Reference Bayon, Giresse, Chen, Rouget, Gueguen and Moizinho2023).
Nevertheless, field and experimental studies using the isotopic composition of clay minerals to reconstruct marine element cycles and weathering/alteration pathways occurring in various sedimentary environments are currently on the increase. This is because marine authigenic clay minerals (1) are probably more resistant to post-depositional alteration compared to the more reactive carbonates, (2) incorporate more elements of interest (e.g. K, Si and Li) and in greater abundance than carbonates (which may also be affected by as-yet unknown vital effects), allowing for the development of more new proxies (Li et al., Reference Li, Liu, Wang, McManus, Haley and Takahashi2022a, Reference Li, Liu, Wang, Hu, Suzuki and Yoshimura2022b), (3) represent important sinks for many elements, meaning that the isotopic characterization of authigenic clay minerals can potentially better constrain modern and past marine elemental budgets via isotope mass balance approaches (e.g. Zheng et al., Reference Zheng, Beard, Neuman, Fahnestock, Bryce and Johnson2022), and (4) are datable through conventional K-Ar and 40Ar-39Ar analysis or advanced in situ Rb-Sr analysis by laser ablation collision cell inductively coupled plasma mass spectrometry (LA-ICP-MS/MS; e.g. Kelley, Reference Kelley2002; Clauer et al., Reference Clauer, Zwingmann, Liewig and Wendling2012; Hogmalm et al., Reference Hogmalm, Zack, Karlsson, Sjöqvist and Garbe-Schönberg2017; Scheiblhofer et al., Reference Scheiblhofer, Moser, Löhr, Wilmsen, Farkaš and Gallhofer2022). This is particularly the case for glauconite, for which new isotope proxies are actively being developed (Fig. 9), taking advantage of the relative ease with which typically millimetre-sized glauconite pellets can be separated from their host sediments and purified using standard sieving, acid leaching and magnetic separation techniques or can be directly targeted for in situ analyses.
Illustration showing potential environmental geochemical and isotopic proxies recorded in glauconite and surrounding sediments. Red text indicates primary proxies of high significance; blue text indicates secondary proxies of lower significance (i.e. these are often influenced by other environmental controls and therefore difficult to interpret). Ap = apatite; Dol = dolomite; Ilt = illite.
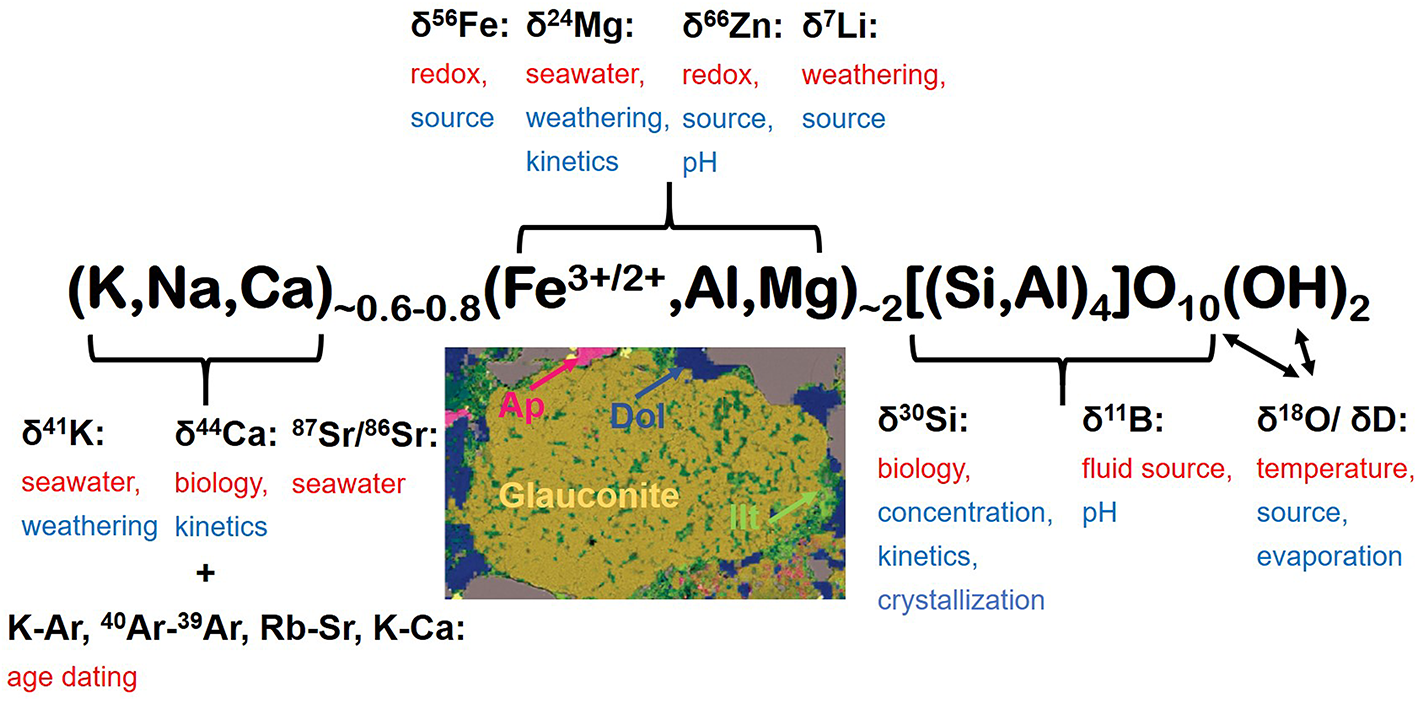
Clay minerals, as hydrous phyllosilicates, contain water (i.e. δ18O and δD isotopic systems) that can be (1) adsorbed on the external surface sites, (2) weakly bound in the interlayer sheet in the case of swellable clay minerals and (3) more strongly bound as hydroxyls in the octahedra. Whereas the first two types of water can isotopically exchange with ambient fluids within minutes to days, structurally bound water frequently shows slow isotope exchange rates depending on temperature and the type of clay mineral (e.g. Clauer et al., Reference Clauer, Williams and Fallick2014). Importantly, the re-equilibration of H isotopes takes place faster at lower temperatures than that of O isotopes, so that the δ18O and δD signatures of clay minerals can provide insights into the nature of mineral reactions (e.g. Schön et al., Reference Schön, Mittermayr, Leis, Mischak and Dietzel2016). For example, the temperature of smectite formation, its post-depositional (re)equilibration/transformation into illite and the kinetics of isotopic exchange between glauconite, host sediment and pore fluid can be resolved (e.g. Savin & Epstein, Reference Savin and Epstein1970).
