Depression affects more than 300 million people worldwide.1 Evidence supports the use of both pharmacological and psychological therapies,Reference Gartlehner, Dobrescu and Chapman2,Reference Cuijpers, Noma, Karyotaki, Vinkers, Cipriani and Furukawa3 but guidelines recommend initiating antidepressant medications for adults with moderate to severe depression.Reference Gartlehner, Dobrescu and Chapman2–7 Antidepressants are commonly used: in England alone, 79.4 million antidepressant prescriptions were issued to 7.87 million people in 2020–2021.8
In randomised controlled trials (RCTs), 26% of people on average discontinue antidepressants for any reason, including inefficacy (i.e. acceptability), while approximately 10% stop them for side effects (i.e. safety) that cannot be tolerated (i.e. tolerability) after 2 months of treatment.Reference Cipriani, Furukawa and Salanti9 Of those who continue taking antidepressants, only about 50% respond to first-line treatment (i.e. efficacy).Reference Rush, Trivedi and Wisniewski10 However, RCTs often exclude individuals with concurrent illness and medication use, despite the high prevalence of multimorbidity (68%) and polypharmacy (63–98%) in those who will subsequently require treatment outside the trial setting (i.e. real-world patients).Reference Tan, Papez and Chang11 Exclusion of these large populations may limit the generalisability and transportability of RCT findings to clinical practice.Reference Rothwell12 This may contribute to an ‘efficacy-effectiveness gap’, where efficacy relates to the performance of drugs in RCTs, and effectiveness to real-world settings.Reference Eichler, Abadie, Breckenridge, Flamion, Gustafsson and Leufkens13 Recently, real-world and RCT data on the effectiveness of antipsychotics for schizophrenia have shown high congruency.Reference Katona, Bitter and Czobor14 It is currently unclear, however, whether the same holds for antidepressants in people with depression.
This study aims to assess the overall and comparative acceptability, tolerability, safety and effectiveness of antidepressants in a large, representative sample of adults diagnosed with depressive disorders. Furthermore, we compare real-world estimates with pooled, summary-level data available from equivalent RCTs,Reference Cipriani, Furukawa and Salanti9 to assess the degree of agreement between the two sources of evidence, and the existence of an efficacy-effectiveness gap in antidepressants.
Method
The study methods are reported in full in the published protocol.Reference De Crescenzo, Garriga and Tomlinson15
Population
We extracted an open cohort of individuals registered for at least 12 months with eligible general practices between 1 January 1998 and 15 August 2020 from the QResearch primary care research registry, version 45. QResearch is a large, consolidated database derived from the anonymised health records from general practices using the Egton Medical Information Systems (EMIS) of over 35 million people in England (www.QResearch.org). We included people aged 18–100 years who had a newly recorded diagnosis of a depressive disorder according to their ‘Read codes’, using case definitionsReference De Crescenzo, Garriga and Tomlinson15 that have been applied in earlier studies.Reference Coupland, Dhiman and Morriss16,Reference Coupland, Hill and Morriss17 Participants included those who had been started on any licensed antidepressant monotherapy and were followed up for 12 months.
We excluded people with a diagnosis of bipolar disorder or schizophrenia spectrum disorder; those with a diagnosis of postpartum depression made within 180 days before or up to 180 days after the first diagnosis of depression; those with more than one antidepressant prescribed at baseline; those who took antidepressants more than 60 days before the diagnosis of depression; those with an antipsychotic or mood stabiliser prescription; and those with a previous antidepressant prescription or diagnosis of depression before the cohort entry date.
The final cohort for analysis included an exposed group of participants on each antidepressant monotherapy.
Drug exposure
The exposure of interest was the prescription of any one of 30 antidepressants licensed in the UK – from the full list previously published.Reference De Crescenzo, Garriga and Tomlinson15 Fluoxetine was utilised as the index comparator, as this drug was the first selective serotonin reuptake inhibitor (SSRI) approved by regulatory agencies internationally and it has been the most used active comparator in antidepressant clinical trials.Reference Cipriani, Furukawa and Salanti9
Information was extracted from all prescriptions of antidepressants issued during the 12-month follow-up according to previously validated criteria.Reference Coupland, Dhiman and Morriss16,Reference Coupland, Hill and Morriss17 The duration of each prescription was calculated by dividing the number of tablets prescribed by the number of tablets to be taken each day. If information on ‘tablets per day’ was missing or not sufficiently detailed, we estimated the duration of the prescription considering the most used prescription, i.e. once daily. Participants were considered as continually exposed to an antidepressant for periods with no gaps of more than 30 days between the end of one prescription and the beginning of the next.
Outcomes
According to the protocol, the outcomes of interest were measured at 2 months (i.e. short term) and 12 months (i.e. long term) after the initial prescription of antidepressants, as follows:
(a) Acceptability of treatment was measured as the proportion of people who dropped out because of any cause. We defined treatment dropout as follows:Reference De Crescenzo, Garriga and Tomlinson15 (i) a person had a gap of more than 30 days between the end of a prescription of an antidepressant and the start of the next prescription; (ii) a person switched to another antidepressant (i.e. switch strategy); (iii) a person was prescribed an additional antidepressant (i.e. combination strategy); (iv) a person was prescribed a mood stabiliser or an antipsychotic (i.e. augmentation strategy).
(b) Tolerability of treatment was measured as the proportion of people who dropped out because of any adverse event.
