Introduction
Pneumonia and lower respiratory tract infections (LRTI) are among the most frequent healthcare-associated infections globally. Reference Zingg, Metsini and Balmelli1–3 The prevalence of nonventilator-associated hospital-acquired pneumonia (nvHAP) is roughly double that of ventilator-associated pneumonia (VAP). Reference Magill, Edwards and Bamberg2,3 Hospital-acquired pneumonia (HAP) in ventilated and non-ventilated patients together is responsible for most disability-adjusted life years (DALY) of all hospital-acquired infections (HAI). Reference Cassini, Plachouras and Eckmanns4 Mortality from nvHAP is up to 30%, Reference Davis and Finley5–Reference Amodio, Schreiber, Faes Hesse and Wolfensberger7 and nvHAP generates significant hospital costs. Reference Davis and Finley5,Reference Kaier, Wolkewitz and Heister8
Because all hospitalized patients are at risk for nvHAP, surveillance expands to the entire hospital population. The University Hospital Zurich (USZ) has developed and validated a semi-automated surveillance system for nvHAP in 2017/2018. Reference Wolfensberger, Jakob and Faes Hesse9 The preselecting classification algorithm with five indicators – radiology procedure, radiology report, leukocytes, body temperature, intubation – closely mirrors the European Centre for Disease Prevention and Control (ECDC) HAP criteria, 10 and reduced the number of patients for manual review at USZ by 93% with high sensitivity. Reference Wolfensberger, Jakob and Faes Hesse9
All automated surveillance systems use routine data from electronic health records (EHR), which are increasingly being introduced in the United States 11 and Europe. 12 Still, some hospitals lack complete electronic records or have insufficient resources in information technology (IT) for data processing. In the USZ patient population we found that certain single indicators and selection algorithms with fewer indicators were able to decrease the number of records to manually review by >90%. Reference van Mourik, van Rooden and Abbas13,Reference Mueller, Pfister, Faes Hesse, Zingg, Wolfensberger and Swissnoso14 This suggested that the USZ nvHAP preselection algorithm could be used in an adapted form in hospitals with limited datasets or limited IT-resources.
In view toward a national nvHAP surveillance in Switzerland, we conducted a feasibility study to evaluate the implementation and performance of an adaptable nvHAP selection algorithm in pilot hospitals. In addition to sensitivity and percentage of records to manually review, the workload for technical implementation, the time for manual review of preselected records, and contextual implementation determinants of the semi-automated surveillance were assessed.
Methods
Study setting and patients under surveillance
For this feasibility study, seven Swiss acute care hospitals were purposefully selected. Two academic tertiary-care centers, two public general hospitals, and three community hospitals were included. One hospital was French-speaking, the others were German-speaking. The hospitals were free to choose the patient population for nvHAP-surveillance.
Adaptable preselection algorithm and minimum data set
The USZ algorithm represented the adaptable “standard algorithm.” It included the indicators “chest radiology procedure,” “chest radiology report,” “leucocyte count,” “body temperature,” and “intubation.” “Chest radiology procedure” (type and date of procedure) was the only minimally required indicator, alongside admission and discharge dates. Patients were preselected if they had at least one “chest radiology procedure of relevance.” A radiology procedure was considered “relevant” if conducted >48 h after hospital admission (or anytime in patients who were readmitted within 10 days), and when it was in a predefined temporal relationship with leucocyte count, body temperature, and respiratory device (if this data was available) (Appendix 1). Radiology procedures were classified as non-relevant if the corresponding radiology report explicitly ruled out pneumonia, based on classification of the report according to a simple text analysis, identifying terms excluding pneumonia, i.e. “no” and “infiltrate” in one sentence, without a “restricting term” (e.g., “but”, “left”) in the same sentence (Appendix 2a). For hospitals unable to implement the standard algorithm, modified versions with less indicators were allowed. For the hospital in French-speaking Switzerland, customizing/translating the radiology text analysis was required.