Stable silicon isotopes (δ30Si) offer a sensitive proxy for studying dissolution–precipitation reactions of silicate minerals in marine sediments (e.g. Rahman et al., Reference Rahman, Aller and Cochran2017). Light Si isotopes (28Si) are preferentially taken up over heavy Si isotopes (30Si) from the pore fluid during precipitation of authigenic clay minerals, shifting pore fluid δ30Si to higher values, often exceeding seawater values (e.g. Ehlert et al., Reference Ehlert, Doering, Wallmann, Scholz, Sommer and Grasse2016; Geilert et al., Reference Geilert, Frick, Garbe-Schönberg, Scholz, Sommer and Grasse2023). Elevated pore fluid δ30Si is thus commonly inferred to reflect authigenic clay mineral formation. However, present constraints on marine authigenic clay mineral δ30Si derive almost entirely from sequential leaching approaches (e.g. Pickering et al., Reference Pickering, Cassarino, Hendry, Wang, Maiti and Krause2020; Huang et al., Reference Huang, Sun, Somelar, Kirsimäe, Pickering and Kim2023), with inherent ambiguity related to the potential dissolution of non-target phases, such as detrital clay minerals. Alternative estimates of authigenic clay mineral δ30Si based on isotope mass balance are helpful (e.g. Rahman & Trower, Reference Rahman and Trower2023) but are impacted by uncertainties in the present-day marine Si cycle. More direct constraints from in situ laser ablation analysis of authigenic clay minerals (e.g. Geilert et al., Reference Geilert, Frick, Garbe-Schönberg, Scholz, Sommer and Grasse2023) or conventional solution-based analyses of authigenic clay mineral separates (e.g. glauconite pellets; Geilert et al., Reference Geilert, Frick, Abbott and Löhr2024) have the potential to substantially reduce these uncertainties and therefore greatly improve quantification of benthic Si fluxes, alkalinity and cation budgets related to marine silicate alteration (e.g. Wallmann et al., Reference Wallmann, Aloisi, Haeckel, Tishchenko, Pavlova and Greinert2008; Reference Wallmann, Geilert and Scholz2023; Geilert et al., Reference Geilert, Frick, Abbott and Löhr2024; Farkaš et al., Reference Farkaš, Wallmann, Mosley, Staudigel, Zheng, Leyden, Anbar and Weis2025). For example, the δ30Si signatures of a bulk sediment rich in diatoms, its porewater, the terrigenous fraction and the authigenic Fe-smectite or glauconite-smectite, obtained by laser ablation, were recently used to demonstrate rapid marine silicate alteration and related clay mineral formation during a coastal El Niño extreme weather event at the Peruvian margin (Geilert et al., Reference Geilert, Frick, Garbe-Schönberg, Scholz, Sommer and Grasse2023). This case study illustrated the fact that clay mineral authigenesis can impact the marine element cycle and the CO2 cycle on timescales of weeks to months.