(c) Safety of treatment was measured as the proportion of people with at least one adverse event, irrespective of whether they dropped out. We selected ‘Read codes’ for 67 adverse events that have been identified in clinical trials as severe and frequently associated with antidepressants,Reference Tomlinson, Efthimiou, Boaden, New, Mather and Salanti18 or shown to be important to patients, carers and healthcare professionals in a recent large survey.Reference Chevance, Ravaud and Tomlinson19
(d) To compare real-world estimates with outcome data from RCTs, effectiveness of treatment at the timepoint closest to 2 and 12 months was measured as response (i.e. 50% reduction on the Patient Health Questionnaire (PHQ)-9Reference Kroenke, Spitzer and Williams20) and remission (i.e. scoring less than 5 on the PHQ-9), by dichotomising the PHQ-9 scores using validated methods. Other depression scales were transformed to PHQ-9 scores, which range from 0 to 27, following Wahl et al.Reference Wahl, Löwe and Bjorner21 If PHQ-9 data were missing at follow-up, we used the PHQ-9 score closest to a specific follow-up timepoint within a predefined time window. This approach ensured that the most relevant data informed the analysis, while still allowing for flexibility in the timing of follow-up assessments. By using the value closest to the specified timepoint, we aimed to provide a more accurate representation of the individual's status at that follow-up period.
Confounders
Based on previous studies of antidepressants in QResearch,Reference Coupland, Dhiman and Morriss16,Reference Coupland, Hill and Morriss17 we considered several confounding baseline variables, i.e. possible risk factors for the outcomes of interest, potentially also associated with the likelihood of receiving a particular antidepressant treatment. Suspected confounders included: gender, age, baseline severity, depression subtype, year of diagnosis, body mass index (BMI), smoking status, daily alcohol intake, ethnicity, Townsend quintile, regions of England, comorbidities and use of other drugs. See Table 4 for the detailed list of confounders.
Statistical analyses
We described the study population by reporting baseline characteristics and antidepressant use. We used multiple imputation by chained equations to impute data when actual values were not available.Reference Rubin22 For each imputation, we generated ten imputed datasets and combined coefficient estimates across these using Rubin's rule. All confounding and outcome variables were included in the multiple imputation process (see Supplementary Appendix 1 available at https://doi.org/10.1192/bjp.2024.194).
To estimate relative effects between antidepressant monotherapy and fluoxetine (index comparator), we used multivariable logistic regression models. These models were clustered by general practices and adjusted for all confounders. We used a separate model for each of the four outcomes, i.e. acceptability, tolerability, safety and effectiveness, and estimated adjusted-odds ratios (aOR) with 99% CIs (99% CIs). We used intention-to-treat (ITT) analyses, i.e. we analysed each person according to the drug they were prescribed, irrespective of whether they received it to the end. We excluded people who switched antidepressants prior to entering the study cohort. However, for those who switched antidepressants during follow-up, they were analysed as part of that treatment group regardless of subsequent changes in their medication. We visualised results using a Kilim plot, where rows indicated drugs, columns indicated outcomes and each cell showed effects versus the reference, for the corresponding outcome; cells were coloured according to P-values, to visualise the strength of statistical evidence against the null hypothesis of no difference versus the reference.Reference Seo, Furukawa, Veroniki, Pillinger, Tomlinson and Salanti23