Team composition, material, training, and support
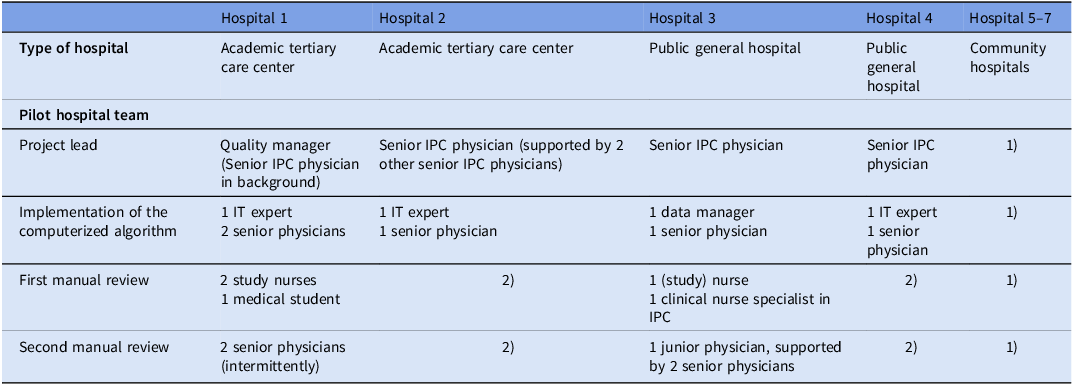
Four pilot hospital teams were established with a project lead, IT staff for technical implementation of the algorithm, and data collectors for manual review (Table 1). The community hospitals were networked with one of the public general hospitals, whose team was responsible for implementing and conducting the surveillance. The teams received all materials to implement the computerized algorithm, including a Python code, an Excel data-entry form, an analysis/reporting tool, and manuals. The Python code was designed to be configurable, allowing to include indicators that were available in the respective hospital. The IT specialist of the coordination center provided ongoing support. For manual review, hospitals were provided with a handbook and received a mandatory two-hour training session. The coordination team was available to support the hospital and provide training as needed.
Pilot hospitals and team composition

Abbreviations: IPC, infection prevention and control; IT, Information Technology.
1) performed by team from public general hospital 2) no manual surveillance conducted.
Pneumonia surveillance definitions and manual review
The pneumonia definition criteria of this study aligned with the ECDC criteria for HAP. 10 Pneumonia was defined with the presence of radiological signs, fever or elevated or reduced leucocyte counts, and clinical signs and symptoms such as cough, dyspnea, or worsening of gas exchange (Appendix 3). Pneumonia in patients who were continuously intubated during the full 48h before symptom onset were considered VAP, and thus not subject of this study. We introduced indeterminate HAP (iHAP) as a novel classification. Patients never intubated in the 48 hours before symptom onset were classified as nvHAP, whereas patients who were both intubated and extubated within the preceding 48 hours were classified as iHAP. In the following sections, nvHAP is used as an umbrella term and includes iHAP.
All preselected patients underwent manual review by a first reviewer, typically someone with a nursing background. In cases of uncertainty, a second reviewer with expertise in infectious diseases provided additional assessment.