The fractionation behaviour of boron isotopes (δ11B) of clay minerals depends on temperature, B speciation, fluid δ11B composition, pH, fluid/rock ratio and (re)equilibration time between the solid and fluid phase (e.g. Williams et al., Reference Williams, Hervig and Hutcheon2001; Deyhle & Kopf, Reference Deyhle and Kopf2005). Boron isotopes have been used for reconstructing the fluid source(s) involved in the formation of ash-derived bentonite deposits in marine and terrestrial low-temperature environments (e.g. Köster et al., Reference Köster, Williams, Kudejova and Gilg2019; Clauer et al., Reference Clauer, Williams and Uysal2022). Moreover, B concentrations and δ11B signatures of diagenetically modified and rejuvenated glauconites can record either ancient seawater δ11B composition or incorporation of organic-derived light 10B into glauconite during diagenesis, which enables reconstruction of the evolutionary history of glauconite (Środoń et al., Reference Środoń, Williams, Szcerba, Zaitseva, Bojanowski and Marciniak-Maliszewska2023).
Another isotope system that has seen great interest in recent years is the stable potassium system (δ41K; e.g. Farkaš et al., Reference Farkaš, Wallmann, Mosley, Staudigel, Zheng, Leyden, Anbar and Weis2025). The balance between silicate weathering and marine clay mineral authigenesis acts as an important control on Earth’s long-term carbon cycle. Since these processes also represent important influences on the marine K cycle (e.g. Li et al., Reference Li, Liu, Wang, McManus, Haley and Takahashi2022a) and are thought to be associated with substantial K isotopic fractionations (e.g. Li et al., Reference Li, Li, Beard, Raymo, Wang, Chen and Chen2019b, Reference Li, Liu, Hu, Teng and Hu2021c; Teng et al., Reference Teng, Hu, Ma, Wei and Rudnick2020), the δ41K signature of seawater has the potential to become an important new tool for understanding long-term changes to the carbon cycle (e.g. Li et al., Reference Li, Li and Beard2019c; Hu et al., Reference Hu, Teng, Plank and Chauvel2020). Preferential uptake of the lighter K isotope (39K) during authigenic clay mineral formation or during adsorption processes by clay minerals in the marine sediment is likely to be a key factor explaining the surprisingly heavy isotopic composition of present-day seawater (∼0.12‰ ± 0.07‰), considering the K isotope difference between modern seawater and Bulk Silicate Earth of ∼0.6‰ (e.g. Li et al., Reference Li, Liu, Wang, McManus, Haley and Takahashi2022a). The large K isotopic difference between seawater and Earth’s silicate crust (i.e. upper continental crust; –0.44‰ ± 0.05‰; Huang et al., Reference Huang, Teng, Rudnick, Chen, Hu, Liu and Wu2020) remains enigmatic because modern seawater shows a significant excess of the heavy 41K isotope compared to the main input fluxes of K into the ocean. We note, however, that no direct measurements of modern or recent authigenic clay mineral δ41K have been published to date (only from bulk sediment), which is due to the difficulties in isolating these authigenic clay minerals from the host sediment. However, pellet-forming authigenic clay minerals, such as glauconite and celadonite (e.g. Santiago Ramos et al., Reference Santiago Ramos, Coogan, Murphy and Higgins2020), are promising targets and have the potential to become an important archive for the reconstruction of seawater δ41K changes over time. Alternative sedimentary archives, which may also record seawater δ41K, include biogenic carbonates and K-rich evaporite salts, but these are either convoluted by vital effects and low K contents or show a very patchy temporal distribution (e.g. Li et al., Reference Li, Liu, Wang, McManus, Haley and Takahashi2022a). Direct measurement of authigenic clay mineral δ41K is thus a key research priority that can be used to assess the relative impact on seawater δ41K caused by changes in continental weathering intensity (e.g. Hu et al., Reference Hu, Teng, Plank and Chauvel2020; Li et al., Reference Li, Liu, Wang, McManus, Haley and Takahashi2022a), the rates of low-temperature seafloor alteration and the magnitude of authigenic clay mineral formation in marine sediments (e.g. Wang et al., Reference Wang, Li, Li, Tian, Koefoed and Zheng2021).