To assess the robustness of our real-world observational estimates of relative (i.e. versus fluoxetine) treatment effects, we examined them in comparison with findings obtained from the largest network meta-analysis of RCTs to date (Group of Researchers Investigating Specific Efficacy of Individual Drugs for Acute depression: GRISELDA).Reference Cipriani, Furukawa and Salanti9 We compared our estimates for all outcomes at 2 months (i.e. short-term) with estimates for acceptability, tolerability, response and remission available. We could not compare safety data as these were not published in GRISELDA. For this analysis, we used only the antidepressants that were studied in both databases, and we focused on the most frequently used antidepressants in QResearch for which we could develop a logistic regression model, i.e. amitriptyline, trazodone, citalopram, escitalopram, paroxetine, sertraline, duloxetine, mirtazapine and venlafaxine. We calculated for each drug the ratio-of-odds ratio (ROR) versus fluoxetine: real-world odds ratio of each drug versus fluoxetine, compared with RCTs odds ratio of the same drug versus fluoxetine, together with 95% CI Stata MP 16.0 software24 (StataCorp, Texas, USA; see https://www.stata.com/support/faqs/resources/citing-software-documentation-faqs/) was used for the statistical analyses.
Institutional review board (IRB) approval
This project was independently peer-reviewed by the QResearch scientific committee and approved on 1 July 2019 with reference 18/EM/0400.
Registration
QResearch scientific committee reference 18/EM/0400, 1 July 2019.
Results
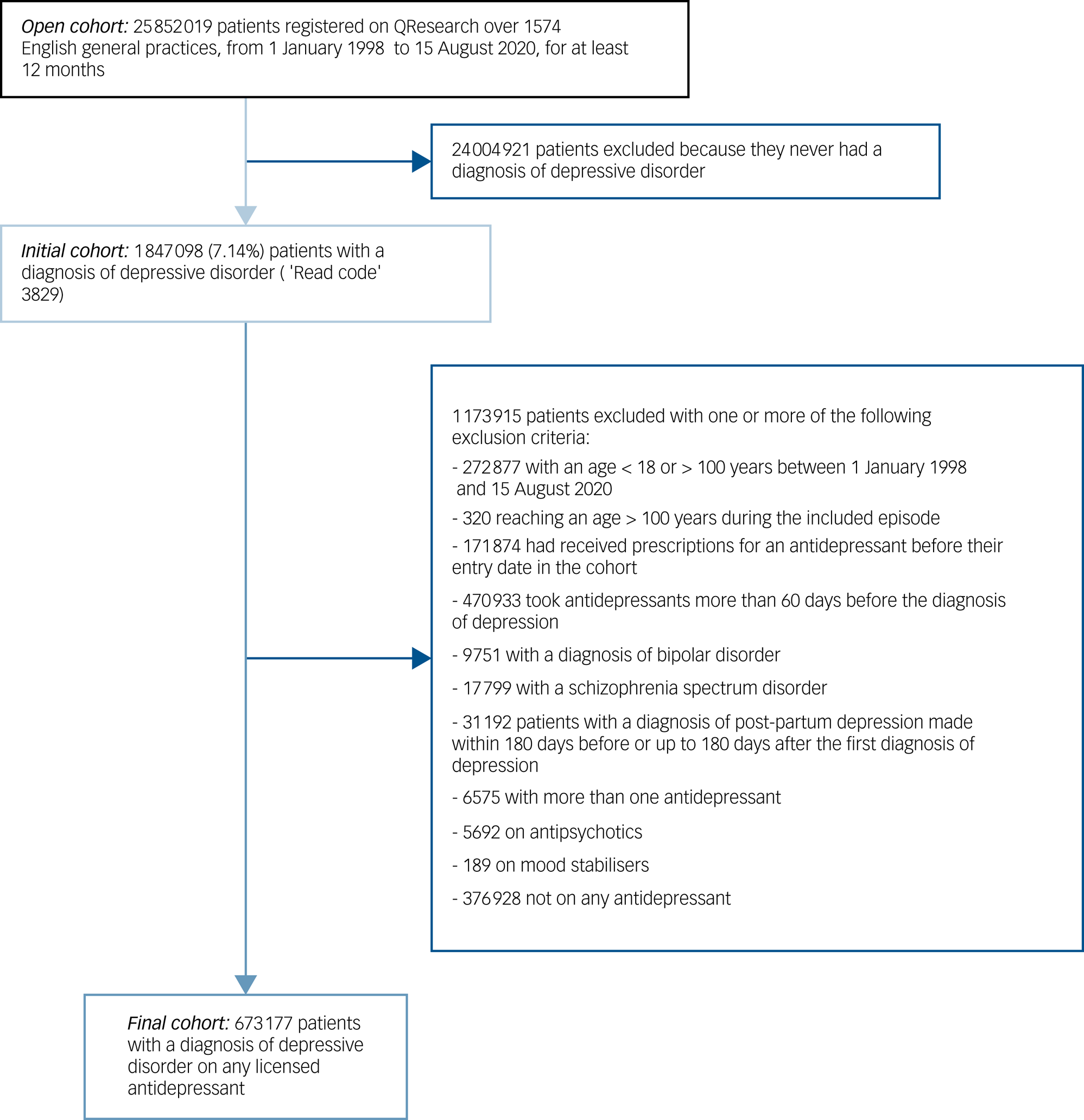
From an open cohort of 25 852 019 people registered on QResearch over 1574 English general practices from 1 January 1998 to 15 August 2020 for at least 12 months, we identified an initial cohort of 1 847 098 participants (7.1%) with a first incident diagnosis of depressive disorder. Following the application of exclusion criteria, we obtained a final cohort of 673 177 people with a diagnosis of depressive disorder on any licensed antidepressant monotherapy. Of note, we excluded from the analysis 376 928 people who were not on antidepressants, but they were included in the multiple imputation process as they all carry information about missing values. The flowchart of the study cohort is shown in Fig. 1.
Flowchart of the study cohort.
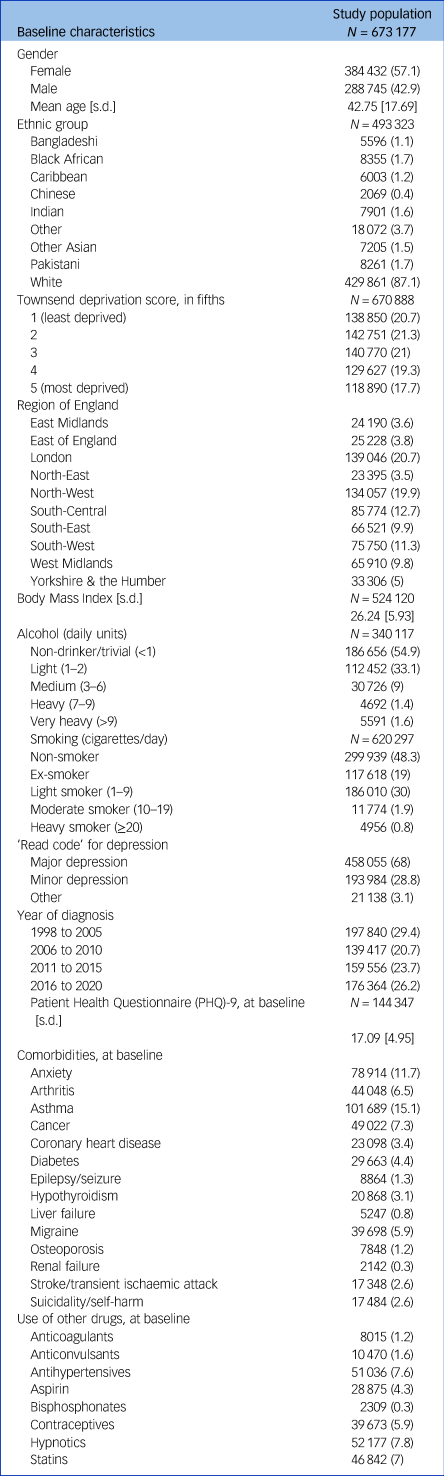
Baseline characteristics of the study population (N = 673 177) are in Table 1. Most participants were females (57.1%), with a mean age of 42.8 years (s.d. 17.7). Mean PHQ-9 score at baseline was 17.1 (s.d. 5.0), consistent with moderately severe depression.Reference Kroenke, Spitzer and Williams20
Baseline characteristics of the study population. Values are numbers (percentages) unless stated otherwise
Exposures for people starting an antidepressant are in Table 2. Most were prescribed SSRIs (85.7%), while a minority were prescribed other antidepressants (7.4%) and tricyclic antidepressants (TCAs) (6.9%), and only few were on monoamine oxidase inhibitors (MAOIs) (0.01%). The four antidepressants most prescribed, i.e. citalopram (36.1%), fluoxetine (22.3%), sertraline (20.2%) and mirtazapine (5.8%) accounted for 84.4% of total prescriptions.
Antidepressants exposure of the study population, divided by antidepressant category. Values are numbers (percentages) unless stated otherwise

MAOIs, monoamine oxidase inhibitors; SSRIs, selective serotonin reuptake inhibitors; TCAs, tricyclic antidepressants.
a. Amoxapine, isocarboaxazid, tranylcypromine and tryptophan had fewer than 5 participants and were therefore excluded from the table to comply with QResearch data management guidelines.