Performance characteristics of algorithms
Sensitivity of the algorithm was assessed using full manual surveillance as the reference standard. A sample size of 73 patients with nvHAP per hospital was estimated sufficient to demonstrate a sensitivity of 95% with a ±5% error margin. As nvHAP is a rare event, we selected a validation cohort with patients at higher probability for HAP, i.e. patients with a discharge diagnostic code for HAP (U69.0x), and - in smaller hospitals - complimented by 200 randomly selected patients with a hospital stay of at least two weeks. Sensitivity was calculated as follows:
 $$\small{\eqalign{& {\rm{Sensitivity\;of\;algorithm\;}} \cr\!& \!\!= {\rm{}}{{{\rm{Patients\;identified\;by\;manual\;review\;AND\;on\;the\;preselection\;list}}} \over {{\rm{Patients\;identified\;by\;manual\;review}}}}\cr& {\quad\rm{\times}}100{\rm{\;}}\left( {{\rm{in\;\% }}} \right)}}$$
$$\small{\eqalign{& {\rm{Sensitivity\;of\;algorithm\;}} \cr\!& \!\!= {\rm{}}{{{\rm{Patients\;identified\;by\;manual\;review\;AND\;on\;the\;preselection\;list}}} \over {{\rm{Patients\;identified\;by\;manual\;review}}}}\cr& {\quad\rm{\times}}100{\rm{\;}}\left( {{\rm{in\;\% }}} \right)}}$$
Percentage of records to manually review was calculated as follows:
 $${\rm{Percentage\;of\;records\;to\;manually\;review\;}} $$$$= {\rm}{{{\rm{number\;of\;patients\;preselected\;from\;algorithm}}} \over {{\rm{all\;patients}}}}{\rm{\times}}100{\rm{\;}}\left( {{\rm{in\;\% }}} \right)$$
$${\rm{Percentage\;of\;records\;to\;manually\;review\;}} $$$$= {\rm}{{{\rm{number\;of\;patients\;preselected\;from\;algorithm}}} \over {{\rm{all\;patients}}}}{\rm{\times}}100{\rm{\;}}\left( {{\rm{in\;\% }}} \right)$$
Finally, the number of patients needed to be screened from the list of preselected patients to detect one patient with nvHAP was calculated.
Time investment and implementation determinants
All pilot hospital team members kept track of hours spent on the project. To identify determinants (i.e., barriers and facilitators) for technical implementation and manual review, two 20-minute interviews were conducted with members of the pilot teams at the start and the end of the project by a member of the coordination team (MFH) (interview guide see Appendix 4). Notes were coded deductively according to constructs of the updated consolidated framework for implementation research (CFIR). Reference Damschroder, Reardon, Widerquist and Lowery15,Reference Damschroder, Aron, Keith, Kirsh, Alexander and Lowery16 CFIR is a comprehensive, meta-theoretical framework including five domains (i.e., Innovation, Outer Setting, Inner Setting, Individuals, and Implementation Process) which is used to help understand the factors that influence how well an innovation is implemented in real-world settings. Deductive coding was supplemented by additional inductively created codes to capture nuances in the data.
Ethics
The project was a quality improvement project and the necessity for formal ethical evaluation was waived from all responsible cantonal ethical committees (BASEC Req-2023-00148). All individual patient data remained in the pilot hospitals; only aggregate data were reported to the coordination center.
Results
The 5-indicator standard algorithm was implemented in one hospital (hospital 1); the other six hospitals implemented adapted algorithms, with four, two or one indicator (Table 2). One hospital included only patients from two departments with patients at intrinsically higher risk for nvHAP, while the other six hospitals included all hospitalized patients (none served psychiatric, and only one (hospital 4) served pediatric patients). All hospitals included patients discharged between the months of January and June 2023. The German-speaking hospitals used the text-analysis provided by the coordination center, while the hospital in French-speaking Switzerland translated the text-analysis into French (Appendix 2b). Manual patient review was conducted in five hospitals by two teams, i.e. one team from the public general hospital and its associated network of three community hospitals, and another team from a tertiary care center.
