1. Introduction
Plants move. This overlooked truth has come to light again thanks to the recent study of spectacular ultra-fast motions (Forterre et al., Reference Forterre, Marmottant, Quilliet and Noblin2016). For example, the snapping of the Venus flytrap (Forterre et al., Reference Forterre, Skotheim, Dumais and Mahadevan2005; Sachse et al., Reference Sachse, Westermeier, Mylo, Nadasdi, Bischoff, Speck and Poppinga2020) and the catapulting of fern spores (Noblin et al., Reference Noblin, Rojas, Westbrook, Llorens, Argentina and Dumais2012) both require high speed cameras to be recorded. At the opposite side of the timescales spectrum, plants move through their growth. The observation of these slow observation necessitates time-lapse imaging. After Darwin (Darwin, Reference Darwin1897), they started to be historically investigated with the development of photography (Gaycken, Reference Gaycken2012). But we are still evidencing nowadays a variety of exciting new motions (Derr et al., Reference Derr, Bastien, Couturier and Douady2018; Rivière et al., Reference Rivière, Derr and Douady2017, Reference Rivière, Corre, Peaucelle, Derr and Douady2020). They can either be nastic motions, or tropisms, depending on whether the direction of the motion is imposed by factors internal or external to the plant, respectively. The movement is defined as autonomic (respectively, paratonic) depending on whether the triggering signal is internal to the plant or not. They can finally be reversible or linked to irreversible growth. These three dichotomies define the traditional classification of slow plant motions (Rivière et al., Reference Rivière, Derr and Douady2017). Within this framework, the status of one remarkable movement called nutation is still undecided (Baskin, Reference Baskin2015; Mugnai et al., Reference Mugnai, Azzarello, Masi, Pandolfi and Mancuso2015; Rivière et al., Reference Rivière, Derr and Douady2017; Stolarz, Reference Stolarz2009)
Nutation is the phenomenon that causes the orientation of the long axis of an elongated growing plant to vary over time in a pseudo-periodical way. It was already observed for climbing plants by British botanists of the 17th century (Webster, Reference Webster1966) and began to be studied by Hugo von Mohl and Ludwig Palm in the first part of the 19th century (Baillaud, Reference Baillaud1957). To the best of our knowledge, the term ‘nutation’ was first mentioned by Charles Bonnet (Bonnet, Reference Bonnet1754) although he acknowledges that this term had been named before him, by physicists who knew the phenomenon. They probably saw this motion as a botanical analog to the astronomical nutation.
Darwin later introduced the idea that nutation had an endogenous origin and many plant motions were actually modified nutations (Darwin, Reference Darwin1897). The very origin of nutation was a source of debate at the time nonetheless (Baillaud, Reference Baillaud1957), and it remains so up to this date (Brown, Reference Brown1993; Migliaccio et al., Reference Migliaccio, Tassone and Fortunati2013; Mugnai et al., Reference Mugnai, Azzarello, Masi, Pandolfi and Mancuso2015). Part of the community backs up Darwin’s idea of an internal oscillator (Brown et al., Reference Brown, Chapman, Lewis and Venditti1990; Johnsson et al., Reference Johnsson, Jansen, Engelmann and Schuster1999). Others ascribe this oscillating behaviour to inertial overshooting of the plant occurring during its straightening process (Agostinelli et al., Reference Agostinelli, Lucantonio, Noselli and DeSimone2020; Gradmann, Reference Gradmann1922; Israelsson & Johnsson, Reference Israelsson and Johnsson1967; Johnsson & Israelsson, Reference Johnsson and Israelsson1968). Finally, the compromise solution calling for a combination of these two hypotheses gathers more and more support (Agostinelli et al., Reference Agostinelli, DeSimone and Noselli2021; Johnsson, Reference Johnsson1997; Johnsson et al., Reference Johnsson, Jansen, Engelmann and Schuster1999; Orbović & Poff, Reference Orbović and Poff1997; Stolarz, Reference Stolarz2009). The one thing making consensus is that nutation is a macroscopic manifestation of multicellular microscopic growth.
Plant growth results from a subtle balance between the strong internal osmotic pressure and the resisting rheology of the cell wall (Tomos et al., Reference Tomos, Malone and Pritchard1989). Although growth and plasticity are very distinct processes, and growth doesn’t involve viscosity (Goriely, Reference Goriely2017), Lockhart used a Bingham plastic framework to formalise plant growth (Lockhart, Reference Lockhart1965). Lockhart’s model received good experimental support at the single cell level (Cosgrove, Reference Cosgrove1985; Green et al., Reference Green, Erickson and Buggy1971; Zhu & Boyer, Reference Zhu and Boyer1992). Still, some shortcomings need to be addressed (Jordan & Dumais, Reference Jordan and Dumais2010), and the origin of the cell wall-loosening mechanism remains unclear (Höfte et al., Reference Höfte, Peaucelle and Braybrook2012; Kroeger et al., Reference Kroeger, Zerzour and Geitmann2011; Micheli, Reference Micheli2001; Palin & Geitmann, Reference Palin and Geitmann2012). The cell wall is considered here to be an inactive gel but it was demonstrated that elements of the cell wall, the homogalacturonans (HG) can transform chemical modification into mechanical expansion through cell controlled enzymatic demethylesterification (Haas et al., Reference Haas, Wightman, Meyerowitz and Peaucelle2020). The precise role of elasticity that was added to Lockhart model later on by Ortega (Ortega, Reference Ortega1985) is then subject to debate (Haas et al., Reference Haas, Wightman, Meyerowitz and Peaucelle2020; Kierzkowski et al., Reference Kierzkowski, Nakayama, Routier-Kierzkowska, Weber, Bayer, Schorderet, Reinhardt, Kuhlemeier and Smith2012). Finally, the multi-cellular aspect of the biophysics of growth remains to be understood (Boudon et al., Reference Boudon, Chopard, Ali, Gilles, Hamant, Boudaoud, Traas and Godin2015). In particular, dynamical aspects related to water fluxes between cells have just started to be taken into account, either numerically (Cheddadi et al., Reference Cheddadi, Génard, Bertin and Godin2019) or even more recently theoretically with the development of a hydromechanical field theory for plant morphogenesis (Oliveri & Cheddadi, Reference Oliveri and Cheddadi2025). These new theoretical concepts will be key to understand the complex spatio-temporal behaviour observed in plant nutation.
The seminal work on the spatio-temporal characterization of nutation has been performed by Berg and Peacock (Berg & Peacock, Reference Berg and Peacock1992) where they evidenced strong fluctuations and traveling waves of the axial elongation rate in the sunflower hypocotyl. They even measured negative rates, suggesting local contractions. At the time, they acquired data with a single camera, and their growth measurements were necessarily biased by strong projection artifacts due to the three-dimensional nature of the motion.