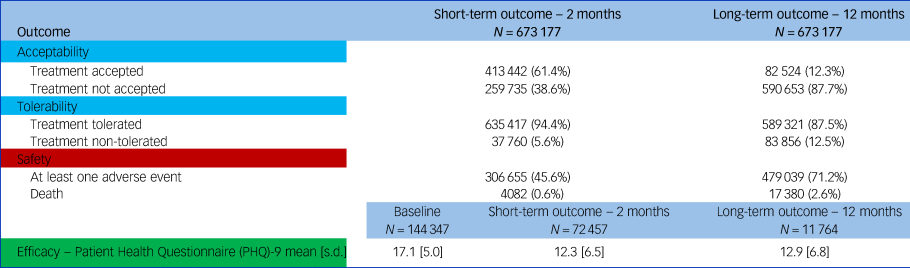
Study outcomes are divided between short term (i.e. 2 months) and long term (i.e. 12 months), and descriptive statistics for them are reported in Table 3.
Descriptive statistics of outcomes at 2 months (short term) and at 12 months (long term)
Short-term outcomes – 2 months
At 2 months, antidepressant treatment was discontinued by 259 735 individuals (38.6%) because of any cause, of which 37 760 (5.6%) were because of an adverse event. In terms of safety, the number of people experiencing at least one adverse event was 306 655 (45.6%), and 4082 (0.6%) individuals died. For effectiveness, PHQ-9 scores decreased from ‘moderately severe’ (17.1, s.d. 5.0) to ‘moderate’ (12.3, s.d. 6.5) – Table 3.
Adjusted analyses comparing any antidepressant against fluoxetine at 2 months are in Table 4. Fluoxetine was more acceptable than all TCAs, trimipramine, paroxetine, duloxetine, venlafaxine, mirtazapine, reboxetine and trazodone. The aORs ranged between 1.09 (99% CI 1.05 to 1.14) for paroxetine and 3.04 (99% CI 1.08 to 8.51) for mianserin. Only citalopram was associated with fewer dropouts than fluoxetine (aOR 0.95, 99% CI 0.92 to 0.97).
Adjusteda analyses comparing any antidepressant with fluoxetine at 2 months (short term) and 12 months (long term)
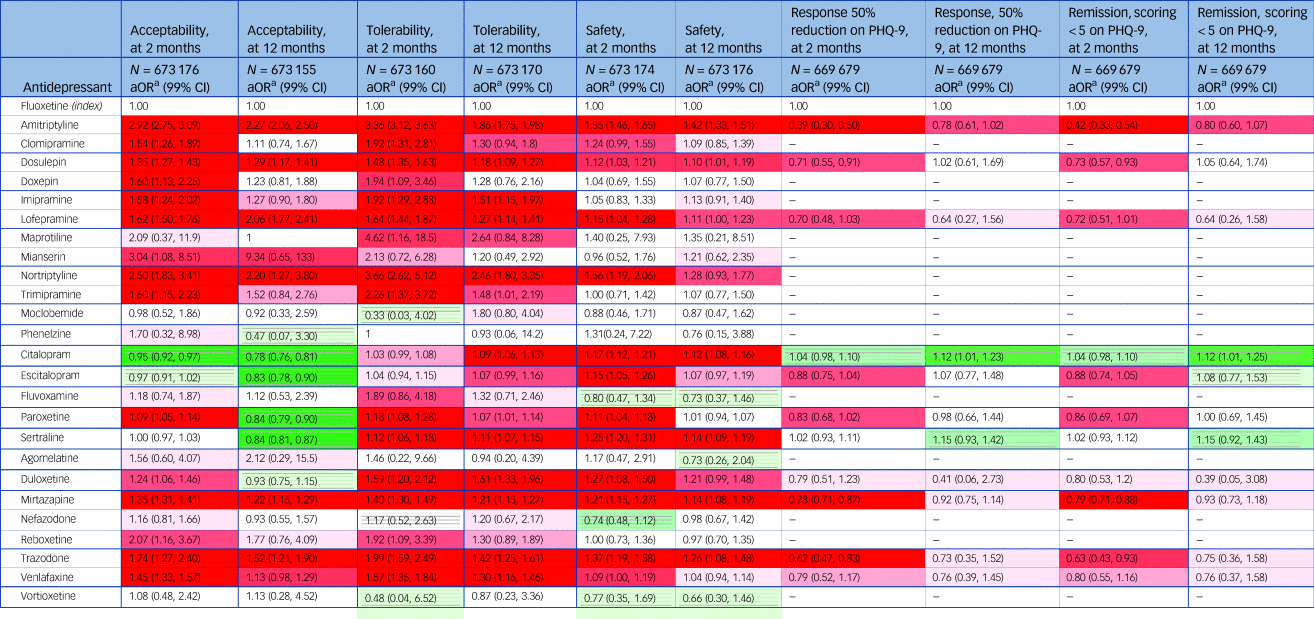
a. The numbers in each cell correspond to the estimated adjusted odds ratios (aORs) for each outcome and antidepressant treatment compared to fluoxetine. This was adjusted for clusters in general practices and for gender, age, baseline severity, depression subtype, year of diagnosis, body mass index (BMI), smoking status, daily alcohol intake, ethnicity, Townsend quintile, region of England, comorbidities at baseline (coronary heart disease, stroke/transient ischaemic attack, diabetes, cancer, epilepsy/seizures, hypothyroidism, osteoarthritis/rheumatoid arthritis, suicidal ideation/behaviour or self-harm, asthma/chronic obstructive pulmonary disease [COPD] , osteoporosis, liver disease, renal disease, anxiety) and use of other drugs at baseline (anticonvulsants, hypnotics/anxiolytics, antihypertensive drugs, aspirin, statins, anticoagulants, non-steroidal anti-inflammatory drugs, bisphosphonates, oral contraceptives/hormone replacement therapy). Amoxapine, isocarboaxazid, tranylcypromine and tryptophan, which had fewer than five patients, were excluded from the table to comply with QResearch data management guidelines. The shading corresponds to the strength of statistical evidence regarding the effects versus fluoxetine. A dark cell with background lines indicates strong evidence (P < 0.001) that the corresponding drug performs better than fluoxetine for the corresponding outcome. Conversely, a dark cell without lines indicates strong evidence (P < 0.001) that the drug performs worse than fluoxetine. Colours closer to white indicate lack of evidence on whether the drug performs better or worse than fluoxetine (see legend below).