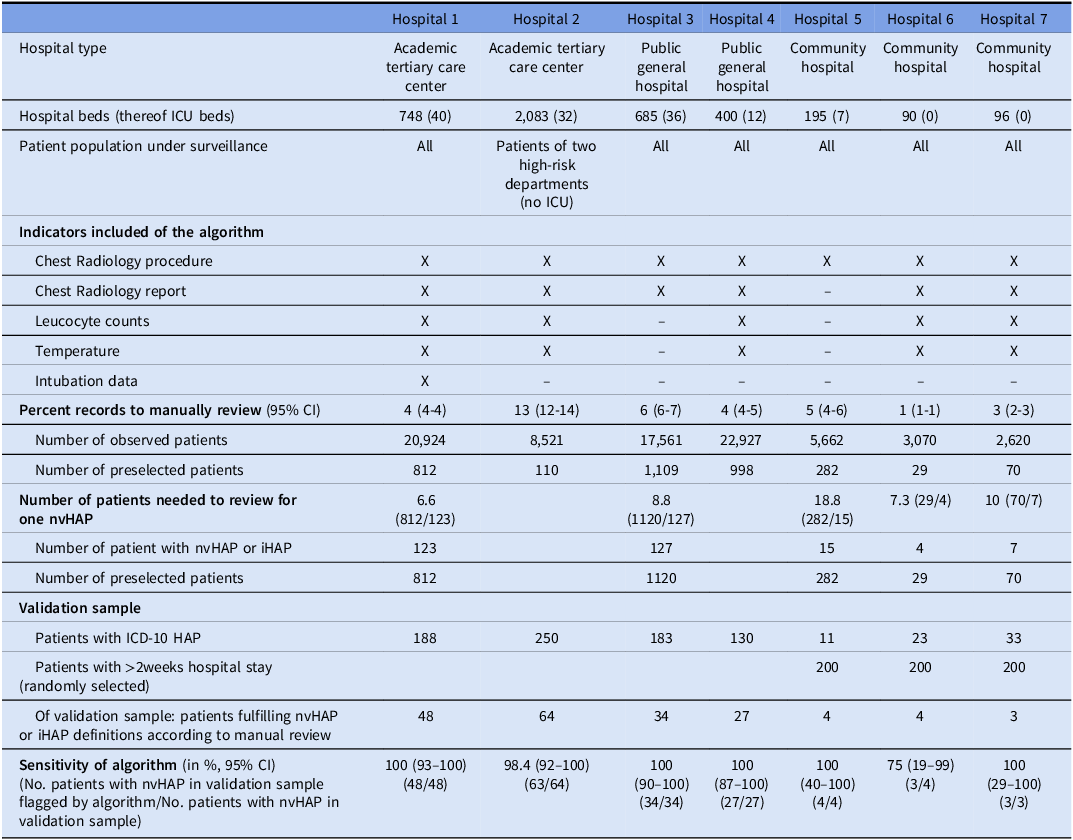
Components and performance characteristics of algorithms per hospital

Abbreviations: CI, confidence interval; HAP, hospital-acquired pneumonia; ICD, International Statistical Classification of Diseases and Related Health Problems; X, indicator included in algorithm.
Performance characteristics of the selection algorithms
Table 2 summarizes the performance characteristics of the algorithms. Sensitivity of the algorithms implemented in the larger pilot hospitals (No. 1–4) – including 2, 4 and 5 indicators - ranged from 98.4% (95% CI: 91.6–100) to 100% (95% CI: 92.6–100). Due to the small number of patients in the validation sample of the community hospitals (Hospitals 5–7), precise quantification of sensitivity was not possible.
Percentage of records to manually review was highest, i.e. 13% (95% CI: 12.2–13.6), in the hospital that applied the algorithm on patients from two high-risk departments. It was lowest, i.e. 1% (95% CI: 0.6–1.4), in a community hospital that applied a 4-indicator algorithm on all hospitalized patients. The mean number of patients needed to be manually reviewed to detect one patient with nvHAP from the list of preselected patients ranged from 6.6 to 18.8.