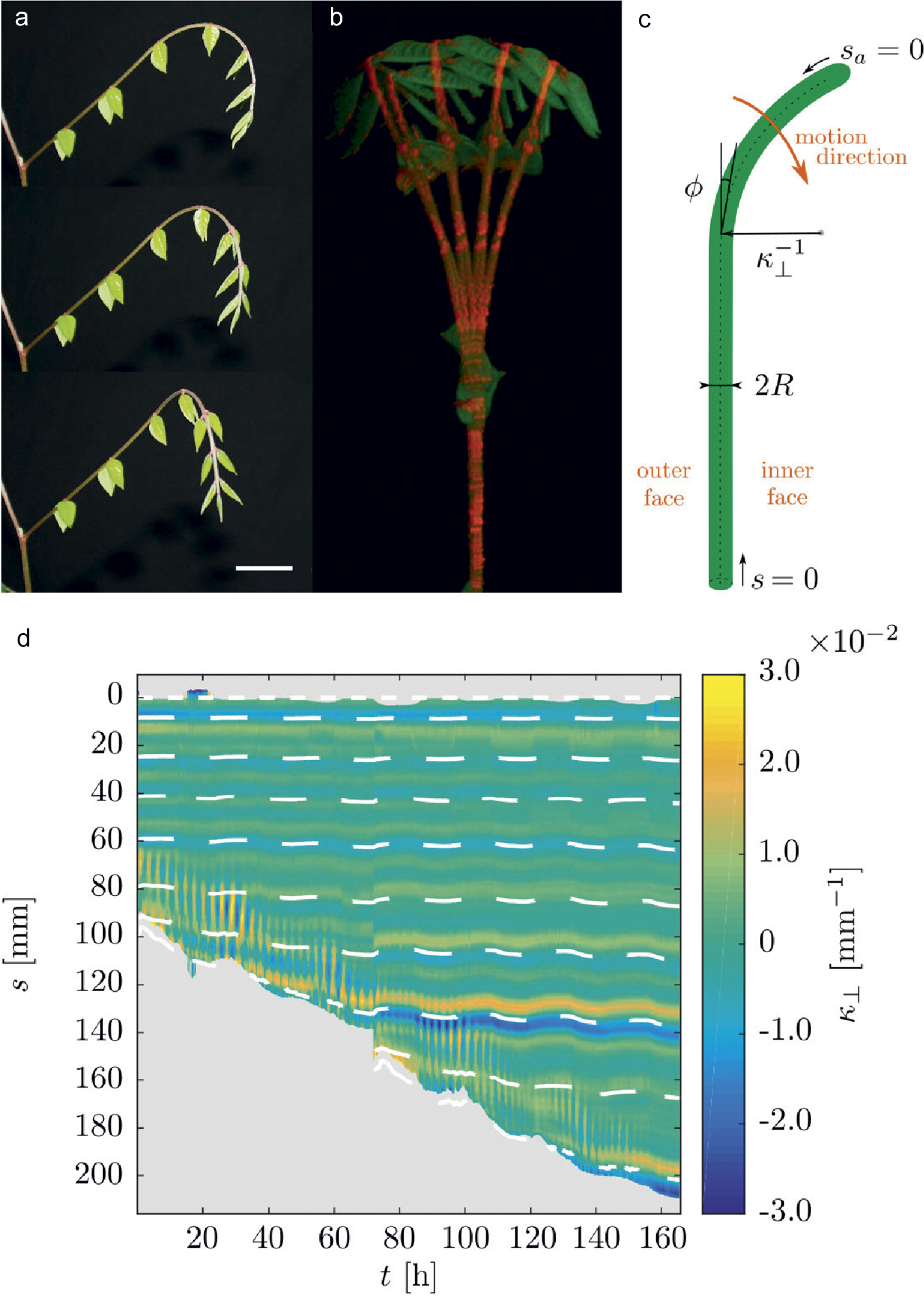
Here, we aim to revisit in detail the phenomenon of nutation. By carefully quantifying the motion of nutation (taking into account the 3D nature of the motion), we will gain knowledge on the nature of this puzzling mechanism. In this article, we focus on the plant Averrhoa carambola, a plant known for exhibiting ample nutation (see Figure 1a, b) and other growth motions (Rivière, Reference Rivière2017; Rivière et al., Reference Rivière, Derr and Douady2017, Reference Rivière, Corre, Peaucelle, Derr and Douady2020).
Nutation movement of an Averrhoa carambola compound leaf. (a) Side view, 30 minutes between pictures from top to bottom. The hook shape gradually comes out of the plane towards the observer. The scale bar indicates 3 cm which is the typical length scale between mature leaflets (b) Top view, 15 min between pictures (nutation period usually varies between 1.5 and 4 hours). The distal end of the leaf oscillates in a pendulum-like fashion, orthogonal to the rachis’ axis. After a full period, the leaf has elongated. (c) Geometrical parameters describing the rachis and nutation: arclengths s and
![]() $s_a$
(from the base or the apex, respectively), local angle
$s_a$
(from the base or the apex, respectively), local angle
![]() $\phi $
, local curvature
$\phi $
, local curvature
![]() $\kappa _\bot $
and radius R. The direction of motion defines the outer and inner faces of the rachis. (d) Spatiotemporal diagram of the curvature
$\kappa _\bot $
and radius R. The direction of motion defines the outer and inner faces of the rachis. (d) Spatiotemporal diagram of the curvature
![]() $\kappa _\bot (s,t)$
along the rachis obtained from a top-view time lapse movie. Oscillations of
$\kappa _\bot (s,t)$
along the rachis obtained from a top-view time lapse movie. Oscillations of
![]() $\kappa _\bot (s,t)$
are visible close to the apex. Dashed white lines mark the position of leaflets.
$\kappa _\bot (s,t)$
are visible close to the apex. Dashed white lines mark the position of leaflets.

The manuscript is organised as follows. We start by characterising the kinematics of nutation at the scale of the whole leaf, and emphasise the spatial organisation of growth. Our measurements allow to characterise the growth law of nutation and highlight a relationship between mean growth and differential growth. We then zoom in on the bending zone and, thanks to a kinematics model, analyse contraction events. Finally, we put our results in perspective with the microscopic properties (elasticity and chemical content) of the plant cell wall.
2. Materials and methods
2.1. Growth conditions of plants
Averrhoa carambola seeds were directly obtained from commercially available fruits and sown into all-purpose compost. Young seedlings were first kept inside a small lab greenhouse. Older plants (
![]() $>$
6 months) were then moved to the experimentation room. There, plants were submitted to a 12/12 light cycle under ORTICA 200W 2700K culture lamps. The temperature and relative humidity rate were monitored with a DHT22 sensor. Temperature was usually comprised between
$>$
6 months) were then moved to the experimentation room. There, plants were submitted to a 12/12 light cycle under ORTICA 200W 2700K culture lamps. The temperature and relative humidity rate were monitored with a DHT22 sensor. Temperature was usually comprised between
![]() $20^{o}C$
and
$20^{o}C$
and
![]() $24^{o}C$
. The relative humidity rate was around
$24^{o}C$
. The relative humidity rate was around
![]() $60\%$
. All methods were performed in accordance with the relevant guidelines and regulations.
$60\%$
. All methods were performed in accordance with the relevant guidelines and regulations.
2.2. Kinematics: sample preparation
The rachis of interest was carefully coated with fluorescent pigments with a brush. For curvature and coarse elongation measurements, the top of the rachis was coated homogeneously with orange pigments. Small blue fluorescent dots were added to mark the nodes and the petiole. For fine measurements of local growth, the orange pigments were deposited on the face of a few interfoliolar segments so that they form highly textured and contrasted patterns. In both cases, because of growth, pigments needed to be added manually on a regular basis to compensate the dilution of the signal over time.
2.3. Kinematics: image acquisition
The kinematics of nutation were captured using time-lapse photography with a DSLR camera controlled with the open-source software gPhoto2. The camera was firmly fixed to a rigid structure to avoid any displacement or rotation. The built-in flash of the camera was covered with LEE Moss green filter and set to the lowest intensity to keep light input minimal during nights. For curvature and coarse growth kinematics, top-views were taken every 2.5 min. For local growth measurements, side-views were taken every minute.
2.4. Kinematics: data analysis
The midline, or skeleton, of the rachis was obtained by first thresholding the red channel of the pictures. A cloud of points was obtained and then reduced to a smooth line with a moving median filter. The curvature of the rachis
![]() $\kappa _\bot $
in the plane of interest was obtained by locally fitting the midline to a circle. The position of the leaflets was retrieved by thresholding the blue channel. Because of growth, blues dots dilated, lost intensity in time and sometimes even split. The global unfurling motion of the rachis sometimes resulted in a temporary occlusion of some blue dots. Simple rules on the conservation of these dots, distance between consecutive dots and displacements values could overcome a majority of tracking failures. Manual correction was still needed in some special cases. Finally, the presented spatiotemporal graphs were smoothed with 2D averaging and median filters.
$\kappa _\bot $
in the plane of interest was obtained by locally fitting the midline to a circle. The position of the leaflets was retrieved by thresholding the blue channel. Because of growth, blues dots dilated, lost intensity in time and sometimes even split. The global unfurling motion of the rachis sometimes resulted in a temporary occlusion of some blue dots. Simple rules on the conservation of these dots, distance between consecutive dots and displacements values could overcome a majority of tracking failures. Manual correction was still needed in some special cases. Finally, the presented spatiotemporal graphs were smoothed with 2D averaging and median filters.