Legend 
Fluoxetine was more tolerable than all TCAs but mianserin (aOR 2.13, 99% CI 0.72 to 6.28), and also more tolerable than trimipramine, paroxetine, sertraline, duloxetine, venlafaxine, mirtazapine, reboxetine and trazodone. The aORs ranged between 1.12 (99% CI 1.06 to 1.18) for sertraline and 4.62, (99% CI 1.16 to 18.46) for maprotiline.
Fluoxetine was found to be safer than amitriptyline, dosulepin, lofepramine and nortriptyline among the TCAs, as well as mirtazapine, trazodone, venlafaxine and duloxetine. Further, fluoxetine was found to be the safest of all SSRIs except for fluvoxamine, for which less information was available (aOR 0.80, 99% CI 0.47 to 1.34).
For effectiveness, amitriptyline, dosulepin, trazodone and mirtazapine were less efficacious in terms of response and remission than fluoxetine, with aORs ranging from 0.39 (99% CI 0.30 to 0.50) and 0.42 (99% CI 0.33 to 0.54) for amitriptyline, and 0.78 (99% CI 0.71 to 0.89) and 0.79 (99% CI 0.71 to 0.88) for mirtazapine. No drug was found to be more efficacious.
Long-term outcomes – 12 months
At 12 months, antidepressant treatment was discontinued by 590 653 individuals (87.7%), of which 83 856 (12.5%) were because of adverse events. In terms of safety, the number of people experiencing at least one side-effect was again high at 479 039 (71.2%), and all-cause mortality was recorded for 14 172 (2.1%) people. For effectiveness, PHQ-9 scores decreased from ‘moderately severe’ at baseline (17.1, s.d. 5.0) to ‘moderate’ (12.9, s.d. 6.8) – Table 3.
Adjusted analyses comparing any antidepressant against fluoxetine at 12 months are in Table 4.
Fluoxetine was more acceptable than amitriptyline, dosulepin, lofepramine, nortriptyline, mirtazapine and trazodone, with aORs ranging between 2.27 (99% CI 2.06 to 2.50) for amitriptyline and 1.22 (99% CI 1.16 to 1.29) for mirtazapine. Four antidepressants were more acceptable than fluoxetine at 12 months: citalopram (aOR 0.78, 99% CI 0.76 to 0.81), escitalopram (aOR 0.83, 99% CI 0.78 to 0.90), paroxetine (aOR 0.84, 99% CI 0.79 to 0.90) and sertraline (aOR 0.84, 99% CI 0.79 to 0.90).
Fluoxetine was more tolerable than amitriptyline, dosulepin, imipramine, lofepramine and nortriptyline among the TCAs, as well as trimipramine, duloxetine, venlafaxine, mirtazapine and trazodone. All SSRIs but escitalopram and fluvoxamine were less tolerable than fluoxetine. The aORs ranged between 2.46 (99% CI 1.80 to 3.35) for nortriptyline and 1.07 (99% CI 1.01 to 1.14) for paroxetine.
Fluoxetine was safer than amitriptyline, dosulepin, citalopram, sertraline, mirtazapine and trazodone with aORs ranging between 1.42 (99% CI 1.33 to 1.51) for amitriptyline and 1.10 (99% CI 1.01 to 1.19) for dosulepin.
For effectiveness, we only found evidence that citalopram was more efficacious than fluoxetine for both response (aOR 1.12, 99% CI 1.01 to 1.23) and remission (aOR 1.12, 99% CI 1.01 to 1.25).
Comparison of real-world versus RCTs data
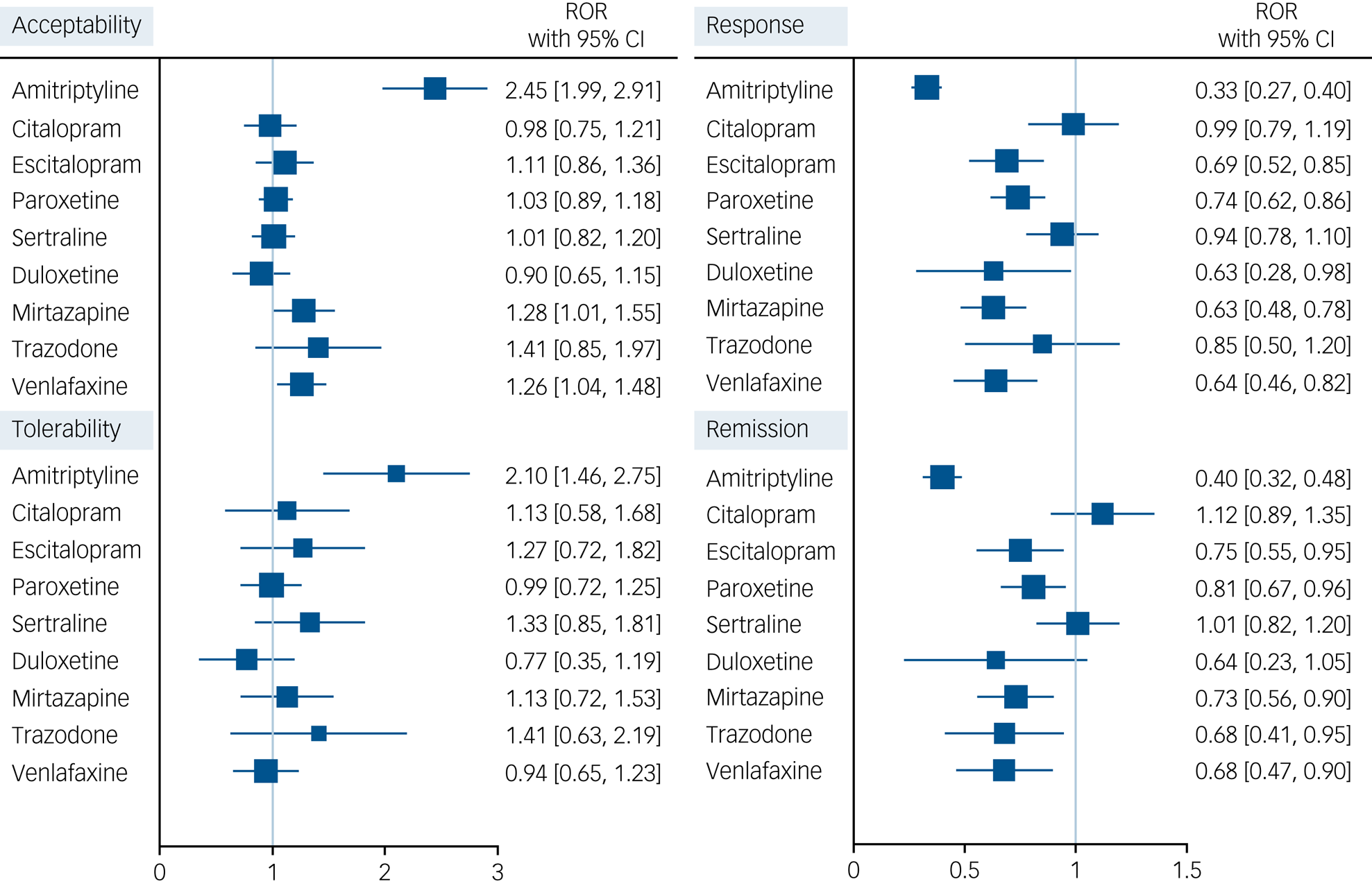
We calculated RORs with 95% CI estimated at 2 months for each antidepressant (fluoxetine as index comparator), comparing real-world with randomised data in Fig. 2. For acceptability, amitriptyline, mirtazapine and venlafaxine fared better in RCTs than in real-world data, with RORs ranging from 1.26 (95% CI 1.06 to 1.49) for venlafaxine to 2.45 (95% CI 2.03 to 2.95) for amitriptyline. Amitriptyline also showed to be better tolerated in RCTs than in the real world (ROR 2.10, 95% CI 1.55 to 2.84). For response and remission, amitriptyline, escitalopram, paroxetine, mirtazapine and venlafaxine were superior in RCTs, with RORs ranging from 0.33 (95% CI 0.27 to 0.40) and 0.40 (95% CI 0.32 to 0.49) for amitriptyline to 0.74 (95% CI 0.63 to 0.87) and 0.81 (95% CI 0.68 to 0.97) for paroxetine.
Forest plot showing ratio-of-odds ratios (ROR) with 95% CI for each antidepressant, with fluoxetine as index comparator, comparing real-world versus clinical trials (Group of Researchers Investigating Specific Efficacy of Individual Drugs for Acute Depression [GRISELDA] study) data. For acceptability and tolerability, a ROR >1 for drug X indicates that the effect of X versus fluoxetine is larger in randomised controlled trials (RCTs) as compared with the real world. For response and remission, a ROR <1 for drug X indicates that the effect of X versus fluoxetine is larger in RCTs as compared with the real world.
Discussion
This study investigated the comparative effects of antidepressants in a cohort of 673 177 participants with a diagnosis of depressive disorder, in a large community-based sample from UK primary care, followed up for 12 months in real-world conditions. The size and detail of available data allowed us to control for numerous potential confounders and to assess several outcomes, including antidepressant acceptability, tolerability, safety (overall and for individual adverse events) and effectiveness (response and remission).