Time investment
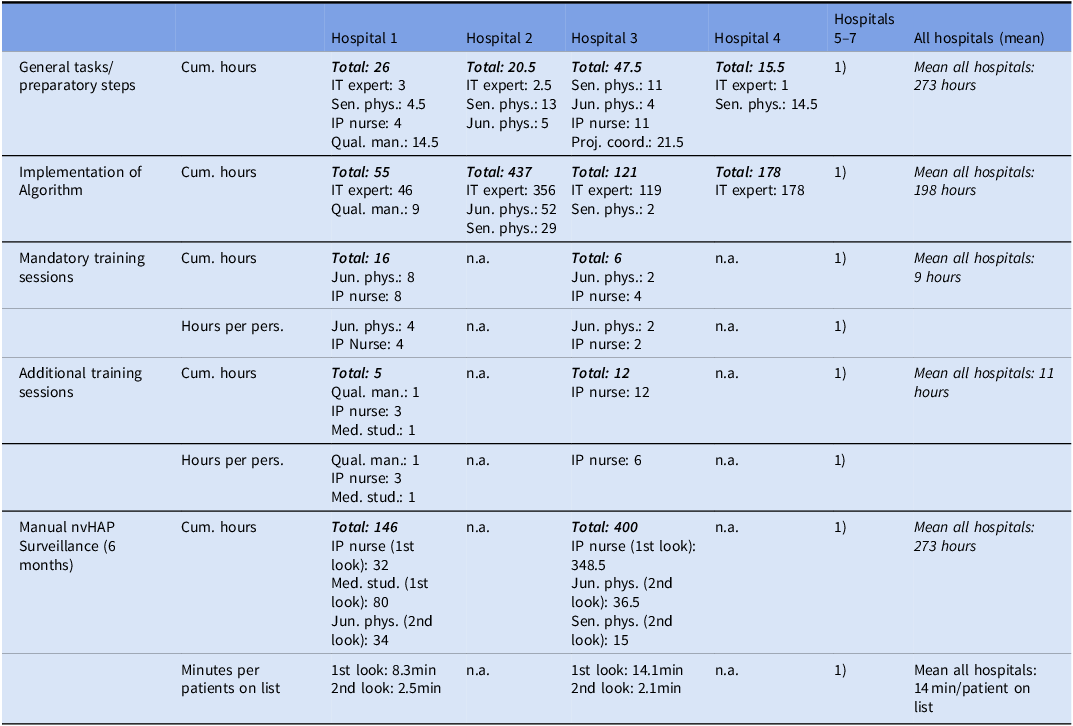
Table 3 summarizes time spent by pilot hospital and task. To technically implement the algorithm, a mean of 198 hours (from 55 to 437 hours) was spent, including tasks like data extraction and processing, data verification, implementation of the Python code, testing the algorithm performance, and iterative improvement steps. In all hospitals, validating the surveillance based on a manually reviewed patient cohort revealed the necessity to check back input data. Among the two pilot hospital teams conducting manual nvHAP review, the first reviewer spent a mean of 11.2 minutes and the second reviewer a mean 2.3 minutes per preselected patient.
Time investment

Caption: Time investment per professional group, project task and hospital. Cumulative hours are hours in total spent by each professional group and the team. Hours per person are mean hours per professional group and project task, relevant in hospitals that made the decision to involve more than one team member per professional group. Minutes per patients on list are mean minutes spent to manually review one preselected patient, divided in first reviewer (usually with nursing background) and – if necessary – second reviewer (usually an infectious disease physician).
Abbreviations: Cum. Hours, Cumulative hours; IP nurse, infection prevention nurse; IT expert, Information technology expert; Jun. phys., junior physician; Med. stud, medical student; min, minutes; per pers., per person; Proj. coord., project coordinator; Qual. man., quality manager; Sen. phys.; senior physician.