2.5. Kinematics: fine measurements
We obtained the skeleton of the rachis by a simple geometric transformation of the upper contour which is less altered by leaflet motions. We measured the elongation field along the rachis by using a previously published image-to-image correlation algorithm (Bastien et al., Reference Bastien, Legland, Martin, Fregosi, Peaucelle, Douady, Moulia and Höfte2016). The time-frequency analysis of the elongation signals was done by using MATLAB’s continuous wavelet transform toolbox. We used the ‘cgau2’ mother wavelet (second-order derivative of the complex Gaussian). For each location of the rachis,
![]() $\dot {\varepsilon }(t)$
was wavelet-transformed. From the resulting complex coefficients
$\dot {\varepsilon }(t)$
was wavelet-transformed. From the resulting complex coefficients
![]() $C_{a,b}$
we extracted information on the weight of each scale/frequency in the signal by computing an ‘energy’:
$C_{a,b}$
we extracted information on the weight of each scale/frequency in the signal by computing an ‘energy’:
![]() $E(a) = \sum _b \left | C_{a,b} \right |{}^2 / \sum _a \sum _b \left | C_{a,b} \right |{}^2$
, where a and b are the scale and shift parameters of the wavelet transform. This information was then re-aggregated and re-arranged to build kymographs displaying the weight of frequencies in the elongation signal along the rachis.
$E(a) = \sum _b \left | C_{a,b} \right |{}^2 / \sum _a \sum _b \left | C_{a,b} \right |{}^2$
, where a and b are the scale and shift parameters of the wavelet transform. This information was then re-aggregated and re-arranged to build kymographs displaying the weight of frequencies in the elongation signal along the rachis.
2.6. Kinematic model of nutation
The rachis is modelled by a two-dimensional beam of width
![]() $2R$
(see Figure S1 in the Supplementary Material) and of total length
$2R$
(see Figure S1 in the Supplementary Material) and of total length
![]() $L_{tot}$
. The geometry of the midline is then described with the same quantities than the actual leaf (see Figure 1c). The model contains only a few essential ingredients:
$L_{tot}$
. The geometry of the midline is then described with the same quantities than the actual leaf (see Figure 1c). The model contains only a few essential ingredients:
-
1. We define the elongation rate
 $\dot \varepsilon $
as the relative local growth rate of an element. For example, at arclength s, an element of size
$\dot \varepsilon $
as the relative local growth rate of an element. For example, at arclength s, an element of size
 $\delta s$
as the following local relative growth rate: (1)The lateral faces of the beam can have different elongation rates
$\delta s$
as the following local relative growth rate: (1)The lateral faces of the beam can have different elongation rates $$ \begin{align} \dot \varepsilon (s) = \frac{1}{\delta s}\frac{d \delta s}{dt}. \end{align} $$
$$ \begin{align} \dot \varepsilon (s) = \frac{1}{\delta s}\frac{d \delta s}{dt}. \end{align} $$
 $\dot {\varepsilon }_L$
and
$\dot {\varepsilon }_L$
and
 $\dot {\varepsilon }_R$
, giving rise to differential elongation
$\dot {\varepsilon }_R$
, giving rise to differential elongation
 $\dot {\delta }$
. We assume that the profile of elongation is linear in the bulk of the rachis: (2)
$\dot {\delta }$
. We assume that the profile of elongation is linear in the bulk of the rachis: (2) $$ \begin{align} \begin{cases} \dot{\varepsilon} = \left( \dot{\varepsilon}_R + \dot{\varepsilon}_L \right)/2 \\ \dot{\delta} = \left( \dot{\varepsilon}_R - \dot{\varepsilon}_L \right)/2. \end{cases} \end{align} $$
$$ \begin{align} \begin{cases} \dot{\varepsilon} = \left( \dot{\varepsilon}_R + \dot{\varepsilon}_L \right)/2 \\ \dot{\delta} = \left( \dot{\varepsilon}_R - \dot{\varepsilon}_L \right)/2. \end{cases} \end{align} $$
-
2. An apical growth zone of length
 $L_{gz}$
of constant length. The elongation rate of the midline
$L_{gz}$
of constant length. The elongation rate of the midline
 $\dot {\varepsilon }$
is thus independent of time and given by (3)where
$\dot {\varepsilon }$
is thus independent of time and given by (3)where $$ \begin{align} \dot{\varepsilon}\left(s_a \right) = \dfrac{\dot{\varepsilon}_0}{2}\left( 1 - \tanh \left(\dfrac{s_a-L_{gz}}{\Delta L}\right) \right), \end{align} $$
$$ \begin{align} \dot{\varepsilon}\left(s_a \right) = \dfrac{\dot{\varepsilon}_0}{2}\left( 1 - \tanh \left(\dfrac{s_a-L_{gz}}{\Delta L}\right) \right), \end{align} $$
 $s_a$
is the arc length starting from the apex, and
$s_a$
is the arc length starting from the apex, and
 $\Delta L$
the typical length scale of variation of
$\Delta L$
the typical length scale of variation of
 $\dot {\varepsilon }$
.
$\dot {\varepsilon }$
.
-
3. Differential elongation occurs where the mean elongation rate drops, within a bending zone of length
 $2\Delta L$
(for justification, see Results). Because nutation is a periodic oscillatory motion, differential elongation is modulated by a sine of period
$2\Delta L$
(for justification, see Results). Because nutation is a periodic oscillatory motion, differential elongation is modulated by a sine of period
 $2\pi /\omega $
: (4)
$2\pi /\omega $
: (4) $$ \begin{align} \dot{\delta}(s_a,t) = \dot{\delta}_0 \left( 1 - \tanh^2 \left( \dfrac{s_a-L_{gz}}{\Delta L} \right) \right) \sin{\omega t}. \end{align} $$
$$ \begin{align} \dot{\delta}(s_a,t) = \dot{\delta}_0 \left( 1 - \tanh^2 \left( \dfrac{s_a-L_{gz}}{\Delta L} \right) \right) \sin{\omega t}. \end{align} $$
-
4. We assume differential elongation is the unique driver of the bending of the rachis. In our case, since the period of nutation is much smaller than the typical time scale of elongation, we furthermore neglect the advection of curvature. In this case, differential elongation rates (
 $\dot \delta $
) and the rate of change of curvature (
$\dot \delta $
) and the rate of change of curvature (
 $d\kappa /dt$
) have been shown to be equivalent (Jensen & Forterre, Reference Jensen and Forterre2022; Silk, Reference Silk1984). Their relationship is purely geometric and can be simplified in the case
$d\kappa /dt$
) have been shown to be equivalent (Jensen & Forterre, Reference Jensen and Forterre2022; Silk, Reference Silk1984). Their relationship is purely geometric and can be simplified in the case
 $R \kappa _\perp \ll 1$
(for us,
$R \kappa _\perp \ll 1$
(for us,
 $R \kappa _\perp \sim 10^{-2}$
). We follow the kinematic calculation provided by Bastien (equation A.43 in Bastien (Reference Bastien2010)) with second-order correction in
$R \kappa _\perp \sim 10^{-2}$
). We follow the kinematic calculation provided by Bastien (equation A.43 in Bastien (Reference Bastien2010)) with second-order correction in
 $R \kappa _\perp $
to write (5)Interestingly, equation 5 does not display the dilution of curvature due to average growth. Chavarría showed that the dilution effect is compensated by curvature creation (Chavarría-Krauser, Reference Chavarría-Krauser2006).