In terms of acceptability, antidepressants dropout rates were high (38.6% at 2 months, 87.7% at 12 months). In RCTs, based on a network meta-analysis of 522 trials, 26% of people on average discontinued antidepressants because of any cause at 2 months.Reference Cipriani, Furukawa and Salanti9 In the Sequenced Treatment Alternatives to Relieve Depression (STAR*D) trial, discontinuation rate in the short term was 27%.Reference Rush, Trivedi and Wisniewski10 Some studies performed in similar naturalistic settings are in line with our results, with discontinuation rates around 43% at 6 weeks and 73% at 6 months,Reference Demyttenaere, Enzlin and Dewé25-Reference Olfson, Marcus, Tedeschi and Wan28 although other epidemiology surveys have reported much lower rates.Reference Samples and Mojtabai29 It is also possible that some of the recorded discontinuations from antidepressant treatment would have occurred in individuals whose depressive symptoms had resolved – such could occur in both clinical trials (e.g. individuals who are lost to follow-up) and observational studies. In our study, this occurrence might also be a consequence of including people with diagnostic codes for ‘minor depression’, as done in similar previous studies on QResearch.Reference Coupland, Dhiman and Morriss16,Reference Coupland, Hill and Morriss17 However, this should not be a major issue in this study of primary care patients, because clinical guidelines followed by general practitioners (GPs) in the UK advise continuing antidepressant medication for at least six months after symptomatic remission.7 The fact that antidepressants dropout rates were higher in real-world data than in trials could be because of a variety of factors. In naturalistic settings, people are often prescribed medication with limited focus on ensuring adherence with treatment; conversely, RCTs tend to enrol participants who are highly motivated to comply with medication,Reference Sackett30 which might contribute to lower external validity of trial results. In the routine clinical practice world, high discontinuation rates of antidepressants can have serious consequences for people, as they are associated with a high risk of relapse.Reference Lewis, Marston and Duffy31 The difficulty of accessing treatment is another significant factor contributing to the high discontinuation rates of antidepressants. In the UK National Health System, access to antidepressant medications through general practitioners can be challenging for some individuals because of limited consultation time and stigma, as well as prescription restrictions, both due to reimbursement constraints and primary care guidelines that limit the choice of antidepressants.Reference Castaldelli-Maia, Scomparini and Andrade32 Our observed low acceptability for antidepressants therefore suggests the importance of improving shared decision-making between patients and healthcare professionals to increase access to appropriate antidepressant treatments and reduce discontinuation rates in routine clinical practice.Reference Ford, Thomas, Byng and McCabe33
Antidepressant treatment was stopped because of adverse events in a non-negligible proportion of participants (5.6% at 2 months, 12.5% at 12 months). Most suffered at least one adverse event (45.6% at 2 months, 71.2% at 12 months), and 2.1% of them died during the 12-month follow-up. Our findings on the tolerability and safety of antidepressants complement the body of research carried out to date using QResearch. Previous work has focused on the association between use of antidepressants and important adverse events such as falls, fractures, upper gastrointestinal bleed, road traffic accidents, adverse drug reactions, all-cause mortality,Reference Coupland, Hill and Morriss17 epilepsy and seizure,Reference Hill, Coupland and Morriss34 myocardial infarction, stroke or transient ischaemic attack, arrhythmia,Reference Coupland, Hill and Morriss35 and suicide and attempted suicide or self-harm,Reference Coupland, Hill and Morriss36 as well as referral for an adverse event in older people.Reference Coupland, Dhiman and Morriss16 Nevertheless, adverse events appeared to be a minor reason for discontinuation, which is in line with RCT data where 10% of people discontinue antidepressants due to any adverse event (i.e. tolerability) within 2 months.Reference Cipriani, Furukawa and Salanti9
For effectiveness, a reduction in the PHQ-9 score was observed, with average depression severity decreasing from ‘moderately severe’ at baseline to ‘moderate’ at 2 months and 12 months. There was an overall improvement, but moderate depressive symptoms were still present at both 2 months and 12 months. This result is in line with the modest effect sizes for antidepressants seen in RCTs.Reference Cipriani, Furukawa and Salanti9
We further assessed the credibility of our findings obtained using real-world data by comparing them with results from a large data-set of clinical trials, GRISELDA. Clinical trials are the most reliable source of information when comparing relative treatment effects,Reference Kennedy-Martin, Curtis and Faries37 but their rigorous experimental setting and restrictive inclusion/exclusion criteria may potentially limit their generalisability to clinical practice.Reference Rothwell12 For example, RCTs often exclude individuals with chronic multimorbidity and those who are taking multiple medications due to the need to control for variables that could affect study outcomes.Reference Tan, Papez and Chang11 If such characteristics are strong effect modifiers, i.e. if they modify relative treatment effects, then the transportability of RCT findings to real-world settings may be jeopardised. On this