We have already mentioned the important role of reverse weathering in the marine REE budget, with the sedimentary REE fluxes and the related composition of the marine bottom waters being controlled by the dissolution of detrital clay minerals versus the uptake of REEs by authigenic clay minerals (e.g. Haley et al., Reference Haley, Du, Abbott and McManus2017; Abbott et al., Reference Abbott, Löhr and Trethewy2019; Bayon et al., Reference Bayon, Giresse, Chen, Rouget, Gueguen and Moizinho2023). This has important implications for the REE concentration and the Nd isotopic composition of water masses, which have been shown to evolve over distance, location and time (e.g. Abbott et al., Reference Abbott, Löhr and Trethewy2019). It has been proposed that authigenic clay minerals, such as glauconite, may be promising recorders of bottom water or porewater Nd isotopic compositions (e.g. Giresse et al., Reference Giresse, Bayon, Tallobre and Loncke2021), with potential applications for reconstructing changes to ocean circulation over time. Similarly, radiogenic Sr isotope (87Sr/86Sr) signatures have been used to track the evolution of authigenic clay minerals, showing that 87Sr/86Sr of detrital clay minerals is progressively lost as they are replaced by authigenic clay minerals, causing 87Sr/86Sr to approach a composition that is more similar to that of seawater (e.g. Clauer et al., Reference Clauer, Keppens and Stille1992). This observation is also commonly used to justify anchoring of Rb-Sr isochrons using expected seawater Sr isotope values (e.g. Redaa et al., Reference Redaa, Farkaš, Gilbert, Collins, Löhr and Vasegh2023).
Other promising isotope systems that have only rarely been applied to marine authigenic clay minerals include (1) magnesium (δ26Mg) and calcium (δ44Ca) for obtaining insights into biogeochemically coupled processes, weathering rates and marine element budgets (e.g. Dunlea et al., Reference Dunlea, Murray, Santiago Ramos and Higgins2017; Huang et al., Reference Huang, Shen, Wang, Ma, Li and Zhou2024), (2) lithium (δ7Li) to quantify continental palaeo-weathering regimes versus rates of oceanic crust alteration (e.g. Zhang et al., Reference Zhang, Awolayo, Nightingale and Tutolo2022a; Yin et al., Reference Yin, Wei, Pogge von Strandmann, Wei, Pogge von Strandmann Philip and Lechte2023) and (3) zinc (δ66Zn), iron (δ56Fe) and chromium (δ53Cr) to constrain palaeo-redox conditions during clay mineral precipitation (cf. Fig. 9).
Another (re)emerging application for authigenic clay minerals that is likely to gain importance aims to determine the absolute depositional ages of marine sedimentary rocks. The most widely used radiometric techniques for obtaining absolute depositional ages are presently U-Pb zircon dating of volcanic ash (e.g. Linnemann et al., Reference Linnemann, Ovtcharova, Schaltegger, Gärtner, Hautmann and Geyer2018), 40Ar/39Ar dating of lava flows (e.g. Jourdan et al., Reference Jourdan, Féraud, Bertrand, Kampunzu, Tshoso, Watkeys and Le Gall2005) and Re-Os dating of organic-rich black shales (e.g. Rooney et al., Reference Rooney, Strauss, Brandon and Macdonald2015; Millikin et al., Reference Millikin, Strauss, Halverson, Bergmann, Tosca and Rooney2022). Because these target lithologies are often restricted to specific tectonic or environmental settings, dating sedimentary sequences remains among the most challenging tasks in geochronology. Glauconite geochronology is a potential alternative approach where ash beds or black shales are absent. In fact, ∼40% of all ages used to constrain the geological timescale of the last 250 Myr up until the mid-1990s were obtained from glauconite (Smith et al., Reference Smith, Evenson, York and Odin1998). Although glauconite bulk separates have long been dated using the Rb-Sr, K-Ca, K-Ar and 40Ar-39Ar techniques, frequent mismatches with independent chronostratigraphic constraints (e.g. Selby, Reference Selby2009; Cecil & Ducea, Reference Cecil and Ducea2011) mean that glauconite geochronology is no longer widely employed. Erroneous ages have been attributed to factors including the presence of detrital Rb- and K-bearing phases in glauconite separates, the presence of younger and isotopically distinct clay mineral overgrowths, as well as post-depositional disruption or resetting of the Rb-Sr isotope system by exposure to increased temperatures and/or fluid–rock interactions (e.g. Rafiei et al., Reference Rafiei, Löhr, Alard, Baldermann, Farkaš and Brock2023). Various sample preparation and screening approaches, based on improved mineral separation and more aggressive chemical cleaning, have been developed to address these problems, but with varying degrees of success (e.g. Derkowski et al., Reference Derkowski, Środoń, Franus, Uhlík, Banaś and Zieliński2009).