$R \kappa _\perp $
to write (5)Interestingly, equation 5 does not display the dilution of curvature due to average growth. Chavarría showed that the dilution effect is compensated by curvature creation (Chavarría-Krauser, Reference Chavarría-Krauser2006). $$ \begin{align} \dfrac{\partial \kappa_\bot}{\partial t} \simeq \dfrac{1-R^2 \kappa^2_\bot}{R} \dot{\delta}. \end{align} $$
$$ \begin{align} \dfrac{\partial \kappa_\bot}{\partial t} \simeq \dfrac{1-R^2 \kappa^2_\bot}{R} \dot{\delta}. \end{align} $$
The model was implemented numerically with discretised versions of the kinematic equations 3, 4 and 5. When and where
![]() $\dot {\varepsilon } < \lvert \dot {\delta } \rvert $
, local contractions will occur along the lateral faces of the rachis – i.e., either
$\dot {\varepsilon } < \lvert \dot {\delta } \rvert $
, local contractions will occur along the lateral faces of the rachis – i.e., either
![]() $\dot {\varepsilon }_R<0$
or
$\dot {\varepsilon }_R<0$
or
![]() $\dot {\varepsilon }_L<0$
over a finite spatial extent (see Figure S1 in the Supplementary Material). This depends on the relative values of
$\dot {\varepsilon }_L<0$
over a finite spatial extent (see Figure S1 in the Supplementary Material). This depends on the relative values of
![]() $\dot {\delta }_0$
and
$\dot {\delta }_0$
and
![]() $\dot {\varepsilon }_0$
and the exact threshold depends on the spatial functions chosen to describe
$\dot {\varepsilon }_0$
and the exact threshold depends on the spatial functions chosen to describe
![]() $\dot {\varepsilon }$
and
$\dot {\varepsilon }$
and
![]() $\dot {\delta }$
. Here, a sufficient condition for contractions is
$\dot {\delta }$
. Here, a sufficient condition for contractions is
![]() $\dot {\varepsilon }_0 \leq \dot {\varepsilon }_c = 4\dot {\delta }_0$
. Finally, the apparent elongation
$\dot {\varepsilon }_0 \leq \dot {\varepsilon }_c = 4\dot {\delta }_0$
. Finally, the apparent elongation
![]() $\dot {\varepsilon }_\bot $
observed by a camera is obtained by measuring the orthogonal projection of the simulated rachis onto the plane of observation (see Figure S2 in the Supplementary Material).
$\dot {\varepsilon }_\bot $
observed by a camera is obtained by measuring the orthogonal projection of the simulated rachis onto the plane of observation (see Figure S2 in the Supplementary Material).
3. Results
3.1. Characterizing nutation
As they grow, Averrhoa carambola compound leaves exhibit pronounced growth motions. Putting aside the leaflets, the motion of the rachis can be broken down into two different motions, depending on their plane of occurrence (for anatomical terms, see Figure S3 in the Supplementary Material). The unfurling motion of the rachis of Averrhoa carambola mostly takes place in a principal plane (Rivière et al., Reference Rivière, Derr and Douady2017). The rachis unfolds steadily while propagating a hook shape (Rivière et al., Reference Rivière, Corre, Peaucelle, Derr and Douady2020). This hook shape is visible in Figure 1a. This motion is also accompanied by out-of-plane curvature variations. The rachis bends and unbends in a pseudo-periodical way, as if it were oscillating around a rectilinear state. The oscillations can already be seen in Figure 1a. In Figure 1b, we see the same motion from the top and on a slightly longer time range. The period of oscillation varies greatly between 1.5 and 4 hours, typically between 2 and 3 hours, while the typical amplitude is of the order of 25 degrees. Supporting movie 1 in the Supplementary Material shows a time-lapse movie of a typical nutation motion, seen from both sides. To properly describe the nutation motion, we define: the base-to-apex arc length s, and
![]() $s_a$
its apex-to-base counterpart;
$s_a$
its apex-to-base counterpart;
![]() $\phi $
the local angle with respect to the average direction of the rachis; and the curvature
$\phi $
the local angle with respect to the average direction of the rachis; and the curvature
![]() $\kappa _{\bot }$
(see Figure 1c). Figure 1d shows the quantification of
$\kappa _{\bot }$
(see Figure 1c). Figure 1d shows the quantification of
![]() $\kappa _{\bot }$
in both time and space.
$\kappa _{\bot }$
in both time and space.
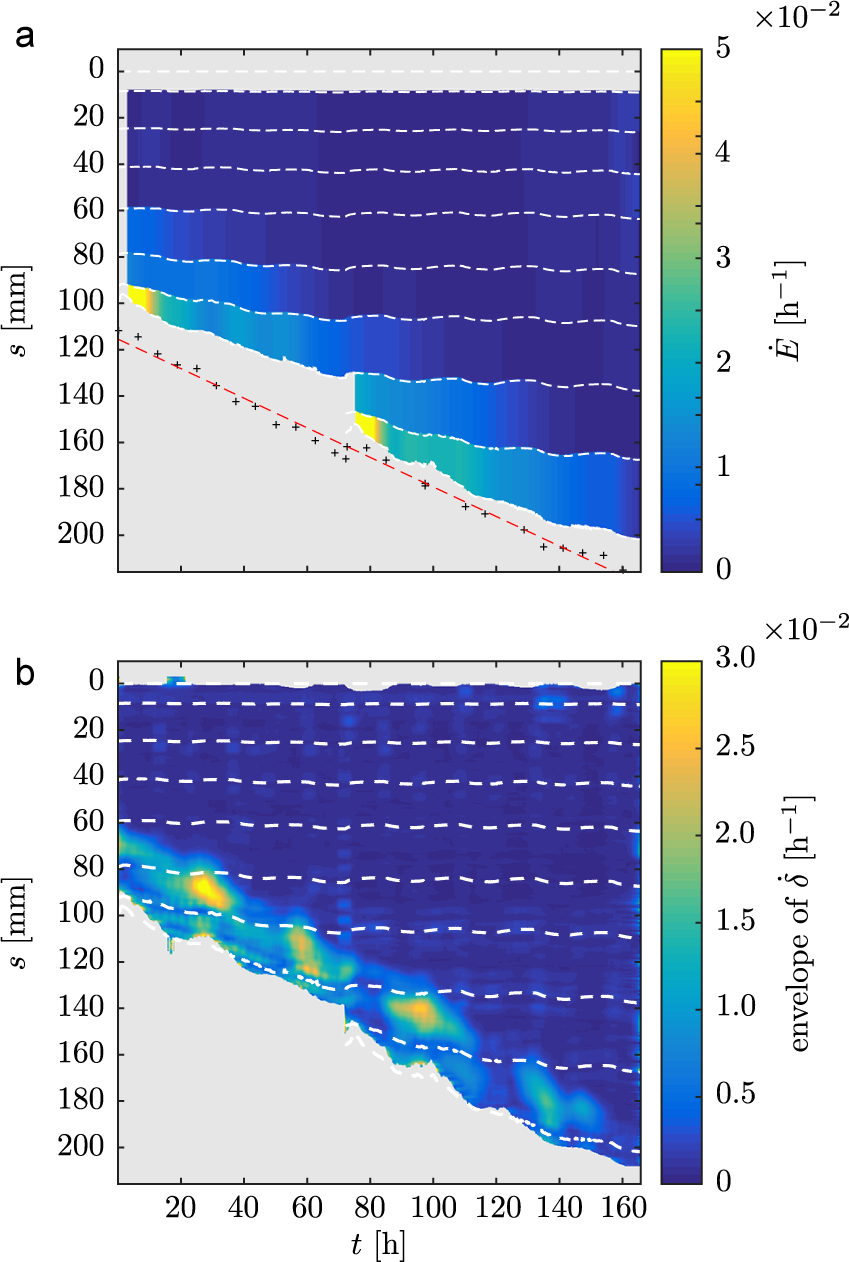
3.2. Elongation and bending are localised
We measured the average elongation rate
![]() $\dot {E}$
of each of the successive interfoliolar segments by tracking the position of the successive nodes. The spatiotemporal diagram of
$\dot {E}$
of each of the successive interfoliolar segments by tracking the position of the successive nodes. The spatiotemporal diagram of
![]() $\dot {E}$
shows that only the apical-most region of the rachis elongates, defining a growth zone near the apex (see Figure 2a).