basis, the comparison of the findings from our study with those of the clinical trials data-set GRISELDA supports our conclusions while also providing novel insights. In our analyses, we found that several of the most used antidepressants (i.e. trazodone, citalopram, escitalopram, paroxetine, sertraline and duloxetine, compared with fluoxetine) showed a similar effect size in both data-sets, with citalopram and sertraline displaying very similar results in the real world and in RCTs. However, amitriptyline, mirtazapine and venlafaxine showed a far better profile in the short-term treatment of depression in RCTs compared with real-world data, where they fared poorly. These conflicting findings could be due to several factors, including selective inclusion and exclusion criteria in RCTs, variable placebo effect observed in antidepressant trials,Reference Furukawa, Cipriani and Atkinson38 unmeasured confounding in real-world studies, missing outcomes being not random in the real world, and the low number of RCTs directly comparing these drugs in GRISELDA.Reference Cipriani, Furukawa and Salanti9 Furthermore, it should be noted that our cohort study included individuals using first-line antidepressant monotherapy, while amitriptyline, mirtazapine and venlafaxine are more commonly used in primary care as second-line treatments, usually after one or more courses of SSRIs. Nevertheless, the overlap of effect sizes between observational and randomised data strengthens the available body of evidence on antidepressant effects. It also highlights how large, detailed and rigorous observational studies, which follow a methodology akin to clinical trials,Reference Hernán39 can complement evidence provided by the latter while addressing some of their limitations. Future research should move from a population-based perspective, toward a personalised-based approach to tailor antidepressants to people's individual characteristics.
Strengths and limitations
This study includes the largest and most detailed cohort of people with depressive disorder investigated under real-world conditions to assess the comparative effectiveness of antidepressant monotherapy. The use of an active index comparator (i.e. fluoxetine) fills an important evidence gap and has been previously advocated.Reference Cipriani, Ioannidis and Rothwell40 Despite its observational nature, this study focuses on clinically relevant outcomes that are similarly evaluated in RCTs,Reference Cipriani, Furukawa and Salanti9 while also including an in-depth evaluation of side effects and a longer follow-up (i.e. 12 months). Moreover, a representative cohort of adults with depression in England could be examined, including people with comorbidities and concomitant medication use, who are usually excluded from clinical trials. Including such individuals constitutes a significant added value of this study, whose results aim to be generalised to the wider population (i.e. high external validity). However, people with certain comorbid conditions such as cognitive impairment (though unlikely to be common for our sample with a mean age of ~42 years) may show responses to antidepressants that are substantially different from those of others.
The main limitation of this study involves a weaker internal validity compared with RCTs, primarily because of potential indication bias. RCTs that aim to estimate causal effects of interventions are mostly at lower risk of bias when compared with observational investigations.Reference Dragioti, Solmi and Favaro41 In our study, we attempted to minimise biases by only including individuals with a diagnosis of depression and by controlling for potentially confounding variablesReference Coupland, Dhiman and Morriss16,Reference Coupland, Hill and Morriss17 recorded in considerable detail on QResearch.Reference Hippisley-Cox, Vinogradova, Coupland and Pringle42 Nevertheless, we cannot exclude the possibility of important differences in unmeasured effect modifiers between participants prescribed different antidepressant medications. Such differences would introduce bias to the associations between the drug exposures and the outcomes, and perhaps explain some of the observed differences with RCT data. Channelling bias, which occurs when treatments with similar indications are prescribed to different patient segments based on a different perceived prognosis, must also be considered.Reference Acton, Willis and Hennessy43 Although all participants in our sample had been newly diagnosed with depression, those with lower depressive symptoms scores on the PHQ-9 may have been treated differently from those with higher scores. Second, the effectiveness of some antidepressants could not be assessed in view of the low number of PHQ-9 scores available at baseline and at study endpoints for these drugs. The PHQ-9 is a validated self-rating scale, which is reliable for measuring depressive symptoms over time but not good for diagnosis.Reference Kroenke, Spitzer and Williams20 The availability of other depression scales more commonly employed in RCTs of antidepressants (e.g. Montgomery-Åsberg Depression Scale or Hamilton Depression Rating Scale) would allow better comparison and stronger consistency between real-world and RCTs data, but these observer-rating scales are not used in real-world practice.