Determinants of implementation success
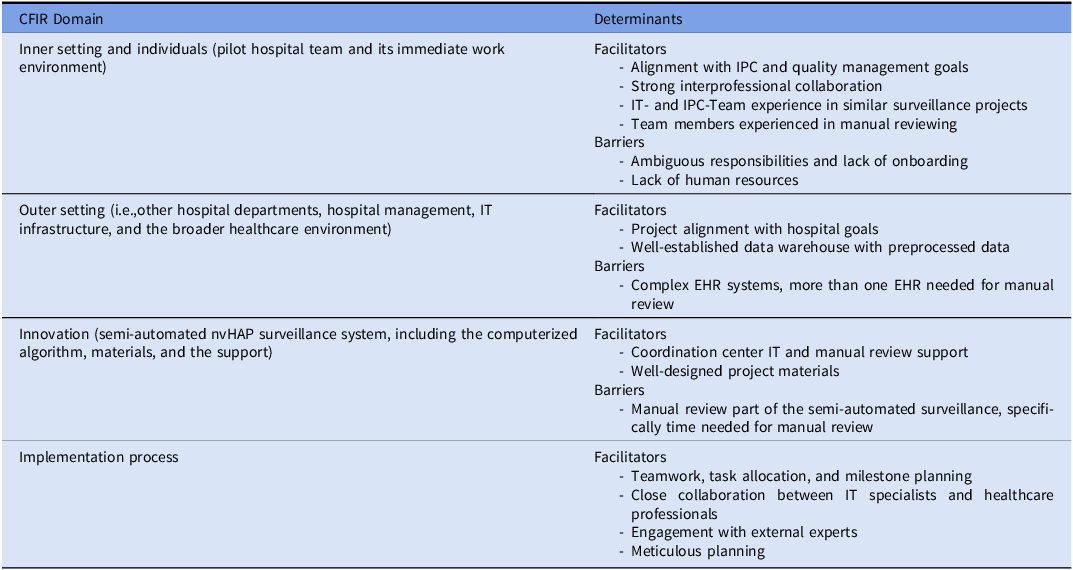
Twenty-five interviews were conducted, 13 at baseline and 12 at the end of the project. A total of 232 note segments were coded. Barriers and facilitators for adoption and implementation of the surveillance system are listed in Table 4.
Determinants (barriers and facilitators) for adoption, implementation and conduct of surveillance

Abbreviations: CFIR, Consolidated framework for implementation research; EHR, electronic health record; IPC, infection prevention and control; IT, information technology; nvHAP, nonventilator hospital acquired pneumonia.
Within the inner setting domain, defined as the pilot hospital team and its immediate work environment, the alignment with IPC and quality management goals, and the strong interprofessional collaboration facilitated implementation. Teams with prior experience in similar projects had an advantage, while staff turnover, insufficient onboarding, and ambiguous responsibilities were implementation barriers. A lack of human resources prevented manual review in some hospitals. In the individuals domain, IT- and IPC team experience in HAI surveillance was a key facilitator. Coordination center IT support helped address knowledge gaps, but implementation remained time-consuming when expertise was limited. Increasing experience in manual review boosted the confidence of the reviewers and expedited the manual part of the surveillance. Nurses found it easier to adhere to surveillance criteria, whereas some physicians struggled with personal clinical judgment interfering with the ECDC definition concept. For the implementation process domain, teamwork, task allocation, and milestone planning were crucial. Close collaboration between IT specialists and healthcare professionals ensured data completeness, while engagement with external experts and meticulous planning accelerated implementation. In the outer setting domain – e.g. other hospital departments, hospital management, IT infrastructure, and the broader healthcare environment – project alignment with hospital goals facilitated adoption. A well-established data warehouse with preprocessed data supported technical implementation, whereas the need for intensive data preparation (e.g., radiologic reports) and complex EHR structures slowed it down significantly. The manual review process was easier when all data were accessible within a single (compared to multiple) EHR system, and the hospital did not use paper-based documentation. In the innovation domain – referring to the semi-automated nvHAP surveillance system, including the computerized algorithm, materials, and the support from the coordination center team – time investment for manual review was the most important challenge for implementation. Manual review was considered challenging due to subjectivity in the interpretation of patient data, but training and support from the coordination center helped mitigate uncertainty. Well-designed project materials and ongoing technical support from the coordination center facilitated implementation.