$\dot {E}$
shows that only the apical-most region of the rachis elongates, defining a growth zone near the apex (see Figure 2a).
Elongation and estimated differential elongation during nutation. (a) Spatiotemporal diagram of the elongation rate
![]() $\dot {E}$
of each interfoliolar segment estimated from the leaflets’ trajectories (white dotted lines). The black crosses show the position of the leaf apex estimated from side-view pictures. The red dashed line is a linear fit of the apex position. (b) Spatiotemporal diagram of the envelope of differential elongation
$\dot {E}$
of each interfoliolar segment estimated from the leaflets’ trajectories (white dotted lines). The black crosses show the position of the leaf apex estimated from side-view pictures. The red dashed line is a linear fit of the apex position. (b) Spatiotemporal diagram of the envelope of differential elongation
![]() $\dot {\delta }$
estimated from the curvature diagram (nutation amplitude).
$\dot {\delta }$
estimated from the curvature diagram (nutation amplitude).

We then estimated the profile of differential elongation
![]() $\dot {\delta }$
along the rachis from the transverse curvature
$\dot {\delta }$
along the rachis from the transverse curvature
![]() $\kappa _{\bot }$
measurement, thanks to the several hypotheses described in the Material and Methods section. Its envelope was estimated via a method based on the Hilbert transform (Kincaid, Reference Kincaid1966) (for more details, see Supplementary Material). The evolution in time and space of the envelope of
$\kappa _{\bot }$
measurement, thanks to the several hypotheses described in the Material and Methods section. Its envelope was estimated via a method based on the Hilbert transform (Kincaid, Reference Kincaid1966) (for more details, see Supplementary Material). The evolution in time and space of the envelope of
![]() $\dot {\delta }$
is displayed in Figure 2b. We see that the differential growth – hence the bending – is spatially limited to a zone downstream of the apex. Similarly to what is done for the elongation, it is thus possible to define a bending zone.
$\dot {\delta }$
is displayed in Figure 2b. We see that the differential growth – hence the bending – is spatially limited to a zone downstream of the apex. Similarly to what is done for the elongation, it is thus possible to define a bending zone.
This bending zone is at a roughly constant distance from the apex, similarly to the constant length of elongation zone from the apex (see 2B). Finally, going a step further in the description of nutation, we notice that the amplitude of the differential elongation – or of the bending – varies in time, reaching a maximum of
![]() $3\times 10^{-2}~h^{-1}$
. These slow amplitude modulations of nutation are, however, not in the scope of the present study.
$3\times 10^{-2}~h^{-1}$
. These slow amplitude modulations of nutation are, however, not in the scope of the present study.
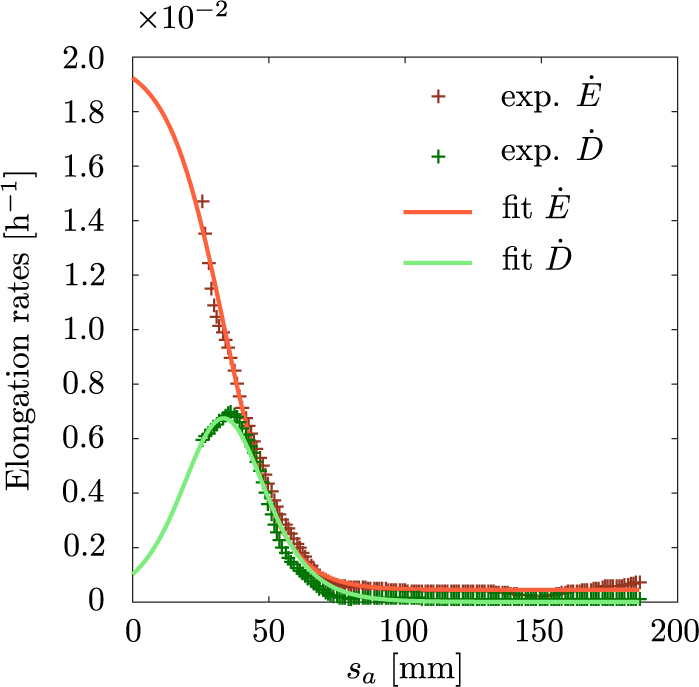
3.3. Differential elongation peaks where elongation drops
Because the growth spatial profile is almost steady in the frame of reference attached to the apex, we can average the measured quantities in time. The averaged quantities
![]() $\dot {E}$
and
$\dot {E}$
and
![]() $\dot {D}$
corresponding to mean elongation and differential elongation rates of interfoliolar segments are plotted in Figure 3. Both profiles confirm the existence of a localised growth zone. The typical length scale is about 50 mm, and beyond 100 mm growth is not detectable at all. The mean elongation rate looks like a sigmoid function. In the growth zone the typical elongation rate is of the order of
$\dot {D}$
corresponding to mean elongation and differential elongation rates of interfoliolar segments are plotted in Figure 3. Both profiles confirm the existence of a localised growth zone. The typical length scale is about 50 mm, and beyond 100 mm growth is not detectable at all. The mean elongation rate looks like a sigmoid function. In the growth zone the typical elongation rate is of the order of
![]() $10^{-2} \text {h}^{-1}$
, consistently to typical averaged values found in the literature (Lambers & Poorter, Reference Lambers and Poorter1992; Poorter & Remkes, Reference Poorter and Remkes1990), and then decays to zero. Interestingly, the differential elongation rate behaves differently. It is non-monotonic and its maximum coincides with the edge of the growing zone, where the mean elongation rate drops. A simple mathematical description of these sigmoid and peaked shapes is well fitted with the hyperbolic functions as in equations 3 and 4. The results are displayed in Figure 3. In this case the derivative of the fit of the longitudinal elongation rate matches well our experimental measurements of the differential elongation rate, with its amplitude remaining a free parameter (see Supplementary Material).
$10^{-2} \text {h}^{-1}$
, consistently to typical averaged values found in the literature (Lambers & Poorter, Reference Lambers and Poorter1992; Poorter & Remkes, Reference Poorter and Remkes1990), and then decays to zero. Interestingly, the differential elongation rate behaves differently. It is non-monotonic and its maximum coincides with the edge of the growing zone, where the mean elongation rate drops. A simple mathematical description of these sigmoid and peaked shapes is well fitted with the hyperbolic functions as in equations 3 and 4. The results are displayed in Figure 3. In this case the derivative of the fit of the longitudinal elongation rate matches well our experimental measurements of the differential elongation rate, with its amplitude remaining a free parameter (see Supplementary Material).
Average spatial profiles of elongation rate and differential elongation rate. The two profiles were fitted, respectively, to a sigmoid (red line) and to its derivative (green line). The complete profiles cannot be measured from a top-view because of the hook shape of the leaf.