The advent of in situ Rb-Sr and K-Ca dating offers a promising new approach that has the potential to resolve many of these limitations, leading to a recent revival in authigenic clay mineral-based geochronology (e.g. Zack & Hogmalm, Reference Zack and Hogmalm2016; Hogmalm et al., Reference Hogmalm, Zack, Karlsson, Sjöqvist and Garbe-Schönberg2017; Laureijs et al., Reference Laureijs, Coogan and Spence2021a; Redaa et al., Reference Redaa, Farkaš, Gilbert, Collins, Löhr and Vasegh2023). Unlike traditional solution-based Rb-Sr dating, which requires the dissolution of bulk sample powders followed by wet chemical separation of Rb and Sr, reaction cell mass spectrometry resolves the spectral overlap of, for example, 87Rb and 87Sr via the addition of a reaction gas (e.g. N2O) to a reaction cell (e.g. Hogmalm et al., Reference Hogmalm, Zack, Karlsson, Sjöqvist and Garbe-Schönberg2017; Gorojovsky & Alard, Reference Gorojovsky and Alard2020; Redaa et al., Reference Redaa, Farkaš, Gilbert, Collins, Löhr and Vasegh2023). In the case of the Rb-Sr system, Rb is measured based on mass while Sr isotopes are measured as mass-shifted oxides (i.e. produced by reaction with the gas), allowing quantitative online separation of 87Rb and 87Sr. When combined with recent advances in sedimentary petrography (Rafiei et al., Reference Rafiei, Löhr, Baldermann, Webster and Kong2020; Han et al., Reference Han, Löhr, Abbott, Baldermann, Farkaš and McMahon2022a) this approach permits rapid in situ Rb-Sr dating of carefully screened glauconite or celadonite grains, which opens up a range of new applications (e.g. Laureijs et al., Reference Laureijs, Coogan and Spence2021a; Scheiblhofer et al., Reference Scheiblhofer, Moser, Löhr, Wilmsen, Farkaš and Gallhofer2022; Rafiei et al., Reference Rafiei, Löhr, Alard, Baldermann, Farkaš and Brock2023; Redaa et al., Reference Redaa, Farkaš, Gilbert, Collins, Löhr and Vasegh2023). For Precambrian mudrocks rich in authigenic clay minerals, in situ dating of marine authigenic illite has also shown great promise (e.g. Subarkah et al., Reference Subarkah, Blades, Collins, Farkaš, Gilbert and Löhr2022, Reference Subarkah, Nixon, Gilbert, Collins, Blades and Simpson2024).
Clay minerals in geoengineering: porewater alkalinity enhancement
A major challenge facing today’s society is to limit anthropogenically induced global warming to 1.5°C above the pre-industrial value (e.g. United Nations Framework Convention on Climate Change, 2015). As a result, various strategies for CO2 removal at an industrial scale have been proposed over the past three to four decades, such as carbon capture and storage or carbon capture and utilization methods.