Moreover, as pre-specified in the published protocol,Reference De Crescenzo, Garriga and Tomlinson15 we used 10 imputed datasets to account for missing data. This number of imputations is the same as previous studies using QResearch;Reference Vinogradova, Coupland and Hippisley-Cox44,Reference De Giorgi, De Crescenzo, Cowen, Harmer and Cipriani45 however, we acknowledge that a larger number of imputations could have further increased the precision and the reproducibility of the result estimates.Reference White, Royston and Wood46 Third, we decided to dichotomise the PHQ-9 scores. The dichotomisation of a continuous variable leads to a potential loss of information. However, dichotomisation was required in our analysis to allow the comparison with the GRISELDA data-set. Fourth, the use of Read Codes for identification of adverse events may have resulted in data omission where such events were recorded in clinical encounter notes. Under-recording or miscoding of these data may have also occurred as this is a common issue with studies using routinely collected electronic health records; however, we have no reason to believe that this problem, if present, was differentiated by type of antidepressant. Fifthly, we decided not to employ a Cox proportional-hazard model for the analyses, as initially planned,Reference De Crescenzo, Garriga and Tomlinson15 because the proportional-hazard assumption for the outcomes measured was found to be implausible in our data. Instead, we employed multiple logistic regression models at pre-specified timepoints, as this analysis does not rely on proportional-hazard assumption, while also allowing comparison with previous clinical trials.Reference Cipriani, Furukawa and Salanti9 Nevertheless, a time-to-event analysis would provide better information about varying lengths of follow-up; this might be pursued in future research.
Additionally, our data-set is based primarily on prescription orders rather than actual medication dispensing. Although such missing data could affect the magnitude of observed changes across the entire sample, they were unlikely to impact the comparative analysis of relative changes between individual antidepressants and fluoxetine.
Finally, we could not assess the dose of initiated antidepressants, meaning that some medications (e.g. amitriptyline) may have been prescribed below usual antidepressant dosage.
Conclusion
Our study included a comprehensive analysis of real-world data from a cohort of 673 177 depressed individuals receiving first-line treatment with antidepressants in England. We found evidence of low acceptability, good tolerability and safety, and small-to-moderate effectiveness of antidepressants. SSRIs, including citalopram, fluoxetine and sertraline had the most favourable benefit/risk profile with good tolerability, safety and small-to-moderate effectiveness in both short- and long-term use. Additionally, our comparison of real-world estimates with data from RCTs showed good agreement for the most frequently prescribed antidepressants.
Supplementary material
Supplementary material is available online at https://doi.org/10.1192/bjp.2024.194.
Data availability
The data that support the findings of this study are not publicly available because they are based on de-identified national clinical records. Because of national and organisational data privacy regulations, individual-level data such as those used for this study cannot be shared openly.
Acknowledgements
We acknowledge the contribution of EMIS practices who contribute to QResearch and EMIS Health, and the Universities of Nottingham and Oxford for expertise in establishing, developing or supporting the QResearch database. This project involves data derived from patient-level information collected by the National Health Service (NHS), as part of the care and support of people with cancer. The Hospital Episode Statistics data and civil registration mortality data are used by permission from NHS England who retain the copyright in these data. NHS England bears no responsibility for the analysis or interpretation of the data.
Author contributions
F.D.C. and A.C. conceived and designed the study. C.G., Q.L., S.F., O.E. and J.H.-C. contributed to the design of the project. F.D.C., C.G., Q.L. and O.E. contributed to the statistical analyses. F.D.C. and R.D.G. drafted the manuscript, and all authors critically revised the manuscript and approved the final version.
Funding
This study was funded by a National Institute for Health and Care Research (NIHR) Research Professorship to A.C. (RP-2017-08-ST2-006) and the NIHR Oxford Health Biomedical Research Centre (NIHR203316). The funders did not have any role in the study design, the collection, analysis and interpretation of data, the writing of the report and the decision to submit the article for publication. The study authors are independent from the funders; they had full access to all the data (including statistical reports and tables), are responsible for the integrity of the data and the accuracy of the data analysis, and accept responsibility to submit for publication.
Declaration of interest
F.D.C. was supported by the NIHR Research Professorship to A.C. (grant RP-2017-08-ST2–006) and by the NIHR Oxford Health Biomedical Research Centre (grant BRC-1215-20005), and he is now an employee of Boehringer-Ingelheim International. A.C. has received research, educational and consultancy fees from INCiPiT (Italian Network for Paediatric Trials), Cariplo Foundation, Lundbeck and Angelini Pharma. He is supported by the NIHR Oxford Cognitive Health Clinical Research Facility, by an NIHR Research Professorship (grant RP-2017-08-ST2-006), by the NIHR Oxford and Thames Valley Applied Research Collaboration, by the NIHR Oxford Health Biomedical Research Centre (grant NIHR203316) and by the Wellcome Trust (Global Alliance for Living Evidence on Anxiety, Depression and Psychosis [GALENOS] Project). The views expressed are those of the authors and not necessarily those of the UK National Health Service, the NIHR or the UK Department of Health and Social Care. R.D.G. is a member of the British Journal of Psychiatry editorial board, and did not take part in the review or decision-making process of this paper. The other authors declare no conflicts of interest.









eLetters
No eLetters have been published for this article.