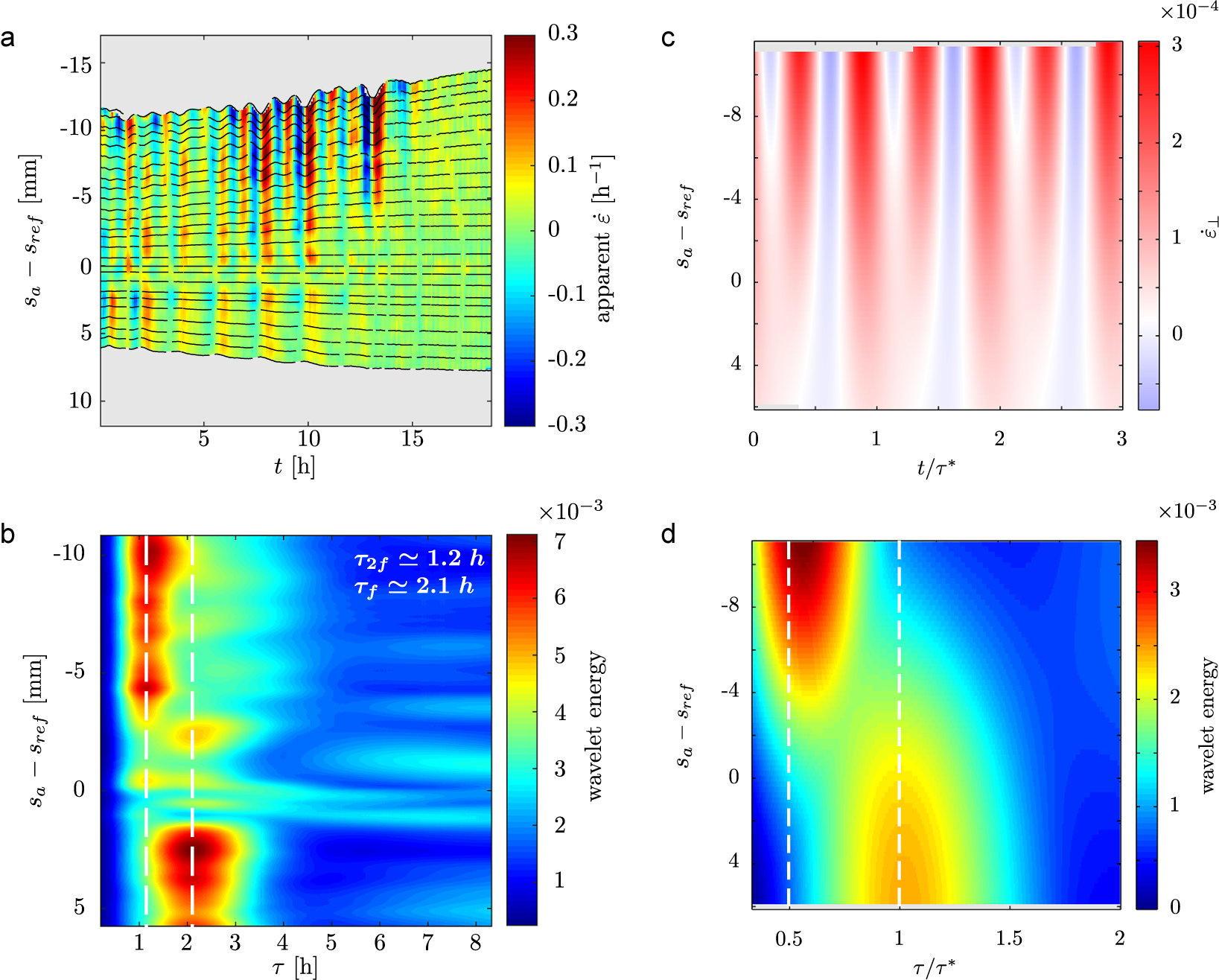
3.4. The elongation profile in the growth zone is compatible with local contractions
We used techniques inspired from digital image correlation (see Materials and Methods) to quantify the elongation profile within the bending zone. However, as the nutation moves the rachis towards or away from the camera, we can only measure an apparent elongation rate
![]() $\dot {\varepsilon }_\bot $
(see Figure S4 in the Supplementary Material and associated text). Strong projection artifacts indeed affect our measurements: we see oscillations and even negative values of
$\dot {\varepsilon }_\bot $
(see Figure S4 in the Supplementary Material and associated text). Strong projection artifacts indeed affect our measurements: we see oscillations and even negative values of
![]() $\dot {\varepsilon }_\bot $
(see Figure 4a).
$\dot {\varepsilon }_\bot $
(see Figure 4a).
(a) Spatiotemporal diagram showing an experimental measurement of the apparent local elongation rate
![]() $\dot {\varepsilon }$
in the bending zone from a side-view time lapse movie. Because of the oscillatory motion of the rachis, the elongation rate measured is affected by projection effects. (b) Wavelet decomposition of the experimental spatiotemporal diagram of apparent elongation rate. The decomposition shows that two dominant modes in the signal:
$\dot {\varepsilon }$
in the bending zone from a side-view time lapse movie. Because of the oscillatory motion of the rachis, the elongation rate measured is affected by projection effects. (b) Wavelet decomposition of the experimental spatiotemporal diagram of apparent elongation rate. The decomposition shows that two dominant modes in the signal:
![]() $\tau _{2f} \approx 1.2 \text {h}$
and
$\tau _{2f} \approx 1.2 \text {h}$
and
![]() $\tau _f \approx 2.1 \text {h},$
respectively, close to the apical and basal ends of the observed section of the rachis. (c) and (d): Best fit of the kinematics model to the experimental data;
$\tau _f \approx 2.1 \text {h},$
respectively, close to the apical and basal ends of the observed section of the rachis. (c) and (d): Best fit of the kinematics model to the experimental data;
![]() $\Delta \phi = 8^{\circ }, \text {L}_{gz} = 20.6 \text { mm}, \ \Delta L = 12.2\text {mm}, \ \dot {\delta _0} = 4.5\times 10^{-3} \text {h}^{-1}$
(
$\Delta \phi = 8^{\circ }, \text {L}_{gz} = 20.6 \text { mm}, \ \Delta L = 12.2\text {mm}, \ \dot {\delta _0} = 4.5\times 10^{-3} \text {h}^{-1}$
(
![]() $\dot {\varepsilon _0} = 1.4\times 10^{-2} \ \text {h}^{-1}, \text {R} = 0.26 \ \text {mm} $
were measured and fixed before fitting). This set of parameters allows local contractions.
$\dot {\varepsilon _0} = 1.4\times 10^{-2} \ \text {h}^{-1}, \text {R} = 0.26 \ \text {mm} $
were measured and fixed before fitting). This set of parameters allows local contractions.

Strikingly, the period of oscillation depends on position (see Figure 4a). Oscillations are faster at the apical end of the sample (top on graph), and slower at its basal end (bottom on graph). A wavelet transform evidences two distinct dominant modes with periods in a 2:1 ratio (see Figure 4b). We measured
![]() $\tau _f \approx 2.1 \text {h}$
at the basal end – corresponding to the nutation period – and
$\tau _f \approx 2.1 \text {h}$
at the basal end – corresponding to the nutation period – and
![]() $\tau _{2f} \approx 1.2 \text {h}$
at the apical end. In an attempt to rationalise these artifacts, and to work around them, we built a simple model based on the experimental kinematic features of nutation and also accounting for projection effects (see Materials and Methods). This model first provides an order of magnitude for differential growth. Indeed, it can be shown that
$\tau _{2f} \approx 1.2 \text {h}$
at the apical end. In an attempt to rationalise these artifacts, and to work around them, we built a simple model based on the experimental kinematic features of nutation and also accounting for projection effects (see Materials and Methods). This model first provides an order of magnitude for differential growth. Indeed, it can be shown that
This can be understood as
![]() $\dot {\delta }_0 / \omega $
being the total differential growth over one period of nutation, which divided by the radius R gives the local curvature of the rachis, and integrated over the bending zone length
$\dot {\delta }_0 / \omega $
being the total differential growth over one period of nutation, which divided by the radius R gives the local curvature of the rachis, and integrated over the bending zone length
![]() $2\Delta L$
, gives the final deviation of the apex (see Supplementary Material for formal derivation). By injecting estimations in this relationship (
$2\Delta L$
, gives the final deviation of the apex (see Supplementary Material for formal derivation). By injecting estimations in this relationship (
![]() $\Delta \Phi \sim \pi /6,\; \text {2}\pi /\omega \sim 2$
h,
$\Delta \Phi \sim \pi /6,\; \text {2}\pi /\omega \sim 2$
h,
![]() $R\sim 0.25$
mm and
$R\sim 0.25$
mm and
![]() $\Delta L \sim 50 $
mm), we find
$\Delta L \sim 50 $
mm), we find
![]() $\dot {\delta }_0 \sim 7.5 \times 10^{-3} \text {h}^{-1} \sim 10^{-2} \text {h}^{-1}$
matching the order of magnitude of the measured average growth, thus confirming the possibility of contractions.
$\dot {\delta }_0 \sim 7.5 \times 10^{-3} \text {h}^{-1} \sim 10^{-2} \text {h}^{-1}$
matching the order of magnitude of the measured average growth, thus confirming the possibility of contractions.