A novel and sustainable solution to decrease atmospheric CO2 concentrations is to bind carbon in near-surface marine sediments and directly in the oceans (e.g. Morrow et al., Reference Morrow, Thompson, Anderson, Batres, Buck and Dooley2020; Meier et al., Reference Meier, Rickels, Quaas and Traeger2022) by enhancing the chemical weathering rates of silicates and the resulting alkalinity of marine porewaters (e.g. Bellamy et al., Reference Bellamy, Chilvers, Vaughan and Lenton2012; Bach et al., Reference Bach, Gill, Rickaby, Gore and Renforth2019; Fakhraee et al., Reference Fakhraee, Li, Planavsky and Reinhard2023; Hartmann et al., Reference Hartmann, Suitner, Lim, Schneider, Marín-Samper and Arístegui2023; Iglesias-Rodríguez et al., Reference Iglesias-Rodríguez, Rickaby, Singh and Gately2023; Dale et al., Reference Dale, Geilert, Diercks, Fuhr, Perner, Scholz and Wallmann2024; Marín-Samper et al., Reference Marín-Samper, Arístegui, Hernández-Hernández, Ortiz, Archer, Ludwig and Riebsell2024). As described earlier, silicate mineral weathering achieves this by releasing cations and binding CO2 (i.e. HCO3– and CO22– ions), thus facilitating the uptake of atmospheric CO2 by the ocean (e.g. Feng et al., Reference Feng, Koeve, Keller and Oschlies2017). The spreading of crushed mafic rocks, such as basalt, and mafic minerals (here: forsterite, Mg2SiO4) in marine settings is increasingly considered in geoengineering to stabilize Earth’s climate through long-term binding and storage of atmospheric CO2 following Equation 10 (e.g. Wood & Kleppa, Reference Wood and Kleppa1981; Taylor et al., Reference Taylor, Quirk, Thorley, Kharecha, Hansen and Ridgwell2016):
During this irreversible dissolution reaction, 1 mol of olivine sequesters 4 mol of CO2, releasing stoichiometric amounts of HCO3– and dissolved metal (cat)ions to the marine pore fluid. However, this process can subsequently trigger marine clay mineral authigenesis and hence reverse weathering (Fig. 10). Depending on the species formed and the reaction pathway, these reactions may lead to the recycling or release of CO2, meaning that the net carbon mass balance remains unknown. Although such a CO2 sequestration technique still requires verification and optimization of the total CO2 binding capacity at individual test sites, as well as estimation of the costs and potential environmental risks, this approach has great potential for application on a large scale (e.g. Montserrat et al., Reference Montserrat, Renforth, Hartmann, Leermakers, Knops and Meysman2017).
(a) Field-scale (carbonate) alkalinity enhancement test site situated in the pioneer vegetation zone of salt marshes in Portugal. (b) Illustration showing mineral reactions and element transfer at the interface between (1) seawater and substrate (deployment: reactive mafic rock rich in olivine), (2) substrate and biologically active top layer and (3) top layer and marsh sediments. Olivine dissolution consumes CO2 and liberates carbonate alkalinity and elements, causing smectite formation. (c) Evolution of the alkalinity profiles in sediment porewater with time at control sites without deployments (marked by the geogenic background curve with a low alkalinity) and at deployment sites (marked by the green curve with a ∼2.5-fold higher alkalinity due to enhanced olivine weathering). Note that the deployment sites approach the geogenic background alkalinity level in a relatively short timeframe, which suggests that reverse weathering takes place (i.e. CO2 release).
Based on CO2 mass balance calculations (Equation 10), olivine deployments in high-energy marine–coastal environments have been proposed (e.g. Meysman & Montserrat, Reference Meysman and Montserrat2017). On main shipping routes, olivine release from vessel tanks is currently under consideration in the open ocean (e.g. Köhler et al., Reference Köhler, Abrams, Völker, Hauck and Wolf-Gladrow2013). Laboratory and modelling studies have demonstrated the great potential of alkalinity enhancement by chemical weathering of silicates, with recognition of mineral-specific CO2 uptake rates, nutrient release rates and ecological effects (e.g. Monserrat et al., Reference Montserrat, Renforth, Hartmann, Leermakers, Knops and Meysman2017; Bach et al., Reference Bach, Gill, Rickaby, Gore and Renforth2019; Fuhr et al., Reference Fuhr, Geilert, Schmidt, Liebetrau, Vogt, Ledwig and Wallmann2022).