Discussion
In this feasibility study, seven Swiss acute care pilot hospitals successfully implemented a semi-automated, locally adaptable surveillance system for nvHAP. Sensitivity is the most important performance indicator in semi-automated surveillance with a proposed threshold of 90%. Reference van Mourik, van Rooden and Abbas13 In the academic tertiary care centers and the public general hospitals, the implemented algorithms achieved sensitivities close to or at 100%, with lower 95% confidence interval margins consistently near or above 90%. Although the sample size in the community hospitals was too small to precisely quantify sensitivity, the available data and the algorithm’s design suggest that the algorithms likely performed comparably as in larger hospitals. Restricting the algorithm to indicators included as mandatory in the ECDC criteria implies that algorithms with fewer indicators may have equal or higher – but not lower – sensitivity. Thus, irrespective of the algorithm included, nvHAP cases could be missed by the algorithm as a result of technical implementation errors (e.g., nonconsideration of certain chest radiography types), text interpretation of radiology reports leading to erroneous exclusion of relevant chest images, or in rare instances of a pulmonary infiltrate being detected in a radiologic procedure not primarily targeting the chest (e.g., pneumonia detected in abdominal CT scan). The former was often the case when initially validating the performance of the algorithms during the technical implementation process, but meticulous overwork of input data finally led to sensitivities above 90%. This finding highlights the urgent need to validate an automated surveillance system after implementation and repetitively afterwards.
The second most important performance characteristic is the percentage of records to manually review, as it directly impacts the feasibility of manual nvHAP review in notoriously resource-constrained hospitals. A lower number of indicators included in the algorithm comes with the downside of a higher percentage of patients to manually review. Reference Mueller, Pfister, Faes Hesse, Zingg, Wolfensberger and Swissnoso14 This became evident in our project when comparing community hospitals 6 and 7, both using a four-indicator algorithm, which had a lower percentage of patients to manually review than hospital 5, which used one indicator only. Second, the patient population under surveillance plays an important role. Hospital 2 performed the study in two high-risk departments and although using a four-indicator algorithm, a high percentage of preselected patients had to be reviewed. The same algorithm resulted in a lower percentage of preselected patients in smaller hospitals, likely reflecting a lower-risk patient population with fewer diagnostic procedures performed.
When discussing feasibility of a semi-automated nvHAP surveillance, the time investment for technically implementing the algorithm is relevant. In our project, it ranged from 55 to 437 hours, with faster implementation observed in teams with prior experience with similar data-driven projects and access to preprocessed indicator variables. Qualitative interviews revealed that the need for detailed data preprocessing significantly slowed down the implementation process. The “onetime” investment to setup the computerized part of the surveillance is independent from hospital size and smaller hospitals often have limited IT resources. Thus, it likely is more difficult to absorb the upfront effort needed for tasks such as data extraction, verification, and algorithm implementation. On the other hand, manual review time is a critical factor for the sustained, long-term conduct of nvHAP surveillance. It is influenced by (1) the percentage of preselected records (see above), and (2) the time the pilot hospital team spends reviewing per record. In the pilot hospitals, the average time spent on one patient was 13.5 minutes. This is significantly longer than the 4 minutes reported by the coordination center team in 2017 reviewing USZ patients. Reference Wolfensberger, Clack and von Felten17 This discrepancy may be due to the coordination center team’s figure representing a one-year average and the manual review conducted by three well-experienced team members, while pilot hospital teams reported data over six months, with work distributed among up to five first-time nvHAP reviewers. Qualitative interviews indicated that individuals with greater experience felt more confident and were able to expedite surveillance, suggesting that most differences in performance were attributable to limited surveillance practice. It is anticipated that manual review teams will significantly enhance their efficiency over time.
To our knowledge, this is the first study to evaluate the implementation and performance of a semi-automated nvHAP surveillance system across multiple acute care hospitals. While offering valuable insights to hospitals considering implementing such a surveillance, it has several limitations. First, although pilot hospitals were selected to reflect diverse hospital sizes, IT infrastructures, and linguistic regions, the sample does not fully capture the variability of hospitals within Switzerland. Conducted in a high-income country without smaller hospital teams implementing the algorithm, the study leaves open whether hospitals with fewer resources would be able to implement and conduct the surveillance. However, our findings suggest that even hospitals with minimal data infrastructure can operate semi-automated nvHAP surveillance based solely on radiologic procedures. Second, algorithm sensitivity was assessed using a six month validation period, resulting in unmet sample size goals. While lower 95% confidence interval limits were consistently near or above 90% in larger hospitals, the low number of nvHAP cases in community hospitals prevented accurate sensitivity quantification. Additionally, the validation cohort consisted of a preselected patient population of patients enriched for nvHAP risk (patients with ICD-10 HAP or hospital stay >2 weeks); thus, selection bias and overestimation of sensitivity cannot be excluded. Third, only two hospital teams conducted manual surveillance in five hospitals, limiting time assessment for manual review. Finally, interviews to identify implementation determinants were conducted by the coordinating team, introducing potential social desirability and observer bias. All efforts were made to foster trust between interviewer and interviewee, and to maintain reflexivity throughout data analysis to minimize these biases.