Second, simulations of our model reproduce the observed pattern of
![]() $\dot {\varepsilon }_\bot $
(see Figure 4c,d). Our model indeed shows that the two main oscillating contributions to
$\dot {\varepsilon }_\bot $
(see Figure 4c,d). Our model indeed shows that the two main oscillating contributions to
![]() $\dot {\varepsilon }_\bot $
are brought by: (i) projection (geometrical) effects, with frequency double that of nutation, maximum at the apical end of the rachis; and (ii) the differential elongation itself, with frequency equal to that of nutation, peaking around
$\dot {\varepsilon }_\bot $
are brought by: (i) projection (geometrical) effects, with frequency double that of nutation, maximum at the apical end of the rachis; and (ii) the differential elongation itself, with frequency equal to that of nutation, peaking around
![]() $s_a = L_{gz}$
(see Supplementary Material for more details). While oscillations of
$s_a = L_{gz}$
(see Supplementary Material for more details). While oscillations of
![]() $\dot {\varepsilon }_\bot $
at
$\dot {\varepsilon }_\bot $
at
![]() $\tau _{2f}$
are expected in any case (see Figure S4 in the Supplementary Material), oscillations with period
$\tau _{2f}$
are expected in any case (see Figure S4 in the Supplementary Material), oscillations with period
![]() $\tau _f$
are a direct signature of differential elongation.
$\tau _f$
are a direct signature of differential elongation.
Finally, we fit the wavelet transform spatiotemporal diagram as a way to estimate the unknown experimental parameters. The best fit is presented in Figure 4c,d. The corresponding parameters
![]() $\dot {\delta }_0 = 4.5\times 10^{-3} \text {h}^{-1}$
and
$\dot {\delta }_0 = 4.5\times 10^{-3} \text {h}^{-1}$
and
![]() $\dot {\varepsilon }_0 = 1.4 \times 10^{-2} h^{-1}$
indicate that the rachis must locally contract to explain our experimental measurements.
$\dot {\varepsilon }_0 = 1.4 \times 10^{-2} h^{-1}$
indicate that the rachis must locally contract to explain our experimental measurements.
4. Discussion
4.1. The nutation zone is spatially linked to the growing zone and undergoes ‘stop and go’ phenomena
The kinematics of nutation presented here are consistent with our previous study on the same system and confirm the presence of a steady growth zone, extending from the apex over a constant length (Rivière et al., Reference Rivière, Corre, Peaucelle, Derr and Douady2020). This is also in agreement with growth spatial profiles observed in roots (Chavarría-Krauser et al., Reference Chavarría-Krauser, Nagel, Palme, Schurr, Walter and Scharr2008; Quiros et al., Reference Quiros, Bogeat-Triboulot, Couturier and Kolb2022; Silk et al., Reference Silk, Lord and Eckard1989; Walter et al., Reference Walter, Spies, Terjung, Küsters, Kirchgessner and Schurr2002), and several cylindrical aerial organs (Bastien et al., Reference Bastien, Guayasamin, Douady and Moulia2018; Peters & Tomos, Reference Peters and Tomos2000; Silk, Reference Silk1992).
We also show that the basal end of the growth zone coincides with the nutation zone – i.e., fluctuations of the differential elongation rate. The spatial coincidence of the maximum of the differential elongation rate with the region of steepest decrease of the average elongation rate is consistent with previous observations on Arabidopsis thaliana roots (Chavarría-Krauser et al., Reference Chavarría-Krauser, Nagel, Palme, Schurr, Walter and Scharr2008). This phenomenon could be compatible with the existence of a maximum value for the elongation rate, likely set by a combination of environmental factors and inner physiological constraints. Close to the apex, growth-regulating signals could be so strong that the elongation saturates by far. Small perturbations of these signals in space or time would not affect the saturated elongation rate and would get edged out. Conversely, when and where they are not strong enough to saturate elongation anymore, any perturbation on the growth-regulating signals could directly affect the elongation rate and would eventually translate into oscillations. The basal end of the growth zone would then be the location most prone to such variations. The same interpretation could apply to oscillations during the gravitropic straightening of wheat coleoptiles (Bastien et al., Reference Bastien, Guayasamin, Douady and Moulia2018): as the coleoptile bends towards the vertical, the differential growth signal is at its maximum, and no oscillation is observed. On the contrary, when the coleoptile approaches a vertical posture, the signal decreases, and nutation of the tip becomes visible again.
Quantitatively, when and where the differential elongation rate is maximum, its amplitude is also comparable to the local average elongation rate (see Figure 3) making the total growth of one side close to zero or even possibly negative. This could be schematized as a ‘stop and go’ phenomenon, where each side of the rachis grows alternately, before growth and motions cease altogether. This alternate growth behavior was already apparent in pea’s epicotyls observation (Baskin, Reference Baskin1986).
4.2. Contraction events during plant growth
In all generality, the spatial arrangement of the average elongation rate
![]() $\dot {\varepsilon }$
and the differential elongation rate
$\dot {\varepsilon }$
and the differential elongation rate
![]() $\dot {\delta }$
can lead to local contractions within the bending zone depending on their relative amplitudes (see Figure 3d). Our local measurements of
$\dot {\delta }$
can lead to local contractions within the bending zone depending on their relative amplitudes (see Figure 3d). Our local measurements of
![]() $\dot {\varepsilon }$
in the bending zone (see Figure 4a,b), interpreted by taking projection effects into account, indirectly revealed that nutation in Averrhoa carambola rachis is compatible with local contraction events – i.e., negative elongation rates over finite spatial extent – (see Figure 4). These results are in line with previous reports of contraction events in the circumnutating stems of several other species (Baskin, Reference Baskin1986; Berg & Peacock, Reference Berg and Peacock1992; Caré et al., Reference Caré, Nefed’ev, Bonnet, Millet and Badot1998; Stolarz et al., Reference Stolarz, Krol, Dziubinska and Zawadzki2008), both at the cell and tissue levels. It was also observed that contractions are circumscribed to either the basal end of the growth zone – where the average elongation rate decays – (Berg & Peacock, Reference Berg and Peacock1992), or to the bending zone (Caré et al., Reference Caré, Nefed’ev, Bonnet, Millet and Badot1998), consistently with our findings.
$\dot {\varepsilon }$
in the bending zone (see Figure 4a,b), interpreted by taking projection effects into account, indirectly revealed that nutation in Averrhoa carambola rachis is compatible with local contraction events – i.e., negative elongation rates over finite spatial extent – (see Figure 4). These results are in line with previous reports of contraction events in the circumnutating stems of several other species (Baskin, Reference Baskin1986; Berg & Peacock, Reference Berg and Peacock1992; Caré et al., Reference Caré, Nefed’ev, Bonnet, Millet and Badot1998; Stolarz et al., Reference Stolarz, Krol, Dziubinska and Zawadzki2008), both at the cell and tissue levels. It was also observed that contractions are circumscribed to either the basal end of the growth zone – where the average elongation rate decays – (Berg & Peacock, Reference Berg and Peacock1992), or to the bending zone (Caré et al., Reference Caré, Nefed’ev, Bonnet, Millet and Badot1998), consistently with our findings.
Reports of contractions and negative growth rates go beyond the sole context of nutation. They have indeed been observed during shoot apical meristem morphogenesis (Kwiatkowska, Reference Kwiatkowska2006; Kwiatkowska & Dumais, Reference Kwiatkowska and Dumais2003; Kwiatkowska & Routier-Kierzkowska, Reference Kwiatkowska and Routier-Kierzkowska2009; Long et al., Reference Long, Cheddadi, Mosca, Mirabet, Dumond, Kiss, Traas, Godin and Boudaoud2020) and the growth of simple leaves (Armon et al., Reference Armon, Moshe and Sharon2021), both at the cellular and organ scales.