Despite the significant interest, field-scale experiments in marginal marine environments (under natural conditions) are still scarce, and the counteracting role of any reverse weathering reaction (e.g. Wallmann et al., Reference Wallmann, Geilert and Scholz2023) remains largely unknown. In a recent study, He & Tyka (Reference He and Tyka2023) proposed that at favourable near-coastal sites, such as in north Madagascar, California, Brazil, Peru and locations close to the Southern Ocean, a carbon uptake efficiency of ∼0.6–0.8 mol CO2 per mol of alkalinity could be reached after 3–4 years. Moreover, Mendes et al. (unpublished data) are currently conducting alkalinity enhancement experiments by deploying picro-basalt and basanite in the pioneer vegetation zone of salt marshes in Portugal and monitoring their impacts on the ecosystem and porewater chemistry (Fig. 10). Preliminary results indicate that: (1) porewater alkalinity increases by a factor of three within a few days of the deployments, maintaining an alkalinity that is ∼10–20% higher compared to that at control sites (without deployments) after 6 months; and (2) the release of dissolved Si(OH4), Mg2+ and Fe2+ ions from olivine, pyroxene and feldspar dissolution triggers the formation of smectite at as-yet unknown rates, confirming the importance of reverse weathering reactions in marine settings (e.g. Han et al., Reference Han, Löhr, Abbott, Baldermann, Voigt and Yu2022b; Mendes et al., unpublished data).
However, before this type of geoengineering can be applied to marine settings on a larger scale (e.g. gigaton scale; He & Tyka, Reference He and Tyka2023), potential side effects and consequences have to be identified and carefully monitored, such as: (1) the formation of distinct chemical and microbial environments on deployed mineral particles and grain surfaces; (2) the response of the biological community to the nutrients released by the deployments; (3) the evolution of porewater geochemistry; (4) the long-term links between increased carbonate alkalinity, porewater pH and carbonate mineral saturation indices; (5) the rate of CO2 re-equilibration with the atmosphere; and (6) the impact of smectite authigenesis on the physicochemical properties of the marine sediment (e.g. Simon et al., Reference Simon, Grossart, Schweitzer and Ploug2002; van der Jagt et al., Reference Van der Jagt, Friese, Stuut, Fischer and Iversen2018; Hartmann et al., Reference Hartmann, Suitner, Lim, Schneider, Marín-Samper and Arístegui2023).
Clay minerals as slow-release fertilizers
Clay minerals that formed during continental chemical weathering, as well as during reverse weathering, are increasingly being used in agriculture for soil conditioning and soil environmental improvement. The addition of these minerals helps mitigate the accumulation of excess nutrients in soils and water bodies worldwide caused by the application of conventional, easily soluble (K-N-P-based) fertilizers. The overuse of common fertilizers leads to burning or nitrate poisoning of plants and to the increased eutrophication of freshwater bodies, as well as to increased emissions of the greenhouse gas nitrous oxide. Hence, modern agricultural practices focus on the efficient use of sustainable ‘green’ fertilizer technologies (e.g. Tilman et al., Reference Tilman, Fargione, Wolff, D’Antonio, Dobson and Howarth2001). One solution to this challenge is the use of functional fertilizers, which enable the controlled or slow release of nutrients (e.g. Sharma, Reference Sharma1979; Liu et al., Reference Liu, Price, Bennett, Maxwell, Zhao, Walker and Bunt2022). Slowly dissolving silicate-based fertilizers, such as glauconite (or greensand), aim to deliver nutrients to plants in a targeted (and directed) manner, thus increasing crop yields while reducing environmental pollution. These controlled-release products typically consist of nutrients (e.g. N, P or other macro- and micronutrients) that are loaded onto a clay mineral-based carrier (e.g. glauconite), whose properties determine the release rate of the nutrients (e.g. Fu et al., Reference Fu, Wang, Chen, Huang and Chen2018; Rahman et al., Reference Rahman, Haque and Khan2021; Duan et al., Reference Duan, Jiang, Chen, Li, Ma and Song2023; Moradi et al., Reference Moradi, Babapoor, Ghanbarlou, Kalashgarani, Salahshoori and Seyfaee2023). Moreover, it has been found that, in the long term, additional secondary clay mineral assemblages may form, such as ferruginous smectite, illite-smectite and metal (hydr)oxides (e.g. Pestitschek et al., Reference Pestitschek, Gier, Essa and Kurzweil2012), which help to maintain the favourable physicochemical properties of the soil, such as a greater pH buffer capacity, an elevated organic matter content and increased water retention and ion-exchange capacities (e.g. Li et al., Reference Li, Zhou, Fiore and Yu2019a).