In conclusion, implementation and conduct of an nvHAP semi-automated surveillance proved to be feasible. The adaptability of the standard algorithm was important. Regardless of the algorithm implemented and the patient population under surveillance, algorithms were sufficiently sensitive and resulted in a significant reduction of workload for manual reviewers. Due to the very high sensitivity of the algorithm including 5 indicators, working with algorithms including fewer indicators will not relevantly affect comparability between hospitals, but will come with the disadvantage of higher workload for manual reviewers. As sufficient time must be allocated for the initial technical setup, validation, and manual review, implementation of semi-automated nvHAP surveillance can be particularly recommended for hospitals that have indicator data available in a preprocessed form and have IT and infection prevention teams experienced with similar projects.
Supplementary material
To view supplementary material for this article, please visit https://doi.org/10.1017/ice.2026.10416.
Acknowledgements
We would like to thank the pilot hospital teams for their dedication and effort in implementing the semi-automated surveillance system, conducting manual surveillance, and providing valuable insights into the challenges and opportunities encountered during the project. We especially thank Anne-Flore Combaz, Marianne Rousseau-Schadegg, and Benedikt Reuthebuch for their manual review work in the pilot hospitals. We also thank Nicole Stoller, Simone Kessler, and Selina Bilger for their organizational support. We thank all members of the Swissnoso group and Mohamed Abbas for his valuable subject-matter input.
Members of the Swissnoso group are: Dr. Carlo Balmelli, Lugano; Dr. Delphine Berthod, Sion; PD Dr. Niccolò Buetti, Geneva; Prof. Stephan Harbarth, Geneva; Dr. Philipp Jent, Bern; Prof. Jonas Marschall, Bern; Prof. Hugo Sax, Zürich; Dr. Matthias Schlegel, St. Gallen; Dr. Alexander Schweiger, Zug; PD Dr. Laurence Senn, Lausanne; Prof. Rami Sommerstein, Luzern; Prof. Nicolas Troillet, Sion; Prof. Sarah Tschudin-Sutter, Basel; Dr. Danielle Vuichard Gysin, Frauenfeld; Prof. Andreas Widmer, Basel; PD Dr. Aline Wolfensberger, Zürich; PD Dr. Walter Zingg, Zürich
Author contribution
AW, DV, MFH, MP, and WZ designed the study. The pilot hospital teams AR, BW, DB, DF, EC, FG, GC, IS, JM, LEK, MA, MCZ, SH, SK, STS, USL technically implemented the algorithm, coordinated and/or performed manual surveillance in the pilot hospitals. MP technically assisted the pilot hospitals in the implementation of the algorithm. AM, CR and MFH validated the manual part of the surveillance in the pilot hospitals. MZ conducted sample size calculations. AW and MZ conducted quantitative data analysis. AW and MFH conducted qualitative data analysis. AW, DV, MFH, MP, and WZ interpreted the data. AW and MFH drafted the manuscript. All co-authors edited and reviewed the manuscript.
Financial support
The Swiss Federal office of public health funded this pilot-study on implementation of a semi-automated surveillance system. Writing of the manuscript was supported by institutional funding.
Competing interests
None to declare for all authors.
Use of artificial intellingence (AI) tools
The authors used AI-based tools (DeepL and ChatGPT) to refine grammar; all substantive content was written and verified by the authors.