The interpretation of negative growth rates is still a matter of debate in the community. In 1992, Berg and Peacock, attributed tissue contractions to a purely elastic behaviour (Berg & Peacock, Reference Berg and Peacock1992). In 1998, Care et al. showed that tissue contractions were not artifacts but instead due to local cell contraction driven by osmotic changes (Caré et al., Reference Caré, Nefed’ev, Bonnet, Millet and Badot1998). Only recently, theories describing both elasticity and osmotic water fluxes between cells in plants (Cheddadi et al., Reference Cheddadi, Génard, Bertin and Godin2019; Oliveri & Cheddadi, Reference Oliveri and Cheddadi2025) have shown that effects due to water transport are central in plant morphogenesis: a growing tissue acts as a sink and extracts water from neighbouring cells which acts like a source. In our case, during the nutation movement, the growing side could get water from the opposite side, leading to contractions of the latter.
4.3. A window on the physiological implications of nutation and growth
We believe that growth motions, and nutation in particular, offer an experimental framework to probe growth at the microscopic scale. Its oscillatory nature combined with a clear spatial pattern allow to probe a variety of cell wall mechanics, cell wall chemical status and macroscopic growth rates combinations. A full microscopic investigation goes beyond the scope of this article, but we provide in Supplementary Material a set of preliminary experiments constituting a proof of concept.
The first possible experiment is to use our nutating system to probe cell wall elasticity in growing or not growing tissue. Our preliminary experiments seem to indicate a strong correlation between elasticity and growth: the growing side is found softer than the non-growing side (see Figure S4 in the Supplementary Material, and corresponding text). This belongs to a long series of observations correlating growth with changes in cell wall elasticity, by suggesting that growth is faster where the Young’s modulus is lower. This phenomenon was evidenced in growing pollen tips (Zerzour et al., Reference Zerzour, Kroeger and Geitmann2009), maize roots elongation zone (Abeysekera & McCully, Reference Abeysekera and McCully1994; Kozlova et al., Reference Kozlova, Petrova, Ananchenko and Gorshkova2019), Arabidopsis shoot meristem before primordia formation (Milani et al., Reference Milani, Gholamirad, Traas, Arnéodo, Boudaoud, Argoul and Hamant2011; Peaucelle et al., Reference Peaucelle, Braybrook, Le Guillou, Bron, Kuhlemeier and Höfte2011). Similarly, we can probe the changes in chemical status during growth, and our preliminary experiments seem to indicate a change in the methylesterification status of the pectins if the tissue is growing or not (see Figure S5 in the Supplementary Material and associated text).
In our system it is difficult to disentangle the reversible and irreversible contributions to growth as it was done by Proseus et al. for the single-cell algae Chara (Proseus et al., Reference Proseus, Ortega and Boyer1999). It has also been shown in the case of the shoot apical meristem that elastic inhomogeneities (or differences in stress stiffening) could lead to differential growth (Kierzkowski et al., Reference Kierzkowski, Nakayama, Routier-Kierzkowska, Weber, Bayer, Schorderet, Reinhardt, Kuhlemeier and Smith2012). Therefore, to discuss the missing link between the observed microscopic properties and the macroscopic contractions, we propose two different hypothetic scenarios.
First, one should consider the reversible processes as they have already been found to be involved in nutation and growth. As mentioned before, Cheddadi et al. recently formalised the water fluxes coupling in multicellular organs. They showed in particular that new types of lateral inhibitory mechanisms could amplify growth heterogeneities (Cheddadi et al., Reference Cheddadi, Génard, Bertin and Godin2019): softer tissues are favored to become sinks for water at the expense of the neighbouring cells. In order to investigate this scenario further, one will need to extend the model to incorporate mechanical aspects. Recently, Moulton et al. generalised the analytical results of Timoshenko about the growth of 2D bimetallic strips (Timoshenko, Reference Timoshenko1925) to filaments in 3D (Moulton et al., Reference Moulton, Lessinnes and Goriely2020a). This new framework, which already proved successful to reproduce plant tropism (Moulton et al., Reference Moulton, Oliveri and Goriely2020b), is an exciting new line of investigation for nutation.
From our preliminary observations, one could also propose a second hypothetical scenario for the temporal events: on the growing side, HG are actively addressed to the cell wall in their native methylated way. Then growth turns to the other side of the rachis following an external or internal signal, and HG are sparsely degraded or recycled by endoglucanase explaining the reduction in staining observed in methylated and demethylated pectins. Here we can indicate that the time scale could be as fast as 30 minutes. Haas et al. (Reference Haas, Wightman, Meyerowitz and Peaucelle2020) proposed that the expansion part could be solely due to HG filament expansion following the de-methylesterification. In addition, the partial removal of the highly charged polymers following their recycling could as well lead to cell wall compaction in link with the observed tissue contraction.
5. Conclusion
To sum up, we provided on a new biological model case (Averrhoa carambola), a complete kinematic description of the nutation motion paying especially attention to the 3D effects. Thanks to a kinematic model we could disentangle the projection artifacts, and prove that contractions really happen during nutation. Nutation is found to occur as a steady propagation spatial growth pattern showing co-localisation of the peak of differential growth with the onset of the growing region. Finally, we showed that this macroscopic behaviour can be used as a tool to investigate microscopic properties of the dynamically alternating growing tissues.
Open peer review
To view the open peer review materials for this article, please visit http://doi.org/10.1017/qpb.2025.10013.
Acknowledgements
We thank Elliot Meyerowitz and Raymond Wightman for their support and help on the immunolabelling experiments. We are also grateful to Olivier Hamant and Emmanuel de Langre for continuous feedback on our works.
Competing interests
The authors declare no competing interests exist.
Data availability statement
The datasets generated during and/or analysed during the current study are available in the Zenodo repository at https://doi.org/10.5281/zenodo.7994913.
Author contributions
All authors designed the research. M.R. and J.D. performed kinematics experiments. M.R. and A.P. performed microscopic investigation. M.R. and S.D. designed the model. M.R. performed numerical simulations. M.R. and J.D. analysed the data. All authors discussed the results. M.R. and J.D. drafted the article. All authors edited the manuscript.
Funding statement
This research received no specific grant from any funding agency, commercial or not-for-profit sectors. M.R. is grateful to ‘Ecole Doctorale Frontières du Vivant - Programme Bettencourt’ for financial support. IJPB benefits from the support of Saclay Plant Sciences-SPS (ANR-17-EUR-0007).
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1017/qpb.2025.10013.
















Comments
Dear Editor,
We are pleased to submit our manuscript “Plant Nutation Relies on Steady Propagation of Spatial Asymmetric Growth Pattern” for consideration as a research article in Quantitative Plant Biology. This work was conducted jointly by Mathieu Rivière, Alexis Peaucelle, Julien Derr and Stéphane Douady.
Nutation is an intriguing oscillatory motion displayed by most growing plant organs. Although nutation has been discussed and debated in the literature, there is no consensus about the ins and outs of this phenomenon. In particular, its internal biophysical
mechanisms remain to be understood.
In this article, we experimentally characterized the nutation of a typical compound leaf, Averrhoa carambola. We provide a complete kinematics description of the nutation motion at the scale of the organ. Our research differs from prior works on nutation by
taking the three-dimensional nature of this motion into account. Thanks to a minimal kinematics model, we go beyond the strong projection artifacts that would otherwise distort growth measurements. This allows us to draw the three following outcomes:
1. We evidence that nutation characterizes as the steady propagation of a robust spatial growth pattern. This pattern shows co-localization of a peak of differential growth with the onset of the growth zone. We discuss in the article the
consequences and possible reasons of this pattern.
2. We show that contraction events occur during nutation, supporting a number of scattered reports of contractions during plant growth in the literature.
3. We propose a set of experiments, constituting a proof of concept that nutation can be used as a tool to probe microscopic properties of the plant cell wall under different growth regimes.
Nutation is fundamentally linked to growth and its dynamics; the implications of our work thus go beyond the sole scope of plant movement or shape regulation and shed a new light on the dynamical aspects of growth itself.
We believe our approach and findings would be of interest to both specialists of plant growth and to a much broader audience of biophysicists and biologists interested in morphogenesis and shape regulation in living systems.
Thank you very much for considering our manuscript.
Sincerely yours,
Julien Derr, on behalf of all the
co-authors