Clay minerals such as smectites (e.g. Pereira et al., Reference Pereira, da Cruz, Solomon, Le, Cavigelli and Ribeiro2015; Ren et al., Reference Ren, Zhao, Cui, Wang, Zhang and Guo2024), kaolinite (e.g. Lei et al., Reference Lei, Cagnetta, Li, Qu, Li, Zhang and Huang2018; AlShamaileh et al., Reference AlShamaileh, Alrbaihat, Moosa, Abu-Afifeh, Al-Fayyad, Hamadneh and Al-Rawajfeh2022), halloysite (e.g. Maximov et al., Reference Maximov, Dasi, Kalinina, Ruban, Pokidko and Rudmin2023) and glauconite (e.g. Vakal et al., Reference Vakal, Yanovska, Vakal, Artyukhov, Shkola and Yarova2020; Rudmin et al., Reference Rudmin, Makarov, López-Quirós, Maximov, Lokteva and Ibraeva2023b) have been investigated in the search for cost-effective and environmentally friendly nutrient-carrier materials. Current research has focused on glauconite because this mineral can act as a carrier for nitrogen (N) and as a natural source of K. For example, glauconite loaded with ammonium (NH4+) ions, carbamide (urea) or ammonium dihydrogen phosphate has been proposed as a promising slow-release fertilizer (e.g. Rudmin et al., Reference Rudmin, Makarov, López-Quirós, Maximov, Lokteva and Ibraeva2023b). It was found that the adsorption of NH4+ ions into the micropores of glauconite or in the interlayer sites of smectite within interstratified glauconite-smectite occurs immediately after the preparation of the mixtures. It was suggested that the degree of NH4+ adsorption by globular glauconite depends on the distribution and abundance of surface-accessible pores and on the smectite content. Importantly, such glauconite fertilizers exhibited prolonged release times for N and K, making them suitable for diverse slow-release fertilizing purposes (e.g. Rudmin et al., Reference Rudmin, Makarov, López-Quirós, Maximov, Lokteva and Ibraeva2023b). These findings advance our understanding of the influence of environmental conditions on reverse weathering. Future studies should make use of isotopes to quantify the rates of silicate weathering versus near-surface clay mineral neoformation and also explore how the application of large volumes of slow release clay-based fertilizers to soils may eventually, after chemical degradation and transport, impact reverse weathering processes in marine environments.
Conclusion and perspectives
Reverse weathering processes produce a suite of authigenic clay minerals in marine sediments over comparatively short geological timescales. Despite the generally sluggish reaction kinetics of near-surface clay mineral authigenesis, there is increasing evidence from modelling, isotopic and experimental studies for the occurrence of faster reactions and substantial element sequestration (K, Si, Mg, REEs, Li, B, etc.) associated with reverse weathering. This takes place in shallow-marine and deep-marine depocentres, as well as in marine hydrothermal surroundings. A complex interplay occurs between global climate change and elemental fluxes to the oceans, which are intimately related to the rates of continental and seafloor chemical weathering, the evolution of seawater biogeochemistry and elemental uptake fluxes associated with marine clay mineral sinks. Novel analytical approaches used to quantify clay mineral reactions are advancing, which will allow better estimations of the efficiency of reverse weathering. These advances are driving applications and proposals for clay mineral-related geoengineering for CO2 sequestration and for slow-release fertilizers as soil amendments. However, further progress in understanding the links between silicate weathering and reverse weathering is needed, which requires continued field studies, advanced laboratory approaches and numerical modelling. Such advancement underlines the great relevance of reverse weathering reactions on past and present marine element cycles and Earth’s climate.
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1180/clm.2024.27.
Author contributions
AB, SB and AC wrote the first manuscript draft. All authors contributed to the writing and revision of the paper.
Acknowledgements
We greatly acknowledge J. Cuadros (Natural History Museum, London, UK) for his review invitation and M. Dietzel and F.M. Stamm (Graz University of Technology, Graz, Austria) for scientific discussions. The valuable comments from the associate editor, J. Cuadros, and from two anonymous reviewers are also acknowledged.
Financial support
This study was partly funded by the NAWI Graz Geocenter (Graz University of Technology) and the ARC Discovery Project (DP210100462; grant to AB and SCL) titled ‘Glauconite: Archive Recording the Timing and Triggers of Cambrian Radiation’. Open access funding was from Graz University of Technology.
Conflicts of interest
The authors declare no conflicts of interest.











